Abstract
Insect pathogenic fungi produce a plethora of insecticidally and pharmaceutically active compounds, including 39 cyclohexadepsipeptide destruxins (dtxs). Even though dtxs were first discovered more than 50 y ago, the genes responsible for their biosynthesis were unknown until this study. Based on our comparative genomic information and targeted gene disruptions, we report the gene cluster for dtx biosynthesis in the insect pathogen Metarhizium robertsii. The nonribosomal peptide synthetase DtxS1 has six adenylation domains, two of which are capable of selecting different amino acids to synthesize dtx B and its analogs. The cytochrome P450 enzyme DtxS2 converts dtx B into other dtxs by a chain of reactions, each producing a new derivative. The aldo-keto reductase DtxS3 and aspartic acid decarboxylase DtxS4 are responsible for the conversion and provision of the first and last substrates for the dtx assembly line, respectively. Insect bioassays showed that dtxs could suppress both cellular and humoral immune responses thereby assisting fungal propagation in insects. The differing abilities of Metarhizium species to produce toxins is dependent on the presence of the dtxS1 gene. The toxigenic species are capable of killing multiple orders of insects, whereas the nontoxigenic Metarhizium spp. have narrow host ranges. Thus, the acquisition or retention of the dtx biosynthesis gene cluster in Metarhizium lineages has been coordinated with the evolution of fungal host specificity. The data from this study will facilitate the development of dtxs as bioinsecticides or pharmaceuticals.
Keywords: virulence, insect biocontrol, tailoring enzyme
Insect pathogenic fungi produce a plethora of bioactive metabolites, including 39 cyclic hexadepsipeptide destruxins (dtxs) (1, 2). These insecticidal compounds were first identified more than 50 y ago (3), but the genes responsible for their biosynthesis have been unknown. Dtxs contain an α-hydroxyisocaproic acid (HIC) and 5-aa residues, i.e., l-proline (Pro) or pipecolic acid (Pip), l-isoleucine (Ile) or l-valine (Val), N-methyl-l-valine (MeVal), N-methyl-l-alanine (MeAla), and β-alanine (β-Ala) (Fig. 1). The known dtxs are categorized into six major groups: the dtxs A through F and their derivatives (2, 4). No single fungal species or strain produces all of these dtxs. The insect-pathogenic fungus Metarhizium anisopliae, however, produces most dtxs (2). Anecdotal evidence suggests some other fungi, including the insect pathogens Beauveria felina (5), Lecanicillium longisporum (6), and Aschersonia spp (7) can also produce some dtxs. Dtx B and its derivatives were found in the plant pathogens Alternaria brassicae (8) and Ophiosphaerella herpotricha (9) as having host selective phytotoxicity.
Fig. 1.
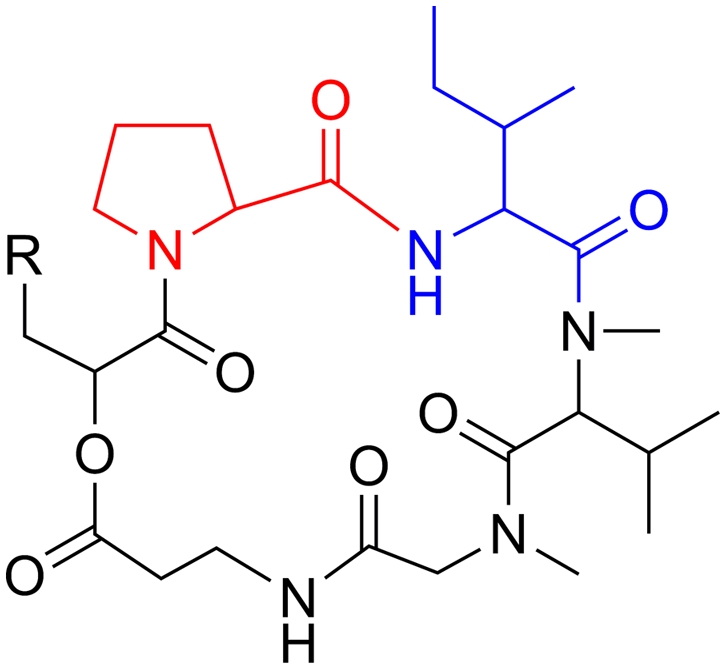
Chemical structures of the dtxs. For dtx A through F, R is –CH=CH2, –CH(CH3)2, –CH(CH3)CH2OH, –CH(CH3) –COOH, –CH(O)CH2, and –CHCH3OH, respectively. When the residue Pro (in red) is replaced by a pipecolic acid, the dtxs are named dtx A1 through E1, whereas when replacing Ile (in blue) with a Val, the series are named dtx A2 through E2. Dtx F1 and F2 have not been reported previously.
Dtxs induce flaccid paralysis and visceral muscle contraction in insects. These and cytotoxic effects on epithelial cells are thought to involve targeting the Ca2+ channel and vacuolar-type ATPase (4, 10). Dtxs are recognized as important virulence determinants in Metarhizium spp (11–13). The Metarhizium genus contains a dozen species (14), including M. anisopliae and M. robertsii, which can kill hundreds of insect species, and M. acridum, which can only kill locusts and grasshoppers (15, 16). It has frequently been observed that dtx production correlates with host specificity (12, 17). Besides their insecticidal activity, dtxs have also been explored as potential pharmaceuticals to treat cancer, osteoporosis, Alzheimer's disease, and hepatitis B (18–20).
