GRK1 is a key enzyme in deactivation of photoreceptor visual pigment. Its role and contribution in recovery of rod photoreceptors is examined in mice expressing varying levels of the enzyme.
Abstract
Purpose.
Rod photoreceptors are exquisitely sensitive light detectors that function in dim light. The timely inactivation of their light responses is critical for the ability of rods to reliably detect and count photons. A key step in the inactivation of the rod transduction is the phosphorylation of the rod visual pigment, rhodopsin, catalyzed by G-protein-dependent receptor kinase 1 (GRK1). Absence of GRK1 greatly prolongs the photoreceptors' light response and enhances their susceptibility to degeneration. This study examined the light responses from mouse rods expressing various levels of GRK1 to evaluate how their function is modulated by rhodopsin inactivation.
Methods.
Transretinal and single-cell rod electrophysiological recordings were obtained from several strains of mice expressing GRK1 at 0.3- to 3-fold the wild-type levels. The effect of GRK1 expression level on the function of mouse rods was examined in darkness and during background adaptation.
Results.
Altering the expression of GRK1 from 0.3- to 3-fold that in wild-type rods had little effect on the single photon response amplitude. Notably, increasing the expression level of GRK1 accelerated the dim flash response shut off but had no effect on the saturated response shut off. Additionally, GRK1 excess abolished the acceleration of saturated responses shut off during light adaptation.
Conclusions.
These results demonstrate that rhodopsin inactivation can modulate the kinetics of recovery from dim light stimulation. More importantly, the ratio of rhodopsin kinase to its modulator recoverin appears critical for the proper adaptation of rods and the acceleration of their response shut off in background light.
Rod photoreceptors are exquisitely sensitive light detectors that are perfectly suited for function in dim light.1 The detection of light and the conversion of its energy into an electric signal take place at the membrane discs in the outer segments of rod photoreceptors. Phototransduction is initiated by the absorption of a photon by a molecule of visual pigment, rhodopsin.2 In its active state, rhodopsin (R*) binds to a G protein, transducin, triggering the exchange of guanosine-5′-triphosphate (GTP) for guanosine-5′-diphosphate (GDP) on its α-subunit (Tα). In turn, the activated Tα-GTP binds to an effector enzyme, cGMP phosphodiesterate (PDE) which eventually results in closure of cGMP-gated channels in the outer segment. The amplification of the rod signal is produced by two phototransduction components: a single R* activating multiple Tα subunits, and a single Tα/PDE complex hydrolyzing multiple cGMP molecules. Response termination requires the timely inactivation of both R* and Tα/PDE. First, R* is partially inactivated on phosphorylation by rhodopsin kinase (GRK1),3,4 a reaction inhibited in darkness by recoverin.5 Phosphorylated rhodopsin is then completely inactivated on binding to arrestin.6 Transducin is inactivated in a GTP hydrolysis reaction catalyzed by regulator of G protein signalling 9 (RGS9)7 which returns transducin into its inactive GDP-bound state.
The molecular mechanisms that rate-limit the inactivation of the transduction cascade and dominate the light response shut off have been an active area of research but are still subjects of debate. While early studies indicated that shut off of the transduction cascade is controlled by the inactivation of R*8, a recent study demonstrated that the inactivation of the Tα/PDE complex is the rate-limiting step in the shut off of the light response in mouse rods.9 The same study stated that overexpression of rhodopsin kinase does not affect the termination of the light response and concluded that the inactivation of R* is very rapid (≤80 ms) and substantially faster than that of Tα/PDE (see also Ref. 10). However, whether the inactivation of R* by rhodopsin kinase is slow enough to modulate the overall response kinetics in rods remains controversial.11 More importantly, it is not known whether rhodopsin phosphorylation affects the function of rods during light adaptation.
We recently generated transgenic mice with rods and cones overexpressing GRK1 driven by the full length rhodopsin kinase promoter12 in preparation for studying how GRK1 expression modulates cone function. We performed initial recordings from the rods of these mice to confirm that, as previously suggested, overexpression of GRK1 in mouse rods does not affect the kinetics of their responses.9 Surprisingly, we observed a notable acceleration of rod response shut off in rods overexpressing GRK1. We proceeded to characterize in detail the effect of GRK1 expression level on the function of mouse rods in darkness and during background adaptation. Our results demonstrate that R* inactivation by rhodopsin kinase affects the kinetics of the single-photon response and plays a role in the background adaptation of mammalian rods.
Methods
Animals
Transgenic mice were generated, genotyped, and maintained on the C57BL6 background as previously described.12 All experimental protocols were in accordance with the Guide for the Care and Use of Laboratory Animals and were approved by the institutional animal care and use committees at Schepens Eye Research Institute, Washington University, State University of New York at Buffalo, and Roswell Park Cancer Institute.
Analysis of Expression Levels
Quantitative RT-PCR and immunoblot analysis were carried out as previously described to estimate the levels of GRK1 in overexpressing mice.12 Briefly, total RNA and protein fractions were isolated from whole globes or retinas using the Trizol method. RNA samples from each line were reverse transcribed, and the amplification of a Grk1 cDNA was primed and monitored in real time in the presence of SYBR green as previously described13 to determine the cycle to detection threshold (Ct). Comparison of the Ct values or those corrected for the levels of actin, or beta 2 microglobulin (ΔCt) served as the basis for the calculation of the relative Grk1 RNA content across the samples. The reverse transcription and amplifications were performed in triplicate using RNA of four individual animals per strain.
For protein blotting, Trizol pellets were solubilized in isoelectric focusing buffer or and applied to gel at serial dilutions for immunoblot analysis and subsequent chemiluminescent signal detection as previously described.12 Monoclonal GRK1 antibodies (G8 or D11; Abcam, Boston, MA), polyclonal anti-recoverin (kindly provided by Alexander M. Dizhoor, Salus University) and RGS9-1 antibody (kindly provided by Ching-Kang Jason Chen, Virginia Commonwealth University) or anti-mouse beta actin (Abcam) were used as primary antibodies. Actin band signal served as the internal standard against which the signal for GRK1 band was normalized before comparison across samples using the previously described algorithm.12 At least four different pools of eyes were examined per strain to determine the relative levels of expression.
