Abstract
Precise localization of various ion channels into proper subcellular compartments is crucial for neuronal excitability and synaptic transmission. Axonal K+ channels that are activated by depolarization of the membrane potential participate in the repolarizing phase of the action potential, and hence regulate action potential firing patterns, which encode output signals. Moreover, some of these channels can directly control neurotransmitter release at axonal terminals by constraining local membrane excitability and limiting Ca2+ influx. K+ channels differ not only in biophysical and pharmacological properties, but in expression and subcellular distribution as well. Importantly, proper targeting of channel proteins is a prerequisite for electrical and chemical functions of axons. In this review, we first highlight recent studies that demonstrate different roles of axonal K+ channels in the local regulation of axonal excitability. Next, we focus on research progress in identifying axonal targeting motifs and machinery of several different types of K+ channels present in axons. Regulation of K+ channel targeting and activity may underlie a novel form of neuronal plasticity. This research field can contribute to generating novel therapeutic strategies through manipulating neuronal excitability in treating neurological diseases, such as multiple sclerosis, neuropathic pain, and Alzheimer’s disease.
Keywords: axonal targeting, action potential, neurotransmitter release, voltage-gated potassium channel, axon initial segment, neuronal polarity
1. Introduction
Neurons are highly polarized cells, typically with multiple dendrites and a long axon, important for inputs and outputs of signals, respectively. In the mammalian central nervous system (CNS), neuronal dendrites often receive and integrate thousands of synaptic inputs. Then the synaptic potentials are converted into action potentials (APs) at the axon initial segment (AIS). APs are conveyed along axons and trigger neurotransmitter release at axonal terminals. Released neurotransmitters activate their receptors located on postsynaptic membranes of the target cells.
Output functions of axons require concerted actions of various types of ion channels, including voltage-gated sodium (Nav) channels, voltage-gated potassium (Kv) channels, and voltage-gated calcium (Cav) channels. Different types of neurons in nervous systems differ in their axon/dendrite morphology and AP firing patterns. The number and activity of these channels at specific axonal locations are fundamental to the axonal functions for various neurons. The biophysical properties of most of these channels have been extensively characterized in native cells and expression systems. Besides critical roles in synaptic transmission, the importance of axonal ion channel targeting in axonal electrical computation was proposed by the studies using computer simulations (Debanne, 2004; Li and Ascoli, 2006). Given the complex branching patterns of dendrites and axons of central neurons, especially interneurons, precise prediction by simulation is very difficult, if not impossible, when the targeting patterns of key ion channels are not known. Therefore, it is important to understand the mechanisms underlying the precise targeting of channel proteins and how they affect neuronal excitability, such as the input-output relationship. Elucidating the modulation of neuronal excitability, a new form of neuronal plasticity, is an essential step for understanding the dynamics and function of neural circuits.
Recently, progress has been made in understanding the local function of axonal ion channels, and identifying the targeting motifs and interacting proteins critical for targeting channel proteins into axons and at specific axonal sites. These studies have been carried out with contemporary patch clamp recording, molecular and cell biology, and protein biochemistry techniques. In this review, we focus on the function and mechanism of four types of K+ channels that exist in axons and are activated by membrane potentials. When activated, these channel complexes selectively allow K+ ions to flux out of the cell following the electrochemical gradient of K+ ion and hence hyperpolarize the membrane potential back to the resting level (towards K+ equilibrium). Therefore, they are involved in regulating AP initiation, waveform, frequency and uni-directional propagation, and neurotransmitter release.
2. Potassium channel superfamily
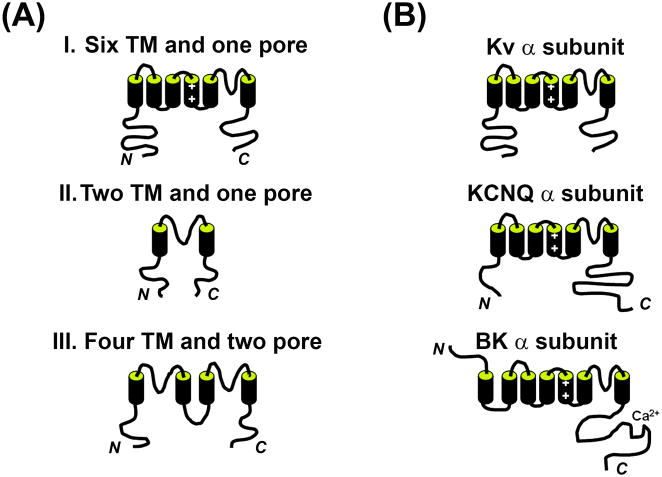
K+ channels are probably the most diverse family of ion channels. K+ channel superfamily contains approximately 80 different genes encoding various pore-forming subunits. Based on the secondary structure of channel subunits, they can be grouped into three major families (Fig. 1A). Kv channels belong to the first family. Each subunit of this family contains six to seven transmembrane (TM) segments and one pore-forming loop between the last two TM segments. Each channel complex contains four such pore-forming subunits. In this family, there are also members that are activated by both the membrane potential and intracellular chemical signals. We collectively call them, “voltage-sensitive K+ channels”. Inwardly rectifying (conducting current more efficiently in the inward direction) K+ channels belong to the second family. The members of this family have the simplest secondary structure, two TM domains and one pore-forming loop. They also form tetramers and often co-assemble with other membrane proteins. Two-pore domain K+ channels belong to the third family. Each subunit contains four TM segments and two pore-forming loops. In this family, two subunits form a channel complex. Most members of this third family generate currents with little voltage or time dependence, creating leak K+ channels.
Fig. 1. Structural diagram of potassium channel α subunits.
(A) Structure diagram of the α subunits of three major groups in the potassium channel superfamily. The α subunits in the group I contain six to seven transmembrane (TM) segments and one pore-forming loop. The α subunits in the group II contain two TM segments and one pore-forming loop. The α subunits in the group III contain four TM segments and two pore-forming loops. “N” and “C” indicate the amino and carboxyl termini of the protein, respectively. (B) Structure diagram of the α subunits of voltage-gated K+ (Kv) channels (Kv1 to Kv4), KCNQ (Kv7) channels, and the BK (Slo1) channel.
2.1. Voltage-sensitive potassium channels
Kv channels form a large and diverse family, which can be further divided into many subfamilies. We focus on the voltage-sensing and pore-forming α subunits. The prototypical Kv channels are divided into four subfamilies based on the similarity to their homologues in fruit fly Drosophila, Kv1 (Shaker; KCNA), Kv2 (Shab; KCNB), Kv3 (Shaw; KCNC), and Kv4 (Shal; KCND) (Fig. 1B). Since the initial cloning of these four subfamilies, eight other subfamilies (Kv5 to Kv12) have been identified to date (Chandy, 1991; Gutman et al., 2005). Among all subfamilies, Kv5 (KCNF), Kv6 (KCNG), Kv8 (KCNV), and Kv9 (KCNS) α subunits have not been shown to yield functional channels by themselves in expression systems, but can modulate other functional Kv channels (Gutman et al., 2005; Vacher et al., 2008).
The four subfamilies of Kv1 to Kv4 express extensively in nervous systems. Their channel activities, pharmacological properties, and expression and distribution patterns have been extensively studied. The Kv7 (KCNQ) subfamily was identified relatively more recently (Fig. 1B). It appears to be a very important subfamily. Mutations in four out of five KCNQ genes (KCNQ1 to KCNQ5) cause diseases in humans including cardiac arrhythmias, deafness and epilepsy (Jentsch, 2000). The Kv10-Kv12 channels are categorized into the KCNH subfamily. Members of this subfamily are also expressed in nervous systems and play important roles in regulating neuronal excitability of nervous systems.
There are two other groups of K+ channels that can be categorized into this family, calcium-activated K+ channels, although they are sometimes assigned to a separate group. One is the large conductance Ca2+-regulated, “big” K+ (BK) channels encoded by the Slo1 gene (a name derived from the conserved gene that encodes this channel, first cloned from fruit fly Drosophila) family, which is among the largest and most complex of the K+ channel superfamily (Fig. 1B). The BK channel is gated by both voltage and intracellular Ca2+. Its α subunit contains seven TM segments and one pore-forming loop. The BK and related channels resemble Kv channels, but possess a large conserved carboxyl terminus, which allows channel gating to be altered in response to the direct sensing of several different intracellular ions, and by other second messenger systems (Salkoff et al., 2006). In this subfamily, there are three other members, Slo2.1 (Slick), Slo2.2 (Slack), and Slo3. They differ with the BK channel in gating properties and Ca2+ regulation (Salkoff et al., 2006). There are four beta subunits for Slo1, β1 to β4 (KCNMB1 to KCNMB4) (Table I). They have two membrane-spanning segments, with N and C termini located on the cytoplasmic side. They regulate Slo1 activity and trafficking. The second group includes small- and intermediate-conductance calcium-regulated K+ channels. Their subunits contain six TM segments and one pore-forming loop, structurally similar to Kv channels, but their activation is regulated by intracellular Ca2+ and insensitive to the membrane potential (Weatherall et al., 2010).
Table I.
Four subfamilies of voltage-sensitive K+ channels
| Subfamily | Channel | Gene | Chromosom al localization | Blockers | Openers | Auxiliary subunits |
|---|---|---|---|---|---|---|
| Kv1 (Shaker) | Kv1.1* | KCNA1 | 12p13.3 | DTX, TEA, 4-AP | Kvβ1 (KCNAB1) Kvβ2 (KCNAB2) Kvβ3 (KCNAB3) |
|
| Kv1.2 | KCNA2 | 1p13 | ||||
| Kv1.3 | KCNA3 | 1p13.3 | TEA, 4-AP | |||
| Kv1.4 | KCNA4 | 11p14.3-15.2 | 4-AP | |||
| Kv1.5* | KCNA5 | 12p13.3 | 4-AP | |||
| Kv1.6 | KCNA6 | 12p13.3 | DTX, TEA,4-AP | |||
| Kv1.7 | KCNA7 | 19q13.3 | 4-AP | |||
| Kv1.8 | KCNA10 | 1p13.3-22.1 | ||||
| Kv3 (Shaw) | Kv3.1 | KCNC1 | 11p14.3-15.2 | TEA, 4-AP | ||
| Kv3.2 | KCNC2 | 19q13.3-13.4 | ||||
| Kv3.3* | KCNC3 | 19q13.3 | ||||
| Kv3.4 | KCNC4 | 1p21 | ||||
| KCNQ (Kv7) | Kv7.1* | KCNQ1 | 11p15.5 | Linopirdine, XE991, | Flupirtine retigabine | |
| Kv7.2* | KCNQ2 | 20q13.3 | ||||
| Kv7.3* | KCNQ3 | 8q24.22-q24.3 | ||||
| Kv7.4* | KCNQ4 | 1p34 | ||||
| Kv7.5 | KCNQ5 | 6q14 | ||||
| BK (Slo1) | KCa1.1* | KCNMA1 | 10q22 | Iberiotoxin, charybdoto xin, (+)-tubocurarin e, kaliotoxin, penitrmA, TEA | NS004 NS1619 DHS1 |
β1(KCNMB1) β2(KCNMB2) β3(KCNMB3) β4(KCNMB4) |
Mutations have been identified to cause diseases in humans.
2.2. Inward-rectifying potassium channels
In contrast to Kv channels, inwardly rectifying K+ channels (Kir; gene symbol: KCNJ) allow K+ ions to more efficiently flow into, rather than out of, the cell. Each Kir subunit contains two TM segments and one pore-forming loop and each Kir channel complex contains four such subunits. To date, fifteen Kir subunit genes have been identified and grouped into seven subfamilies (Kir1.x to Kir7.x). These subfamilies can be further categorized into four functional groups: (1) classical Kir channels (Kir2.x); (2) G protein-gated Kir channels (Kir3.x); (3) ATP-sensitive K+ channels (Kir6.x); (4) K+-transport channels (Kir1.x, Kir4.x, Kir5.x, and Kir7.x) (Hibino et al., 2010)). Kir channels not only control the passive and active electrical properties of cells, but they are also involved in G protein-coupled receptor signaling, and they may link cellular metabolic state and membrane excitability in vivo (see more extensive reviews about Kir channels (Bichet et al., 2003; Hibino et al., 2010; Loussouarn et al., 2002; Luscher and Slesinger, 2010)). The trafficking and function of Kir channels in nervous systems is beyond the scope of this review.
2.3. Two-pore domain potassium channels
Leak K+ channels contribute to the resting membrane potential and are involved in regulating neuronal excitability. The first channel, TWIK (tandem of pore domains in a weak inward rectifying K+ channel, now called TWIK-1), was identified in 1996 (Lesage et al., 1996). Currently, there are fifteen members in the KCNK family in human genome (KCNK1 to KCNK18, but KCNK8, KCNK11, and KCNK14 do not exist). These two-pore domain K+ channels can be grouped into six subfamilies, TWIK, THIK, TREK, TASK, TALK, and TRESK. These channels can be modulated by a host of different endogenous and clinical compounds such as neurotransmitters and anesthetics, and by other voltage-independent factors such as temperature, pH, oxygen tension, and osmolarity. Many of these channels are expressed in mammalian brains and participate in homeostatic regulation of physicochemical processes (see more extensive reviews about two-pore domain channels (Enyedi and Czirjak, 2010; Goldstein et al., 2001; Talley et al., 2003)). The axon-dendrite targeting of this channel family has not yet been established.
This review focuses on the function and mechanism of four subfamilies of axonal K+ channels that are directly involved in regulating neuronal excitability and synaptic transmission. They include Kv1, Kv3, KCNQ and BK channel subfamilies (Table I). Among them, Kv3 channels have the highest activation threshold of membrane potentials (~ − 10 mV), fastest activation (activation constant, the time for half-maximal conductance, < 1 ms) and deactivation (deactivation constant, ~ 2 ms) kinetics, the biophysical features thought to be critical to enable rapid spiking of some central neurons (Jonas et al., 2004; Rudy and McBain, 2001). In contrast, KCNQ and BK channels operate at much slower kinetics (activation constant, > 100 ms; deactivation is very slow) and are subject to the regulation of various intracellular factors (Salkoff et al., 2006). The activation and deactivation kinetics of Kv1 are intermediate between the two groups (activation constant, ~ 5 – 10 ms; deactivation constant, ~ 10 – 40 ms). The activation threshold of Kv1 and KCNQ channels is more negative (~ − 40 mV) and close to the resting membrane potential. Therefore, the biophysical properties of these channels differentially impact AP initiation, waveform, frequency, and invasion into axonal terminals. Their precise localization along axons allows them to profoundly regulate the electrical signal computation in axons.
3. Physiological functions of axonal targeting of voltage-sensitive potassium channels
3.1. Axonal compartments and ion channel functions at the axon initial segment
An axon is usually a long and slender projection of a neuron that conducts electrical impulses away from neuronal soma. It contains several structurally distinct regions that play key roles in axonal electrical and chemical functions. The AIS, adjacent to the axon hillock, is the site where APs are initiated in most neurons due to the presence of high-density Nav channels. Its position varies between neurons and is subject to regulation. Nodes of Ranvier are the gaps (~ 1 μm in length) between the myelin sheaths formed along myelinated axons, where Nav channels are also highly concentrated. The nodal region is actively involved in the saltatory conduction of APs. Axonal terminals are the sites where neurotransmitters are released upon activation. AISs, nodes of Ranvier, axonal branching points, and axonal terminals are strategic sites for electrical signal computation of axons (Debanne, 2004). Axonal localization of various ion channels profoundly affects AP initiation, propagation and invasion to nerve terminals, and neurotransmitter release.
The AIS is not only the AP initiation site but also the gate where the entry of axonal proteins and organelles is controlled. Analysis of the structure and function of the AIS attracted much attention lately. Localization of AISs including key molecular components can vary in different cell types (Lorincz and Nusser, 2008) and be regulated by neuronal activity, which may constitute a novel form of neuronal plasticity (Grubb and Burrone, 2010; Kuba et al., 2010). Different types of ion channels are localized at the AIS and carry out different functions. For instance, the differential targeting of Nav1.2 and Nav1.6 at the AIS controls uni-direction propagation of APs (Boiko et al., 2003; Hu et al., 2009), and the T- and R-type Cav channels at the AIS shape spike bursts (Bender and Trussell, 2009). Kv channels also play critical roles at the AIS.
