Abstract
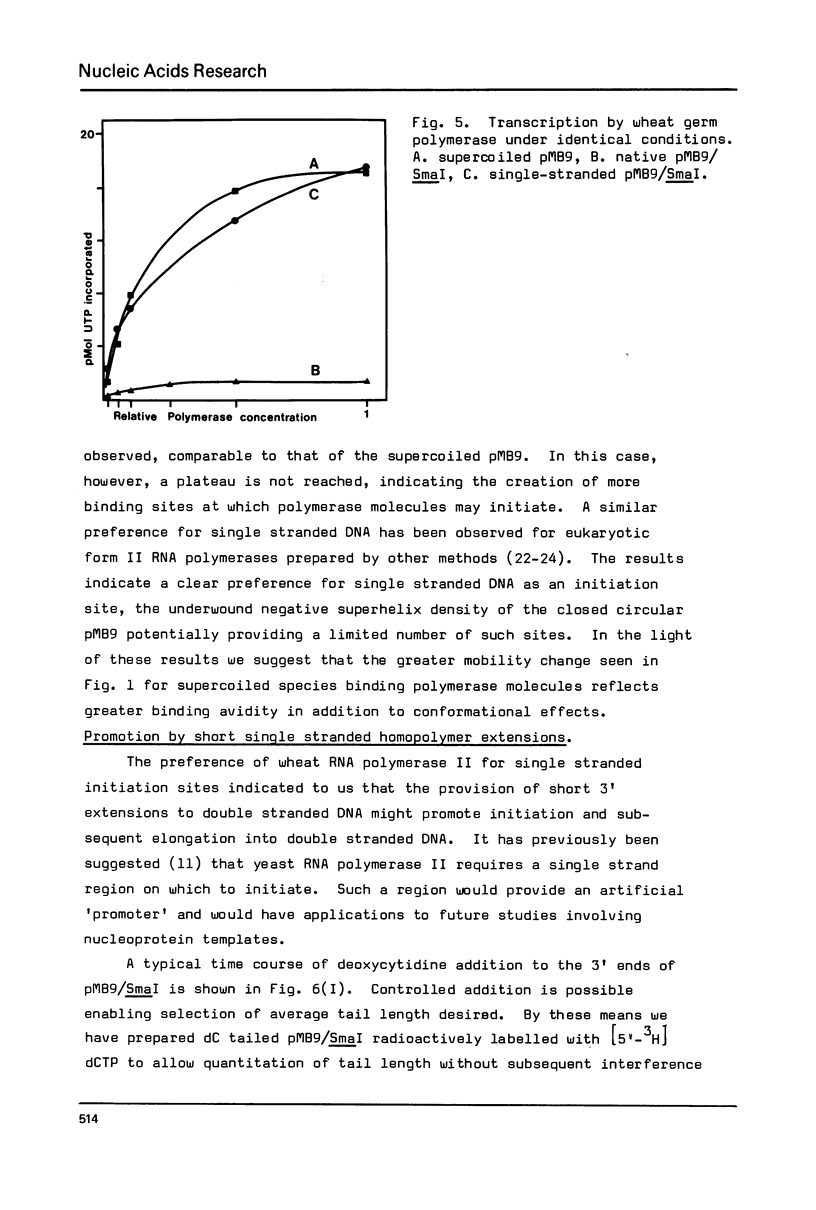
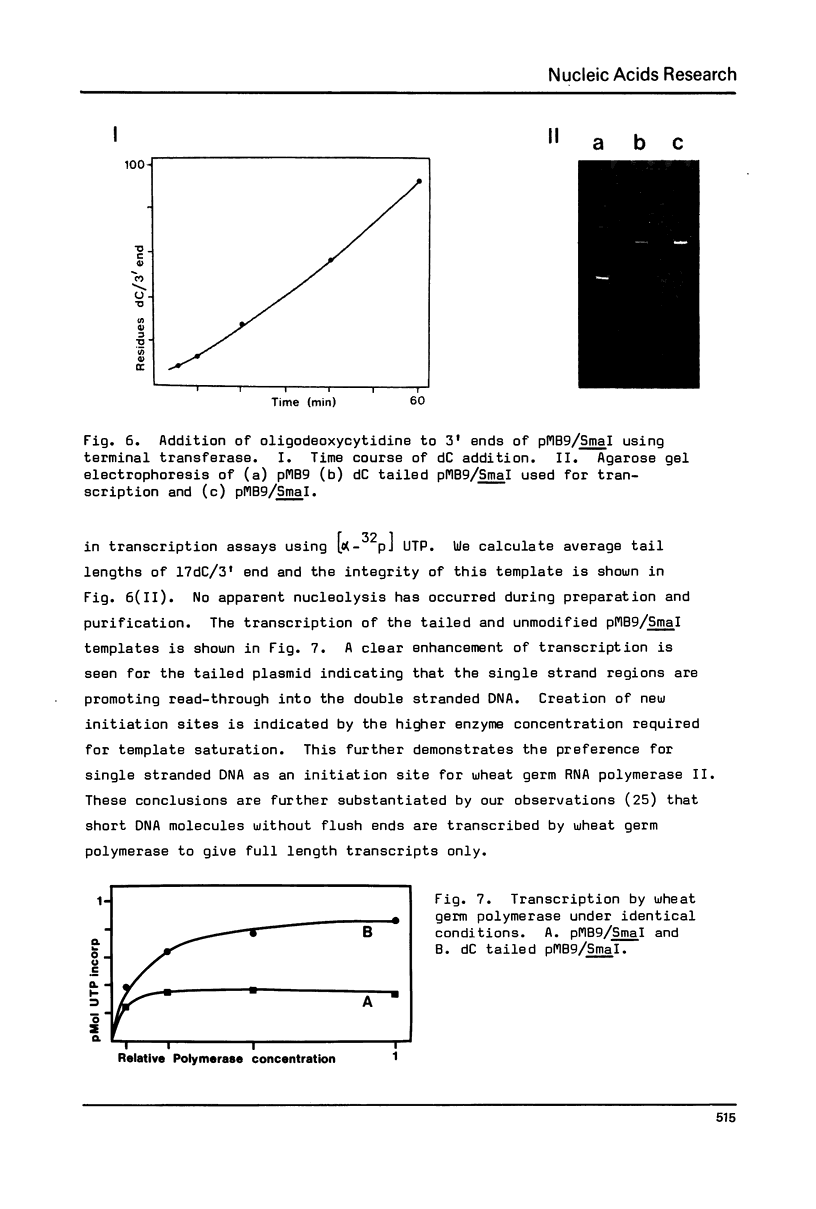
Interactions between a plant RNA polymerase II and ColE1 based plasmid DNA templates have been studied. Gel electrophoresis indicates that the enzyme binds to both supercoiled and linear species. Using the totally double stranded pMB9/SmaI fragment it is shown that transcription of completely base paired DNA is ten-fold lower than that of denatured or supercoiled plasmid, and reflects the presence of fewer initiation sites. A small proportion of the transcript remains tightly bound to supercoiled templates. 3' oligodeoxycytidine extensions on pMB9/SmaI serve to promote transcription of the linear double stranded form. Using restriction kinetics it is shown that there is a small enhancement of polymerase binding at the pMB9 tetracycline promoter, but that the selectivity of binding at this locus is lower than for the natural bacterial polymerase.