The chemical synthesis of dtxs has been reported before (4). However, the mode of dtx biosynthesis by fungi was a puzzle. The involvement of multifunctional nonribosomal peptide synthetase (NRPS) has been suggested but not successfully verified (21). Based on our comparative genomic study of M. anisopliae (the strain now reclassified as M. robertsii) and M. acridum (15), we report the identification of a NRPS gene cluster responsible for the biosynthesis of dtxs. Insect bioassays confirmed that dtxs could suppress insect cellular and humoral immune responses and contribute to fungal virulence. We also show that the presence or absence of the dtxS1 gene in Metarhizium lineages is associated with the evolution of fungal host specificity.
Results
Prediction of dtx Synthetase Gene.
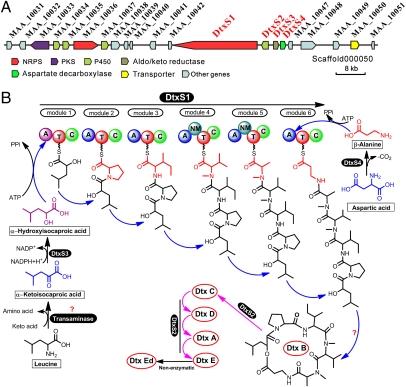
As reported previously (17, 22), our preliminary analysis confirmed that the M. robertsii strain ARSEF 23 produced dtxs in Czapek Dox (CD) broth, whereas the M. acridum strain CQMa 102 did not. Comparative genomic analysis indicated that the genome structures of M. robertsii and M. acridum are largely syntenic (15). Thus, nonproduction of dtxs by M. acridum suggests that its genome does not contain the NRPS gene cluster involved in dtx biosynthesis. Based on this assumption, a phylogenetic analysis was conducted for 15 NRPS proteins from M. robertsii and 13 NRPSs from M. acridum (Table S1 and Fig. S1A). The results showed that MAA_10043 and MAA_06559 loci in M. robertsii do not have orthologs in M. acridum. The modulation analysis demonstrated that the NRPS MAA_06559 only contains one module of adenylation (A), thiolation (T), and condensation (C) domains (Table S1). In contrast, MAA_10043 has six ATC modules (designated as DtxS1), as would be predicted for the biosynthesis of dtx hexadepsipeptide structures. The predicted NRPS DtxS1 has 7,898 aa and the gene contains two introns. Like other fungal NRPS genes (23), dtxS1 is within a cluster that contains genes encoding one or more cytochrome P450, aldo/keto reductase, and decarboxylase enzymes (Fig. 2A) (15).
Fig. 2.
Genes involved in dtx biosynthesis in M. robertsii. (A) Schematic map of the dtx biosynthetic gene cluster. (B) Biosynthetic model for hydroxyl and amino acids incorporation and assembly. The dtx B2 instead of dtx B will be synthesized when the amino acid Val but not Ile is selected by the A3 domain. The question mark indicates that the enzyme(s) involved in the reaction have not been determined.
Gene Deletion and Elucidation of the dtx Biosynthesis Pathway.
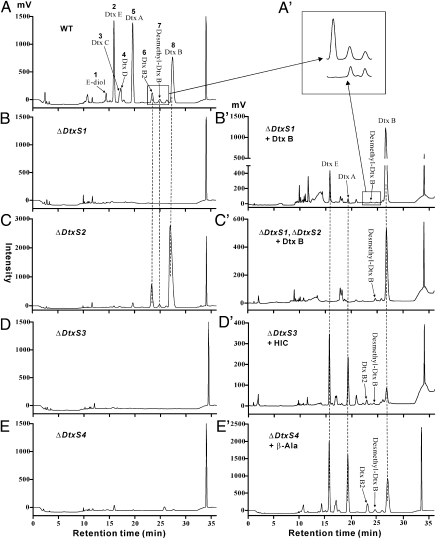
To verify the functions of dtxS1 and its clustered genes, serial genes were deleted by homologous replacements. Because some dtxs have the same molecular masses (2, 4), we performed a combination of liquid chromatography (LC)-MS (Fig. S2 A–G), NMR (Table S2), and HPLC analysis to profile the dtxs produced by the wild-type (WT) and mutant strains. The data indicated that during growth in CD medium the WT strain produces eight dtxs, including dtx E-diol (ED), E, C, D, A, B2, desmethyl-B, and B (Fig. 3A). Consistent with previous studies (13, 22), dtx E, A, and B were produced in the largest amounts (Fig. 3A and Table S3). As predicted, the dtxS1-null mutants were unable to produce dtxs either in the CD medium (Fig. 3B) or in silkworm larvae (Fig. S2H). Deletions of dtxS1 upstream genes (Fig. 2A) did not affect dtx production by the null mutants compared with the WT strain (Fig. S3), indicating that these genes are not involved in production and modification of dtxs. However, deletions of genes downstream of dtxS1 led to partial or complete loss of dtx production (Fig. 3 C–E).
Fig. 3.
HPLC profiling of dtx production by the WT and gene deletion mutants of M. robertsii. When grown in CD broth, the WT strain could produce at least eight dtx derivatives (A). After the deletion of the NRPS gene dtxS1, the resulting mutant failed to synthesize the dtxs (B). The null mutant of dtxS2 could only produce dtx B, B2, and desmethyl-B (C). The addition of dtx B to the cultures of ΔdtxS1 enabled the mutant to produce dtx E and A (B′) but not the dtx B2 (A′). Dtx B did not allow ΔdtxS1ΔdtxS2 to produce dtx E and A (C′). The clustered dtxS3 gene functions in the conversion of α-ketoisocaproic acid to hydroxyisocaproic acid (HIC), the first substrate for dtx assembly. A mutant deleted in the dtxS3 gene did not produce dtxs (D). Conversely, the addition of HIC to ΔdtxS3 culture restored its ability to produce dtxs (D′). The dtxS4 gene was verified to involve in the decarboxylation of aspartic acid into β-Ala (E and E′), i.e., the null mutant produced dtxs only when β-Ala was added in medium.
