Abstract
Peroxisome proliferator-activated receptor γ (PPARγ) is a critical factor for adipogenesis and glucose metabolism, but accumulating evidence demonstrates the involvement of PPARγ in skeletal metabolism as well. PPARγ agonists, the thiazolidinediones (TZDs), have been widely used for the treatment of type 2 diabetes mellitus owing to their effectiveness in lowering blood glucose. However, the use of TZDs has been associated with bone loss and fractures. TZD-induced alterations in the bone marrow milieu—that is, increased marrow adiposity with suppression of osteogenesis—could partially explain the pathogenesis of TZD-induced bone loss Furthermore, several lines of evidence place PPARγ at the center of a regulatory loop between circadian networks and metabolic output. PPARγ exhibits a circadian expression pattern that is magnified by consumption of a high-fat diet. One of the circadian-regulated genes expressed in peripheral tissues, nocturnin (Noc), has been shown to enhance PPARγ activity. Importantly Noc-deficient mice are protected from diet-induced obesity, exhibit impaired circadian expression of PPARγ and have increased bone mass. This Review focuses on new findings regarding the role of PPARγ in adipose tissue and skeletal metabolism and summarizes the emerging role of PPARγ as an integral part of a complex circadian regulatory system that modulates food storage, energy consumption and skeletal metabolism.
Introduction
Since the identification of peroxisome proliferator-activated receptor γ (PPARγ) as a master regulator of adipogenesis,1 newer insights have emerged that demonstrate an even more critical role for PPARγ as a central integrator of glucose metabolism and energy homeostasis.2–4 A synthetic class of compounds, the thiazolidinediones (TZDs), bind to PPARγ and have widely been used to treat type 2 diabetes mellitus.5–9 Despite improvement in glucose and lipid metabolism and variable effects on the cardiovascular system, a growing body of evidence demonstrates unexpected adverse effects of these drugs on skeletal metabolism.10–17 Although the skeleton controls mineral homeostasis, novel evidence also suggests it acts as an active modulator of glucose metabolism. Several lines of evidence both in clinical and animal studies imply that altered mesenchymal stem cell fate could be the principal mechanism of TZD-induced bone loss. For example, PPARγ activation by TZDs suppresses several key osteogenic transcription factors in both mice and humans,18,19 and these changes contribute to a decrease in bone mass. In addition, TZDs can stimulate bone resorption, further uncoupling the remodeling unit.
The circadian system of gene expression is now widely accepted as an important modulator of metabolism. Indeed a number of adipogenic and skeletal genes possess circadian expression profiles.20 Nevertheless, how peripheral tissues sense alterations in external cues, such as nutrient availability, is still not known. The link between the external environment and cellular metabolic responses is mediated via nuclear receptor activation; many of these factors exhibit circadian expression profiles and are critical for glucose and lipid metabolism.21 In line with this finding, Pparg (the gene that encodes the murine Pparγ protein) exhibits a remarkable circadian expression pattern in liver and adipose tissue, and this rhythmicity is magnified by consumption of a high-fat diet.21,22 This Review will focus on the emerging concept of PPARγ as a regulator of skeletal metabolism and summarize novel findings concerning the relationship of skeletal metabolism to central and peripheral circadian networks.
PPARγ
PPARγ is a member of the PPAR family of transcriptional factors and nuclear receptors and plays a pivotal part in cell fate determination, lipid biosynthesis, inflammation and insulin sensitivity.2–4 Differential promoters and alternative splicing produce PPARγ variants, including two major forms of the protein, PPARγ1 and PPARγ2. PPARγ1 is expressed in a wide range of tissues, including the liver, skeletal muscle, adipose tissue and bone. PPARγ2, which contains 30 additional amino acids in its N-terminus compared with PPARγ1, is expressed mainly in adipogenic cells. However, some evidence suggests expression of PPARγ2 in marrow stromal cells, which includes those of osteogenic origin.1,2,23,24 Unsurprisingly, given its broad expression profile, PPARγ has a profound effect on a wide range of metabolic circuits. In addition to its effect on lipid and glucose metabolism, PPARγ is involved in inflammation and may play a part in neoplastic growth.2 PPARγ is activated by naturally occurring ligands, such as fatty acids, eicosanoids,43 as well as compounds and ligands derived from arachidonic acid.3,44
Adipogenesis consists of integrated cascades that involve several transcription factors. The initial step of adipogenesis is the lineage commitment of mesenchymal stem cells (MSCs) followed by the expansion of preadipocytes. PPARγ is a critical component in adipogenesis, as indicated by the fact that loss of Pparg expression in murine embryonic fibroblasts leads to a complete absence of adipogenic capacity.25 Haploinsufficiency of Pparg, on the other hand, increases bone mass.26 Upon ligand binding, PPARγ forms a dimer with the retinoic acid receptor α and initiates transcription by binding to the promoter of its target genes Multiple regulatory controls act to modulate activity of this nuclear receptor and transcriptional factor.
Regulation of PPARγ activity
Transcriptional regulation
A number of transcription factors are involved in the regulation of PPARG expression and function. Transcription factors of the CCAAT/enhancer-binding protein (C/EBP) family, C/EBPs α, β and δ, stimulate PPARG transcription by directly binding to the promoter region.27 Given that adipocytes share common precursor progenitor cells with osteoblasts, entrance into the adipocytic or osteoblastic lineages is thought to be mutually exclusive. The homeobox protein MSX2, which stimulates osteoblastogenesis, represses adipogenesis via inhibition of C/EBPα-driven PPARG transcriptional activity.33,34 Similarly, activation of PPARγ by rosiglitazone leads to enhanced marrow adiposity and suppressed osteoblastogenesis. However, under certain circumstances, mesenchymal cells might conceivably enter both lineages simultaneously, resulting in high bone mass and marrow adipogenesis.
PPARG expression and function is also positively regulated by other transcription factors during adipogenesis, including sterol regulatory element-binding protein 1c (SREBP1c), Krueppel-like factor (KLF) 5, KLF15, Zinc finger protein 423 (ZFP423) and transcription factor COE1 (also known as early B cell factor; EBF1). KLF2 and GATA-binding protein 2 and 3, on the other hand, regulate PPARG expression negatively.4,28–32
Histone modification
Another level of regulation of PPARγ’s transcriptional activity is its ability to modify histones. In the absence of ligand binding, PPARγ forms a protein complex with co-repressors such as nuclear receptor corepressor 1 (NCOR1) and NCOR2 (also known as silencing mediator of retinoic acid and thyroid hormone receptor; SMRT), as well as histone deacetylases (HDACs), as a result of which PPARγ is transcriptionally silent.35 Upon ligand binding, HDACs are dissociated from this protein complex and coactivators such as CREB-binding protein (CBP) and histone acetyltransferases (HATs) are recruited.
