Abstract
Paper spray is a newly developed ambient ionization method that has been applied for direct qualitative and quantitative analysis of biological samples. The properties of the paper substrate and spray solution have a significant impact on the release of chemical compounds from complex sample matrices, the diffusion of the analytes through the substrate, and the formation of ions for mass spectrometry analysis. In this study, a commercially available silica-coated paper was explored in an attempt to improve the analysis of therapeutic drugs in dried blood spots (DBS). The dichloromethane/isopropanol solvent has been identified as an optimal spray solvent for the analysis. The comparison was made with paper spray using chromatography paper as substrate with methanol/water as solvent for the analysis of verapamil, citalopram, amitriptyline, lidocaine and sunitinib in dried blood spots. It has been demonstrated the efficiency of recovery of the analytes was notably improved with the silica coated paper and the limit of quantitation (LOQ) for the drug analysis was 0.1 ng mL−1 using a commercial triple quadrupole mass spectrometer. The use of silica paper substrate also resulted in a sensitivity improvement of 5-50 fold in comparison with chromatography papers, including the Whatmann ET31 paper used for blood card. Analysis using a handheld miniature mass spectrometer Mini 11 gave LOQs of 10~20 ng mL−1 for the tested drugs, which is sufficient to cover the therapeutic ranges of these drugs.
Keywords: paper spray, silica coated paper, dichloromethane/isopropanol, drug, dried blood spot, ambient ionization, mass spectrometry, point-of-care analysis
Introduction
Accurate measurement of therapeutic drugs and their metabolites in blood plays an important role in drug discovery and disease therapy. Storing whole blood samples as the dried blood spots (DBS) on paper is being adopted for the analysis of drugs in blood. In comparison with conventional way of collecting blood with test tubes, DBS has some special advantages including small sample volume (typically less than 50 μL), improved chemical stability for many analytes in blood and easy sample storage and transfer at ambient temperatures.1, 2 DBS analysis can be used for a wide range of applications, including toxicology3 and pharmacokinetics studies4-6 for drug discovery as well as therapeutic drug monitoring (TDM) to assist in dosage optimization during therapy.7-17
Regardless of how blood samples are collected or stored, similar procedures have been applied for the chemical analysis of the therapeutic drugs in blood. The analytes are extracted from the blood sample using organic solvents, then separated using chromatography and analyzed using mass spectrometry (MS)1, 12-14, ultraviolet (UV),7-9 fluorescence (FL)10, 11 or immunoassay.15-17 Liquid chromatography-tandem mass spectrometry (LC-MS/MS) has been the mainstream method for the quantitation of drugs in blood.18-21 High sensitivity and selectivity are obtained with LC-MS/MS for quantitative analysis of therapeutic drugs in blood. The standard procedure involves complex sample preparation and analyte separation prior to the MS analysis. These steps are essential for minimizing matrix effects and improving the detection limits,22, 23 but could take about 30 minutes to several hours. This time frame might not be rate determining for in-lab analysis of many samples where parallel high throughput approaches can be used, but could be so when small numbers of samples are analyzed and rapid decision making is required. Used in this way, LC-MS/MS can be applied to a wide range of drug compounds; however, this method must be performed in analytical laboratories and only by experienced chemists.
Recent efforts in the development of ambient ionization methods24-30 attempt to minimize the need for sample preparation and separation prior to MS analysis of complex mixtures by combining the sampling and ionization processes into a single step in direct analysis.31-38 Some of the ambient ionization methods have been applied for analysis of dried blood spots.39, 40 For example, desorption electrospray ionization (DESI), combined with MS/MS measurement, is readily applied to the identification and quantitation of various analytes in DBS including amino acids, acylcarnitines, bile acids, glucose, creatinine, and bilirubin, etc.39, 41 Similarly, another method using the sealing surface sampling probe (SSSP), in combination with an electrospray ionization (ESI) or an atmospheric pressure chemical ionization (APCI) source, has also been successfully applied in the direct analysis of drugs (e.g., sitamaquine, acetaminophen) in DBS.40
Paper spray is a recently developed ambient sampling ionization method for MS analysis, which has been successfully applied for rapid analysis of a variety of compounds from biological samples, including urine, blood and tissue.42-47 Characteristic ions are produced directly from analytes present in complex mixtures on a paper substrate by applying a high voltage and a small volume (10 to 50 μL) of solvent to the paper. The geometry of the paper substrate, the onset voltage for spray and the sample load have been investigated for their effects on the ionization efficiency of paper spray.47 The capability of paper spray in analyzing target analytes with different sample matrices has been systematically characterized.45 Several procedures for quantitation of therapeutic drugs from DBSs have been developed and compared.44 Typical precision and accuracy values are better than 10% for a series of oncology drugs at therapeutic concentration levels in experiments that take a few minutes.48 Paper spray with various solvents, including polar, non-polar and their mixtures, has also been studied.42-44, 46 In the work reported here, the modification of the paper substrate is investigated.
Paper spray is not just an ionization process, but a complex process involving real time sample extraction with the optional possibilities of chromatographic separation and chemical reactions.42-47 For analysis of therapeutic drugs in DBS using paper spray, the drug compounds are extracted from the blood matrix by the wetting solution. The extent of the extraction, relative to other compounds eluting from the DBS, has a direct impact on the sensitivity of the analysis. The affinity of the drug compounds to a paper substrate also affects the extraction efficiency and causes loss of compound during transport to the paper tip by the spray solvent and inefficiency in ionization. The chemical properties of the solvent obviously need to be optimized for the extraction and transfer process, but they also need to be suitable for the spray ionization process, which is important to the MS analysis sensitivity. The selection of the combination of a paper substrate and a solvent is critical to the overall performance of the paper spray MS analysis.