The null mutants of MAA_10044 gene (designated as dtxS2), putatively encoding a cytochrome P450 (CYP) subfamily 65AF enzyme, could only produce dtx B, B2, and desmethyl-B (Fig. 3C and Table S2). DtxS2 expressed using either a bacterium or yeast expression system lacked activity. To verify DtxS2 function, we generated a double mutant by deleting both dtxS1 and dtxS2 genes. When ΔdtxS1 (dtxS2 functional) and ΔdtxS1ΔdtxS2 were grown in medium supplemented with dtx B, ΔdtxS1 could convert dtx B into dtx E, C, D, and A, whereas ΔdtxS1ΔdtxS2 could not (Fig. 3 B′ and C′). Dtx B2 was not produced by either ΔdtxS1 or ΔdtxS1ΔdtxS2 in dtx B feeding assays (Fig. 3A′). However, both mutants produced desmethyl-B. Thus, DtxS1 synthesizes dtx B and B2, whereas DtxS2 is required to convert dtx B into other dtx derivatives (Fig. 2B).
The locus MAA_10045 (designated as dtxS3) putatively encodes an aldo-keto reductase and MAA_10046 (named as dtxS4) is predicted to be an aspartate decarboxylase. The null mutants of these two genes failed to produce any dtxs (Fig. 3 D and E). We hypothesized that DtxS3 converts α-ketoisocaproic acid from deaminated leucine into α-hydroxyisocaproic acid (HIC), the first substrate for dtx assembly (Fig. 2B). This was confirmed by adding HIC into the ΔdtxS3 culture to restore dtx production (Fig. 3D′). Decarboxylation of aspartic acid into β-Ala by DtxS4 was confirmed by adding β-Ala to the mutant culture because this rescued dtx biosynthesis by ΔdtxS4 (Fig. 3E′). Our previous study has shown that dtx E is not stable (24), and the degradation of dtx E to E-diol is a nonenzymatic process (Fig. 2B).
The detection of both dtx B and B2 in the ΔdtxS2 cultures confirmed that the A3 domain could interchangeably incorporate either Ile (highly biased) for dtx B or Val for B2 biosynthesis. Phylogenetic analysis of DtxS1 A domains demonstrated that they clustered according to substrate structures, e.g., A3 (for Val/Ile) and A4 (Val) grouped together (Fig. S1B). The C domains of NRPSs catalyze peptide bond formation (25). The topology of DtxS1 C domains is different from the A domain tree (Fig. S1C).
Effects of dtxs on Fungal Virulence.
Injection of culture filtrates of the WT, ΔdtxS1, and ΔdtxS2 confirmed the insecticidal effects of dtxs (Fig. S4A). To verify the contribution of dtx to fungal virulence, insect bioassays were conducted by injecting locusts and silkworms with spore suspensions of the WT and mutants (Fig. S4 B and C). Median lethal times (LT50) for locusts were estimated at 105.4 ± 1.4, 106.5 ± 1.7, and 112.4 ± 3.2 h for the WT, ΔdtxS1, and ΔdtxS2, respectively. Log-rank tests revealed that the difference between the WT and ΔdtxS1 was not significant (χ2 = 0.26; P = 0.61), whereas the differences between the WT and ΔdtxS2 (χ2 = 5.91; P = 0.015) and between ΔdtxS1 and ΔdtxS2 (χ2 = 5.91, P = 0.043) were significant. A similar trend was observed in bioassays using silkworms as LT50 values were 79.3 ± 0.5, 81.0 ± 0.6 and 88.0 ± 1.7 h for the WT, ΔdtxS1, and ΔdtxS2, respectively. However, differences were significant between the WT and ΔdtxS1 (χ2 = 4.92; P = 0.027), as well as between the WT and ΔdtxS2 (χ2 = 17.12; P = 0) and ΔdtxS1 and ΔdtxS2 (χ2 = 15.13; P = 0). Thus, the effects of dtx on fungal virulence varied with insect species but, intriguingly, the differences were significantly greater between the WT and ΔdtxS2 than between the WT and ΔdtxS1, i.e., ΔdtxS2 (with the ability to produce dtx B and B2) was less virulent than ΔdtxS1.
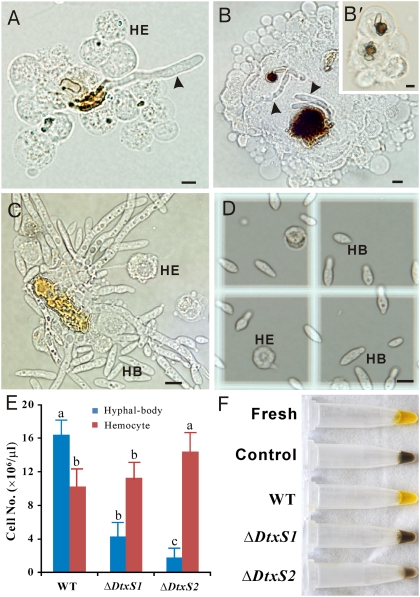
Microscopic observations indicated that the dtxs could counteract insect host immune defenses (Fig. 4). Following injection of silkworm larvae, Metarhizium spores were rapidly encapsulated by hemocytes. The germinated WT spores in silkworm hemocoel quickly escaped (16–20 h postinjection) from hemocyte encapsulation (Fig. 4A), but spores from both the ΔdtxS1 and ΔdtxS2 mutants were unable to escape (Fig. 4 B and B′). Thus, the WT cells propagated more quickly than the mutants (Fig. 4 C and D). Forty-eight hours postinjection, the average number of WT cells in insects was fourfold higher than in the treatment with ΔdtxS1 and eightfold higher than in the insects treated with ΔdtxS2. Furthermore, the hemocyte numbers were higher (P < 0.05) in ΔdtxS2-treated insects than those in insects injected with the WT and ΔdtxS1 spores, respectively (Fig. 4E). The data were generally consistent with the bioassay results, suggesting that ΔdtxS2 is less virulent than the WT and ΔdtxS1. In addition, unlike insects infected with the mutants, the hemolymph collected from the WT-injected insects lost melanization activity (Fig. 4F). Thus, the dtxs, especially A and E, may suppress both cellular and humoral immune responses to facilitate fungal colonization in insects.