Histone methylation also plays a critical part in the regulation of PPARγ activation as evidenced by the fact that noncanonical Wnt pathways, activated by Wnt-5a, suppress PPARγ transcriptional activation via histone-lysine N-methyltransferase SETDB1.36
Post-translational modification
Post-translational modifications of PPARγ may also be important to regulate its activity. Phosphorylation of PPARγ2 at Ser112 by secretory factors such as epidermal growth factor (EGF) and platelet-derived growth factor (PDGF), results in impaired PPARγ2 transcriptional activity.37–39 In addition to phosphorylation, sumoylation and ubiquitination confer a different level of post-transcriptional PPARγ regulation.40–42.
Thiazolidinediones Effects on adipose tissue
TZDs, a synthetic class of compounds, also activate PPARγ and have been shown to improve insulin resistance in rodents and humans.5–9 The mechanisms by which TZDs improve insulin resistance involve multiple pathways. TZDs increase PPARG expression and stimulate PPARγ transcriptional activity in adipose tissue, resulting in the upregulation of genes involved in lipid metabolism, such as CD36 (which encodes platelet glycoprotein 4, also known as fatty acid translocase), FABP4 (which encodes fatty acid binding protein 4) and LPL (which encodes lipoprotein lipase).3 These changes in gene expression enhance the incorporation of free fatty acids (FFAs) into adipose tissue, which eventually decreases serum FFAs levels and improves lipid deposition in liver and skeletal muscle. Lipid redistribution from visceral to subcutaneous depots by TZDs also has an important role for the improvement of insulin sensitivity, because visceral adipose tissue is more metabolically active than subcutaneous adipose tissue.45,46 In addition, PPARγ activation by TZDs leads to an increase in the number of small adipocytes as a result of apoptosis of hypertrophic adipocytes.47 As small adipocytes are associated with improved insulin sensitivity—in part through increased expression of insulin-sensitizing secretory factors such as adiponectin and decreased expression of genes that cause insulin resistance such as tumor necrosis factor (TNF)—one underlying mechanism of TZD action could be altering adipose tissue composition.
Effects on liver and skeletal muscle
PPARG is also expressed in the liver and in skeletal muscle, albeit at lower levels than in adipose tissue. TZD treatment might stimulate lipid accumulation in the liver, as suggested by findings that activation of hepatic Pparγ enhances the accumulation of lipids and that deletion of Pparg expression in liver reduced lipid accumulation in a diabetic mouse model.48 But the lipid and glucose-lowering effect of TZDs through modulation of adipose tissue metabolism potentially predominates over the local TZD effects in liver. In other words, TZDs increase the production of adiponectin in adipocytes, and as a result of reduced adipocyte size, lipid oxidation in the liver may be enhanced. The role of PPARγ in skeletal muscle, however, is still controversial.49,50 Nevertheless, the net effect of TZDs is to improve lipid and glucose metabolic status.
Marrow adiposity and the skeleton
Effects of metabolic disruption on bone
The skeleton is a dynamic organ in which bone formation and resorption are tightly coupled. Bone formation is a result of activated osteoblasts, whereas bone resorption occurs following osteoclast differentiation. Bone is the largest reservoir of minerals such as calcium and phosphate in the body and plays a pivotal part in mineral homeostasis.51 Disruption of bone remodeling is often observed in physiological and pathological conditions. For example, in states of calorie deprivation, the skeleton maintains mineral homeostasis by favoring bone resorption over bone formation.51 In addition to this canonical function, bone also possesses an important role in glucose homeostasis. Growing evidence suggests that the skeleton is a target of the adipose-tissue-derived, secretory factor leptin, via a hypothalamic–sympathetic nervous system relay. This endocrine regulation may occur through the secretion of undercarboxylated osteocalcin,52,53 which regulates glucose metabolism by acting on adipose tissue and pancreatic β cells. Insulin signaling in osteoblasts has been implicated in osteocalcin-mediated regulation of glucose metabolism.54,55 These lines of evidence highlight the clinical observations that bone is adversely affected by metabolic disruptions. For example, in type 1 diabetes mellitus, fracture rates are high, and fractures heal poorly. Similarly, patients with obesity and type 2 diabetes mellitus are at a higher risk of fractures than the general population despite their obesity. Thus, impaired glucose utilization can profoundly affect the skeleton.
Effects of TZDs on bone loss
Use of TZDs increases fracture risk in postmenopausal women,10–14,16,17,56 because these drugs induce an imbalance between bone formation and resorption, as shown in one short-term randomized, placebo-controlled trial.11 In this study, rosiglitazone reduced hip bone mass, and this phenomenon was accompanied by a decrease in serum osteocalcin levels.11 Habib et al.57 reported that TZD treatment in type 2 diabetes mellitus increased the risk of fracture in women >65 years of age. Because PPARG is expressed in MSCs and is an important regulator of MSC specification toward adipogenesis versus osteogenesis, activation of PPARγ by TZDs could alter the fate of MSCs through the suppression of osteogenic transcription factors such as homeobox protein Dlx5, Runt-related transcription factor 2 (Runx2) and Osterix.18,58 In support of this tenet, haploinsufficiency of Pparg is associated with increased bone mass and reduced marrow adiposity, as well as increased osteoblast number and bone formation rates in mice.26 In addition, Pparγ activation has been shown to enhance bone resorption in vivo, in part through recruitment of other coactivators of Pparγ, Pgc1β and estrogen-related receptor 1 (Err1).59–62 The effect of TZDs on osteoclastogenesis could also be mediated by the increased expression of Rankl (which encodes receptor activator of nuclear factor κB ligand) in an age-dependent manner;63 however, the exact role of PPARγ in osteoclastogenesis needs further clarification.64,65
Effects of TZDs on marrow adiposity
Aside from bone loss, activation of Pparγ is associated with increased marrow adiposity in rodent models, although the effect of PPARγ agonists on increased marrow adiposity is strain-specific and drug-specific.66 Marrow adipogenesis has gained increasing attention among researchers, because of its possible role as a contributor to physiologic and pathogenic conditions. Marrow adipocytes have long been considered inert and were thought to result from a default pathway of MSC differentiation. In addition, marrow adipocytes are found in abundance in states such as aplastic anemia, which suggests that adipogenesis inhibits hematopoiesis.67
The process of marrow adipogenesis is probably governed by the same transcriptional cascades observed in white adipocyte differentiation, and, hence, PPARγ is certain to play a pivotal part. Streptozotocin-induced type 1 diabetic mice exhibit increased marrow adiposity; which, however, can be inhibited by treatment with the PPARγ inhibitor bisphenol A diglycidyl ether (BADGE).68 Correspondingly, BADGE also suppresses the marrow adipogenesis following irradiation in mice.67 Interestingly, gene expression profiling analysis revealed the existence of genes involved in thermogenesis and lipid metabolism in marrow adipocytes, suggesting that marrow adipocytes could be metabolically active (unpublished work, C. J. Rosen). With aging, marrow adiposity increases in association with bone loss and enhanced Pparg expression.
