Abstract
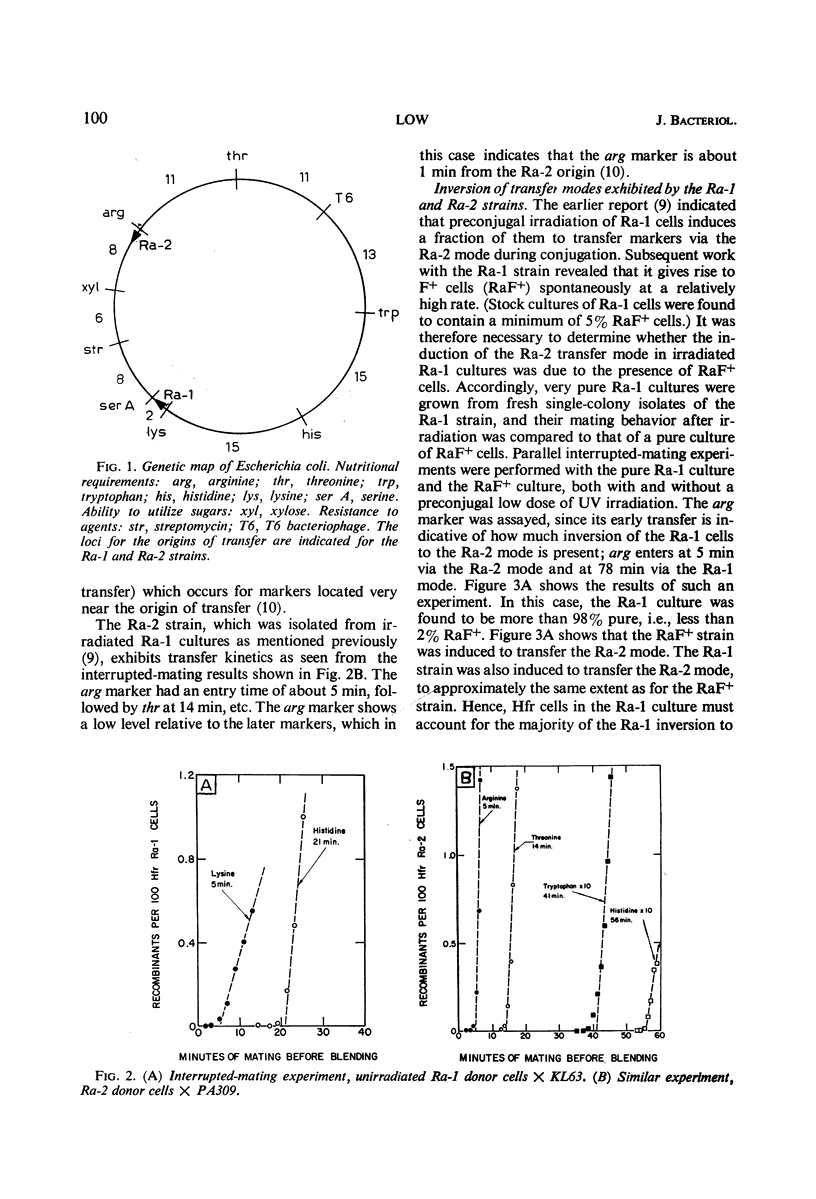
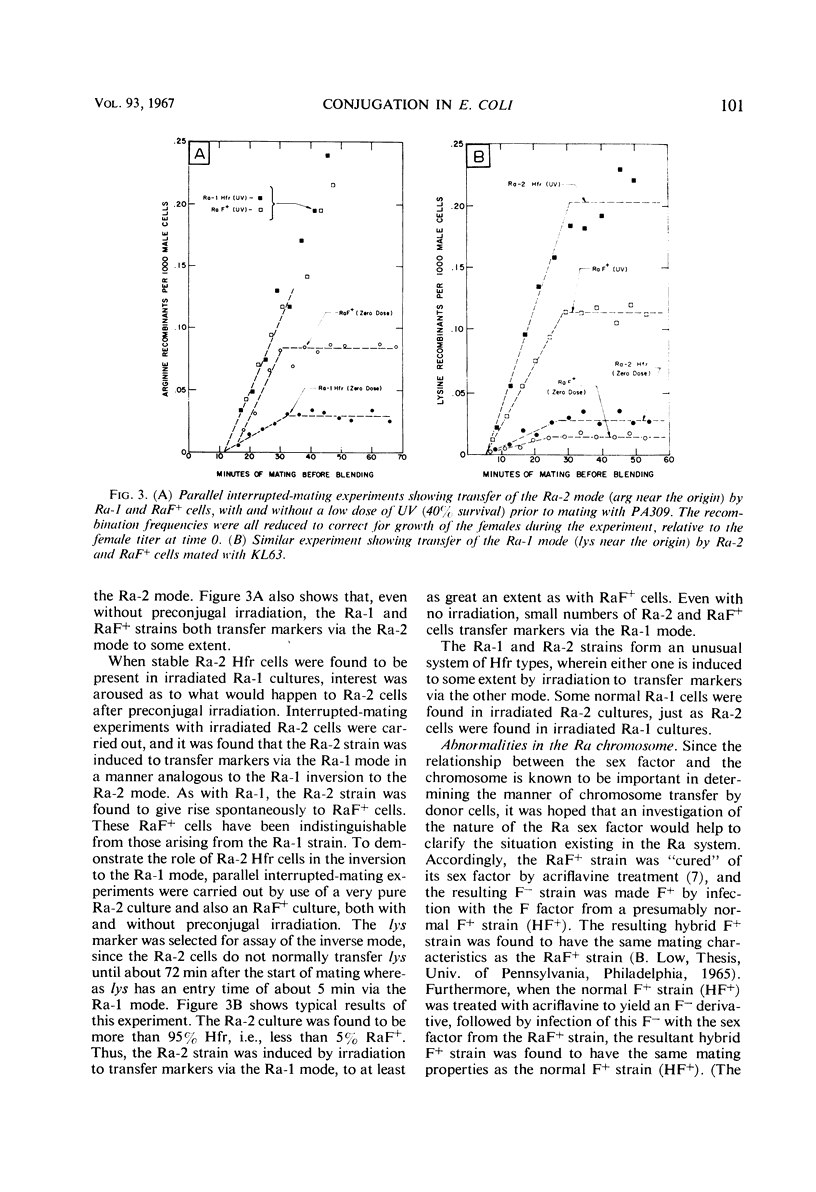
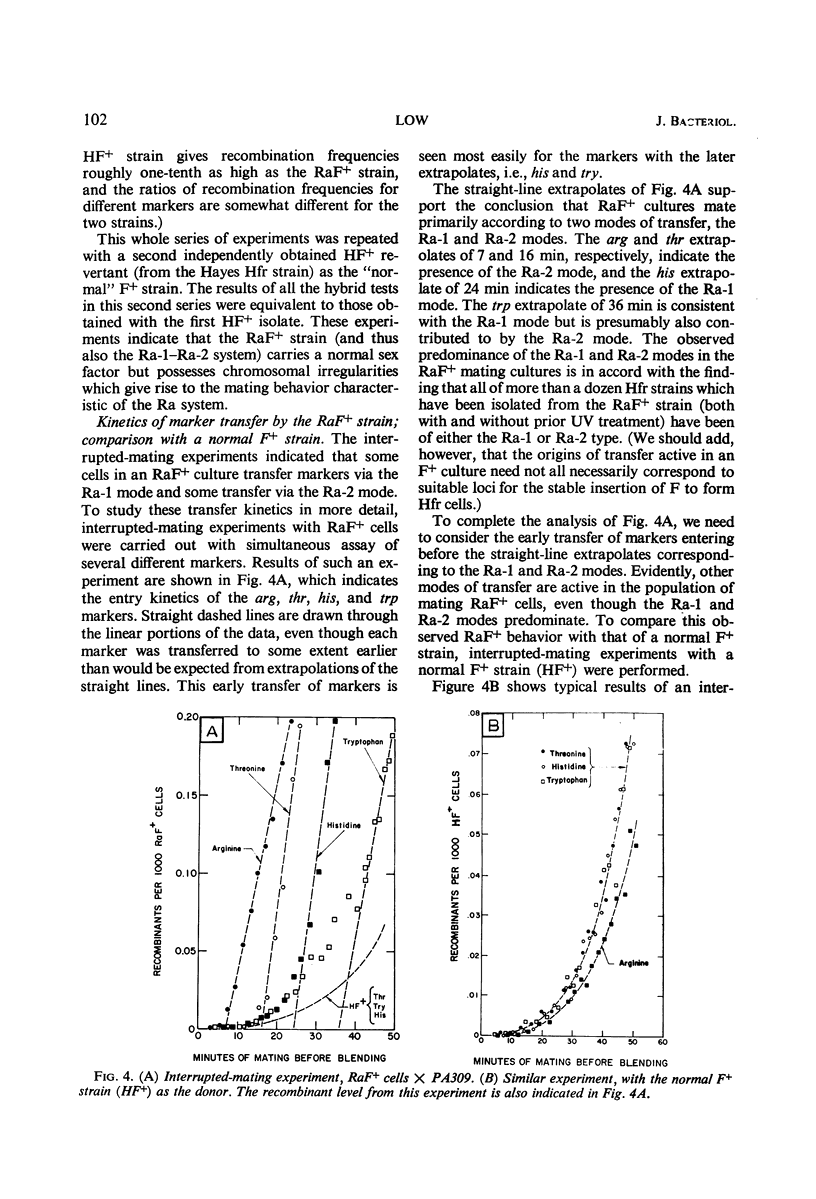
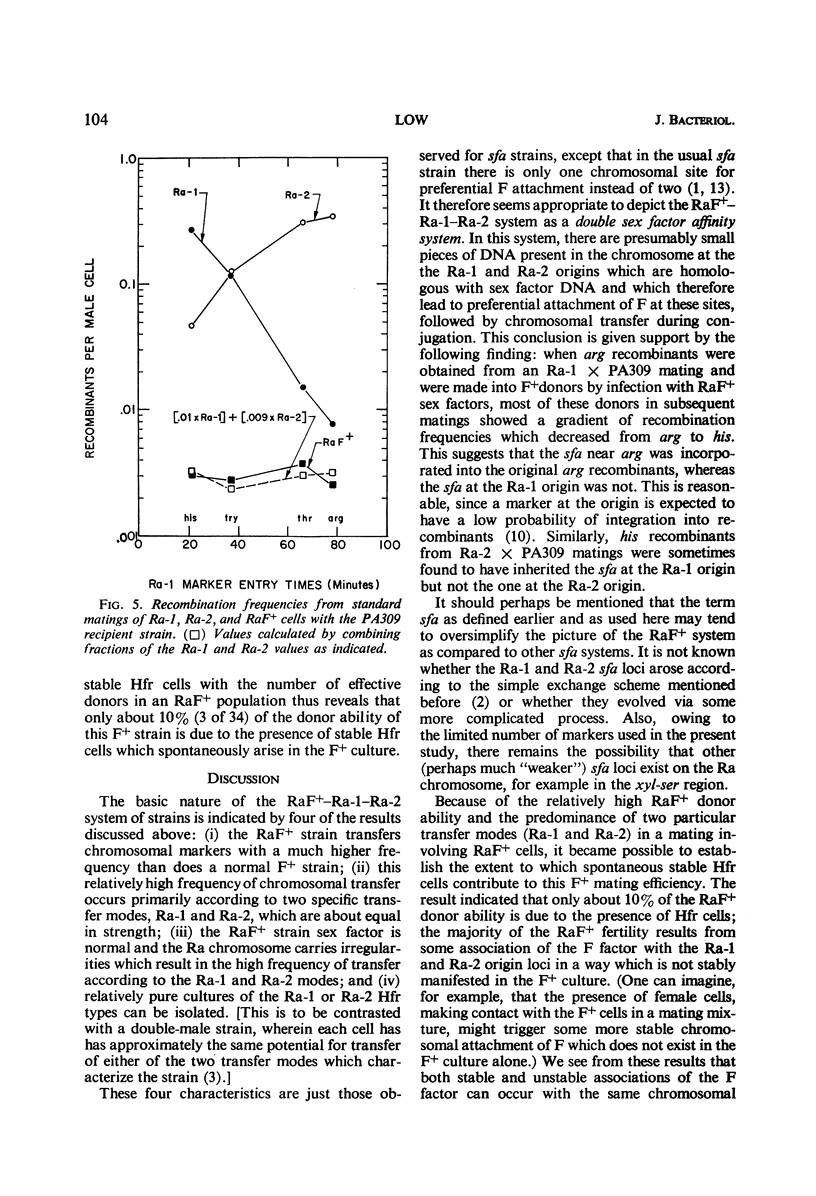
A study was made of the mating properties of an unusual system of interconvertible donor strains of Escherichia coli K-12: Ra-1, Ra-2, and RaF+. The Ra-1 and Ra-2 strains are Hfr strains whose origins are widely separated on the chromosome and whose transfer modes proceed in the opposite direction from one another. When Ra-1 cells were mated with females, a small fraction of the donors transferred markers via the Ra-2 mode. This effect was enhanced by preconjugal ultraviolet (UV) treatment of the Ra-1 cells. Among the survivors of UV-treated Ra-1 cells, a few stable Ra-2 cells were found. When Ra-2 cells were used as the donors, some of them were found to mate via the Ra-1 mode, in analogy with the Ra-1 to Ra-2 alteration with inversion of F mentioned above. Related