Abstract
The wogonin-containing herb Scutellaria baicalensis has successfully been used for curing various diseases in traditional Chinese medicine. Wogonin has been shown to induce apoptosis in different cancer cells and to suppress growth of human cancer xenografts in vivo. However, its direct targets remain unknown. In this study, we demonstrate for the first time that wogonin and structurally related natural flavones, for example, apigenin, chrysin and luteolin, are inhibitors of cyclin-dependent kinase 9 (CDK9) and block phosphorylation of the carboxy-terminal domain of RNA polymerase II at Ser2. This effect leads to reduced RNA synthesis and subsequently rapid downregulation of the short-lived anti-apoptotic protein myeloid cell leukemia 1 (Mcl-1) resulting in apoptosis induction in cancer cells. We show that genetic inhibition of Mcl-1 or CDK9 expression by siRNA is sufficient to mimic flavone-induced apoptosis. Pull-down and in silico docking studies demonstrate that wogonin directly binds to CDK9, presumably to the ATP-binding pocket. In contrast, wogonin does not inhibit CDK2, CDK4 and CDK6 at doses that inhibit CDK9 activity. Furthermore, we show that wogonin preferentially inhibits CDK9 in malignant compared with normal lymphocytes. Thus, our study reveals a new mechanism of anti-cancer action of natural flavones and supports CDK9 as a therapeutic target in oncology.
Keywords: anti-cancer drug, apoptosis, CDK9, Mcl-1, transcription
Flavonoids are naturally occurring polyphenolic compounds present in fruits, vegetables and some medicinal plants. A number of flavones and flavonols have been found to possess anti-tumor activities. We and others have shown that wogonin, one of the active flavones of the most popular Chinese herbal remedy Huang-Qin (Scutellaria baicalensis Georgi), induces apoptosis in a wide spectrum of human tumor cells in vitro and inhibits tumor growth in vivo in different mouse tumor models.1, 2, 3, 4, 5, 6 In addition, Scutellaria extracts were successfully tested in patients with advanced breast cancer in early clinical trials.7, 8 Importantly, at doses lethal to tumor cells, wogonin showed no or little toxicity for normal cells and had also no obvious toxicity in animals.2, 3, 4, 5, 6
Although many studies have demonstrated that wogonin preferentially kills tumor cells, little is known about the molecular mechanisms. We have previously shown that wogonin is a potent anti-oxidant capable to scavenge •O2− and, thereby, shifts the cellular redox potential to the more reduced state H2O2.6 H2O2 in turn serves as a signaling molecule to activate phospholipase Cγ1 (PLCγ1) and triggers a PLCγ1-regulated and Ca2+-dependent apoptosis.3 Although the crucial role of Ca2+ in wogonin-induced apoptosis was largely confirmed, we noticed that inhibiting Ca2+ transport did not completely inhibit apoptosis induction.3 Thus, other unknown mechanisms may be involved in wogonin-mediated apoptosis.
Targeting apoptotic pathways is one of the therapeutic strategies against cancer.9, 10 In the intrinsic apoptosis pathway, death and life of cells are largely controlled by pro-apoptotic, for example, Bax and Bak, and anti-apoptotic proteins, for example, Bcl-2, Bcl-xL, XIAP and myeloid cell leukemia 1 (Mcl-1).9 Strong evidence has linked the anti-apoptotic Bcl-2 family proteins to drug resistance and poor treatment outcome in a variety of tumor types.10 Among the anti-apoptotic proteins, Mcl-1 has been considered to be the most relevant therapeutic target in multiple types of cancer because it differs from other members of the Bcl-2 family by a short half-life.11 Inhibition of Mcl-1 expression alone via RNA interference has been shown to be sufficient to promote mitochondrial membrane depolarization and apoptosis in leukemic cells.12
In this study, we show that wogonin and structurally related flavones, for example, apigenin, chrysin and luteolin, are inhibitors of cyclin-dependent kinase (CDK) 9. Unlike other CDKs, which primarily control cell cycle progression, CDK7 and CDK9 have a major role in regulation of transcription. CDK7 is a component of the transcription factor TFIIH, which phosphorylates Ser5 residues in the heptad repeats of the carboxy-terminal domain (CTD) of RNA polymerase II (RNAPII) to facilitate transcription initiation.13, 14 CDK9, the core component of the positive transcription elongation factor b, phosphorylates Ser2 residues in the CTD of RNAPII, which is required for transcript elongation.13, 14, 15 We show that inhibition of CDK9 activity by wogonin, apigenin, chrysin and luteolin prevents phosphorylation of RNAPII and thereby inhibits transcription. This event leads to the downregulation of the short-lived anti-apoptotic protein Mcl-1 and, consequently, to the induction of apoptosis. We also found that wogonin, at a concentration that inhibits CDK9, does not inhibit activities of the cell cycle-regulating kinases CDK2, CDK4 and CDK6. Furthermore, we demonstrate that wogonin preferentially inhibits CDK9 in malignant compared with normal lymphocytes.
Results
Wogonin downregulates Mcl-1 expression in malignant cells
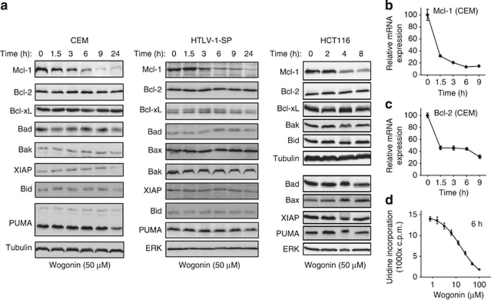
To investigate the molecular mechanisms by which wogonin induces apoptosis in cancer cells, we systematically analyzed expression levels of pro- and anti-apoptotic proteins after wogonin treatment in three tumor cell lines: the human colorectal carcinoma cell line HCT116, the human leukemic T-cell line CEM and the adult T-cell leukemic cell line SP derived from a human T-cell leukemia/lymphoma virus 1 (HTLV-1)-infected patient. Consistent with the previous study,3 wogonin treatment resulted in apoptotic cell death in HCT116, CEM and SP cells in a dose- and time-dependent manner (Supplementary Figure S1). Western blot analysis of the pro- and anti-apoptotic proteins revealed that only the Mcl-1 protein expression levels were rapidly downregulated on wogonin treatment (Figure 1a). The 3 h wogonin treatment already resulted in >50% reduction in Mcl-1 protein levels. In contrast, other pro- and anti-apoptotic proteins such as Bcl-2, Bcl-xL, Bad, Bak and Bax remained unaffected until 24 h of treatment (Figure 1a). A reduction in XIAP and PUMA protein expression was also detected in wogonin-treated cells but only after 24 h of treatment (Figure 1a). However, apoptosis was initiated by wogonin already at earlier time points (Supplementary Figure S1A).
Figure 1.