To date, no studies have analyzed the effect of TZDs on marrow adiposity in humans, but studies in mouse models have provided insights regarding their role in the development of marrow adiposity.66 Rosiglitazone-induced bone loss in C57BL/6J mice is associated with increased infiltration of the marrow with large adipocytes and is more pronounced in older, female mice.66 Histomorphometric analysis reveals suppressed bone formation and increased bone resorption, consistent with a profound imbalance in bone remodeling. Given the widely accepted tenet that adipose tissue produces a number of secretory factors that affect a wide range of metabolism, it comes as no surprise that marrow adipocytes, especially under pathogenic conditions such as aging and treatment with TZDs, also produce secretory factors which function in the bone marrow millieu. These factors may then act in a paracrine manner to impair osteoblast differentiation and/or function. Further investigations are, however, needed to support this hypothesis, as some evidence suggests that mice which lack marrow adipocytes have normal bone mass.69
Physiological versus pathogenic conditions
Marrow adipocyte infiltration is often associated with bone loss, but evidence also exists of a positive correlation between marrow adiposity and bone mass in humans and animal models. For example, the C3H/HeJ mouse strain exhibits higher bone mass and increased marrow adiposity compared with the C57BL/6J mouse strain.70 In humans, marrow adiposity robustly increases during puberty when skeletal acquisition is maximized.71 These lines of evidence suggest that in physiological situations marrow adipocytes have a distinctly different role from pathogenic conditions. Marrow adipocytes may be an important component of the bone marrow niche and establish a favorable skeletal microenvironment for osteoblast differentiation by functioning as a source of secretory factors and a depot for energy utilization. The role of adipocytes in hematopoiesis needs further delineation.72 However, the adipogenesis that occurs in bone marrow after transplantation or injury may serve not as a true antagonist of hematopoiesis but rather as a ‘place-holder’, to maintain hematopoietic progenitors in a stem cell state awaiting signals that trigger their differentiation.
Circadian regulation of PPARG
Several lines of evidence demonstrate the tight connection between circadian networks and metabolic outputs, thereby placing PPARγ at the center of another regulatory system.73–77 For example, night shift workers who have disrupted circadian cycles showed a higher incidence of metabolic syndrome and cardiovascular complications.78 Mutations in circadian genes are associated with sleep disorders, which can also affect metabolic status and body composition.79 In addition, drugs such as glucocorticoids and second-generation antipsychotics have been shown to result in the disruption of endogenous circadian rhythms and the development of metabolic phenotypes including insulin resistance both in humans and mice.75–77,80
Components of the circadian clock
Central components
The circadian clock network is composed of central and peripheral components. The master pacemaker is located in the suprachiasmatic nucleus in the hypothalamus81 and governs peripheral rhythm by processing retinal, hormonal, nutritional and neuronal signals.20 The 24-h rhythm is integrated by a series of transcriptional, translational and post-translational mechanisms which involve a number of genes, including CLOCK (which encodes circadian locomoter output cycles protein kaput), BMAL1 (which encodes brain and muscle ARNT-like 1; also known as ARNTL), PER (which encodes period circadian protein) and CRY (which encodes cryptochrome).82 CLOCK heterodimerizes with BMAL1 and activates transcription of PER and CRY through enhancer elements called E-box. The complex consisting of PER and CRY in turn suppresses CLOCK and BMAL1 transcriptional activitythus forming a 24-h feedback loop. Besides this core feedback loop, growing evidence suggest the existence of other factors involved in 24-h rhythms. For example, the nuclear receptors REV-ERBα and RORα, whose transcription is driven by the CLOCK and BMAL1 complex, are shown to suppress and enhance BMAL1 transcription.83 As most of the physiological activities, such as feeding and energy expenditure, are regulated in a circadian manner, the disruptions of this feedback loop affect the rhythmicity of these activities, which results in alterations in metabolic status. Indeed, mutant mice which lack the Clock gene exhibits an impaired diurnal feeding rhythm and hyperphagia associated with metabolic derangements.80
Peripheral components
The central clock network has a well-established, critical role in the regulation of metabolism; however, a growing body of evidence suggests that peripheral tissues also possess cell-autonomous regulatory systems that are independent of the central clock, but use the same machinery of genes in the same temporal pattern. Given that entrainment to external stimuli, such as food intake, needs to be accomplished in a way that peripheral tissues change their metabolic status in a timely manner, it comes as no surprise that these tissues can function independently (but in coordination with the central clock) through alterations in gene expression. Kornmann et al.84 analyzed gene expression in transgenic mice with a conditionally active liver clock, in which Rev-erbα repressed Bmal1 transcription under the control of tetracycline. The researchers identified 351 circadian-regulated genes in liver, including genes involved in energy and glucose metabolism. The majority of these genes lost their rhythmicity when the liver oscillator was suppressed. On the other hand, 31 genes were still rhythmic in the absence of liver oscillation. These data imply that the liver oscillator is critical for the regulation of rhythmic gene expression.
Linking metabolism and the circadian clock
Accumulating evidence clearly demonstrates the link between circadian clock genes and metabolic output in peripheral tissues. The mechanisms by which metabolic status is controlled by the circadian clock is, however, still largely unknown. Nuclear receptors could have an important role in this regulation, because metabolic physiology, such as lipid and glucose disposal, as well as adipogenesis, requires activation of these factors.85 Importantly, many nuclear receptors exhibit rhythmic circadian patterns in peripheral tissues.21 For example, PPARγ, shows circadian expression in liver and adipose tissue. Interestingly, the hepatic oscillation is enhanced by a high-fat diet.22 In addition, the TZD pioglitazone has been shown to shift the circadian rhythm of blood pressure in patients with type 2 diabetes mellitus.86 Thus, PPARγ, activated by TZDs, might conceivably affect the circadian rhythm, which may result in the alteration of metabolic status.
PGC1α
The inducible PPARγ coactivator 1α (PGC1α) also exhibits a circadian expression pattern. The Pgc1α-deficient mouse strain shows disrupted locomotor activity, diurnal oscillation of body temperature and energy metabolism. Clock, Bmal1 and Per1 gene expression, as well as metabolic factors, such as phosphoenolpyruvate carboxykinase (Pck) and pyruvate dehydrogenase kinase 4 (Pdk4), are also altered by the lack of Pgc1α in the liver.87 Restricted feeding reversed the circadian expression pattern of Pgc1α, Bmal1, Cry1 and Cry2 in livers from wild-type mice, but this adaptation of gene expression was disrupted in the liver from the Pgc1α-deficient mouse.87 Thus, PGC1α may be one factor linking external stimuli and cellular outputs by modulating clock genes and the transcriptional activity of nuclear receptors.
