Abstract
The endemic human pathogenic fungus Histoplasma capsulatum is a major fungal pathogen with a broad variety of clinical presentations, ranging from mild, focal pulmonary disease to life-threatening systemic infections. Although azoles, such as itraconazole and voriconazole, and amphotericin B have significant activity against H. capsulatum, about 1 in 10 patients hospitalized due to histoplasmosis die. Hence, new approaches for managing disease are being sought. Over the past 10 years, studies have demonstrated that monoclonal antibodies (mAbs) can modify the pathogenesis of histoplasmosis. Disease has been shown to be impacted by mAbs targeting either fungal cell surface proteins or host co-stimulatory molecules. This review will detail our current knowledge regarding the impact of antibody therapy on histoplasmosis.
Keywords: Histoplasma capsulatum, histoplasmosis, antibody, histone 2B, heat shock protein 60, M antigen, co-stimulation
Introduction
The most commonly encountered endemic mycoses in the Americas are due to Histoplasma capsulatum, Blastomyces dermatitidis, Paracoccidioides brasiliensis, and Coccidioides immitis/posadasii (Lockhart et al., 2009; Prado et al., 2009). As is the case in other endemic fungi, H. capsulatum infection is typically acquired by inhalation of fungal propagules after disturbances of contaminated soil or excreta (Guimaraes et al., 2006). The clinical manifestation of the disease range from asymptomatic infection or a mild influenza-like illness to a disseminated sepsis form that may involve virtually any tissue (Meloan, 1952; Goodwin and Des Prez, 1978; Fojtasek et al., 1994; Bradsher, 1996). These manifestations depend mainly on the magnitude of exposure (i.e., the number of fungal particles inhaled), the immunological status of the host (i.e., patients with AIDS or individuals receiving steroids or chemotherapy), and the virulence of the infective strain, indicating that environmental and genetic factors influence the manifestation of disease (Goodwin et al., 1981; Kauffman, 2007). The vast majority of infected persons have either no symptoms or a very mild illness that is never recognized as being histoplasmosis (Wheat et al., 2007). In fact, 95–99% of the primary infections are not recognized or detected in immunologically normal hosts in endemic areas (Saliba and Beatty, 1960; Isbister et al., 1976; Goodwin et al., 1981). Although the majority of symptomatic infections follow primary exposures to H. capsulatum, reactivation of latent infection can result in significant disease, particularly in the setting of immunosuppression (Kauffman, 2007). Furthermore, reactivation disease can be developed in liver transplant recipients with disease originating from latent infections in the transplanted organs (Limaye et al., 2000). Additionally, reactivation histoplasmosis has increasingly occurred in patients receiving anti-cytokine therapies, especially inhibitors of INF-γ and TNF-α (Deepe, 2005; Deepe et al., 2005; Scheckelhoff and Deepe, 2005).
As infection with H. capsulatum is not a mandatory reportable event, the actual incidence of clinically significant histoplasmosis is not known. Epidemiological studies have estimated that 500,000 individuals acquire H. capsulatum annually in the USA and over 80% of young adults in endemic areas have been infected with the fungus (Edwards et al., 1969). A national survey of hospital discharge diagnoses from 2002 identified 3,370 patients hospitalized for histoplasmosis in the USA with a crude mortality rate of 8% (Chu et al., 2006). Notably only 14% of the patients were immunocompromised and this percentage was similar in those who died. Given the nature of the survey, it only represented a “fraction of the burden of all morbidity and mortality” (Chu et al., 2006) related to H. capsulatum. This study also documents that hospital charges for the identified patients were well over $100 million. Hence, histoplasmosis is a significant and costly cause of morbidity and mortality in otherwise healthy individuals and in immunodeficient patients. Despite the potency of current antifungal drugs, they nevertheless fail to prevent mortality in nearly 1 in 10 patients hospitalized with histoplasmosis.
Although H. capsulatum has previously been considered to consist of three varieties, capsulatum, duboisii, and farciminosum (Darling, 1906; Dodd and Thompkins, 1934; Medoff et al., 1987), recent molecular work has shown that these distinctions are phylogenetically meaningless, but instead, there are genetically distinct geographical populations or phylogenetic species (Kasuga et al., 2003). H. capsulatum is a dimorphic fungal pathogen with two distinct morphological forms, filamentous and yeast, depending on the nutritional factors and temperature (Maresca and Kobayashi, 1989). H. capsulatum is found in nature primarily as a saprophytic mold, and exists in soils enriched with organic nitrogen sources such as animal excrements, or when grown in the laboratory at less than 35°C (Emmons, 1950, 1956a,b; Zeidberg et al., 1952; Alteras, 1966; Emmons et al., 1966; Disalvo et al., 1970; Smith, 1971a,b). The mold form is composed of hyaline septate hyphae that produce two different asexual reproduction structures, macroconidia and microconidia. The microconidia are the purported infectious propagule, as their size, 2–6 μm, is well suited for deposition into distal alveoli. Upon entry to a susceptible host, the microconidia rapidly convert to the pathogenic single, budding yeast-like form, which can also be cultivated in laboratory medium at 37°C.
As a facultative intracellular parasite, the interaction of H. capsulatum with macrophage cells is a critical component of the host response to infection (Newman, 2005) and is a complex and obscure phenomenon. Heat shock protein 60 (Hsp60) is the major H. capsulatum surface ligand that engages CD11b/CD18 (CR3) integrin on the surface of phagocytes resulting in phagocytosis (Long et al., 2003; Habich et al., 2006). H. capsulatum yeasts have critical interactions with inflammatory neutrophils, and with dendritic cells (DCs) in the lung and other organs. Indeed, recent new evidence suggests that DCs may be the key antigen-presenting cells that initiate cell-mediated immunity (Deepe et al., 2008). H. capsulatum yeast cells must survive and/or subvert the hostile antimicrobial environmental within phagocytes (Allendoerfer et al., 1997), including fungicidal mechanisms such as reactive oxygen species and products of the nitric oxide synthase (NOS) pathway (Eissenberg and Goldman, 1987). The yeast form actively inhibits phagolysosomal fusion, thereby preventing exposure to the acidic hydrolytic enzymes of the lysosomes. H. capsulatum also prohibits accumulation of vacuolar ATPase, which is important for proton accumulation in phagosomes, and the fungus can actively alkalinize phagosomal pH to 6.5 (Strasser et al., 1999). Within the phagocytic cells, viable yeast may travel to hilar and mediastinal lymph nodes where they gain access to the blood circulation for dissemination to various organs, such as the liver and spleen (Wheat and Kauffman, 2003).