Fig. 4.
Time point observations of fungal development and insect immune responses to the WT and mutant strains. Within sixteen hours of injection, WT spores germinated and the germ tube (arrow) escaped from hemocyte encapsulation (A). However, the ΔdtxS1 spores were heavily melanized, and most of the germ tubes (arrow) were continuously attacked by hemocytes (B). ΔdtxS2 showed similar observations (B′). In contrast to mutants, the WT demonstrated massive growth and yeast-type budding producing hyphal-bodies (HB) up to 36 h postinjection (C). After 48 h, the production of HBs was counted using a hemocytometer (D). In comparison with the untreated insects (4.2 × 107 hemocytes/μL), the number of hemocytes was reduced fourfold in the insects injected with the WT or ΔdtxS1 spores and threefold in ΔdtxS2 treated insects (E). At this time point (48 h), the insects were bled and the hemolymph was individually collected and left exposed to air for 1 h (F). The blood collected from 80% insects infected with the WT remained unmelanized and resembled freshly collected hemolymph from the untreated larvae (the first tube). However, the blood collected from 75% of ΔdtxS1 and 70% of ΔdtxS2 mutant-treated insects was melanized to a similar extent as controls (hemolymph collected from untreated insects and left for 1 h exposed to air). HE, hemocyte; HB, hyphal-body. (Scale bar, 5 μm.)
Association of Toxin Production with the Evolution of Metarhizium Host Specificity.
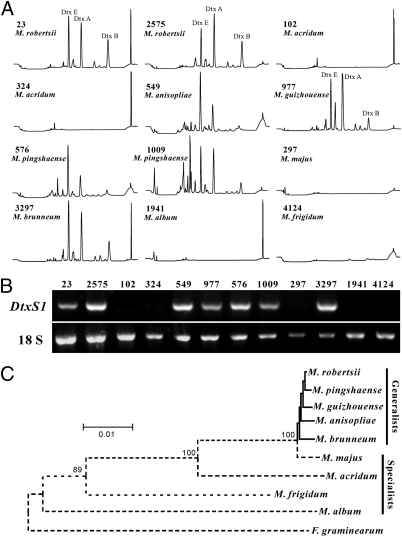
The most recent taxonomic revision estimated that the Metarhizium genus contains at least 13 species, including the dtx-nonproducing M. acridum (14). To further verify the association of dtx production with the presence or absence of the dtxS1 gene cluster, we conducted HPLC analysis and PCR verifications for different strains of nine Metarhizium species (Table S4). Dtx profiles were detected in CD cultures of M. robertsii, M. guizhouense, M. anisopliae, M. pingshaense, and M. brunneum but not in the cultures of M. acridum, M. majus, M. album, and M. frigidum (Fig. 5A). This is consistent with the presence or absence of dtxS1 genes in these species (Fig. 5B). Our previous phylogenomic analysis suggested that Metarhizium is closely related to the plant endophyte Epichloe festucae and the plant pathogen Fusarium graminearum (15). The well-supported phylogenetic tree rooted with F. graminearum demonstrated that dtx-nonproducing Metarhizium spp. evolved before dtx-producing species (Fig. 5C). Given that dtx production has not been reported from Epichloe spp. or Fusarium spp., it suggests that the dtx-producing Metarhizium species acquired the toxin gene cluster, by an as yet unknown mechanism, during evolution.
Fig. 5.
Association of dtx production with the evolution of Metarhizium spp. host range. (A) HPLC analysis of dtx production by different Metarhizium species and strains. The strain number and species are provided in each panel. (B) PCR detection of the presence/absence of a dtxS1 gene in different Metarhizium species and strains. (C) Phylogenetic relationship of different Metarhizium species. The consensus neighbor-joining tree was generated using the internal transcribed spacer sequences. The dashed lines represent the lack of dtxS1 gene in the lineages.
Among the dtx-nonproducing species, M. acridum is a well-known acridid-specific fungus (15, 16), whereas M. majus is specific for Coleopteran insects, principally scarabs (26), and M. album is specific for Homopteran insects (27). It is evident, therefore, that dtx-nonproducing species are characterized by having a narrow host range, whereas the toxin-producing species have broad host ranges covering multiple orders of insects (Table S5). The data suggest that the acquisition of the dtx gene cluster in Metarhizium lineages is coordinated with the evolution of fungal host specificity.
Discussion
Based on the information derived from the genome sequences of M. robertsii and M. acridum (15), we found the gene cluster responsible for dtx biosynthesis in M. robertsii. The NRPS DtxS1 is a typical linear NRPS with six modules for the sequential addition of hydroxyl and amino acids during dtx biosynthesis. In silico analysis of NRPSs based on sequence similarity/structure identified many A domains with a predicted specificity for multiple amino acids (28). However, this has only been experimentally demonstrated for a few fungal NRPSs (29). In this study, we found that the A3 domain of DtxS1 is capable of selecting either Ile or Val for the biosynthesis of dtx B or B2. Dtx A1 through E1 were not detected in this study, although there is a single report of dtx A through E, A1 through E1, and A2 through E2 derivatives being identified in a strain of M. anisopliae (30). Their presence would mean that the DtxS1 A2 domain could also select either Pro or Pip to synthesize dtx B or B1. Like many fungal NRPSs (23, 31), the carboxyl terminal of DtxS1 does not contain a thioesterase (TE) domain. Thus, the terminal C domain (C6) may be involved in cyclization of dtx B and B2. In support of this, the DtxS1 C6 domain contains a SHAQYDG motif, which is very much like the active site signature (SHALYDG) in the terminal C domains of BbBEAS and BbBSLS involved in cyclization of depsipeptides beauvericin and bassianolide in the insect pathogenic fungus B. bassiana (32, 33). However, the identification of a linear protodestruxin from the cultures of M. anisopliae (34) could mean that an external TE may function for dtx cyclization.
