Abstract
Background
Recent evidence suggests that autonomic nervous system activity could be involved in the pathophysiology of sickle cell disease, but it is unclear whether differences in autonomic nervous system activity are detectable during steady state in patients with mild and severe disease. The aim of the present study was to compare the autonomic nervous system activity, blood rheology, and inflammation in patients with sickle cell anemia according to the frequency of acute pain crisis.
Design and Methods
Twenty-four healthy volunteers, 20 patients with sickle cell anemia with milder disease, and 15 patients with sickle cell anemia with more severe disease were recruited. Milder disease was defined as having no pain crisis within the previous year. More severe disease was defined as having had within the previous year three or more pain crises which were documented by a physician and required treatment with narcotics. The autonomic nervous system activity was determined by spectral analysis of nocturnal heart rate variability. Blood viscosity determination and measurements of several inflammatory markers (interleukin-6, soluble vascular cell adhesion molecule-1, soluble CD40 ligand and sL-selectin) were made on blood samples collected in steady-state conditions.
Results
Results showed that: 1) patients who had suffered more frequent pain crises had lower parasympathetic activity and greater sympatho-vagal imbalance than both controls and patients with milder disease. However, when adjusted for age, no significant difference was detected between the two sickle cell anemia patient groups; 2) patients who had suffered more frequent pain crises had higher blood viscosity than patients with milder disease, and this was not dependent on age.
Conclusions
Results from the present study indicate that both the autonomic nervous system activity and blood viscosity are impaired in patients with sickle cell anemia exhibiting high frequency of pain crisis in comparison with those who did not experience a crisis within the previous year.
Keywords: sickle cell anemia, autonomic function, inflammation, hemoglobin, blood viscosity
Introduction
Sickle cell anemia (SCA) is the most common genetic disease in Africa, the Caribbean, the Americas, the Middle East, and India. Although it is a monogenic disorder, its phenotypic expression and severity are highly variable from one patient to another. It has been established that blood rheology alterations,1 chronic vascular inflammation and abnormal adhesion processes,2,3 and vascular dysfunction4 play an important role in the occurrence of SCA complications. It is still not clear how these mechanisms interact to modulate the phenotypic expression of SCA, but the occurrence of two main clinical sub-phenotypes is now the subject of much debate: 1) a hemolysis-endothelial dysfunction phenotype; and 2) a viscosity-vaso-occlusive phenotype, the latter being associated with vaso-occlusive pain crisis, acute chest syndrome and osteonecrosis.5,6
Over the past two decades, there has been a greater interest in the study of the autonomic nervous system (ANS) activity in several chronic diseases. A decrease or imbalance in ANS activity is considered a powerful and independent predictor of cardiovascular and cerebrovascular adverse events, as well as death from any cause, in the general population and in patients with established heart disease or metabolic syndrome.7,8 In SCA patients, Inamo et al.9 found depressed mean ANS activity when compared to healthy controls but the level of ANS activity depression varied greatly from one patient to another. Pearson et al.10 and Romero Mestre et al.11 suggested a possible relationship between autonomic reactivity and the clinical severity of SCA. Pearson et al.10 speculated that autonomic dysregulation could alter autonomic tone in patients with SCA and could, therefore, exacerbate pain episodes by increasing peripheral vasoconstriction. Romero-Mestre et al.11 speculated that the sudden deaths reported in sickle cell disease (SCD) could be partly related to the cardiovascular autonomic nervous dysfunction. However, although it has been suggested that ANS activity could be involved in the pathophysiology of SCA, the mechanisms by which ANS imbalance might modulate the clinical severity of SCA are still poorly understood.12,13
In an effort to gain further insight into the relationship between ANS activity and clinical expression of SCA, we compared the ANS activity pattern of SCA patients with frequent severe pain crises within the previous year with that of SCA patients who had not experienced any pain crises during this same time, and with a control group made up of healthy subjects. In addition, since associations between blood rheology, inflammation and ANS activity have been proposed in other diseases,14,15 hematologic, blood viscosity and inflammatory markers were compared between the three groups.
