Abstract
Aims
Exercise training (ExT) is a recommended adjunct to many pharmaceutical antihypertensive therapies. The effects of chronic ExT on the development of hypertension-induced renal injury remain unknown. We examined whether ExT would preserve renal hemodynamics and structure in the spontaneously hypertensive rat (SHR), and whether these effects were mediated by improved redox status and decreased inflammation. Normotensive WKY rats and SHR underwent moderate-intensity ExT for 16 weeks. One group of SHR animals was treated with hydralazine to investigate the pressure-dependent/independent effects of ExT. Acute renal clearance experiments were performed prior to sacrifice. Tissue free radical production rates were measured by electron paramagnetic resonance; gene and protein expression were measured by real time RT-PCR and Western blot or immunofluorescence, respectively. Plasma angiotensin II levels and kidney antioxidants were assessed. Training efficacy was assessed by citrate synthase activity assay in hind-limb muscle. Results: ExT delayed hypertension, prevented oxidative stress and inflammation, preserved antioxidant status, prevented an increase in circulating AngII levels, and preserved renal hemodynamics and structure in SHR. In addition, exercise-induced effects, at least, in part, were found to be pressure-independent. Innovation: This study is the first to provide mechanistic evidence for the renoprotective benefits of ExT in a model of hypertension. Our results demonstrate that initiation of ExT in susceptible patients can delay the development of hypertension and provide renoprotection at the functional and ultrastructural level. Conclusion: Chronic ExT preserves renal hemodynamics and structure in SHR; these effects are partially mediated by improved redox status and decreased inflammation. Antioxid. Redox Signal. 16, 139–152.
Introduction
Hypertension-induced renal dysfunction is a significant cause of morbidity and mortality in hypertensive patients, and remains a leading cause of end-stage renal disease in the United States (34). Current antihypertensive treatments are mostly effective in reducing the severity of hypertensive renal disease; however, its progressive clinical course underscores the need for new therapeutic approaches. The benefits of nonpharmacological interventions, such as diet and exercise, on several chronic diseases are well-established. To date, no detailed reports exist that examine the effects of exercise training (ExT) on renal redox status, renal hemodynamics, or renal structure in the hypertensive condition. Therefore, we chose to examine the effects of ExT on renal function and injury and also examined ExT-induced changes in oxidative, nitrosative, and inflammatory parameters in the spontaneously hypertensive rat (SHR), a genetically hypertensive rat model that exhibits many features of human essential hypertension. Additonally, in order to investigate whether exercise-mediated effects on hypertensive kidneys are pressure-dependent or independent, a group of SHR animals were given hydralazine for the duration of ExT.
We and others have demonstrated that proinflammatory cytokine (PIC) production and several renin–angiotensin system (RAS) components are increased in the hypertensive kidney (18, 23, 40). Reactive oxygen species (ROS) production is also increased in the renal tissue of the SHR. Further, PICs and RAS components have been found to increase ROS production (9, 36, 38), which in turn can activate various intracellular signaling pathways, including that of the transcription factor nuclear factor-kappa B (NF-κB) (18). Activation of NF-κB induces transcription of PIC genes, leading to further increases in ROS production and fostering a cyclic positive feedback mechanism, thereby accelerating the progression of hypertension and its associated renal changes.
Innovation.
Exercise is currently recommended as an adjunct to many pharmacological therapies for several chronic diseases, and has been shown to benefit end organs, including the kidney, in hypertension. However, the mechanistic basis for the positive effects of exercise on the kidney has not yet been elucidated. This study is the first to provide mechanistic evidence that exercise training can delay the development of hypertension and resulted in structural and functional improvements in the kidneys of hypertensive rats by decreasing oxidative stress, inflammation, and renin–angiotensin system activation in the kidney itself. Here, it was found that the early initiation of ExT in a rat model of hypertension delayed the development of hypertension by preventing oxidative stress, preserving antioxidant status, and decreasing inflammation. These benefits of ExT are likely responsible for the preservation of renal hemodynamics and structure in this model. Importantly, these findings on hydralazine-treated SHR animals suggest that exercise-induced benefits in hypertensive kidneys, at least, in part, are pressure independent. Although, a direct cause-effect relationship between various parameters studied could not be established, this study is clinically significant in that it demonstrates that exercise may be a viable nonpharmacological approach to delay hypertension and preserve renal function as well as renal structure in patients that are predisposed to hypertension.
Several previous studies have investigated the effects of exercise on hypertension and kidney diseases; however, most of the studies were performed on patients or animals with established disease or the exercise was combined with other interventions (7, 8, 14, 21, 47). However, the effects of ExT on delaying or preventing the progression of hypertension-induced renal injury have not yet been elucidated. The mechanisms by which chronic ExT may affect renal function are unknown, although various mechanisms have been proposed, which include a direct lowering of blood pressure, leading to reduced peripheral vascular resistance (39) and reductions in oxidative stress (21). Here, we hypothesized that chronic ExT would preserve renal structure and function by modulating oxidative stress and inflammation in the SHR model of hypertension.
Results
Body weights
There were no significant differences in body weights between WKY and SHR groups at baseline (Table 1). However, body weights were significantly lower in both WKY and SHR exercise groups at study end when compared to their sedentary counterparts (Table 2).
Table 1.
Baseline Hemodynamic Data for Wistar-Kyoto and Spontaneously Hypertensive Rats
| WKY (n=5) | SHR (n=5) | |
|---|---|---|
| BW (g) | 193.9±1.61 | 189.4±2.83 |
| SBP (mmHg) | 120.6±3.32 | 124.7±1.99 |
| MAP (mmHg | 106.3±2.94 | 106.8±2.12 |
| GFR (ml/min/g KW) | 1.01±0.04 | 0.94±0.05 |
| RBF (ml/min/g KW) | 6.97±0.33 | 6.25±0.77 |
| RVR (mmHg/ml/min/g KW) | 11.41±1.01 | 14.89±0.51 |
Rats were obtained at 7 weeks of age.
Data are presented as mean±SEM.
BW, body weight; g, grams; GFR, glomerular filtration rate; KW, kidney weight; MAP, mean arterial pressure; mmHg, millimeters of mercury; RBF, renal blood flow; RVR, renal vascular resistance; SBP, systolic blood pressure.
Table 2.
Mean Body Weights and Hemodynamic Measurements of Rats from All Groups
| Parameter | WKY-S (n=6) | WKY-E (n=6) | SHR-S (n=6) | SHR-E (n=6) | SHR-H (n=8) |
|---|---|---|---|---|---|
| BW (g) | 372.4 ± 4.61 | 323.0 ± 7.64$ | 370.8 ± 2.63† | 314.4 ± 11.69* | 374.2 ± 10.14 |
| SBP (mmHg) | 121.1 ± 2.37 | 130.7 ± 2.36 | 189.2 ± 3.47$ | 144.8 ± 3.50* | 126.3 ± 4.22* |
| MAP (mmHg) | 93.9 ± 1.83 | 98.6 ± 2.21 | 163.4 ± 9.30$ | 120.0 ± 4.73* | 114.5 ± 3.28* |
| GFR (ml/min/g KW) | 0.90 ± 0.09 | 0.90 ± 0.05 | 0.45 ± 0.08$ | 0.73 ± 0.05* | 0.71 ± 0.11* |
| RBF (ml/min/g KW) | 8.26 ± 1.11 | 7.24 ± 0.79 | 3.58 ± 0.45$ | 7.51 ± 0.58* | 8.83 ± 2.85* |
| RVR (mmHg/ml/min/g KW) | 13.61 ± 1.61 | 16.22 ± 0.49 | 42.49 ± 7.81$ | 15.96 ± 2.84* | 19.26 ± 1.95* |
Data were obtained at conclusion of study. Data are presented as mean±SEM.
p<0.05 vs. WKY-S; *p<0.05 vs. SHR-S; †p<0.05 vs. SHR-E.
BW, body weight; g, grams; GFR, glomerular filtration rate; KW, kidney weight; MAP, mean arterial pressure; mmHg, millimeters of mercury; RBF, renal blood flow; RVR, renal vascular resistance; SBP, systolic blood pressure.