Immunostaining
Cryosections were taken of the eyes from various mouse strains and stained with a combination of peanut neuroagglutinin (PNA) and GRK1 polyclonal antibody 8585 as previously described.14
Light Stimulation
For both transretinal and single-cell recordings, test flashes at 500 nm were delivered from a calibrated light source via computer-controlled shutters. Flash intensity was set by a combination of neutral density filters. Calibrated 520-nm light was used for background stimulation.
Transretinal Recordings
Mice were maintained in 12/12 hour light/dark cycle and dark-adapted overnight before experiments. After euthanatization, the eyes were removed under dim red light. All subsequent manipulations were done under infrared light. A quarter of the isolated retina was mounted on filter paper photoreceptor-side up and placed on the recording chamber with an electrode connected to the bottom. A reference electrode was placed above the retina. The perfusion Locke solution (112 mM NaCl, 3.6 mM KCl, 2.4 mM MgCl2, 1.2 mM CaCl2, 10 mM HEPES, 20 mM NaHCO3, 3 mM Na2-succinate, 0.5 mM Na-glutamate, 10 mM glucose) was equilibrated with 95% O2/5% CO2, heated to 34–37°C, and contained, in addition, 2 mM l-glutamic acid to block higher order components of photoresponse.15 The electrode solution contained 140 mM NaCl, 3.6 mM KCl, 2.4 mM MgCl2, 1.2 mM CaCl2, 3 mM HEPES, 10 mM glucose, pH 7.4. The electrode under the retina contained, in addition, 10 mM BaCl2 to suppress glial components.16,17
Rod Single-Cell Recordings
For single-cell recordings retinas were prepared as for transretinal recordings. Recordings were done from small pieces of retina placed in a recording chamber fit to an inverted microscope and perfused at 34–37°C. The perfusion Locke solution contained 112 mM NaCl, 3.6 mM KCl, 2.4 mM MgCl2, 1.2 mM CaCl2, 10 mM HEPES, 20 mM NaHCO3, 3 mM Na2-succinate, 0.5 mM Na-glutamate, 10 mM glucose, and was equilibrated with 95% O2/5% CO2, pH 7.4. Membrane current was recorded from the outer segment of a single rod photoreceptor protruding from the retina and drawn into the electrode. The suction electrode was filled with solution containing 140 mM NaCl, 3.6 mM KCl, 2.4 mM MgCl2, 1.2 mM CaCl2, 3 mM HEPES, 10 mM glucose, pH 7.4. Responses were amplified by a current-to-voltage converter (Axopatch 200B; Molecular Devices, Sunnyvale, CA), low-pass filtered by an eight-pole Bessel filter with a cut off frequency of 30 Hz (Model 3382; Krohn-Hite), digitized at 1 kHz and stored on a computer using acquisition software (pClamp 8.2; Molecular Devices).
The kinetics of dim flash responses were determined as follows: time to peak was measured from the mid-point of the test flash; integration time was estimated as the time integral of the dim flash response after normalization of its amplitude to unity; recovery time constant (τrec) was estimated from a single exponential fit to the second half of the response shut off phase. Finally, the dominant time constant (τD) was determined from the slope of the time in saturation versus flash intensity plot for saturated flash responses. Rod sensitivity was monitored from the shift in the flash intensity required to produce half maximal response (Io). The rod single photon response was estimated by dividing the amplitude of a dim flash response by the flash intensity and the rod collecting area 0.5 μm2.18 We used the two-tailed unpaired Student's t-test to determine the significance of difference unless otherwise noted.
Results
The Expression Level of Rhodopsin Kinase in Mouse Rods Can Be Modulated over a 10-Fold Range
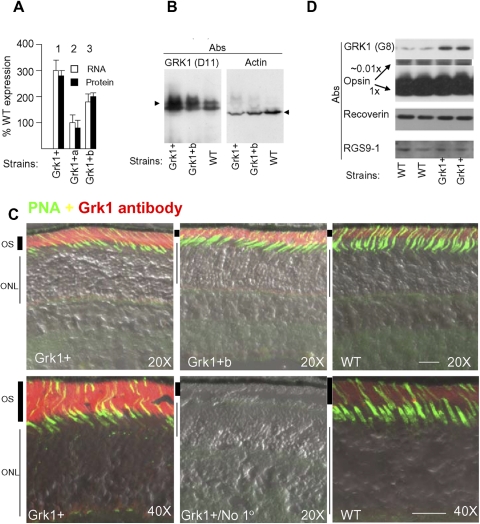
We generated several transgenic mouse lines with varying levels of rhodopsin kinase (GRK1) expression to study the impact of rhodopsin phosphorylation on the rod light response shut off. The lines carried copies of a bacterial artificial chromosome (BAC) construct in their genome containing the mouse Grk1 gene along with the entire 5′ and 3′ flanking sequences.12 Of the three lines with the full transgene, lines 1 and 3 designated Grk1+ and Grk1+b overexpressed GRK1 based on quantitative RT-PCR and immunoblot-based criteria (Figs. 1A and 1B), whereas line 2 (Grk1+a) did not overexpress GRK1 (Fig. 1A). In Grk1+b and Grk1+ lines, expressions were at 200% and 300% of the WT background, respectively (see also Ref. 12). The retinas overexpressing GRK1 showed no evidence of degeneration at least until 18 months of age. Retinal cryosections colabeled with antibodies recognizing GRK1 and the cone sheath marker peanut neuroagglutinin (PNA) showed remarkably uniform expression of GRK1 in rods and cones in both lines (Fig. 1C). Because of the higher kinase expression levels of the Grk1+ line and its markedly greater fertility, our studies focused primarily on this line. To expand the range of GRK1 expression levels, we also used heterozygous Grk1 knockout mice (Grk1+/−), with rods expressing GRK1 at 30% of WT rods as previously quantified.11 Together, the Grk1+/− and Grk1+ animals gave us a 10-fold range of GRK1 expression levels, from 0.3 to 3-fold the WT levels. Alterations in Grk1 expression did not affect the levels of other key molecules involved in regulation of recovery including recoverin, RGS9, or arrestin as seen in Figure 1D and as documented previously.11,19
Figure 1.
