Abstract
Because naïve T cells are unable to import cystine due to the absence of cystine transporters, it has been suggested that T cell activation is dependent on cysteine generated by antigen presenting cells. The aim of this study was to determine at which phases during T cell activation exogenous cystine/cysteine is required and how T cells meet this requirement. We found that early activation of T cells is independent of exogenous cystine/cysteine, whereas T cell proliferation is strictly dependent of uptake of exogenous cystine/cysteine. Naïve T cells express no or very low levels of both cystine and cysteine transporters. However, we found that these transporters become strongly up-regulated during T cell activation and provide activated T cells with the required amount of cystine/cysteine needed for T cell proliferation. Thus, T cells are equipped with mechanisms that allow T cell activation and proliferation independently of cysteine generated by antigen presenting cells.
The tripeptide glutathione (GSH) is a ubiquitous intracellular peptide with diverse functions that include antioxidant defense, maintenance of thiol status, and modulation of cell proliferation. GSH plays an essential role in T cell function, and several studies have found that an increase in intracellular GSH during T cell activation is required for T cell proliferation1,2,3,4. GSH is synthesized in the cytosol in a tightly regulated manner with cysteine (Cys) being the rate-limiting amino acid5. Theoretically, T cells can obtain Cys either by transport of Cys or the oxidized, dimeric form of Cys called cystine (Cys2) from the extracellular space or by endogenous production of Cys from methionine (Met) via the transsulfuration pathway6. Whether the transsulfuration pathway actually exists in T cells is controversial. Some studies support the presence of the transsulfuration pathway in T cells6,7 whereas other studies have concluded that T cells lack this pathway8,9.
Due to the oxidizing environment approximately 90% of Cys is found as oxidized Cys2 in the extracellular space. Thus, plasma contains 50 – 100 µM Cys2, whereas the concentration of Cys is very low compared to other amino acids10,11. Cys is transported over the plasma membrane predominantly by the neutral amino acid transporters ASCT1 and ASCT2, whereas Cys2 exclusively is transported by the xc− cystine/glutamate antiporter12,13. Imported Cys2 is rapidly reduced to Cys in the cytosol due to the reducing intracellular milieu. By measurement of the uptake of Cys and Cys2 previous studies have indirectly indicated that T cells express ASCT1 and/or ASCT2 transporters but not xc−14,15. Based on these observations it was suggested that a sufficient high concentration of exogenous Cys in the microenvironment is provided to T cells by activated antigen presenting cells (APC), and that this APC-generated Cys is required for T cell activation16,17. Recently, it was confirmed that naïve unstimulated T cells do not express the xc− cystine/glutamate antiporter but interestingly it was also found that T cell activation induced the expression of xc−6,18. This indicated that T cells are released from their need for APC-generated Cys early after activation, and it was suggested that APC-generated Cys is essential to start T cell activation18. Thus, the model predicted that in the early phases of an immune response APC nurse T cells by taking up oxidized Cys2 and releasing Cys. The released Cys is then internalized by the naïve T cells via their ASCT transporters. Later on, the activated T cells can meet their requirement for Cys by uptake of Cys2 via the induced expression of xc− and thereby become independent of Cys generated by the APC19,20. However, the proposed dependency on APC-generated Cys for early T cell activation is in conflict with other studies demonstrating that purified T cells can be fully activated in the absence of APC4,21. The aim of this study was to determine at which phases during activation of purified T cells Cys is required and how T cells can meet this requirement. We found that early T cell activation as measured by CD25 and CD69 expression and secretion of IL-2 was independent of Cys, whereas T cell proliferation was strictly dependent on Cys. T cell activation led to a strong up-regulation of both the Cys2 transporter xc− and the Cys transporters ASCT1 and ASCT2 independently of Cys. The T cells met their requirement of Cys completely independently of APC by importing either Cys2 or Cys via the activation-induced xc−, ASCT1 and ASCT2 transporters.