Assessment of training efficacy
Citrate synthase (CS) activity in soleus muscle was used as a marker of training efficacy. After the period of 16 weeks of exercise, the CS activity was significantly higher in SHR and WKY rats compared with their sedentary control groups, indicating the efficacy of the exercise protocol (Fig. 1). CS activity was higher in WKY rats compared with SHR both in the exercise and the sedentary group.
FIG.1.
Citrate synthase activity (nmol/min/mg of protein) in soleus muscle of sedentary or exercised SHR and WKY as measured by citrate synthase activity assay kit (n=8 per group). After the period of 16 weeks of exercise, the activity of citrate synthase in the soleus muscle was significantly higher in SHR, as well as in WKY rats compared with their sedentary control groups, indicating the efficacy of the exercise protocol. ***p<0.001.
ExT preserves renal hemodynamics in SHR
No differences in systolic blood pressure (SBP) or mean arterial pressure (MAP) were noted between WKY and SHR at baseline (Table 1); however, baseline SBP in SHR (124.7±1.99 mmHg) was slightly higher than WKY (120.6±3.32), suggesting that the BP began to increase. At study end, the pressures for SHR-E animals were significantly lower than those for SHR-S animals (Table 2). We observed significantly lowered GFR and RBF and elevated RVR values in SHR-S rats when compared to SHR-E at the study end. However, there was no significant difference in GFR, RBF, or RVR between WKY-S and WKY-E rats. Additionally, significantly higher urine albumin and lower creatinine levels were found in SHR-S than in WKY-S (Figs. 2G and 2H) and ExT resulted in reveral of urine albumin and creatinine levels in SHR.
FIG. 2.
Assessment of glomerular injury, and urinary albumin and creatinine levels. (A–E) Representative photomicrographs for scoring of glomerular injury; scale bars=50 μm. (A) Grade 0: glomerulus with no lesions; (B) Grade 1: 0%–25% of glomerular area affected; (C) Grade 2: 25%–50% of glomerular area affected; (D) Grade 3: 50%–75% of glomerular area affected; (E) Grade 4: 75%–100% of glomerular area affected. (F) Average glomerular lesion scores for each experimental group (n=5–6 per group). SHR-S rats had more glomeruli with greater degrees of injury; these changes were prevented with ExT. (G, H) Urinary albumin and creatinine levels. SHR-S in comparison with WKY-S had significantly increased levels of albumin and creatinine in urine, indicating impairment in renal function, whereas SHR-E had significantly reduced levels when compared to SHR-S. $p<0.05 vs. WKY-S; *p<0.05 vs. SHR-S; †p<0.05 vs. SHR-E. (To see this illustration in color the reader is referred to the web version of this article at www.liebertonline.com/ars).
ExT preserves glomerular morphology and structure in the SHR kidney
PAS-stained kidney sections from rats (n=5 from each group) were examined by a veterinary pathologist who was blinded to the experimental conditions. One hundred glomeruli from each section were scored. Representative photomicrographs of glomeruli for each lesion score appear in Figures 2A–2E. There was no significant difference in glomerular lesion scores for WKY or SHR at baseline (WKY score 27±1.44 and SHR score 34±2.61). SHR-S rats had significantly higher glomerular lesion scores than WKY-S and SHR-E rats at the completion of the study (Fig. 2F).
Additonally, kidney sections stained with Masson's trichrome (Fig. 3A) revealed more arterial myointimal hyperplasia, tubular degeneration, ectasia, interstitial, periglomerular and periarterial fibrosis, and glomerular parietal metaplasia in SHR-S when compared to WKY-S. Interestingly, when compared to SHR-S, SHR-E exhibited less arterial myointimal hyperplasia, tubular degeneration, ectasia, interstitial, periglomerular and periarterial fibrosis, and glomerular parietal metaplasia. There were no appreciable differences between WKY-S and WKY-E in aforementioned renal structure except presence for less tubular degeneration and ectasia in WKY-E. Exercise-induced reduction in renal fibrosis was further confirmed, at the molecular level, by assessment of collagen I protein expression. Collagen I levels in SHR-S were nearly 2-fold higher than WKY-S and exercise was found to significantly reduce Collagen I in SHR-S (Fig. 3B).
FIG. 3.
Effects of ExT on renal tissue architecture and fibrosis. (A) Assessment of kidney tissue architecture, and interstitial and perivascular fibrosis by Masson's trichrome staining (n=5–6 per group), scale bars=200 μm. In comparison to WKY-S, SHR-S have more arterial myointimal hyperplasia (arrow), more tubular degeneration and ectasia (arrowhead), more interstitial, periglomerular, and periarterial fibrosis (light blue staining areas), and more glomerular parietal metaplasia (asterisk). Whereas, when compared to SHR-S, SHR-E rats exhibited less arterial myointimal hyperplasia, less tubular degeneration and ectasia, less interstitial, periglomerular, and periarterial fibrosis, and less glomerular parietal metaplasia. In WKY, there were no appreciable differences between WKY-S and WKY-E in aforementioned renal structural features except presence of less tubular degeneration and ectasia in WKY-E in comparison with WKY-S. (B) A representative Western blot and densitometric analyses of protein expression of collagen I in renal cortex. Our results revealed increased protein levels of collagen I in SHR-S in comparison with WKY-S, and ExT resulted in significant reduction in its level in SHRs. **p<0.05; ***p<0.001. (To see this illustration in color the reader is referred to the web version of this article at www.liebertonline.com/ars).
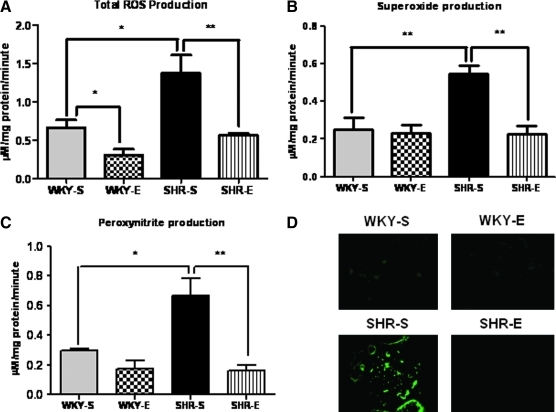
ExT decreases total ROS and superoxide production in the SHR kidney
We measured total ROS and superoxide production rates using EPR spectroscopy in cortical tissues from rats (n=6 per group) in all experimental groups. Production rates of both species were significantly decreased in cortical tissues of SHR-E rats when compared to SHR-S rats (Figs. 4A and 4B). No significant differences in total ROS or superoxide measurements were found between WKY-S and WKY-E rats or between WKY-E and SHR-E rats.
FIG. 4.
ExT attenuates production of oxidants in the kidney of SHRs. (A) Total reactive oxygen species (ROS), (B) superoxide, and (C) peroxynitrite production rates in renal cortical tissues of rats from each experimental group (n=6 per group), as determined by electron paramagnetic resonance spectroscopy. Chronic ExT prevented the increases in total ROS, superoxide, and peroxynitrite production rates normally seen in SHR. (D) Immunofluorescence (n=5–6 per group) for 3-nitrotyrosine, an indirect indicator of peroxynitrite formation. Exercise intervention prevented the increases in 3-NT normally seen in SHR. *p<0.01; **p<0.05. (To see this illustration in color the reader is referred to the web version of this article at www.liebertonline.com/ars).
ExT decreases peroxynitrite production and NT formation in the SHR kidney
We measured cortical peroxynitrite levels in all rat groups (n=6 per group) using EPR spectroscopy. We also examined expression of 3-nitrotyrosine (NT), a footprint of peroxynitrite formation, via immunofluoresence. In the SHR-E rats, peroxynitrite production rates were significantly decreased and NT expression levels were lower when compared to those of SHR-S rats (Figs. 4C and 4D, respectively).