Grk1 expression in transgenic mouse lines. (A) The mouse lines were generated by transgenesis with a BAC containing the full-length mouse Grk1 gene and its entire flanking sequences. The eye expression levels were quantified in the three strains using real-time RT-PCR (empty bars) and quantitative immunoblotting (filled bars). For real-time RT-PCR and quantitative immunoblots the data from individual mouse samples in triplicates were averaged at various RNA or protein loads to ensure consistency. The error bars represent SEM. Relative Grk1 expression in Grk1+/− were quantified previously by Doan et al. at 0.32 ± 0.03 the WT levels using quantitative immunoblots.11 (B) Immunoblot shows overexpression of GRK1 bands in the overexpressing Grk1+ and Grk1+b strains. The immunoblot was probed with monoclonal antibody D11 against an amino terminal domain epitope, which recognizes both full-length and presumably the alternatively spliced truncated form of GRK1 previously described.37,38 For comparison the same blot probed with actin antibody is shown. The WT sample was intentionally overloaded to qualitatively demonstrate the GRK1 overexpression in the transgenic samples. Immunoblots from six animals were compared with WT in this manner. (C) Cryosections of eyes from Grk1+, Grk1+b, and WT strains were reacted with 8585 polyclonal anti-GRK1 antibody (red) and the cone sheath marker peanut neuroagglutinin (PNA, green). Images show uniform overexpression of GRK1 with correct localization in outer segments (OS) across photoreceptors. Sections 10-μm thick were incubated with identical cocktail of primary antibodies except the one control that lacked any primary reactants (No 1o). They were subsequently incubated with the secondary Alexa- 488 and 568 antibodies. The same excitation and acquisition parameters were used on a confocal microscope (Zeiss meta LSM; Carl Zeiss, Germany) for obtaining 12 bit depth images with the sensitivities set so that the signal intensities from Grk1+ tissue did not saturate the photomultiplier. The images were processed with the software designed for the confocal microscope. Objective lens magnifications indicated by 20× and 40×. Twenty micrometer scale bar is shown on each of the image panels (bottom). ONL, outer nuclear layer. (D) Immunoblot of rod outer segments showing that other key components of the recovery pathway including opsin, Rgs9-1, and recoverin are unaffected by GRK1 overexpression.
Overexpression of Rhodopsin Kinase Modulates the Overall Rod Retina Response
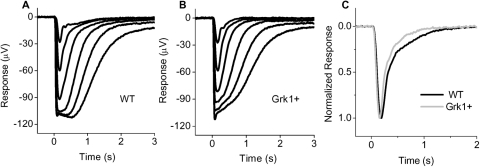
We initially compared the light responses of Grk1+ retinas with those of WT retinas to evaluate the functional impact of increasing GRK1 on rod function. Figures 2A and 2B show representative families of transretinal flash responses obtained from isolated dark-adapted wild-type (WT) and GRK1+ retinas. These traces were obtained under conditions that pharmacologically suppress synaptic transmission and postsynaptic activity (see Methods). As a result, the light-induced electrical deflections reflect the en mass rod activity with minimal contribution from second-order neurons or cones. As seen in Table 1, the mean rod sensitivities and response amplitudes across the strains remained statistically indistinguishable based on comparison of the intensities required to produce half maximal response (Io) and the maximal response amplitudes (Rmax). These results are consistent with our morphologic studies and demonstrate that increasing by threefold the expression level of GRK1 in mouse rods from animals raised in cyclic light conditions does not adversely affect photoreceptor morphology (see also Ref. 12). However, in the course of these experiments, we observed a notable acceleration of the response shut off in rods overexpressing GRK1 (Fig. 2C). A more systematic examination revealed a significant (P < 0.001) shortening of the response duration (Tint) from 370 ms to 259 ms as a result of the GRK1 overexpression (Table 1). The time to peak was also significantly (P = 0.01) reduced in the Grk1+ strain at 166 ms compared with 184 ms in WT rods (Table 1).
Figure 2.
Typical transretinal flash response families from (A) WT and (B) GRK1 overexpressing (Grk1+) retinas evoked by 500-nm flashes of 20 ms duration. Flashes of incremental intensities, in 0.5 log steps, were delivered at time 0. The dimmest flashes delivered 27 photons μm-2. (C) Averaged normalized rod dim flash responses for WT (black; n = 8) and Grk1+ (gray; n = 6) retinas.
Table 1.
Parameters of Transretinal ERG Recordings
| WT Retina | Grk1+ Retina | |
|---|---|---|
| Sensitivity, photons μm−2 | 70 ± 7 (8) | 72 ± 8 (6) |
| Rmax, μV | 97 ± 13 (8) | 115 ± 13 (6) |
| Time to peak, ms | 184 ± 2 (8) | 166 ± 6 (6)* |
| Integration time, ms | 370 ± 18 (8) | 259 ± 13 (6)† |
Values given as mean ± SEM (n).
Significantly different at 5% level.
Significantly different at 1% level.