Full text
PDF














Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Beard P., Morrow J. F., Berg P. Cleavage of circular, superhelical simian virus 40 DNA to a linear duplex by S1 nuclease. J Virol. 1973 Dec;12(6):1303–1313. doi: 10.1128/jvi.12.6.1303-1313.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beebee T. J., Butterworth P. H. Transcription specificity of Xenopus laevis RNA polymerase A. FEBS Lett. 1974 Oct 15;47(2):304–306. doi: 10.1016/0014-5793(74)81035-5. [DOI] [PubMed] [Google Scholar]
- Benyajati C., Worcel A. Isolation, characterization, and structure of the folded interphase genome of Drosophila melanogaster. Cell. 1976 Nov;9(3):393–407. doi: 10.1016/0092-8674(76)90084-2. [DOI] [PubMed] [Google Scholar]
- Bolivar F., Rodriguez R. L., Betlach M. C., Boyer H. W. Construction and characterization of new cloning vehicles. I. Ampicillin-resistant derivatives of the plasmid pMB9. Gene. 1977;2(2):75–93. doi: 10.1016/0378-1119(77)90074-9. [DOI] [PubMed] [Google Scholar]
- Botchan P., Wang J. C., Echols H. Effect of circularity and superhelicity on transcription from bacteriophagelambda DNA. Proc Natl Acad Sci U S A. 1973 Nov;70(11):3077–3081. doi: 10.1073/pnas.70.11.3077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown K. D., Bennett G. N., Lee F., Schweingruber M. E., Yanofsky C. RNA polymerase interaction at the promoter--operator region of the tryptophan operon of Escherichia coli and Salmonella typhimurium. J Mol Biol. 1978 May 15;121(2):153–177. doi: 10.1016/s0022-2836(78)80003-5. [DOI] [PubMed] [Google Scholar]
- Burgess R. R., Jendrisak J. J. A procedure for the rapid, large-scall purification of Escherichia coli DNA-dependent RNA polymerase involving Polymin P precipitation and DNA-cellulose chromatography. Biochemistry. 1975 Oct 21;14(21):4634–4638. doi: 10.1021/bi00692a011. [DOI] [PubMed] [Google Scholar]
- Campbell A. M. Conformational analysis of deoxyribonucleic acid from PM2 bacteriophage. The effect of size on supercoil shape. Biochem J. 1976 Apr 1;155(1):101–105. doi: 10.1042/bj1550101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campbell A. M., Jolly D. J. Light-scattering studies on supercoil unwinding. Biochem J. 1973 Jun;133(2):209–226. doi: 10.1042/bj1330209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chambon P. Eukaryotic nuclear RNA polymerases. Annu Rev Biochem. 1975;44:613–638. doi: 10.1146/annurev.bi.44.070175.003145. [DOI] [PubMed] [Google Scholar]
- Cook P. R., Brazell I. A. Supercoils in human DNA. J Cell Sci. 1975 Nov;19(2):261–279. doi: 10.1242/jcs.19.2.261. [DOI] [PubMed] [Google Scholar]
- Davidson N. Effect of DNA length on the free energy of binding of an unwinding ligand to a supercoiled DNA. J Mol Biol. 1972 May 14;66(2):307–309. doi: 10.1016/0022-2836(72)90482-2. [DOI] [PubMed] [Google Scholar]
- Dezelee S., Wyers F., Darlix J. L., Sentenac A., Fromageot P. DNA binding, condensing and unwinding properties of yeast RNase H1. J Biol Chem. 1977 Dec 25;252(24):8935–8944. [PubMed] [Google Scholar]
- Dezélée S., Sentenac A., Fromageot P. Role of deoxyribonucleic acid-ribonucleic acid hybrids in eukaryotes. Study of the template requirements of yeast ribonucleic acid polymerases and nature of the ribonucleic acid product. J Biol Chem. 1974 Sep 25;249(18):5971–5977. [PubMed] [Google Scholar]
- Dezélée S., Sentenac A., Fromageot P. Role of deoxyribonucleic acid-ribonucleic acid hybrids in eukaryotes. Synthetic ribo- and deoxyribopolynucleotides as template for yeast ribonucleic acid polymerase B (or II). J Biol Chem. 1974 Sep 25;249(18):5978–5983. [PubMed] [Google Scholar]
- Dezélée S., Sentenac A. Role of DNA-RNA hybrids in eukaryotes. Purification and properties of yeast RNA polymerase B. Eur J Biochem. 1973 Apr 2;34(1):41–52. doi: 10.1111/j.1432-1033.1973.tb02726.x. [DOI] [PubMed] [Google Scholar]