We found that a single tailoring enzyme CYP65 DtxS2 is involved in modifying dtx B into other dtxs, probably in the order dtx B → C → D → A → E. Family 65 is one of the largest CYP families in filamentous fungi (35). The genome of M. robertsii contains three CYP65 genes, and M. acridum has six (15). Fusarium spp. express a CYP65 trichothecene C-15 hydroxylase (Tri11, 35% identity with DtxS2) involved in trichothecene biosynthesis (36). The Aspergillus parasiticus CYP65 VerB (33% identity with DtxS2) in the aflatoxin biosynthesis cluster is involved in desaturation of versicolorin B to versicolorin A (37). DtxS2 also shows an identity (35%) with the CYP65 RadP from the endophytic fungus Chaetomium chiversii that can epoxidize the antibiotic radicicol (38). These similarities suggest that DtxS2 probably functions as a multiple functional enzyme for metabolite hydroxylation, desaturation, oxidation, and/or epoxidation. Thus, hydroxylation of dtx B would lead to the formation of dtx C, which will be further oxidized into dtx D by DtxS2. Oxidation and desaturation of dtx D would produce dtx A, and the latter could be further epoxidized by DtxS2 to form dtx E (Fig. 2B). The lower ratios of dtx C and D in culture media (Fig. 3A) would support the hypothesis that they are the intermediate products of DtxS2 metabolism. These assumptions are consistent with the negatively correlated relationships of dtx production levels, e.g., the decrease of dtx A is paralleled with the increase of dtx E in media (17). The steroselectivity and sterochemistry of DtxS2 have yet to be determined.
Within the dtxS1 gene cluster, we verified that dtxS3 (aldo-keto reductase) and dtxS4 (aspartic acid decarboxylase) are responsible for the conversion and provision of the first (HIC) and last (β-Ala) substrates for the dtx assembly line, respectively. Assuming leucine is the initial precursor of HIC, an as yet unidentified leucine transaminase will be required for deamination of Leu into an α-ketoisocaproic acid (Fig. 2B). The tailoring enzyme involved in dtx demethylation and the regulatory genes controlling dtx biosynthesis also remain to be investigated. Additional studies are ongoing, including verifying whether a downstream ATP-binding cassette transporter (MAA_10048) is involved in pumping dtxs out of fungal cells.
The insecticidal effects of dtxs have been well studied (2, 4). However, our bioassays showed that the effects of dtxs on fungal virulence differed between locusts and caterpillars. This could reflect differences in the insect physiology of the target sites for dtx activity and/or dtx detoxification, as reported for plants that break down dtx B (2, 8). Locusts may have a greater ability to detoxify dtxs than silkworms because no significant differences were observed between the WT and ΔdtxS1. Although dtx B is less insecticidal than Dtx A or E (11), it is still hard to explain at this stage why the dtx B/B2-producing mutant ΔdtxS2 was significantly less virulent than ΔdtxS1 toward both examined insect species. However, this result is consistent with the ΔdtxS2 mutant being less able to colonize the insect hemocoel compared with the WT and ΔdtxS1.
The evolution of pathogen host specificity is a longstanding issue in evolutionary biology (39). Given that the dtxs are insecticidal to a large variety of insects (2, 4), it is not surprising that, unlike Metarhizium generalists, the specialist species do not have the dtxS1 gene to produce dtxs. We showed that the specialists evolved before the generalist species. Thus, the lateral acquisition of the dtxS1 gene cluster could have driven, at least in part, the evolution of Metarhizium generalist species able to kill multiple orders of insects. In support of this interpretation, several ribosomal and nonribosomal toxins involved in virulence are believed to have been transferred by horizontal gene transfer (HGT) between plant pathogenic fungi (40). For example, interspecific transfer of a host-selective toxin gene (toxA) from the wheat pathogen Stagonospora nodorum to Pyrenophora tritici-repentis enabled the latter to cause a serious disease in wheat (41). Our phylogenomic analysis indicated that the Metarhizium lineage diverged from the plant pathogens or endophytes (15). The dtx B and its derivatives are associated with host selective phytotoxicity in plant pathogens (8, 9), indicating that the gene clusters responsible for dtx biosynthesis originated in plant pathogens. We suggest, therefore, that the dtxS1 gene cluster in the generalist species of Metarhizium may have been acquired from a plant pathogen through interspecific HGT and that it evolved further with the acquisition of dtxS2 that allowed production of other dtxs. However, it cannot be ruled out that the ancestor of the Metarhizium lineage possessed the gene cluster and that it has been lost in specialist species.
In this report, we present the mechanism of dtx biosynthesis in M. robertsii. The toxins are able to suppress insect immune responses and, thus, assist fungal propagation in insects. It is interesting to find that the presence/absence of the dtx biosynthesis gene cluster in the Metarhizium lineage is correlated with the evolution of fungal host specificity. Our data will facilitate future exploration of the gene cluster to develop dtxs as environmentally friendly insecticides and to provide dtx resources for pharmaceutical studies.
Materials and Methods
Fungal Strains and Culture Maintenance.