Wogonin inhibits transcription and downregulates expression of the anti-apoptotic protein Mcl-1 in malignant cells. (a) Wogonin downregulates Mcl-1 protein expression in malignant cells. CEM, HTLV-1-SP and HCT116 cells were treated with 50 μM wogonin for indicated time periods. The expression levels of the indicated proteins were examined by western blot analysis. Data are representative of two independent experiments. (b and c) Wogonin downregulates Mcl-1 and Bcl-2 mRNA expression in malignant cells. CEM cells were treated with 50 μM wogonin for indicated time periods. Mcl-1 and Bcl-2 mRNA expression levels were examined by real-time PCR. Means±S.D. are shown. Data are representative of three independent experiments performed in triplicates. (d) Wogonin suppresses RNA synthesis in malignant cells. CEM cells were treated with different concentrations of wogonin for 6 h as indicated. RNA synthesis was measured by [3H]-uridine-incorporation. Means±S.D. are shown. Data are representative of two independent experiments performed in triplicates
Wogonin suppresses Mcl-1 expression at the transcriptional level
To investigate the molecular mechanisms by which wogonin suppresses Mcl-1 expression, we first examined the mRNA expression levels of Mcl-1 in CEM and SP cells following wogonin treatment. Real-time PCR revealed that wogonin, at the concentrations that reduced Mcl-1 protein levels, suppressed Mcl-1 mRNA expression in a time-dependent manner (Figure 1b and Supplementary Figure S2A). However, we found that Bcl-2 mRNA expression was also downregulated by wogonin although the Bcl-2 protein level was not affected (Figures 1a and c). Therefore, we asked whether wogonin inhibits transcription. To investigate this question, we carried out a [3H]-uridine-incorporation assay in the absence or presence of wogonin. These experiments showed that wogonin suppressed RNA synthesis in a dose- and time-dependent manner (Figure 1d and Supplementary Figure S2B). Thus, wogonin seems to be an inhibitor of transcription.
Mcl-1 is known to be a target of proteasomal degradation.11 To study whether the downregulation of Mcl-1 protein expression by wogonin also involves a proteasome-dependent mechanism, SP cells were treated with wogonin in the presence or absence of the proteasome inhibitor MG-132. MG-132 could only rescue the already existing Mcl-1 protein but not the loss of Mcl-1 in response to wogonin (Supplementary Figure S3A). To further investigate whether wogonin treatment enhances the rate of Mcl-1 protein turnover, CEM cells were treated with cycloheximide (CHX), a potent inhibitor of translation, or wogonin for different time periods. This experiment showed that wogonin treatment did not accelerate the Mcl-1 turnover rate (Supplementary Figure S3B). These results indicate that the effect of wogonin on Mcl-1 protein expression is not mediated by enhanced degradation.
Wogonin inhibits CDK9 activity
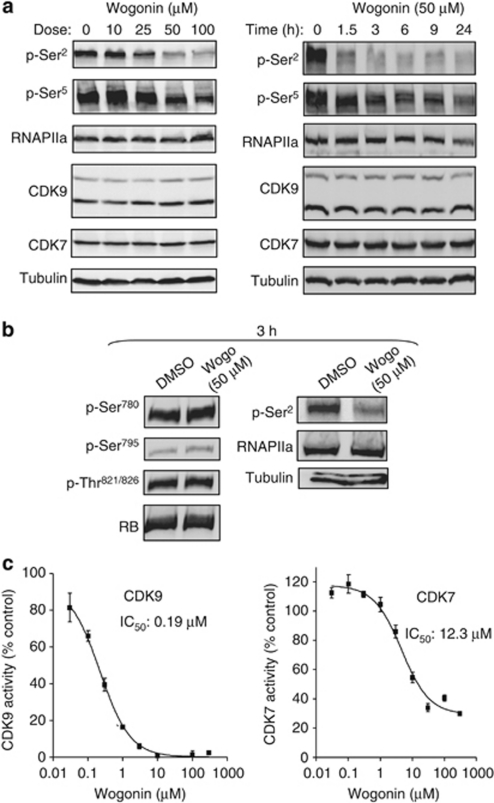
CDK7 and CDK9 are two well-known components of basal transcription factor complexes important for transcription initiation and transcription elongation, respectively.13, 14, 15 Therefore, we asked whether wogonin-mediated transcriptional inhibition involves inactivation of CDK7 and CDK9. We first investigated the effects of wogonin on phosphorylation of the CTD of RNAPII at both Ser2 and Ser5 sites. Western blot analysis of CEM cells treated with different concentrations of wogonin showed that Ser2 phosphorylation was diminished in a dose-dependent manner after 2 h of wogonin treatment (Figure 2a, left panel). In comparison, only weak reductions in the phosphorylation of the Ser5 site were seen (Figure 2a). Kinetic analysis showed that Ser2 phosphorylation was rapidly inhibited by wogonin already within 1.5 h of treatment, whereas Ser5 phosphorylation was reduced with much slower kinetics compared with Ser2 (Figure 2a, right panel). In contrast, at the concentration that inhibits RNAPII phosphorylation, wogonin did not inhibit phosphorylation of retinoblastoma (RB) protein, the target of CDK2, CDK4 and CDK6 (Figure 2b). Also, neither an arrest of the cells in the G1 phase nor in the G2/M phase was seen in wogonin-treated cells (Supplementary Figure S4). The wogonin concentrations required for CDK9 inhibition correlated with the doses for Mcl-1 downregulation and apoptosis induction (Figure 1a and Supplementary Figure S1). Above data demonstrate that wogonin preferentially inhibits CDK9-mediated phosphorylation of RNAPII.
Figure 2.
Wogonin is a naturally occurring inhibitor of CDK7 and CDK9. (a) Wogonin inhibits phosphorylation of the CTD of RNAPII at Ser2 and Ser5 sites. CEM cells were treated with different concentrations of wogonin for 2 h (left panel) or for different time periods with 50 μM wogonin (right panel) and the cells were analyzed for the status of RNAPII phosphorylation by western blot analysis using antibodies specific for phosphorylated CTD of RNAPII Ser2 and Ser5 sites. One representative experiment of three is shown. (b) Wogonin does not inhibit phosphorylation of the retinoblastoma (RB) protein. CEM cells were treated with 50 μM wogonin (Wogo) for 3 h. Cells were lysed and total RB was immunoprecipitated and phosphorylated RB was examined by western blot using phospho-specific antibodies as indicated (left panel). As a control, the same cell lysates were analyzed for the status of phosphorylation of RNAPII at the Ser2 residue (right panel). Data are representative of three independent experiments. (c) Wogonin inhibits CDK7 and CDK9 kinase activity determined by incorporation of [33P]. CDK7/cyclinH/MAT1 or CDK9/cyclinT was incubated with substrate peptide and [33P]-ATP in the presence of different doses of wogonin as indicated. The kinase activity is described as % of [33P]-phosphorylated substrate peptide. Means±S.D. are shown. The half-maximal inhibitory concentrations (IC50) are indicated
To further investigate whether wogonin directly inhibits CDK9 activity, we carried out two different cell-free kinase assays. In the first assay, the effect of wogonin on CDK9 activity was examined by monitoring the levels of [33P]-incorporation into the phosphorylated substrate peptide. The experiment showed that wogonin inhibited CDK9 activity in a dose-dependent manner (Figure 2c). Wogonin also reduced CDK7 activity but with much less efficacy (Figure 2c). These findings could be confirmed in a second assay, in which phosphorylation of a substrate by recombinant CDK7 or CDK9 was detected using phospho-specific antibodies (Supplementary Figure S2C).