Results
Early T cell activation is independent of Cys and Met whereas T cell proliferation is strictly dependent on Cys
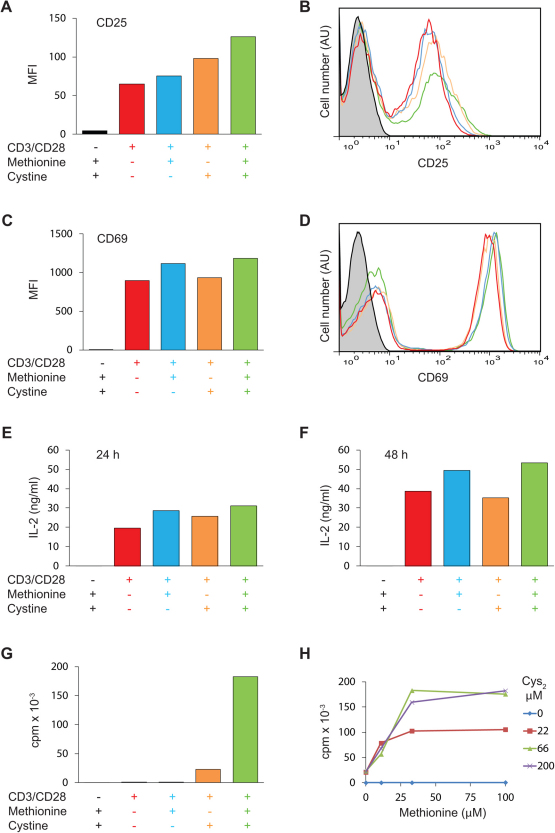
To study a homogeneous population of T cells we purified antigen-inexperienced naïve CD4+ T cells from blood samples obtained from healthy blood donors. The resulting cell population consisted of ∼ 98 % CD4+, CD45RA+, CD25−, CD69− T cells (Supplementary Fig. 1S). To study the requirements of T cell activation for Cys and Met we stimulated the purified T cells with anti-CD3 and anti-CD28 coated Dynabeads in Cys- and Met-free medium that was either not supplemented or supplemented with Met, Cys2, or a combination of the two. We found that early T cell activation as measured by CD25 and CD69 expression was independent of Cys2 and Met, although the CD25 expression was somewhat higher in cells stimulated in medium supplemented with Cys2 (Fig. 1 A–D). Likewise, IL-2 production was independent of Cys2 and Met (Fig. 1 E & F). In contrast, T cell proliferation as measured by 3H-thymidine incorporation was strictly dependent on Cys2 and was optimal in the presence of both Cys2 and Met (Fig. 1G). To determine the minimal concentrations of Cys2 and Met required for optimal T cell proliferation we next performed a checkerboard titration of Cys2 and Met. We found that the minimal concentrations of Cys2 and Met required for optimal T cell proliferation were 66 and 33 µM, respectively (Fig. 1H). Thus, the normal plasma Cys2 concentration of 50 – 100 µM seems to fully satisfy the Cys2 requirement for T cell proliferation. In the absence of Cys2, T cell proliferation did not take place irrespective of the Met concentrations tested. In contrast, in the absence of Met, supplementation of the medium with just 22 µM of Cys2 resulted in significant T cell proliferation (Fig. 1H).
Figure 1. Early T cell activation is independent of Cys and Met whereas T cell proliferation is strictly dependent on Cys.
Purified naïve CD4+ T cells were incubated in Met- and Cys-free DMEM medium without stimulation (black columns and lines in A–G) or were stimulated with Dynabeads Human T-Activator CD3/CD28 in Met- and Cys-free DMEM medium (red columns and lines in A–G) or in Met- and Cys-free DMEM medium supplemented with either 33 µM Met (blue columns and lines in A–G), 66 µM Cys2 (orange columns and lines in A–G), or 33 µM Met plus 66 µM Cys2 (green columns and lines in A–G). The expression levels of CD25 and CD69 were determined by flow cytometry after 24 hours of cell culture and depicted as mean fluorescence intensity (MFI) (A and C) and as FACS profiles (B and D). The production of IL-2 was measured by ELISA following 24 (E) and 48 (F) hours of cell culture. T cell proliferation was measured as3H-thymidine incorporation for the last six hours of a total of 72 hours of cell culture (G and H). The results shown are representative for at least three separate experiments.