Nocturnin
Green et al.88 previously identified a circadian-regulated gene, nocturnin (Noc), in the xenopus retina. Noc expression peaks at around the time that light is turned off in most tissues, including liver, kidney, skeleton and spleen.89 Noc belongs to a family of proteins which includes transcription factors, deadenylases and phosphatases.90–92 It shares sequence similarities with the inducible yeast gene Ccr4p, which functions as a transcription factor and deadenylase. Noc, however, lacks the transactivation domain that Ccr4p possesses.88,92–93 Noc is also an inducible gene and is upregulated in response to fetal calf serum and insulin in NIH3T3 and 3T3-L1 cells.90,94 Hepatic Noc expression is also enhanced with aging.95 Importantly, Pparγ activation in bone marrow stromal cells induced Noc gene expression nearly 30-fold.18,96 Offspring from mice fed a western-like fat diet develop obesity over four generations, and Noc is one of the most upregulated genes in the stromal vascular fractions of adipose tissue from these mice.97 Thus, Noc appears to be important in lipid homeostasis.
To investigate the metabolic role of Noc, detailed analyses of mice with a knockout of the Noc gene were performed.22 Noc-deficient mice showed normal phase-shifts to light pulses and normal food intake, but were protected from high-fat diet induced fatty liver and obesity.22 Interestingly, the circadian expression pattern of Pparg on high-fat diet in the liver disappeared in Noc-deficient mice and was accompanied by a decrease in expression of those genes involved in lipid metabolism, including Srebp1a, Scd1 (which encodes stearoyl-CoA desaturase 1) and Fabpl (which encodes liver fatty acid-binding protein). These data imply that Noc regulates lipid metabolism by modulating Pparγ activity. To support this notion, Noc has been shown to stimulate adipogenesis of 3T3-L1 cells with enhanced expression of C/EBPα, Pparg2, aP2 and Lpl. In addition, Noc was found to physically bind to Pparγ and enhance its transcriptional activity.94 Surprisingly, Noc protein expression is exclusively limited to the cytoplasm and nuclear membrane, ruling out the possibility of Noc as a coactivator of Pparγ; however, detailed analyses using cell fractionation and immunofluorescence revealed that Noc stimulates the nuclear translocation of Pparγ. Interestingly, this effect is independent of its deadenylase activity.94 Consistent with these findings, Noc-deficient mice exhibit low marrow adiposity and high bone mass, supporting the function of Noc as a positive modulator of Pparγ activity.94 Because Noc expression is inducible by fetal calf serum and insulin, Noc may be a key factor linking external stimuli and metabolic output by activating Pparγ.
Conclusions
Advances in PPARγ biology have provided new insights into an emerging field of metabolism and its relationship to skeletal homeostasis via circadian-regulated factors. As is increasingly recognized, PPARγ activation entrains multiple pathways, some of which may be beneficial, whereas others may be harmful. Nevertheless, TZDs are widely prescribed for the treatment of type 2 diabetes mellitus. New insights into the multifunctional nature of PPARγ may allow for the development of novel agents which specifically target adipocyte progenitors with minimal or no adverse effects to the skeleton. Concomitantly, PPARγ antagonists specific to the osteoblastic lineage could be an attractive candidate for the treatment of osteoporosis. Further translational studies are needed to bridge results from bench studies into the clinical arena. But careful study of the highly integrated connection between circadian rhythms and metabolic output should provide a head start on drug development for both obesity and osteoporosis.
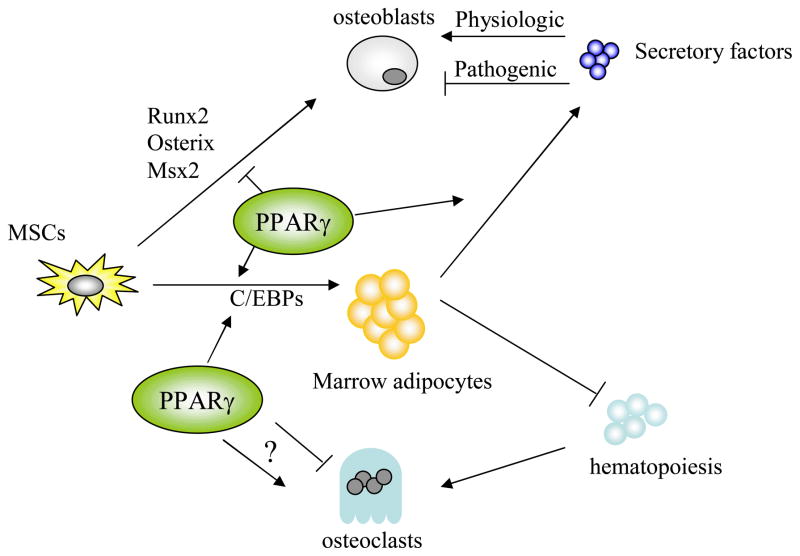
Figure 1. Schematic model of the role of PPARγ in bone marrow.
PPARγ regulates the specification of mesenchymal stem cells (MSCs) toward the adipogenic lineage and activation of PPARγ by specific ligands leads to increased marrow adiposity. The role of PPARγ in osteoclast differentiation is still controversial and needs to be clarified. Marrow adipocytes produce a number of secretory factors and PPARγ regulates the expression of these genes. Such factors could have a significant role in osteoblast differentiation and /or function. In pathogenic conditions, these determinants could impact osteoblasts in a negative direction, whereas in physiological conditions these factors may have a different role from the one observed in the pathogenic conditions. There is also an evidence of the role for marrow adipocytes as an inhibitory factor for hematopoiesis. PPARγ: Peroxisome proliferator-activated receptor-gamma. C/EBP: CCAAT enhancer binding protein. Runx2: Runt-related transcription factor 2. Msx2: Muscle segment homeobox homolog of 2.
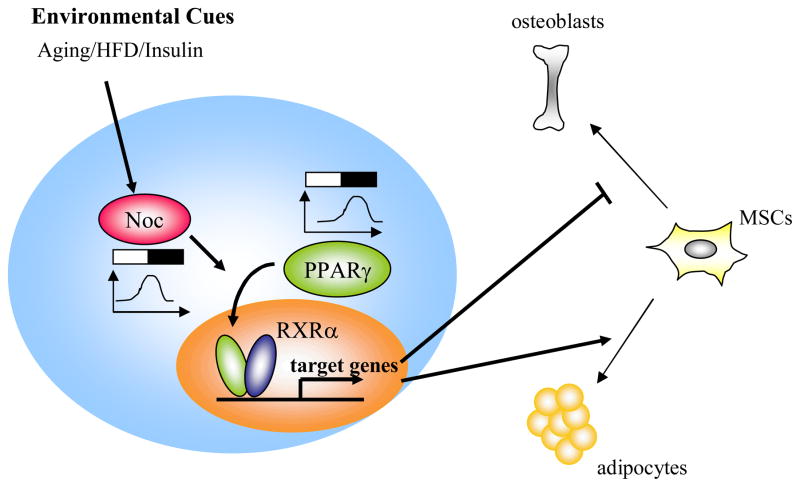
Figure 2. Nocturnin regulates PPARγ circadian profile and activity.