To exclude the possibility that wogonin-mediated inhibition of RNAPII phosphorylation and Mcl-1 expression is the result of apoptosis induction, we investigated the effect of wogonin in the presence or absence of the pan-caspase inhibitor zVAD-FMK. Inhibition of apoptosis by zVAD-FMK did not prevent wogonin-mediated inhibition of CDK9 activity and Mcl-1 expression (Supplementary Figure S5). Thus, cells that ultimately do not undergo apoptosis following treatment with wogonin still have suppressed CDK9 activity.
Binding of wogonin to the ATP-binding pocket of CDK9
To better understand the molecular mechanism of action of wogonin, an in silico molecular docking study with wogonin on the crystal structure of human CDK9 was performed. Wogonin was shown to dock into the ATP-binding pocket of CDK9 in a cluster at 98 out of 100 runs with a mean docking energy (ΔG) of −7.65 kcal/mol. The wogonin-binding site was predicted to consist of 16 residues using MultiBind webserver (Figure 3). This docking analysis further supports CDK9 as potential target of wogonin.
Figure 3.
In silico docking analysis of wogonin on the crystal structure of CDK9. (a) Computer docking simulation of the crystal structure of human CDK9 in complex with wogonin. (b) MultiBind webserver analysis predicted wogonin-binding residues in CDK9. The residues involved in the binding of wogonin to CDK9 are indicated by triangles
Inhibition of RNAPII phosphorylation by the natural flavones apigenin, chrysin and luteolin
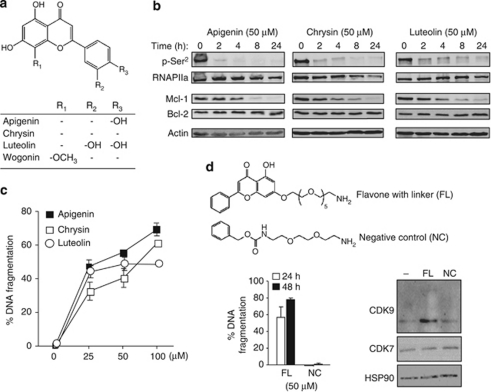
In recent years, several natural flavones have been found to have anti-carcinogenic activities. However, their modes of function as anti-cancer agents are still largely unknown. To investigate whether other flavones besides wogonin also target CDK9, we randomly tested three naturally occurring anti-tumor flavones, namely apigenin, chrysin and luteolin (Figure 4a).16, 17 All flavones tested were shown to inhibit the phosphorylation of the CTD of RNAPII at the Ser2 sites that correlated with the reduction of Mcl-1 protein expression and induction of apoptosis in leukemic CEM cells (Figures 4b and c). These experiments demonstrate that flavones that have chemical structures similar to wogonin are potential inhibitors of CDK9.
Figure 4.
Several natural flavones inhibit CDK9 function. (a) Chemical structures of apigenin, chrysin, luteolin and wogonin. (b) Apigenin, chrysin and luteolin inhibit phosphorylation of RNAPII and downregulate Mcl-1 expression. Leukemic CEM cells were treated with 50 μM of different flavones for indicated time periods. Cell lysates were subjected to western blot analysis using antibodies as indicated. Results are representative of two independent experiments. (c) Apigenin, chrysin and luteolin induce apoptosis in leukemic cells. Leukemic CEM cells were treated with different concentrations of the indicated flavones for 24 h. Apoptotic cell death was determined by DNA fragmentation. Means±S.D. are shown. Results are representative of two independent experiments. (d) Flavones directly interact with CDK9. Upper panel, a flavone backbone coupled to Affi-Gel-10 and the Affi-Gel coupled to the linker only were used to analyze flavone-binding proteins. Lower panel left, leukemic CEM cells were treated with 50 μM of flavone with linker (FL) or negative control (NC) for 24 and 48 h. Apoptotic cell death was determined by DNA fragmentation. Means±S.D. are shown. Lower panel right, western blot analysis of proteins pulled down from the flavone-coupled or the negative control-coupled Affi-Gel beads. HSP90 was used to control for equal unspecific binding. The result is representative of two independent pull-down experiments
To examine whether flavones can directly bind to CDK9 in cancer cells, a pull-down experiment was carried out with flavone-conjugate-coupled Affi-Gel beads (see Materials and Methods) or Affi-Gel beads containing only the linker as a negative control (Figure 4d, upper panel). The flavone coupled to the linker still exerted cytotoxicity for malignant cells as demonstrated by apoptosis induction in CEM cells (Figure 4d, lower panel, left). Western blot analysis of eluted proteins showed that CDK9 was bound to the flavone-Affi-Gel beads but not to the control beads (Figure 4d, lower panel, right). Although wogonin could inhibit CDK7 activity in cell-free in vitro assays, we could not detect CDK7 in this pull-down experiment.
Wogonin inhibits Mcl-1 expression in different types of cancer cells
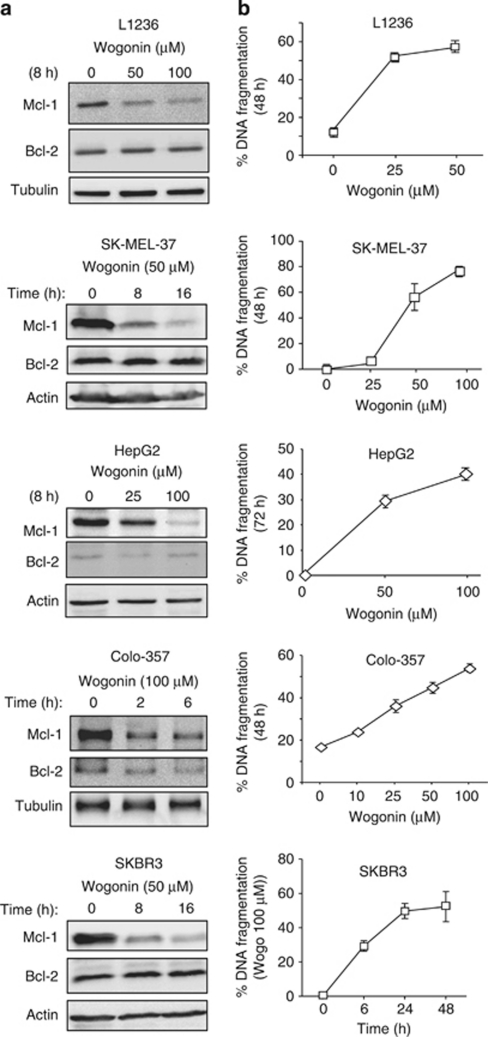
To further study whether wogonin inhibits Mcl-1 expression in other cancer cells, we examined five different types of malignant cell lines including the human Hodgkin's lymphoma cell line L1236, the human melanoma cell line SK-MEL-37, the human hepatocellular carcinoma cell line HepG2, the human pancreatic carcinoma cell line Colo-357 and the human breast cancer cell line SKBR3. For all cell lines tested, wogonin was shown to inhibit Mcl-1 expression (Figure 5a). Inhibition of Mcl-1 expression by wogonin correlated with apoptosis induction in these cells (Figure 5b). Thus, in general wogonin can suppress Mcl-1 expression in tumor cells and inhibition of Mcl-1 expression may account for one of the mechanisms of wogonin-induced cell death in tumor cells.