experiments suggested that the inversion occurs by detachment of the F factor from one Hfr origin locus, followed by reassociation of the F factor with the other Hfr origin locus. Both the Ra-1 and Ra-2 strains reverted spontaneously to an F+ strain, called RaF+. Cultures of RaF+ cells were found to mate primarily according to the Ra-1 and Ra-2 transfer modes, with smaller contributions also coming from transfer modes with origins elsewhere on the chromosome in a way which is similar to the transfer of markers from a normal F+ strain. The RaF+ sex factor was found to be wild type, whereas the chromosome was found to carry irregularities (sex factor affinity loci) at the locations of the Ra-1 and Ra-2 origins. Only about 10% of the donor capacity of the RaF+ strain was due to stable spontaneous Hfr cells in cultures of RaF+ cells.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- ADELBERG E. A., BURNS S. N. Genetic variation in the sex factor of Escherichia coli. J Bacteriol. 1960 Mar;79:321–330. doi: 10.1128/jb.79.3.321-330.1960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- ADELBERG E. A., PITTARD J. CHROMOSOME TRANSFER IN BACTERIAL CONJUGATION. Bacteriol Rev. 1965 Jun;29:161–172. doi: 10.1128/br.29.2.161-172.1965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- CLARK A. J. Genetic analysis of a "double male" strain of Escherichia coli K-12. Genetics. 1963 Jan;48:105–120. doi: 10.1093/genetics/48.1.105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- FALKOW S., CITARELLA R. V. MOLECULAR HOMOLOGY OF F-MEROGENOTE DNA. J Mol Biol. 1965 May;12:138–151. doi: 10.1016/s0022-2836(65)80288-1. [DOI] [PubMed] [Google Scholar]
- Gross J. D., Caro L. Genetic transfer in bacterial mating. Science. 1965 Dec 24;150(3704):1679–1684. doi: 10.1126/science.150.3704.1679. [DOI] [PubMed] [Google Scholar]