Circadian-regulated gene, Nocturnin, which is induced by external cues such as aging, high-fat diet (HFD) and insulin regulates the circadian expression pattern of PPARγ. In addition, nocturnin enhances the PPARγ transcriptional activity in part by stimulating the nuclear translocation of PPARγ. Enhanced activity of PPARγ by nocturnin may result in increased marrow adiposity and bone loss. White bar and black bar represents light and dark cycle, respectively. Noc: nocturnin, PPARγ: Peroxisome proliferator-activated receptor-gamma. RXRα : Retinoid X receptor alpha. MSCs: nesenchymal stem cells.
Acknowledgments
C. J. Rosen is supported by NIH grants R24DK084970 and AR45433.
Footnotes
Activation of PPARγ is a therapeutic target in type 2 diabetes mellitus
Activation of PPARγ is a risk factor for osteoporosis
PPARγ is a key factor in the determination of mesenchymal stem cell fate
A role for marrow adipocytes in skeletal metabolism is emerging
PPARγ provides a link between the circadian clock system and metabolic output
Competing interests
The authors declare no competing interests.
Author contributions
To be added.
References
- 1.Tontonoz P, Hu E, Graves RA, Budavari AI, Spiegelman BM. mPPAR gamma 2: tissue-specific regulator of an adipocyte enhancer. Genes Dev. 1994;8:1224–1234. doi: 10.1101/gad.8.10.1224. [DOI] [PubMed] [Google Scholar]
- 2.Tontonoz P, Spiegelman BM. Fat and beyond: the diverse biology of PPARgamma. Annu Rev Biochem. 2008;77:289–312. doi: 10.1146/annurev.biochem.77.061307.091829. [DOI] [PubMed] [Google Scholar]
- 3.Kawai M, Sousa KM, MacDougald OA, Rosen CJ. The many facets of PPARgamma: novel insights for the skeleton. Am J Physiol Endocrinol Metab. 2010;299:E3–9. doi: 10.1152/ajpendo.00157.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Rosen ED, MacDougald OA. Adipocyte differentiation from the inside out. Nat Rev Mol Cell Biol. 2006;7:885–896. doi: 10.1038/nrm2066. [DOI] [PubMed] [Google Scholar]
- 5.Iwamoto Y, et al. Effect of new oral antidiabetic agent CS-045 on glucose tolerance and insulin secretion in patients with NIDDM. Diabetes Care. 1991;14:1083–1086. doi: 10.2337/diacare.14.11.1083. [DOI] [PubMed] [Google Scholar]
- 6.Fujiwara T, Yoshioka S, Yoshioka T, Ushiyama I, Horikoshi H. Characterization of new oral antidiabetic agent CS-045. Studies in KK and ob/ob mice and Zucker fatty rats. Diabetes. 1988;37:1549–1558. doi: 10.2337/diab.37.11.1549. [DOI] [PubMed] [Google Scholar]
- 7.Ciaraldi TP, Gilmore A, Olefsky JM, Goldberg M, Heidenreich KA. In vitro studies on the action of CS-045, a new antidiabetic agent. Metabolism. 1990;39:1056–1062. doi: 10.1016/0026-0495(90)90166-a. [DOI] [PubMed] [Google Scholar]
- 8.Suter SL, Nolan JJ, Wallace P, Gumbiner B, Olefsky JM. Metabolic effects of new oral hypoglycemic agent CS-045 in NIDDM subjects. Diabetes Care. 1992;15:193–203. doi: 10.2337/diacare.15.2.193. [DOI] [PubMed] [Google Scholar]
- 9.Nolan JJ, Ludvik B, Beerdsen P, Joyce M, Olefsky J. Improvement in glucose tolerance and insulin resistance in obese subjects treated with troglitazone. N Engl J Med. 1994;331:1188–1193. doi: 10.1056/NEJM199411033311803. [DOI] [PubMed] [Google Scholar]
- 10.Schwartz AV, et al. Thiazolidinedione use and bone loss in older diabetic adults. J Clin Endocrinol Metab. 2006;91:3349–3354. doi: 10.1210/jc.2005-2226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Grey A, et al. The peroxisome proliferator-activated receptor-gamma agonist rosiglitazone decreases bone formation and bone mineral density in healthy postmenopausal women: a randomized, controlled trial. J Clin Endocrinol Metab. 2007;92:1305–1310. doi: 10.1210/jc.2006-2646. [DOI] [PubMed] [Google Scholar]
- 12.Kahn SE, et al. Glycemic durability of rosiglitazone, metformin, or glyburide monotherapy. N Engl J Med. 2006;355:2427–2443. doi: 10.1056/NEJMoa066224. [DOI] [PubMed] [Google Scholar]
- 13.Home PD, et al. Rosiglitazone evaluated for cardiovascular outcomes in oral agent combination therapy for type 2 diabetes (RECORD): a multicentre, randomised, open-label trial. Lancet. 2009;373:2125–2135. doi: 10.1016/S0140-6736(09)60953-3. [DOI] [PubMed] [Google Scholar]
- 14.Yaturu S, Bryant B, Jain SK. Thiazolidinedione treatment decreases bone mineral density in type 2 diabetic men. Diabetes Care. 2007;30:1574–1576. doi: 10.2337/dc06-2606. [DOI] [PubMed] [Google Scholar]
- 15.Rosen CJ. The rosiglitazone story--lessons from an FDA Advisory Committee meeting. N Engl J Med. 2007;357:844–846. doi: 10.1056/NEJMp078167. [DOI] [PubMed] [Google Scholar]
- 16.Aubert RE, Herrera V, Chen W, Haffner SM, Pendergrass M. Rosiglitazone and pioglitazone increase fracture risk in women and men with type 2 diabetes. Diabetes Obes Metab. 2010;12:716–721. doi: 10.1111/j.1463-1326.2010.01225.x. [DOI] [PubMed] [Google Scholar]
- 17.Dormuth CR, Carney G, Carleton B, Bassett K, Wright JM. Thiazolidinediones and fractures in men and women. Arch Intern Med. 2009;169:1395–1402. doi: 10.1001/archinternmed.2009.214. [DOI] [PubMed] [Google Scholar]
- 18.Shockley KR, et al. PPARgamma2 nuclear receptor controls multiple regulatory pathways of osteoblast differentiation from marrow mesenchymal stem cells. J Cell Biochem. 2009;106:232–246. doi: 10.1002/jcb.21994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lecka-Czernik B, et al. Activation of peroxisome proliferator-activated receptor gamma (PPARgamma) by rosiglitazone suppresses components of the insulin-like growth factor regulatory system in vitro and in vivo. Endocrinology. 2007;148:903–911. doi: 10.1210/en.2006-1121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Green CB, Takahashi JS, Bass J. The meter of metabolism. Cell. 2008;134:728–742. doi: 10.1016/j.cell.2008.08.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Yang X, et al. Nuclear receptor expression links the circadian clock to metabolism. Cell. 2006;126:801–810. doi: 10.1016/j.cell.2006.06.050. [DOI] [PubMed] [Google Scholar]