Figure 5.
Wogonin inhibits Mcl-1 expression and induces apoptosis in different malignant cell lines. (a) Wogonin inhibits Mcl-1 expression in different malignant cell lines. Hodgkin's lymphoma L1236, melanoma SK-MEL-37, hepatocellular carcinoma HepG2, pancreatic carcinoma Colo-357 and breast cancer SKBR3 cells were incubated with different concentrations of wogonin for indicated time periods. The protein expression levels of Mcl-1 and Bcl-2 were examined by western blot. (b) Wogonin induces apoptosis in different malignant cell lines. Different tumor cell lines were treated with different concentrations of wogonin for different time periods as indicated. Apoptotic cells were determined by measuring DNA fragmentation. Means±S.D. are shown. Results are representative of two (western blot) to three (apoptosis) independent experiments
Genetic inhibition of Mcl-1 or CDK9 expression is sufficient to mimic wogonin-induced apoptosis
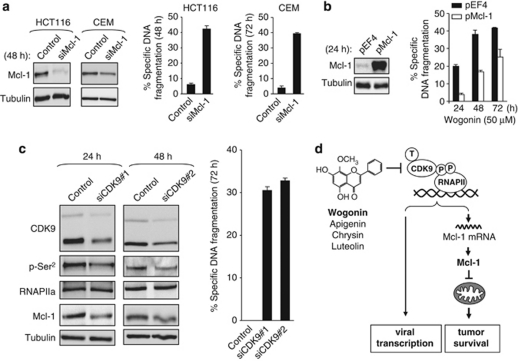
To investigate the role of Mcl-1 in regulation of life and death of malignant cells, we performed a siRNA knockdown experiment using the colon carcinoma cell line HCT116 and the leukemic T-cell line CEM as a model system. Specific knockdown of Mcl-1 expression by RNA interference resulted in induction of apoptosis in both cell lines (Figure 6a). Similar results were also obtained using the B-cell lymphoma cell line Raji (Supplementary Figure S6). To further examine the role of Mcl-1 in wogonin-mediated apoptosis, Mcl-1 was introduced into HCT116 cells by transient transfection of plasmids expressing the Mcl-1 protein. Ectopic expression of the Mcl-1 protein resulted in significant resistance to wogonin-induced apoptosis (Figure 6b). Of note, the Mcl-1 overexpressing tumor cells also started to die by wogonin treatment at later time points compared with control-transfected cells. This is in line with the observation that the overexpressed Mcl-1 protein was also downregulated by wogonin (Supplementary Figure S7).
Figure 6.
Inhibition of Mcl-1 or CDK9 expression is sufficient to mimic wogonin-induced apoptosis. (a) Knockdown of Mcl-1 induces apoptosis. HCT116 and CEM cells were transfected with either siRNA specific for Mcl-1 or scrambled siRNA as described in Materials and Methods. Knockdown efficiency was controlled by western blot at 48 h after transfection. Apoptotic cell death was determined by DNA fragmentation at 48 h and 72 h after transfection for HCT116 and CEM, respectively. Means±S.D. are shown. The result is representative of two independent knockdown experiments. (b) Overexpression of Mcl-1 inhibits wogonin-induced apoptosis. HCT116 cells were transfected with either the Mcl-1 expressing plasmid (pMcl-1) or the parental control plasmid (pEF4) as described in Materials and Methods. The overexpression efficiency was controlled by western blot at 24 h after transfection. Cells were treated with 50 μM wogonin for 24, 48 and 72 h as indicated. Apoptotic cell death was determined by DNA fragmentation. Means±S.D. are shown. The result is representative of two independent experiments. (c) Knockdown of CDK9 is sufficient to induce apoptosis. CEM cells were transfected with either siRNA #1 or #2 specific for CDK9 or control siRNA. Knockdown efficiency was controlled by western blot at 24 or 48 h after transfection. Apoptotic cell death was determined by DNA fragmentation at 72 h after transfection. The result is representative of two independent knockdown experiments. Means±S.D. are shown. (d) Model showing the mechanism of the anti-tumor and anti-viral effect of flavones
To further investigate the role of CDK9 as a drug target, we also examined the effect of CDK9 knockdown on apoptosis induction. Specific knockdown of CDK9 expression in CEM cells by RNA interference resulted in induction of apoptosis to a similar extent as the knockdown of Mcl-1 (Figure 6c). Thus, targeting CDK9 seems to be a promising approach to induce apoptosis in tumor cells. Collectively, our data suggest that suppression of CDK9 activity and downregulation of Mcl-1 is an important mechanism of flavone-induced apoptosis (Figure 6d).
Wogonin acts differentially on CDK9 activity in malignant versus normal T cells
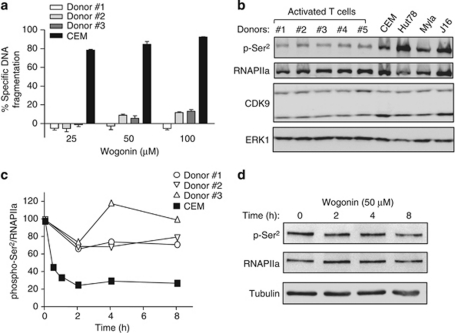
As many studies have shown that wogonin preferentially kills tumor cells,1 we wondered whether wogonin exerts different effects on CDK9 activity in leukemic and normal T lymphocytes. To investigate this question, T cells were isolated from peripheral blood of healthy donors. As proliferating cells are more susceptible to drug treatment, freshly isolated peripheral blood T cells were activated by PHA overnight and further cultured in IL-2-containing medium for 5 days to generate proliferating T cells (named activated T cells). CEM cells and activated T cells were treated with wogonin in parallel. Consistent with previous studies,3 wogonin showed no toxicity for normal peripheral blood T cells at concentrations of 25–100 μM (Figure 7a). Comparison of the status of phosphorylated RNAPII between leukemic and normal T cells revealed that RNAPII was phosphorylated at Ser2 to a greater extent in leukemic cells indicating that CDK9 activity is higher in malignant compared with normal T cells (Figure 7b). Kinetic analysis showed that wogonin only moderately inhibited CDK9 activity (approximately 20%) in normal T cells (Figures 7c and d). In contrast, 70% inhibition of RNAPII phosphorylation was seen in CEM cells in parallel experiments (Figure 7c). Thus, wogonin may preferentially inhibit CDK9 activity in tumor versus normal T cells.
Figure 7.