The therapeutic approach to patients with histoplasmosis is well documented in a 2007 “practice guideline” by the Infectious Diseases Society of America (Wheat et al., 2007). Azole drugs, such as itraconazole and voriconazole, and amphotericin B are the drugs of choice for clinically significant disease. However, as detailed above, these potent therapeutics fail to prevent mortality in a significant proportion (∼10%) of hospitalized patients. Additionally, the antifungal agents are given for protracted periods or even life-long in settings of ongoing immunocompromise. Hence, new therapeutic approaches have been investigated. One of the different avenues of study has been the application of antibodies to modify the pathogenesis of histoplasmosis.
Antibodies in Histoplasmosis
Passive immunization with polyclonal antibodies is controversial for mycoses (Louria and Kaminski, 1965). However, there is a growing consensus that antibodies collaborate with phagocytic cells and T cells for the enhancement of the immune response during systemic mycosis (Yuan et al., 1997; Vecchiarelli and Casadevall, 1998; Huffnagle and Deepe, 2003). Moreover, experiments with polyclonal sera have produced conflicting results that have raised questions regarding antibody efficacy in fungal disease (Louria and Kaminski, 1965; Mukherjee et al., 1992; Casadevall, 1998). In the 1970s, adoptive transfer experiments of serum from mice immunized with Hc ribosomes or live yeast cells failed to protect mice infected with the fungus, whereas transfer of filtered spleen or peritoneal cells from the immunized animals were protective (Tewari et al., 1977).
Recently, studies with monoclonal antibodies (mAbs) strongly have suggested that divergent results obtained with polyclonal antibodies preparations may be a result of the relative proportions of protective, non-protective, and inhibitory antibodies in immune sera (Dromer et al., 1987; Mukherjee et al., 1992) since animal experiments with mAbs to the capsular polysaccharide of Cryptococcus neoformans (Cn) have revealed the existence of protective, non-protective, and disease-enhancing mAbs. A protective mAb may even have a reduced efficacy if administered in high amounts, due to a prozone-like effect (Taborda and Casadevall, 2001; Taborda et al., 2003). In addition, protective efficacy of mAbs is determined by several variables including pathogen inoculum (Taborda et al., 2003), genetic background of both microbe (Mukherjee et al., 1995b) and host (Lendvai and Casadevall, 1999; Zaragoza et al., 2007), host immunological status (Yuan et al., 1997), both epitope specificity and isotype of mAb (Mukherjee et al., 1992, 1995a), timing of antibody administration (Casadevall, 1998; Casadevall et al., 1998), and route of infection (Briles et al., 1992a,b).
The antibody response to infection with H. capsulatum has been characterized. In humans, infection induces an increase in IgM by 2 weeks, followed by rising titers of IgA and IgG (Chandler et al., 1969). The IgG fraction contains complement-fixing and precipitating antibodies (Chandler et al., 1969). Murine experiments show that Histoplasma-specific serum immunoglobulin levels peak by day 21 (Fojtasek et al., 1993). Studies on the inflammatory reactions in the lungs of mice infected with H. capsulatum demonstrate that the number of B cells increase in the first week of infection, albeit to a lesser degree than other myeloid cells lines (Fojtasek et al., 1993; Cain and Deepe, 1998). Subsequently, the number of B cells continues to increase as other myeloid lines decrease (Cain and Deepe, 1998). The number of B cells in the spleen does not significantly change until the end of the second week of infection when all cell subsets nearly double (Fojtasek et al., 1993). In histoplasmosis, the current paradigm for host control of infection relies most heavily on activation of cellular immunity, since, in the absence of effector cells, progressive disease with dissemination occurs (Allendorfer et al., 1999). However, B-lymphocytes can impact histoplasmosis. Depletion of CD4+ and CD8+ T cells in B-lymphocyte knockout mice induced a markedly higher H. capsulatum burden in organs when compared with T cell depletion in wild-type animals in a secondary histoplasmosis model (Allen and Deepe, 2006), corroborating previous studies showing that antibodies can modify the pathogenesis of mycoses (Pirofski and Casadevall, 1996; Casadevall, 1998; Casadevall et al., 1998).
Histoplasma capsulatum Fungal Cell Surface: Targets for Antibody
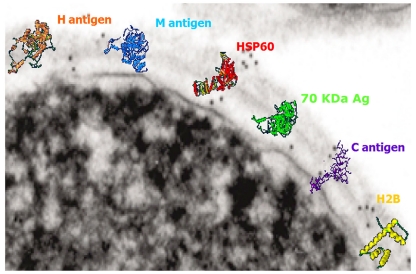
The fungal cell wall is rich in targets for the immune system (Nimrichter et al., 2005). As with most other pathogenic fungi, H. capsulatum possesses a rigid, polysaccharide composed cell wall structure, with four different glycans: soluble galactomannan, α-1,3-glucan, β-1,3-glucan, and a fibrillar chitin skeleton (Domer, 1971; San-Blas et al., 1978). Displayed on the surface are a variety of proteins that have been associated with virulence or utilized in diagnosis of the fungus (Figure 1). MAbs have been generated that bind H. capsulatum cell wall antigens, including melanin (Nosanchuk et al., 2002), histone 2B (Nosanchuk et al., 2003), Hsp60 (Guimaraes et al., 2009), M antigen (Guimaraes et al., 2008), and a 70-kDa protein (Lopes et al., 2010).