The WT strain M. robertsii ARSEF23 is a generalist insect pathogen and M. acridum CQMa 102, a locust-specific pathogen. Both species have been genome-sequenced (15). Other Metarhizium species were also included for comparative analysis of dtx production (Table S4). Fungal cultures were maintained on potato dextrose agar (PDA) (Difco) at 25 °C in the dark until sporulation and then kept at 4 °C.
Analysis of NRPS Genes and Gene Deletion.
The preliminary analysis showed that in contrast to M. robertsii strain ARSEF 23, the M. acridum strain CQMa 102 does not produce dtxs. Using the published genome data (15), we performed comparative sequence analysis to determine the nonorthologous NRPS genes in the M. robertsii genome. The predicted NRPS genes were collected from each fungal species, and the A domain(s) from each NRPS were aligned with Clustal X 2.0 and subjected to phylogenetic analysis with MAGA 5.0 (42) using a Jones–Taylor–Thornton substitution model, a pair-wised deletion for gaps/missing data, and 1,000 bootstrap replications test. To verify gene function and determine the biosynthetic pathway, serial gene deletions were conducted through homologous replacements (43). Briefly, the flanking regions of the target genes were amplified using different primer pairs (Table S6) and ligated into the binary vector of pDHt containing the herbicide resistance bar gene for Agrobacterium-mediated transformations.
Dtx Induction.
Different strains/species of Metarhizium (Table S4) and gene knockout mutants were cultured in CD broth for induction of dtx biosynthesis (13, 44). To verify the functions of DtxS2, DtxS3, and DtxS4 activities, the substrates of dtx B, α-HIC (Sigma), and β-Ala (Sigma) were added into the cultures of corresponding null mutants at a final concentration of 10 mM. After 7 d of incubation at 25 °C and 200 rpm, the culture filtrates were collected by filtration through Whatman No. 1 filter paper for dtx extraction as described previously (44). The crude extracts were dissolved in 1 mL of neat methanol and filtrated through a 0.22-μm membrane before HPLC and HPLC-MS analysis. The experiments were repeated twice with three replicates per treatment. To detect in insecta dtx production, the mycosed larvae silkworm were freeze-dried and ground to powder for toxin extraction.
HPLC, MS, and NMR Analysis.
The chromatographic profiles of crude extract samples were detected with a LC-20AD HPLC system (Shimadzu Scientific Instruments), equipped with a C18 reverse phase column (particle size: 5μm; length: 4.6 × 250 mm) and a SPD-20A UV/Vis detector. The mobile phase was a linear gradient of deionized water and methanol from 90:10 for 2 min, 55:45 for 2 min, 39:61 for 22 min, 5:95 for 5 min, and 90:10 for 8 min at a flow rate of 0.1 mL/min and was monitored at a wavelength of 215 nm (13). Quantification of dtxs was determined by integration with calibration curves obtained from dtx A standard (Sigma) and purified dtx B and Dtx E. Detection of in insecta dtxs production was performed by high-performance ion chromatography. The molecular masses of different components were determined by Q-TOF LC-MS analysis using the Agilent G6520A accurate-mass system with a poroshell 300 sb-C8 column as follows: drying gas temperature 325 °C, drying gas flow 10 L/mL, and nebulizer pressure 30 psi. To verify the structures of ΔdtxS2 products, H and C NMR spectra were recorded on a Bruker ARX 600 spectrometer (600/150 MHz) and a Bruker AM-400 (400/100 MHz) instrument with tetramethylsilane as a calibration standard.
Insect Bioassays.
Two insect species were included in virulence assays: fifth instar larvae of the silkworm (Bombyx mori) and adult locusts (Locusta migratoria). The silkworms were each injected with 10 μL of suspension of 5 × 105 WT or mutant spores/mL through the second proleg, and the locust were injected with 10 μL of 1 × 106 spores/mL through the intersegmental membrane between two abdominal terga. Each treatment had 3 replicates with 10 insects each, and the experiments were repeated twice. Mortality was recorded every 12 h. Extra silkworm larvae were injected for bleeding for microscopic observation of encapsulation and fungal development within the insect hemocoel (43). The median lethal time (LT50) of different fungal strains was estimated and compared with the Kaplan–Meier method (log-rank pairwise comparison) using the SPSS 17 program (SPSS).
PCR Detection of dtxS1 Gene and Phylogenetic Analysis of Metarhizium Species.
Identification of the dtxS1 gene in diverse strains of Metarhizium species (Table S4) was conducted by PCR using the primers dtxF (AGATTTGCCGCAGCTACCTA) and dtxR (CACCAGATGCGAGTTCTCAA). The internal transcribed spacer (ITS) regions were amplified using the primer-pairs itsF (TTACGTCCCTGCCCTTTGTA) and itsR (ACCTCACCAAAAGCATCCTC). The acquired sequences plus the ITS sequence from the plant pathogen F. graminearum (HQ176435) were aligned with CLUSTAL X 2.0, and a neighbor-joining phylogenetic tree was generated with the program MEGA 5.0 (42) using 1,000 bootstrap replicates and a Jukes–Cantor substitution model with a pairwise deletion for gap/missing data.
Supplementary Material
Acknowledgments
We thank Prof. Raymond J. St. Leger (University of Maryland) for critical reading of the manuscript. The study was supported by Ministry of Science and Technology of China Grants 2009CB118904 and 2009AA10Z112 and Chinese Academy of Sciences Grants KSCX2-EW-N-06, DBSH-2011-047, and KSCX2-EW-G-16.
Footnotes
The authors declare no conflict of interest.
*This Direct Submission article had a prearranged editor.
This article contains supporting information online at www.pnas.org/lookup/suppl/doi:10.1073/pnas.1115983109/-/DCSupplemental.