3.2. Regulation of action potential firing by voltage-sensitive potassium channels
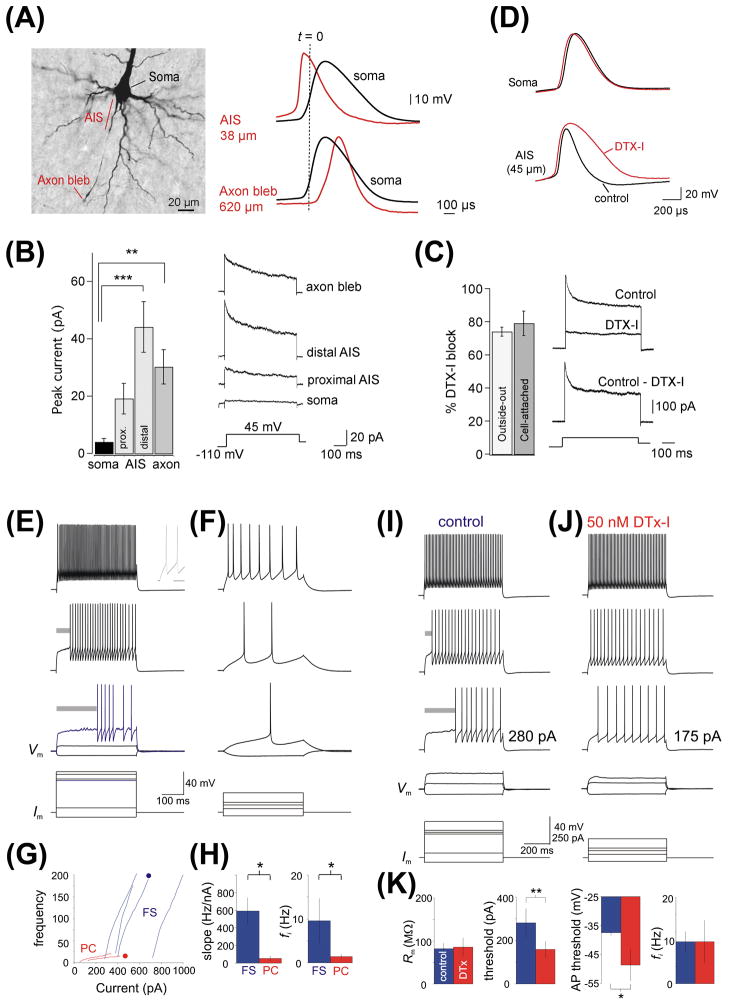
Kv1 channels at the AIS of pyramidal neurons in brain contribute to a low threshold, slowly inactivating, outward current during the AP generation, which shortens the AP duration (Kole et al., 2007; Shu et al., 2007) (Fig. 2A–D). Local application of the specific blocker of Kv1 channels, α-dendrotoxin, at the AIS but not the soma broadened the APs (Kole et al., 2007; Shu et al., 2007) (Fig. 2C,D). In rapid-spiking GABAergic interneurons of the neocortex, Kv1 channels at the AIS control the AP threshold so that the cells respond preferentially to large inputs (Goldberg et al., 2008). Blocking Kv1 channels with α-dendrotoxin shortens the delay of the first spike, and hence converts the delay-type spiking of the cells to continuous spiking without influencing the high-frequency firing (Goldberg et al., 2008) (Fig. 2E–K). KCNQ channels at the AIS of hippocampal pyramidal neurons regulate the inherent spontaneous firing via controlling axonal resting membrane potential and AP threshold (Shah et al., 2008). Kv2.1 and Kv2.2 channels are present at the AIS (Johnston et al., 2008; Sarmiere et al., 2008). Kv2.2 may play a crucial role in maintaining AP amplitude by regulating the interspike potential during high frequency firing in medial nucleus of the trapezoid body neurons (Johnston et al., 2008). Currently, it remains unclear whether Kv3 and Slo1 channels play an important role at AISs. In the neurons of spinal cord, it is shown that Kv3.1b is concentrated at nodes of Ranvier but not at AISs (Devaux et al., 2003).
Fig. 2. Regulation of action potential (AP) firing by Kv1 channels localized at axon initial segments.
(A) AP is initiated at the AIS of pyramidal neurons. Photograph of a biocytin-filled layer 5 pyramidal neuron indicates the soma, the unmyelinated AIS, a myelinated axonal region, and an axonal bleb site accessible for whole-cell recording (left). Example traces show APs evoked by somatic current injection, and recorded at the AIS 38 μm from the axon hillock (red) and at the soma (black) (top), and recorded at an axon bleb 620 μm from the hillock (bottom). (B) Mean amplitude of slowly inactivating K+ currents evoked by voltage steps from −110 to +45 mV recorded in cell-attached patches from the soma, proximal AIS (5–30 mm from hillock), distal AIS (35–55 mm), and axonal sites (70–400 mm). Asterisks indicate significant increases in K+ peak amplitudes compared to the soma (*** p<0.001 and ** p<0.01). Traces show examples of slow-inactivating K+ current at the indicated locations (averages of four to six patches). (C) Outside-out recordings of K+ currents from AIS or axonal blebs before and after puffs with 500 nM DTX-1. Subtracted traces show the DTX-sensitive current. (D) Impact of bath application of 50–100 nM DTX-1 on somatic (top) and axonal (bottom) APs before (black) and several minutes after DTX-1 (red) ((A)–(D) modified from Kole et al., 2007 with copyright permission from Neuron). (E) Discharge pattern of a fast-spiking (FS) neuron in layer 2/3 barrel cortex. Shown are voltage responses to 600 ms current injections of, from bottom to top, −100, 100, 345, 360, and 440 pA. Gray bars highlight delay to first spike. Inset in the topmost trace shows the first two spikes on an expanded timescale (Scale bar, 10 ms). (F) Firing pattern of an adjacent pyramidal neuron. (G) f–I curves for a subset of representative FS cells (blue) and PCs (red), with the cells in (E) and (F) indicated by dots. (H) Summary data illustrating initial firing frequency and the slope of the f–I relation for FS cells and PCs. (I) Firing pattern of an FS cell in layer 2/3 barrel cortex to current injections. (J) Same cell as in (I), after bath application of 50 nM DTX-1. (K) Summary data. DTX-1 produces no change in Rm or fi but decreased ITH and shifted the voltage threshold for AP generation to hyperpolarized potentials ((E)–(K) modified from Goldberg et al., 2008 with copyright permission from Neuron).
K+ channels along the axonal shaft regulate AP waveform, frequency, and uni-directional propagation. In particular, axonal Kv3 may play a critical role in rapid spiking of APs in some GABAergic interneurons (Jonas et al., 2004; Rudy and McBain, 2001). The unique channel properties of Kv3 channels, high activation threshold, fast activation and deactivation kinetics (Coetzee et al., 1999; Rudy and McBain, 2001), allow neurons to fire APs at high frequency, up to several hundred Hz. In contrast, BK channels operate with much slower kinetics and contribute to the formation of afterhyperpolarization (AHP), which lasts several tens of milliseconds. BK channels also play important roles in spike frequency adaptation. During repetitive AP firing, increased Ca2+ entry results in greater activation of BK channels and hence reduction of AP firing. Low-threshold, slowly inactivating K+ currents, containing Kv1.2, play a key role in the flexible properties of intracortical axons and may contribute significantly to intracortical processing (Shu et al., 2007).
In myelinated axons, Kv1 channels are concealed under the myelin sheath, thus they may contribute to maintaining the AP fidelity by preventing repetitive and aberrant firing. In contrast, Kv3.1b and KCNQ channels are found at nodes of Ranvier and may be directly involved in the saltatory conduction of APs. Moreover, localized ion channels may play critical roles in axonal branch points during AP failure and frequency change (Debanne, 2004). However, the exact roles of ion channels in axonal branching points have not yet been experimentally established.
3.3. Regulation of neurotransmitter release by voltage-sensitive potassium channels
All axonal voltage-sensitive K+ channels can regulate neurotransmitter release. The action can be either directly through regulating AP invasion into axonal terminals and limiting Ca2+ influx via Cav channels by hyperpolarizing axonal membranes, indirectly via regulating AP propagation, or both.
Kv1 channels regulate neurotransmitter release in various neurons. Kv1.1 channels regulate neurotransmitter release at motor nerve terminals. Deleting Kv1.1 produces a striking temperature-sensitive excitability change at nerve terminals (Zhou et al., 1999; Zhou et al., 1998). Here, Kv1.1 channels most likely exert their function by regulating AP propagation rather than directly regulating neurotransmitter release, since Kv1.1 channels are not present in terminal membranes (Zhou et al., 1998). On the other hand, Kv1 channels directly regulate neurotransmitter release at the cerebellar basket cell terminals, since they are highly concentrated there (McNamara et al., 1993; Rhodes et al., 1995; Rhodes et al., 1997; Wang et al., 1993; Wang et al., 1994). A recent study shows that axonal Kv1 channels are found to regulate synaptic strength at proximal axonal terminals of layer 5 pyramidal neurons (Kole et al., 2007). Furthermore, in hippocampal mossy fiber boutons fast inactivating K+ channels dynamically regulate presynaptic Ca2+ influx, contributing to the control of efficacy of a glutamatergic synapse in the cortex (Geiger and Jonas, 2000). Kv1 channels suppress terminal hyperexcitability during the depolarizing after-potential (Dodson et al., 2002; Dodson et al., 2003).
Kv3 channels are directly involved in neurotransmitter release in nerve terminals of various neurons, including rapid-spiking GABAergic interneurons (Goldberg et al., 2005), cerebellar basket cell nerve terminals (Southan and Robertson, 2000), the calyx of Held terminals (a synapse required for the binaural processing of sound-source localization) (Elezgarai et al., 2003; Ishikawa et al., 2003), and motor nerve terminals (Brooke et al., 2004a; Brooke et al., 2004b). At the calyx of Held terminal, Kv3 channels (including Kv3.1 and Kv3.4) directly regulate evoked transmitter release, whereas Kv1 channels reduce nerve terminal excitability, thereby preventing aberrant transmitter release (Ishikawa et al., 2003).
KCNQ channels give rise to the non-inactivating M-current in neurons (Brown and Adams, 1980). Presynaptic M-channels on hippocampal CA3 neurons regulate axonal excitability and neurotransmitter release (Vervaeke et al., 2006). M-channels act pre- and postsynaptically to dampen neuronal excitability (Peretz et al., 2007). Presynaptic M-channels (KCNQ2) inhibit transmitter release by reducing Ca2+ entry through P/Q-type voltage-gated Ca2+ channels in the cortex (Luisi et al., 2009), and modulate neurotransmitter release from rat hippocampal nerve terminals (Martire et al., 2004).
In axonal terminals, BK channels are activated by both Ca2+ entry and depolarization of membrane potentials during AP invasion. This results in acceleration of the repolarization of the terminal membrane, and thereby exert negative feedback on the release process (Berkefeld and Fakler, 2008; Berkefeld et al., 2006; Grimes et al., 2009; Petersen and Maruyama, 1984; Raffaelli et al., 2004; Robitaille et al., 1993).
4. Axonal targeting of Kv1 (Shaker) channels
4.1. Axonal targeting of endogenous Kv1 channels
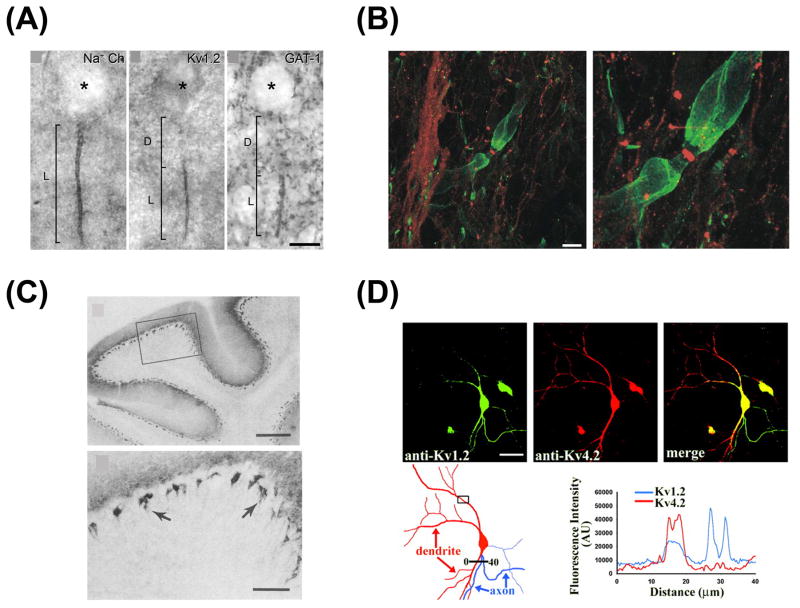
Among the eight α subunits of Kv1 channels (Kv1.1 to Kv1.8), Kv1.4 is the only one carrying transient (inactivating, or A type) current when expressed alone. Kv1.1, Kv1.2, and Kv1.4, the three most abundant Kv1 α subunits in mammalian brain, primarily localize in axons and nerve terminals (Trimmer and Rhodes, 2004). Kv1.1 and Kv1.2 colocalize at AISs of various central neurons (Dodson et al., 2002; Inda et al., 2006; Van Wart et al., 2007), the juxtaparanodal (JXP) regions of myelinated axons in spinal cord and cortex (Rasband, 2004; Rasband et al., 2002; Rasband and Trimmer, 2001; Rasband et al., 1999; Rasband et al., 1998; Rhodes et al., 1995; Rhodes et al., 1997; Vabnick et al., 1999; Wang et al., 1993; Wang et al., 1994), and axonal terminals of cerebellar basket cells (McNamara et al., 1993; Rhodes et al., 1995; Rhodes et al., 1997; Wang et al., 1993; Wang et al., 1994) and other neurons (Dodson et al., 2003) (Fig. 3). Kv1.4 colocalizes with Kv1.1 in mossy fiber axons in the hippocampus (Cooper et al., 1998; Sheng et al., 1992; Wang et al., 1993; Wang et al., 1994). Kv1.4 also localizes in small diameter axonal fibers of dorsal root ganglion neurons, which are involved in pain sensing (Rasband et al., 2001).
Fig. 3. Kv1 channels are mainly localized in axon initial segments, juxtaparanodal regions, axonal terminals in nervous systems.
(A) Distribution of Nav and Kv1.2 channels in the AIS of pyramidal neurons and their relationship with chandelier cell axonal terminals (modified from Inda et al., 2006 with copyright permission from Proc. Natl. Acad. Sci. U.S.A.) (B) Double labeling by using anti-Kv1.2 (green) and anti-Nav channel (red) antibodies in myelinated rat spinal cord (modified from Rasband and Trimmer, 2001 with copyright permission from J. Comp. Neurol.). Scale bar, 10 μm. (C) Localization of Kv1.1 in mouse cerebellar basket cell terminals. General distribution of Kv1.1 in mouse cerebellum (top) and higher magnification of the boxed region (bottom) (modified from Wang et al., 1994 with copyright permission from the J. Neurosci.) (D) Double labeling reveals segregation of endogenous Kv1.2 and Kv4.2 in mature hippocampal neurons (21 DIV) (modified from Gu et al., 2003 with copyright permission from Science). Immunofluorescence intensity profiles, along a 40-mm line (black, lower left) were measured from the original 16-bit TIFF images with NIH Image J (lower right). AU, arbitrary unit.
Kv1.3 and Kv1.6 are less abundant in neurons. Kv1.3 is highly expressed in the parallel fiber axons of cerebellar granule cells (Koch et al., 1997; Veh et al., 1995), and localizes in presynaptic terminals of brainstem auditory neurons (Gazula et al., 2010). Kv1.6 is found primarily in interneurons (Rhodes et al., 1997). Other Kv1 channels, including Kv1.5, Kv1.7 and Kv1.8, are either not present in neurons or not known (Chittajallu et al., 2002; Kalman et al., 1998; Pannasch et al., 2006; Vacher et al., 2008). It is important to note that dendritic staining of some Kv1 channels, albeit less abundant, is seen in the brain, which may play an important role in dendrite integration. The functions of various Kv channels in dendrites are beyond the scope of this review.
Kv1 α subunits can coassemble into either homotetramers or heterotetramers with distinct channel properties. Furthermore, assembly with auxiliary (Kvβ1 to Kvβ3) subunits can markedly affect expression, targeting, and activity of the channel complex (Li et al., 2006; Pongs et al., 1999; Pongs and Schwarz, 2010). For instance, the presence of Kvβ1.1 can convert the Kv1.1 or Kv1.2-containing channel complexes from carrying sustained currents to carrying transient currents (Rettig et al., 1994). Among three isoforms of Kv1 auxiliary subunits, Kvβ1 and Kvβ2 are extensively expressed in the brain, and generally coexpressed and colocalized with Kv1 α subunits (Monaghan et al., 2001; Rettig et al., 1994; Rhodes et al., 1995; Rhodes et al., 1996; Rhodes et al., 1997). In particular, Kvβ2 appears to colocalize with most Kv1-containing channel complexes. Immunostaining for Kvβ2 shows it is present in almost every location where immunoreactivity for Kv1 α subunits is observed, including AISs, the JXP regions, and cerebellar basket cell terminals (Rasband et al., 1999; Rhodes et al., 1996; Rhodes et al., 1997). However, there is also extensive immunostaining for Kvβ2 in soma and dendrites of neurons.
Owing to complex neuronal structure and other surrounding cells in the brain, it is not always easy to unequivocally determine the amount and localization of Kv1 channels using immunohistochemistry, especially in the hippocampus. Using an excitotoxin lesion strategy combined with immunohistochemistry, it was confirmed that Kv1 channel complexes predominantly localize on axons and nerve terminals in the hippocampus (Monaghan et al., 2001). Axonal localization of Kv1 channels has also been confirmed with cultured hippocampal neurons (Grosse et al., 2000; Gu et al., 2003) (Fig. 3D).
4.2. Mechanisms underlying intrinsic Kv1 axonal targeting
Kv1 T1 domains have been identified as the axonal targeting domain for Kv1 channels. When over-expressed in cultured hippocampal neurons, the HA-tagged Kv1.2 is enriched on axonal membranes (Gu et al., 2003). A non-polarized reporter protein, CD4, can be enriched on axonal membranes when fused with the Kv1.2 T1 domain but not with the Kv1.2 C-terminus (Gu et al., 2003). In addition, fusing the T1 domains from Kv1.1, Kv1.2, Kv1.3 or Kv1.4, but not from Kv4.2, to a dendritic reporter protein, the transferrin receptor, results in axonal localization of the fusion protein (Gu et al., 2003; Rivera et al., 2005). Owing to the high homology of T1 domains within the Kv1 subfamily members, the evidence is compelling that the Kv1 T1 domain is the axonal targeting domain.