Comparative studies of the effects of wogonin on CDK9 activity in leukemic and normal T cells. (a) Wogonin induces apoptosis in malignant T cells and not in normal activated T cells. CEM and activated T cells isolated from peripheral blood of three representative healthy donors were treated with different concentrations of wogonin for 48 h. Apoptotic cell death was determined by DNA fragmentation. Means±S.D. are shown. (b) Malignant T cells have higher levels of phosphorylated RNAPII compared with normal activated T cells. Phosphorylation of RNAPII at Ser2 in peripheral blood T cells from five healthy donors and different malignant T cell lines was analyzed using western blot. (c) Wogonin exerts stronger inhibitory effects on CDK9 in malignant than in normal T cells. Peripheral blood T cells from donors in a were treated with 50 μM of wogonin for different time periods as indicated. The effects of wogonin on CDK9 activity were examined by western blot for phosphorylation of RNAPII at Ser2. CEM cells were treated in parallel as a control. Results are presented as amount of Ser2 phosphorylated RNAPII relative to total RNAPIIa. (d) Representative western blot from donor 1 is shown
Discussion
In this study, we demonstrate that CDK9 is a direct target of wogonin. Inhibition of CDK9 activity by wogonin results in reduced RNA synthesis and subsequently rapid downregulation of the short-lived anti-apoptotic protein Mcl-1 leading to apoptosis induction in cancer cells. Furthermore, we also show that other natural flavones such as apigenin, chrysin and luteolin inhibit CDK9 function, which correlates with downregulation of Mcl-1 expression and induction of apoptosis in malignant cells. This new finding provides, at least in part, the molecular mechanisms of the medicinal function of natural flavones.
Wogonin was shown to reduce phosphorylation of the CTD of RNAPII at Ser5 to a lesser extent than Ser2. In a cell-free assay, wogonin also inhibited CDK7 activity but with a strongly reduced efficacy compared with CDK9 (>60-fold higher half-maximal inhibitory concentration (IC50) than for CDK9). These results indicate that wogonin preferentially inhibits CDK9. This indication is further supported by the fact that we could not detect binding of CDK7 to flavones in the pull-down experiment. In addition, wogonin did not inhibit the activity of other members of the CDK family, for example, CDK2/4/6 in tumor cells (Figure 2b).
Targeting apoptotic pathways is one of the therapeutic strategies against cancer.9, 10 Among the anti-apoptotic proteins, Mcl-1 has been found to be upregulated in numerous hematological malignancies and in solid tumors, and its upregulation has been shown to be an important factor in resistance to apoptosis.11, 18 One of the unique features of Mcl-1 compared with other members of the Bcl-2 family is its very short half-life.11 Specific knockdown of Mcl-1 expression by RNA interference has been shown to be sufficient or at least required for the induction of apoptosis in different types of cancer cells.12, 19, 20 In this study, we confirmed by knockdown and overexpression experiments that Mcl-1 is a key survival factor for tumors. Thus, Mcl-1, which is efficiently targeted by wogonin, may be the most relevant therapeutic target in multiple types of cancer.11, 18
Wogonin has been shown to have no or little toxicity for normal cells and had also no obvious toxicity in animals.1, 2, 3, 4, 5, 6 The tumor selectivity of wogonin may involve several mechanisms. First, tumors show an increased metabolism and often a metabolic switch to aerobic glycolysis (Warburg effect) known to influence the redox status.21 Thus, tumors, particularly in advanced stage, produce elevated levels of reactive oxygen species and show an altered redox status.22 This biochemical property of cancer cells can be exploited to achieve therapeutic activity and selectivity. We have previously shown that wogonin is a strong anti-oxidant that differentially regulates the cellular redox status of tumor and normal cells by neutralizing •O2− to the more reduced form H2O2.3, 6 This effect leads to stronger activation of PLCγ in malignant T cells and, consequently, triggers prolonged (or/and stronger) intracellular Ca2+ mobilization and Ca2+-mediated apoptosis. In this study, we show that wogonin differentially inhibits CDK9 activity in malignant versus normal T lymphocytes. This action may account for another mechanism of the selectivity of wogonin. Although CDK9 is a general regulator of transcription, recent studies indicate that CDK9 activity is rather involved in pathological cellular processes than in normal cellular functions.13, 23 In addition, the phosphorylation of CTD of RNAPII at Ser2 has been shown not to be required for basal transcription in vitro.24 A deregulation of the CDK9-related pathway has been suggested, similar as, for example, growth factor receptor signaling, to be involved in the establishment and maintenance of a malignant cell phenotype.23 Consequently, transformed cells with high oncogenic stress are ‘addicted' to CDK9 activity due to the requirement for continuous production of anti-apoptotic proteins.
The semi-synthetic flavone flavopiridol, a pan-inhibitor of CDKs including CDK9, has been shown to effectively kill different tumor cells by apoptosis induction.13, 14 Currently, flavopiridol is tested in clinical trials as anti-cancer drug. Although encouraging efficacy has been reported, >40% of patients showed side effects including tumor lysis syndrome, electrolyte and liver function abnormalities, fatigue, diarrhea and cytopenias leading to infections.25 Recently, a study, which aimed to analyze the direct effects of CDK9 inhibition in global gene expression by using a dominant negative form of CDK9 (DN-CDK9), showed that DN-CDK9 targets fewer genes than flavopiridol, suggesting that flavopiridol has additional effects.26 As flavopiridol shows little selectivity between cancer and normal cells,27 our study may help to develop more specific CDK9 inhibitors by chemical modifications.
Besides their anti-cancer effects, wogonin, apigenin, chrysin and luteolin have also been shown to have anti-viral activity.28, 29 It is well known that viruses depend on their infected host for the transcription of their genome. CDK9 has been shown to be critical for viral replication of, for example, human immunodeficiency virus (HIV)-1 and HIV-2 in human cells.13, 23 Therefore, our study may partially explain the anti-viral effect of flavones.
In this study, wogonin was shown to inhibit CDK9 at a concentration of 25–50 μM in cell culture experiments. The effective concentrations are relatively high. However, in the cell-free assay, wogonin was shown to inhibit CDK9 activity with an IC50 of about 200 nM (Figure 2c). The discrepancy between the concentrations used in cell culture and in cell-free assays may be because of the poor bioavailability of wogonin in cell culture partially because of binding to bovine serum albumin. Several independent research groups have demonstrated that at a dose of 10–20 mg/kg wogonin efficiently inhibited tumor growth in vivo in several mouse tumor models although higher concentrations of wogonin (25–200 μM) were needed to achieve the same effects in cell culture experiments.2, 4, 5 The mouse data demonstrate that the amount of wogonin required to achieve anti-cancer effects in vivo is in a suitable range. Toxicological studies in experimental animals (rat and dog) showed that up to 60 mg/kg/day wogonin had no organ toxicity when intravenously administered for 90 days.30, 31 Thus, wogonin may be an attractive new anti-cancer compound that offers relative safety for long term therapies.
Taken together, our results demonstrate that wogonin and related natural flavones are CDK9 inhibitors. The current knowledge about the biology of CDK9 strongly suggests that targeting CDK9 is a promising therapeutic strategy in oncology and virology.13 Thus, our finding provides a new mechanism for the anti-cancer as well as the anti-viral activities of natural flavones (Figure 6d).
Materials and Methods
Cell lines and culture
The human malignant cell lines used in this study are the T-cell leukemic cell lines CEM and Jurkat (J16), the T-cell lymphoma cell lines Hut78 and Myla, the adult T-cell leukemic cell line SP derived from a HTLV-1-infected patient,32 the B-cell lymphoma cell line Raji, the Hodgkin lymphoma cell line L1236, the melanoma cell line SK-MEL-37, the hepatocellular carcinoma cell line HepG2, the pancreatic carcinoma cell line Colo-357, the breast cancer cell line SKBR3 and the colon carcinoma cell line HCT116. All cells were cultured in RPMI 1640 or DMEM medium (Gibco laboratories, Grand Island, NE, USA), respectively, supplemented with 10% FCS, 100 U/ml penicillin (Gibco), 100 μg/ml streptomycin (Gibco) and 2 mM -glutamine (Gibco) at 37° C and 5% CO2.