References
- 1.Molnár I, Gibson DM, Krasnoff SB. Secondary metabolites from entomopathogenic Hypocrealean fungi. Nat Prod Rep. 2010;27:1241–1275. doi: 10.1039/c001459c. [DOI] [PubMed] [Google Scholar]
- 2.Liu BL, Tzeng YM. Development and applications of destruxins: A review. Biotechnol Adv. 2011 doi: 10.1016/j.biotechadv.2011.10.006. [DOI] [PubMed] [Google Scholar]
- 3.Kodaira Y. Toxic substances to insects, produced by Aspergillus ochraceus and Oospora destructor. Agric Biol Chem. 1961;25:261–262. [Google Scholar]
- 4.Pedras MSC, Irina Zaharia L, Ward DE. The destruxins: Synthesis, biosynthesis, biotransformation, and biological activity. Phytochemistry. 2002;59:579–596. doi: 10.1016/s0031-9422(02)00016-x. [DOI] [PubMed] [Google Scholar]
- 5.Lira SP, et al. New destruxins from the marine-derived fungus Beauveria felina. J Antibiot (Tokyo) 2006;59:553–563. doi: 10.1038/ja.2006.76. [DOI] [PubMed] [Google Scholar]
- 6.Butt TM, et al. Mass spectrometry as a tool for the selective profiling of destruxins; their first identification in Lecanicillium longisporum. Rapid Commun Mass Spectrom. 2009;23:1426–1434. doi: 10.1002/rcm.4018. [DOI] [PubMed] [Google Scholar]
- 7.Krasnoff SB, Gibson DM. New destruxins from the entomopathogenic fungus Aschersonia sp. J Nat Prod. 1996;59:485–489. [Google Scholar]
- 8.Pedras MS, Zaharia IL, Gai Y, Zhou Y, Ward DE. In planta sequential hydroxylation and glycosylation of a fungal phytotoxin: Avoiding cell death and overcoming the fungal invader. Proc Natl Acad Sci USA. 2001;98:747–752. doi: 10.1073/pnas.021394998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Venkatasubbaiah P, Tisserat NA, Chilton WS. Metabolites of Ophiosphaerella herpotricha, a cause of spring dead spot of bermudagrass. Mycopathologia. 1994;128:155–159. [Google Scholar]
- 10.Ruiz-Sanchez E, Lange AB, Orchard I. Effects of the mycotoxin destruxin A on Locusta migratoria visceral muscles. Toxicon. 2010;56:1043–1051. doi: 10.1016/j.toxicon.2010.07.012. [DOI] [PubMed] [Google Scholar]
- 11.Dumas C, Robert P, Pais M, Vey A, Quiot JM. Insecticidal and cytotoxic effects of natural and hemisynthetic destruxins. Comp Biochem Physiol Pharmacol Toxicol Endocrinol. 1994;108:195–203. doi: 10.1016/1367-8280(94)90031-0. [DOI] [PubMed] [Google Scholar]
- 12.Kershaw MJ, Moorhouse ER, Bateman R, Reynolds SE, Charnley AK. The role of destruxins in the pathogenicity of metarhizium anisopliae for three species of insect. J Invertebr Pathol. 1999;74:213–223. doi: 10.1006/jipa.1999.4884. [DOI] [PubMed] [Google Scholar]
- 13.Wang CS, Skrobek A, Butt TM. Concurrence of losing a chromosome and the ability to produce destruxins in a mutant of Metarhizium anisopliae. FEMS Microbiol Lett. 2003;226:373–378. doi: 10.1016/S0378-1097(03)00640-2. [DOI] [PubMed] [Google Scholar]
- 14.Bischoff JF, Rehner SA, Humber RA. A multilocus phylogeny of the Metarhizium anisopliae lineage. Mycologia. 2009;101:512–530. doi: 10.3852/07-202. [DOI] [PubMed] [Google Scholar]
- 15.Gao Q, et al. Genome sequencing and comparative transcriptomics of the model entomopathogenic fungi Metarhizium anisopliae and M. acridum. PLoS Genet. 2011;7:e1001264. doi: 10.1371/journal.pgen.1001264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wang S, Fang W, Wang CS, St Leger RJ. Insertion of an esterase gene into a specific locust pathogen (Metarhizium acridum) enables it to infect caterpillars. PLoS Pathog. 2011;7:e1002097. doi: 10.1371/journal.ppat.1002097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Amiri-Besheli B, Khambay B, Cameron S, Deadman ML, Butt TM. Inter- and intra-specific variation in destruxin production by insect pathogenic Metarhizium spp., and its significance to pathogenesis. Mycol Res. 2000;104:447–452. [Google Scholar]
- 18.Chen HC, Chou CK, Sun CM, Yeh SF. Suppressive effects of destruxin B on hepatitis B virus surface antigen gene expression in human hepatoma cells. Antiviral Res. 1997;34:137–144. doi: 10.1016/s0166-3542(97)01031-0. [DOI] [PubMed] [Google Scholar]
- 19.Nakagawa H, et al. Destruxins, cyclodepsipeptides, block the formation of actin rings and prominent clear zones and ruffled borders in osteoclasts. Bone. 2003;33:443–455. doi: 10.1016/s8756-3282(03)00201-1. [DOI] [PubMed] [Google Scholar]
- 20.Itoh N, et al. Destruxin E decreases Beta-amyloid generation by reducing colocalization of beta-amyloid-cleaving enzyme 1 and beta-amyloid protein precursor. Neurodegener Dis. 2009;6:230–239. doi: 10.1159/000236902. [DOI] [PubMed] [Google Scholar]
- 21.Bailey AM, et al. Cloning and sequence analysis of an intron-containing domain from a peptide synthetase-encoding gene of the entomopathogenic fungus Metarhizium anisopliae. Gene. 1996;173:195–197. doi: 10.1016/0378-1119(96)00212-0. [DOI] [PubMed] [Google Scholar]