The Overexpression of Rhodopsin Kinase Has No Effect on Light Sensitivity in Rods
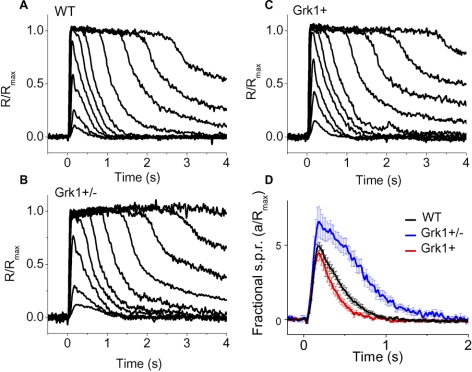
Even though transretinal recordings provide a reliable and convenient measure of photoreceptor sensitivity,20 residual response components from second-order neurons and Müller cells make a careful analysis of response kinetics difficult. Thus, to evaluate the effect of varying GRK1 levels on sensitivity and response kinetics, we performed suction electrode recordings from individual mouse rods expressing different levels of the GRK. The response waveforms from WT rods (Fig. 3A) were comparable with those from rods with GRK1 deficit (Fig. 3B) or excess (Fig. 3C). Both Grk1+ and Grk1+/− rods produced saturating responses with amplitudes comparable with those in WT rods (Table 2). Thus, altering the kinase expression level did not affect the number of open cGMP-gated channels in darkness or the basal activity of the phototransduction cascade consistent with our transretinal recordings (Fig. 2) and with the absence of degeneration in the transgenic retinas.12 Comparison of the half-saturating flash intensity (Io), derived from the intensity-response function for each rod, showed no overexpression effect on the light sensitivity (Table 2). Consistent with this, the estimated amplitudes of their single-photon responses remained unaffected in the GRK1-overexpressing rods (Fig. 3D). In contrast, the Grk1+/− rods showed a slight but statistically significant (P < 0.0001) decrease in Io (Table 1) indicating an increase in sensitivity. The corresponding fractional single photon response amplitude (a/Rmax) was also increased in Grk1+/− compared with WT rods (Fig. 3D). This result is consistent with the observations of Doan et al.11 and of Chen et al.19
Figure 3.
Typical flash response families from (A) WT, (B) GRK1 overexpressing (Grk1+), and (C) Grk1+/− rods evoked by 500-nm flashes of 20 ms duration. Flashes of incremental intensities, in 0.5 log steps, were delivered at time 0. The dimmest flashes delivered 3.9 photons μm-2. (D) Mean fractional single photon responses (s.p.r.), a/Rmax, for WT (n = 36; black line), Grk1+ (n = 21; red line), and Grk1+/− (n = 15; blue line) rods. Single photon responses were estimated by dividing the dim flash response by the number of photoisomerizations estimated using a rod collecting area of 0.5 μm2.18 Error bars indicate SEM.
Table 2.
Response Parameter of Single-Cell Recordings
| WT | Grk1+/− | Grk1+ | Grk1+b | |
|---|---|---|---|---|
| Dark current, pA | 14.9 ± 0.6 (36) | 13.7 ± 0.9 (16) | 13.8 ± 0.6 (21) | 14.9 ± 0.6 (24) |
| Sensitivity Io, photons μm−2 | 27.5 ± 2.0 (36)* | 17.0 ± 1.2 (15)† | 27.3 ± 2.0 (21)* | 31.4 ± 2.8 (24)‡ |
| Time to peak, ms | 176 ± 4 (36) | 187 ± 5 (15) | 180 ± 6 (21) | 175 ± 3 (24) |
| Integration time, ms | 356 ± 21 (36)‡ | 570 ± 50 (15)§ | 254 ± 15 (21)‡§ | 338 ± 22 (24)‡ |
| Recovery time constant, τrec, ms | 223 ± 9 (36)‡ | 343 ± 38 (15)§ | 182 ± 16 (21)†‡ | 211 ± 16 (24)‡ |
| Dominant time constant of recovery, τD, ms | 271 ± 13 (26) | 271 ± 28 (11) | 274 ± 17 (14) | 251 ± 11 (16) |
Values given as mean ± SEM (n). Recovery time constant (τrec) was determined by fitting a single exponential function to the second half of the shut off of the dim flash response. Dominant recovery time constant of recovery (τD) was determined by the slope of Pepperberg plot. Statistical analysis was carried out by one-way ANOVA with the post hoc Tukey Honestly Significant Difference test.
Significantly different from Grk1+/− at 5% level.
Significantly different from WT at 5% level.
Significantly different from Grk1+/− at 1% level.
Significantly different from WT at 1% level.
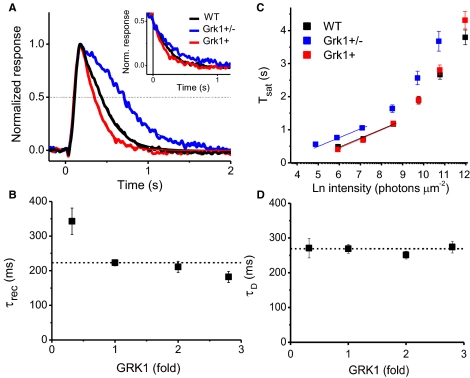
The Expression Level of Rhodopsin Kinase Modulates the Response Shut Off Kinetics for Dim Flashes but Not for Saturating Flashes
It has been previously suggested that the overexpression of GRK1 in mouse rods does not affect the kinetics of their light responses.9 However, examination of the transretinal (Fig. 2C) and single-photon (Fig. 3D) responses of our Grk1+ rods revealed a surprising acceleration of response termination compared with WT rods. The effect of rhodopsin kinase expression level on the kinetics of response termination of mouse rods could be better appreciated from their normalized dim flash responses (Fig. 4A). The threefold reduction in rhodopsin kinase on the deletion of one copy of Grk1 slowed down the response shutoff, as previously shown.11 On the other hand, contrary to the report by Krispel et al.,9 the threefold overexpression of rhodopsin kinase in the Grk1+ mice resulted in notable acceleration of the response shut off. Accordingly, the integration time was also significantly higher in Grk1+/− rods (P < 0.005) and lower in Grk1+ rods (P < 0.0001) compared with WT controls (Table 2). The recovery time constant (τrec), believed to reflect the rate-limiting step of the rod phototransduction inactivation, was also affected by the expression level of rhodopsin kinase. It was significantly increased in Grk1+/− rods (P < 0.0005) and decreased in Grk1+ rods (P < 0.05) (Table 2, and Fig. 4B). To ensure that our measurements reflect the recovery time constant and are not affected by reactions early on in the response shut off, we used only the second half of the response shut off for the single exponential fit determining τrec (Fig 4A, inset). Together, these results demonstrate that, unexpectedly, the dim flash response shut off accelerates with increasing expression of GRK1 leading to a reduction in the recovery time constant. Grk1+b rods expressing rhodopsin kinase at twice the WT level also showed a trend of accelerated response shut off, though the difference with WT rods did not reach statistical significance (Table 1, and Fig. 4B). Thus, the effect of GRK1 overexpression appears to be dose-dependent so that a clear physiological phenotype becomes detectable only when GRK levels become sufficiently higher than in WT rods.