Preparation of human T cells from peripheral blood
Human peripheral T cells were prepared as described previously6 and were >90% CD3 positive. For activation, freshly isolated T cells were cultured at 2 × 106 cells/ml and were activated with 1 μg/ml PHA overnight. Activated T cells were then washed three times and cultured for additional 5 days in the presence of 25 U/ml IL-2 (activated T cells).
Determination of apoptosis
Cells were treated with different concentrations of wogonin (Biotrend Chemicals AG, Wangen, Switzerland), apigenin, chrysin, luteolin (Sigma-Aldrich, St. Louis, MO, USA) solved in dimetyl sulfoxide (Roth, Karlsruhe, Germany) at a stock concentration of 50 mM, zVAD-FMK (Bachem, Weil am Rhein, Germany), MG-132 (Calbiochem, Darmstadt, Germany) or CHX (Chem Service, West Chester, PA, USA) for the indicated periods of time. Apoptotic cell death was examined by analysis of DNA fragmentation as previously described.3, 6 Results are presented as % specific DNA fragmentation using the formula: (percentage of experimental apoptosis−percentage of spontaneous apoptosis)/(100−percentage of spontaneous apoptosis) × 100.
Western blot analysis
For each sample, 1 × 106 cells were lysed as previously described.3, 6 Equal amounts of protein were separated on 5–13% SDS-PAGE depending on the molecular sizes of the proteins, blotted onto a nitrocellulose membrane (Amersham Biosciences, Little Chalfon, UK) as previously described.6, 7 The following antibodies were used: RNAPIIa (hypo-phosphorylated form) and phospho-RNAPII (Ser2 and Ser5) are made by our laboratory;33 Bad, Bax, Bcl-xL, Bid, CDK7, CDK9, ERK1, PUMA, RB, phospho-RB (Ser780 and Ser795) and XIAP from Cell Signaling Technology (Danvers, MA, USA); Bcl-2 (sc-509), HSP90, Mcl-1 (sc-819) and phospho-RB (Thr821/826) from Santa Cruz Biotechnology (Heidelberg, Germany); Mcl-1 from BD Biosciences (Erembodegem, Belgium) and Tubulin from Sigma-Aldrich.
Quantitative real-time PCR
RNA was isolated using the RNeasy kit (Qiagen, Hilden, Germany) according to the manufacturer's instructions. A 1 μg of total RNA was reverse transcribed using the Perkin Elmer GeneAmp RNA PCR kit (Foster City, CA, USA). For TaqMan quantitative real-time PCR, the conditions and the sequences of primers and fluorescent-labeled probes for the human Mcl-1, Bcl-2 and 18S rRNA were described previously.34, 35, 36 The level of mRNA, relative to 18S rRNA, was calculated using the formula: Relative mRNA expression=2–(Ct of Mcl-1/Bcl-2−Ct of 18S rRNA) whereby Ct is the respective threshold cycle value.
[3H]-uridine-incorporation
CEM cells (2 × 105 cells/well) were treated with wogonin serially diluted across the plate. After addition of 1 μCi/well [5,6-3H]-uridine (Perkin Elmer, Waltham, MA, USA), cells were incubated for 2 h and 6 h at 37° C and 5% CO2 in a final volume of 200 μl. At the end of the incubation time, the cells were aspirated onto glass fiber filters using a multiple automated harvester (Tomtec, Perkin Elmer). The filter mats were washed with distilled water to remove non-adherent material, dried and the amount of [5,6-3H]-uridine incorporated into the retained RNA was measured using a Wallac Microbeta Trilux scintillation counter (Perkin Elmer).
Kinase assay
To determine the effect of wogonin on CDK7 and CDK9 activities, CDK7/cyclinH/MAT1 or CDK9/cyclinT (Millipore, Dundee, UK) and the substrate peptide (Millipore) were incubated with 8 mM MOPS (pH 7.0) 0.2 mM EDTA, 10 mM Mg-acetate and 10 μM γ-[33P]-ATP. The reaction was initiated by the addition of the MgATP mix. After incubation for 40 min at room temperature, the reaction was stopped by the addition of a 3% phosphoric acid solution. An aliquot of the reaction was then spotted onto a P30 filtermat and washed three times for 5 min in 75 mM phosphoric acid and once in methanol before drying and scintillation counting. Alternatively, the inhibitory effect of wogonin on kinase activity was examined by incubating recombinant CDK7/cyclinH/MAT1 or CDK9/cyclinT (Proqinase GmbH, Freiburg, Germany) and recombinant substrate RBER-CHKtide (Proqinase) in the presence of different concentrations of wogonin in 60 mM HEPES-NaOH (pH 7.5) 3 mM MgCl2, 3 mM MnCl2, 3 μM Na-orthovanadate, 1.2 mM DTT, 50 μg/ml PEG20000 and 1 μM ATP for 2 h at 30°C. The amount of phosphorylated substrate was determined by western blot analysis using phospho-specific antibodies.
Molecular docking studies of wogonin on CDK9
The X-ray structure of human CDK9/cyclinT1 in complex with flavopiridol (PDB CODE:3BLR)37 was taken as docking structure template. Docking calculations were performed using the AutoDOCK program. An energy-minimized 3D structure of wogonin compatible for docking was used throughout the docking operation. Before the start of the docking operation, essential hydrogens and Gasteiger chargers were added to the macromolecules CDK9. To sample the binding site, a grid of 120 × 120 × 120 Å (in case of blind docking) and 60 × 60 × 60 Å (in case of specific docking) with a spacing of 0.375 Å was first computed. In total, 100 cycles of flexible ligand docking with approximately 250 000 energy evaluations in each cycle without any flexibility constraints on the ligand were performed in the grid representation of the receptor binding site, followed by scoring the ligand–receptor interaction. AutoDock clustering was performed based on similarities in binding modes and affinities in these cycles. The AutoDock docking output contains solutions ranked according to the scoring functions with information about the frequency of occurrence, mean energies (ΔG), inhibition constant and root mean square deviations (RMSDs) within the cluster each defined by its corresponding 3D coordinates. PyMOL was used as a visualization tool to further achieve a deeper insight into the binding modes and to calculate the RMSD.38 Residues involved in protein–ligand interactions and their chemical binding patterns were analyzed using MultiBind online web-server, http://bioinfo3d.cs.tau.ac.il/MultiBind/.39
Pull-down assay
The pull-down assay was performed using a flavone-conjugate coupled to Affi-Gel-10 agarose beads (Bio-Rad laboratories, Hercules, CA, USA). CEM cells (1 × 108) were washed in phosphate-buffered saline and lysed in 2 ml lysis buffer containing 50 mM Tris/HCl (pH 8.0), 120 mM NaCl, 1% NP-40, 5 mM DTT, 200 μM Na-orthovanadate, 25 mM NaF and protease inhibitor cocktail (Roche Diagnostics, Mannheim, Germany). Cellular debris was removed by centrifugation at 10 000 × g for 30 min. A 500 μg of total protein extract was incubated for 12 h at 4°C with 40 μl of flavone-coupled, negative control-coupled or uncoupled Affi-Gel beads. The beads were extensively washed with lysis buffer and bound proteins were eluted by SDS sample buffer containing 20 mM Tris/HCl (pH 6.8), 1% SDS, 10% glycerol, 3% β-mercaptoethanol and bromophenol blue. Eluted proteins were recovered from the beads by centrifugation and subjected to SDS-PAGE and western blot analysis.