- 22.Green CB, et al. Loss of Nocturnin, a circadian deadenylase, confers resistance to hepatic steatosis and diet-induced obesity. Proc Natl Acad Sci USA. 2007;104:9888–9893. doi: 10.1073/pnas.0702448104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Rosen ED, Spiegelman BM. PPARgamma: a nuclear regulator of metabolism, differentiation, and cell growth. J Biol Chem. 2001;276:37731–37734. doi: 10.1074/jbc.R100034200. [DOI] [PubMed] [Google Scholar]
- 24.Braissant O, Foufelle F, Scotto C, Dauça M, Wahli W. Differential expression of peroxisome proliferator-activated receptors (PPARs): tissue distribution of PPAR-alpha, -beta, and -gamma in the adult rat. Endocrinology. 1996;137:354–366. doi: 10.1210/endo.137.1.8536636. [DOI] [PubMed] [Google Scholar]
- 25.Kubota N, et al. PPAR gamma mediates high-fat diet-induced adipocyte hypertrophy and insulin resistance. Mol Cell. 1999;4:597–609. doi: 10.1016/s1097-2765(00)80210-5. [DOI] [PubMed] [Google Scholar]
- 26.Akune T, et al. PPARgamma insufficiency enhances osteogenesis through osteoblast formation from bone marrow progenitors. J Clin Invest. 2004;113:846–855. doi: 10.1172/JCI19900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Darlington GJ, Ross SE, MacDougald OA. The role of C/EBP genes in adipocyte differentiation. J Biol Chem. 1998;273:30057–30060. doi: 10.1074/jbc.273.46.30057. [DOI] [PubMed] [Google Scholar]
- 28.Mori T, et al. Role of Krüppel-like factor 15 (KLF15) in transcriptional regulation of adipogenesis. J Biol Chem. 2005;280:12867–12875. doi: 10.1074/jbc.M410515200. [DOI] [PubMed] [Google Scholar]
- 29.Oishi Y, et al. Krüppel-like transcription factor KLF5 is a key regulator of adipocyte differentiation. Cell Metab. 2005;1:27–39. doi: 10.1016/j.cmet.2004.11.005. [DOI] [PubMed] [Google Scholar]
- 30.Gupta RK, et al. Transcriptional control of preadipocyte determination by Zfp423. Nature. 2010;464:619–623. doi: 10.1038/nature08816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Banerjee SS, et al. The Krüppel-like factor KLF2 inhibits peroxisome proliferator-activated receptor-gamma expression and adipogenesis. J Biol Chem. 2003;278:2581–2584. doi: 10.1074/jbc.M210859200. [DOI] [PubMed] [Google Scholar]
- 32.Tong Q, Tsai J, Tan G, Dalgin G, Hotamisligil GS. Interaction between GATA and the C/EBP family of transcription factors is critical in GATA-mediated suppression of adipocyte differentiation. Mol Cell Biol. 2005;25:706–715. doi: 10.1128/MCB.25.2.706-715.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Cheng SL, Shao JS, Charlton-Kachigian N, Loewy AP, Towler DA. MSX2 promotes osteogenesis and suppresses adipogenic differentiation of multipotent mesenchymal progenitors. J Biol Chem. 2003;278:45969–45977. doi: 10.1074/jbc.M306972200. [DOI] [PubMed] [Google Scholar]
- 34.Ichida F, et al. Reciprocal roles of MSX2 in regulation of osteoblast and adipocyte differentiation. J Biol Chem. 2004;279:34015–34022. doi: 10.1074/jbc.M403621200. [DOI] [PubMed] [Google Scholar]
- 35.Guan HP, Ishizuka T, Chui PC, Lehrke M, Lazar MA. Corepressors selectively control the transcriptional activity of PPARgamma in adipocytes. Genes Dev. 2005;19:453–461. doi: 10.1101/gad.1263305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Takada I, et al. A histone lysine methyltransferase activated by non-canonical Wnt signalling suppresses PPAR-gamma transactivation. Nat Cell Biol. 2007;9:1273–1285. doi: 10.1038/ncb1647. [DOI] [PubMed] [Google Scholar]
- 37.Hu E, Kim JB, Sarraf P, Spiegelman BM. Inhibition of adipogenesis through MAP kinase-mediated phosphorylation of PPARgamma. Science. 1996;274:2100–2103. doi: 10.1126/science.274.5295.2100. [DOI] [PubMed] [Google Scholar]
- 38.Camp HS, Tafuri SR. Regulation of peroxisome proliferator-activated receptor gamma activity by mitogen-activated protein kinase. J Biol Chem. 1997;272:10811–10816. doi: 10.1074/jbc.272.16.10811. [DOI] [PubMed] [Google Scholar]
- 39.Hosooka T, et al. Dok1 mediates high-fat diet-induced adipocyte hypertrophy and obesity through modulation of PPAR-gamma phosphorylation. Nat Med. 2008;14:188–193. doi: 10.1038/nm1706. [DOI] [PubMed] [Google Scholar]
- 40.Yamashita D, et al. The transactivating function of peroxisome proliferator-activated receptor gamma is negatively regulated by SUMO conjugation in the amino-terminal domain. Genes Cells. 2004;9:1017–1029. doi: 10.1111/j.1365-2443.2004.00786.x. [DOI] [PubMed] [Google Scholar]
- 41.Hauser S, et al. Degradation of the peroxisome proliferator-activated receptor gamma is linked to ligand-dependent activation. J Biol Chem. 2000;275:18527–18533. doi: 10.1074/jbc.M001297200. [DOI] [PubMed] [Google Scholar]
- 42.Floyd ZE, Stephens JM. Interferon-gamma-mediated activation and ubiquitin-proteasome-dependent degradation of PPARgamma in adipocytes. J Biol Chem. 2002;277:4062–4068. doi: 10.1074/jbc.M108473200. [DOI] [PubMed] [Google Scholar]
- 43.Xu HE, et al. Molecular recognition of fatty acids by peroxisome proliferator-activated receptors. Mol Cell. 1999;3:397–403. doi: 10.1016/s1097-2765(00)80467-0. [DOI] [PubMed] [Google Scholar]
- 44.Forman BM, et al. 15-Deoxy-delta 12, 14-prostaglandin J2 is a ligand for the adipocyte determination factor PPAR gamma. Cell. 1995;83:803–812. doi: 10.1016/0092-8674(95)90193-0. [DOI] [PubMed] [Google Scholar]
- 45.Akazawa S, Sun F, Ito M, Kawasaki E, Eguchi K. Efficacy of troglitazone on body fat distribution in type 2 diabetes. Diabetes Care. 2000;23:1067–1071. doi: 10.2337/diacare.23.8.1067. [DOI] [PubMed] [Google Scholar]
- 46.Shadid S, Jensen MD. Effects of pioglitazone versus diet and exercise on metabolic health and fat distribution in upper body obesity. Diabetes Care. 2003;26:3148–3152. doi: 10.2337/diacare.26.11.3148. [DOI] [PubMed] [Google Scholar]