Figure 4.
(A) Normalized dim flash responses of WT (black), Grk1+ (red), and Grk1+/− (blue) rods. The recovery time constant for each cell was estimated by fitting the second half of the recovery phase of dim flash responses with a single exponential function (inset). The recovery phases of dim flash response for each genotype aligned at the point of the half-peak amplitude. (B) Plot of the recovery time constant, τrec, as a function of GRK1 expression, scaled to WT levels. The recovery time constant was shortened with increasing GRK1 expression. (C) Time in saturation of responses against the flash intensities for WT (black), Grk1+ (red), and Grk1+/− (blue) rods. The dominant time constant of recovery was estimated from the slopes of fitting line for the first phase. (D) Plot of the dominant time constant, τD, as a function of GRK1 expression, scaled to WT levels. The dominant time constant was unaffected by GRK1 expression levels.
In addition to τrec, we measured a second kinetic parameter, the dominant time constant (τD) used to describe the inactivation of the phototransduction cascade. Whereas τrec is derived from dim flashes responses not exceeding 30% of the saturating response of the rod, τD is derived from saturating responses. In WT rods, these two parameters have similar values and are believed to reflect the same reaction, namely the rate-limiting step of the phototransduction shut off. Accordingly, next we examined the effect of rhodopsin kinase expression level on τD. The dominant time constant of recovery was calculated from the slope of the duration of signaling at >80% saturation (Tsat) plotted against test flash intensity (Fig. 4C). We found that the expression level of GRK1 does not modulate τD (Fig. 4D). Thus, whereas the recovery time constant demonstrated a clear dependency on the expression level of rhodopsin kinase, the dominant time constant was not affected by its expression levels. Unlike our result on τrec, the lack of effect on GRK1 expression on τD is consistent with the findings of Krispel et al.9
The Expression Level of Rhodopsin Kinase Modulates the Rod Adaptation to Background Light
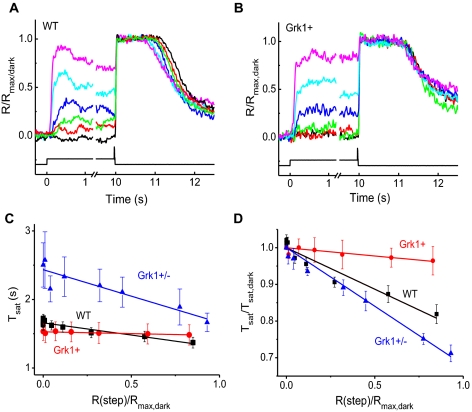
The activity of rhodopsin kinase in rods is modulated by the calcium-binding protein recoverin.21,22 We investigated how the recoverin to rhodopsin kinase ratio modulates rhodopsin shut off and background adaptation. The effect of rhodopsin shut off during background adaptation can be studied using the time of saturation, Tsat, of bright flash responses. As a saturating flash always reduces intracellular calcium to its lowest level, a decrease in Tsat in background light would reflect a reduction in the light-evoked phosphodiesterase activity due to shortened lifetime of photoactivated rhodopsin.23 Thus, we determined how background light affects the time of saturation of bright flash responses in Grk1+/−, WT, and Grk1+ mouse rods (Fig. 5).
Figure 5.
The effect of background light on the shut off of saturating responses was tested by the step-flash protocol, where steady background lights of various intensities were delivered to lower the rod's calcium, followed by a saturating test flash of fixed intensity. Typical response families of (A) WT and (B) Grk1+ rods. Background light shortened the time in saturation robustly for WT rods, but had little to no effect in Grk1+ rods. (C) Duration in saturation (Tsat) of the test flash as a function of step light amplitude for WT (black; n =10), Grk1+ (red; n = 7), and Grk1+/− (blue; n = 6) rods. (D) Plot of Tsat normalized to its dark-adapted value (Tsat,dark) as a function of step light amplitude for WT (black), Grk1+ (red), and Grk1+/− (blue).
As expected,22 in WT rods, exposure to background light of increasing intensity resulted in progressively faster response shut off with a corresponding decline in Tsat consistent with phosphodiesterase (PDE) adaptation mediated by recoverin (Fig. 5A). The value of Tsat declined linearly with the increase in the amplitude of the steady response produced by the step of light (Fig. 5C). Reducing the expression level of rhodopsin kinase to 0.3-fold that of WT rods in Grk1+/− rods or in effect raising the recoverin:GRK1 ratio by 3 folds led to a steeper Tsat decline consistent with a stronger PDE adaptation and kinase modulation by recoverin (Figs. 5C and 5D). Conversely, a threefold overexpression of rhodopsin kinase in Grk1+ rods, or in effect reducing the recoverin:GRK1 ratio by 3 folds, essentially blocked PDE adaptation and rendered Tsat insensitive to modulation by background light (Fig. 5B). The lack of PDE adaptation in Grk1+ rods was also apparent from the independence of Tsat from the amplitude of the background response (Figs. 5C and 5D). Together, these results indicate that the ratio of rhodopsin kinase to its modulator recoverin is critical for the proper adaptation of rods and the acceleration of their response shutoff in background light.