In our previous studies, chromatography paper has mainly been used for paper spray substrates with methanol/water as the wetting and spray solvent. In this study, we explore the use of silica-coated paper for the analysis of therapeutic drugs in dried blood spots. Silica is by far the most widely used matrix for chromatographic separation owing to its chemical and mechanical stability, variable pore size, well documented chemistry for surface modification, and excellent demonstrated performance for separations. Previous studies49-51 have shown that silica-coated paper is an excellent substrate for resolving a wide variety of compounds. In this study, paper coated with silica gel was adopted as the paper spray substrate and relatively less polar solvents were used. The selection of the substrate/solvent systems was first optimized for drug analysis using a commercial triple quadrupole mass spectrometer and the performance using a handheld ion trap mass spectrometer (Mini 1152-54) was then characterized for a set of therapeutic drugs from DBSs. The combination of the paper spray with a miniature mass spectrometer is of a great interest for point-of-care applications where small-size equipment and simple operational procedures are highly desirable. The improvement in sensitivity with optimized paper spray conditions is more important for the miniature mass spectrometers than for the commercial lab-scale instrument, since the performance of miniature instruments is compromised by the need for smaller pumping systems and simpler electronics to achieve reductions in size and weight.55, 56 In the Mini 11 handheld mass spectrometer, a single stage discontinuous atmospheric pressure interface (DAPI)57 was used with an ion trap. A TSQ (Thermo Scientific, Inc., San Jose, CA) with multiple differential pumping stages and triple quadrupole mass analyzers can provide 50 times better LOQ for the analysis of therapeutic drugs in blood using paper spray.
Experimental
Paper spray
The experimental procedure of paper spray used in this work was similar to that previously reported.42-45 Briefly, the standards used for these experiments, including verapamil, sunitinib, citalopram, amitriptyline, and lidocaine, were prepared as follows: drug solutions at 100× concentration were prepared by dilution of the stock solutions into 1:1 methanol/water. The 100× standards were then spiked into bovine whole blood (with sodium citrate as anticoagulant, Innovative Research, Novi, MI) by pipetting 5 μL of the standard into 495 μL of blood. The blood samples with lower concentrations of drugs were prepared serially by diluting 40 μL of higher drug content blood samples with 360 μL of blood. The concentrations of the drugs in the final blood samples were 0.01, 0.1, 1, 10, 1000, and 10,000 ng mL−1, respectively. DBS samples were prepared by spotting a fixed volume (5 μL) of blood onto the paper substrate and drying for at least 4 h at room temperature. Samples were stored at room temperature in a sealed plastic bag containing desiccant. For paper spray, the DBS substrate was cut into a triangle (10 mm height and 5 mm base width). A copper clip was used to hold the paper triangle and to apply the high voltage needed for the spray. The distance between the tip of the paper triangle and the inlet to the mass spectrometer was about 5 mm. The silica-coated ion-exchange paper Grade SG81 (0.27 mm thick) and the chromatography paper Grade 4 (0.21 mm thick) and Grade ET31 (0.50 mm thick) were purchased from Whatman International Ltd. (Maidstone, England) and used without further chemical treatment. The Grade 4 chromatography paper is of similar thickness to the silica-coated paper while the thicker Grade ET31 is used for making the commercial blood cards (Whatman FTA DMPK-C card). The 9:1 methanol/water (v/v) solvent has been found to be generally optimal for analysis of drugs in DBS on the cellulose filter papers. The MS analysis was carried out in positive ion mode with a spray voltage at 3.5 kV. The LOQ value for each drug was defined as the lowest concentration within the set of linear responses. Three replicate measurements were made for each sample.
Characterization of paper substrates
Images of surfaces of the chromatography paper (Whatman Grade 4, 0.21 mm thick) and the silica-coated paper (Whatman Grade SG81, 0.27 mm thick)) were recorded using a FEI NOVA nanoSEM field emission scanning electron microscope (SEM, FEI Company, Hillsboro, Oregon). The substrates were sputter-coated with platinum for 1.0 min before the analysis and the accelerating voltage for the Everhart-Thornley detector (ETD, routine imaging) or through-the-lens detector (TLD, high magnification/resolution imaging) was 5 kV with a working distance of about 5.0 mm.