Knockdown and overexpression studies
CEM cells (2 × 106) or Raji cells (3 × 106) were transfected in Nucleofector solution (Nucleofector kit C or V, respectively, Amaxa Biosystems, Cologne, Germany) with 1–2 μM of nonsense siRNA (Qiagen), Mcl-1 siRNA (5′-CGCCGAAUUCAUUAAUUUATT-3′ Qiagen), CDK9 siRNA #1 (5′-UGAGAUUUGUCGAACCAAATT-3′ Applied Biosystems, Warrington, UK) or CDK9 siRNA #2 (5′-GGCACAGUUUGGUCCGUUATT-3′ Qiagen) using the Amaxa Nucleofector apparatus and the program X-01. Cells were collected at indicated time points after transfection for apoptosis measurement and Mcl-1 protein expression analysis. Transfection of HCT116 cells was carried out using Lipofectamine 2000 (Invitrogen, Paisley, UK) and 33 nM of Mcl-1 siRNA according to the manufacturer's instructions. Cells were collected at 48 h after transfection for apoptosis measurement and Mcl-1 protein expression analysis. For Mcl-1 overexpression, HCT116 cells were transfected using Lipofectamine 2000 with the specific expression vector pEF4Mcl-1 or empty vector as described previously.40
Acknowledgments
This work was supported by the Helmholtz Alliance on Immunotherapy of Cancer in the Helmholtz association.
Glossary
- CDK
cyclin-dependent kinase
- CTD
carboxy-terminal domain
- HIV
human immunodeficiency virus
- HTLV-1
human T-cell leukemia/lymphoma virus 1
- IC50
half-maximal inhibitory concentration
- Mcl-1
myeloid cell leukemia 1
- PLCγ1
phospholipase Cγ1
- RB
retinoblastoma protein
- RNAPII
RNA polymerase II
The authors declare no conflict of interest.
Footnotes
Supplementary Information accompanies the paper on Cell Death and Disease website (http://www.nature.com/cddis)
Edited by G Melino
Supplementary Material
References
- Li-Weber M. New therapeutic aspects of flavones: the anticancer properties of Scutellaria and its main active constituents wogonin, baicalein and baicalin. Cancer Treat Rev. 2009;35:57–68. doi: 10.1016/j.ctrv.2008.09.005. [DOI] [PubMed] [Google Scholar]
- Wang W, Guo QL, You QD, Zhang K, Yang Y, Yu J, et al. The anticancer activities of wogonin in murine sarcoma S180 both in vitro and in vivo. Biol Pharm Bull. 2006;29:1132–1137. doi: 10.1248/bpb.29.1132. [DOI] [PubMed] [Google Scholar]
- Baumann S, Fas SC, Giaisi M, Muller WW, Merling A, Gulow K, et al. Wogonin preferentially kills malignant lymphocytes and suppresses T-cell tumor growth by inducing PLCgamma1- and Ca2+-dependent apoptosis. Blood. 2008;111:2354–2363. doi: 10.1182/blood-2007-06-096198. [DOI] [PubMed] [Google Scholar]
- Chung H, Jung YM, Shin DH, Lee JY, Oh MY, Kim HJ, et al. Anticancer effects of wogonin in both estrogen receptor-positive and -negative human breast cancer cell lines in vitro and in nude mice xenografts. Int J Cancer. 2008;122:816–822. doi: 10.1002/ijc.23182. [DOI] [PubMed] [Google Scholar]
- Lu N, Gao Y, Ling Y, Chen Y, Yang Y, Gu HY, et al. Wogonin suppresses tumor growth in vivo and VEGF-induced angiogenesis through inhibiting tyrosine phosphorylation of VEGFR2. Life Sci. 2008;82:956–963. doi: 10.1016/j.lfs.2008.02.013. [DOI] [PubMed] [Google Scholar]
- Fas SC, Baumann S, Zhu JY, Giaisi M, Treiber MK, Mahlknecht U, et al. Wogonin sensitizes resistant malignant cells to TNFalpha- and TRAIL-induced apoptosis. Blood. 2006;108:3700–3706. doi: 10.1182/blood-2006-03-011973. [DOI] [PubMed] [Google Scholar]
- Perez AT, Arun B, Tripathy D, Tagliaferri MA, Shaw HS, Kimmick GG, et al. A phase 1B dose escalation trial of Scutellaria barbata (BZL101) for patients with metastatic breast cancer. Breast Cancer Res Treat. 2010;120:111–118. doi: 10.1007/s10549-009-0678-5. [DOI] [PubMed] [Google Scholar]
- Rugo H, Shtivelman E, Perez A, Vogel C, Franco S, Tan Chiu E, et al. Phase I trial and antitumor effects of BZL101 for patients with advanced breast cancer. Breast Cancer Res Treat. 2007;105:17–28. doi: 10.1007/s10549-006-9430-6. [DOI] [PubMed] [Google Scholar]
- Galluzzi L, Larochette N, Zamzami N, Kroemer G. Mitochondria as therapeutic targets for cancer chemotherapy. Oncogene. 2006;25:4812–4830. doi: 10.1038/sj.onc.1209598. [DOI] [PubMed] [Google Scholar]
- Adams JM, Cory S. The Bcl-2 apoptotic switch in cancer development and therapy. Oncogene. 2007;26:1324–1337. doi: 10.1038/sj.onc.1210220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Akgul C. Mcl-1 is a potential therapeutic target in multiple types of cancer. Cell Mol Life Sci. 2009;66:1326–1336. doi: 10.1007/s00018-008-8637-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hussain SR, Cheney CM, Johnson AJ, Lin TS, Grever MR, Caligiuri MA, et al. Mcl-1 is a relevant therapeutic target in acute and chronic lymphoid malignancies: down-regulation enhances rituximab-mediated apoptosis and complement-dependent cytotoxicity. Clin Cancer Res. 2007;13:2144–2150. doi: 10.1158/1078-0432.CCR-06-2294. [DOI] [PubMed] [Google Scholar]
- Wang S, Fischer PM. Cyclin-dependent kinase 9: a key transcriptional regulator and potential drug target in oncology, virology and cardiology. Trends Pharmacol Sci. 2008;29:302–313. doi: 10.1016/j.tips.2008.03.003. [DOI] [PubMed] [Google Scholar]
- Shapiro GI. Cyclin-dependent kinase pathways as targets for cancer treatment. J Clin Oncol. 2006;24:1770–1783. doi: 10.1200/JCO.2005.03.7689. [DOI] [PubMed] [Google Scholar]
- Sims RJ, III, Belotserkovskaya R, Reinberg D. Elongation by RNA polymerase II: the short and long of it. Genes Dev. 2004;18:2437–2468. doi: 10.1101/gad.1235904. [DOI] [PubMed] [Google Scholar]
- Patel D, Shukla S, Gupta S. Apigenin and cancer chemoprevention: progress, potential and promise (review) Int J Oncol. 2007;30:233–245. [PubMed] [Google Scholar]