Figure 1.
Transmission electron micrograph of the surface of a H. capsulatum yeast cell with overlaying cartoons depicting the hypothetical structures of the H antigen, M antigen, heat shock protein 60, 70 kDa antigen, C antigen, and histone 2B. The antigen structures are based on molecular modeling as described in Guimaraes et al. (2008).
Histone 2B
Immunization with heat-killed H. capsulatum yeast cells resulted in the generation of a panel of IgM isotype mAbs that specifically bound cell surface expressed histone 2B (Nosanchuk et al., 2003). Although the identification of histone 2B as a cell surface antigen was surprising, histones are increasingly described on the cell surface of diverse host cells as well as pathogens; such as the cell surface of Mycobacterium leprae (Pessolani et al., 1993; Marques et al., 2000, 2001) and Mycobacterium smegmatis (Pethe et al., 2001) that are associated with binding to host cells and, specifically in the case of M. leprae, cell invasion (Shimoji et al., 1999). The administration of the IgM mAbs to H. capsulatum histone 2B was hindered by the large molecular weight of the mAbs and the difficulty with their entry into the alveolar space. However, co-incubation of mAb with H. capsulatum yeast cells prior to infection of mice revealed that the mAbs reduced the severity of disease. The protective effect was augmented by the administration of sub-inhibitory concentrations of amphotericin B, which stimulates host immune responses by engaging TLR2 and CD14 (Sau et al., 2003). Overall, the IgM mAbs to histone 2B reduced fungal burdens, decreased pulmonary inflammatory damage, and prolonged murine survival.
The protective effect of these mAbs was associated with an alteration in the intracellular fate of H. capsulatum (Nosanchuk et al., 2003; Shi et al., 2008). The mAbs reduced the capacity of the fungus to replicate intracellularly. Although H. capsulatum typically tightly regulates the phagosomal pH of macrophages, yeast opsonized with mAb to histone 2B were unable to maintain a neutral pH in their host macrophage cells. The inability to regulate the pH resulted in fungal cell killing, increased processing of fungal antigens and increased T cell activation (Shi et al., 2008).
Heat Shock Protein 60
As described above, Hsp60 is the surface protein that is the major ligand that mediates attachment of H. capsulatum to macrophage CR3 (CD11b/CD18; Long et al., 2003). Hsp60 is also an immunodominant antigen expressed on the surface of H. capsulatum yeasts and is able to induce protective immunity upon vaccination (Deepe et al., 1996; Deepe and Gibbons, 2002; Scheckelhoff and Deepe, 2002). Using recombinant Hsp60, we generated IgG isotype mAbs (Guimaraes et al., 2009). Interestingly, IgG2a and IgG1 mAbs to Hsp60 were protective whereas a IgG2b was disease-enhancing. Protection was characterized by a reduction of fungal burden, decreased tissue damage, and prolongation of survival. Cytokine analyses revealed that the protective mAbs induced a strong Th1-type host response. Notably, the IgG2b recognized the same region on the protein to which a protective IgG1 also bound in a competitive manner, suggesting that protection was regulated by isotype. This finding was supported by data from mice treated with methamphetamine that developed significant increases in their IgG2b levels and also had accelerated and exacerbated histoplasmosis (Martinez et al., 2009).
As with the mAbs to histone 2B, the protective mAbs to Hsp60 modified the intracellular fate of the yeast cells. Phagosomal maturation was significantly increased in the presence of the protective mAbs and this correlated with a reduction in intracellular yeast cell survival. In contrast, yeast cultured with the disease-enhancing IgG2b mAb replicated at an enhanced rate compared to controls.
Interestingly, the protective mAbs were noted to induce aggregation of H. capsulatum yeast cells (Guimaraes et al., 2011a). Agglutination of yeast cells caused by antibodies had been reported previously for C. neoformans (Kozel and McGaw, 1979). However, agglutination was evaluated with IgG-opsonized strain 602 of C. neoformans, by using a monospecific antiserum to IgG heavy chains, in order to confirm association of IgG antibodies to the yeast and specifically determine that these molecules were binding directly at the yeast surface. With H. capsulatum we observed that agglutination occurred only when cells are brought together due to a result of a Brownian movement during which cellular collision permits interaction. However, the surface of H. capsulatum yeast is typically negatively charged due to the high amounts of α-1,3-glucans on the cell wall. The negative charge increases the electrostatic potential surrounding the cells leading to repulsion between cells. H. capsulatum yeast aggregation was an effect of concentration, but the magnitude of aggregation efficiency was dependent on the dissociation constant of each mAb characterized. Additionally, we used an optical tweezer to measure real-time interactions between single cells in the presence of opsonins and found a correlation of time for aggregation (characteristic time) and binding constant, with the protective mAb being more effective than the non-protective mAb. Interestingly, blockade of CR3 receptors resulted in an additional drop of phagocytosis rate of larger aggregates, suggesting a cooperative function of Fc and CR3 receptor for the phagocytosis of large particles. Overall, it is unclear what the impact of agglutination potential of the antibodies is during infection. However, it is possible that the antibodies may keep replicating cells agglutinated, which can reduce the dissemination of the fungus, and these clusters of cells may be more effectively targeted by host responses.
We have also recently demonstrated that Hsp60 is involved in the presentation of a diverse range of proteins on the fungal cell surface, including proteins associated with oxidative stress responses (Guimaraes et al., 2011c). Hence, the binding of Hsp60 by antibody may dysregulate the chaperone functions of the protein. Disruption of this interactome, especially during stress response conditions, could impact the capacity of the fungus to cause disease.