MS analysis
A TSQ Quantum Access Max (Thermo Scientific, San Jose, CA), operated in the selected reaction monitoring (SRM) mode was used and specific product ions produced by collision-induced dissociation (CID) were monitored. The Xcalibur™ software was used for control of the TSQ Quantum Access Max MS system and data acquisition. Argon gas (99.995% purity) was used as collision gas. The temperature MS inlet capillary was 300 °C. The SRM and instrumental parameters used for the drug compounds were as follows: verapamil: m/z 455 → 303, tube lens 97 V, Q2 offset (collision energy) 28 V; sunitinib: m/z 399 → 283, tube lens 116 V, Q2 offset 28 V; citalopram: m/z 325 → 109, tube lens 121 V, Q2 offset 28 V; amitriptyline: m/z 278 → 233, tube lens 108 V, Q2 offset 17 V; lidocaine: m/z 235 → 86, tube lens 94 V, Q2 offset 18 V. The Mini 11,52-54 a homebuilt handheld rectilinear ion trap mass spectrometer, has a discontinuous atmospheric pressure interface (DAPI)52, 53 and a pumping system with a 10 L/s trubomolecular pump (Pfeiffer HiPace 10, Pfeiffer Vacuum Inc., Nashua, NH) and a 5 L/min diaphragm pump (1091-N84.0-8.99, KNF Neuberger Inc., Trenton, NJ). The flow restricting capillary in the DAPI is of 5 cm length and 250 μm I.D. The DAPI was opened for 12 ms to introduce the ions generated by paper spray and the mass analysis was performed 600 ms after the DAPI was closed and the vacuum had reached an approximate level. The precursor ions were trapped at an RF voltage of 275 Vp-p at 1.0 MHz and the product ions were analyzed with a RF scan from 206 Vp-p to 4,500 Vp-p and a resonance ejection at q = 0.70. The frequency and the amplitude of the excitation signal for the CID were as follows: amitriptyline, 99.40 kHz and 0.39 Vp-p; citalopram: 84.60 kHz and 0.63 Vp-p; lidocaine: 78.76 kHz and 1.22 Vp-p; sunitinib: 84.89 kHz and 0.73 Vp-p; verapamil: 80.58 kHz and 0.37 Vp-p.
Results and Discussion
SEM images of the substrate surfaces are shown in Figure 1. The chromatography paper (Figure 1a) was shown to have a framework of cellulosic fibers, each of a diameter 10 - 20 μm. As previously reported by Roberts,58 strong connections at the points of contact between two cellulose fibers were observed, which are due to the hydrogen bonding between the polysaccharides at the fiber surfaces. The silica-coated paper (Figure 1b) has a similar cellulosic framework but the pores are filled with silica gel particles of diameters 1-5 μm (Figure 1c). Bovine blood of 5.0 μL was deposited onto the paper substrates to form dried blood spots (ca. 7.0 mm diameter). The cellulosic framework in the chromatography paper could still be seen except that some small pores were blocked by the dried blood (Figure 1d); however, the surface of the silica coated paper was completely covered by the dried blood (Figure 1e). Diffusion of the blood through the paper substrates was also examined as shown in Figure 1f - i. The colors of the top and bottom sides of the paper substrate within the DBS area are similar for the chromatography paper (Figure 1f and g), but significantly different for the silica-coated paper (Figure 1h and i). The color of the top side of the silica-coated paper substrate is much darker than that of the bottom side, which is due to a poor diffusion of the blood through the substrate with the framework pores blocked by the silica gel particles. A relatively large percentage of the blood sample stayed on the top side of the silica-coated paper substrate, which helped to improve the efficiency of analyte elution during the paper spray process. Though the blood distribution was less homogenous on this substrate, no unexpected sample degradation was observed after they had been stored in a sealed plastic bag for days.
Figure 1.
SEM and photographic images of Grade 4 chromatography and silica coated papers without and with dried blood spots: SEM images of (a) chromatography and (b) silica coated paper, and (c) close-up image of the selected area in (b) without dried blood spots; top view of (d) chromatography and (e) silica coated paper with dried blood spots; photograph images of the (f) top and (g) back sides of chromatography paper, and (h) top and (i) back sides of silica coated paper with blood spots.
As discussed above, the paper spray process involves the analyte elution as well as spray ionization. The solvent properties affect not only the extraction of the analytes from the dried blood spots, but also their transfer across the paper as well as the ion formation during the spray. For the silica-coated paper, pure solvents with a range of polarities were first investigated, including water, hexane, dichloromethane, methanol, ethanol, isopropanol, butyl alcohol. Figure 2(a) compares efficiency of sampling and ionization of verapamil ((M+H)+, m/z 455) in dried blood spots (500 ng mL−1) using the silica-coated paper substrate. The intensity of the fragment ion m/z 303 was monitored using the triple quadrupole operated in SRM mode. With a spray voltage of 3.5 kV, the lowest signal intensities were observed with water and hexane, which have the highest and lowest solvent polarities, respectively. Signal intensities two orders of magnitude greater were observed with dichloromethane, while the best intensities were obtained with the alcohols, viz. methanol, ethanol, isopropanol, and butyl alcohol. Among these solvents, isopropanol was the best as a pure solvent for the efficient extraction and spray ionization of verapamil.
Figure 2.
(a) Effect of spray solvent on the analysis of verapamil [(M+H)+, m/z 455, product ion, m/z 303] and (b) effect of isopropanol percentage in dichloromethane on the signal of verapamil with a triple quadrupole. Silica-coated paper substrate used. The peak intensity is an average of total ion chronogram values. The concentration of verapamil in the blood sample was 500 ng mL−1.