ExT improves antioxidant status in the SHR kidney
We measured glutathione peroxidase (GPx) and reduced and oxidized glutathione (GSH and GSSG, respectively) in cortical tissues from each experimental group (n=8 per group), using commercially available kits (Figs. 5A–5D). Levels of GPx and GSH were significantly lower in SHR-S when compared to WKY-S. In addition, SHR-S exhibited significantly increased GSSG levels and reduced GSH/GSSG ratio (an important marker of cellular redox balance (43)) in comparison with WKY-S. ExT increased GPx, GSH, and decreased GSSG levels and GSH/GSSG ratio in SHRs. Furthermore, our Western blot analyses did not show any significant difference in the basal levels of renal Cu/Zn-superoxise dismutase (Cu/Zn-SOD) expression between WKY and SHR (Fig. 6). ExT significantly increased SOD and GPx levels in WKY but not in SHRs. Additionally, HO-1 levels were not significantly different among all four groups; however, SHR had slightly higher levels compared to WKY rats (Fig. 6).
FIG. 5.
ExT improves antioxidant levels in the kidney of SHRs. Levels of (A) reduced glutathione (GSH), (B) oxidized glutathione (GSSG), (C) GSH/GSSG ratio, and (D) glutathione peroxidase (GPx) as measured in cortical tissues from 8 rats from each experimental group. GSH, GSSG, and GPx levels were significantly reduced in SHR-S animals; levels were normalized in SHR-E animals. *p<0.01; **p<0 .05; ***p<0.001.
FIG. 6.
Protein levels of Cu/Zn-SOD and heme-oxygenase-1 (HO-1) in renal cortical tissue from each group (n=6 per group) as measured by Western blot analyses. There was no significant difference in the basal levels of renal Cu/Zn-SOD expression between WKY-S and SHR-S, however, WKY-E had significantly increased Cu/Zn-SOD levels compared with WKY-S. HO-1 levels were not significantly different among all four groups; however, SHR animals exhibited slightly higher levels compared to WKY rats.*p<0.01.
ExT decreases NF-κB activity and TNF-α expression in the SHR kidney
Renal cortical NF-κB p65 DNA binding activity was measured in tissues from all groups (n=8 per group). Rats from the SHR-S group had significantly higher cortical NF-κB activity than WKY-S rats (Fig. 7A). The SHR-E rats had NF-κB activity levels comparable to the WKY-S and WKY-E rats, suggesting that chronic ExT prevents the increase in cortical NF-κB activity seen in SHR. Since the proinflammatory cytokine tumor necrosis factor-alpha (TNF-α) acts through a NF-κB-dependent pathway, we measured protein and gene expression of this cytokine in the cortical tissues of animals from all experimental groups. Expression of TNF-α was significantly increased in SHR-S rats, but was completely normalized in SHR-E rats (Figs. 7B, 7C, and Supplementary Fig. S1).
FIG. 7.
Effects of ExT on NF-κB p65 DNA binding activity and TNF-α levels in the kidney. (A) DNA binding activity of NF-κB p65 in renal cortical tissues of rats from each experimental group (n=6–8 per group), as determined by ELISA. $p<0.05 vs. WKY-S; *p<0.05 vs. SHR-S; †p<0.05 vs. SHR-E. Increased NF-κB p65 DNA binding activity in SHR-S was significantly reduced by ExT. SHR-H exhibited no change in NF-κB activity in comparison with SHR-S; however, significant difference was noticed between SHR-H and SHR-E. (B) A representative Western blot showing TNF-α protein expression and (C) mRNA expression of TNF-α. TNF-α levels were significantly higher in SHR-S rats than in other groups; no significant increases were seen in these parameters in the SHR-E animals. *p<0.01.
ExT alters NO production and NOS isoform expression in the SHR kidney
Protein and gene expression levels of eNOS and iNOS were measured in all experimental groups at study completion (Figs. 8A, 8B, 8D, and Supplementary Fig. S1). Cortical nitrate/nitrite levels (indirect indicator of NO production) were also measured (Fig. 8C). Both protein and gene expression levels of eNOS, along with NO production, were decreased in SHR-S rats, while iNOS levels were significantly increased. These effects were normalized in the SHR-E animals.
FIG. 8.
Effects of ExT on renal cortical nitric oxide production. mRNA (n=8 per group) expression of (A) eNOS and (B) iNOS. (C) NO production as assessed by nitrate/nitrite measurement (n=8 per group). (D) A representative Western blot showing protein expression of eNOS and iNOS, in kidney cortical tissues from each group (n=6 per group). Expression of eNOS was very low and iNOS expression was increased in SHR-S animals, while SHR-E animals exhibited increased eNOS and normalized iNOS levels. (E) Representative immunoblots of low-temperature SDS-PAGE showing eNOS monomer and dimer formation (n=6 per group). (F) Bands were analyzed and quantified by densitometry and the eNOS dimer/monomer ratio was evaluated. SHR-S predominantly had eNOS monomer and reduced dimer/monomer ration when compared to WKY-S, whereas SHR-E had more of eNOS dimer in comparison with SHR-S, indicating exercise reduces eNOS uncoupling in SHRs. *p<0.01; **p<0.05; ***p<0.001.
ExT attenuates eNOS uncoupling in SHR
Previous studies have shown that the formation of eNOS homodimers is necessary for eNOS activity (48); therefore, we examined the eNOS dimer and monomer levels by low-temperature SDS-PAGE (LT-PAGE). In sedentary SHRs, eNOS was found to be present predominantly as the monomer; whereas, in WKY-S it was predominantly in the active homodimeric form, suggesting low eNOS activity in SHR-S (Fig. 8E). Densitometric analyses showed that total quantity of homodimeric eNOS and the ratio of dimer:monomer band intensities were more in WKY-S and WKY-E when compared to SHR-S (Fig. 8F). Interestingly, the dimer:monomer ratio was high in SHR-E in comparison with SHR-S. These results suggest that ExT attenuates eNOS uncoupling in SHR, suggesting increased eNOS activity.
ExT alters expression of RAS components in the SHR kidney
Protein and gene expression (Figs. 9A, 9B, and Supplementary Fig. S1) levels of ACE, ACE2, AT-1 receptor (AT-1R), and Mas receptor (MasR) were measured in all experimental groups at study completion. Circulating AngII levels were also measured in plasma samples from animals from all experimental groups at study end (Fig. 9C). Both protein and gene expression levels of ACE and AT-1R were higher in SHR-S rats, while ACE2 and MasR levels were significantly lower. Similarly, AngII levels were also elevated in SHR-S animals. In SHR-E animals, ACE2 and MasR expression levels were elevated, while ACE and AT-1R expression levels were decreased. Circulating AngII levels were also lower in SHR-E animals.
FIG. 9.
Effects of ExT on renal cortical RAS components. (A) Protein (n=6 per group) and (B) mRNA (n=8 per group) expression of various renin–angiotensin system components and (C) plasma ANGII levels (n=8 per group). Expression of ACE and AT-1R, and plasma AngII levels, were increased in SHR-S rats, while ACE2 and MasR were decreased. These changes were attenuated in SHR-E animals. **p<0.05; ***p<0.001.
The effects of ExT on the kidney are, in part, pressure independent
To investigate whether effects of exercise on hypertensive kidneys are pressure-dependent or pressure-independent, a group (n=8) of SHR rats (SHR-H) were treated with hydralazine for the duration of 16 weeks to clamp the BP. Although hydralazine treatment normalized MAP and SBP in SHR (Table 2), it did not appreciably alter the activity of NF-κB (Fig. 7A), the most important redox-sensitive transcription factor. Additionally, similar to SHR-E, SHR-H exhibited significantly improved GFR, RBF, and RVR when compared to SHR-S, and these values were not statistically different between SHR-H and SHR-E (Table 2). Surprisingly, however, urinary albumin levels in SHR-H were slightly increased and creatinine was reduced when compared to SHR-S, however, these values were still significantly different from SHR-E (Figs. 2G and 2H). Importantly, glomerular lesion scores in SHR-H were significantly lower in comparison with SHR-S, although these values were still higher than SHR-E (Fig. 2F). Taken together, these findings suggest that hydralazine treatment in SHR normalizes BP and moderately improves renal function but does not alters glomerular structural changes and NF-κB activity to the level of SHR-E.