Discussion
We investigated whether the expression level of rhodopsin kinase modulates the phototransduction amplification and the kinetics of the light response in mouse rods. This is one of the very few studies known to the authors in which the impact of varying expression levels of individual signaling proteins have been determined. Previous studies have focused on rhodopsin,24,25 transducin,26 RGS9,9 and arrestin.11,27,28
In our study, we systematically varied the expression level of GRK1 at 30% (Grk1+/−), 100% (WT), 200% (Grk1+b), and 300% (Grk1+) of the WT levels in mice and characterized the function of their rods. Single-cell suction recordings revealed that increasing the expression level of rhodopsin kinase accelerates the shut off of the rod dim flash response. Thus, the lifetime of photoactivated rhodopsin must be sufficiently long to affect the inactivation of the phototransduction cascade and the shutoff of dim flash responses in mammalian rods. In addition, we found that the level of rhodopsin kinase affects the kinetics of saturated response shut off during background adaptation. Thus, the modulation of photoactivated rhodopsin lifetime is important for rod adaptation to background light.
GRK1 Expression and Rod Sensitivity
We found that sensitivity was only subtly impacted by the expression level of GRK1. A threefold overexpression of Grk1+ in rods had no significant effect on their sensitivity or single photon response amplitude (Tables 1 and 2, and Fig. 3D). This lack of overexpression effect on rod sensitivity indicates that WT rods express a sufficiently high level of GRK1 to allow efficient inactivation of rhodopsin under dim light conditions. In contrast, a reduction of rhodopsin kinase expression in Grk1+/− rods to 30% of its WT level did increase the sensitivity of mouse rods (Tables 1 and 2). Accordingly, their single photon responses were also larger in amplitude (Fig. 3D). Thus, reducing rhodopsin kinase expression threefold hindered the inactivation of rhodopsin enough to subtly but significantly affect the amplitude of the dim-flash response. A similar result was recently reported by Doan et al.,11 as well as by Chen et al.,29 who found only a small increase in rod sensitivity even when the expression of rhodopsin kinase was reduced to 15% of the WT level. Notably, even the complete deletion of rhodopsin kinase in the Grk1−/− rods results in only a two-fold increase in the dim flash response amplitude.19 Whole-retina recordings have also demonstrated the lack of significant increase of sensitivity in mouse rods lacking GRK1.30 Thus, it appears that the expression level of rhodopsin kinase has only a minor effect on the amplitude of the response. As the peak of the response reflects the balance of cGMP hydrolysis by PDE and its synthesis by guanylyl cyclase, its amplitude is controlled by the strong activation of cGMP synthesis induced by the reduction in intracellular calcium. Induced during the light response, this negative feedback produces a substantially larger 5- to 6-fold change in the response amplitude and a more dramatic change in its shut off kinetics than the deletion of rhodopsin kinase.31,32
GRK1 Expression and the Shut Off Kinetics of Rod Dim Flash Responses
The recovery of the dark current after light stimulation requires inactivation of all phototransduction intermediates, including photoactivated rhodopsin, R*, and the active form of transducin bound to PDE, Tα/PDE. The relative rates of inactivation of these two intermediates and which one represents the rate-limiting step for response shut off has been a subject of debate. One recent study examined this question using mouse rods overexpressing either RGS9 or GRK1 to speed up the inactivation of Tα/PDE or R*, respectively.9 This study clearly demonstrated that acceleration of Tα/PDE inactivation produces a dramatic reduction in both τrec and τD and concluded that the shut off of the flash response in both dim light (as measured by τrec) and in bright light (as measured by τD) is rate-limited by the inactivation of the Tα/PDE complex. Notably, the authors also stated that overexpression of GRK1 had no effect on the rod dim flash or saturated response shut off. The conclusion drawn from these results was that the lifetime of R* is significantly shorter than that of Tα/PDE so that acceleration of R* shutoff would not affect the overall response kinetics. Indeed, a more recent study27 suggested that the time constant of R* inactivation is only ∼40 ms compared with ∼250 ms for the inactivation of Tα/PDE.9
However, in contrast to the results by Krispel et al.,9 our results clearly demonstrate that the expression level of rhodopsin kinase does modulate the shut off kinetics of dim flash response. We find that reducing the level of rhodopsin kinase threefold in Grk1+/− rods results in a substantial slowing of the dim flash response termination and a significant increase in the recovery time constant. More importantly, increasing kinase expression level threefold in Grk1+ rods accelerates their dim flash response termination and decreases significantly the recovery time constant (Table 2, and Figs. 4A and 4B). The disparity could originate in our use of the full-length BAC rhodospin kinase promoter which produced uniform GRK1 expression across photoreceptors (Fig. 1) as opposed to the opsin promoter used by Krispel et al.9 which may have a less uniform activity profile.33,34 Alternatively, the discrepancy between our results could be due to species differences as we used mouse GRK1 in our study in contrast to Krispel et al. who used bovine GRK1.
Though it is possible that rhodopsin kinase somehow directly modulates the inactivation of Tα/PDE, the most likely interpretation of our results is that the overexpression of rhodopsin kinase speeds up the inactivation of rhodopsin by phosphorylation to result in faster response shut off. Thus, while rhodopsin inactivation is clearly not the rate-limiting reaction for phototransduction inactivation, its rate does appear to be close enough to the one of Tα/PDE inactivation to affect the overall response shut off kinetics. If Tα/PDE decay dominates the response shut off after R* inactivation, then increasing or decreasing the lifetime of R* by varying the expression level of rhodopsin kinase might shift the shut off to the right or to the left, respectively, thus affecting the overall kinetics of the response.