It is well-known that the electrospray process is highly dependent on the polarity and the volatility of the solvent. Typically solvent mixtures are used to optimize the spray process for analysis of target analytes.59-62 Addition of a non- or less-polar solvent component of low boiling point helps to enhance the generation of droplets of suitable size during the spray and facilitates their subsequent desolvation.63 Paper spray is expected to share similar spray characteristics with electrospray but the efficiency of the analyte extraction step needs also to be considered in the selection of solvent composition. After finding isopropanol as the best pure solvent for paper spray with silica-coated substrates, dichloromethane was mixed with isopropanol to produce solvents with increased volatility. The direct sampling ionization of the verapamil in the dried blood spots was performed using a set of isopropanol/dichloromethane solvents in different ratios, as shown in Figure 2b. The best signal intensity was observed with 10% (v/v) of isopropanol in the solvent mixture for the silica-coated paper. Mixtures of dichloromethane with methanol, ethanol, and butyl alcohol have also been tested and similar trends were observed. Among these solvents, 9:1 dichloromethane/isopropanol (v/v) gave the optimal performance with silica-coated paper for verapamil, citalopram, amitriptyline, lidocaine and sunitinib. These solvents were also tested with chromatography paper substrates (Grade 4 and ET31 papers) but the signal intensity was one order of magnitude lower than that with 9:1 methanol/water solvent in this case.
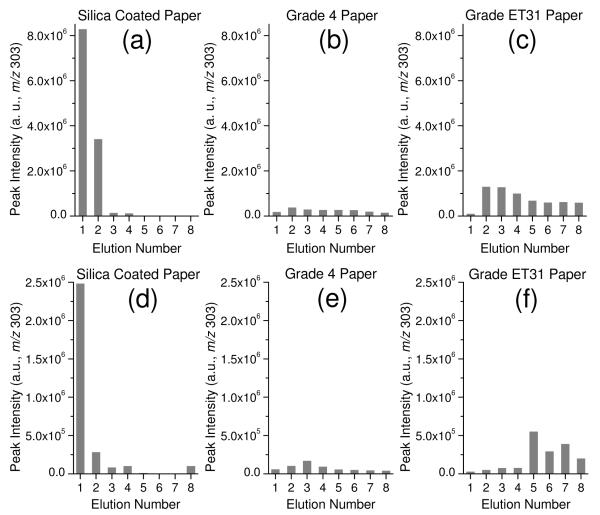
The elution efficiency was also characterized for the three solvent/substrate systems, 9:1 dichloromethane/isopropanol (v/v) for silica-coated paper and 9:1 methanol/water (v/v) for Grade 4 and Grade ET31 chromatography papers, using both pure verapamil sample spots and dried blood spots containing verapamil. The pure verapamil sample spots were prepared by dropping 5 μL water solution containing verapamil (500 ng mL−1) onto the paper and drying the substrate completely. The voltage was applied and solvent added multiple times to produce many paper spray events using the same substrate bearing a single sample spot while the signal of fragment ion m/z 303 was monitored. Spray solvent of 25 μL was used each time and the solvent was not added until the monitored ion signal decreased to a minimum, when the spray solvent was also exhausted. Solvent consumption took 5-8 s for dichloromethane/isopropanol on silica-coated paper but 40-70 s for methanol/water on chromatography paper substrates. For both pure analytes and blood samples, much higher peak intensities were was observed for the silica-coated paper substrate (Figure 3a and d). Most (~70%) of the verapamil, was eluted during the first elution step of the paper spray process. A relatively even elution pattern was observed for the Grade 4 chromatography paper with low peak intensities for verapamil (Figure 3b and e), although an increasing and then decreasing trend exists. Interestingly, a significant increase in analyte in the 2nd (Figure 3c) and 5th (Figure 3f) elution was observed with the Grade ET31 paper for the pure and the blood samples, respectively. Presumably, this is due to the relatively larger substrate volume, which requires sufficient wetting of the substrate and transfer of the analytes to the substrate tip for the paper spray.
Figure 3.
Comparison of the elution behavior of verapamil with silica coated paper and chromatography papers: (i) verapamil in pure water (5 μL, 500 ng mL−1) deposited onto surface of (a) silica coated paper, (b) Grade 4 chromatography paper and (c) ET31 chromatography paper; (ii) verapamil in blood (5 μL, 500 ng mL−1) deposited onto surface of (d) silica coated paper and (e) Grade 4 chromatography paper and (f) Grade ET31 chromatography paper. Note: the experiments were performed after the paper had dried. Solvent for silica coated paper was 9:1 dichloromethane/isopropanol, chromatography paper 9:1 methanol/water. a.u. : arbitrary units.
Similar procedure was applied to dried blood spots containing verapamil. The blood spots were each prepared with 5 μL of blood sample containing 500 ng mL−1 verapamil. The matrix in the dried blood spots is much more complex than that in the pure verapamil sample spots. For silica-coated paper with 9:1 dichloromethane/isopropanol, analyte release during the first paper spray event was still dominant, although the peak intensity for verapamil decreased about 3 times, presumably due to the matrix effect. For chromatography papers with methanol/water, no significant difference in elution pattern was observed except that the maxima of the verapamil detected appeared at later elutions (Figure 3d and f). There was also about an order of magnitude difference in the best signal intensities observed for these three systems in favor of the hydrophobic substrate.
Several factors could account for the difference between these two substrate/solvent systems and the improvement with the silica-coated paper substrate. The blocking of the pores in the cellulosic framework by the silica resulted in a more concentrated sample on the top surface of the substrate and also a less binding interaction between the analyte and the cellulose. This helps to improve analyte elution during the paper spray process. The polarity of the solvent systems might also play an important role. The polarity of methanol/water solvent (9:1, v/v) is higher than that of dichloromethane/isopropanol solvent (9:1, v/v) (polarity index: water 10.2, methanol 5.1, isopropanol 3.9 and dichloromethane 3.1). Low polarity organic compounds, such as verapamil, dissolve better in dichloromethane/isopropanol solvent, which makes the extraction of these chemicals from dried blood more efficient.