Design and Methods
Participants and protocol
This study was designed to compare inflammatory, hemorheological, and ANS activity markers in: i) 15 Jamaican SCA patients with at least three episodes of acute vaso-occlusive pain crises requiring day care16 or hospital admission, and opioid analgesia within the previous year (frequent crisis, FSCA group); ii) 20 Jamaican SCA patients who had not experienced any pain crises during the year prior to recruitment (infrequent crisis, IFSCA group); and (iii) 24 healthy Jamaican subjects without hemoglobinopathy (controls, CONT group). All the SCA patients (SS homozygotes) had benefited from medical follow up at the Sickle Cell Unit of the Tropical Medicine Research Institute in Kingston, Jamaica, for several years and were in steady-state condition at the time of their recruitment to the study. Steady state was defined as a period without blood transfusions within the previous four months and the absence of acute episodes (infection or vaso-occlusive crisis) for at least one month before enrolment in the study. The stratification of SCA patients according to the frequency of crisis was similar to that used by Phillips et al.17 and Platt et al.,18 except that visits to the Sickle Cell Unit during which narcotics were prescribed for an episode of pain crisis were assessed as equivalent to visits to an Accident and Emergency Department. It is currently used by physicians from the Sickle Cell Unit of the Tropical Medicine Research Institute in Kingston, Jamaica, to decide whether or not they propose hydroxyurea therapy to SCA patients who are exposed to frequent adverse acute medical events (painful vasoocclusive crisis or acute chest syndrome). An acute event was scored as a vaso-occlusive crisis if the pain episode lasted for longer than one hour, the patients felt that the pain was secondary to vaso-occlusion (‘typical’ pain for them) and no other etiology of pain was apparent, and if patients were admitted to either the Accident and Emergency Department17,18 or the Sickle Cell Unit16 for an acute visit. The Jamaican Sickle Cell Unit offers comprehensive care, including acute care in an 8-bed day care unit equivalent to an Accident and Emergency Department five days a week, so during the week patients come to the Unit rather than going to an Accident and Emergency Department.16 The vaso-occlusive episodes were documented in the patient’s chart by a physician as severe pain crisis. All the information regarding the vaso-occlusive episodes has been collected retrospectively and independently by a physician who was not aware of the results of the ANS, inflammation and blood viscosity studies. Documentation of pain crises covered a period of one year before the time that holters electrocardiograms were placed and samples were drawn for the different studies performed. Exacerbation of chronic painful conditions, such as avascular necrosis of bone or painful leg ulcers, were not considered as a vaso-occlusive crisis. Exclusion criteria were ongoing pregnancy, known narcotic addiction, recent blood transfusion, acute chest syndrome within the previous month, HIV infection, ischemic heart disease, or diabetes mellitus. Although half of the SCA participants were classified as FSCA, none of them were receiving hydroxyurea therapy. This study was approved by the Ethics Committee of the University of the West Indies, Mona Campus, Kingston, Jamaica, and all subjects gave their informed consent in accordance with the declaration of Helsinki.
Hematology and blood viscosity