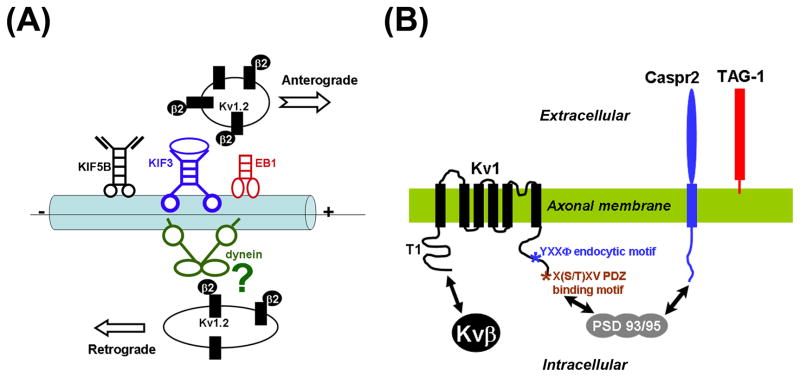
Kvβ subunits likely play a critical role in mediating the axonal targeting of Kv1 channels. Most importantly, Kvβ directly binds to the Kv1 T1 domains. Co-expression of Kvβ2 markedly enhances axonal targeting of Kv1.2 (Gu et al., 2006). Using live cell imaging technique, Kvβ2 was found to markedly enhance the velocity of anterograde transport of YFP-tagged Kv1.2 (Gu and Gu, 2010). Surprisingly, coexpressed Kvβ2 also enhanced the retrograde transport of YFP-Kv1.2 (Gu and Gu, 2010). Overall, the effect of Kvβ2 is more biased towards the anterograde direction and hence promotes Kv1.2 axonal targeting. The effect of Kvβ2 may act via the functions of a plus end tracking protein EB1 and KIF3A/kinesin II, both of which associate with Kvβ2 (Gu et al., 2006). Disrupting the function of either EB1 or KIF3A impairs both the axonal targeting and anterograde transport of Kv1 channels (Gu et al., 2006; Gu and Gu, 2010) (Fig. 4A).
Fig. 4. Mechanisms underlying Kv1 channel targeting in axons.
(A) Axonal transport of Kv1 channels. The study using live cell imaging on hippocampal neuron axons expressing YFP-Kv1.2 suggests that Kv1/Kvβ channel complexes reside in small round vesicles moving anterogradely along axons. These vesicles are carried by KIF3A/kinesin II motors, where plus end tracking protein EB1 also plays a critical role. It is also possible that KIF5B might transport Kv1 channels anterogradely under some conditions. Kv1 channel-containing vesicles can also be transported retrogradely, which is potentially mediated by dynein complex. How Kv1 channel complex is linked to dynein remains unknown. (B) Targeting of Kv1 channels into the JXP regions in meylinated axons. TAG-1 expressed on myelinating glial and axonal membranes can form homomultimers in trans. TAG-1 can bind to Caspr2 on axonal membranes in cis. Since both Kv1 channels and Caspr2 have PDZ-binding ligand at their C-termini, PDZ domain-containing proteins, such as PSD-95 and PSD-93, may cluster Kv1 channel complexes and Caspr2 together. Therefore, Kv1, Caspr2 and TAG-1 co-cluster in the juxtaparanodal regions of myelinated axons.
KIF5B/kinesin I is also implicated in the axonal targeting of Kv1.3 channels (Rivera et al., 2007) and in forward trafficking of cardiac Kv1.5 channels (Zadeh et al., 2009). How these channels are loaded onto the KIF5B motor remains unclear. The association and transport of different cargos of even the same KIF5 motor can be differentially regulated (Hirokawa et al., 2009; Hirokawa and Takemura, 2005). Moreover, electron microscopy shows multiple tiny bridges linking an intracellular vesicle to microtubules, suggesting that a vesicle cargo can be transported by more than one type of kinesin motor (Hirokawa and Takemura, 2005). Therefore, it is possible that a Kv channel complex can be transported by different motors simultaneously, or by different types of motors in different types of cells, developmental stages and activity states (Fig. 4A).
Members of the MAGUK (membrane-associated guanylate kinase) family, including postsynaptic density protein 95 (PSD-95) and postsynaptic density protein 93 (PSD-93), also play important roles in Kv1 channel trafficking along axons. The interaction between Kv1.4 and PSD-95 was proposed to be critical for the axonal targeting of Kv1.4 channels (Arnold and Clapham, 1999). PSD-93 clusters Kv1 channels at AISs independently of contactin-associated protein 2 (Caspr2) (Ogawa et al., 2008).
4.3. Regulation of Kv1 channel axonal targeting
Targeting of axonal Kv1 channels can be further regulated by various factors. In myelinated axons of both CNS and peripheral nervous system (PNS), Kv1 channels are clustered into JXP regions, controlling the fidelity of AP propagation (Hille, 2001; Salzer, 2003; Wang et al., 1993; Zhou et al., 1998). In the knockout mice of Caspr2 or transient axonal glycoprotein-1 (TAG-1), a glycosylphosphatidylinosital (GPI)-anchored membrane protein, the JXP targeting of Kv1 channels was largely eliminated. Therefore, it was hypothesized that Caspr2 interacts with TAG-1 and clusters Kv1 channels via a PDZ domain-containing protein (Poliak et al., 2003; Traka et al., 2003). Whereas Caspr2 is expressed only in neurons, TAG-1 is expressed in both neurons and myelin cells (Poliak et al., 1999; Traka et al., 2002; Traka et al., 2003). The function of Caspr2/TAG-1 interaction is somewhat analogous to that of Caspr/contactin interaction required for the paranodal formation (Traka et al., 2003) (Fig. 4B). Interestingly, Caspr2 but not Caspr has a PDZ domain ligand at the C-terminus (Poliak et al., 1999; Poliak and Peles, 2003; Salzer, 2003), while both interact with protein 4.1B, which may link the paranodal and JXP adhesion complexes to the axonal cytoskeleton (Horresh et al., 2010). A recent study suggests that the expression of TAG-1 in myelin cells is sufficient to rescue the JXP complex and phenotypes of TAG-1 knockout mice (Savvaki et al., 2010). Thus, in addition to the regulation of axonal and dendritic bundling (Barry et al., 2010), polarized targeting of cell adhesion molecules can regulate the localization of ion channel proteins in response to alteration of cellular environment.
Deleting other proteins with different functions in mice also disrupted precise Kv1 JXP clustering (Bhat et al., 2001; Dupree et al., 1999; Ishibashi et al., 2002; Schaeren-Wiemers et al., 2004; Tanaka et al., 2009; Wang et al., 1995). This suggests their involvement in the Kv1 JXP targeting. Furthermore, a recent study shows that a disintegrin and metalloproteinase 22 (ADAM22) is an axonal component of the Kv1 channel complex that recruits PSD-93 and PSD-95 to juxtaparanodes (Ogawa et al., 2010). However, deleting either ADAM22, PSD-93, or PSD-95 in mice had no effect on the Kv1 JXP targeting (Horresh et al., 2008; Ogawa et al., 2010). The mechanism underlying Kv1 clustering in the JXP regions remains to be fully elucidated.
A recent study shows that cyclin-dependent kinase (Cdk)-mediated phosphorylation of the Kvβ2 subunit regulates Kv1 channel axonal targeting via disrupting the Kvβ2/EB1 interaction (Vacher et al., 2011). Kv1 channel trafficking may also be regulated by other interacting proteins, although whether these regulations occur in mammalian axons remains to be determined in future studies. Axonal Kv1 channels can potentially be regulated by G-protein-coupled receptor initiated signaling. Activation of muscarinic acetycholine receptors activates tyrosine kinase, which phosphorylates Kv1.2 and induces channel endocytosis mediated by cortactin and dynamin, to suppress the channel function (Cachero et al., 1998; Hattan et al., 2002; Nesti et al., 2004; Tsai et al., 1997). Calnexin, an endoplasmic reticulum chaperone, promotes the surface expression of Kv1.2, but not Kv1.1 or Kv1.6, likely through the same forward-trafficking pathway that is facilitated by Kvβ subunits (Manganas and Trimmer, 2004). The leucine-rich glioma inactivated gene 1, recently identified as an associated protein of Kv1.1 channel complex and involved in lateral temporal lobe epilepsy, selectively prevents N-type inactivation mediated by the Kvβ1 subunit (Schulte et al., 2006). Another recent study shows that a GPI-anchored protein, SLEEPLESS (a Ly-6.neurotoxin family member), plays important roles in regulating the levels, localization and activity of Drosophila Shaker channels (Koh et al., 2008; Wu et al., 2010).
5. Axonal targeting of Kv3 (Shaw) channels
5.1. Axonal targeting of endogenous Kv3 channels
The Kv3 subfamily consists of four genes, Kv3.1, Kv3.2, Kv3.3, and Kv3.4. The Kv3.1 and Kv3.2 channels carry sustained currents, whereas the Kv3.3 and Kv3.4 channels carry transient currents (Rudy et al., 1999). They can also form heterotetramers with intermediate gating characteristics (Rudy et al., 1999). Furthermore, Kv3 mRNAs are subjected to extensive alternative splicing, which exclusively gives rise to different C-termini (Luneau et al., 1991). The alternative splicing usually does not affect channel biophysical properties, but affects channel protein targeting.
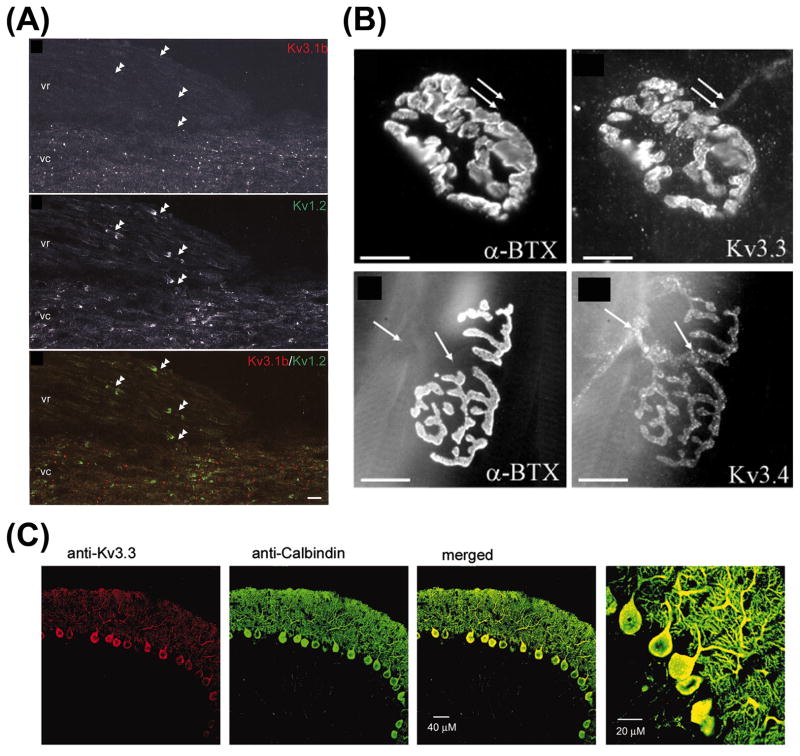
Unlike Kv1 channels, Kv3 channels have prominent presence in both axons and dendrites of neurons in the brain. The axonal and dendritic targeting of Kv3 channels appears to rely on the neuronal type, channel isoform, and alternative splicing. Kv3.1b is found in proximal dendrites, soma, axons, and axonal terminals (Chow et al., 1999; Elezgarai et al., 2003; Goldberg et al., 2005; Ishikawa et al., 2003; Sekirnjak et al., 1997; Weiser et al., 1995). In axons, Kv3.1b is found at some nodes of Ranvier in the CNS but not in the AIS in spinal cord (Devaux et al., 2003) (Fig. 5A). In contrast, Kv3.1a proteins are predominantly expressed in the axons of some of the same neurons expressing Kv3.1b (Ozaita et al., 2002). The shorter form, Kv3.1a, is less abundant, and is down-regulated and replaced gradually by Kv3.1b channels in adults (Gan and Kaczmarek, 1998; Perney et al., 1992).
Fig. 5. Axon-dendrite targeting of Kv3 channels.
(A) Differential localization of Kv3.1b and Kv1.2 in axons of central nervous systems. These are images of longitudinal sections of the ventral column (vc) of unfixed lumbar spinal cord from adult mice. In the spinal cord, the narrow band of nodal Kv3.1b staining (top) is flanked by two broader regions of JXP Kv1.2 staining (middle). The merged image is at the bottom. Note the relative lack of labeling for Kv3.1b in the nodes of peripheral nervous systems (double arrowheads) of the ventral rootlet (vr) (modified from Devaux et al., 2003 with copyright permission from J. Neurosci.). (B) Kv3.3 (flexor digitorum brevis)(top) and Kv3.4 (transverses abdominus)(bottom) localize in presynaptic motor nerve terminals revealed by double labeling with the postsynaptic nicotinic acetylcholine receptors by FITC-α bungarotoxin (left). The presynaptic localization of Kv3.3 and Kv3.4 was further confirmed by electron microscopy studies (modified from Brooke et al., 2004 with copyright permission from Eur. J. Neurosci.). (C) Presence of Kv3.3 in soma and dendrites of Purkinje neurons in rat cerebellum (modified from Martina et al., 2003 with copyright permission from J. Neurosci.).
Kv3.2 is found in non-fast-spiking, somatostatin- and calbindin-containing interneurons (Chow et al., 1999). Kv3.2 is also found in the terminals of basket cells that synapse onto Purkinje cell AISs (Boiko et al., 2003). In a study using Madin-Darby canine kidney cells as a model system, the targeting of Kv3.2 splice variants has been examined (Ponce et al., 1997). Whereas Kv3.2b and Kv3.2c are expressed predominantly in the apical membrane (corresponding to the axonal compartment (Dotti and Simons, 1990)), Kv3.2a localizes to the basolateral membrane (corresponding to the somatodendritic membrane) (Ponce et al., 1997). Therefore, similar to Kv3.1, alternative splicing of Kv3.2 generates functionally similar variants of the same channel with altered subcellular distribution.
Co-localization of different Kv3 subunits in different neurons further generates Kv3 heterotetramer channel complexes with distinct channel properties and targeting patterns. Kv3.3 coexpresses with Kv3.1b in parvalbumin-positive interneurons and is also found in mossy fiber axons of hippocampal dentate granule cells (Chang et al., 2007). Kv3.3 not only exists in axons of cerebellar Purkinje cells and motor nerve terminals (Fig. 5B), but also coexpresses and co-localizes with Kv3.4 in the somatodendritic regions of Purkinje cells to regulate large depolarization events (Chang et al., 2007; Martina et al., 2003) (Fig. 5C). At the calyx of Held terminal, a synapse required for the binaural processing of sound-source localization, Kv3.4 and Kv3.1 channels directly regulate evoked transmitter release (Ishikawa et al., 2003). Kv3.4 is located in nerve terminals of other principal neurons (Brooke et al., 2004a; Brooke et al., 2004b; Rettig et al., 1992; Weiser et al., 1994).
5.2. Mechanisms underlying intrinsic Kv3 axonal targeting
Compared to Kv1 channels, the trafficking mechanism for Kv3 channels is less studied. The C-terminal PDZ domain of Kv3.3 is required for Kv3.3 targeting into distal dendrites of pyramidal neurons of Apteronotus leptorhynchus (Deng et al., 2005). Given the divergence of Kv3 C-terminus, it is possible that other motifs are involved in axonal targeting of Kv3 channel protein complex. By combining hippocampal neuron culture, mutagenesis, and protein biochemistry, we have identified a conditional axonal targeting motif (ATM) in the Kv3.1 C-terminus near the 6th TM segment (Xu et al., 2007). In cultured hippocampal neurons, although the two splice variants of Kv3.1, Kv3.1a and Kv3.1b, are differentially targeted to the somatodendritic and axonal membrane, respectively. They surprisingly share the same lysine-rich and conditional ATM. The ATM directly binds to the T1 domain in a Zn2+-dependent manner, and binds to the ankyrin-repeat domain of ankyrin G. Interestingly, the full-length channel proteins of Kv3.1b display stronger association to ankyrin G than those of Kv3.1a, suggesting that the unique splice domain at Kv3.1b C terminus influences ATM binding to T1 and ankyrin G (Xu et al., 2007).
Because ankyrin G resides at the AIS, where all axonal proteins have to pass through, we propose that ankyrin G may function as a conditional barrier for axon-dendrite targeting of Kv3.1 channels (Xu et al., 2007). Ankyrin G, a critical adaptor protein localized at the AIS, contains 24 N-terminal ankyrin repeats (Bennett and Baines, 2001; Bennett and Chen, 2001; Bouzidi et al., 2002), which are 33-residue motifs involved in protein recognition. Since these ankyrin repeats bind to membrane proteins, this region of ankyrin G is also called the membrane-binding (MB) domain. The ankyrin repeats are followed by a spectrin-binding domain, providing a link to the actin cytoskeleton (Bennett and Chen, 2001). Thus, ankyrin G links the spectrin-based membrane skeleton to integral membrane proteins, including the anion exchanger, the Na+/Ca2+ exchanger, the Na+/K+ ATPase, members of L1-CAM family, Nav channels, and KCNQ channels (Boiko et al., 2003; Davis et al., 1992; Garrido et al., 2003; Jenkins and Bennett, 2001; Pan et al., 2006). Ankyrin G also interacts with Kv3.1b channels. Kv3.1b channels and ankyrin G co-immunoprecipitate from rat brain lysates and co-localize at nodes of Ranvier in the CNS (Devaux et al., 2003).
Consistent with our hypothesis that ankyrin G plays an important role in differential targeting of Kv3.1 splice variants, disrupting ankyrin G function either by over-expressing a dominant-negative mutant or by RNAi knockdown decreases polarized axon-dendrite targeting of both Kv3.1a and Kv3.1b (Xu et al., 2007). Indeed, recent studies show that ankyrin G may generally play a critical role in the maintenance of neuronal axon-dendrite polarity (Hedstrom et al., 2008; Sobotzik et al., 2009; Song et al., 2009). Knocking down endogenous ankyrin G with siRNA in cultured hippocampal neurons allows dendritic proteins to traffic into axons and makes axons adopt some dendritic features including some dendritic spine-like structures (Hedstrom et al., 2008; Song et al., 2009). When ankyrin G is deleted in mouse cerebellum, some Purkinje cell axons develop spine-like structures, which even form asymmetric synapses with other presynaptic axons revealed by electron microscopy (Sobotzik et al., 2009). Therefore, there is compelling evidence supporting our hypothesis that the conditional ATM masked by the T1 domain in Kv3.1a is exposed by the splice domain in Kv3.1b, and is subsequently recognized by ankyrin G to target Kv3.1b into axons (Xu et al., 2007).