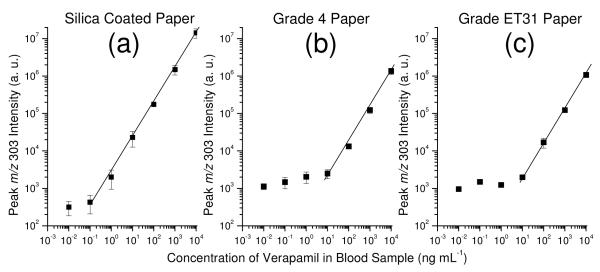
The performance of the improved substrate/solvent system was characterized for quantitative analysis of therapeutic drugs in whole blood samples using paper spray. Dried blood spots on paper spray substrates were prepared by depositing 5 μL whole blood containing verapamil at a concentration from 0.01 ng mL−1 to 10,000 ng mL−1 and drying the sample on substrate completely. MS analysis was performed using the triple quadrupole mass spectrometer and the peak intensity of m/z 303 from verapamil ((M+H)+, m/z 455) was recorded. As shown in Figure 4a, an LOQ of 0.1 ng mL−1 was observed for silica-coated substrate with 9:1 (v/v) dichloromethane/isopropanol, which is about two orders of magnitude better than that for Grade 4 (Figure 4b) and Grade ET31 (Figure 4c) chromatography papers with 9:1 (v/v) methanol/water. Other therapeutic drugs in whole blood, including sunitinib, citalopram, amitriptyline, and lidocaine, were also analyzed and an LOQ of 0.1 ng mL−1 was obtained for each of them using paper spray with silica-coated paper.
Figure 4.
Comparison of the LOQ and linear dynamic range of verapamil with (a) silica coated paper (0.27 mm thick; 9:1 dichloromethane/isopropanol), (b) Grade 4 chromatography paper (0.21 mm thick; 9:1 methanol/water), and (c) Grade ET31 chromatography paper (0.50 mm thick; 9:1 methanol/water) . Note: 5 μl of blood sample was used, product ion m/z 303 of verapamil was monitored.
As discussed above, one of the major goals of developing the paper spray42-46 method is to eventually allow miniature paper spray MS analysis system to be developed for in-situ applications such as point-of-care diagnosis. A sample cartridge47 can be developed with a paper substrate or even with internal standards preprinted on it to allow blood samples taken by a finger prick to be subjected to immediate analysis. The improvement of the front end paper spray ionization module for the quantitation of the therapeutic drugs in whole blood is of particular significance for analysis using miniature mass spectrometers, since the performance of the miniature instruments is necessarily compromised by the use of smaller mass analyzers, simpler control electronics, and significantly smaller vacuum systems. In this study, paper spray using the silica paper substrates was characterized using a miniature rectilinear ion trap mass spectrometer, Mini 11.52 This was done by quantitative analysis of verapamil, sunitinib, citalopram, amitriptyline, and lidocaine in dried blood spots. MS/MS analysis was performed using the procedure previously described52-54 to acquire the intensities of the characteristic fragment ions from the drug compounds.
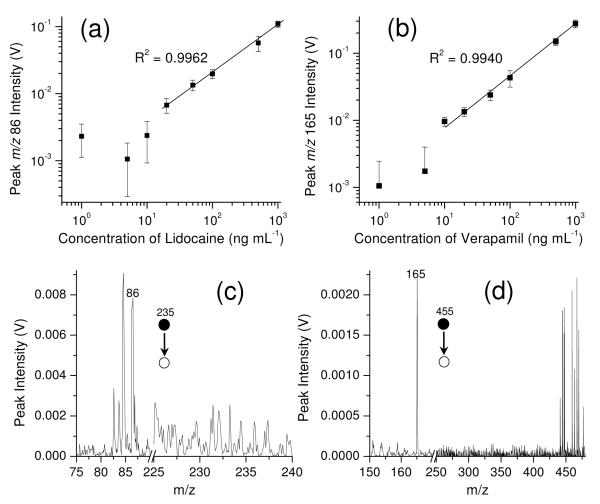
Figure 5a and b shows the low concentration regions (1 ng mL−1 to 1,000 ng mL−1) of the linear ranges for quantitation of lidocaine (fragment ion m/z 86) and verapamil (fragment ion m/z 165). LOQ values for lidocaine and verapamil are 20 ng mL−1 and 10 ng mL−1, respectively, approximately two orders of magnitude higher than achieved using the benchtop triple quadruple. The spectra recorded at LOQs for these two drugs are shown in Figure 5c and d. Good linearity was also obtained in the ranges from their LOQs to 1 μg/mL.
Figure 5.
Linear dynamic range for (a) lidocaine and (b) verapamil, and typical spectra of lidocaine (c) and verapamil (d) with concentrations in blood of 20 ng mL−1 and 10 ng mL−1, respectively, obtained with Mini 11. Silica-coated paper (0.27 mm thick) with 9:1 dichloromethane/isopropanol.