Venepuncture was performed between 9:00 a.m. and 10:00 a.m. the day after holter recording. EDTA blood samples were immediately used for measurements of hemoglobin concentration (Hb), hematocrit (Hct), mean corpuscular volume (MCV), mean corpuscular hemoglobin concentration (MCHC), red blood cell distribution width (RDW), platelet (PLT) and white blood cell (WBC) counts, and percentage of reticulocytes (RET) (Max M-Retic, Coulter, Miami, FL, USA). Fetal hemoglobin (HbF) level was determined by high performance liquid chromatography (VARIANTTM, Bio-Rad Laboratories, Hercules, CA, USA). Whole blood viscosity (ηb) was measured at moderate and high shear rate (46 and 230 s−1) by a cone and plate viscometer (Brookfield DVII+, Middleboro, MA, USA) within the first hour following phlebotomy, at a temperature of 37°C and according to the recommendation of the recent guidelines on hemorheology.19 Full oxygenation of blood was performed before blood measurement of viscosity using the recommended methods.19 The Hct/ηb ratio was calculated at both shear rates and is considered a hemorheological index of oxygen transport potential.20
Inflammatory markers
Plasma from blood collected in heparin tubes was immediately obtained by centrifugation (1,000 g, 4°C, 5 min) and stored at −80°C for subsequent analysis. ELISA kits were used to assay the plasma concentrations of the following cytokines and soluble adhesion molecules: interleukin-6 (IL-6), tumor necrosis factor α (TNF-α), soluble vascular cell adhesion molecule (sVCAM-1), soluble CD40 ligand (sCD40L) and soluble L-selectin (sL-selectin) (Diaclone Systems, Besançon, France; high sensitivity kits for IL-6 and TNF-α), according to the manufacturer’s instructions. The sVCAM-1, sCD40L and sL-selectin reflected endothelial, platelet, and leukocyte activation, respectively. The inflammatory markers assessed in the present study were those known to play a role in the pathophysiological mechanisms of acute SCA complications.2,21–23
Autonomic nervous system activity
A holter electrocardiograph was placed on the chest of each patient after clinical examination and recording had been taken from 6:00 p.m. to 8:00 a.m. The electrocardiographic Holter system (Novacor system, Duosoft, Rueil-Malmaison, France) was used during the night to extract the RR interval list with a precision of 0.008 s. Each RR interval was validated before analysis. Only the night periods were analyzed (midnight to 7 am) to avoid variations arising from differences in the subject’s daily environment.24 The mean heart rate (beats per minute, bpm), and the standard deviation of all normal RR intervals (SDNN), were calculated first. After fast Fourier transform, the power spectrum indices were calculated as recommended by the Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology.7 The high frequency peak of the spectrum (HF, 0.15–0.40 Hz) is known to represent parasympathetic activity, and the low frequency (LF, 0.04–0.15 Hz) represents both parasympathetic and sympathetic activities. Additional calculations included: the very low frequency power (0–0.04 Hz), the LF/HF ratio and the total frequency power (Ptot). SDNN and Ptot reflected the global autonomic activity.7 The very low frequency of spectrum (VLF) partially contains parasympathetic activity and is predictive of clinical prognosis in cardiac patients.25 The LF/HF ratio has been proposed as a marker for autonomic nervous system balance.7
Statistical analysis
ANS activity and biological parameters were compared between the three groups using a one-way analysis of variance (ANOVA) and differences between two groups were tested using the Newman-Keuls test. The Hct and Hct/ηb ratios were compared between the three groups using a non-parametric ANOVA (Kruskal-Wallis test) and differences between two groups were tested using the Dunn’s post test. Analysis of covariance (ANCOVA) was performed on ANS parameters and ηb in SCA patients only, while controlling for age and gender. Correlations between age, biological and ANS parameters were tested by Pearson’s test both in SCA patients only and in all subjects. Values are expressed as means ± SD or median (25th, 75th). The significance level was defined as P<0.05 (Statistica, v. 5.5, Statsoft, Tulsa, OK, USA).
Results
Hematologic and hemorheological parameters
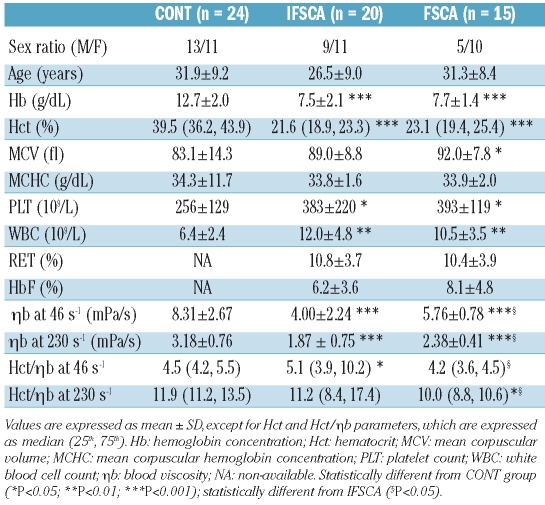