- Dreyer C., Hausen P. On the initiation of mammalian RNA polymerase at single-strand breaks in DNA. Eur J Biochem. 1976 Nov 1;70(1):63–74. doi: 10.1111/j.1432-1033.1976.tb10956.x. [DOI] [PubMed] [Google Scholar]
- Espejo R. T., Canelo E. S., Sinsheimer R. L. DNA of bacteriophage PM2: a closed circular double-stranded molecule. Proc Natl Acad Sci U S A. 1969 Aug;63(4):1164–1168. doi: 10.1073/pnas.63.4.1164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flashner M. S., Katopes M. A., Lebowitz J. Inhibition of transcription of supercoiled PM2 DNA by carbodiimide modification. Nucleic Acids Res. 1977 Jun;4(6):1713–1726. doi: 10.1093/nar/4.6.1713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flint S. J., De Pomerai D. I., Chesterton C. J., Butterworth P. H. Template specificity of eucaryotic DNA-dependent RNA polymerases. Effect of DNA structure and integrity. Eur J Biochem. 1974 Mar 1;42(2):567–579. doi: 10.1111/j.1432-1033.1974.tb03372.x. [DOI] [PubMed] [Google Scholar]
- Hayashi M. A DNA-RNA complex as an intermediate of in vitro genetic transcription. Proc Natl Acad Sci U S A. 1965 Dec;54(6):1736–1743. doi: 10.1073/pnas.54.6.1736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hecht R. M., Birnstiel M. L. Integrity of the DNA template, a prerequisite for the faithful transcription of Xenopus rDNA in vitro. Eur J Biochem. 1972 Sep 25;29(3):489–499. doi: 10.1111/j.1432-1033.1972.tb02013.x. [DOI] [PubMed] [Google Scholar]
- Honjo T., Reeder R. H. Transcription of Xenopus chromatin by homologous ribonucleic acid polymerase: aberrant synthesis of ribosomal and 5S ribonucleic acid. Biochemistry. 1974 Apr 23;13(9):1896–1899. doi: 10.1021/bi00706a018. [DOI] [PubMed] [Google Scholar]
- Hossenlopp P., Oudet P., Chambon P. Animal DNA-dependent RNA polymerases. Studies on the binding of mammalian RNA polymerases AI and B to Simian virus 40 DNA. Eur J Biochem. 1974 Jan 16;41(2):397–411. doi: 10.1111/j.1432-1033.1974.tb03281.x. [DOI] [PubMed] [Google Scholar]
- Houghton M., Cox R. F. The purification and properties of hen oviduct form B DNA-dependent RNA polymerase. Nucleic Acids Res. 1974 Feb;1(2):299–308. doi: 10.1093/nar/1.2.299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ide T., Nakane M., Anzai K., Ando T. Supercoiled DNA folded by non-histone proteins in cultured mammalian cells. Nature. 1975 Dec 4;258(5534):445–447. doi: 10.1038/258445a0. [DOI] [PubMed] [Google Scholar]
- Jaehning J. A., Roeder R. G. Transcription of specific adenovirus genes in isolated nuclei by exogenous RNA polymerases. J Biol Chem. 1977 Dec 10;252(23):8753–8761. [PubMed] [Google Scholar]
- Jendrisak J. J., Burgess R. R. A new method for the large-scale purification of wheat germ DNA-dependent RNA polymerase II. Biochemistry. 1975 Oct 21;14(21):4639–4645. doi: 10.1021/bi00692a012. [DOI] [PubMed] [Google Scholar]
- Katz L., Kingsbury D. T., Helinski D. R. Stimulation by cyclic adenosine monophosphate of plasmid deoxyribonucleic acid replication and catabolite repression of the plasmid deoxyribonucleic acid-protein relaxation complex. J Bacteriol. 1973 May;114(2):577–591. doi: 10.1128/jb.114.2.577-591.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lescure B., Chestier A., Yaniv M. Transcription of polyoma virus DNA in vitro. II. Transcription of superhelical and linear polyoma DNA by RNA polymerase II. J Mol Biol. 1978 Sep 5;124(1):73–85. doi: 10.1016/0022-2836(78)90148-1. [DOI] [PubMed] [Google Scholar]
- Lescure B., Dauguet C., Yaniv M. Transcription of polyoma virus DNA in vitro. III. Localization of calf thymus RNA polymerase II binding sites. J Mol Biol. 1978 Sep 5;124(1):87–96. doi: 10.1016/0022-2836(78)90149-3. [DOI] [PubMed] [Google Scholar]