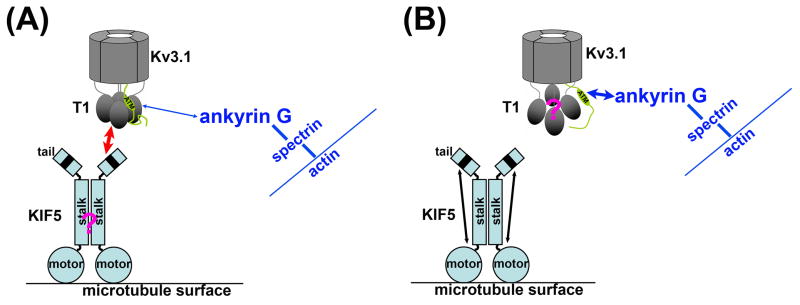
The next important question is how Kv3.1b channels are transported down the axon. There are 45 members in the mammalian kinesin superfamily. Conventional Kinesin I, a major anterograde motor in axons, is composed of a heavy chain (KIF5) dimer and two light chains (KLC). The heavy chains, including KIF5A, KIF5B and KIF5C, have an N-terminal motor domain, followed by a stalk domain responsible for dimerization through coiled-coil regions, and a C-terminal tail domain containing cargo-binding sites (Asbury et al., 2003; Gennerich and Vale, 2009; Goldstein, 2001; Hirokawa and Noda, 2008). KLCs directly bind to a region close to the junction of the stalk and tail domains, mediating the transport of many cargos in axons (Glater et al., 2006; Hirokawa and Takemura, 2005; Setou et al., 2002). In a recent study, we show that KIF5/kinesin I transports Kv3.1 channels through the AIS via direct binding (Xu et al., 2010). We have found that the Kv3.1 N-terminal T1 domain directly binds to a conserved region in KIF5 tail domains. Proper T1 tetramerization is required for the binding. Over-expression of the KIF5B tail domains or suppression of the endogenous KIF5B level by RNA interference markedly reduced Kv3.1b axonal levels. Interestingly, co-expression of YFP-tagged KIF5B assists dendritic Kv3.1a and even mutants with a disrupted ATM to penetrate the AIS. Finally, fluorescently tagged Kv3.1 channels co-localize and co-move with KIF5B along axons revealed by two-color time-lapse imaging. Our data suggest that the binding to KIF5 ensures transport of properly assembled and functioning Kv3.1 channels into axons (Xu et al., 2010).
Because of the high sequence homology, the direct interactions between Kv3.1 C-terminal region and ankyrin G (Xu et al., 2007), and between Kv3.1 T1 domain and the 70-residue region in KIF5 tail (Xu et al., 2010), are likely evolutionarily conserved. Moreover, by interacting with both ankyrin G at the AIS (Xu et al., 2007) and the KIF5 motor, Kv3.1 channels are in a unique position to provide insights into the collaboration of the AIS filter and kinesin motors (Fig. 6). Although Kv3.1 is the only known protein interacting with both ankyrin G and kinesin, the targeting strategy may be shared by many other axonal proteins. The transport efficacy of the motor-cargo complex, but not the cargo or the motor alone, was suggested to determine the axonal entry (Song et al., 2009). The cargo binding to the motor may change the conformation of the motor and affect the recognition of microtubules and the direction of transport (Hirokawa and Takemura, 2005; Setou et al., 2002). Therefore, the binding between the Kv3.1 C-terminus and ankyrin G may build enough tension to change the conformation and hence the activity of KIF5 motors (Fig. 6). The following two interesting questions remain to be determined in future investigation. How does Kv3.1 shift between actin- and microtubule-based trafficking? How might the N/C-terminal interaction of Kv3.1 be involved in this process?
Fig. 6. Local trafficking and long-distance transport of axonal Kv3.1 channels.
A proposed model for the potential interplay of KIF5 and ankyrin G during transportation of Kv3.1 channels through the AIS. (A) A KIF5 motor directly binds to the Kv3.1 T1 tetramer via its C-terminal tail and transports the channel tetramer. Kv3.1 channel may or may not also bind to ankyrin G through its C-terminus assembled together with the T1 tetramer. For clarification, only one C-terminal tail is shown here. (B) Disassembly or conformational change of the T1 tetramer dissociates the channel from KIF5 and allows a stronger binding of ankyrin G to the C-terminus. The process may involve more intermediate states besides the two shown here (modified from Xu et al., 2010 with copyright permission from J. Neurosci.).
5.3. Regulation of Kv3 channel axonal targeting
How the axonal targeting of Kv3 channels is regulated remains unclear. However, there are some clues regarding the regulation of Kv3 channel trafficking. The next important step is to determine whether these regulations influence Kv3 channel axonal targeting. Protein phosphorylation has been shown to regulate the activity and trafficking of Kv3 channels. Whether protein phosphorylation can regulate Kv3 axonal targeting is an intriguing question for future studies. Protein kinase C (PKC) has been implicated in modulating the firing patterns of auditory neurons to adjust to the acoustic environment (Macica et al., 2003; Song et al., 2005; Strumbos et al., 2010). Casein kinase 2 has been shown to alter the voltage dependence of Kv3.1, but the specific residue(s) phosphorylated remains unknown (Macica and Kaczmarek, 2001). Protein kinase A (PKA) has been implicated in histamine-dependent regulation of high-frequency oscillation of hippocampal interneurons (Atzori et al., 2000). The PKC phosphorylation site in Kv3.1b C-terminus is S503 (Macica et al., 2003). A putative PKA phosphorylation site in Kv3.2 channels has also been identified (Rudy and McBain, 2001). Interestingly, a recent study suggested that oscillation of Kv3.1b channel protein levels may alter the AP firing pattern in pacemaker neurons of the suprachiasmatic nucleus (SCN) across the circadian cycle (Itri et al., 2005). During day-to-night transition, Kv3.1b levels decrease, resulting in decreased firing rate of SCN neurons; the decrease may involve endocytosis of channel proteins, leading to altered targeting and possibly degradation of channel proteins. During night-to-day transition, Kv3.1b levels increase, resulting in faster firing of SCN neurons; the increase may involve channel protein synthesis and forward trafficking to the plasma membrane (Itri et al., 2005).
Kv3.1b channel axonal targeting can be further regulated by myelin. It has been shown that Kv3.1b co-localizes with ankyrin G at nodes of Ranvier of central neurons (Devaux et al., 2003). Ankyrin G clusters several proteins, including Nav channels, neurofascin, etc. It has been shown that phosphorylation of Nav channels by casein kinase regulates the interaction with ankyrin G (Brechet et al., 2008). How ankyrin G clusters Kv3.1b channels in response to myelin remains unclear. Taken together, there are several interesting and important questions that remain to be resolved in future studies.
6. Difference and similarity of axonal targeting of Kv1 and Kv3 channels
Kv1 channels are broadly expressed in different cells of both CNS and PNS. Kv3 channels are mainly expressed in GABAergic interneurons as well as some principal neurons. Kv1 and Kv3 are coexpressed in some neurons, such as interneurons, cerebellar Purkinje neurons, auditory neurons, motor neurons, etc. The patterns of their polarized axon-dendrite targeting are different. Kv1 channels are predominantly localized in axons. The presence of Kv1 channels in dendrites is usually restricted to a few particular neurons and at a lower level. In contrast, Kv3 channels are localized in both dendrites and axons, where they play critical roles.
Recent studies suggest that the mechanisms governing Kv1 and Kv3 their axonal transport are likely very different. Whereas EB1 and KIF3A are involved in transporting Kv1 channels, KIF5 appears to be the major anterograde motor transporting Kv3.1 channels (Gu et al., 2006; Gu and Gu, 2010; Xu et al., 2007; Xu et al., 2010). Despite their differences, Kv1 and Kv3 channels do share a common strategy. They are linked to kinesin motor proteins via their T1 domains for long-distance and microtubule-based trafficking, in which proper T1 tetramerization is required (Gu et al., 2003; Gu et al., 2006; Xu et al., 2010). The important role of the N-terminal T1 domains of Kv channels in long-distance trafficking is consistent with the notion that Kv channel T1 domains can form tetramers right after emerging from the ribosomal exiting tunnel once synthesis of the T1-S1 linker is completed (Kosolapov et al., 2004). Their C-terminal domains are linked to local retention or trafficking, either through ankyrin G and the actin cytoskeleton (Xu et al., 2007), or through a tyrosine-based endocytic motif and a PDZ-domain ligand (Lai and Jan, 2006). In contrast, dendritic ion channels are more likely linked to different kinesin motors via their C-termini (Chu et al., 2006; Setou et al., 2000; Setou et al., 2002).
7. Axonal targeting of KCNQ (Kv7) channels
7.1. Axonal targeting of endogenous KCNQ channels
All five members of KCNQ channel family can form homotetramer channels in vitro, but the formation of heterotetramers appears to be restricted to certain combinations (Kubisch et al., 1999; Lerche et al., 2000; Schroeder et al., 2000; Schroeder et al., 1998). The major KCNQ subunits expressed in mammalian brain include KCNQ2, KCNQ3, and KCNQ5. KCNQ4 is found only in a few nuclei and tracts mainly in the brainstem (Kharkovets et al., 2000). KCNQ1 is restricted to the heart, peripheral epithelial and smooth muscle cells.
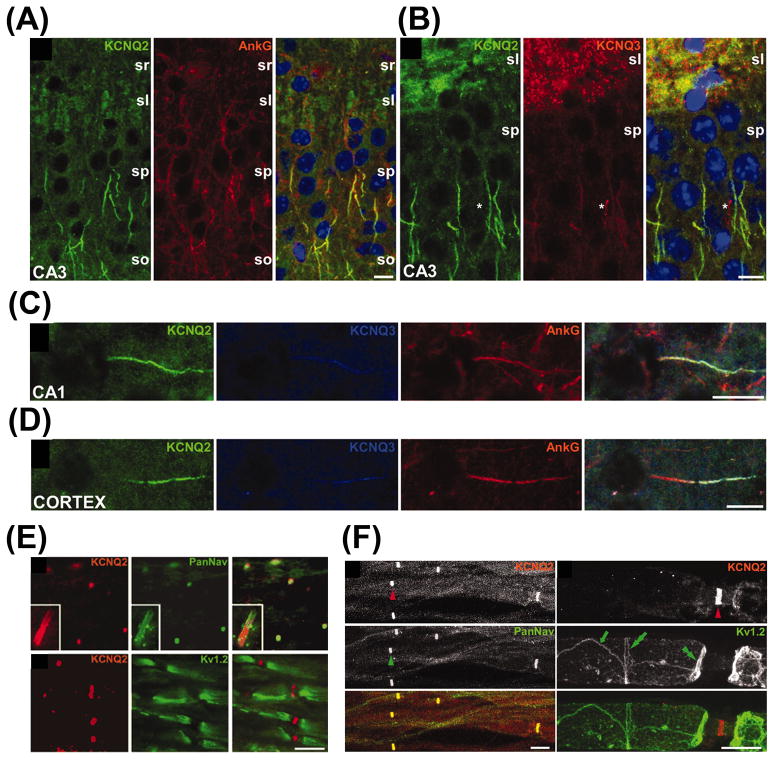
KCNQ2 and KCNQ3 predominantly localize in axons as revealed by immunohistochemistry studies. Previous studies show that they mainly localize in mossy fiber axons but not in dentate granule cell somas in rodent hippocampus (Cooper et al., 2001; Roche et al., 2002). A subset of GABAergic interneurons in hippocampus and some neurons in basal ganglia also express KCNQ2 (Cooper et al., 2001; Roche et al., 2002). KCNQ2 is found at AISs of hippocampal and cortical pyramidal neurons, and at nodes of Ranvier in spinal cord and optic nerves (Devaux et al., 2004; Rasmussen et al., 2007) (Fig. 7). However, in the cerebellum, KCNQ2 appears to localize in Purkinje cell soma and dendrites (Cooper et al., 2001).
Fig. 7. Axonal localization of KCNQ channels.
(A)–(D) Colocalization of KCNQ2 and KCNQ3 in AISs in cortex. These are images of horizontal sections of unfixed mouse brain immunolabeled for KCNQ2, KCNQ3, and ankyrin G; DAPI was used as a nuclear counterstain in (A) and (B). In the CA3 region of the hippocampus, many AISs in the stratum pyramidale (sp), as well as the mossy fibers of the stratum lucidum (sl), are strongly KCNQ2 positive (A). The stratum radiatum (sr) and stratum oriens (so) are indicated. KCNQ3 was found with KCNQ2 in the AISs of some pyramidal cells in CA3 but also in the mossy fibers (B). KCNQ3 colocalized with both ankyrin G and KCNQ2 in the AIS of neurons from the CA1 (C) and temporal neocortex (D). The asterisk in (B) marks a KCNQ3-positive blood vessel. (E) KCNQ2 localized to CNS nodes and initial segments. Anti-KCNQ2 (red), anti-panNav (green, top), and anti-Kv1.2 (green, bottom) antibodies were used to stain rat spinal cord. (F) KCNQ2 is localized to PNS nodes of Ranvier. The staining was performed on unfixed teased fibers from adult rat sciatic nerves (modified from Devaux et al., 2004 with copyright permission from J. Neurosci.).
KCNQ3 displays similar localization patterns to KCNQ2, which is consistent with the notion that KCNQ2 and KCNQ3 form heterotetramers as M channel complexes. KCNQ3 also localizes at AISs and nodes of Ranvier (Devaux et al., 2004; Pan et al., 2006; Rasmussen et al., 2007).
KCNQ5 is widely distributed in the brain, including the hippocampus (Jensen et al., 2005; Lerche et al., 2000; Schroeder et al., 2000; Tzingounis et al., 2010). KCNQ5 is found in presynaptic terminals of the auditory nerve, as well as in dendrites of rat auditory system (Caminos et al., 2007). KCNQ5 also localizes in synaptic endings in auditory brainstem (Garcia-Pino et al., 2010).
7.2. Mechanisms underlying intrinsic KCNQ axonal targeting
A short motif, common to both KCNQ2 and KCNQ3, which mediates ankyrin G interaction and retention of KCNQ2/3 at the AIS, has been identified (Pan et al., 2006). This motif contains several acidic residues and is nearly identical to the sequence in Nav α subunits binding to ankyrin G (Pan et al., 2006). This common ankyrin G-based mechanism, which retains Nav and KCNQ channels at the AIS, was shown to have evolved separately, first Nav, then KCNQ (Hill et al., 2008). Deletion of the ankyrin G-binding motif of KCNQ2 and KCNQ3 disrupts the channel AIS localization (Rasmussen et al., 2007). In the KCNQ2/KCNQ3 heterotetramer channels, the ankyrin G-binding motif of KCNQ3 appears to be dominant over that of KCNQ2 (Rasmussen et al., 2007).
Interestingly, using hippocampal neuron culture, another study shows that KCNQ2/3 channel complexes are preferentially localized to axonal membranes at both proximal and distal axons (Chung et al., 2006). While the AIS targeting of surface KCNQ channels is mediated by the ankyrin G binding motifs of KCNQ2 and KCNQ3, sequences mediating targeting to more distal portion of the axon reside in the membrane proximal and A domains of the KCNQ2 C-terminal tail (Chung et al., 2006) (Fig. 8).
Fig. 8. Motifs of KCNQ channels responsible for axonal targeting.
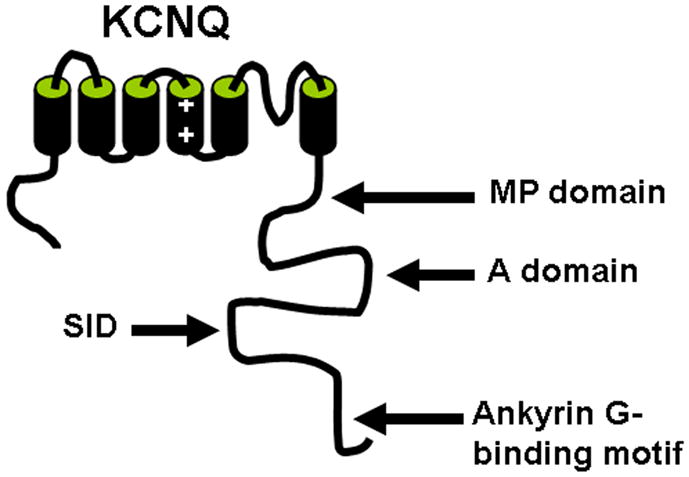
A structure diagram of KCNQ subunit shows the important trafficking motifs in the C-terminus. MP domain, membrane proximal domain (amino acids 323–500); A domain, amino acids 501–579; SID, subunit interaction domain (amino acids 580–623); Ankyrin G-binding domain, amino acids 624–844.
7.3. Regulation of KCNQ axonal targeting
Much less is known regarding the potential regulation of the axonal targeting of KCNQ channels. They may share the regulatory mechanisms for other channel proteins. For instance, like Nav channels, KCNQ channels localize at the nodes of Ranvier, suggesting that myelin potentially regulates the distribution of KCNQ channels via ankyrin G. Protein phosphorylation has been shown to regulate the interaction between ankyrin G and Nav channels (Brechet et al., 2008). Whether protein phosphorylation also regulates KCNQ channel localization remains to be determined. Many other proteins have been identified as regulators of KCNQ channel activity and trafficking, such as calmodulin, phosphatidylinositol 4,5-bisphosphate, the A-kinase anchor proteins 150 and Yotiao, src tyrosine kinase (Dai et al., 2009; Delmas and Brown, 2005). Their potential roles in regulating the channel targeting and functions in axons remain unknown. This may be an interesting topic for future investigation.