Discussion
Primary (essential) hypertension remains a major cause of morbidity and mortality in Western society, and is the second leading cause of end-stage renal disease in the United States (46). The most effective way to avoid the development of hypertension-induced renal injury is to prevent hypertension or to delay its progression. Current pharmacological therapies have proven beneficial in the treatment of hypertension, but the focus has recently shifted to include nonpharmacological approaches, such as exercise, as adjunct therapies to prevent or mitigate hypertension and its end-organ effects. Here, we examined the effects of ExT on renal function and injury and assessed exercise-induced changes in oxidative, nitrosative, and inflammatory parameters in SHR. We initiated chronic ExT at 7 weeks of age, an age when SHR began to develop hypertension and alterations in renal hemodynamic indices that is shown to be preceded by proteinuria (19, 30). We also performed baseline measurements of arterial pressures and renal hemodynamic parameters in 7-week-old WKY and age-matched SHR, with no statistically significant differences found between strains for any of the parameters measured (Table 1). In addition, a group of SHR rats was given hydralazine for 16 weeks, the same length of time that the exercise group was subjected to exercise, to investigate whether exercise-mediated effects are pressure-dependent/independent. The first and foremost finding of this study was that besides significantly reducing SBP and MAP in SHRs, chronic regular ExT preserved renal structure and renal hemodynamics in these animals. Second, moderate-intensity ExT prevents renal oxidative stress and inflammation, improves renal antioxidant defense, and modulates both intrarenal and extrarenal renin–angiotensin system (RAS) components in SHR. Third, ExT attenuates elevated NFκB–DNA binding activity in hypertensive rats. Finally, this study revealed that hydralazine treatment, although able to normalize MAP and SBP in SHR, did not appreciably alter NF-κB activity (Fig. 7A and Table 2). Taken together, these findings clearly suggest that chronic regular exercise reduces BP, causes structural and functional improvements in hypertensive kidneys, and improves RAS components, possibly via reduction in oxidative stress and NF-κB activity in SHRs. In addition, exercise-induced effects in this study, at least in part, were found to be pressure independent.
Our findings revealed that hydralazine treatment in SHR normalizes BP and moderately improves renal function but does not alters glomerular structural changes and NF-κB activity to the level of SHR-E, indicating that factors other than reduction in BP may have contributed to exercise-induced renal changes in SHRs. It is apparent from hydralazine-induced improvements in renal functional parameters that effects of exercise on kidneys of SHR could, partially, be pressure dependent. However, in this study, hydralazine treatment was started from the beginning of the study (at 7 weeks of age) when SHR animals began to develop hypertension and hence, this chronic treatment may have resulted in renal function improvement. In addition, being an antihypertensive drug, hydralazine not only clamps the pressure but also has several other effects on heart, vasculature, and kidney (4, 11). Therefore, it is plausible to suggest that using a compound other than hydralazine that does not have significant effects on renal function would be more appropriate to prove or rule out the pressure-independent effects of exercise in hypertensive kidneys. Nevertheless, lower glomerular scores and albuminuria, higher urinary creatinine levels, and attenuated NF-κB activity in SHR-E in comparison with SHR-H provide evidence that chronic exercise exerts its effects on hypertensive kidneys, at least, in part, might be pressure independent.
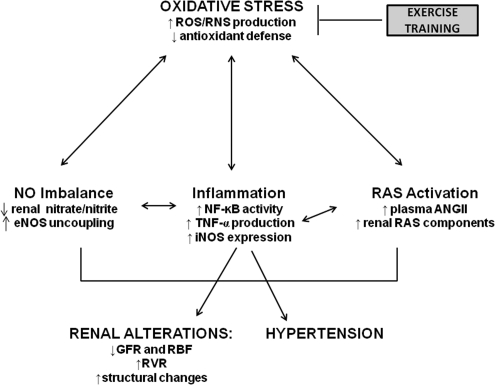
Oxidative stress is characteristic of the adult SHR, and has been shown to precede the development of hypertension in this rat strain by several weeks (31, 37). Conversely, hypertension has been shown to cause oxidative stress in the kidney (51). This self-perpetuating cycle, if left unchecked, can lead to progressive renal disease. Several key mediators of renal oxidative stress have been identified, including PIC and the effector peptide of the renin-angiotensin system, AngII; both can cause activation of the key redox-sensitive transcription factor, NF-κB, and increase production of ROS and reactive nitrogen species (RNS), such as superoxide and peroxynitrite, respectively. These ROS/RNS themselves can increase NF-κB activity, leading to further oxidative/nitrosative insult and RAS activation, which perpetuates this vicious positive feedback cycle and accelerates hypertension-induced renal damage (18). A schematic representation of this cycle appears in Figure 10.
FIG. 10.
Schematic depiction of the interactions of oxidative stress, inflammation, RAS activation, and NO imbalance in the pathogenesis of hypertensive renal injury, and the effects of exercise training on this vicious positive feedback cycle.
NF-κB can induce and respond to oxidative stress; when activated, NF-κB can activate the promoters of two proinflammatory molecules: TNF-α (12) and iNOS (56). A key finding in the present study is that NF-κB p65 DNA binding activity, along with cortical protein and gene expression levels of iNOS and TNF-α, did not increase in the SHR kidney when ExT was initiated prior to development of hypertension, while all three parameters increased significantly in sedentary SHR. Conversely, gene and protein expression levels of eNOS were significantly higher in SHR-E animals than in SHR-S animals. Further, chronic ExT not only prevented the increases normally seen in cortical total ROS, superoxide, and peroxynitrite production rates in SHR but also significantly upregulated the eNOS protein levels. Our current finding that decreased oxidative stress is associated with decreased TNF-α expression and decreased NF-κB activity in SHR-E rats raises the possibility that decreased PICs might be responsible for the exercise-induced decrease in oxidative stress in SHR.
Oxidative stress, by definition, involves an excess of free radicals; in the case of renal oxidative stress during hypertension, the predominant free radical is superoxide, which is mostly produced by NADPH oxidases (13). We have previously demonstrated an upregulation in NADPH oxidase expression and activity in the cortical tissues of SHR; this was associated with a decline in renal hemodynamic parameters and with increased arterial pressure (18). In addition to its direct detrimental effects, superoxide can interact with NO to form the highly cytotoxic peroxynitrite radical. Peroxynitrite can then react with tyrosine residues in various proteins to generate NT (6,25). Although NT can be formed from reactions that do not include NO (17), the contribution of these reactions to total tissue NT abundance is minimal; thus, tissue NT abundance is largely considered a function of the interaction between superoxide and NO (6). In this study, the increase in arterial blood pressure and alterations in renal hemodynamics seen in sedentary SHR were accompanied by cortical accumulation of NT and increased tissue production rates of total ROS, superoxide, and peroxynitrite. These findings were also accompanied by a marked reduction in renal cortical NO metabolites, which is suggestive of diminished NO bioavailability, likely resulting from an enhancement of NO inactivation by superoxide. Given the current findings that SHR-S had higher iNOS levels than WKY-S and because iNOS can itself increase superoxide production (54, 55), it is highly likely that superoxide-mediated degradation of NO may have contributed to reduced nitrate/nitrite levels in SHR-S. However, concomitant downregulation of eNOS may have also contributed to reduced nitrate/nitrite in SHRs. Regardless, reduced eNOS accompanied by elevated free radical production and decreased nitrate/nitrite levels in SHR-S suggest uncoupling of eNOS. In support of this statement, our densitometric analyses of LT-PAGE clearly showed that total quantity of homodimeric eNOS and the ratio of dimer:monomer were significantly lowered in SHR-S when compared to WKY-S; whereas,ExT in SHR resulted in higher eNOS dimer:monomer ratio, suggesting that chronic exercise attenuates eNOS uncoupling in SHR. Since tetrahydrobiopterin (BH4) is an essential cofactor for eNOS activity and has been shown to be required for promoting and stabilizing NOS protein monomers into the active homodimeric form (45, 50), the eNOS uncoupling observed in SHR-S could be due to a decrease in BH4 levels. Nonetheless, exercise-induced reduction in free radical production along with upregulation of dimeric eNOS may have contributed to renoprotective effects of exercise in SHR as observed in the present study.