A comparison study was done for silica-coated paper and chromatography paper (Whatman Grade ET31, 0.50 mm thick) using paper spray and the Mini 11 for analysis of therapeutic drugs in dried blood spots. The LOQs obtained and the therapeutic windows for lidocaine, amitriptyline, sunitinib, verapamil, and citalopram are listed in Table I. Using the chromatography paper with 9:1 methanol/water, the performance of the paper spray/Mini 11 system was only good enough to cover the therapeutic range for lidocaine, but not for the other 5 drugs in DBSs. With the silica paper substrate and 9:1 dicholoramethane/isopropanol, improvements of 5-50 fold were obtained in LOQs for these drugs, which made the performance of the miniature systems adequate for their quantitative analysis from dried blood spots.
Table 1.
Comparison of LOQ valuesa for some typical drugs between ET 31 chromatography paper and silica coated paper with Mini 11.
| Drug | Precursor ion |
Product ion |
LOQ with Chromatography paper (ng mL−1) |
LOQ with silica coated paper (ng mL−1) |
Therapeutic range (ng mL−1) |
|---|---|---|---|---|---|
| lidocaine | 235 | 86 | 100 | 20 | 1,000-6,000 |
| amitriptyline | 278 | 233 | 100 | 10 | 50-200 |
| sunitinib | 399 | 326 | 500 | 10 | 20-200 |
| verapamil | 455 | 165 | 100 | 10 | 50-250 |
| citalopram | 325 | 109 | 100 | 10 | 10-200 |
Conclusions
Paper spray with silica coated paper substrate was characterized using a commercial triple quadrupole and a home-built miniature ion trap mass spectrometer for analysis of therapeutic drugs in dried blood spots. The overall analysis efficiency can be greatly improved by using low boiling and low polarity solvent, 9:1 dichloromethane/isopropanol (v/v). The LOQ of analysis of a set of therapeutic drugs in dried blood spots, including verapamil, citalopram, amitriptyline, lidocaine, and sunitinib, was obtained as low as 0.1 ng mL−1 with the commercial triple quadrupole and 10~20 ng mL−1 with a miniature ion trap mass spectrometer. The triple quadrupole is inherently better suited to SRM analysis than is an ion trap instrument so this factor as well as the reduced performance of the Mini MS is responsible for the difference. Compared to chromatography paper, in each case there is a 5-50 fold improvement with the silica paper substrate.
Acknowledgment
This work was supported by the National Science Foundation (CHE 0847205 and CHE 0848650), National Science Foundation Instrumentation Development for Biological Research (DBI 0852740), National Institutes of Health (1R21RR031246-01 and 1R21EB009459-01), National Natural Science Foundation of China (20728505) and the Alfred Mann Institute at Purdue.
References
- (1).Rao RN, Maurya PK, Ramesh M, Srinivas R, Agwane SB. Biomed. Chromat. 2010;24:1356–1364. doi: 10.1002/bmc.1450. [DOI] [PubMed] [Google Scholar]
- (2).Li WK, Zhang J, Tse FLS. Biomed. Chromat. 2011;25:258–277. doi: 10.1002/bmc.1572. [DOI] [PubMed] [Google Scholar]
- (3).Barfield M, Spooner N, Lad R, Parry S, Fowles S. J. Chromatogr. B: Analyt. Technol. Biomed. Life Sci. 2008;870:32–37. doi: 10.1016/j.jchromb.2008.05.025. [DOI] [PubMed] [Google Scholar]
- (4).Lawson G, Tanna S, Mulla H, Pandya H. J. Pharm. Pharmacol. 2009;61:A33. [Google Scholar]
- (5).Suyagh MF, Laxman KP, Millership J, Collier P, Halliday H, McElnay JC. J. Chromatogr. B: Analyt. Technol. Biomed. Life Sci. 2010;878:769–776. doi: 10.1016/j.jchromb.2010.01.031. [DOI] [PubMed] [Google Scholar]
- (6).Spooner N, Lad R, Barfield M. Anal. Chem. 2009;81:1557–1563. doi: 10.1021/ac8022839. [DOI] [PubMed] [Google Scholar]
- (7).Ntale M, Mahindi M, Ogwal-Okeng JW, Gustafsson LL, Beck O. J. Chromatogr. B: Anal. Technol. Biomed. Life Sci. 2007;859:137–140. doi: 10.1016/j.jchromb.2007.09.012. [DOI] [PubMed] [Google Scholar]
- (8).Lejeune D, Souletie I, Houze S, Le Bricon T, Le Bras J, Gourmel B, Houze P. J. Pharm. Biomed. Anal. 2007;43:1106–1115. doi: 10.1016/j.jpba.2006.09.036. [DOI] [PubMed] [Google Scholar]