M Antigen
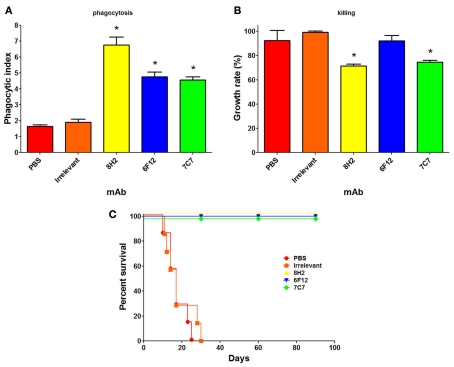
The M antigen is a glycoprotein that is well known as a diagnostic antigen for acute histoplasmosis as it induces the first precipitins during disease (Pizzini et al., 1999). We have documented that live fungal cells secrete only small amounts of the protein and interestingly, we demonstrated that the M antigen was also expressed at the H. capsulatum cell surface and that it functions as a catalase (Guimaraes et al., 2008). In the course of this study, we produced three mAbs to the M antigen, one IgM and two IgG2a isotype mAbs. Opsonization of H. capsulatum with the IgM or IgG2a mAbs to the M antigen enhances yeast cell phagocytosis by macrophages and two of the three mAbs also promote host cell cidal activity (Figures 2A,B). All three mAbs also altered the pathogenesis of experimental murine histoplasmosis as mice challenged with opsonized yeast cells uniformly survived a lethal challenge with H. capsulatum (Figure 2C).
Figure 2.
Monoclonal antibodies (mAbs) to the M antigen can modify the pathogenesis of experimental histoplasmosis. The generation of the mAbs is described in Guimaraes et al. (2008). The mAbs produced are mAb 7C7 (IgM isotype), mAb 6F12 (IgG2a), and mAb 8H2 (IgG2a). Using methods described in Guimaraes et al. (2011b), the phagocytosis rates (A), and killing capacity (B) of J774.16 macrophage-like cells reveal that the mAbs significantly enhanced the uptake of yeast cells by the phagocytes (p < 0.05 vs. controls) and that two of the three mAbs enabled the killing of yeast cells by the J774.16 cells (p < 0.05 for mAbs 8H2 and 7C7 vs. controls). (C) C57Bl/6 mice infected with 1.25 × 107 opsonized yeast cells had a 100% survival whereas control infected mice died within 1 month after infection (p < 0.001 for mAbs 8H2, 6F12, and 7C7 vs. controls).
70 kDa Antigen
In addition to the fact that the IgG2b mAb to Hsp60 was not protective, we have demonstrated that an IgG1 mAb specific for a 70-kDa cell surface antigen was non-protective (Lopes et al., 2010). The mAb was previously shown to be highly specific for H. capsulatum and is a candidate for use in the serological diagnosis and management of histoplasmosis (Gomez et al., 1997, 1999). The finding that the IgG1 to the 70-kDa antigen was non-protective indicates that isotype is not the only determinant for a protective response, but that the antigen target can also influence the outcome of disease.
Discussion
The incidence of clinically relevant mycoses is rising, mainly due to recent advances in modern medicine, including the use of intravascular devices, broad spectrum antibiotics, organ transplantation, the use of chemotherapeutic and anti-inflammatory drugs, and the increasing number of individuals with HIV infection (Pfaller and Diekema, 2010). Given the difficulties in combating severe fungal diseases with our current antifungal medications, novel approaches, including administration of mAbs, are being pursued. Studies with antibody and H. capsulatum reveal that protective and non-protective antibodies exist. This finding is also extremely relevant to vaccine development, as an adverse outcome of vaccination might be the generation of disease-enhancing antibodies, even if the latter are a subset of those induced.
The protective mAbs to H. capsulatum identified to date mediate protection by modifying the intracellular fate of the yeast cells. H. capsulatum is remarkable for its capacity to tightly regulate the intracellular milieu of macrophage phagosomes. The protective mAbs to histone 2B and Hsp60 enhance macrophage fungistatic/fungicidal responses, subverted H. capsulatum’s ability to modify the phagosomal environment, resulting in augmented antigen processing and T cell activation, and control of the fungal disease. The opsonization by the antibody on H. capsulatum can also impact the fungus’ ability to traffic surface proteins. Future research is needed to better characterize the fundamental influences on protection of antibody in histoplasmosis, including defining the importance of isotype on the protective efficacy, determining the role of the quantity of surface antigen on protection, and the direct effect of antibody on H. capsulatum transcriptional regulation.
In addition to exploring fungal antigens as targets for antibody therapy, we have examined the impact of antibody on host cell co-stimulation as a new alternative for controlling the pathogenesis of histoplasmosis. We determined that H. capsulatum altered the PD-L expression on macrophages, which resulted in a dysregulation of T cell activation (Lazar-Molnar et al., 2008). The PD-1/PD-L pathway is involved in maintenance of self-tolerance and T cell regulation. Mice deficient in PD-1 were protected against lethal challenges of H. capsulatum. Administration of mAb that blocks PD-1 similarly protected wild-type mice against lethal infection. Hence, antibody targeting host responses can modify histoplasmosis.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
- Allen H. L., Deepe G. S., Jr. (2006). B cells and CD4-CD8-T cells are key regulators of the severity of reactivation histoplasmosis. J. Immunol. 177, 1763–1771 [DOI] [PubMed] [Google Scholar]
- Allendoerfer R., Biovin G. P., Deepe G. S., Jr. (1997). Modulation of immune responses in murine pulmonary histoplasmosis. J. Infect. Dis. 175, 905–914 10.1086/513989 [DOI] [PubMed] [Google Scholar]