The number of pain crises requiring parenteral opioid analgesia in the FSCA group ranged from 3 to 8 (3.9±1.4). No age difference was observed between the three groups and although FSCA patients were slightly older than IFSCA patients, the difference was not significant between the two groups (P=0.10). Both the IFSCA and the FSCA groups had lower values for Hb and Hct compared with the CONT group (Table 1). Consequently, ηb measured at both shear rates was significantly reduced in the two groups of SCA patients but the FSCA group showed significantly greater values than the IFSCA group. Age and ηb were neither significantly correlated in the combined SCA group (FSCA and IFSCA patients; r=0.019 and 0.059, and P=0.92 and 0.76, for ηb at 46 s−1 and 230 s−1, respectively), nor in the three groups (r=0.026 and 0.074, and P=0.85 and 0.60, for ηb at 46 s−1 and 230 s−1, respectively). No difference in ηb was observed between males and females. When controlled for age and gender, the differences in ηb between the two SCA groups remained significant, indicating that frequency of pain crisis was associated with elevated blood viscosity. The Hct/ηb ratio at the shear rate of 46 s−1 was higher in the IFSCA group than in the FSCA group and did not differ from the CONT group. Although the FSCA group had a lower value than the CONT group, the difference between these two groups did not reach statistical significance. At the shear rate of 230 s−1, the Hct/ηb ratio was reduced in the FSCA group as compared with both the IFSCA and the CONT groups, with no difference between the two latter groups. There was no difference in MCHC between the three groups. There was no statistical difference in MCV between the IFSCA group and the CONT group whereas FSCA patients showed greater values than the CONT group. When the two SCA groups were combined, SCA patients had greater MCV than the CONT group. White blood cell and platelet counts were higher in the two SCA groups with no difference between them. There was no difference in RET and HbF levels between the IFSCA and FSCA groups.
Table 1.
Hematologic and hemorheological parameters in the three studied groups.
Parameters of autonomic nervous system activity
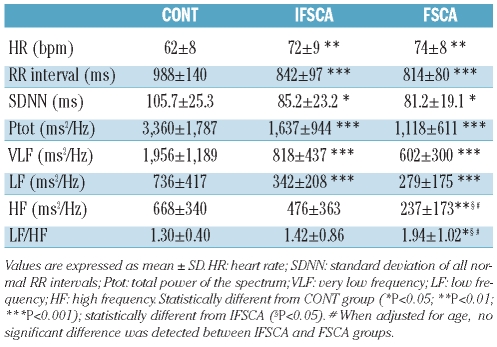
Heart rate was higher in the two SCA groups than in the CONT group, as confirmed by the decrease in RR intervals of the IFSCA and the FSCA groups (Table 2). The indices reflecting the global autonomic nervous system activity (i.e. SDNN and Ptot), as well as VLF and LF, were decreased in the two groups of SCA patients compared to the CONT group, with no significant difference between the two SCA groups. There was no significant difference in the index of parasympathetic activity (HF) between the IFSCA and the CONT groups (P=0.09) but HF was lower in the FSCA group compared with the IFSCA and the CONT groups (Table 2). The index of autonomic nervous system balance (LF/HF) was also greater in the FSCA group than both the CONT and the IFSCA groups. Controlling for gender had no impact on the associations observed between the SCA groups with HF and LF/HF. When adjusted for age, however, there was no significant difference in HF or LF/HF between the SCA groups. HF was significantly related with age in the combined SCA group (r = −0.48; P<0.01) and in the three groups (r = −0.31; P<0.05). No correlation was found between hemorheological parameters and ANS activity in the SCA groups.
Table 2.
Parameters of autonomic nervous system activity in the three studied groups.
Inflammatory markers
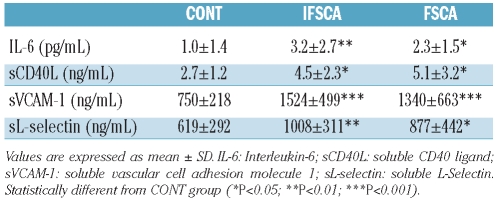
Levels of inflammatory markers are shown in Table 3. The two groups of SCA patients had greater values than the CONT group, but no significant difference was observed between the FSCA and the IFSCA groups. TNF-α levels were very low in the two SCA groups with no difference observed between them (data not shown). None of the inflammatory molecules were correlated with ANS activity in any of the SCA patients (data not shown).
Table 3.
Plasmatic concentrations of inflammatory markers in the three studied groups.