In addition to increased free radical production, SHR-S exhibited decreased GPx levels and GSH/GSSG ratio in renal cortex, which is indicative of deranged antioxidant status. Lee and colleagues recently found similar impairments in the glutathione system in SHR from the age of 8 weeks (32). However, we did not observe any significant difference in the basal levels of renal Cu/Zn-SOD between WKY and SHR. This is not unexpected because not all enzymes upregulate their expression in response to an increase in substrate. Consistent with our findings, previous reports have also shown no difference in renal cortical SOD levels between normotensive and hypertensive rats (1, 20, 57). Similarly, previous studies have shown that SOD levels and activity differed in the liver but not in the myocardium of sedentary and exercising WKY and SHR (53). Therefore, the discrepancies in these results could be due to the variations in tissue types studied. Nevertheless, increased superoxide production in absence of any increases in SOD levels in SHR further highlight the point that oxidative stress caused by overproduction of oxidants and impaired antioxidant defense system exist in SHR and may be responsible for observed renal damage in these rats. Unlike SOD, HO-1 levels were not different among all four groups; however, SHRs had slightly higher levels than WKY. HO-1 is known to be induced by various stressors (eg., hypoxia, poisons, and chemicals); therefore, it is likely that increased HO-1 in SHRs could be a compensatory mechanism in response to increased oxidative stress. Interestingly, however, we observed increases in SOD and GPx levels in exercised WKY when compared to WKY-S. Similar results have also been previously reported (5, 27, 29). These findings explain absence of exercise-induced oxidative stress in trained animals or individuals. Our results suggest that increased free radical production accompanied with impaired antioxidant defense system in SHR continues through age 24 weeks and that ExT prevents these alterations in antioxidant status.
In exercised SHR, there was a delayed, moderate increase in arterial pressure and a normalization of cortical NO metabolites. Further, SHR-E exhibited no evidence of cortical NT accumulation and no appreciable increases in tissue production rates of total ROS, superoxide, or peroxynitrite. These results were associated with a preservation of renal hemodynamics and renal antioxidant status in these animals, and with alterations in cortical gene and protein expression levels of iNOS and eNOS (29). Our results are in line with a previous report from our laboratory, where the same ExT regimen resulted in preserved cardiac function, decreased ROS formation, decreased iNOS expression, and increased NO metabolites in the SHR myocardium (2). Taken together, these observations suggest that chronic ExT can decrease the severity of hypertension and its associated alterations in renal hemodynamics in SHR by ameliorating the renal oxidative stress known to exist in these animals. These beneficial effects seem to involve preservation of redox status and an improvement in NO bioavailability.
Activation of the RAS and the resulting AngII-induced pressor response are key mediators of renal damage (26, 42), and have renal pro-oxidant and proinflammatory effects which negatively alter renal hemodynamic parameters (26, 52). For example, AngII can antagonize the effects of NO (59), potentiate superoxide production (52), and activate NF-κB (33); these actions of AngII represent another arm of the vicious positive feedback cycle involved in hypertension-induced renal injury. We measured protein and gene expression levels of several RAS components (AT-1R, ACE, ACE2, and MasR) and circulating AngII levels to assess the possible involvement of the RAS in the renoprotection afforded by ExT in this study. We found downregulation of AT-1R and ACE and decreased plasma AngII, with concomitant upregulation of ACE2 and MasR, in exercised SHR when compared to SHR-S animals. These findings suggest that exercise-induced improvement in renal function may be attributed, at least in part, to improved cortical RAS components. The exact mechanisms by which improvement in RAS may have contributed to exercise-mediated benefits are still unclear. However, RAS-mediated upregulation of renal COX2 has been shown to play an important role in pathogenesis of hypertensive renal injury (28). Therefore, the possibility that downregulation of renal COX2 mediated by reduced vasoconstrictor RAS may have contributed to exercise-induced improvement in renal function and BP, cannot be ignored. Nevertheless, our results indicate a role for decreased RAS activation in the renoprotective beneficial effects of ExT in SHR.
The normal parietal tissue of Bowman's capsule consists of simple squamous epithelium (24). Tubular metaplasia of Bowman's capsule has been well-characterized in SHR and in aging male Sprague-Dawley rats (10, 22, 24); however, the exact cause of such metaplasia remains uncertain. Metaplasia usually occurs in response to chronic inflammation and allows for substitution of cells that are better able to survive under circumstances in which a more fragile cell type might succumb. Although the normal squamous epithelium that lines Bowman's capsule is robust and more resistant to many insults than is cuboidal epithelium, local changes in cytokine, growth factor, or extracellular matrix components arising from either increased glomerular pressure or proteinuria may favor metaplasia to renal tubular epithelium (49). In this study, a higher incidence of glomerular parietal metaplasia, arterial myointimal hyperplasia, and tubular degeneration were seen in SHR-S than any other group whereas the incidence of aforementioned renal structural changes in SHR-E were similar to that of WKY-S and WKY-E animals. These differences were not present in WKY or SHR that were sacrificed at 7 weeks of age, suggesting that the metaplasia seen in SHR-S glomeruli at study completion may indeed be the result of increased pressure and increased inflammation, as previously suggested (24). Besides the abovementioned changes, ExT resulted in reduced periglomerular and periarterial fibrosis as well as collagen I levels in SHRs. It has previously been shown that ACE2 upregulation inhibits Ang II- induced tubulointerstitial fibrosis; whereas the loss of ACE2 enhances Ang II-mediated increase in collagen type I expression and fibrosis (58). Therefore, in light of these reports and our current findings that exercise reduces AngII levels and upregulates ACE2 in SHRs, it is plausible to suggest that exercise-mediated improvement in RAS may have contributed to observed attenuation of renal fibrosis in SHR-E. The absence of significant structural alterations in SHR-E kidneys was associated with improved renal hemodynamics and decreased inflammation, reduced proteinuria, while the increased incidence of metaplasia in SHR-S kidneys was associated with a decline in renal hemodynamics, increased proteinuria, and an increase in inflammation. These results suggest that ExT has ability to improve renal function and structure at physiological and molecular level, possibly through reduction in oxidative stress and attenuated inflammatory cytokines.
We have previously reported an association of PICs and their transcription factor, NF-κB, with oxidative stress and hemodynamic alterations in the adult SHR kidney (18). Given the results from that report and our current results, it is plausible to suggest that the lower NF-κB activity levels seen in SHR-E animals may be attributable to reductions of PIC and ROS production and of RAS activation, and thus, lead to disruption of the positive feedback cycle involved with hypertension-induced renal dysfunction. This study is the first to provide mechanistic evidence surrounding the effects of chronic exercise on NF-κB activity, oxidative stress, hemodynamics, and structure in the SHR kidney. Our results suggest a major role for exercise training in modulating hypertensive renal injury via decreases in inflammation, oxidative stress, and RAS activation. This study also provides evidence that exercise-induced effects in hypertensive kidneys are, in part, pressure-independent as indicated by hydralazine treatment in SHR. The present study clearly suggests that chronic, moderate-intensity ExT may be a cost-effective nonpharmacological approach to preventing hypertension and preserving renal hemodynamics in susceptible patients.
Materials and Methods
For expanded materials and methods, please see the accompanying data supplement. (Supplementary information is available online at www.liebertonline.com/ars.). All procedures in this study were approved by the Louisiana State University Institutional Animal Care and Use Committee and were performed in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Animals and experimental design
Seven-week-old male normotensive Wistar-Kyoto (WKY) and spontaneously hypertensive rats (SHR), from colonies maintained at the Louisiana State University School of Veterinary Medicine, were used in this study. The original source of the breeder animals was Harlan (Indianapolis, IN). Prior to the initiation of any experimental protocols, five WKY and five SHR were subjected to acute renal clearance experiments as previously published, to obtain baseline values for glomerular filtration rate (GFR) and renal plasma flow (RPF) (18); immediately following clearance experiments, animals were euthanized by thiobutabarbital overdose.