8. Axonal targeting of large conductance Ca2+-regulated potassium (BK) channels
8.1. Axonal targeting of endogenous BK channels
BK channels are widely distributed in the nervous systems, where they are localized to neuronal somas, dendrites and presynaptic terminals (Knaus et al., 1996; Marrion and Tavalin, 1998) (Fig. 9). They are powerful regulators of synaptic transmission in central neurons. Deletion of postsynaptic BK channels leads to cerebellar ataxia (abnormal locomotion and motor incoordination) along with Purkinje cell dysfunction in mice (Sausbier et al., 2004). This is most likely due to depolarization-induced inactivation of APs, since BK channels provide the main negative feedback mechanism for timing the bursts of Ca2+ APs, such as those generated in the dendrites of cerebellar Purkinje neurons (Swensen and Bean, 2003; Womack and Khodakhah, 2004). BK channels contribute to the AHP in the soma of pyramidal neurons of the hippocampal CA1 region (Faber and Sah, 2003; Lancaster and Nicoll, 1987). The AHP is a main determinant of the refractory period and hence the overall firing rate of a neuron. In presynaptic terminals, BK channels provide a homeostatic mechanism for regulating synaptic transmission by limiting the influx of extracellular Ca2+ through presynaptic voltage-gated Ca2+ channels (Hu et al., 2001; Raffaelli et al., 2004; Robitaille et al., 1993; Sun et al., 2004; Wang et al., 2001). Moreover, BK channels are also found to modulate pre- and postsynaptic signaling at reciprocal synapses in retina (Grimes et al., 2009).
Fig. 9. Axon-dendrite targeting of BK channels.
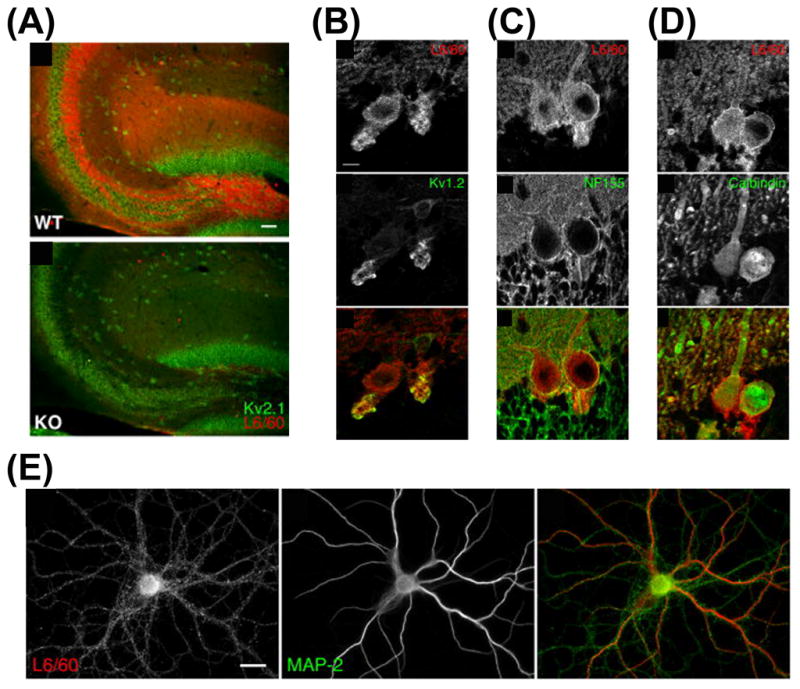
(A) Immunolocalization of BK channels in hippocampus. The monoclonal anti-BK antibody (L6/60) (red) was used to stain the hippocampal sections from wild type mouse (top) and from BK-knockout mouse (bottom). The anti-Kv2.1 staining (green) was used as control. (B)–(D) In rat cerebellar Purkinje cell layer, BK channels are localized in Kv1.2-positive basket cell terminals (B), around the neurofilament-positive AISs (C), and in calbindin-positive Purkinje cell soma and dendrites (D). (E) BK channel staining associates with axons and dendrites of cultured hippocampal neurons (modified from Misonou et al., 2006 with copyright permission from J. Comp. Neurol.).
The existence of BK channels at presynaptic terminals has also been confirmed by immunocytochemistry studies. BK channels are found enriched in axons and nerve terminals in rat brain (Knaus et al., 1996). Using cultured hippocampal neurons and combined with electron microscopy, BK channel staining is found in both presynaptic terminals in axons and postsynaptic sites in dendrites (Sailer et al., 2006). BK channels are associated with glutamatergic synapses in hippocampus and GABAergic synapses in cerebellum (Misonou et al., 2006) (Fig. 9).
8.2. Potential mechanism underlying BK channel axonal targeting
The molecular mechanism underlying the axonal and presynaptic targeting of BK channels remains unknown. With the advance of the research field in channel axonal targeting, we expect that the mystery should be resolved in the near future. Among four types of β subunits (β1-β4), β4 subunits are almost exclusively expressed in the brain (Brenner et al., 2000). β2 and β3 subunits are expressed in the brain and other tissues (Brenner et al., 2000; Wallner et al., 1996), whereas the β1 subunit is primarily localized in smooth muscle cells (Jiang et al., 1999). β subunits not only regulate BK channel activity, including kinetic behavior, voltage dependence, calcium sensitivity, and sensitivity to channel blockers (such as iberiotoxin and charybdotoxin) (Dworetzky et al., 1996; Lippiat et al., 2003), but also regulate channel protein trafficking (Torres et al., 2007). It would be of interest in future studies to determine whether β subunits play a role in targeting BK channels into axons. Furthermore, extensive alternative splicing occurs in BK channels (Fodor and Aldrich, 2009). Whether alternative splicing plays an important role in BK channel targeting is also an interesting question for future studies.
To fully elucidate the basic mechanism governing BK channel axonal targeting, two major questions need to be addressed. First, how are BK channels transported over long distance along axons? The potential motor protein carrying BK channels should be identified first. It would be interesting to determine whether BK channels share the same kinesin motors with Kv1 or Kv3 channels. Second, how are BK channels localized in presynaptic terminals? In this case, potential cytoskeletion associating proteins may be involved. Several such proteins have been identified to interact with BK channels, such as ankyrin-repeat family A protein and microtubule associated protein 1A (Lim and Park, 2005; Park et al., 2004). Whether these interactions are critical for BK channel presynaptic targeting remains unknown. Furthermore, since BK channels are functionally coupled with presynaptic Cav channels, it would be interesting to determine whether BK channels share similar mechanisms with Cav channels in axonal targeting. Once the basic mechanism of BK channel axonal targeting is established, it would be of interest to understand how the channel targeting is regulated in response to neuronal activity.
Many other proteins have been identified to regulate BK channel activity and trafficking, such as membrane-associated guanylate kinase with inverted orientation protein-1, nephrin, the β1 subunit of Na+/K+-ATPase, caveolin-1, canonical transient receptor potential channels, c-Src tyrosine kinase, PKC adaptor protein RACK1, leucine-rich repeat-containing protein 26, PKA, etc (Alioua et al., 2008; Dai et al., 2009; Jha and Dryer, 2009; Kim et al., 2009; Kim et al., 2008; Lim and Park, 2005; Park et al., 2004; Ridgway et al., 2009; Yan and Aldrich, 2010). Whether they play an important role in BK channel axonal targeting remains unknown, but they are good candidates for initial investigation.
9. Future perspective
Despite much progress, an immediate key question in this field is how axonal distribution of various voltage-gated ion channels exactly contributes to electrical signal computation, such as input-output relationship, AP initiation, frequency and failure at branch points, and AP invasion of nerve terminals to trigger neurotransmitter release. Different types of neurons express a distinct set of channel proteins and target them to a different set of subcellular compartments. Therefore, the electrical function of channel targeting might be specific for a particular neural circuit. Unfortunately, this research endeavor is hindered by major technical challenges. Most axons are too small for electrophysiological recording. Optic imaging techniques developed for recording rapid electrical events demand fluorescent probes with high temporal resolution and signal-to-noise ratio. With the rapid advance of electrical and optic recording techniques, this question should become attainable in the near future.
The second major question is to understand how each channel protein is precisely targeted into distinct locations along axons. New mechanistic insights into the axonal targeting of Kv1 and Kv3 channels that have been provided in recent studies (Gu et al., 2003; Gu et al., 2006; Rivera et al., 2007; Rivera et al., 2005; Xu et al., 2007; Gu and Gu, 2010; Xu et al., 2010) may serve as a framework for studies of the mechanism and function of other key axonal ion channels, in understanding long-distance transport and local trafficking/retention. The answer to this question is highly attainable. Over the next ten years, we should have enough information to categorize axonal ion channels and receptors into different groups in terms of the axonal targeting mechanisms utilized. The third question is how the targeting can be regulated during neural development, in an activity-dependent manner, and during pathogenic processes of various neurodegenerative disorders. The long-term impact of this research field may be its contribution to the efforts in drug discovery for curing neurological diseases via modulating axonal electrical and chemical functions.
Acknowledgments
This work was supported by a Career Transition Fellowship Award from the National Multiple Sclerosis Society (Grant TA3012A1) and a grant from National Institute of Neurological Disorders and Stroke/National Institutes of Health (R01NS062720) to C.G.. We thank P. Jukkola for assisting the editing of the manuscript. We apologize to authors whose work is not included in this review due to space constraints.
Abbreviations
- K+
potassium ions
- Ca2+
calcium ions
- CNS
central nervous system
- AP
action potential
- AIS
axon initial segment
- Nav channel
voltage-gated sodium channel
- Kv channel
voltage-gated potassium channel
- Cav channel
voltage-gated calcium channel
- TM
transmembrane
- BK
large conductance calcium-regulated potassium channel
- Kir channel
inwardly rectifying potassium channel
- AHP
afterhyperpolarization
- JXP
juxtaparanodal
- MAGUK
membrane-associated guanylate kinase
- PSD-95
postsynaptic density protein 95
- PSD-93
postsynaptic density protein 93
- Caspr2
contactin-associated protein 2
- PNS
peripheral nervous system
- TAG-1
transient axonal glycoprotein-1
- ADAM22
a disintegrin and metalloproteinase 22
- ATM
axonal targeting motif
- KLC
kinesin light chain
- PKC
protein kinase C
- PKA
protein kinase A
- SCN
suprachiasmatic nucleus
- KIF3
kinesin family member 3
- KIF5
kinesin family member 5
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Alioua A, Lu R, Kumar Y, Eghbali M, Kundu P, Toro L, Stefani E. Slo1 caveolin-binding motif, a mechanism of caveolin-1-Slo1 interaction regulating Slo1 surface expression. J Biol Chem. 2008;283:4808–4817. doi: 10.1074/jbc.M709802200. [DOI] [PubMed] [Google Scholar]
- Arnold DB, Clapham DE. Molecular determinants for subcellular localization of PSD-95 with an interacting K+ channel. Neuron. 1999;23:149–157. doi: 10.1016/s0896-6273(00)80761-8. [DOI] [PubMed] [Google Scholar]
- Asbury CL, Fehr AN, Block SM. Kinesin moves by an asymmetric hand-over-hand mechanism. Science. 2003;302:2130–2134. doi: 10.1126/science.1092985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Atzori M, Lau D, Tansey EP, Chow A, Ozaita A, Rudy B, McBain CJ. H2 histamine receptor-phosphorylation of Kv3.2 modulates interneuron fast spiking. Nat Neurosci. 2000;3:791–798. doi: 10.1038/77693. [DOI] [PubMed] [Google Scholar]
- Barry J, Gu Y, Gu C. Polarized targeting of L1-CAM regulates axonal and dendritic bundling in vitro. Eur J Neurosci. 2010;32:1618–1631. doi: 10.1111/j.1460-9568.2010.07447.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bender KJ, Trussell LO. Axon initial segment Ca2+ channels influence action potential generation and timing. Neuron. 2009;61:259–271. doi: 10.1016/j.neuron.2008.12.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bennett V, Baines AJ. Spectrin and ankyrin-based pathways: metazoan inventions for integrating cells into tissues. Physiol Rev. 2001;81:1353–1392. doi: 10.1152/physrev.2001.81.3.1353. [DOI] [PubMed] [Google Scholar]
- Bennett V, Chen L. Ankyrins and cellular targeting of diverse membrane proteins to physiological sites. Curr Opin Cell Biol. 2001;13:61–67. doi: 10.1016/s0955-0674(00)00175-7. [DOI] [PubMed] [Google Scholar]
- Berkefeld H, Fakler B. Repolarizing responses of BKCa-Cav complexes are distinctly shaped by their Cav subunits. J Neurosci. 2008;28:8238–8245. doi: 10.1523/JNEUROSCI.2274-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berkefeld H, Sailer CA, Bildl W, Rohde V, Thumfart JO, Eble S, Klugbauer N, Reisinger E, Bischofberger J, Oliver D, Knaus HG, Schulte U, Fakler B. BKCa-Cav channel complexes mediate rapid and localized Ca2+-activated K+ signaling. Science. 2006;314:615–620. doi: 10.1126/science.1132915. [DOI] [PubMed] [Google Scholar]
- Bhat MA, Rios JC, Lu Y, Garcia-Fresco GP, Ching W, St Martin M, Li J, Einheber S, Chesler M, Rosenbluth J, Salzer JL, Bellen HJ. Axon-glia interactions and the domain organization of myelinated axons requires neurexin IV/Caspr/Paranodin. Neuron. 2001;30:369–383. doi: 10.1016/s0896-6273(01)00294-x. [DOI] [PubMed] [Google Scholar]
- Bichet D, Haass FA, Jan LY. Merging functional studies with structures of inward-rectifier K+ channels. Nat Rev Neurosci. 2003;4:957–967. doi: 10.1038/nrn1244. [DOI] [PubMed] [Google Scholar]
- Boiko T, Van Wart A, Caldwell JH, Levinson SR, Trimmer JS, Matthews G. Functional specialization of the axon initial segment by isoform-specific sodium channel targeting. J Neurosci. 2003;23:2306–2313. doi: 10.1523/JNEUROSCI.23-06-02306.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bouzidi M, Tricaud N, Giraud P, Kordeli E, Caillol G, Deleuze C, Couraud F, Alcaraz G. Interaction of the Nav1.2a subunit of the voltage-dependent sodium channel with nodal ankyrinG. In vitro mapping of the interacting domains and association in synaptosomes. J Biol Chem. 2002;277:28996–29004. doi: 10.1074/jbc.M201760200. [DOI] [PubMed] [Google Scholar]
- Brechet A, Fache MP, Brachet A, Ferracci G, Baude A, Irondelle M, Pereira S, Leterrier C, Dargent B. Protein kinase CK2 contributes to the organization of sodium channels in axonal membranes by regulating their interactions with ankyrin G. J Cell Biol. 2008;183:1101–1114. doi: 10.1083/jcb.200805169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brenner R, Jegla TJ, Wickenden A, Liu Y, Aldrich RW. Cloning and functional characterization of novel large conductance calcium-activated potassium channel beta subunits, hKCNMB3 and hKCNMB4. J Biol Chem. 2000;275:6453–6461. doi: 10.1074/jbc.275.9.6453. [DOI] [PubMed] [Google Scholar]
- Brooke RE, Atkinson L, Batten TF, Deuchars SA, Deuchars J. Association of potassium channel Kv3.4 subunits with pre- and post-synaptic structures in brainstem and spinal cord. Neuroscience. 2004a;126:1001–1010. doi: 10.1016/j.neuroscience.2004.03.051. [DOI] [PubMed] [Google Scholar]
- Brooke RE, Moores TS, Morris NP, Parson SH, Deuchars J. Kv3 voltage-gated potassium channels regulate neurotransmitter release from mouse motor nerve terminals. Eur J Neurosci. 2004b;20:3313–3321. doi: 10.1111/j.1460-9568.2004.03730.x. [DOI] [PubMed] [Google Scholar]
- Brown DA, Adams PR. Muscarinic suppression of a novel voltage-sensitive K+ current in a vertebrate neurone. Nature. 1980;283:673–676. doi: 10.1038/283673a0. [DOI] [PubMed] [Google Scholar]
- Cachero TG, Morielli AD, Peralta EG. The small GTP-binding protein RhoA regulates a delayed rectifier potassium channel. Cell. 1998;93:1077–1085. doi: 10.1016/s0092-8674(00)81212-x. [DOI] [PubMed] [Google Scholar]
- Caminos E, Garcia-Pino E, Martinez-Galan JR, Juiz JM. The potassium channel KCNQ5/Kv7.5 is localized in synaptic endings of auditory brainstem nuclei of the rat. J Comp Neurol. 2007;505:363–378. doi: 10.1002/cne.21497. [DOI] [PubMed] [Google Scholar]
- Chandy KG. Simplified gene nomenclature. Nature. 1991;352:26. doi: 10.1038/352026b0. [DOI] [PubMed] [Google Scholar]
- Chang SY, Zagha E, Kwon ES, Ozaita A, Bobik M, Martone ME, Ellisman MH, Heintz N, Rudy B. Distribution of Kv3.3 potassium channel subunits in distinct neuronal populations of mouse brain. J Comp Neurol. 2007;502:953–972. doi: 10.1002/cne.21353. [DOI] [PubMed] [Google Scholar]
- Chittajallu R, Chen Y, Wang H, Yuan X, Ghiani CA, Heckman T, McBain CJ, Gallo V. Regulation of Kv1 subunit expression in oligodendrocyte progenitor cells and their role in G1/S phase progression of the cell cycle. Proc Natl Acad Sci U S A. 2002;99:2350–2355. doi: 10.1073/pnas.042698399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chow A, Erisir A, Farb C, Nadal MS, Ozaita A, Lau D, Welker E, Rudy B. K+ channel expression distinguishes subpopulations of parvalbumin- and somatostatin-containing neocortical interneurons. J Neurosci. 1999;19:9332–9345. doi: 10.1523/JNEUROSCI.19-21-09332.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chu PJ, Rivera JF, Arnold DB. A role for Kif17 in transport of Kv4.2. J Biol Chem. 2006;281:365–373. doi: 10.1074/jbc.M508897200. [DOI] [PubMed] [Google Scholar]