- HIROTA Y., LIJIMA T. Acriflavine as an effective agent for eliminating F-factor in Escherichia coli K-12. Nature. 1957 Sep 28;180(4587):655–656. doi: 10.1038/180655a0. [DOI] [PubMed] [Google Scholar]
- Hewitt R., Billen D. Reorientation of chromosome replication after exposure to ultraviolet light of Escherichia coli. J Mol Biol. 1965 Aug;13(1):40–53. doi: 10.1016/s0022-2836(65)80078-x. [DOI] [PubMed] [Google Scholar]
- LOW B., WOOD T. H. A QUICK AND EFFICIENT METHOD FOR INTERRUPTION OF BACTERIAL CONJUGATION. Genet Res. 1965 Jul;6:300–303. doi: 10.1017/s001667230000416x. [DOI] [PubMed] [Google Scholar]
- Low B. Low recombination frequency for markers very near the origin in conjugation in E. coli. Genet Res. 1965 Nov;6(3):469–473. doi: 10.1017/s0016672300004341. [DOI] [PubMed] [Google Scholar]
- TAYLOR A. L., THOMAN M. S. THE GENETIC MAP OF ESCHERICHIA COLI K-12. Genetics. 1964 Oct;50:659–677. doi: 10.1093/genetics/50.4.659. [DOI] [PMC free article] [PubMed] [Google Scholar]