- Allendorfer R., Brunner G. D., Deepe G. S., Jr. (1999). Complex requirements for nascent and memory immunity in pulmonary histoplasmosis. J. Immunol. 162, 7389–7396 [PubMed] [Google Scholar]
- Alteras I. (1966). First Romanian isolation of Histoplasma capsulatum from the soil. Dermatol. Int. 5, 69–71 10.1111/j.1365-4362.1966.tb05188.x [DOI] [PubMed] [Google Scholar]
- Bradsher R. W. (1996). Histoplasmosis and blastomycosis. Clin. Infect. Dis. 22, S102–S111 10.1093/clinids/22.Supplement_2.S102 [DOI] [PubMed] [Google Scholar]
- Briles D. E., Crain M. J., Gray B. M., Forman C., Yother J. (1992a). Strong association between capsular type and virulence for mice among human isolates of Streptococcus pneumoniae. Infect. Immun. 60, 111–116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Briles D. E., Forman C., Crain M. (1992b). Mouse antibody to phosphocholine can protect mice from infection with mouse-virulent human isolates of Streptococcus pneumoniae. Infect. Immun. 60, 1957–1962 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cain J. A., Deepe G. S., Jr. (1998). Evolution of the primary immune response to Histoplasma capsulatum in murine lung. Infect. Immun. 66, 1473–1481 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Casadevall A. (1998). Antibody-mediated protection against intracellular pathogens. Trends Microbiol. 6, 102–107 10.1016/S0966-842X(98)01208-6 [DOI] [PubMed] [Google Scholar]
- Casadevall A., Cassone A., Bistoni F., Cutler J. E., Magliani W., Murphy J. W., Polonelli L., Romani L. (1998). Antibody and/or cell-mediated immunity, protective mechanisms in fungal disease: an ongoing dilemma or an unnecessary dispute? Med. Mycol. 36(Suppl. 1), 95–105 [PubMed] [Google Scholar]
- Chandler J. W., Jr., Smith T. K., Newberry W. M., Jr., Chin T. D., Kirkpatrick C. H. (1969). Immunology of the mycoses. II. Characterization of the immunoglobulin and antibody responses in histoplasmosis. J. Infect. Dis. 119, 247–254 10.1093/infdis/119.3.247 [DOI] [PubMed] [Google Scholar]
- Chu J. H., Feudtner C., Heydon K., Walsh T. J., Zaoutis T. E. (2006). Hospitalizations for endemic mycoses: a population-based national study. Clin. Infect. Dis. 42, 822–825 10.1086/500405 [DOI] [PubMed] [Google Scholar]
- Darling S. T. (1906). A protozoan general infection producing pseudotubercles in the lungs and focal necrosis in the liver, spleen and lymph nodes. J. Am. Med. Assoc. 46, 1283–1285 10.1001/jama.1906.62510440037003 [DOI] [Google Scholar]
- Deepe G. S., Jr. (2005). Modulation of infection with Histoplasma capsulatum by inhibition of tumor necrosis factor-alpha activity. Clin. Infect. Dis. 41(Suppl. 3), S204–S207 10.1086/498035 [DOI] [PubMed] [Google Scholar]
- Deepe G. S., Jr., Gibbons R., Brunner G. D., Gomez F. J. (1996). A protective domain of heat-shock protein 60 from Histoplasma capsulatum. J. Infect. Dis. 174, 828–834 10.1093/infdis/174.4.828 [DOI] [PubMed] [Google Scholar]
- Deepe G. S., Jr., Gibbons R. S. (2002). Cellular and molecular regulation of vaccination with heat shock protein 60 from Histoplasma capsulatum. Infect. Immun. 70, 3759–3767 10.1128/IAI.70.7.3759-3767.2002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deepe G. S., Jr., Gibbons R. S., Smulian A. G. (2008). Histoplasma capsulatum manifests preferential invasion of phagocytic subpopulations in murine lungs. J. Leukoc. Biol. 84, 669–678 10.1189/jlb.0308154 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deepe G. S., Jr., Smelt S., Louie J. S. (2005). Tumor necrosis factor inhibition and opportunistic infections. Clin. Infect. Dis. 41(Suppl. 3), S187–S188 10.1086/498035 [DOI] [PubMed] [Google Scholar]
- Disalvo A. F., Bigler W. J., Ajello L., Johnson J. E., Palmer J. (1970). Bat and soil studies for sources of histoplasmosis in Florida. Public Health Rep. 85, 1063–1069 10.2307/4594044 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dodd K., Thompkins E. H. (1934). Case of histoplasmosis of darling in a infant. Am. J. Trop. Med. Hyg. 14, 127–137 [Google Scholar]
- Domer J. E. (1971). Monosaccharide and chitin content of cell walls of Histoplasma capsulatum and Blastomyces dermatitidis. J. Bacteriol. 107, 870–877 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dromer F., Charreire J., Contrepois A., Carbon C., Yeni P. (1987). Protection of mice against experimental cryptococcosis by anti-Cryptococcus neoformans monoclonal antibody. Infect. Immun. 55, 749–752 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edwards L. B., Acquaviva F. A., Livesay V. T., Cross F. W., Palmer C. E. (1969). An atlas of sensitivity to tuberculin, PPD-B, and histoplasmin in the United States. Am. Rev. Respir. Dis. 99(Suppl.), 1–132 [PubMed] [Google Scholar]
- Eissenberg L. G., Goldman W. E. (1987). Histoplasma capsulatum fails to trigger release of superoxide from macrophages. Infect. Immun. 55, 29–34 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Emmons C. W. (1950). Histoplasmosis: animal reservoirs and other sources in nature of the pathogenic fungus Histoplasma capsulatum. Am. J. Public Health 40, 436–440 10.2105/AJPH.40.4.436 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Emmons C. W. (1956a). Histoplasmosis in animals. Public Health Monogr. 70, 272–273 [PubMed] [Google Scholar]
- Emmons C. W. (1956b). Isolation of Histoplasma capsulatum from soil. Public Health Monogr. 70, 237–239 [PubMed] [Google Scholar]