- Chung HJ, Jan YN, Jan LY. Polarized axonal surface expression of neuronal KCNQ channels is mediated by multiple signals in the KCNQ2 and KCNQ3 C-terminal domains. Proc Natl Acad Sci U S A. 2006;103:8870–8875. doi: 10.1073/pnas.0603376103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coetzee WA, Amarillo Y, Chiu J, Chow A, Lau D, McCormack T, Moreno H, Nadal MS, Ozaita A, Pountney D, Saganich M, Vega-Saenz de Miera E, Rudy B. Molecular diversity of K+ channels. Ann N Y Acad Sci. 1999;868:233–285. doi: 10.1111/j.1749-6632.1999.tb11293.x. [DOI] [PubMed] [Google Scholar]
- Cooper EC, Harrington E, Jan YN, Jan LY. M channel KCNQ2 subunits are localized to key sites for control of neuronal network oscillations and synchronization in mouse brain. J Neurosci. 2001;21:9529–9540. doi: 10.1523/JNEUROSCI.21-24-09529.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cooper EC, Milroy A, Jan YN, Jan LY, Lowenstein DH. Presynaptic localization of Kv1.4-containing A-type potassium channels near excitatory synapses in the hippocampus. J Neurosci. 1998;18:965–974. doi: 10.1523/JNEUROSCI.18-03-00965.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dai S, Hall DD, Hell JW. Supramolecular assemblies and localized regulation of voltage-gated ion channels. Physiol Rev. 2009;89:411–452. doi: 10.1152/physrev.00029.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis LH, Davis JQ, Bennett V. Ankyrin regulation: an alternatively spliced segment of the regulatory domain functions as an intramolecular modulator. J Biol Chem. 1992;267:18966–18972. [PubMed] [Google Scholar]
- Debanne D. Information processing in the axon. Nat Rev Neurosci. 2004;5:304–316. doi: 10.1038/nrn1397. [DOI] [PubMed] [Google Scholar]
- Delmas P, Brown DA. Pathways modulating neural KCNQ/M (Kv7) potassium channels. Nat Rev Neurosci. 2005;6:850–862. doi: 10.1038/nrn1785. [DOI] [PubMed] [Google Scholar]
- Deng Q, Rashid AJ, Fernandez FR, Turner RW, Maler L, Dunn RJ. A C-terminal domain directs Kv3.3 channels to dendrites. J Neurosci. 2005;25:11531–11541. doi: 10.1523/JNEUROSCI.3672-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Devaux J, Alcaraz G, Grinspan J, Bennett V, Joho R, Crest M, Scherer SS. Kv3.1b is a novel component of CNS nodes. J Neurosci. 2003;23:4509–4518. doi: 10.1523/JNEUROSCI.23-11-04509.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Devaux JJ, Kleopa KA, Cooper EC, Scherer SS. KCNQ2 is a nodal K+ channel. J Neurosci. 2004;24:1236–1244. doi: 10.1523/JNEUROSCI.4512-03.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dodson PD, Barker MC, Forsythe ID. Two heteromeric Kv1 potassium channels differentially regulate action potential firing. J Neurosci. 2002;22:6953–6961. doi: 10.1523/JNEUROSCI.22-16-06953.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dodson PD, Billups B, Rusznak Z, Szucs G, Barker MC, Forsythe ID. Presynaptic rat Kv1.2 channels suppress synaptic terminal hyperexcitability following action potential invasion. J Physiol. 2003;550:27–33. doi: 10.1113/jphysiol.2003.046250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dotti CG, Simons K. Polarized sorting of viral glycoproteins to the axon and dendrites of hippocampal neurons in culture. Cell. 1990;62:63–72. doi: 10.1016/0092-8674(90)90240-f. [DOI] [PubMed] [Google Scholar]
- Dupree JL, Girault JA, Popko B. Axo-glial interactions regulate the localization of axonal paranodal proteins. J Cell Biol. 1999;147:1145–1152. doi: 10.1083/jcb.147.6.1145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dworetzky SI, Boissard CG, Lum-Ragan JT, McKay MC, Post-Munson DJ, Trojnacki JT, Chang CP, Gribkoff VK. Phenotypic alteration of a human BK (hSlo) channel by hSlobeta subunit coexpression: changes in blocker sensitivity, activation/relaxation and inactivation kinetics, and protein kinase A modulation. J Neurosci. 1996;16:4543–4550. doi: 10.1523/JNEUROSCI.16-15-04543.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elezgarai I, Diez J, Puente N, Azkue JJ, Benitez R, Bilbao A, Knopfel T, Donate-Oliver F, Grandes P. Subcellular localization of the voltage-dependent potassium channel Kv3.1b in postnatal and adult rat medial nucleus of the trapezoid body. Neuroscience. 2003;118:889–898. doi: 10.1016/s0306-4522(03)00068-x. [DOI] [PubMed] [Google Scholar]
- Enyedi P, Czirjak G. Molecular background of leak K+ currents: two-pore domain potassium channels. Physiol Rev. 2010;90:559–605. doi: 10.1152/physrev.00029.2009. [DOI] [PubMed] [Google Scholar]
- Faber ES, Sah P. Calcium-activated potassium channels: multiple contributions to neuronal function. Neuroscientist. 2003:181–194. doi: 10.1177/1073858403009003011. [DOI] [PubMed] [Google Scholar]
- Fodor AA, Aldrich RW. Convergent evolution of alternative splices at domain boundaries of the BK channel. Annu Rev Physiol. 2009;71:19–36. doi: 10.1146/annurev.physiol.010908.163124. [DOI] [PubMed] [Google Scholar]
- Gan L, Kaczmarek LK. When, where, and how much? Expression of the Kv3.1 potassium channel in high-frequency firing neurons. J Neurobiol. 1998;37:69–79. doi: 10.1002/(sici)1097-4695(199810)37:1<69::aid-neu6>3.0.co;2-6. [DOI] [PubMed] [Google Scholar]
- Garcia-Pino E, Caminos E, Juiz JM. KCNQ5 reaches synaptic endings in the auditory brainstem at hearing onset and targeting maintenance is activity-dependent. J Comp Neurol. 2010;518:1301–1314. doi: 10.1002/cne.22276. [DOI] [PubMed] [Google Scholar]
- Garrido JJ, Giraud P, Carlier E, Fernandes F, Moussif A, Fache MP, Debanne D, Dargent B. A targeting motif involved in sodium channel clustering at the axonal initial segment. Science. 2003;300:2091–2094. doi: 10.1126/science.1085167. [DOI] [PubMed] [Google Scholar]
- Gazula VR, Strumbos JG, Mei X, Chen H, Rahner C, Kaczmarek LK. Localization of Kv1.3 channels in presynaptic terminals of brainstem auditory neurons. J Comp Neurol. 2010;518:3205–3220. doi: 10.1002/cne.22393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geiger JR, Jonas P. Dynamic control of presynaptic Ca2+ inflow by fast-inactivating K+ channels in hippocampal mossy fiber boutons. Neuron. 2000;28:927–939. doi: 10.1016/s0896-6273(00)00164-1. [DOI] [PubMed] [Google Scholar]
- Gennerich A, Vale RD. Walking the walk: how kinesin and dynein coordinate their steps. Curr Opin Cell Biol. 2009;21:59–67. doi: 10.1016/j.ceb.2008.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glater EE, Megeath LJ, Stowers RS, Schwarz TL. Axonal transport of mitochondria requires milton to recruit kinesin heavy chain and is light chain independent. J Cell Biol. 2006;173:545–557. doi: 10.1083/jcb.200601067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldberg EM, Clark BD, Zagha E, Nahmani M, Erisir A, Rudy B. K+ channels at the axon initial segment dampen near-threshold excitability of neocortical fast-spiking GABAergic interneurons. Neuron. 2008;58:387–400. doi: 10.1016/j.neuron.2008.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldberg EM, Watanabe S, Chang SY, Joho RH, Huang ZJ, Leonard CS, Rudy B. Specific functions of synaptically localized potassium channels in synaptic transmission at the neocortical GABAergic fast-spiking cell synapse. J Neurosci. 2005;25:5230–5235. doi: 10.1523/JNEUROSCI.0722-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldstein LS. Kinesin molecular motors: transport pathways, receptors, and human disease. Proc Natl Acad Sci U S A. 2001;98:6999–7003. doi: 10.1073/pnas.111145298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldstein SA, Bockenhauer D, O’Kelly I, Zilberberg N. Potassium leak channels and the KCNK family of two-P-domain subunits. Nat Rev Neurosci. 2001;2:175–184. doi: 10.1038/35058574. [DOI] [PubMed] [Google Scholar]
- Grimes WN, Li W, Chavez AE, Diamond JS. BK channels modulate pre- and postsynaptic signaling at reciprocal synapses in retina. Nat Neurosci. 2009;12:585–592. doi: 10.1038/nn.2302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grosse G, Draguhn A, Hohne L, Tapp R, Veh RW, Ahnert-Hilger G. Expression of Kv1 potassium channels in mouse hippocampal primary cultures: development and activity-dependent regulation. J Neurosci. 2000;20:1869–1882. doi: 10.1523/JNEUROSCI.20-05-01869.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grubb MS, Burrone J. Activity-dependent relocation of the axon initial segment fine-tunes neuronal excitability. Nature. 2010;465:1070–1074. doi: 10.1038/nature09160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gu C, Jan YN, Jan LY. A conserved domain in axonal targeting of Kv1 (Shaker) voltage-gated potassium channels. Science. 2003;301:646–649. doi: 10.1126/science.1086998. [DOI] [PubMed] [Google Scholar]
- Gu C, Zhou W, Puthenveedu MA, Xu M, Jan YN, Jan LY. The microtubule plus-end tracking protein EB1 is required for Kv1 voltage-gated K+ channel axonal targeting. Neuron. 2006;52:803–816. doi: 10.1016/j.neuron.2006.10.022. [DOI] [PubMed] [Google Scholar]
- Gu Y, Gu C. Dynamics of Kv1 channel transport in axons. PLoS One. 2010;5:e11931. doi: 10.1371/journal.pone.0011931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gutman GA, Chandy KG, Grissmer S, Lazdunski M, McKinnon D, Pardo LA, Robertson GA, Rudy B, Sanguinetti MC, Stuhmer W, Wang X. International Union of Pharmacology. LIII. Nomenclature and molecular relationships of voltage-gated potassium channels. Pharmacol Rev. 2005;57:473–508. doi: 10.1124/pr.57.4.10. [DOI] [PubMed] [Google Scholar]
- Hattan D, Nesti E, Cachero TG, Morielli AD. Tyrosine phosphorylation of Kv1.2 modulates its interaction with the actin-binding protein cortactin. J Biol Chem. 2002;277:38596–38606. doi: 10.1074/jbc.M205005200. [DOI] [PubMed] [Google Scholar]
- Hedstrom KL, Ogawa Y, Rasband MN. AnkyrinG is required for maintenance of the axon initial segment and neuronal polarity. J Cell Biol. 2008;183:635–640. doi: 10.1083/jcb.200806112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hibino H, Inanobe A, Furutani K, Murakami S, Findlay I, Kurachi Y. Inwardly rectifying potassium channels: their structure, function, and physiological roles. Physiol Rev. 2010;90:291–366. doi: 10.1152/physrev.00021.2009. [DOI] [PubMed] [Google Scholar]
- Hill AS, Nishino A, Nakajo K, Zhang G, Fineman JR, Selzer ME, Okamura Y, Cooper EC. Ion channel clustering at the axon initial segment and node of Ranvier evolved sequentially in early chordates. PLoS Genet. 2008;4:e1000317. doi: 10.1371/journal.pgen.1000317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hille B. Ion Channels of Excitable Membranes. 3. Ion Channels of Ecitable Membranes; 2001. [Google Scholar]
- Hirokawa N, Noda Y. Intracellular transport and kinesin superfamily proteins, KIFs: structure, function, and dynamics. Physiol Rev. 2008;88:1089–1118. doi: 10.1152/physrev.00023.2007. [DOI] [PubMed] [Google Scholar]
- Hirokawa N, Noda Y, Tanaka Y, Niwa S. Kinesin superfamily motor proteins and intracellular transport. Nat Rev Mol Cell Biol. 2009;10:682–696. doi: 10.1038/nrm2774. [DOI] [PubMed] [Google Scholar]
- Hirokawa N, Takemura R. Molecular motors and mechanisms of directional transport in neurons. Nat Rev Neurosci. 2005;6:201–214. doi: 10.1038/nrn1624. [DOI] [PubMed] [Google Scholar]
- Horresh I, Bar V, Kissil JL, Peles E. Organization of myelinated axons by Caspr and Caspr2 requires the cytoskeletal adapter protein 4.1B. J Neurosci. 2010;30:2480–2489. doi: 10.1523/JNEUROSCI.5225-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horresh I, Poliak S, Grant S, Bredt D, Rasband MN, Peles E. Multiple molecular interactions determine the clustering of Caspr2 and Kv1 channels in myelinated axons. J Neurosci. 2008;28:14213–14222. doi: 10.1523/JNEUROSCI.3398-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu H, Shao LR, Chavoshy S, Gu N, Trieb M, Behrens R, Laake P, Pongs O, Knaus HG, Ottersen OP, Storm JF. Presynaptic Ca2+-activated K+ channels in glutamatergic hippocampal terminals and their role in spike repolarization and regulation of transmitter release. J Neurosci. 2001;21:9585–9597. doi: 10.1523/JNEUROSCI.21-24-09585.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu W, Tian C, Li T, Yang M, Hou H, Shu Y. Distinct contributions of Na(v)1.6 and Na(v)1.2 in action potential initiation and backpropagation. Nat Neurosci. 2009;12:996–1002. doi: 10.1038/nn.2359. [DOI] [PubMed] [Google Scholar]
- Inda MC, DeFelipe J, Munoz A. Voltage-gated ion channels in the axon initial segment of human cortical pyramidal cells and their relationship with chandelier cells. Proc Natl Acad Sci U S A. 2006;103:2920–2925. doi: 10.1073/pnas.0511197103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ishibashi T, Dupree JL, Ikenaka K, Hirahara Y, Honke K, Peles E, Popko B, Suzuki K, Nishino H, Baba H. A myelin galactolipid, sulfatide, is essential for maintenance of ion channels on myelinated axon but not essential for initial cluster formation. J Neurosci. 2002;22:6507–6514. doi: 10.1523/JNEUROSCI.22-15-06507.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ishikawa T, Nakamura Y, Saitoh N, Li WB, Iwasaki S, Takahashi T. Distinct roles of Kv1 and Kv3 potassium channels at the calyx of Held presynaptic terminal. J Neurosci. 2003;23:10445–10453. doi: 10.1523/JNEUROSCI.23-32-10445.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Itri JN, Michel S, Vansteensel MJ, Meijer JH, Colwell CS. Fast delayed rectifier potassium current is required for circadian neural activity. Nat Neurosci. 2005;8:650–656. doi: 10.1038/nn1448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jenkins SM, Bennett V. Ankyrin-G coordinates assembly of the spectrin-based membrane skeleton, voltage-gated sodium channels, and L1 CAMs at Purkinje neuron initial segments. J Cell Biol. 2001;155:739–746. doi: 10.1083/jcb.200109026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jensen HS, Callo K, Jespersen T, Jensen BS, Olesen SP. The KCNQ5 potassium channel from mouse: a broadly expressed M-current like potassium channel modulated by zinc, pH, and volume changes. Brain Res Mol Brain Res. 2005;139:52–62. doi: 10.1016/j.molbrainres.2005.05.007. [DOI] [PubMed] [Google Scholar]
- Jentsch TJ. Neuronal KCNQ potassium channels: physiology and role in disease. Nat Rev Neurosci. 2000;1:21–30. doi: 10.1038/35036198. [DOI] [PubMed] [Google Scholar]
- Jha S, Dryer SE. The beta1 subunit of Na+/K+-ATPase interacts with BKCa channels and affects their steady-state expression on the cell surface. FEBS Lett. 2009;583:3109–3114. doi: 10.1016/j.febslet.2009.08.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiang Z, Wallner M, Meera P, Toro L. Human and rodent MaxiK channel beta-subunit genes: cloning and characterization. Genomics. 1999;55:57–67. doi: 10.1006/geno.1998.5627. [DOI] [PubMed] [Google Scholar]
- Johnston J, Griffin SJ, Baker C, Skrzypiec A, Chernova T, Forsythe ID. Initial segment Kv2.2 channels mediate a slow delayed rectifier and maintain high frequency action potential firing in medial nucleus of the trapezoid body neurons. J Physiol. 2008;586:3493–3509. doi: 10.1113/jphysiol.2008.153734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jonas P, Bischofberger J, Fricker D, Miles R. Interneuron Diversity series: Fast in, fast out--temporal and spatial signal processing in hippocampal interneurons. Trends Neurosci. 2004;27:30–40. doi: 10.1016/j.tins.2003.10.010. [DOI] [PubMed] [Google Scholar]
- Kalman K, Nguyen A, Tseng-Crank J, Dukes ID, Chandy G, Hustad CM, Copeland NG, Jenkins NA, Mohrenweiser H, Brandriff B, Cahalan M, Gutman GA, Chandy KG. Genomic organization, chromosomal localization, tissue distribution, and biophysical characterization of a novel mammalian Shaker-related voltage-gated potassium channel, Kv1.7. J Biol Chem. 1998;273:5851–5857. doi: 10.1074/jbc.273.10.5851. [DOI] [PubMed] [Google Scholar]
