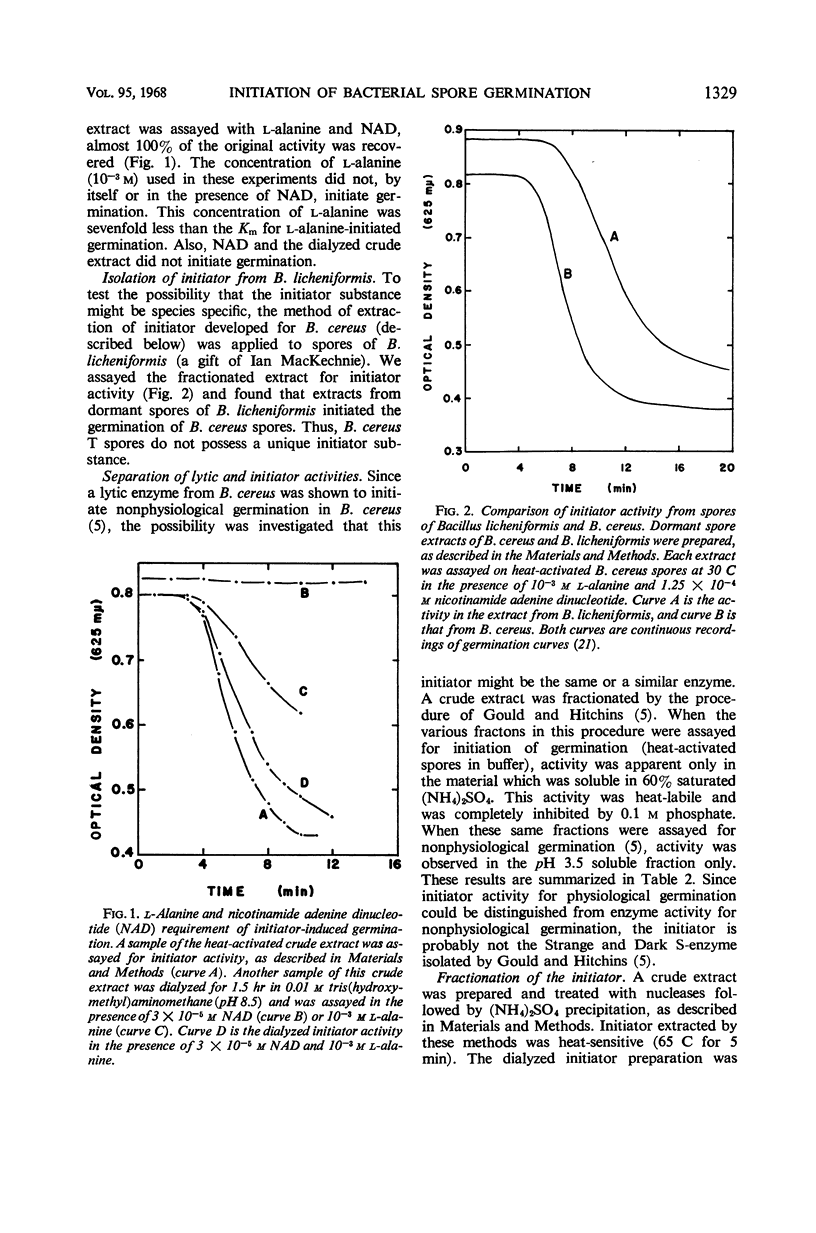
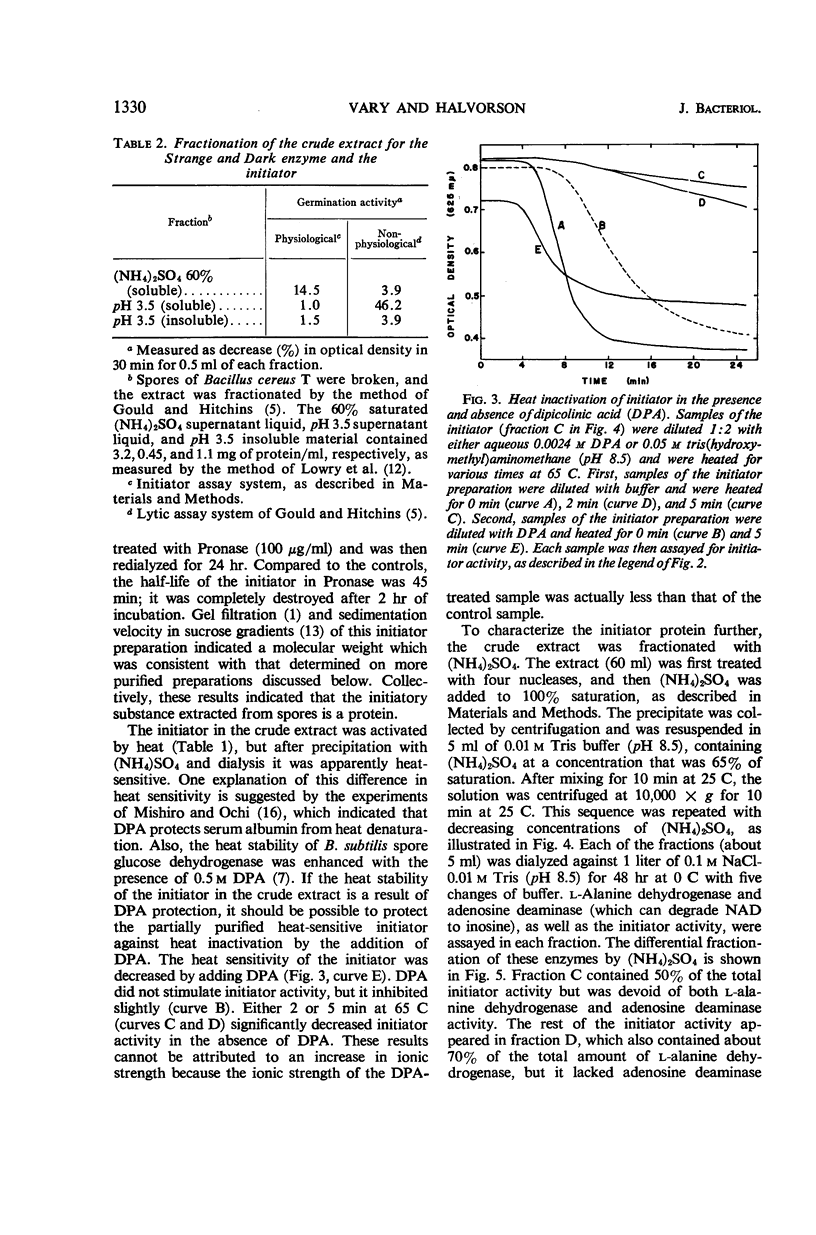
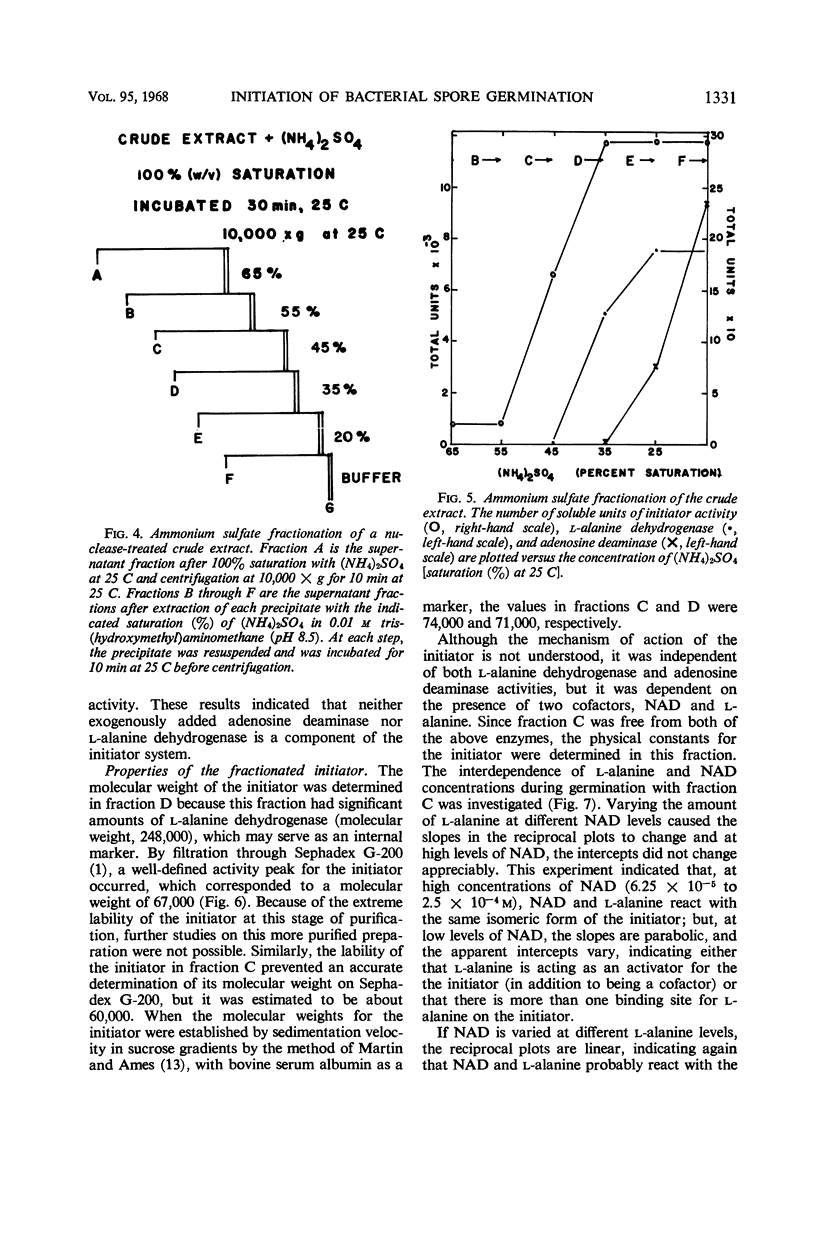
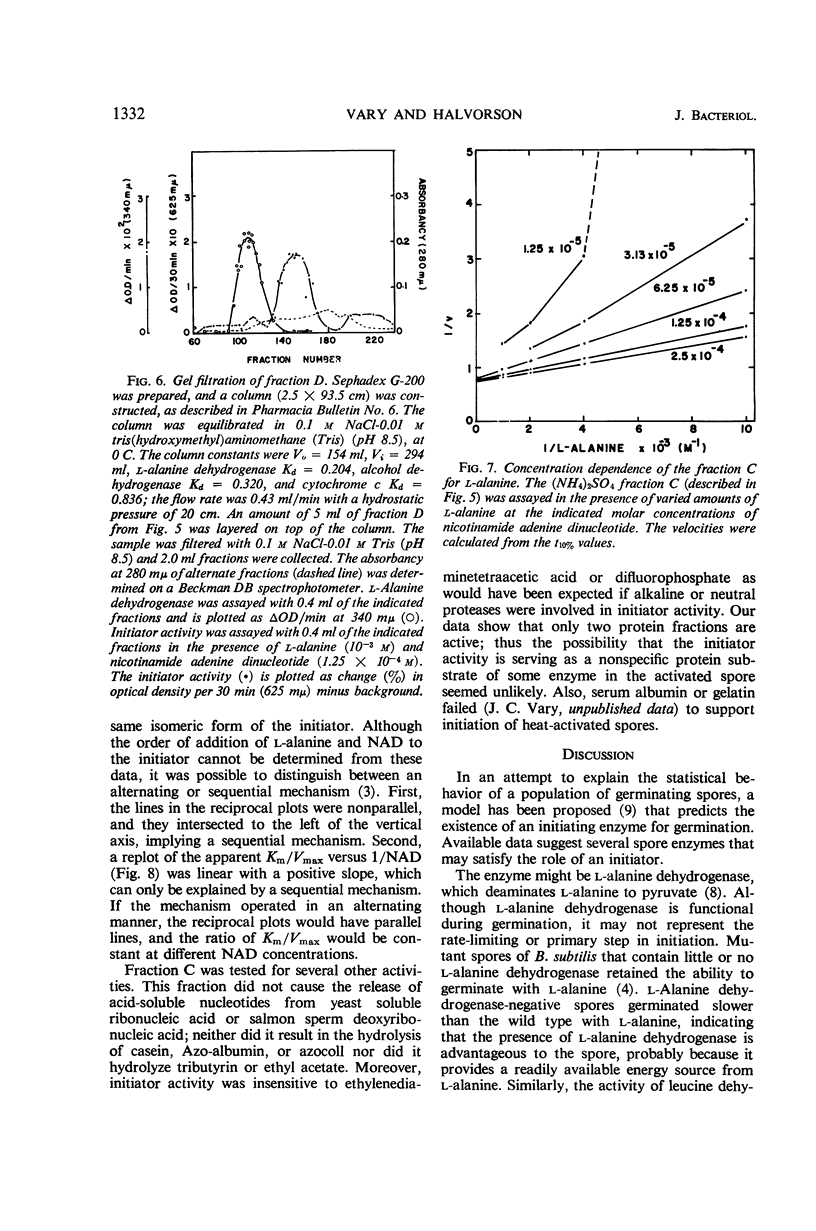
Abstract
To investigate the problem of initiation in bacterial spore germination, we isolated, from extracts of dormant spores of Bacillus cereus strain T and B. licheniformis, a protein that initiated spore germination when added to a suspension of heat-activated spores. The optimal conditions for initiatory activity of this protein (the initiator) were 30 C in 0.01 to 0.04 m NaCl and 0.01 m tris(hydroxymethyl)aminomethane (pH 8.5). The initiator was inhibited by phosphate but required two co-factors, l-alanine (1/7 of Km for l-alanine-inhibited germination) and nicotinamide adenine dinucleotide (1.25 × 10−4m). In the crude extract, the initiator activity was increased 3.5-fold by heating the extract at 65 C for 10 min, but the partially purified initiator preparation was completely heat-sensitive (65 C for 5 min). Heat stability could be conferred on the purified initiator by adding 10−3m dipicolinic acid. A fractionation of this protein that excluded l-alanine dehydrogenase and adenosine deaminase