- Emmons C. W., Klite P. D., Baer G. M., Hill W. B., Jr. (1966). Isolation of Histoplasma capsulatum from bats in the United States. Am. J. Epidemiol. 84, 103–109 [DOI] [PubMed] [Google Scholar]
- Fojtasek M. F., Kleiman M. B., Connolly-Stringfield P., Blair R., Wheat L. J. (1994). The Histoplasma capsulatum antigen assay in disseminated histoplasmosis in children. Pediatr. Infect. Dis. J. 13, 801–805 10.1097/00006454-199409000-00010 [DOI] [PubMed] [Google Scholar]
- Fojtasek M. F., Sherman M. R., Garringer T., Blair R., Wheat L. J., Schnizlein-Bick C. T. (1993). Local immunity in lung-associated lymph nodes in a murine model of pulmonary histoplasmosis. Infect. Immun. 61, 4607–4614 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gomez B. L., Figueroa J. I., Hamilton A. J., Diez S., Rojas M., Tobon A., Restrepo A., Hay R. J. (1999). Detection of the 70-kilodalton Histoplasma capsulatum antigen in serum of histoplasmosis patients: correlation between antigenemia and therapy during follow-up. J. Clin. Microbiol. 37, 675–680 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gomez B. L., Figueroa J. I., Hamilton A. J., Ortiz B. L., Robledo M. A., Restrepo A., Hay R. J. (1997). Development of a novel antigen detection test for histoplasmosis. J. Clin. Microbiol. 35, 2618–2622 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goodwin R. A., Jr., Des Prez R. M. (1978). State of the art: histoplasmosis. Am. Rev. Respir. Dis. 117, 929–956 [DOI] [PubMed] [Google Scholar]
- Goodwin R. A., Loyd J. E., Des Prez R. M. (1981). Histoplasmosis in normal hosts. Medicine (Baltimore) 60, 231–266 [DOI] [PubMed] [Google Scholar]
- Guimaraes A. J., Frases S., Gomez F. J., Zancope-Oliveira R. M., Nosanchuk J. D. (2009). Monoclonal antibodies to heat shock protein 60 alter the pathogenesis of Histoplasma capsulatum. Infect. Immun. 77, 1357–1367 10.1128/IAI.01443-08 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guimaraes A. J., Hamilton A. J., De M. G. H. L., Nosanchuk J. D., Zancope-Oliveira R. M. (2008). Biological function and molecular mapping of M antigen in yeast phase of Histoplasma capsulatum. PLoS ONE 3, e3449 10.1371/journal.pone.0003449 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guimaraes A. J., Frases S., Pontes B., De Cerqueira M. D., Rodrigues M. L., Viana N. B., Nimrichter L., Nosanchuk J. D. (2011a). Agglutination of Histoplasma capsulatum by IgG monoclonal antibodies against Hsp60 impacts macrophage effector functions. Infect. Immun. 79, 918–927 10.1128/IAI.00673-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guimaraes A. J., Martinez L. R., Nosanchuk J. D. (2011b). Passive administration of monoclonal antibodies against H. capsulatum and other fungal pathogens. J. Vis. Exp. Available at: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=21372781 [DOI] [PMC free article] [PubMed]
- Guimaraes A. J., Nakayasu E. S., Sobreira T. J., Cordero R. J., Nimrichter L., Almeida I. C., Nosanchuk J. D. (2011c). Histoplasma capsulatum heat-shock 60 orchestrates the adaptation of the fungus to temperature stress. PLoS ONE 6, e14660. 10.1371/journal.pone.0014660 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guimaraes A. J., Nosanchuk J. D., Zancope-Oliveira R. M. (2006). Diagnosis of histoplasmosis. Braz. J. Microbiol. 37, 1–13 10.1590/S1517-83822006000100001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Habich C., Kempe K., Gomez F. J., Lillicrap M., Gaston H., Van der Zee R., Kolb H., Burkart V. (2006). Heat shock protein 60: identification of specific epitopes for binding to primary macrophages. FEBS Lett. 580, 115–120 10.1016/j.febslet.2005.11.060 [DOI] [PubMed] [Google Scholar]
- Huffnagle G. B., Deepe G. S. (2003). Innate and adaptive determinants of host susceptibility to medically important fungi. Curr. Opin. Microbiol. 6, 344–350 10.1016/S1369-5274(03)00089-4 [DOI] [PubMed] [Google Scholar]
- Isbister J., Elliott M., Nogrady S. (1976). Histoplasmosis: an outbreak occurring among young men who visited one cave. Med. J. Aust. 2, 243–248 [DOI] [PubMed] [Google Scholar]
- Kasuga T., White T. J., Koenig G., Mcewen J., Restrepo A., Castaneda E., Da Silva Lacaz C., Heins-Vaccari E. M., De Freitas R. S., Zancope-Oliveira R. M., Qin Z., Negroni R., Carter D. A., Mikami Y., Tamura M., Taylor M. L., Miller G. F., Poonwan N., Taylor J. W. (2003). Phylogeography of the fungal pathogen Histoplasma capsulatum. Mol. Ecol. 12, 3383–3401 10.1046/j.1365-294X.2003.01995.x [DOI] [PubMed] [Google Scholar]
- Kauffman C. A. (2007). Histoplasmosis: a clinical and laboratory update. Clin. Microbiol. Rev. 20, 115–132 10.1128/CMR.00027-06 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kozel T. R., McGaw T. G. (1979). Opsonization of Cryptococcus neoformans by human immunoglobulin G: role of immunoglobulin G in phagocytosis by macrophages. Infect. Immun. 25, 255–261 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lazar-Molnar E., Gacser A., Freeman G. J., Almo S. C., Nathenson S. G., Nosanchuk J. D. (2008). The PD-1/PD-L costimulatory pathway critically affects host resistance to the pathogenic fungus Histoplasma capsulatum. Proc. Natl. Acad. Sci. U.S.A. 105, 2658–2663 10.1073/pnas.0804453105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lendvai N., Casadevall A. (1999). Monoclonal antibody-mediated toxicity in Cryptococcus neoformans infection: mechanism and relationship to antibody isotype. J. Infect. Dis. 180, 791–801 10.1086/314946 [DOI] [PubMed] [Google Scholar]