Discussion
The major findings of the present study are that, in steady state conditions: i) FSCA patients had higher blood viscosity than IFSCA patients, and this was not dependent on age; ii) patients with SCA had lower parasympathetic activity and greater sympatho-vagal imbalance than controls.
Epidemiological studies have demonstrated that low HbF level and increased Hb and/or Hct level are risk factors for vaso-occlusive crisis.5,18,26 This has been rationalized by the resulting increased viscosity of high hematocrit sickle cell blood that may impair microvascular blood flow and cause tissue infarction.5 Indeed, it has been proposed that SCA patients may exhibit a viscosity-vaso-occlusive phenotype which is predominant in vaso-occlusive pain crisis and other clinical manifestations, such as acute chest syndrome or osteonecrosis.5 Our data support these findings since FSCA patients had higher blood viscosity than IFSCA patients. Surprisingly, Phillips et al.17 did not find any association between blood viscosity (or Hct) and frequency of pain crisis in patients with SCA. That could be due to the limited sampling size in their study (16 SCA subjects analyzed), and also to the fact that viscosity was measured in HEPES-buffered saline solution after washing the red blood cells twice and Hct adjustment to 90%; conditions clearly different to the physiological situation. Although there was no significant difference in Hct between the two SCA groups, the median Hct was slightly greater in the FSCA group than in the IFSCA group. Since Hct is the main determinant of whole blood viscosity, it could be suggested that the higher ηb found in the FSCA patients was attributed to the difference in Hct. In addition, the lower Hct/ηb ratio observed in the FSCA patients supports the hypothesis that other still unidentified blood rheological factors may also be involved in the blood hyperviscosity observed in the FSCA group.
Our results on ANS activity are in agreement with those of Pearson et al.10 and Inamo et al.9 showing that SCA patients present an altered global autonomic nervous system activity (reduced SDNN and Ptot) compared with healthy subjects.9,10 It has been suggested that the decreased VLF and LF indices in SCA are involved in the alteration of the autonomic nervous system activity in SCA patients.9 Our results also demonstrated that SCA patients with frequent pain crises had impaired parasympathetic activity (low HF value) and marked sympatho-vagal imbalance when compared with both the SCA group with no pain crisis during the previous year and the CONT group. Autonomic nervous system imbalance has been observed in various diseases, such as diabetes,27 celiac disease,28 or fibromyalgia,29,30 and has been shown to be related to clinical severity and pain. However, in SCA, it is unclear whether the changes in ANS activity predate the occurrence of pain crises or are a consequence of them. In patients with β-thalassemia major, as in other forms of anemia, the decreased ANS activity and impaired sympa-tho-vagal balance has been attributed to the chronic anemia which may lead to a persistent sinus tachycardia and a sustained decrease in autonomic fluctuations.31 However, this hypothesis does not explain why FSCA patients had lower parasympathetic activity than IFSCA patients; the two groups exhibited similar low Hb and Hct levels. It is important to note that age and HF were significantly and negatively correlated in the combined SCA group, as well as in all three groups. In addition, when controlling for age, there was no longer any difference in HF or LF/HF between the two SCA groups. Although the difference in age between the IFSCA and the FSCA patients did not reach statistical significance, these results suggest that age has played a role in the decline of parasympathetic activity in our SCA patients, as is the case in healthy populations,32,33 indicating that FSCA patients have greater sympatho-vagal imbalance than IFSCA patients. Further studies are needed to investigate whether aging and ANS dysfunction are closely related or not to each other in SCA, and whether they play an independent role in SCA pathophysiology and severity.
Recent studies described a causal link between ANS activity and inflammation modulation.15,34,35 Stimulation of the vagal nerve, for instance, stimulates the release of acetylcholine, which inhibits the production of pro-inflammatory cytokines by white blood cells.36,37 However, we found no association between ANS activity parameters and inflammatory markers in SCA patients, and similarly no difference in inflammation between the FSCA and the IFSCA groups. Thus, although ANS activity impairment could be thought to play a role in the patho-physiology of SCA, the present study did not show a physiological link with inflammation. Further studies are clearly warranted to clarify the relationship between altered ANS activity and SCA severity.