Activated T cells strongly up-regulate transporters for Cys2 and Cys
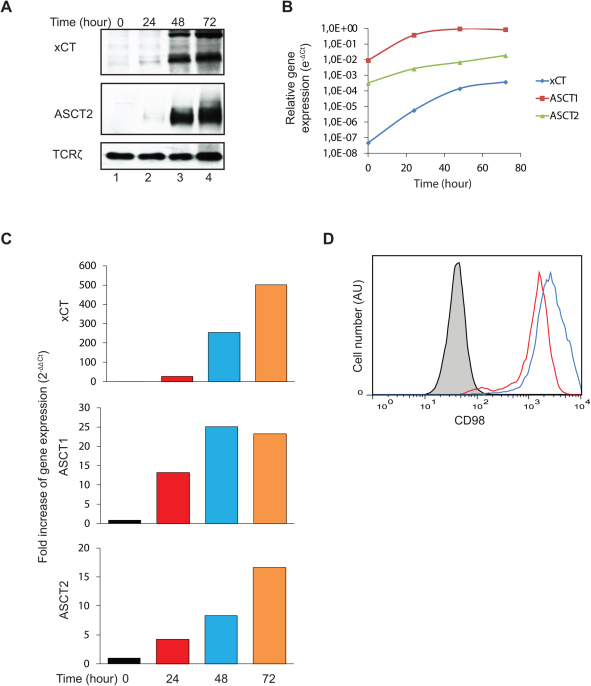
The experiments described above demonstrated that early T cell activation is independent of Cys and Met, whereas T cell proliferation is strictly dependent on Cys. This indicated that T cells have or obtain the ability to transport Cys2 and/or Cys over the plasma membrane during their activation. Furthermore, the experiments demonstrated that if T cells possess the transsulfuration pathway this is not efficient enough to cover the requirement for Cys during T cell proliferation. The xc− cystine/glutamate antiporter that transport Cys2 over the plasma membrane is a disulfide-bonded heterodimer composed of the heavy subunit 4F2hc (CD98) and the light subunit xCT (gene name SLC7A11)13. CD98 is involved in trafficking of the heterodimer to the plasma membrane, whereas xCT confers specificity for Cys2. CD98 acts as a subunit for various amino acid transporters and is therefore commonly expressed. Whereas xc− is the only known transporter for Cys2, several transporters for Cys exist, the most important being the neutral amino acid transporters ASCT1 (gene name SLC1A4) and ASCT2 (gene name SLC1A5)22,23. To determine the expression level of xc−, ASCT1 and ASCT2 on CD4+ T cells we stimulated the cells for zero to 72 hours in X-VIVO 15 medium and measured the level of protein and gene expression for these transporters. We found that un-stimulated naïve CD4+ T cells express no or very low levels of xCT and ASCT2 as determined by Western blot analyses (Fig. 2A). We could not determine the protein level of ASCT1 as no proper anti-ASCT1 antibodies were available for the use in Western blot analyses. T cell stimulation induced strong up-regulation of both xCT and ASCT2 (Fig. 2A). In agreement, gene expression analyses demonstrated that the transporter genes were weakly expressed in naïve T cells and that they became strongly up-regulated following T cell activation (Fig. 2B & C). In particular the gene for xCT was up-regulated approximately 500 fold relative to the housekeeping gene GAPDH (Fig. 2C). In concordance with this, we found a significant up-regulation of CD98 as measured by FACS analysis (Fig. 2D).
Figure 2. Activated T cells strongly up-regulate the xc− cystine/glutamate antiporter and the neutral amino acid transporters ASCT1 and ASCT2.
Purified naïve CD4+ T cells were incubated in X-VIVO 15 medium and stimulated with Dynabeads Human T-Activator CD3/CD28 for the time indicated. The cells were harvested and the expression of xCT (55 and 35 kDa31) and ASCT2 (75 kDa) was determined by Western blot analyses using TCRζ (16 kDa) as loading control (A) or by quantitative RT-PCR (B and C). The gene expression levels of the target genes are given relative to the housekeeping gene GAPDH (B) or as fold increase of the target gene expression relative to GAPDH (C). CD98 expression was measured by FACS analyses following zero (black line), 24 (red line) and 48 (blue line) hours of cell culture (D). The results shown are representative for at least two separate experiments.