- Limaye A. P., Connolly P. A., Sagar M., Fritsche T. R., Cookson B. T., Wheat L. J., Stamm W. E. (2000). Transmission of Histoplasma capsulatum by organ transplantation. N. Engl. J. Med. 343, 1163–1166 10.1056/NEJM200010193431605 [DOI] [PubMed] [Google Scholar]
- Lockhart S. R., Diekema D. J., Pfaller M. A. (2009). “The epidemiology of fungal infections,” in Clinical Mycology, eds Anaissie E. J., McGinnis M. R., Pfaller M. A. (Oxford: Elsevier Inc; ) [Google Scholar]
- Long K. H., Gomez F. J., Morris R. E., Newman S. L. (2003). Identification of heat shock protein 60 as the ligand on Histoplasma capsulatum that mediates binding to CD18 receptors on human macrophages. J. Immunol. 170, 487–494 [DOI] [PubMed] [Google Scholar]
- Lopes L. C., Guimaraes A. J., De Cerqueira M. D., Gomez B. L., Nosanchuk J. D. (2010). A Histoplasma capsulatum-specific IgG1 isotype monoclonal antibody, H1C, to a 70-kilodalton cell surface protein is not protective in murine histoplasmosis. Clin. Vaccine Immunol. 17, 1155–1158 10.1128/CVI.00033-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Louria D. B., Kaminski T. (1965). Passively-acquired immunity in experimental cryptococcosis. Sabouraudia 4, 80–84 10.1080/00362176685190211 [DOI] [PubMed] [Google Scholar]
- Maresca B., Kobayashi G. S. (1989). Dimorphism in Histoplasma capsulatum: a model for the study of cell differentiation in pathogenic fungi. Microbiol. Rev. 53, 186–209 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marques M. A. M., Mahapatra S., Nandan D., Dick T., Sarno E. N., Pessolani M. C. V. (2000). Bacterial and host-derived cationic proteins bind a2-laminins and enhance Mycobacterium leprae attachment to human Schwann cells. Microbes Infect. 2, 1407–1417 10.1016/S1286-4579(00)01294-6 [DOI] [PubMed] [Google Scholar]
- Marques M. A. M., Mahapatra S., Sarno E. N., Santos S., Spencer J. S., Brennan P. J., Pessolani M. C. V. (2001). Further biochemical characterization of Mycobacterium leprae laminin-binding proteins. Braz. J. Med. Biol. Res. 34, 463–470 10.1590/S0100-879X2001000400004 [DOI] [PubMed] [Google Scholar]
- Martinez L. R., Mihu M. R., Gacser A., Santambrogio L., Nosanchuk J. D. (2009). Methamphetamine enhances histoplasmosis by immunosuppression of the host. J. Infect. Dis. 200, 131–141 10.1086/605060 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Medoff G., Kobayashi G. S., Painter A., Travis S. (1987). Morphogenesis and pathogenicity of Histoplasma capsulatum. Infect. Immun. 55, 1355–1358 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meloan E. L. (1952). Histoplasmosis. Miss. Doct. 29, 256–257 [PubMed] [Google Scholar]
- Mukherjee J., Nussbaum G., Scharff M. D., Casadevall A. (1995a). Protective and nonprotective monoclonal antibodies to Cryptococcus neoformans originating from one B cell. J. Exp. Med. 181, 405–409 10.1084/jem.181.1.405 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mukherjee J., Scharff M. D., Casadevall A. (1995b). Variable efficacy of passive antibody administration against diverse Cryptococcus neoformans strains. Infect. Immun. 63, 3353–3359 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mukherjee J., Scharff M. D., Casadevall A. (1992). Protective murine monoclonal antibodies to Cryptococcus neoformans. Infect. Immun. 60, 4534–4541 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newman S. L. (2005). Interaction of Histoplasma capsulatum with human macrophages, dendritic cells, and neutrophils. Methods Mol. Med. 118, 181–191 [DOI] [PubMed] [Google Scholar]
- Nimrichter L., Rodrigues M. L., Rodrigues E. G., Travassos L. R. (2005). The multitude of targets for the immune system and drug therapy in the fungal cell wall. Microbes Infect. 7, 789–798 10.1016/j.micinf.2005.03.002 [DOI] [PubMed] [Google Scholar]
- Nosanchuk J. D., Gomez B. L., Youngchim S., Diez S., Aisen P., Zancope-Oliveira R. M., Restrepo A., Casadevall A., Hamilton A. J. (2002). Histoplasma capsulatum synthesizes melanin-like pigments in vitro and during mammalian infection. Infect. Immun. 70, 5124–5131 10.1128/IAI.70.9.5124-5131.2002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nosanchuk J. D., Steenbergen J. N., Shi L., Deepe G. S., Jr., Casadevall A. (2003). Antibodies to a cell surface histone-like protein protect against Histoplasma capsulatum. J. Clin. Invest. 112, 1164–1175 10.1172/JCI200319361 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pessolani M. C., Hunter S. W., Brennan P. J. (1993). Relationship between host histones and armadillo-derived Mycobacterium leprae. Int. J. Lepr. Other Mycobact. Dis. 61, 381–388 [PubMed] [Google Scholar]
- Pethe K., Puech V., Daffe M., Josenhans C., Drobecq H., Locht C., Menozzi F. D. (2001). Mycobacterium smegmatis laminin-binding glycoprotein shares epitopes with Mycobacterium tuberculosis heparin-binding haemagglutinin. Mol. Microbiol. 39, 89–99 10.1046/j.1365-2958.2001.02206.x [DOI] [PubMed] [Google Scholar]
- Pfaller M. A., Diekema D. J. (2010). Epidemiology of invasive mycoses in North America. Crit. Rev. Microbiol. 36, 1–53 10.3109/10408410903241444 [DOI] [PubMed] [Google Scholar]
- Pirofski L. A., Casadevall A. (1996). Cryptococcus neoformans: paradigm for the role of antibody immunity against fungi? Zentralbl. Bakteriol. 284, 475–495 [DOI] [PubMed] [Google Scholar]