Four groups of eight animals each were used for the experimental protocol: sedentary WKY (WKY-S), sedentary SHR (SHR-S), exercise WKY (WKY-E), and exercise SHR (SHR-E). Animals in exercise groups were subjected to moderate-intensity exercise on a motor-driven treadmill for a period of 16 weeks (2). At the end of the study, six animals from each experimental group were subjected to acute renal clearance experiments (18). Animals in the exercise groups were sacrificed 24 hours after the last exercise session (at age 24 weeks); sedentary animals were sacrificed at the same age. Animals were euthanized immediately after clearance experiments, and kidneys and soleus muscle were collected for further assessment. One kidney from each animal was sectioned and subjected to immunofluorescence studies and histopathological examination. Cortical tissue was separated from the other kidney and stored for later analyses. We performed the following experimental procedures as previously described: real-time RT-PCR, Western blot, electron paramagnetic resonance (EPR) studies, NF-κB p65 DNA binding assay, immunofluorescence, antioxidant assays, and statistical analysis.
In addition, to investigate whether effects of exercise on hypertensive kidneys are pressure-dependent or pressure-independent, a group (n=8) of SHR rats (SHR-H) was treated with hydralazine (25 mg/kg/day) in drinking water daily for 16 weeks. Subsequently, the SHR-H animals were anesthetized, subjected to acute renal experiments, euthanized, and kidneys were collected for NF-κB p65 DNA binding assay and histopathological studies.
Renal clearance experiments
Acute renal clearance experiments were performed according to previously published methods (18).
Glomerular injury scoring
A semi-quantitative glomerular lesion scoring method was used, which was based upon previously published methods for glomerulosclerosis scoring (44) and expanded to include the following criteria: tubular epithelial metaplasia of Bowman's capsule, glomerulosclerosis, mesangial proliferation, and glomerular capillary basement membrane thickening.
Measurement of cortical nitrate/nitrite production
The reaction of nitric oxide (NO) with oxygen can cause the oxidation of NO, leading to nitrate/nitrite production; therefore, levels of nitrate/nitrite are considered an indirect indicator of NO production. Nitrate/nitrite levels were measured in renal cortical tissues of animals from all experimental groups with a commercially available colorimetric assay kit (Cayman Chemical; Ann Arbor, MI), as previously described (2).
Measurement of cortical NF-κB p65 DNA binding activity
Activated NF-κB translocates to the nucleus, where the p65 subunit binds to DNA to promote transcription. The binding activity of free NF-κB p65 in nuclear extracts was assessed with a NF-κB p65 TransAM ELISA kit (Active Motif, Carlsbad, CA) as per manufacturer's instructions, as previously described (35).
Measurement of cortical oxidized/reduced glutathione and glutathione peroxidase levels
Antioxidant status was assessed in renal cortical tissues of animals from all groups by measurement of reduced and oxidized glutathione and glutathione peroxidase, with commercially available colorimetric assay kits (Cayman Chemical, Ann Arbor, MI), as previously described (36).
Analysis of mRNA expression by real-time PCR
Total RNA isolation from renal cortical tissues, cDNA synthesis and real-time RT-PCR were performed as previously described in detail (2, 18).
Analysis of protein expression by Western blotting
Protein expression in renal cortical tissues was determined by Western blot analysis, as described previously in detail (2, 18).
Low-temperature SDS-PAGE (LT-PAGE) for the measurement of eNOS dimer/monomer
To investigate eNOS homodimer formation in kidney cortex, low-temperature SDS-PAGE was employed, as described previously (41, 60).
Electron paramagnetic resonance spectroscopy
Total ROS, superoxide, and peroxynitrite were measured in renal cortical tissues using electron paramagnetic resonance spectroscopy, as described previously in detail (15, 16, 18, 36).
Immunofluorescence
Immunofluorescence detection of 3-nitrotyrosine (Cayman Chemical; 1:100 dilution) in paraffin-embedded kidney sections was conducted as previously described (18).
Statistical analyses
All data are presented as means±SEM. For baseline analyses between WKY and SHR, unpaired t-tests were used. At study completion, unpaired t-tests were used to execute planned comparisons between WKY-S and SHR-S; WKY-E and SHR-E; and SHR-S and SHR-E groups, using Graph Pad Prism software (version 5.0). In all cases, results were considered significant when P<0.05.
Supplementary Material
Abbreviations Used
- ACE
angiotensin converting enzyme
- AngII
angiotensin II
- AT-1R
angiotensin type 1 receptor
- BSA
bovine serum albumin
- eNOS
endothelial nitric oxide synthase
- EPR
electron paramagnetic resonance
- ExT
exercise training
- GFR
glomerular filtration rate
- GPx
glutathione peroxidase
- GSH
reduced glutathione
- GSSG
oxidized glutathione
- Hct
hematocrit
- iNOS
inducible nitric oxide synthase
- MAP
mean arterial pressure
- NF-κB
nuclear factor-kappa B
- NO
nitric oxide
- NT
3-nitrotyrosine
- PAH
para-aminohippurate
- PIC
proinflammatory cytokine
- RAS
renin–angiotensin system
- RBF
renal blood flow
- RNS
reactive nitrogen species
- ROS
reactive oxygen species
- RPF
renal plasma flow
- RVR
renal vascular resistance
- SBP
systolic blood pressure
- SHR
spontaneously hypertensive rat
- SHR-E
exercise SHR
- SHR-S
sedentary SHR
- TNF-α
tumor necrosis factor-alpha
- WKY
Wistar-Kyoto rat
- WKY-E
exercise WKY
- WKY-S
sedentary WKY
Acknowledgments
The authors gratefully acknowledge Sherry Ring for processing, sectioning, and staining tissue sections. This work was supported by NIH National Heart, Lung, and Blood Institute grant HL-80544-04 (J.F.).
Author Disclosure Statement
No competing financial interests exist.
References
- 1.Adler S. Huang H. Oxidant stress in kidneys of spontaneously hypertensive rats involves both oxidase overexpression and loss of extracellular superoxide dismutase. Am J Physiol Renal Physiol. 2004;287:F907–913. doi: 10.1152/ajprenal.00060.2004. [DOI] [PubMed] [Google Scholar]
- 2.Agarwal D. Haque M. Sriramula S. Mariappan N. Pariaut R. Francis J. Role of proinflammatory cytokines and redox homeostasis in exercise-induced delayed progression of hypertension in spontaneously hypertensive rats. Hypertension. 2009;54:1393–1400. doi: 10.1161/HYPERTENSIONAHA.109.135459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.This reference has been deleted
- 4.Anderson PG. Bishop SP. Digerness SB. Vascular remodeling and improvement of coronary reserve after hydralazine treatment in spontaneously hypertensive rats. Circ Res. 1989;64:1127–1136. doi: 10.1161/01.res.64.6.1127. [DOI] [PubMed] [Google Scholar]
- 5.Avula CP. Fernandes G. Modulation of antioxidant enzymes and apoptosis in mice by dietary lipids and treadmill exercise. J Clin Immunol. 1999;19:35–44. doi: 10.1023/a:1020562518071. [DOI] [PubMed] [Google Scholar]
- 6.Beckman JS. Koppenol WH. Nitric oxide, superoxide, and peroxynitrite: The good, the bad, and ugly. Am J Physiol Cell Physiol. 1996;271:C1424–C1437. doi: 10.1152/ajpcell.1996.271.5.C1424. [DOI] [PubMed] [Google Scholar]