These results are consistent with recent findings by Doan et al.11 who demonstrated that modulation of the rate of rhodopsin inactivation by arrestin can also affect the response shut off and estimated the time constant for R* inactivation at ∼150 ms. Notably, our flash responses are comparable to those reported by Gross and Burns,27 both recorded in Locke's solution, and are substantially faster and smaller than those reported by Doan et al.11 who used Ames perfusion solution.
Finally, our results clearly demonstrate that whereas τrec is modulated by the expression level of rhodopsin kinase, τD is not. Thus, whereas in WT rods these two parameters have similar values,35 the two kinetic parameters can be uncoupled and modulated independently of each other in mouse rods so that the expression level of GRK1 modulates τrec but has no effect on τD. As recently argued by Gross and Burns,27 the measurement of τrec might not allow sufficient time for R* to be turned off. In contrast, as the measurement of τD is done a relatively long time after the test flash, its value would reflect only the slowest inactivation step of the phototransduction cascade. Thus, it appears that the lifetime of photoactivated rhodopsin is long enough to modulate τrec but not τD.
GRK Expression and Background Adaptation
The activity of rhodopsin kinase in rods is modulated by recoverin.21,22 In darkness, when intracellular calcium is high, recoverin inhibits the phosphorylation of photoactivated rhodopsin by rhodopsin kinase, thus prolonging its activity. The decline in calcium when the photoreceptor is exposed to light would relieve rhodopsin kinase of its inhibition by recoverin and speed up rhodopsin inactivation. This represents one possible mechanism by which the modulation of the rod phototransduction cascade could occur during background light adaptation. The recoverin content in mouse rods is believed to exceed that of rhodopsin kinase by an order of magnitude.22 Altering this ratio by either increasing or decreasing the level of rhodopsin kinase in rods could potentially reduce or enhance the effectiveness with which recoverin inhibits its activity. Indeed, our results demonstrate that reducing the expression level of GRK1 threefold enhances the modulation of τD in background light (Fig. 5C). Conversely, the threefold overexpression of GRK1 essentially abolishes the modulation of τD by background light (Fig. 5C). Interestingly, this lack of PDE adaptation in rhodopsin kinase overexpressing rods is reminiscent of the case in recoverin null mice.22 Thus, weakening the calcium modulation of rhodopsin kinase by either deleting recoverin or by increasing the ratio of rhodopsin kinase to recoverin in Grk1+ rods has a similar effect on PDE adaptation. Together, our results indicate that the inhibition of rhodopsin inactivation by recoverin modulates the lifetime of R* and that this modulation is important for the ability of mouse rods to adapt to background light. As recent studies have shown the importance of visual pigment phosphorylation in modulating the pathogenesis of retinal disease in animal models,12,30,36 control of visual pigment phosphorylation could also be an important parameter in how photoreceptors respond and adapt to environments of varying light intensity. Finally, the GRK1 transgenic mice will be a valuable resource for future studies of the role of GRK in response shut off in cones.
Acknowledgments
The authors thank Cheryl Craft and Bruce M. Brown for help with immunoblot analysis of the GRK1 overexpressing mice and for critical reading of the manuscript; Kenneth Gross for advice on generation of BAC transgenic mice; and Mary Kay Ellsworth and the Transgenic Facility at Roswell Park Cancer Institute for their generation and excellent care of the animals.
Footnotes
Supported by a challenge grant to the Department of Ophthalmology at State University of New York at Buffalo, a Career Development Award (VJK) from Research to Prevent Blindness, and NIH Grants EY019312 (VJK), EY13600 (SCK), EY002687 to the Department of Ophthalmology and Visual Sciences at Washington University, and EY016662 to the Department of Ophthalmology, State University of New York at Buffalo.
Disclosure: K. Sakurai, None; J.E. Young, None; V.J. Kefalov, None; S.C. Khani, None
References
- 1. Baylor DA. Photoreceptor signals and vision. Proctor lecture. Invest Ophthalmol Vis Sci. 1987;28:34–49 [PubMed] [Google Scholar]
- 2. Lamb TD, Pugh EN., Jr Dark adaptation and the retinoid cycle of vision. Prog Retin Eye Res. 2004;23:307–380 [DOI] [PubMed] [Google Scholar]
- 3. Hurley JB, Spencer M, Niemi GA. Rhodopsin phosphorylation and its role in photoreceptor function. Vision Res. 1998;38:1341–1352 [DOI] [PubMed] [Google Scholar]
- 4. Maeda T, Imanishi Y, Palczewski K. Rhodopsin phosphorylation: 30 years later. Prog Retin Eye Res. 2003;22:417–434 [DOI] [PubMed] [Google Scholar]
- 5. Chen CK. Recoverin and rhodopsin kinase. Adv Exp Med Biol. 2002;514:101–107 [DOI] [PubMed] [Google Scholar]
- 6. Gurevich EV, Gurevich VV. Arrestins: ubiquitous regulators of cellular signaling pathways. Genome Biol. 2006;7:236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Wensel TG. Signal transducing membrane complexes of photoreceptor outer segments. Vision Res. 2008;48:2052–2061 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Pepperberg DR, Cornwall MC, Kahlert M, et al. Light-dependent delay in the falling phase of the retinal rod photoresponse. Vis Neurosci. 1992;8:9–18 [DOI] [PubMed] [Google Scholar]
- 9. Krispel CM, Chen D, Melling N, et al. RGS expression rate-limits recovery of rod photoresponses. Neuron. 2006;51:409–416 [DOI] [PubMed] [Google Scholar]