- Pizzini C. V., Zancope-Oliveira R. M., Reiss E., Hajjeh R., Kaufman L., Peralta J. M. (1999). Evaluation of a western blot test in an outbreak of acute pulmonary histoplasmosis. Clin. Diagn. Lab. Immunol. 6, 20–23 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prado M., Silva M. B., Laurenti R., Travassos L. R., Taborda C. P. (2009). Mortality due to systemic mycoses as a primary cause of death or in association with AIDS in Brazil: a review from 1996 to 2006. Mem. Inst. Oswaldo Cruz 104, 513–521 10.1590/S0074-02762009000300019 [DOI] [PubMed] [Google Scholar]
- Saliba A., Beatty O. A. (1960). Pulmonary histoplasmosis, importance of diagnostic methods, with report of an early case. JAMA 173, 902–904 10.1001/jama.1960.73020260007009c [DOI] [PubMed] [Google Scholar]
- San-Blas G., Ordaz D., Yegres F. J. (1978). Histoplasma capsulatum: chemical variability of the yeast cell wall. Sabouraudia 16, 279–284 [DOI] [PubMed] [Google Scholar]
- Sau K., Mambula S. S., Latz E., Henneke P., Golenbock D. T., Levitz S. M. (2003). The antifungal drug amphotericin B promotes inflammatory cytokine release by a Toll-like receptor- and CD14-dependent mechanism. J. Biol. Chem. 278, 37561–37568 10.1074/jbc.M306137200 [DOI] [PubMed] [Google Scholar]
- Scheckelhoff M., Deepe G. S., Jr. (2002). The protective immune response to heat shock protein 60 of Histoplasma capsulatum is mediated by a subset of V beta 8.1/8.2+ T cells. J. Immunol. 169, 5818–5826 [DOI] [PubMed] [Google Scholar]
- Scheckelhoff M., Deepe G. S., Jr. (2005). A deficiency in gamma interferon or interleukin-10 modulates T-cell-dependent responses to heat shock protein 60 from Histoplasma capsulatum. Infect. Immun. 73, 2129–2134 10.1128/IAI.73.4.2129-2134.2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi L., Albuquerque P. C., Lazar-Molnar E., Wang X., Santambrogio L., Gacser A., Nosanchuk J. D. (2008). A monoclonal antibody to Histoplasma capsulatum alters the intracellular fate of the fungus in murine macrophages. Eukaryot. Cell 7, 1109–1117 10.1128/EC.00036-08 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimoji Y., Ng V., Matsumura K., Fischetti V. A., Rambukkana A. (1999). A 21-kDa surface protein of Mycobacterium leprae binds peripheral nerve laminin-2 and mediates Schwann cell invasion. Proc. Natl. Acad. Sci. U.S.A. 96, 9857–9862 10.1073/pnas.96.17.9857 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith C. D. (1971a). “Nutritional factors that are required for the growth and sporulation of Histoplasma capsulatum,” in Histoplasmosis: Proceedings of the Second National Conference, eds Ajello L., Chick E. W., Furcolow M. L. (Springfield: C. Thomas; ), 64–70 [Google Scholar]
- Smith C. D. (1971b). “The role of birds in the ecology of Histoplasma capsulatum,” in Histoplasmosis: Proceedings of the Second National Conference, eds Ajello L., Chick E. W., Furcolow M. L. (Springfield: C. Thomas; ), 140–148 [Google Scholar]
- Strasser J. E., Newman S. L., Ciraolo G. M., Morris R. E., Howell M. L., Dean G. E. (1999). Regulation of the macrophage vacuolar ATPase and phagosome-lysosome fusion by Histoplasma capsulatum. J. Immunol. 162, 6148–6154 [PubMed] [Google Scholar]
- Taborda C. P., Casadevall A. (2001). Immunoglobulin M efficacy against Cryptococcus neoformans: mechanism, dose dependence, and prozone-like effects in passive protection experiments. J. Immunol. 166, 2100–2107 [DOI] [PubMed] [Google Scholar]
- Taborda C. P., Rivera J., Zaragoza O., Casadevall A. (2003). More is not necessarily better: prozone-like effects in passive immunization with IgG. J. Immunol. 170, 3621–3630 [DOI] [PubMed] [Google Scholar]
- Tewari R. P., Sharma D., Solotorovsky M., Lafemina R., Balint J. (1977). Adoptive transfer of immunity from mice immunized with ribosomes or live yeast cells of Histoplasma capsulatum. Infect. Immun. 15, 789–795 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vecchiarelli A., Casadevall A. (1998). Antibody-mediated effects against Cryptococcus neoformans: evidence for interdependency and collaboration between humoral and cellular immunity. Res. Immunol. 149, 321–333 10.1016/S0923-2494(98)80756-6 [DOI] [PubMed] [Google Scholar]
- Wheat L. J., Freifeld A. G., Kleiman M. B., Baddley J. W., Mckinsey D. S., Loyd J. E., Kauffman C. A. (2007). Clinical practice guidelines for the management of patients with histoplasmosis: 2007 update by the Infectious Diseases Society of America. Clin. Infect. Dis. 45, 807–825 10.1086/521259 [DOI] [PubMed] [Google Scholar]
- Wheat L. J., Kauffman C. A. (2003). Histoplasmosis. Infect. Dis. Clin. North Am. 17, 1–19 10.1016/S0891-5520(02)00039-9 [DOI] [PubMed] [Google Scholar]
- Yuan R. R., Casadevall A., Oh J., Scharff M. D. (1997). T cells cooperate with passive antibody to modify Cryptococcus neoformans infection in mice. Proc. Natl. Acad. Sci. U.S.A. 94, 2483–2488 10.1073/pnas.94.6.2483 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zaragoza O., Alvarez M., Telzak A., Rivera J., Casadevall A. (2007). The relative susceptibility of mouse strains to pulmonary Cryptococcus neoformans infection is associated with pleiotropic differences in the immune response. Infect. Immun. 75, 2729–2739 10.1128/IAI.00094-07 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zeidberg L. D., Ajello L., Dillon A., Runyon L. C. (1952). Isolation of Histoplasma capsulatum from soil. Am. J. Public Health 42, 930–935 10.2105/AJPH.42.8.930 [DOI] [PMC free article] [PubMed] [Google Scholar]