- 7.Bertagnolli M. Schenkel PC. Campos C. Mostarda CT. Casarini DE. Bello-Klein A. Irigoyen MC. Rigatto K. Exercise training reduces sympathetic modulation on cardiovascular system and cardiac oxidative stress in spontaneously hypertensive rats. Am J Hypertens. 2008;21:1188–1193. doi: 10.1038/ajh.2008.270. [DOI] [PubMed] [Google Scholar]
- 8.Boissiere J. Eder V. Machet MC. Courteix D. Bonnet P. Moderate exercise training does not worsen left ventricle remodeling and function in untreated severe hypertensive rats. J Appl Physiol. 2008;104:321–327. doi: 10.1152/japplphysiol.00442.2007. [DOI] [PubMed] [Google Scholar]
- 9.Cai H. Harrison DG. Endothelial dysfunction in cardiovascular diseases: The role of oxidant stress. Circ Res. 2000;87:840–844. doi: 10.1161/01.res.87.10.840. [DOI] [PubMed] [Google Scholar]
- 10.Castelletto L. Goya RG. Sex-related incidence of tubular metaplasia in Bowman's capsule of aging rats. Virchows Arch B Cell Pathol Incl Mol Pathol. 1990;59:79–82. doi: 10.1007/BF02899390. [DOI] [PubMed] [Google Scholar]
- 11.Cooper ME. Allen TJ. O'Brien RC. Papazoglou D. Clarke BE. Jerums G. Doyle AE. Nephropathy in model combining genetic hypertension with experimental diabetes. Enalapril versus hydralazine and metoprolol therapy. Diabetes. 1990;39:1575–1579. doi: 10.2337/diab.39.12.1575. [DOI] [PubMed] [Google Scholar]
- 12.Coward WR. Okayama Y. Sagara H. Wilson SJ. Holgate ST. Church MK. NF-κB and TNF-α: A positive autocrine loop in human lung mast cells? J Immunol. 2002;169:5287–5293. doi: 10.4049/jimmunol.169.9.5287. [DOI] [PubMed] [Google Scholar]
- 13.Datla SR. Griendling KK. Reactive oxygen species, NADPH oxidases, and hypertension. Hypertension. 2010;56:325–330. doi: 10.1161/HYPERTENSIONAHA.109.142422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.De Angelis K. Ogawa T. Sanches ÍC. Vianna Rigatto K. Krieger EM. rigoyen M-C. Impairment on cardiac output and blood flow adjustments to exercise in L-NAME-induced hypertensive rats. J Cardiovasc Pharmacol. 2006;47:371–376. doi: 10.1097/01.fjc.0000210068.70076.e2. [DOI] [PubMed] [Google Scholar]
- 15.Ebenezer PJ. Mariappan N. Elks CM. Haque M. Francis J. Diet-induced renal changes in Zucker rats are ameliorated by the superoxide dismutase mimetic TEMPOL. Obesity (Silver Spring) 2009;17:1994–2002. doi: 10.1038/oby.2009.137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ebenezer PJ. Mariappan N. Elks CM. Haque M. Soltani Z. Reisin E. Francis J. Effects of pyrrolidine dithiocarbamate on high-fat diet-induced metabolic and renal alterations in rats. Life Sci. 2009;85:357–364. doi: 10.1016/j.lfs.2009.06.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Eiserich JP. Butler J. van der Vliet A. Cross CE. Halliwell B. Nitric oxide rapidly scavenges tyrosine and tryptophan radicals. Biochem J. 1995;310:745–749. doi: 10.1042/bj3100745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Elks CM. Mariappan N. Haque M. Guggilam A. Majid DS. Francis J. Chronic NF-kB blockade reduces cytosolic and mitochondrial oxidative stress and attenuates renal injury and hypertension in SHR. Am J Physiol Renal Physiol. 2009;296:F298–305. doi: 10.1152/ajprenal.90628.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Feld LG. Van Liew JB. Brentjens JR. Boylan JW. Renal lesions and proteinuria in the spontaneously hypertensive rat made normotensive by treatment. Kidney Int. 1981;20:606–614. doi: 10.1038/ki.1981.183. [DOI] [PubMed] [Google Scholar]
- 20.Fortepiani LA. Reckelhoff JF. Increasing oxidative stress with molsidomine increases blood pressure in genetically hypertensive rats but not normotensive controls. Am J Physiol Regul Integr Comp Physiol. 2005;289:R763–770. doi: 10.1152/ajpregu.00526.2004. [DOI] [PubMed] [Google Scholar]
- 21.George L. Lokhandwala MF. Asghar M. Exercise activates redox-sensitive transcription factors and restores renal D1 receptor function in old rats. Am J Physiol Renal Physiol. 2009;297:F1174–1180. doi: 10.1152/ajprenal.00397.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Goya RG. Castelletto L. Sosa YE. Plasma levels of growth hormone correlate with the severity of pathologic changes in the renal structure of aging rats. Lab Invest. 1991;64:29–34. [PubMed] [Google Scholar]
- 23.Granger JP. An emerging role for inflammatory cytokines in hypertension. Am J Physiol Heart Circ Physiol. 2006;290:H923–924. doi: 10.1152/ajpheart.01278.2005. [DOI] [PubMed] [Google Scholar]
- 24.Haensly WE. Granger HJ. Morris AC. Cioffe C. Proximal-tubule-like epithelium in Bowman's capsule in spontaneously hypertensive rats. Changes with age. Am J Pathol. 1982;107:92–97. [PMC free article] [PubMed] [Google Scholar]
- 25.Halliwell B. What nitrates tyrosine? Is nitrotyrosine specific as a biomarker of peroxynitrite formation in vivo? FEBS Letters. 1997;411:157–160. doi: 10.1016/s0014-5793(97)00469-9. [DOI] [PubMed] [Google Scholar]
- 26.Hisada Y. Sugaya T. Yamanouchi M. Uchida H. Fujimura H. Sakurai H. Fukamizu A. Murakami K. Angiotensin II plays a pathogenic role in immune-mediated renal injury in mice. J Clin Invest. 1999;103:627–635. doi: 10.1172/JCI2454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hong H. Johnson P. Antioxidant enzyme activities and lipid peroxidation levels in exercised and hypertensive rat tissues. Int J Biochem Cell Biol. 1995;27:923–931. doi: 10.1016/1357-2725(95)00057-v. [DOI] [PubMed] [Google Scholar]
- 28.Jaimes EA. Zhou MS. Pearse DD. Puzis L. Raij L. Upregulation of cortical COX-2 in salt-sensitive hypertension: Role of angiotensin II and reactive oxygen species. Am J Physiol Renal Physiol. 2008;294:F385–392. doi: 10.1152/ajprenal.00302.2007. [DOI] [PubMed] [Google Scholar]
- 29.Johnson P. Antioxidant enzyme expression in health and disease: Effects of exercise and hypertension. Comp Biochem Physiol C Toxicol Pharmacol. 2002;133:493–505. doi: 10.1016/s1532-0456(02)00120-5. [DOI] [PubMed] [Google Scholar]
- 30.Komatsu K. Frohlich ED. Ono H. Ono Y. Numabe A. Willis GW. Glomerular dynamics and morphology of aged spontaneously hypertensive rats. Effects of angiotensin-converting enzyme inhibition. Hypertension. 1995;25:207–213. doi: 10.1161/01.hyp.25.2.207. [DOI] [PubMed] [Google Scholar]
- 31.Lazaro A. Gallego-Delgado J. Justo P. Esteban V. Osende J. Mezzano S. Ortiz A. Egido J. Long-term blood pressure control prevents oxidative renal injury. Antioxid Redox Signal. 2005;7:1285–1293. doi: 10.1089/ars.2005.7.1285. [DOI] [PubMed] [Google Scholar]
- 32.Lee S. Arunkumar S. Sirajudeen K. Singh H. Glutathione system in young spontaneously hypertensive rats. J Physiol Biochem. 2010;66:321–327. doi: 10.1007/s13105-010-0038-2. [DOI] [PubMed] [Google Scholar]
- 33.Li XC. Zhuo JL. Nuclear factor-[kappa]B as a hormonal intracellular signaling molecule: Focus on angiotensin II-induced cardiovascular and renal injury. Curr Opin Nephrol Hypertension. 2008;17:37–43. doi: 10.1097/MNH.0b013e3282f2903c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lloyd-Jones D. Adams R. Carnethon M. De Simone G. Ferguson TB. Flegal K. Ford E. Furie K. Go A. Greenlund K. Haase N. Hailpern S. Ho M. Howard V. Kissela B. Kittner S. Lackland D. Lisabeth L. Marelli A. McDermott M. Meigs J. Mozaffarian D. Nichol G. O'Donnell C. Roger V. Rosamond W. Sacco R. Sorlie P. Stafford R. Steinberger J. Thom T. Wasserthiel-Smoller S. Wong N. Wylie-Rosett J. Hong Y. Heart Disease and Stroke Statistics–2009 Update: A Report From the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation. 2009;119:e21–181. doi: 10.1161/CIRCULATIONAHA.108.191261. [DOI] [PubMed] [Google Scholar]