- Kharkovets T, Hardelin JP, Safieddine S, Schweizer M, El-Amraoui A, Petit C, Jentsch TJ. KCNQ4, a K+ channel mutated in a form of dominant deafness, is expressed in the inner ear and the central auditory pathway. Proc Natl Acad Sci U S A. 2000;97:4333–4338. doi: 10.1073/pnas.97.8.4333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim EY, Alvarez-Baron CP, Dryer SE. Canonical transient receptor potential channel (TRPC)3 and TRPC6 associate with large-conductance Ca2+-activated K+ (BKCa) channels: role in BKCa trafficking to the surface of cultured podocytes. Mol Pharmacol. 2009;75:466–477. doi: 10.1124/mol.108.051912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim EY, Choi KJ, Dryer SE. Nephrin binds to the COOH terminus of a large-conductance Ca2+-activated K+ channel isoform and regulates its expression on the cell surface. Am J Physiol Renal Physiol. 2008;295:F235–246. doi: 10.1152/ajprenal.00140.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knaus HG, Schwarzer C, Koch RO, Eberhart A, Kaczorowski GJ, Glossmann H, Wunder F, Pongs O, Garcia ML, Sperk G. Distribution of high-conductance Ca2+-activated K+ channels in rat brain: targeting to axons and nerve terminals. J Neurosci. 1996;16:955–963. doi: 10.1523/JNEUROSCI.16-03-00955.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koch RO, Wanner SG, Koschak A, Hanner M, Schwarzer C, Kaczorowski GJ, Slaughter RS, Garcia ML, Knaus HG. Complex subunit assembly of neuronal voltage-gated K+ channels. Basis for high-affinity toxin interactions and pharmacology. J Biol Chem. 1997;272:27577–27581. doi: 10.1074/jbc.272.44.27577. [DOI] [PubMed] [Google Scholar]
- Koh K, Joiner WJ, Wu MN, Yue Z, Smith CJ, Sehgal A. Identification of SLEEPLESS, a sleep-promoting factor. Science. 2008;321:372–376. doi: 10.1126/science.1155942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kole MH, Letzkus JJ, Stuart GJ. Axon initial segment Kv1 channels control axonal action potential waveform and synaptic efficacy. Neuron. 2007;55:633–647. doi: 10.1016/j.neuron.2007.07.031. [DOI] [PubMed] [Google Scholar]
- Kosolapov A, Tu L, Wang J, Deutsch C. Structure acquisition of the T1 domain of Kv1.3 during biogenesis. Neuron. 2004;44:295–307. doi: 10.1016/j.neuron.2004.09.011. [DOI] [PubMed] [Google Scholar]
- Kuba H, Oichi Y, Ohmori H. Presynaptic activity regulates Na+ channel distribution at the axon initial segment. Nature. 2010;465:1075–1078. doi: 10.1038/nature09087. [DOI] [PubMed] [Google Scholar]
- Kubisch C, Schroeder BC, Friedrich T, Lutjohann B, El-Amraoui A, Marlin S, Petit C, Jentsch TJ. KCNQ4, a novel potassium channel expressed in sensory outer hair cells, is mutated in dominant deafness. Cell. 1999;96:437–446. doi: 10.1016/s0092-8674(00)80556-5. [DOI] [PubMed] [Google Scholar]
- Lai HC, Jan LY. The distribution and targeting of neuronal voltage-gated ion channels. Nat Rev Neurosci. 2006;7:548–562. doi: 10.1038/nrn1938. [DOI] [PubMed] [Google Scholar]
- Lancaster B, Nicoll RA. Properties of two calcium-activated hyperpolarizations in rat hippocampal neurones. J Physiol. 1987;389:187–203. doi: 10.1113/jphysiol.1987.sp016653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lerche C, Scherer CR, Seebohm G, Derst C, Wei AD, Busch AE, Steinmeyer K. Molecular cloning and functional expression of KCNQ5, a potassium channel subunit that may contribute to neuronal M-current diversity. J Biol Chem. 2000;275:22395–22400. doi: 10.1074/jbc.M002378200. [DOI] [PubMed] [Google Scholar]
- Lesage F, Guillemare E, Fink M, Duprat F, Lazdunski M, Romey G, Barhanin J. TWIK-1, a ubiquitous human weakly inward rectifying K+ channel with a novel structure. EMBO J. 1996;15:1004–1011. [PMC free article] [PubMed] [Google Scholar]
- Li X, Ascoli GA. Computational simulation of the input-output relationship in hippocampal pyramidal cells. J Comput Neurosci. 2006;21:191–209. doi: 10.1007/s10827-006-8797-z. [DOI] [PubMed] [Google Scholar]
- Li Y, Um SY, McDonald TV. Voltage-gated potassium channels: regulation by accessory subunits. Neuroscientist. 2006;12:199–210. doi: 10.1177/1073858406287717. [DOI] [PubMed] [Google Scholar]
- Lim HH, Park CS. Identification and functional characterization of ankyrin-repeat family protein ANKRA as a protein interacting with BKCa channel. Mol Biol Cell. 2005;16:1013–1025. doi: 10.1091/mbc.E04-06-0537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lippiat JD, Standen NB, Harrow ID, Phillips SC, Davies NW. Properties of BK(Ca) channels formed by bicistronic expression of hSloalpha and beta1–4 subunits in HEK293 cells. J Membr Biol. 2003;192:141–148. doi: 10.1007/s00232-002-1070-0. [DOI] [PubMed] [Google Scholar]
- Lorincz A, Nusser Z. Cell-type-dependent molecular composition of the axon initial segment. J Neurosci. 2008;28:14329–14340. doi: 10.1523/JNEUROSCI.4833-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loussouarn G, Rose T, Nichols CG. Structural basis of inward rectifying potassium channel gating. Trends Cardiovasc Med. 2002;12:253–258. doi: 10.1016/s1050-1738(02)00170-6. [DOI] [PubMed] [Google Scholar]
- Luisi R, Panza E, Barrese V, Iannotti FA, Viggiano D, Secondo A, Canzoniero LM, Martire M, Annunziato L, Taglialatela M. Activation of pre-synaptic M-type K+ channels inhibits [3H]D-aspartate release by reducing Ca2+ entry through P/Q-type voltage-gated Ca2+ channels. J Neurochem. 2009;109:168–181. doi: 10.1111/j.1471-4159.2009.05945.x. [DOI] [PubMed] [Google Scholar]
- Luneau CJ, Williams JB, Marshall J, Levitan ES, Oliva C, Smith JS, Antanavage J, Folander K, Stein RB, Swanson R, et al. Alternative splicing contributes to K+ channel diversity in the mammalian central nervous system. Proc Natl Acad Sci U S A. 1991;88:3932–3936. doi: 10.1073/pnas.88.9.3932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luscher C, Slesinger PA. Emerging roles for G protein-gated inwardly rectifying potassium (GIRK) channels in health and disease. Nat Rev Neurosci. 2010;11:301–315. doi: 10.1038/nrn2834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Macica CM, Kaczmarek LK. Casein kinase 2 determines the voltage dependence of the Kv3.1 channel in auditory neurons and transfected cells. J Neurosci. 2001;21:1160–1168. doi: 10.1523/JNEUROSCI.21-04-01160.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Macica CM, von Hehn CA, Wang LY, Ho CS, Yokoyama S, Joho RH, Kaczmarek LK. Modulation of the kv3.1b potassium channel isoform adjusts the fidelity of the firing pattern of auditory neurons. J Neurosci. 2003;23:1133–1141. doi: 10.1523/JNEUROSCI.23-04-01133.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manganas LN, Trimmer JS. Calnexin regulates mammalian Kv1 channel trafficking. Biochem Biophys Res Commun. 2004;322:577–584. doi: 10.1016/j.bbrc.2004.06.182. [DOI] [PubMed] [Google Scholar]
- Marrion NV, Tavalin SJ. Selective activation of Ca2+-activated K+ channels by co-localized Ca2+ channels in hippocampal neurons. Nature. 1998;395:900–905. doi: 10.1038/27674. [DOI] [PubMed] [Google Scholar]
- Martina M, Yao GL, Bean BP. Properties and functional role of voltage-dependent potassium channels in dendrites of rat cerebellar Purkinje neurons. J Neurosci. 2003;23:5698–5707. doi: 10.1523/JNEUROSCI.23-13-05698.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martire M, Castaldo P, D’Amico M, Preziosi P, Annunziato L, Taglialatela M. M channels containing KCNQ2 subunits modulate norepinephrine, aspartate, and GABA release from hippocampal nerve terminals. J Neurosci. 2004;24:592–597. doi: 10.1523/JNEUROSCI.3143-03.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McNamara NM, Muniz ZM, Wilkin GP, Dolly JO. Prominent location of a K+ channel containing the alpha subunit Kv 1.2 in the basket cell nerve terminals of rat cerebellum. Neuroscience. 1993;57:1039–1045. doi: 10.1016/0306-4522(93)90047-j. [DOI] [PubMed] [Google Scholar]
- Misonou H, Menegola M, Buchwalder L, Park EW, Meredith A, Rhodes KJ, Aldrich RW, Trimmer JS. Immunolocalization of the Ca2+-activated K+ channel Slo1 in axons and nerve terminals of mammalian brain and cultured neurons. J Comp Neurol. 2006;496:289–302. doi: 10.1002/cne.20931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Monaghan MM, Trimmer JS, Rhodes KJ. Experimental localization of Kv1 family voltage-gated K+ channel alpha and beta subunits in rat hippocampal formation. J Neurosci. 2001;21:5973–5983. doi: 10.1523/JNEUROSCI.21-16-05973.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nesti E, Everill B, Morielli AD. Endocytosis as a mechanism for tyrosine kinase-dependent suppression of a voltage-gated potassium channel. Mol Biol Cell. 2004;15:4073–4088. doi: 10.1091/mbc.E03-11-0788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ogawa Y, Horresh I, Trimmer JS, Bredt DS, Peles E, Rasband MN. Postsynaptic density-93 clusters Kv1 channels at axon initial segments independently of Caspr2. J Neurosci. 2008;28:5731–5739. doi: 10.1523/JNEUROSCI.4431-07.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ogawa Y, Oses-Prieto J, Kim MY, Horresh I, Peles E, Burlingame AL, Trimmer JS, Meijer D, Rasband MN. ADAM22, a Kv1 channel-interacting protein, recruits membrane-associated guanylate kinases to juxtaparanodes of myelinated axons. J Neurosci. 2010;30:1038–1048. doi: 10.1523/JNEUROSCI.4661-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ozaita A, Martone ME, Ellisman MH, Rudy B. Differential subcellular localization of the two alternatively spliced isoforms of the Kv3.1 potassium channel subunit in brain. J Neurophysiol. 2002;88:394–408. doi: 10.1152/jn.2002.88.1.394. [DOI] [PubMed] [Google Scholar]
- Pan Z, Kao T, Horvath Z, Lemos J, Sul JY, Cranstoun SD, Bennett V, Scherer SS, Cooper EC. A common ankyrin-G-based mechanism retains KCNQ and NaV channels at electrically active domains of the axon. J Neurosci. 2006;26:2599–2613. doi: 10.1523/JNEUROSCI.4314-05.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pannasch U, Farber K, Nolte C, Blonski M, Yan Chiu S, Messing A, Kettenmann H. The potassium channels Kv1.5 and Kv1.3 modulate distinct functions of microglia. Mol Cell Neurosci. 2006;33:401–411. doi: 10.1016/j.mcn.2006.08.009. [DOI] [PubMed] [Google Scholar]
- Park SM, Liu G, Kubal A, Fury M, Cao L, Marx SO. Direct interaction between BKCa potassium channel and microtubule-associated protein 1A. FEBS Lett. 2004;570:143–148. doi: 10.1016/j.febslet.2004.06.037. [DOI] [PubMed] [Google Scholar]
- Peretz A, Sheinin A, Yue C, Degani-Katzav N, Gibor G, Nachman R, Gopin A, Tam E, Shabat D, Yaari Y, Attali B. Pre- and postsynaptic activation of M-channels by a novel opener dampens neuronal firing and transmitter release. J Neurophysiol. 2007;97:283–295. doi: 10.1152/jn.00634.2006. [DOI] [PubMed] [Google Scholar]
- Perney TM, Marshall J, Martin KA, Hockfield S, Kaczmarek LK. Expression of the mRNAs for the Kv3.1 potassium channel gene in the adult and developing rat brain. J Neurophysiol. 1992;68:756–766. doi: 10.1152/jn.1992.68.3.756. [DOI] [PubMed] [Google Scholar]
- Petersen OH, Maruyama Y. Calcium-activated potassium channels and their role in secretion. Nature. 1984;307:693–696. doi: 10.1038/307693a0. [DOI] [PubMed] [Google Scholar]
- Poliak S, Gollan L, Martinez R, Custer A, Einheber S, Salzer JL, Trimmer JS, Shrager P, Peles E. Caspr2, a new member of the neurexin superfamily, is localized at the juxtaparanodes of myelinated axons and associates with K+ channels. Neuron. 1999;24:1037–1047. doi: 10.1016/s0896-6273(00)81049-1. [DOI] [PubMed] [Google Scholar]
- Poliak S, Peles E. The local differentiation of myelinated axons at nodes of Ranvier. Nat Rev Neurosci. 2003;4:968–980. doi: 10.1038/nrn1253. [DOI] [PubMed] [Google Scholar]
- Poliak S, Salomon D, Elhanany H, Sabanay H, Kiernan B, Pevny L, Stewart CL, Xu X, Chiu SY, Shrager P, Furley AJ, Peles E. Juxtaparanodal clustering of Shaker-like K+ channels in myelinated axons depends on Caspr2 and TAG-1. J Cell Biol. 2003;162:1149–1160. doi: 10.1083/jcb.200305018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ponce A, Vega-Saenz de Miera E, Kentros C, Moreno H, Thornhill B, Rudy B. K+ channel subunit isoforms with divergent carboxy-terminal sequences carry distinct membrane targeting signals. J Membr Biol. 1997;159:149–159. doi: 10.1007/s002329900278. [DOI] [PubMed] [Google Scholar]
- Pongs O, Leicher T, Berger M, Roeper J, Bahring R, Wray D, Giese KP, Silva AJ, Storm JF. Functional and molecular aspects of voltage-gated K+ channel beta subunits. Ann N Y Acad Sci. 1999;868:344–355. doi: 10.1111/j.1749-6632.1999.tb11296.x. [DOI] [PubMed] [Google Scholar]
- Pongs O, Schwarz JR. Ancillary subunits associated with voltage-dependent K+ channels. Physiol Rev. 2010;90:755–796. doi: 10.1152/physrev.00020.2009. [DOI] [PubMed] [Google Scholar]
- Raffaelli G, Saviane C, Mohajerani MH, Pedarzani P, Cherubini E. BK potassium channels control transmitter release at CA3-CA3 synapses in the rat hippocampus. J Physiol. 2004;557:147–157. doi: 10.1113/jphysiol.2004.062661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rasband MN. It’s “juxta” potassium channel! J Neurosci Res. 2004;76:749–757. doi: 10.1002/jnr.20073. [DOI] [PubMed] [Google Scholar]
- Rasband MN, Park EW, Vanderah TW, Lai J, Porreca F, Trimmer JS. Distinct potassium channels on pain-sensing neurons. Proc Natl Acad Sci U S A. 2001;98:13373–13378. doi: 10.1073/pnas.231376298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rasband MN, Park EW, Zhen D, Arbuckle MI, Poliak S, Peles E, Grant SG, Trimmer JS. Clustering of neuronal potassium channels is independent of their interaction with PSD-95. J Cell Biol. 2002;159:663–672. doi: 10.1083/jcb.200206024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rasband MN, Trimmer JS. Subunit composition and novel localization of K+ channels in spinal cord. J Comp Neurol. 2001;429:166–176. doi: 10.1002/1096-9861(20000101)429:1<166::aid-cne13>3.0.co;2-y. [DOI] [PubMed] [Google Scholar]
- Rasband MN, Trimmer JS, Peles E, Levinson SR, Shrager P. K+ channel distribution and clustering in developing and hypomyelinated axons of the optic nerve. J Neurocytol. 1999;28:319–331. doi: 10.1023/a:1007057512576. [DOI] [PubMed] [Google Scholar]
- Rasband MN, Trimmer JS, Schwarz TL, Levinson SR, Ellisman MH, Schachner M, Shrager P. Potassium channel distribution, clustering, and function in remyelinating rat axons. J Neurosci. 1998;18:36–47. doi: 10.1523/JNEUROSCI.18-01-00036.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rasmussen HB, Frokjaer-Jensen C, Jensen CS, Jensen HS, Jorgensen NK, Misonou H, Trimmer JS, Olesen SP, Schmitt N. Requirement of subunit co-assembly and ankyrin-G for M-channel localization at the axon initial segment. J Cell Sci. 2007;120:953–963. doi: 10.1242/jcs.03396. [DOI] [PubMed] [Google Scholar]
- Rettig J, Heinemann SH, Wunder F, Lorra C, Parcej DN, Dolly JO, Pongs O. Inactivation properties of voltage-gated K+ channels altered by presence of beta-subunit. Nature. 1994;369:289–294. doi: 10.1038/369289a0. [DOI] [PubMed] [Google Scholar]
- Rettig J, Wunder F, Stocker M, Lichtinghagen R, Mastiaux F, Beckh S, Kues W, Pedarzani P, Schroter KH, Ruppersberg JP, et al. Characterization of a Shaw-related potassium channel family in rat brain. EMBO J. 1992;11:2473–2486. doi: 10.1002/j.1460-2075.1992.tb05312.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rhodes KJ, Keilbaugh SA, Barrezueta NX, Lopez KL, Trimmer JS. Association and colocalization of K+ channel alpha- and beta-subunit polypeptides in rat brain. J Neurosci. 1995;15:5360–5371. doi: 10.1523/JNEUROSCI.15-07-05360.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rhodes KJ, Monaghan MM, Barrezueta NX, Nawoschik S, Bekele-Arcuri Z, Matos MF, Nakahira K, Schechter LE, Trimmer JS. Voltage-gated K+ channel beta subunits: expression and distribution of Kv beta 1 and Kv beta 2 in adult rat brain. J Neurosci. 1996;16:4846–4860. doi: 10.1523/JNEUROSCI.16-16-04846.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rhodes KJ, Strassle BW, Monaghan MM, BekeleArcuri Z, Matos MF, Trimmer JS. Association and colocalization of the Kv beta 1 and Kv beta 2 beta-subunits with Kv1 alpha-subunits in mammalian brain K+ channel complexes. J Neurosci. 1997;17:8246–8258. doi: 10.1523/JNEUROSCI.17-21-08246.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ridgway LD, Kim EY, Dryer SE. MAGI-1 interacts with Slo1 channel proteins and suppresses Slo1 expression on the cell surface. Am J Physiol Cell Physiol. 2009;297:C55–65. doi: 10.1152/ajpcell.00073.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rivera J, Chu PJ, Lewis TL, Jr, Arnold DB. The role of Kif5B in axonal localization of Kv1 K+ channels. Eur J Neurosci. 2007;25:136–146. doi: 10.1111/j.1460-9568.2006.05277.x. [DOI] [PubMed] [Google Scholar]