- (9).Ronn AM, Lemnge MM, Angelo HR, Bygbjerg IC. Therap. Drug Monitor. 1995;17:79–83. doi: 10.1097/00007691-199502000-00013. [DOI] [PubMed] [Google Scholar]
- (10).Tawa R, Hirose S, Fujimoto T. J. Chromatogr. B: Biomed. Appl. 1989;490:125–132. doi: 10.1016/s0378-4347(00)82767-x. [DOI] [PubMed] [Google Scholar]
- (11).Croes K, McCarthy PT, Flanagan RJ. J. Anal. Toxicol. 1994;18:255–260. doi: 10.1093/jat/18.5.255. [DOI] [PubMed] [Google Scholar]
- (12).ter Heine R, Rosing H, van Gorp ECM, Mulder JW, van der Steeg WA, Beijnen JH, Huitema ADR. J. Chromatogr. B: Analyt. Technol. Biomed. Life Sci. 2008;867:205–212. doi: 10.1016/j.jchromb.2008.04.003. [DOI] [PubMed] [Google Scholar]
- (13).Koal T, Burhenne H, Romling R, Svoboda M, Resch K, Kaever V. Rapid Commun. Mass Spectrom. 2005;19:2995–3001. doi: 10.1002/rcm.2158. [DOI] [PubMed] [Google Scholar]
- (14).Cheung CY, van der Heijden J, Hoogtanders K, Christiaans M, Liu YL, Chan YH, Choi KS, van de Plas A, Shek CC, Chau KF, Li CS, van Hooff J, Stolk L. Transplant Int. 2008;21:140–145. doi: 10.1111/j.1432-2277.2007.00584.x. [DOI] [PubMed] [Google Scholar]
- (15).Coombes EJ, Gamlen TR, Batstone GF, Leigh PN. Ann. Clin. Biochem. 1984;21:519–522. doi: 10.1177/000456328402100615. [DOI] [PubMed] [Google Scholar]
- (16).Fujimoto T, Tsuda Y, Tawa R, Hirose S. Clin. Chem. 1989;35:867–869. [PubMed] [Google Scholar]
- (17).Li PK, Lee JT, Conboy KA, Ellis EF. Clin. Chem. 1986;32:552–555. [PubMed] [Google Scholar]
- (18).Taylor PJ, Tai C-H, Franklin ME, Pillans PI. Clin. Biochem. 2011;44:14–20. doi: 10.1016/j.clinbiochem.2010.06.012. [DOI] [PubMed] [Google Scholar]
- (19).Saint-Marcoux F, Sauvage F-L, Marquet P. Anal. Bioanal. Chem. 2007;388:1327–1349. doi: 10.1007/s00216-007-1320-1. [DOI] [PubMed] [Google Scholar]
- (20).Korecka M, Shaw LM. Ann. Transplant. 2009;14:61–72. [PubMed] [Google Scholar]
- (21).Checa A, Oliver R, Hernandez-Cassou S, Saurina J. Anal. Chim. Acta. 2009;647:1–13. doi: 10.1016/j.aca.2009.05.028. [DOI] [PubMed] [Google Scholar]
- (22).Breadmore MC, Theurillat R, Thormann W. Electrophoresis. 2004;25:1615–1622. doi: 10.1002/elps.200305819. [DOI] [PubMed] [Google Scholar]
- (23).Lee ED, Henion JD. Rapid Commun. Mass Spectrom. 1992;6:727–733. [Google Scholar]
- (24).Cooks RG, Ouyang Z, Takats Z, Wiseman JM. Science. 2006;311:1566–1570. doi: 10.1126/science.1119426. [DOI] [PubMed] [Google Scholar]
- (25).Venter A, Nefliu M, Cooks RG. TrAC, Trends Anal. Chem. 2008;27:284–290. [Google Scholar]
- (26).Van Berkel GJ, Pasilis SP, Ovchinnikova O. J. Mass Spectrom. 2008;43:1161–1180. doi: 10.1002/jms.1440. [DOI] [PubMed] [Google Scholar]
- (27).Weston DJ. Analyst. 2010;135:661–668. doi: 10.1039/b925579f. [DOI] [PubMed] [Google Scholar]
- (28).Huang MZ, Yuan CH, Cheng SC, Cho YT, Shiea J. Annu. Rev. Anal. Chem. 2010;3:43–65. doi: 10.1146/annurev.anchem.111808.073702. [DOI] [PubMed] [Google Scholar]
- (29).Ouyang Z, Zhang XR. Analyst. 2010;135:659–660. doi: 10.1039/c003812c. [DOI] [PubMed] [Google Scholar]
- (30).Harris GA, Galhena AS, Fernandez FM. Anal. Chem. 2011;83:4508–4538. doi: 10.1021/ac200918u. [DOI] [PubMed] [Google Scholar]
- (31).Takats Z, Wiseman JM, Gologan B, Cooks RG. Science. 2004;306:471–473. doi: 10.1126/science.1104404. [DOI] [PubMed] [Google Scholar]
- (32).Cody RB, Laramee JA, Durst HD. Anal. Chem. 2005;77:2297–2302. doi: 10.1021/ac050162j. [DOI] [PubMed] [Google Scholar]
- (33).Chen H, Yang S, Wortmann A, Zenobi R. Angew. Chem. Int. Ed. 2007;46:7591–7594. doi: 10.1002/anie.200702200. [DOI] [PubMed] [Google Scholar]
- (34).Shiea J, Lin SY, Huang MZ, Chang HC. Analytical Chemistry. 2007;79:8789–8795. doi: 10.1021/ac070590k. [DOI] [PubMed] [Google Scholar]
- (35).Andrade FJ, Shelley JT, Wetzel WC, Webb MR, Gamez G, Ray SJ, Hieftje GM. Anal. Chem. 2008;80:2654–2663. doi: 10.1021/ac800210s. [DOI] [PubMed] [Google Scholar]