- 35.Mariappan N. Elks CM. Sriramula S. Guggilam A. Liu Z. Borkhsenious O. Francis J. NF-κB-induced oxidative stress contributes to mitochondrial and cardiac dysfunction in type II diabetes. Cardiovasc Res. 2010;85:473–483. doi: 10.1093/cvr/cvp305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Mariappan N. Soorappan RN. Haque M. Sriramula S. Francis J. TNF-alpha-induced mitochondrial oxidative stress and cardiac dysfunction: Restoration by superoxide dismutase mimetic Tempol. Am J Physiol Heart Circ Physiol. 2007;293:H2726–2737. doi: 10.1152/ajpheart.00376.2007. [DOI] [PubMed] [Google Scholar]
- 37.Nabha L. Garbern JC. Buller CL. Charpie JR. Vascular oxidative stress precedes high blood pressure in spontaneously hypertensive rats. Clin Exp Hypertension. 2005;27:71–82. doi: 10.1081/ceh-200044267. [DOI] [PubMed] [Google Scholar]
- 38.Neri M. Cerretani D. Fiaschi AI. Laghi PF. Lazzerini PE. Maffione AB. Micheli L. Bruni G. Nencini C. Giorgi G. D'Errico S. Fiore C. Pomara C. Riezzo I. Turillazzi E. Fineschi V. Correlation between cardiac oxidative stress and myocardial pathology due to acute and chronic norepinephrine administration in rats. J Cell Mol Med. 2007;11:156–170. doi: 10.1111/j.1582-4934.2007.00009.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.O'Rourke MF. Hashimoto J. Mechanical factors in arterial aging: A clinical perspective. J Am Coll Cardiol. 2007;50:1–13. doi: 10.1016/j.jacc.2006.12.050. [DOI] [PubMed] [Google Scholar]
- 40.Peeters AC. Netea MG. Janssen MC. Kullberg BJ. Van der Meer JW. Thien T. Pro-inflammatory cytokines in patients with essential hypertension. Eur J Clin Invest. 2001;31:31–36. doi: 10.1046/j.1365-2362.2001.00743.x. [DOI] [PubMed] [Google Scholar]
- 41.Pfeiffer S. Gorren AC. Pitters E. Schmidt K. Werner ER. Mayer B. Allosteric modulation of rat brain nitric oxide synthase by the pterin-site enzyme inhibitor 4-aminotetrahydrobiopterin. Biochem J. 1997;328:349–352. doi: 10.1042/bj3280349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Polichnowski AJ. Jin C. Yang C. Cowley AW., Jr Role of renal perfusion pressure versus angiotensin II on renal oxidative stress in angiotensin II-induced hypertensive rats. Hypertension. 2010;55:1425–1430. doi: 10.1161/HYPERTENSIONAHA.110.151332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Powers SK. Jackson MJ. Exercise-induced oxidative stress: Cellular mechanisms and impact on muscle force production. Physiol Rev. 2008;88:1243–1276. doi: 10.1152/physrev.00031.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Raij L. Azar S. Keane W. Mesangial immune injury, hypertension, and progressive glomerular damage in Dahl rats. Kidney Int. 1984;26:137–143. doi: 10.1038/ki.1984.147. [DOI] [PubMed] [Google Scholar]
- 45.Reif A. Frohlich LG. Kotsonis P. Frey A. Bommel HM. Wink DA. Pfleiderer W. Schmidt HH. Tetrahydrobiopterin inhibits monomerization and is consumed during catalysis in neuronal NO synthase. J Biol Chem. 1999;274:24921–24929. doi: 10.1074/jbc.274.35.24921. [DOI] [PubMed] [Google Scholar]
- 46.Rosamond W. Flegal K. Furie K. Go A. Greenlund K. Haase N. Hailpern SM. Ho M. Howard V. Kissela B. Kittner S. Lloyd-Jones D. McDermott M. Meigs J. Moy C. Nichol G. O'Donnell C. Roger V. Sorlie P. Steinberger J. Thom T. Wilson M. Hong Y. Heart Disease and Stroke Statistics–2008 Update: A Report From the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation. 2008;117:e25–146. doi: 10.1161/CIRCULATIONAHA.107.187998. [DOI] [PubMed] [Google Scholar]
- 47.Svarstad E. Myking O. Ofstad J. Iversen BM. Effect of light exercise on renal hemodynamics in patients with hypertension and chronic renal disease. Scand J Urol Nephrol. 2002;36:464–472. doi: 10.1080/003655902762467648. [DOI] [PubMed] [Google Scholar]
- 48.Terasaka N. Yu S. Yvan-Charvet L. Wang N. Mzhavia N. Langlois R. Pagler T. Li R. Welch CL. Goldberg IJ. Tall AR. ABCG1 and HDL protect against endothelial dysfunction in mice fed a high-cholesterol diet. J Clin Invest. 2008;118:3701–3713. doi: 10.1172/JCI35470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Tosh D. Slack JMW. How cells change their phenotype. Nat Rev Mol Cell Biol. 2002;3:187–194. doi: 10.1038/nrm761. [DOI] [PubMed] [Google Scholar]
- 50.Tzeng E. Billiar TR. Robbins PD. Loftus M. Stuehr DJ. Expression of human inducible nitric oxide synthase in a tetrahydrobiopterin (H4B)-deficient cell line: H4B promotes assembly of enzyme subunits into an active dimer. Proc Natl Acad Sci USA. 1995;92:11771–11775. doi: 10.1073/pnas.92.25.11771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Vaziri ND. Rodriguez-Iturbe B. Mechanisms of disease: Oxidative stress and inflammation in the pathogenesis of hypertension. Nat Clin Pract Neph. 2006;2:582–593. doi: 10.1038/ncpneph0283. [DOI] [PubMed] [Google Scholar]
- 52.Welch WJ. Angiotensin II-dependent superoxide: Effects on hypertension and vascular dysfunction. Hypertension. 2008;52:51–56. doi: 10.1161/HYPERTENSIONAHA.107.090472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Wilson DO. Johnson P. Exercise modulates antioxidant enzyme gene expression in rat myocardium and liver. J Appl Physiol. 2000;88:1791–1796. doi: 10.1152/jappl.2000.88.5.1791. [DOI] [PubMed] [Google Scholar]
- 54.Xia Y. Roman LJ. Masters BS. Zweier JL. Inducible nitric-oxide synthase generates superoxide from the reductase domain. J Biol Chem. 1998;273:22635–22639. doi: 10.1074/jbc.273.35.22635. [DOI] [PubMed] [Google Scholar]
- 55.Xia Y. Zweier JL. Superoxide and peroxynitrite generation from inducible nitric oxide synthase in macrophages. Proc Natl Acad Sci USA. 1997;94:6954–6958. doi: 10.1073/pnas.94.13.6954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Xie QW. Kashiwabara Y. Nathan C. Role of transcription factor NF-kappa B/Rel in induction of nitric oxide synthase. J Biol Chem. 1994;269:4705–4708. [PubMed] [Google Scholar]
- 57.Zhan CD. Sindhu RK. Pang J. Ehdaie A. Vaziri ND. Superoxide dismutase, catalase and glutathione peroxidase in the spontaneously hypertensive rat kidney: Effect of antioxidant-rich diet. J Hypertens. 2004;22:2025–2033. doi: 10.1097/00004872-200410000-00027. [DOI] [PubMed] [Google Scholar]
- 58.Zhong J. Guo D. Chen CB. Wang W. Schuster M. Loibner H. Penninger JM. Scholey JW. Kassiri Z. Oudit GY. Prevention of angiotensin II-mediated renal oxidative stress, inflammation, and fibrosis by angiotensin-converting enzyme 2. Hypertension. 2011;57:314–322. doi: 10.1161/HYPERTENSIONAHA.110.164244. [DOI] [PubMed] [Google Scholar]
- 59.Zhou MS. Schulman IH. Raij L. Nitric oxide, angiotensin II, and hypertension. Semin Nephrol. 2004;24:366–378. doi: 10.1016/j.semnephrol.2004.04.008. [DOI] [PubMed] [Google Scholar]
- 60.Zou MH. Shi C. Cohen RA. Oxidation of the zinc-thiolate complex and uncoupling of endothelial nitric oxide synthase by peroxynitrite. J Clin Invest. 2002;109:817–826. doi: 10.1172/JCI14442. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.