- Rivera JF, Chu PJ, Arnold DB. The T1 domain of Kv1.3 mediates intracellular targeting to axons. Eur J Neurosci. 2005;22:1853–1862. doi: 10.1111/j.1460-9568.2005.04384.x. [DOI] [PubMed] [Google Scholar]
- Robitaille R, Garcia ML, Kaczorowski GJ, Charlton MP. Functional colocalization of calcium and calcium-gated potassium channels in control of transmitter release. Neuron. 1993;11:645–655. doi: 10.1016/0896-6273(93)90076-4. [DOI] [PubMed] [Google Scholar]
- Roche JP, Westenbroek R, Sorom AJ, Hille B, Mackie K, Shapiro MS. Antibodies and a cysteine-modifying reagent show correspondence of M current in neurons to KCNQ2 and KCNQ3 K+ channels. Br J Pharmacol. 2002;137:1173–1186. doi: 10.1038/sj.bjp.0704989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rudy B, Chow A, Lau D, Amarillo Y, Ozaita A, Saganich M, Moreno H, Nadal MS, Hernandez-Pineda R, Hernandez-Cruz A, Erisir A, Leonard C, Vega-Saenz de Miera E. Contributions of Kv3 channels to neuronal excitability. Ann N Y Acad Sci. 1999;868:304–343. doi: 10.1111/j.1749-6632.1999.tb11295.x. [DOI] [PubMed] [Google Scholar]
- Rudy B, McBain CJ. Kv3 channels: voltage-gated K+ channels designed for high-frequency repetitive firing. Trends Neurosci. 2001;24:517–526. doi: 10.1016/s0166-2236(00)01892-0. [DOI] [PubMed] [Google Scholar]
- Sailer CA, Kaufmann WA, Kogler M, Chen L, Sausbier U, Ottersen OP, Ruth P, Shipston MJ, Knaus HG. Immunolocalization of BK channels in hippocampal pyramidal neurons. Eur J Neurosci. 2006;24:442–454. doi: 10.1111/j.1460-9568.2006.04936.x. [DOI] [PubMed] [Google Scholar]
- Salkoff L, Butler A, Ferreira G, Santi C, Wei A. High-conductance potassium channels of the SLO family. Nat Rev Neurosci. 2006;7:921–931. doi: 10.1038/nrn1992. [DOI] [PubMed] [Google Scholar]
- Salzer JL. Polarized domains of myelinated axons. Neuron. 2003;40:297–318. doi: 10.1016/s0896-6273(03)00628-7. [DOI] [PubMed] [Google Scholar]
- Sarmiere PD, Weigle CM, Tamkun MM. The Kv2.1 K+ channel targets to the axon initial segment of hippocampal and cortical neurons in culture and in situ. BMC Neurosci. 2008;9:112. doi: 10.1186/1471-2202-9-112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sausbier M, Hu H, Arntz C, Feil S, Kamm S, Adelsberger H, Sausbier U, Sailer CA, Feil R, Hofmann F, Korth M, Shipston MJ, Knaus HG, Wolfer DP, Pedroarena CM, Storm JF, Ruth P. Cerebellar ataxia and Purkinje cell dysfunction caused by Ca2+-activated K+ channel deficiency. Proc Natl Acad Sci U S A. 2004;101:9474–9478. doi: 10.1073/pnas.0401702101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Savvaki M, Theodorakis K, Zoupi L, Stamatakis A, Tivodar S, Kyriacou K, Stylianopoulou F, Karagogeos D. The expression of TAG-1 in glial cells is sufficient for the formation of the juxtaparanodal complex and the phenotypic rescue of tag-1 homozygous mutants in the CNS. J Neurosci. 2010;30:13943–13954. doi: 10.1523/JNEUROSCI.2574-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schaeren-Wiemers N, Bonnet A, Erb M, Erne B, Bartsch U, Kern F, Mantei N, Sherman D, Suter U. The raft-associated protein MAL is required for maintenance of proper axon--glia interactions in the central nervous system. J Cell Biol. 2004;166:731–742. doi: 10.1083/jcb.200406092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schroeder BC, Hechenberger M, Weinreich F, Kubisch C, Jentsch TJ. KCNQ5, a novel potassium channel broadly expressed in brain, mediates M-type currents. J Biol Chem. 2000;275:24089–24095. doi: 10.1074/jbc.M003245200. [DOI] [PubMed] [Google Scholar]
- Schroeder BC, Kubisch C, Stein V, Jentsch TJ. Moderate loss of function of cyclic-AMP-modulated KCNQ2/KCNQ3 K+ channels causes epilepsy. Nature. 1998;396:687–690. doi: 10.1038/25367. [DOI] [PubMed] [Google Scholar]
- Schulte U, Thumfart JO, Klocker N, Sailer CA, Bildl W, Biniossek M, Dehn D, Deller T, Eble S, Abbass K, Wangler T, Knaus HG, Fakler B. The epilepsy-linked Lgi1 protein assembles into presynaptic Kv1 channels and inhibits inactivation by Kvbeta1. Neuron. 2006;49:697–706. doi: 10.1016/j.neuron.2006.01.033. [DOI] [PubMed] [Google Scholar]
- Sekirnjak C, Martone ME, Weiser M, Deerinck T, Bueno E, Rudy B, Ellisman M. Subcellular localization of the K+ channel subunit Kv3.1b in selected rat CNS neurons. Brain Res. 1997;766:173–187. doi: 10.1016/s0006-8993(97)00527-1. [DOI] [PubMed] [Google Scholar]
- Setou M, Nakagawa T, Seog DH, Hirokawa N. Kinesin superfamily motor protein KIF17 and mLin-10 in NMDA receptor-containing vesicle transport. Science. 2000;288:1796–1802. doi: 10.1126/science.288.5472.1796. [DOI] [PubMed] [Google Scholar]
- Setou M, Seog DH, Tanaka Y, Kanai Y, Takei Y, Kawagishi M, Hirokawa N. Glutamate-receptor-interacting protein GRIP1 directly steers kinesin to dendrites. Nature. 2002;417:83–87. doi: 10.1038/nature743. [DOI] [PubMed] [Google Scholar]
- Shah MM, Migliore M, Valencia I, Cooper EC, Brown DA. Functional significance of axonal Kv7 channels in hippocampal pyramidal neurons. Proc Natl Acad Sci U S A. 2008;105:7869–7874. doi: 10.1073/pnas.0802805105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sheng M, Tsaur ML, Jan YN, Jan LY. Subcellular Segregation of 2 A-Type K+ Channel Proteins in Rat Central Neurons. Neuron. 1992;9:271–284. doi: 10.1016/0896-6273(92)90166-b. [DOI] [PubMed] [Google Scholar]
- Shu Y, Yu Y, Yang J, McCormick DA. Selective control of cortical axonal spikes by a slowly inactivating K+ current. Proc Natl Acad Sci U S A. 2007;104:11453–11458. doi: 10.1073/pnas.0702041104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sobotzik JM, Sie JM, Politi C, Del Turco D, Bennett V, Deller T, Schultz C. AnkyrinG is required to maintain axo-dendritic polarity in vivo. Proc Natl Acad Sci U S A. 2009;106:17564–17569. doi: 10.1073/pnas.0909267106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song AH, Wang D, Chen G, Li Y, Luo J, Duan S, Poo MM. A selective filter for cytoplasmic transport at the axon initial segment. Cell. 2009;136:1148–1160. doi: 10.1016/j.cell.2009.01.016. [DOI] [PubMed] [Google Scholar]
- Song P, Yang Y, Barnes-Davies M, Bhattacharjee A, Hamann M, Forsythe ID, Oliver DL, Kaczmarek LK. Acoustic environment determines phosphorylation state of the Kv3.1 potassium channel in auditory neurons. Nat Neurosci. 2005;8:1335–1342. doi: 10.1038/nn1533. [DOI] [PubMed] [Google Scholar]
- Southan AP, Robertson B. Electrophysiological characterization of voltage-gated K+ currents in cerebellar basket and Purkinje cells: Kv1 and Kv3 channel subfamilies are present in basket cell nerve terminals. J Neurosci. 2000;20:114–122. doi: 10.1523/JNEUROSCI.20-01-00114.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strumbos JG, Polley DB, Kaczmarek LK. Specific and rapid effects of acoustic stimulation on the tonotopic distribution of Kv3.1b potassium channels in the adult rat. Neuroscience. 2010;167:567–572. doi: 10.1016/j.neuroscience.2010.02.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun XP, Yazejian B, Grinnell AD. Electrophysiological properties of BK channels in Xenopus motor nerve terminals. J Physiol. 2004;557:207–228. doi: 10.1113/jphysiol.2003.060509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Swensen AM, Bean BP. Ionic mechanisms of burst firing in dissociated Purkinje neurons. J Neurosci. 2003;23:9650–9663. doi: 10.1523/JNEUROSCI.23-29-09650.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Talley EM, Sirois JE, Lei Q, Bayliss DA. Two-pore-Domain (KCNK) potassium channels: dynamic roles in neuronal function. Neuroscientist. 2003;9:46–56. doi: 10.1177/1073858402239590. [DOI] [PubMed] [Google Scholar]
- Tanaka H, Ma J, Tanaka KF, Takao K, Komada M, Tanda K, Suzuki A, Ishibashi T, Baba H, Isa T, Shigemoto R, Ono K, Miyakawa T, Ikenaka K. Mice with altered myelin proteolipid protein gene expression display cognitive deficits accompanied by abnormal neuron-glia interactions and decreased conduction velocities. J Neurosci. 2009;29:8363–8371. doi: 10.1523/JNEUROSCI.3216-08.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Torres YP, Morera FJ, Carvacho I, Latorre R. A marriage of convenience: beta-subunits and voltage-dependent K+ channels. J Biol Chem. 2007;282:24485–24489. doi: 10.1074/jbc.R700022200. [DOI] [PubMed] [Google Scholar]
- Traka M, Dupree JL, Popko B, Karagogeos D. The neuronal adhesion protein TAG-1 is expressed by Schwann cells and oligodendrocytes and is localized to the juxtaparanodal region of myelinated fibers. J Neurosci. 2002;22:3016–3024. doi: 10.1523/JNEUROSCI.22-08-03016.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Traka M, Goutebroze L, Denisenko N, Bessa M, Nifli A, Havaki S, Iwakura Y, Fukamauchi F, Watanabe K, Soliven B, Girault JA, Karagogeos D. Association of TAG-1 with Caspr2 is essential for the molecular organization of juxtaparanodal regions of myelinated fibers. J Cell Biol. 2003;162:1161–1172. doi: 10.1083/jcb.200305078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trimmer JS, Rhodes KJ. Localization of voltage-gated ion channels in mammalian brain. Annu Rev Physiol. 2004;66:477–519. doi: 10.1146/annurev.physiol.66.032102.113328. [DOI] [PubMed] [Google Scholar]
- Tsai W, Morielli AD, Peralta EG. The m1 muscarinic acetylcholine receptor transactivates the EGF receptor to modulate ion channel activity. EMBO J. 1997;16:4597–4605. doi: 10.1093/emboj/16.15.4597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tzingounis AV, Heidenreich M, Kharkovets T, Spitzmaul G, Jensen HS, Nicoll RA, Jentsch TJ. The KCNQ5 potassium channel mediates a component of the afterhyperpolarization current in mouse hippocampus. Proc Natl Acad Sci U S A. 2010;107:10232–10237. doi: 10.1073/pnas.1004644107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vabnick I, Trimmer JS, Schwarz TL, Levinson SR, Risal D, Shrager P. Dynamic potassium channel distributions during axonal development prevent aberrant firing patterns. J Neurosci. 1999;19:747–758. doi: 10.1523/JNEUROSCI.19-02-00747.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vacher H, Mohapatra DP, Trimmer JS. Localization and targeting of voltage-dependent ion channels in mammalian central neurons. Physiol Rev. 2008;88:1407–1447. doi: 10.1152/physrev.00002.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vacher H, Yang JW, Cerda O, Autillo-Touati A, Dargent B, Trimmer JS. Cdk-mediated phosphorylation of the Kv{beta}2 auxiliary subunit regulates Kv1 channel axonal targeting. J Cell Biol. 2011;192:813–824. doi: 10.1083/jcb.201007113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Wart A, Trimmer JS, Matthews G. Polarized distribution of ion channels within microdomains of the axon initial segment. J Comp Neurol. 2007;500:339–352. doi: 10.1002/cne.21173. [DOI] [PubMed] [Google Scholar]
- Veh RW, Lichtinghagen R, Sewing S, Wunder F, Grumbach IM, Pongs O. Immunohistochemical Localization of 5 Members of the Kv1 Channel Subunits - Contrasting Subcellular Locations and Neuron-Specific Co-Localizations in Rat-Brain. Eur J Neurosci. 1995;7:2189–2205. doi: 10.1111/j.1460-9568.1995.tb00641.x. [DOI] [PubMed] [Google Scholar]
- Vervaeke K, Gu N, Agdestein C, Hu H, Storm JF. Kv7/KCNQ/M-channels in rat glutamatergic hippocampal axons and their role in regulation of excitability and transmitter release. J Physiol. 2006;576:235–256. doi: 10.1113/jphysiol.2006.111336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wallner M, Meera P, Toro L. Determinant for beta-subunit regulation in high-conductance voltage-activated and Ca2+-sensitive K+ channels: an additional transmembrane region at the N terminus. Proc Natl Acad Sci U S A. 1996;93:14922–14927. doi: 10.1073/pnas.93.25.14922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang H, Allen ML, Grigg JJ, Noebels JL, Tempel BL. Hypomyelination alters K+ channel expression in mouse mutants shiverer and Trembler. Neuron. 1995;15:1337–1347. doi: 10.1016/0896-6273(95)90012-8. [DOI] [PubMed] [Google Scholar]
- Wang H, Kunkel DD, Martin TM, Schwartzkroin PA, Tempel BL. Heteromultimeric K+ channels in terminal and juxtaparanodal regions of neurons. Nature. 1993;365:75–79. doi: 10.1038/365075a0. [DOI] [PubMed] [Google Scholar]
- Wang H, Kunkel DD, Schwartzkroin PA, Tempel BL. Localization of Kv1.1 and Kv1.2, two K+ channel proteins, to synaptic terminals, somata, and dendrites in the mouse brain. J Neurosci. 1994;14:4588–4599. doi: 10.1523/JNEUROSCI.14-08-04588.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang ZW, Saifee O, Nonet ML, Salkoff L. SLO-1 potassium channels control quantal content of neurotransmitter release at the C. elegans neuromuscular junction. Neuron. 2001;32:867–881. doi: 10.1016/s0896-6273(01)00522-0. [DOI] [PubMed] [Google Scholar]
- Weatherall KL, Goodchild SJ, Jane DE, Marrion NV. Small conductance calcium-activated potassium channels: from structure to function. Prog Neurobiol. 2010;91:242–255. doi: 10.1016/j.pneurobio.2010.03.002. [DOI] [PubMed] [Google Scholar]
- Weiser M, Bueno E, Sekirnjak C, Martone ME, Baker H, Hillman D, Chen S, Thornhill W, Ellisman M, Rudy B. The potassium channel subunit KV3.1b is localized to somatic and axonal membranes of specific populations of CNS neurons. J Neurosci. 1995;15:4298–4314. doi: 10.1523/JNEUROSCI.15-06-04298.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weiser M, Vega-Saenz de Miera E, Kentros C, Moreno H, Franzen L, Hillman D, Baker H, Rudy B. Differential expression of Shaw-related K+ channels in the rat central nervous system. J Neurosci. 1994;14:949–972. doi: 10.1523/JNEUROSCI.14-03-00949.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Womack MD, Khodakhah K. Dendritic control of spontaneous bursting in cerebellar Purkinje cells. J Neurosci. 2004;24:3511–3521. doi: 10.1523/JNEUROSCI.0290-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu MN, Joiner WJ, Dean T, Yue Z, Smith CJ, Chen D, Hoshi T, Sehgal A, Koh K. SLEEPLESS, a Ly-6/neurotoxin family member, regulates the levels, localization and activity of Shaker. Nat Neurosci. 2010;13:69–75. doi: 10.1038/nn.2454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu M, Cao R, Xiao R, Zhu MX, Gu C. The axon-dendrite targeting of Kv3 (Shaw) channels is determined by a targeting motif that associates with the T1 domain and ankyrin G. J Neurosci. 2007;27:14158–14170. doi: 10.1523/JNEUROSCI.3675-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu M, Gu Y, Barry J, Gu C. Kinesin I transports tetramerized Kv3 channels through the axon initial segment via direct binding. J Neurosci. 2010;30:15987–6001. doi: 10.1523/JNEUROSCI.3565-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yan J, Aldrich RW. LRRC26 auxiliary protein allows BK channel activation at resting voltage without calcium. Nature. 2010;466:513–516. doi: 10.1038/nature09162. [DOI] [PubMed] [Google Scholar]
- Zadeh AD, Cheng Y, Xu H, Wong N, Wang Z, Goonasekara C, Steele DF, Fedida D. Kif5b is an essential forward trafficking motor for the Kv1.5 cardiac potassium channel. J Physiol. 2009;587:4565–4574. doi: 10.1113/jphysiol.2009.178442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou L, Messing A, Chiu SY. Determinants of excitability at transition zones in Kv1.1-deficient myelinated nerves. J Neurosci. 1999;19:5768–5781. doi: 10.1523/JNEUROSCI.19-14-05768.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou L, Zhang CL, Messing A, Chiu SY. Temperature-sensitive neuromuscular transmission in Kv1.1 null mice: Role of potassium channels under the myelin sheath in young nerves. J Neurosci. 1998;18:7200–7215. doi: 10.1523/JNEUROSCI.18-18-07200.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]