A limitation of our study was the small sample size. This was, however, comparable to many papers concerning ANS activity in the literature.10,13,14,38 None the less, these findings add to the small body of literature concerning SCA disease severity, viscosity and ANS activity.
In conclusion, the present study demonstrates that both ANS activity and blood viscosity are impaired in patients with SCA showing a high frequency of pain crisis. Further studies are needed to address ANS activity in other conditions, such as during vaso-occlusive episodes, to define more precisely its role in the pathophysiological mechanisms of SCA.
Acknowledgments
The authors would like to express their sincere gratitude to all the patients who agreed to participate in the study and to the technical staff of the Laboratory SNA-EPIS (PRES Lyon, University of Saint-Etienne, France) for performing the HRV analyses. This study was supported in part by the French Embassy in Jamaica and by the Université des Antilles et de la Guyane.
Footnotes
Authorship and Disclosures
The information provided by the authors about contributions from persons listed as authors and in acknowledgments is available with the full text of this paper at www.haematologica.org.
Financial and other disclosures provided by the authors using the ICMJE (www.icmje.org) Uniform Format for Disclosure of Competing Interests are also available at www.haematologica.org.
References
- 1.Ballas SK, Mohandas N. Sickle red cell microrheology and sickle blood rheology. Microcirculation. 2004;11(2):209–25. doi: 10.1080/10739680490279410. [DOI] [PubMed] [Google Scholar]
- 2.Etienne-Julan M, Belloy MS, Decastel M, Dougaparsad S, Ravion S, Hardy-Dessources MD. Childhood sickle cell crises: clinical severity, inflammatory markers and the role of interleukin-8. Haematologica. 2004;89(7):863–4. [PubMed] [Google Scholar]
- 3.Stuart MJ, Nagel RL. Sickle-cell disease. Lancet. 2004;364(9442):1343–60. doi: 10.1016/S0140-6736(04)17192-4. [DOI] [PubMed] [Google Scholar]
- 4.Wood KC, Hsu LL, Gladwin MT. Sickle cell disease vasculopathy: a state of nitric oxide resistance. Free Radic Biol Med. 2008;44(8):1506–28. doi: 10.1016/j.freeradbiomed.2008.01.008. [DOI] [PubMed] [Google Scholar]
- 5.Kato GJ, Gladwin MT, Steinberg MH. Deconstructing sickle cell disease: reappraisal of the role of hemolysis in the development of clinical subphenotypes. Blood Rev. 2007;21(1):37–47. doi: 10.1016/j.blre.2006.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hebbel RP. Reconstructing sickle cell disease: a data-based analysis of the "hyperhemolysis paradigm" for pulmonary hypertension from the perspective of evidence-based medicine. Am J Hematol. 2011;86(2):123–54. doi: 10.1002/ajh.21952. [DOI] [PubMed] [Google Scholar]
- 7.TaskForce. Heart rate variability: standards of measurement, physiological interpretation and clinical use. Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Circulation. 1996;93(5):1043–65. [PubMed] [Google Scholar]
- 8.Kleiger RE, Stein PK, Bigger JT., Jr Heart rate variability: measurement and clinical utility. Ann Noninvasive Electrocardiol. 2005;10(1):88–101. doi: 10.1111/j.1542-474X.2005.10101.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Inamo J, Connes P, Barthelemy JC, Dan V, Coates T, Loko G. Pulmonary hypertension does not affect the autonomic nervous system dysfunction of sickle cell disease. Am J Hematol. 2009;84(5):311–2. doi: 10.1002/ajh.21377. [DOI] [PubMed] [Google Scholar]