from the initiator activity was developed. The molecular weight of the initiator was estimated as 7 × 104. The kinetics of germination in the presence of initiator were examined at various concentrations of l-alanine and nicotinamide adenine dinucleotide.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Andrews P. The gel-filtration behaviour of proteins related to their molecular weights over a wide range. Biochem J. 1965 Sep;96(3):595–606. doi: 10.1042/bj0960595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- CHURCH B. D., HALVORSON H., HALVORSON H. O. Studies on spore germination: its independence from alanine racemase activity. J Bacteriol. 1954 Oct;68(4):393–399. doi: 10.1128/jb.68.4.393-399.1954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- CLELAND W. W. The kinetics of enzyme-catalyzed reactions with two or more substrates or products. I. Nomenclature and rate equations. Biochim Biophys Acta. 1963 Jan 8;67:104–137. doi: 10.1016/0006-3002(63)91800-6. [DOI] [PubMed] [Google Scholar]
- Gould G. W., King W. L. Role of amino-acids in the germination of bacterial spores by subtilisn. Nature. 1966 Sep 24;211(5056):1431–1432. doi: 10.1038/2111431a0. [DOI] [PubMed] [Google Scholar]
- Hachisuka Y., Tochikubo K., Yokoi Y., Murachi T. The action of dipicolinic acid and its chemical analogues on the heat stability of glucose dehydrogenase of Bacillus subtilis spores. J Biochem. 1967 May;61(5):659–661. doi: 10.1093/oxfordjournals.jbchem.a128598. [DOI] [PubMed] [Google Scholar]
- Halvorson H. O., Vary J. C., Steinberg W. Developmental changes during the formation and breaking of the dormant state in bacteria. Annu Rev Microbiol. 1966;20:169–188. doi: 10.1146/annurev.mi.20.100166.001125. [DOI] [PubMed] [Google Scholar]
- JANSSEN F. W., LUND A. J., ANDERSON L. E. Colorimetric assay for dipicolinic acid in bacterial spores. Science. 1958 Jan 3;127(3288):26–27. doi: 10.1126/science.127.3288.26. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- MARTIN R. G., AMES B. N. A method for determining the sedimentation behavior of enzymes: application to protein mixtures. J Biol Chem. 1961 May;236:1372–1379. [PubMed] [Google Scholar]
- MCCORMICK N. G. KINETICS OF SPORE GERMINATION. J Bacteriol. 1965 May;89:1180–1185. doi: 10.1128/jb.89.5.1180-1185.1965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mishiro Y., Ochi M. Effect of dipicolinate on the heat denaturation of proteins. Nature. 1966 Sep 10;211(5054):1190–1190. doi: 10.1038/2111190a0. [DOI] [PubMed] [Google Scholar]
- Moberly B. J., Shafa F., Gerhardt P. Structural details of anthrax spores during stages of transformation into vegetative cells. J Bacteriol. 1966 Jul;92(1):220–228. doi: 10.1128/jb.92.1.220-228.1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- STRANGE R. E., DARK F. A. A cell-wall lytic enzyme associated with spores of Bacillus species. J Gen Microbiol. 1957 Feb;16(1):236–249. doi: 10.1099/00221287-16-1-236. [DOI] [PubMed] [Google Scholar]
- Sierra G. Germination of bacterial endospores with subtilopeptidases. Can J Microbiol. 1967 May;13(5):489–501. doi: 10.1139/m67-064. [DOI] [PubMed] [Google Scholar]
- VARY J. C., HALVORSON H. O. KINETICS OF GERMINATION OF BACILLUS SPORES. J Bacteriol. 1965 May;89:1340–1347. doi: 10.1128/jb.89.5.1340-1347.1965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vallee B. L., Hoch F. L. ZINC, A COMPONENT OF YEAST ALCOHOL DEHYDROGENASE. Proc Natl Acad Sci U S A. 1955 Jun 15;41(6):327–338. doi: 10.1073/pnas.41.6.327. [DOI] [PMC free article] [PubMed] [Google Scholar]