- 47.Okuno A, et al. Troglitazone increases the number of small adipocytes without the change of white adipose tissue mass in obese Zucker rats. J Clin Invest. 1998;101:1354–1361. doi: 10.1172/JCI1235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Gavrilova O, et al. Liver peroxisome proliferator-activated receptor gamma contributes to hepatic steatosis, triglyceride clearance, and regulation of body fat mass. J Biol Chem. 2003;278:34268–34276. doi: 10.1074/jbc.M300043200. [DOI] [PubMed] [Google Scholar]
- 49.Hevener AL, et al. Muscle-specific Pparg deletion causes insulin resistance. Nat Med. 2003;9:1491–1497. doi: 10.1038/nm956. [DOI] [PubMed] [Google Scholar]
- 50.Norris AW, et al. Muscle-specific PPARgamma-deficient mice develop increased adiposity and insulin resistance but respond to thiazolidinediones. J Clin Invest. 2003;112:608–618. doi: 10.1172/JCI17305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Rosen CJ. Bone remodeling, energy metabolism, and the molecular clock. Cell Metab. 2008;7:7–10. doi: 10.1016/j.cmet.2007.12.004. [DOI] [PubMed] [Google Scholar]
- 52.Hinoi E, et al. The sympathetic tone mediates leptin’s inhibition of insulin secretion by modulating osteocalcin bioactivity. J Cell Biol. 2008;183:1235–1242. doi: 10.1083/jcb.200809113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Lee NK, et al. Endocrine regulation of energy metabolism by the skeleton. Cell. 2007;130:456–469. doi: 10.1016/j.cell.2007.05.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Fulzele K, et al. Insulin receptor signaling in osteoblasts regulates postnatal bone acquisition and body composition. Cell. 2010;142:309–319. doi: 10.1016/j.cell.2010.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Ferron M, et al. Insulin signaling in osteoblasts integrates bone remodeling and energy metabolism. Cell. 2010;142:296–308. doi: 10.1016/j.cell.2010.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Lecka-Czernik B. Bone as a target of type 2 diabetes treatment. Curr Opin Investig Drugs. 2009;10:1085–1090. [PubMed] [Google Scholar]
- 57.Habib ZA, et al. Thiazolidinedione use and the longitudinal risk of fractures in patients with type 2 diabetes mellitus. J Clin Endocrinol Metab. 2010;95:592–600. doi: 10.1210/jc.2009-1385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Lecka-Czernik B, et al. Inhibition of Osf2/Cbfa1 expression and terminal osteoblast differentiation by PPARgamma2. J Cell Biochem. 1999;74:357–371. [PubMed] [Google Scholar]
- 59.Wan Y, Chong LW, Evans RM. PPAR-gamma regulates osteoclastogenesis in mice. Nat Med. 2007;13:1496–1503. doi: 10.1038/nm1672. [DOI] [PubMed] [Google Scholar]
- 60.Sottile V, Seuwen K, Kneissel M. Enhanced marrow adipogenesis and bone resorption in estrogen-deprived rats treated with the PPARgamma agonist BRL49653 (rosiglitazone) Calcif Tissue Int. 2004;75:329–337. doi: 10.1007/s00223-004-0224-8. [DOI] [PubMed] [Google Scholar]
- 61.Li M, et al. Surface-specific effects of a PPAR-gamma agonist, darglitazone, on bone in mice. Bone. 2006;39:796–806. doi: 10.1016/j.bone.2006.04.008. [DOI] [PubMed] [Google Scholar]
- 62.Wei W, et al. PGC1beta mediates PPARgamma activation of osteoclastogenesis and rosiglitazone-induced bone loss. Cell Metab. 2010;11:503–516. doi: 10.1016/j.cmet.2010.04.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Lazarenko OP, et al. Rosiglitazone induces decreases in bone mass and strength that are reminiscent of aged bone. Endocrinology. 2007;148:2669–2680. doi: 10.1210/en.2006-1587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Bendixen AC, et al. IL-4 inhibits osteoclast formation through a direct action on osteoclast precursors via peroxisome proliferator-activated receptor gamma 1. Proc Natl Acad Sci USA. 2001;98:2443–2448. doi: 10.1073/pnas.041493198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Hounoki H, et al. Activation of peroxisome proliferator-activated receptor gamma inhibits TNF-alpha-mediated osteoclast differentiation in human peripheral monocytes in part via suppression of monocyte chemoattractant protein-1 expression. Bone. 2008;42:765–774. doi: 10.1016/j.bone.2007.11.016. [DOI] [PubMed] [Google Scholar]
- 66.Ackert-Bicknell CL, et al. Strain-specific effects of rosiglitazone on bone mass, body composition, and serum insulin-like growth factor-I. Endocrinology. 2009;150:1330–1340. doi: 10.1210/en.2008-0936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Naveiras O, et al. Bone-marrow adipocytes as negative regulators of the haematopoietic microenvironment. Nature. 2009;460:259–263. doi: 10.1038/nature08099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Botolin S, McCabe LR. Inhibition of PPARgamma prevents type I diabetic bone marrow adiposity but not bone loss. J Cell Physiol. 2006;209:967–976. doi: 10.1002/jcp.20804. [DOI] [PubMed] [Google Scholar]
- 69.Justesen J, et al. Mice deficient in 11beta-hydroxysteroid dehydrogenase type 1 lack bone marrow adipocytes, but maintain normal bone formation. Endocrinology. 2004;145:1916–1925. doi: 10.1210/en.2003-1427. [DOI] [PubMed] [Google Scholar]
- 70.Sheng MH, et al. Histomorphometric studies show that bone formation and bone mineral apposition rates are greater in C3H/HeJ (high-density) than C57BL/6J (low-density) mice during growth. Bone. 1999;25:421–429. doi: 10.1016/s8756-3282(99)00184-2. [DOI] [PubMed] [Google Scholar]
- 71.Kawai M, Rosen CJ. Insulin-like growth factor-I and bone: lessons from mice and men. Pediatr Nephrol. 2009;24:1277–1285. doi: 10.1007/s00467-008-1040-6. [DOI] [PubMed] [Google Scholar]