- Seelinger G, Merfort I, Wolfle U, Schempp CM. Anti-carcinogenic effects of the flavonoid luteolin. Molecules. 2008;13:2628–2651. doi: 10.3390/molecules13102628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Warr MR, Shore GC. Unique biology of Mcl-1: therapeutic opportunities in cancer. Curr Mol Med. 2008;8:138–147. doi: 10.2174/156652408783769580. [DOI] [PubMed] [Google Scholar]
- Jiang CC, Lucas K, Avery-Kiejda KA, Wade M, deBock CE, Thorne RF, et al. Up-regulation of Mcl-1 is critical for survival of human melanoma cells upon endoplasmic reticulum stress. Cancer Res. 2008;68:6708–6717. doi: 10.1158/0008-5472.CAN-08-0349. [DOI] [PubMed] [Google Scholar]
- Schulze-Bergkamen H, Fleischer B, Schuchmann M, Weber A, Weinmann A, Krammer PH, et al. Suppression of Mcl-1 via RNA interference sensitizes human hepatocellular carcinoma cells towards apoptosis induction. BMC Cancer. 2006;6:232. doi: 10.1186/1471-2407-6-232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koppenol WH, Bounds PL, Dang CV. Otto Warburg's contributions to current concepts of cancer metabolism. Nat Rev Cancer. 2011;11:325–337. doi: 10.1038/nrc3038. [DOI] [PubMed] [Google Scholar]
- Trachootham D, Alexandre J, Huang P. Targeting cancer cells by ROS-mediated mechanisms: a radical therapeutic approach. Nat Rev Drug Discov. 2009;8:579–591. doi: 10.1038/nrd2803. [DOI] [PubMed] [Google Scholar]
- Romano G, Giordano A. Role of the cyclin-dependent kinase 9-related pathway in mammalian gene expression and human diseases. Cell Cycle. 2008;7:3664–3668. doi: 10.4161/cc.7.23.7122. [DOI] [PubMed] [Google Scholar]
- Serizawa H, Conaway JW, Conaway RC. Phosphorylation of C-terminal domain of RNA polymerase II is not required in basal transcription. Nature. 1993;363:371–374. doi: 10.1038/363371a0. [DOI] [PubMed] [Google Scholar]
- Abou-Nassar K, Brown JR. Novel agents for the treatment of chronic lymphocytic leukemia. Clin Adv Hematol Oncol. 2011;8:886–895. [PubMed] [Google Scholar]
- Garriga J, Xie H, Obradovic Z, Grana X. Selective control of gene expression by CDK9 in human cells. J Cell Physiol. 2010;222:200–208. doi: 10.1002/jcp.21938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu X, Shi S, Lam F, Pepper C, Fischer PM, Wang S.CDKI-71, a novel CDK9 inhibitor, is preferentially cytotoxic to cancer cells when compared with flavopiridol Int J Cancer 2011. Apr: 11. [DOI] [PubMed]
- Liu AL, Liu B, Qin HL, Lee SM, Wang YT, Du GH. Anti-influenza virus activities of flavonoids from the medicinal plant Elsholtzia rugulosa. Planta Med. 2008;74:847–851. doi: 10.1055/s-2008-1074558. [DOI] [PubMed] [Google Scholar]
- Guo Q, Zhao L, You Q, Yang Y, Gu H, Song G, et al. Anti-hepatitis B virus activity of wogonin in vitro and in vivo. Antiviral Res. 2007;74:16–24. doi: 10.1016/j.antiviral.2007.01.002. [DOI] [PubMed] [Google Scholar]
- Qi Q, Peng J, Liu W, You Q, Yang Y, Lu N, et al. Toxicological studies of wogonin in experimental animals. Phytother Res. 2009;23:417–422. doi: 10.1002/ptr.2645. [DOI] [PubMed] [Google Scholar]
- Peng J, Qi Q, You Q, Hu R, Liu W, Feng F, et al. Subchronic toxicity and plasma pharmacokinetic studies on wogonin, a natural flavonoid, in Beagle dogs. J Ethnopharmacol. 2009;124:257–262. doi: 10.1016/j.jep.2009.04.031. [DOI] [PubMed] [Google Scholar]
- Krueger A, Fas SC, Giaisi M, Bleumink M, Merling A, Stumpf C, et al. HTLV-1 Tax protects against CD95-mediated apoptosis by induction of the cellular FLICE-inhibitory protein (c-FLIP) Blood. 2006;107:3933–3939. doi: 10.1182/blood-2005-06-2567. [DOI] [PubMed] [Google Scholar]
- Chapman RD, Heidemann M, Albert TK, Mailhammer R, Flatley A, Meisterernst M, et al. Transcribing RNA polymerase II is phosphorylated at CTD residue serine-7. Science. 2007;318:1780–1782. doi: 10.1126/science.1145977. [DOI] [PubMed] [Google Scholar]
- Rosato RR, Almenara JA, Kolla SS, Maggio SC, Coe S, Gimenez MS, et al. Mechanism and functional role of XIAP and Mcl-1 down-regulation in flavopiridol/vorinostat antileukemic interactions. Mol Cancer Ther. 2007;6:692–702. doi: 10.1158/1535-7163.MCT-06-0562. [DOI] [PubMed] [Google Scholar]
- Kang MH, Wan Z, Kang YH, Sposto R, Reynolds CP. Mechanism of synergy of N-(4-hydroxyphenyl)retinamide and ABT-737 in acute lymphoblastic leukemia cell lines: Mcl-1 inactivation. J Natl Cancer Inst. 2008;100:580–595. doi: 10.1093/jnci/djn076. [DOI] [PubMed] [Google Scholar]
- Osman F, Rowhani A. Real-time RT-PCR (TaqMan) assays for the detection of viruses associated with Rugose wood complex of grapevine. J Virol Methods. 2008;154:69–75. doi: 10.1016/j.jviromet.2008.09.005. [DOI] [PubMed] [Google Scholar]
- Baumli S, Lolli G, Lowe ED, Troiani S, Rusconi L, Bullock AN, et al. The structure of P-TEFb (CDK9/cyclin T1), its complex with flavopiridol and regulation by phosphorylation. EMBO J. 2008;27:1907–1918. doi: 10.1038/emboj.2008.121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DeLano WL. Unraveling hot spots in binding interfaces: progress and challenges. Curr Opin Struct Biol. 2002;12:14–20. doi: 10.1016/s0959-440x(02)00283-x. [DOI] [PubMed] [Google Scholar]
- Shatsky M, Shulman-Peleg A, Nussinov R, Wolfson HJ. The multiple common point set problem and its application to molecule binding pattern detection. J Comput Biol. 2006;13:407–428. doi: 10.1089/cmb.2006.13.407. [DOI] [PubMed] [Google Scholar]
- Schulze-Bergkamen H, Ehrenberg R, Hickmann L, Vick B, Urbanik T, Schimanski CC, et al. Bcl-x(L) and myeloid cell leukaemia-1 contribute to apoptosis resistance of colorectal cancer cells. World J Gastroenterol. 2008;14:3829–3840. doi: 10.3748/wjg.14.3829. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.