- 10. Burns ME, Pugh EN., Jr RGS9 concentration matters in rod phototransduction. Biophys J. 2009;97:1538–1547 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Doan T, Azevedo AW, Hurley JB, Rieke F. Arrestin competition influences the kinetics and variability of the single-photon responses of mammalian rod photoreceptors. J Neurosci. 2009;29:11867–11879 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Whitcomb T, Sakurai K, Brown BM, et al. Effect of g protein-coupled receptor kinase 1 (Grk1) overexpression on rod photoreceptor cell viability. Invest Ophthalmol Vis Sci. 2010;51:1728–1737 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Young JE, Gross KW, Khani SC. Conserved structure and spatiotemporal function of the compact rhodopsin kinase (GRK1) enhancer/promoter. Mol Vis. 2005;11:1041–1051 [PubMed] [Google Scholar]
- 14. Young JE, Vogt T, Gross KW, Khani SC. A short, highly active photoreceptor-specific enhancer/promoter region upstream of the human rhodopsin kinase gene. Invest Ophthalmol Vis Sci. 2003;44:4076–4085 [DOI] [PubMed] [Google Scholar]
- 15. Sillman AJ, Ito H, Tomita T. Studies on the mass receptor potential of the isolated frog retina. I. General properties of the response. Vision Res. 1969;9:1435–1442 [DOI] [PubMed] [Google Scholar]
- 16. Bolnick DA, Walter AE, Sillman AJ. Barium suppresses slow PIII in perfused bullfrog retina. Vision Res. 1979;19:1117–1119 [DOI] [PubMed] [Google Scholar]
- 17. Nymark S, Heikkinen H, Haldin C, Donner K, Koskelainen A. Light responses and light adaptation in rat retinal rods at different temperatures. J Physiol. 2005;567:923–938 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Nikonov SS, Kholodenko R, Lem J, Pugh EN., Jr Physiological features of the S- and M-cone photoreceptors of wild-type mice from single-cell recordings. J Gen Physiol. 2006;127:359–374 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Chen CK, Burns ME, Spencer M, et al. Abnormal photoresponses and light-induced apoptosis in rods lacking rhodopsin kinase. Proc Natl Acad Sci U S A. 1999;96:3718–3722 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Wang JS, Kefalov VJ. An alternative pathway mediates the mouse and human cone visual cycle. Curr Biol. 2009;19:1665–1669 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Kawamura S. Rhodopsin phosphorylation as a mechanism of cyclic GMP phosphodiesterase regulation by S-modulin. Nature. 1993;362:855–857 [DOI] [PubMed] [Google Scholar]
- 22. Makino CL, Dodd RL, Chen J, et al. Recoverin regulates light-dependent phosphodiesterase activity in retinal rods. J Gen Physiol. 2004;123:729–741 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Fain GL, Lamb TD, Matthews HR, Murphy RL. Cytoplasmic calcium as the messenger for light adaptation in salamander rods. J Physiol. 1989;416:215–243 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Calvert PD, Govardovskii VI, Krasnoperova N, Anderson RE, Lem J, Makino CL. Membrane protein diffusion sets the speed of rod phototransduction. Nature. 2001;411:90–94 [DOI] [PubMed] [Google Scholar]
- 25. Wen XH, Shen L, Brush RS, et al. Overexpression of rhodopsin alters the structure and photoresponse of rod photoreceptors. Biophys J. 2009;96:939–950 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Lobanova ES, Finkelstein S, Herrmann R, et al. Transducin gamma-subunit sets expression levels of alpha- and beta-subunits and is crucial for rod viability. J Neurosci. 2008;28:3510–3520 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Gross OP, Burns ME. Control of rhodopsin's active lifetime by arrestin-1 expression in mammalian rods. J Neurosci. 2010;30:3450–3457 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Song X, Vishnivetskiy SA, Seo J, Chen J, Gurevich EV, Gurevich VV. Arrestin-1 expression level in rods: balancing functional performance and photoreceptor health. Neuroscience. 2011;174:37–49 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Chen CK, Woodruff ML, Chen FS, Chen D, Fain GL. Background light produces a recoverin-dependent modulation of activated-rhodopsin lifetime in mouse rods. J Neurosci. 2010;30:1213–1220 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Fan J, Sakurai K, Chen CK, et al. Deletion of GRK1 causes retina degeneration through a transducin-independent mechanism. J Neurosci. 2010;30:2496–2503 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Mendez A, Burns ME, Sokal I, et al. Role of guanylate cyclase-activating proteins (GCAPs) in setting the flash sensitivity of rod photoreceptors. Proc Natl Acad Sci U S A. 2001;98:9948–9953 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Howes KA, Pennesi ME, Sokal I, et al. GCAP1 rescues rod photoreceptor response in GCAP1/GCAP2 knockout mice. EMBO J. 2002;21:1545–1554 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Zack DJ, Bennett J, Wang Y, et al. Unusual topography of bovine rhodopsin promoter-lacZ fusion gene expression in transgenic mouse retinas. Neuron. 1991;6:187–199 [DOI] [PubMed] [Google Scholar]
- 34. Khani SC, Pawlyk BS, Bulgakov OV, et al. AAV-Mediated Expression Targeting of Rod and Cone Photoreceptors with a Human Rhodopsin Kinase Promoter. Invest Ophthalmol Vis Sci. 2007;48:3954–3961 [DOI] [PubMed] [Google Scholar]
- 35. Pugh EN, Jr, Nikonov S, Lamb TD. Molecular mechanisms of vertebrate photoreceptor light adaptation. Curr Opin Neurobiol. 1999;9:410–418 [DOI] [PubMed] [Google Scholar]
- 36. Yetemian RM, Brown BM, Craft CM. Neovascularization, enhanced inflammatory response, and age-related cone dystrophy in the Nrl-/-Grk1-/- mouse retina. Invest Ophthalmol Vis Sci. 2010;51:6196–6206 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Zhao X, Huang J, Khani SC, Palczewski K. Molecular forms of human rhodopsin kinase (GRK1). J Biol Chem. 1998;273:5124–5131 [DOI] [PubMed] [Google Scholar]
- 38. Mears AJ, Kondo M, Swain PK, et al. Nrl is required for rod photoreceptor development. Nat Genet. 2001;29:447–452 [DOI] [PubMed] [Google Scholar]