- 10.Pearson SR, Alkon A, Treadwell M, Wolff B, Quirolo K, Boyce WT. Autonomic reactivity and clinical severity in children with sickle cell disease. Clin Auton Res. 2005;15(6):400–7. doi: 10.1007/s10286-005-0300-9. [DOI] [PubMed] [Google Scholar]
- 11.Romero Mestre JC, Hernandez A, Agramonte O, Hernandez P. Cardiovascular autonomic dysfunction in sickle cell anemia: a possible risk factor for sudden death? Clin Auton Res. 1997;7(3):121–5. doi: 10.1007/BF02308838. [DOI] [PubMed] [Google Scholar]
- 12.Alexy T, Sangkatumvong S, Connes P, Pais E, Tripette J, Barthelemy JC, et al. Sickle cell disease: selected aspects of pathophysiology. Clin Hemorheol Microcirc. 2010;44(3):155–66. doi: 10.3233/CH-2010-1270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sangkatumvong S, Coates TD, Khoo MC. Abnormal autonomic cardiac response to transient hypoxia in sickle cell anemia. Physiol Meas. 2008;29(5):655–68. doi: 10.1088/0967-3334/29/5/010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Simmonds MJ, Sabapathy S, Gass GC, Marshall-Gradisnik SM, Haseler LJ, Christy RM, et al. Heart rate variability is related to impaired haemorheology in older women with type 2 diabetes. Clin Hemorheol Microcirc. 2010;46(1):57–68. doi: 10.3233/CH-2010-1333. [DOI] [PubMed] [Google Scholar]
- 15.Tracey KJ. The inflammatory reflex. Nature. 2002;420(6917):853–9. doi: 10.1038/nature01321. [DOI] [PubMed] [Google Scholar]
- 16.Ware MA, Hambleton I, Ochaya I, Serjeant GR. Day-care management of sickle cell painful crisis in Jamaica: a model applicable elsewhere? Br J Haematol. 1999;104(1):93–6. doi: 10.1046/j.1365-2141.1999.01160.x. [DOI] [PubMed] [Google Scholar]
- 17.Phillips G, Jr, Coffey B, Tran-Son-Tay R, Kinney TR, Orringer EP, Hochmuth RM. Relationship of clinical severity to packed cell rheology in sickle cell anemia. Blood. 1991;78(10):2735–9. [PubMed] [Google Scholar]
- 18.Platt OS, Thorington BD, Brambilla DJ, Milner PF, Rosse WF, Vichinsky E, et al. Pain in sickle cell disease. Rates and risk factors. N Engl J Med. 1991;325(1):11–6. doi: 10.1056/NEJM199107043250103. [DOI] [PubMed] [Google Scholar]
- 19.Baskurt OK, Boynard M, Cokelet GC, Connes P, Cooke BM, Forconi S, et al. New guidelines for hemorheological laboratory techniques. Clin Hemorheol Microcirc. 2009;42(2):75–97. doi: 10.3233/CH-2009-1202. [DOI] [PubMed] [Google Scholar]
- 20.Tripette J, Alexy T, Hardy-Dessources MD, Mougenel D, Beltan E, Chalabi T, et al. Red blood cell aggregation, aggregate strength and oxygen transport potential of blood are abnormal in both homozygous sickle cell anemia and sickle-hemoglobin C disease. Haematologica. 2009;94(8):1060–5. doi: 10.3324/haematol.2008.005371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Dworkis DA, Klings ES, Solovieff N, Li G, Milton JN, Hartley SW, et al. Severe sickle cell anemia is associated with increased plasma levels of TNF-R1 and VCAM-1. Am J Hematol. 2011;86(2):220–3. doi: 10.1002/ajh.21928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lee SP, Ataga KI, Orringer EP, Phillips DR, Parise LV. Biologically active CD40 ligand is elevated in sickle cell anemia: potential role for platelet-mediated inflammation. Arterioscler Thromb Vasc Biol. 2006;26(7):1626–31. doi: 10.1161/01.ATV.0000220374.00602.a2. [DOI] [PubMed] [Google Scholar]
- 23.Naprawa JT, Bonsu BK, Goodman DG, Ranalli MA. Serum biomarkers for identifying acute chest syndrome among patients who have sickle cell disease and present to the emergency department. Pediatrics. 2005;116(3):e420–5. doi: 10.1542/peds.2004-2107. [DOI] [PubMed] [Google Scholar]