- 72.Naveiras O, et al. Bone-marrow adipocytes as negative regulators of the haematopoietic microenvironment. Nature. 2009;460:259–263. doi: 10.1038/nature08099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Zvonic S, Floyd ZE, Mynatt RL, Gimble JM. Circadian rhythms and the regulation of metabolic tissue function and energy homeostasis. Obesity (Silver Spring) 2007;15:539–543. doi: 10.1038/oby.2007.544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Fu L, Patel MS, Bradley A, Wagner EF, Karsenty G. The molecular clock mediates leptin-regulated bone formation. Cell. 2005;122:803–815. doi: 10.1016/j.cell.2005.06.028. [DOI] [PubMed] [Google Scholar]
- 75.Turek FW, et al. Obesity and metabolic syndrome in circadian Clock mutant mice. Science. 2005;308:1043–1045. doi: 10.1126/science.1108750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Rudic RD, et al. BMAL1 and CLOCK, two essential components of the circadian clock, are involved in glucose homeostasis. PLoS Biol. 2004;2:e377. doi: 10.1371/journal.pbio.0020377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Oishi K, et al. Disrupted fat absorption attenuates obesity induced by a high-fat diet in Clock mutant mice. FEBS Lett. 2006;580:127–130. doi: 10.1016/j.febslet.2005.11.063. [DOI] [PubMed] [Google Scholar]
- 78.Holmbäck U, et al. Endocrine responses to nocturnal eating--possible implications for night work. Eur J Nutr. 2003;42:75–83. doi: 10.1007/s00394-003-0386-6. [DOI] [PubMed] [Google Scholar]
- 79.Cirelli C. The genetic and molecular regulation of sleep: from fruit flies to humans. Nat Rev Neurosci. 2009;10:549–560. doi: 10.1038/nrn2683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Balsalobre A, et al. Resetting of circadian time in peripheral tissues by glucocorticoid signaling. Science. 2000;289:2344–2347. doi: 10.1126/science.289.5488.2344. [DOI] [PubMed] [Google Scholar]
- 81.Ralph MR, Foster RG, Davis FC, Menaker M. Transplanted suprachiasmatic nucleus determines circadian period. Science. 1990;247:975–978. doi: 10.1126/science.2305266. [DOI] [PubMed] [Google Scholar]
- 82.Gallego M, Virshup DM. Post-translational modifications regulate the ticking of the circadian clock. Nat Rev Mol Cell Biol. 2007;8:139–148. doi: 10.1038/nrm2106. [DOI] [PubMed] [Google Scholar]
- 83.Preitner N, et al. The orphan nuclear receptor REV-ERBalpha controls circadian transcription within the positive limb of the mammalian circadian oscillator. Cell. 2002;110:251–260. doi: 10.1016/s0092-8674(02)00825-5. [DOI] [PubMed] [Google Scholar]
- 84.Kornmann B, Schaad O, Bujard H, Takahashi JS, Schibler U. System-driven and oscillator-dependent circadian transcription in mice with a conditionally active liver clock. PLoS Biol. 2007;5:e34. doi: 10.1371/journal.pbio.0050034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Evans RM, Barish GD, Wang YX. PPARs and the complex journey to obesity. Nat Med. 2004;10:355–361. doi: 10.1038/nm1025. [DOI] [PubMed] [Google Scholar]
- 86.Anan F, et al. Pioglitazone shift circadian rhythm of blood pressure from non-dipper to dipper type in type 2 diabetes mellitus. Eur J Clin Invest. 2007;37:709–714. doi: 10.1111/j.1365-2362.2007.01854.x. [DOI] [PubMed] [Google Scholar]
- 87.Liu C, Li S, Liu T, Borjigin J, Lin JD. Transcriptional coactivator PGC-1alpha integrates the mammalian clock and energy metabolism. Nature. 2007;447:477–481. doi: 10.1038/nature05767. [DOI] [PubMed] [Google Scholar]
- 88.Green CB, Besharse JC. Identification of a novel vertebrate circadian clock-regulated gene encoding the protein nocturnin. Proc Natl Acad Sci USA. 1996;93:14884–14888. doi: 10.1073/pnas.93.25.14884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Wang Y, et al. Rhythmic expression of Nocturnin mRNA in multiple tissues of the mouse. BMC Dev Biol. 2001;1:9. doi: 10.1186/1471-213X-1-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Garbarino-Pico E, et al. Immediate early response of the circadian polyA ribonuclease nocturnin to two extracellular stimuli. RNA. 2007;13:745–755. doi: 10.1261/rna.286507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Baggs JE, Green CB. Nocturnin, a deadenylase in Xenopus laevis retina: a mechanism for posttranscriptional control of circadian-related mRNA. Curr Biol. 2003;13:189–198. doi: 10.1016/s0960-9822(03)00014-9. [DOI] [PubMed] [Google Scholar]
- 92.Dupressoir A, et al. Identification of four families of yCCR4- and Mg2+-dependent endonuclease-related proteins in higher eukaryotes, and characterization of orthologs of yCCR4 with a conserved leucine-rich repeat essential for hCAF1/hPOP2 binding. BMC Genomics. 2001;2:9. doi: 10.1186/1471-2164-2-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Tucker M, et al. The transcription factor associated Ccr4 and Caf1 proteins are components of the major cytoplasmic mRNA deadenylase in Saccharomyces cerevisiae. Cell. 2001;104:377–386. doi: 10.1016/s0092-8674(01)00225-2. [DOI] [PubMed] [Google Scholar]
- 94.Kawai M, et al. A circadian-regulated gene, Nocturnin, promotes adipogenesis by stimulating PPAR-gamma nuclear translocation. Proc Natl Acad Sci USA. 2010;107:10508–10513. doi: 10.1073/pnas.1000788107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Cao SX, Dhahbi JM, Mote PL, Spindler SR. Genomic profiling of short- and long-term caloric restriction effects in the liver of aging mice. Proc Natl Acad Sci USA. 2001;98:10630–10635. doi: 10.1073/pnas.191313598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Kawai M, et al. Nocturnin: a circadian target of Pparg-induced adipogenesis. Ann NY Acad Sci. 2010;1192:131–138. doi: 10.1111/j.1749-6632.2009.05221.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Massiera F, et al. A Western-like fat diet is sufficient to induce a gradual enhancement in fat mass over generations. J Lipid Res. 2010;51:2352–2361. doi: 10.1194/jlr.M006866. [DOI] [PMC free article] [PubMed] [Google Scholar]