- 22.Hu QB, Ren SX, Wu JH, Chang JM, Musa PD. Investigation of destruxin A and B from 80 Metarhizium strains in China, and the optimization of cultural conditions for the strain MaQ10. Toxicon. 2006;48:491–498. doi: 10.1016/j.toxicon.2006.06.018. [DOI] [PubMed] [Google Scholar]
- 23.Keller NP, Turner G, Bennett JW. Fungal secondary metabolism - from biochemistry to genomics. Nat Rev Microbiol. 2005;3:937–947. doi: 10.1038/nrmicro1286. [DOI] [PubMed] [Google Scholar]
- 24.Dudley E, Wang CS, Skrobek A, Newton RP, Butt TM. Mass spectrometric studies on the intrinsic stability of destruxin E from Metarhizium anisopliae. Rapid Commun Mass Spectrom. 2004;18:2577–2586. doi: 10.1002/rcm.1659. [DOI] [PubMed] [Google Scholar]
- 25.Rausch C, Hoof I, Weber T, Wohlleben W, Huson DH. Phylogenetic analysis of condensation domains in NRPS sheds light on their functional evolution. BMC Evol Biol. 2007;7:78. doi: 10.1186/1471-2148-7-78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lin KJ, Roberts DW. The production of destruxin by the entomogenous fungus Metarhizium anisopliae var. majus. J Invertebr Pathol. 1986;47:120–122. [Google Scholar]
- 27.Rombach MC, Humber RA, Evans HC. Metarhizium album, a fungal pathogen of leaf- and planthoppers of rice. Trans Br Mycol Soc. 1987;88:451–459. [Google Scholar]
- 28.Röttig M, et al. NRPSpredictor2—a web server for predicting NRPS adenylation domain specificity. Nucleic Acids Res. 2011;39(Web Server issue) doi: 10.1093/nar/gkr323. W362-W367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Stack D, Neville C, Doyle S. Nonribosomal peptide synthesis in Aspergillus fumigatus and other fungi. Microbiology. 2007;153:1297–1306. doi: 10.1099/mic.0.2006/006908-0. [DOI] [PubMed] [Google Scholar]
- 30.Pais M, Das BC, Ferron P. Depsipeptides from Metarhizium anisopliae. Phytochemistry. 1981;20:715–723. [Google Scholar]
- 31.Bushley KE, Turgeon BG. Phylogenomics reveals subfamilies of fungal nonribosomal peptide synthetases and their evolutionary relationships. BMC Evol Biol. 2010;10:26. doi: 10.1186/1471-2148-10-26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Xu Y, et al. Biosynthesis of the cyclooligomer depsipeptide beauvericin, a virulence factor of the entomopathogenic fungus Beauveria bassiana. Chem Biol. 2008;15:898–907. doi: 10.1016/j.chembiol.2008.07.011. [DOI] [PubMed] [Google Scholar]
- 33.Xu Y, et al. Biosynthesis of the cyclooligomer depsipeptide bassianolide, an insecticidal virulence factor of Beauveria bassiana. Fungal Genet Biol. 2009;46:353–364. doi: 10.1016/j.fgb.2009.03.001. [DOI] [PubMed] [Google Scholar]
- 34.Suzuki A, Tamura S. Isolation and structure of protodestruxin from Matarhizium anisopliae. Agric Biol Chem. 1972;36:896–898. [Google Scholar]
- 35.Deng J, Carbone I, Dean RA. The evolutionary history of cytochrome P450 genes in four filamentous Ascomycetes. BMC Evol Biol. 2007;7:30. doi: 10.1186/1471-2148-7-30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ward TJ, Bielawski JP, Kistler HC, Sullivan E, O'Donnell K. Ancestral polymorphism and adaptive evolution in the trichothecene mycotoxin gene cluster of phytopathogenic Fusarium. Proc Natl Acad Sci USA. 2002;99:9278–9283. doi: 10.1073/pnas.142307199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Yu J, Bhatnagar D, Cleveland TE. Completed sequence of aflatoxin pathway gene cluster in Aspergillus parasiticus. FEBS Lett. 2004;564:126–130. doi: 10.1016/S0014-5793(04)00327-8. [DOI] [PubMed] [Google Scholar]
- 38.Wang S, et al. Functional characterization of the biosynthesis of radicicol, an Hsp90 inhibitor resorcylic acid lactone from Chaetomium chiversii. Chem Biol. 2008;15:1328–1338. doi: 10.1016/j.chembiol.2008.10.006. [DOI] [PubMed] [Google Scholar]
- 39.Legros M, Koella JC. Experimental evolution of specialization by a microsporidian parasite. BMC Evol Biol. 2010;10:159. doi: 10.1186/1471-2148-10-159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Mehrabi R, et al. Horizontal gene and chromosome transfer in plant pathogenic fungi affecting host range. FEMS Microbiol Rev. 2011;35:542–554. doi: 10.1111/j.1574-6976.2010.00263.x. [DOI] [PubMed] [Google Scholar]
- 41.Friesen TL, et al. Emergence of a new disease as a result of interspecific virulence gene transfer. Nat Genet. 2006;38:953–956. doi: 10.1038/ng1839. [DOI] [PubMed] [Google Scholar]
- 42.Tamura K, et al. MEGA5: Molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol. 2011;28:2731–2739. doi: 10.1093/molbev/msr121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Wang CS, St Leger RJ. A collagenous protective coat enables Metarhizium anisopliae to evade insect immune responses. Proc Natl Acad Sci USA. 2006;103:6647–6652. doi: 10.1073/pnas.0601951103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Wang CS, Skrobek A, Butt TM. Investigations on the destruxin production of the entomopathogenic fungus Metarhizium anisopliae. J Invertebr Pathol. 2004;85:168–174. doi: 10.1016/j.jip.2004.02.008. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.