- 24.Fortrat JO, Formet C, Frutoso J, Gharib C. Even slight movements disturb analysis of cardiovascular dynamics. Am J Physiol. 1999;277(1 Pt 2):H261–7. doi: 10.1152/ajpheart.1999.277.1.H261. [DOI] [PubMed] [Google Scholar]
- 25.Hadase M, Azuma A, Zen K, Asada S, Kawasaki T, Kamitani T, et al. Very low frequency power of heart rate variability is a powerful predictor of clinical prognosis in patients with congestive heart failure. Circ J. 2004;68(4):343–7. doi: 10.1253/circj.68.343. [DOI] [PubMed] [Google Scholar]
- 26.Baum KF, Dunn DT, Maude GH, Serjeant GR. The painful crisis of homozygous sickle cell disease. A study of the risk factors. Arch Intern Med. 1987;147(7):1231–4. [PubMed] [Google Scholar]
- 27.Stauss HM. Heart rate variability. Am J Physiol Regul Integr Comp Physiol. 2003;285(5):R927–31. doi: 10.1152/ajpregu.00452.2003. [DOI] [PubMed] [Google Scholar]
- 28.Barbato M, Curione M, Amato S, Carbone J, Briani C, Pannone V, et al. Autonomic imbalance in celiac children. Minerva Pediatr. 2010;62(4):333–8. [PubMed] [Google Scholar]
- 29.Staud R. Autonomic dysfunction in fibromyalgia syndrome: postural orthostatic tachycardia. Curr Rheumatol Rep. 2008;10(6):463–6. doi: 10.1007/s11926-008-0076-8. [DOI] [PubMed] [Google Scholar]
- 30.Di Franco M, Iannuccelli C, Alessandri C, Paradiso M, Riccieri V, Libri F, et al. Autonomic dysfunction and neuropeptide Y in fibromyalgia. Clin Exp Rheumatol. 2009;27(5 Suppl 56):S75–8. [PubMed] [Google Scholar]
- 31.Franzoni F, Galetta F, Di Muro C, Buti G, Pentimone F, Santoro G. Heart rate variability and ventricular late potentials in beta-thalassemia major. Haematologica. 2004;89(2):233–4. [PubMed] [Google Scholar]
- 32.Dogru MT, Basar MM, Yuvanc E, Simsek V, Sahin O. The relationship between serum sex steroid levels and heart rate variability parameters in males and the effect of age. Turk Kardiyol Dern Ars. 2010;38(7):459–65. [PubMed] [Google Scholar]
- 33.Umetani K, Singer DH, McCraty R, Atkinson M. Twenty-four hour time domain heart rate variability and heart rate: relations to age and gender over nine decades. J Am Coll Cardiol. 1998;31(3):593–601. doi: 10.1016/s0735-1097(97)00554-8. [DOI] [PubMed] [Google Scholar]
- 34.Pavlov VA, Tracey KJ. Controlling inflammation: the cholinergic anti-inflammatory pathway. Biochem Soc Trans. 2006;34(Pt 6):1037–40. doi: 10.1042/BST0341037. [DOI] [PubMed] [Google Scholar]
- 35.Parrish WR, Rosas-Ballina M, Gallowitsch-Puerta M, Ochani M, Ochani K, Yang LH, et al. Modulation of TNF release by choline requires alpha7 subunit nicotinic acetylcholine receptor-mediated signaling. Mol Med. 2008;14(9–10):567–74. doi: 10.2119/2008-00079.Parrish. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Borovikova LV, Ivanova S, Zhang M, Yang H, Botchkina GI, Watkins LR, et al. Vagus nerve stimulation attenuates the systemic inflammatory response to endotoxin. Nature. 2000;405(6785):458–62. doi: 10.1038/35013070. [DOI] [PubMed] [Google Scholar]
- 37.Wang H, Yu M, Ochani M, Amella CA, Tanovic M, Susarla S, et al. Nicotinic acetyl-choline receptor alpha7 subunit is an essential regulator of inflammation. Nature. 2003;421(6921):384–8. doi: 10.1038/nature01339. [DOI] [PubMed] [Google Scholar]
- 38.Connes P, Martin C, Barthelemy JC, Monchanin G, Atchou G, Forsuh A, et al. Nocturnal autonomic nervous system activity impairment in sickle cell trait carriers. Clin Physiol Funct Imaging. 2006;26(2):87–91. doi: 10.1111/j.1475-097X.2006.00655.x. [DOI] [PubMed] [Google Scholar]