- Lilley D. M. Active chromatin structure. Cell Biol Int Rep. 1978 Jan;2(1):1–10. doi: 10.1016/0309-1651(78)90078-4. [DOI] [PubMed] [Google Scholar]
- Mandel J. L., Chambon P. Animal DNA-dependent RNA polymerases. Analysis of the RNAs synthesized on Simian virus 40 superhelical DNA by mammalian RNA polymerases AI and B. Eur J Biochem. 1974 Jan 16;41(2):379–395. doi: 10.1111/j.1432-1033.1974.tb03280.x. [DOI] [PubMed] [Google Scholar]
- Mandel J. L., Chambon P. Animal DNA-dependent RNA polymerases. Studies on the reaction parameters of transcription in vitro of Simian virus 40 DNA by mammalian RNA polymerases AI and B. Eur J Biochem. 1974 Jan 16;41(2):367–378. doi: 10.1111/j.1432-1033.1974.tb03279.x. [DOI] [PubMed] [Google Scholar]
- Meilhac M., Tysper Z., Chambon P. Animal DNA-dependent RNA polymerases. 4. Studies on inhibition by rifamycin derivatives. Eur J Biochem. 1972 Jul 13;28(2):291–300. doi: 10.1111/j.1432-1033.1972.tb01913.x. [DOI] [PubMed] [Google Scholar]
- Onishi T., Muramatsu M. Inhibition by derivatives of rifamycin of soluble ribonucleic acid polymerase from rat liver. Biochem J. 1972 Aug;128(5):1361–1364. doi: 10.1042/bj1281361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richardson J. P. Attachment of nascent RNA molecules to superhelical DNA. J Mol Biol. 1975 Nov 5;98(3):565–579. doi: 10.1016/s0022-2836(75)80087-8. [DOI] [PubMed] [Google Scholar]
- Richardson J. P. Effects of supercoiling on transcription from bacteriophage PM2 deoxyribonucleic acid. Biochemistry. 1974 Jul 16;13(15):3164–3169. doi: 10.1021/bi00712a025. [DOI] [PubMed] [Google Scholar]
- Roeder R. G., Rutter W. J. Multiple forms of DNA-dependent RNA polymerase in eukaryotic organisms. Nature. 1969 Oct 18;224(5216):234–237. doi: 10.1038/224234a0. [DOI] [PubMed] [Google Scholar]
- Sharp P. A., Sugden B., Sambrook J. Detection of two restriction endonuclease activities in Haemophilus parainfluenzae using analytical agarose--ethidium bromide electrophoresis. Biochemistry. 1973 Jul 31;12(16):3055–3063. doi: 10.1021/bi00740a018. [DOI] [PubMed] [Google Scholar]
- Smith H. O., Wilcox K. W. A restriction enzyme from Hemophilus influenzae. I. Purification and general properties. J Mol Biol. 1970 Jul 28;51(2):379–391. doi: 10.1016/0022-2836(70)90149-x. [DOI] [PubMed] [Google Scholar]
- Van Keulen H., Retèl J. Transcription specificity of yeast RNA polymerase A. Highly specific transcription in vitro of the homologous ribosomal transcription units. Eur J Biochem. 1977 Oct 3;79(2):579–588. doi: 10.1111/j.1432-1033.1977.tb11842.x. [DOI] [PubMed] [Google Scholar]
- Vinograd J., Lebowitz J., Watson R. Early and late helix-coil transitions in closed circular DNA. The number of superhelical turns in polyoma DNA. J Mol Biol. 1968 Apr 14;33(1):173–197. doi: 10.1016/0022-2836(68)90287-8. [DOI] [PubMed] [Google Scholar]
- Wang J. C. Interactions between twisted DNAs and enzymes: the effects of superhelical turns. J Mol Biol. 1974 Aug 25;87(4):797–816. doi: 10.1016/0022-2836(74)90085-0. [DOI] [PubMed] [Google Scholar]
- Wensink P. C., Finnegan D. J., Donelson J. E., Hogness D. S. A system for mapping DNA sequences in the chromosomes of Drosophila melanogaster. Cell. 1974 Dec;3(4):315–325. doi: 10.1016/0092-8674(74)90045-2. [DOI] [PubMed] [Google Scholar]
- Woodworth-Gutai M., Lebowitz J. Introduction of interrupted secondary structure in supercoiled DNA as a function of superhelix density: consideration of hairpin structures in superhelical DNA. J Virol. 1976 Apr;18(1):195–204. doi: 10.1128/jvi.18.1.195-204.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Worcel A., Burgi E. On the structure of the folded chromosome of Escherichia coli. J Mol Biol. 1972 Nov 14;71(2):127–147. doi: 10.1016/0022-2836(72)90342-7. [DOI] [PubMed] [Google Scholar]