- (36).Nemes P, Vertes A. Anal. Chem. 2007;79:8098–8106. doi: 10.1021/ac071181r. [DOI] [PubMed] [Google Scholar]
- (37).Sampson JS, Hawkridge AM, Muddiman DC. J. Am. Soc. Mass Spectrom. 2008;19:1527–1534. doi: 10.1016/j.jasms.2008.06.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (38).Haddad R, Sparrapan R, Kotiaho T, Eberlin MN. Anal. Chem. 2008;80:898–903. doi: 10.1021/ac701960q. [DOI] [PubMed] [Google Scholar]
- (39).Takats Z, Wiseman JM, Cooks RG. J. Mass Spectrom. 2005;40:1261–1275. doi: 10.1002/jms.922. [DOI] [PubMed] [Google Scholar]
- (40).Van Berkel GJ, Kertesz V. Anal. Chem. 2009;81:9146–9152. doi: 10.1021/ac901712b. [DOI] [PubMed] [Google Scholar]
- (41).Wiseman JM, Evans CA, Bowen CL, Kennedy JH. Analyst. 2010;135:720–725. doi: 10.1039/b922329k. [DOI] [PubMed] [Google Scholar]
- (42).Wang H, Liu JJ, Cooks RG, Ouyang Z. Angew. Chem., Int. Ed. 2010;49:877–880. doi: 10.1002/anie.200906314. [DOI] [PubMed] [Google Scholar]
- (43).Wang H, Manicke NE, Yang Q, Zheng L, Shi R, Cooks RG, Ouyang Z. Anal. Chem. 2011;83:1197–1201. doi: 10.1021/ac103150a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (44).Manicke NE, Yang QA, Wang H, Oradu S, Ouyang Z, Cooks RG. Int. J. Mass Spectrom. 2011;300:123–129. doi: 10.1016/j.ijms.2011.05.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (45).Liu J, Wang H, Manicke NE, Lin J-M, Cooks RG, Ouyang Z. Anal. Chem. 2010;82:2463–2471. doi: 10.1021/ac902854g. [DOI] [PubMed] [Google Scholar]
- (46).Li AY, Wang H, Ouyang Z, Cooks RG. Chem. Commun. 2011;47:2811–2813. doi: 10.1039/c0cc05513a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (47).Yang Q, Wang H, Maas JD, Chappell WJ, Manicke NE, Cooks RG, Ouyang Z. Int. J. Mass Spectrom. 2011 doi: 10.1016/j.ijms.2011.05.013. In Press, Corrected Proof. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (48).Manicke N, Abu-Rabie P, Spooner N, Ouyang Z, Cooks R. J. Am. Soc. Mass Spectrom. 2011:1–7. doi: 10.1007/s13361-011-0177-x. [DOI] [PubMed] [Google Scholar]
- (49).Marinetti GV. In: Lipid Chromatographic Analysis. Wuthier RE, editor. Vol. 1. Marcel Dekker; New York: 1976. pp. 59–109. [Google Scholar]
- (50).Valadon LRG, Mummery RS. Phytochemistry. 1972;11:413–414. [Google Scholar]
- (51).Egan RW. Anal. Biochem. 1975;68:654–657. doi: 10.1016/0003-2697(75)90664-8. [DOI] [PubMed] [Google Scholar]
- (52).Gao L, Sugiarto A, Harper JD, Cooks RG, Ouyang Z. Anal. Chem. 2008;80:7198–7205. doi: 10.1021/ac801275x. [DOI] [PubMed] [Google Scholar]
- (53).Hou KY, Xu W, Xu JA, Cooks RG, Ouyang Z. Anal. Chem. 2011;83:1857–1861. doi: 10.1021/ac102962e. [DOI] [PubMed] [Google Scholar]
- (54).Sokol E, Noll RJ, Cooks RG, Beegle LW, Kim HI, Kanik I. Int. J. Mass Spectrom. 2011 In Press, Corrected Proof. [Google Scholar]
- (55).Maas JD, Xu W, Hendricks P, Chappell WJ, Ieee 2010 Ieee Mtt-S International Microwave Symposium Digest; New York: Ieee; 2010. pp. 1636–1639. [Google Scholar]
- (56).Ouyang Z, Cooks RG. Annu. Rev. Anal. Chem. 2009;2:187–214. doi: 10.1146/annurev-anchem-060908-155229. [DOI] [PubMed] [Google Scholar]
- (57).Gao L, Cooks RG, Ouyang Z. Anal. Chem. 2008;80:4026–4032. doi: 10.1021/ac800014v. [DOI] [PubMed] [Google Scholar]
- (58).Roberts JC. The Chemistry of Paper. The Royal Society of Chemistry; Letchworth, UK: 1996. [Google Scholar]
- (59).Tian ZX, Kass SR. J. Am. Chem. Soc. 2008;130:10842–10843. doi: 10.1021/ja802088u. [DOI] [PubMed] [Google Scholar]
- (60).Lavarone AT, Jurchen JC, Williams ER. J. Am. Soc. Mass Spectrom. 2000;11:976–985. doi: 10.1016/S1044-0305(00)00169-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (61).Venter AR, Kamali A, Jain S, Bairu S. Anal. Chem. 2010;82:1674–1679. doi: 10.1021/ac902013x. [DOI] [PubMed] [Google Scholar]
- (62).Li JW, Dewald HD, Chen H. Anal. Chem. 2009;81:9716–9722. doi: 10.1021/ac901975j. [DOI] [PubMed] [Google Scholar]
- (63).Kebarle P, Tang L. Anal. Chem. 1993;65:A972–A986. [Google Scholar]