Both the xc- cystine/glutamate antiporter and the neutral amino acid transporters can supply T cells with the required amounts of Cys
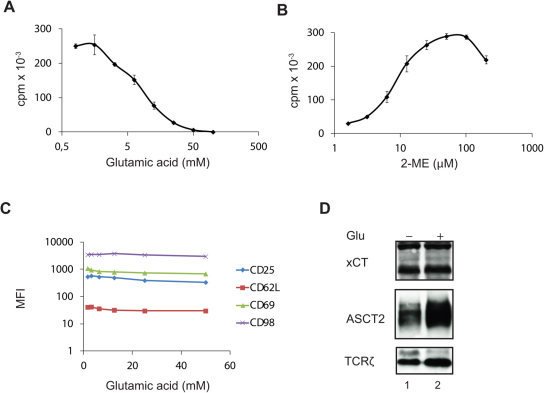
The above studies demonstrated that CD4+ T cells start to express high levels of both Cys2 and Cys transporters following activation. Due to the oxidizing environment the vast majority of Cys is found as oxidized Cys2 in the extracellular space and in most cell growth media; however, approximately 10% is found as Cys in these sites10. To determine whether T cells predominantly meet their Cys requirement by uptake of Cys2 or by uptake of Cys we inhibited the uptake of Cys2 by incubation of the cells in X-VIVO 15 medium supplemented with increasing quantities of glutamic acid (Glu). Glu is the main exchange substrate in the xc− cystine/glutamate antiporter-mediated uptake of Cys2 and is a highly specific inhibitor of Cys2 uptake13. We found that incubation of the cells with 20 mM of Glu inhibited T cell proliferation by more than 90% (Fig. 3A). This indicated that T cell proliferation is dependent on the uptake of Cys2 via the xc− cystine/glutamate antiporter. To exclude the possibility that the observed inhibition of T cell proliferation was caused by toxic side effects of Glu, and to test whether the neutral amino acid transporters could substitute for the xc− transporter we incubated the cells in X-VIVO 15 medium with a fixed concentration of Glu (20 mM) in combination with increasing quantities of 2-mercaptoethanol (2-ME), which reduces the Cys2 in the medium to Cys. Thus, if the cells were unaffected by the high Glu concentration and if the neutral amino acid transporters were functional, T cell proliferation should be reconstituted with increasing concentrations of 2-ME. Indeed, we found that 2-ME reconstituted T cell proliferation and that T cell proliferation could be completely rescued by adding 2-ME at a concentration of 50 – 100 µM (Fig. 3B). Likewise, supplementation of X-VIVO 15 medium containing 20 mM Glu with free thiols in the form of N-acetyl-L-cysteine or with GSH completely rescued T cell proliferation (Supplementary Fig. 2S). Thus, during normal conditions T cell proliferation is strictly dependent on xc− -mediated uptake of Cys2. However, if Cys is present in sufficiently high concentrations in the extracellular environment ASCT1 and ASCT2 can provide adequate Cys uptake required for T cell proliferation.
Figure 3. Both the xc− cystine/glutamate antiporter and the neutral amino acid transporters can supply T cells with the required amounts of Cys.
(A and C) Purified naïve CD4+ T cells were incubated in X-VIVO 15 medium supplemented with the indicated concentrations of Glu and stimulated with Dynabeads Human T-Activator CD3/CD28 for 72 h (A) or 48 h (C). T cell proliferation was measured by incorporation of3H-thymidine for the last 6 h of cell culture (A). The expression levels of activation markers were measured by FACS analyses (C). (B) Purified naïve CD4+ T cells were incubated in X-VIVO 15 medium supplemented with 20 mM of Glu and the indicated concentrations of 2-ME and stimulated with Dynabeads Human T-Activator CD3/CD28 for 72 h. T cell proliferation was measured by incorporation of3H-thymidine for the last 6 h of incubation. (D) Purified naïve CD4+ T cells were incubated in X-VIVO 15 medium that was either not supplemented (lane 1) or supplemented with 20 mM of Glu (lane 2) and stimulated with Dynabeads Human T-Activator CD3/CD28 for 72 h. The expression of xCT and ASCT2 was determined by Western blot analyses using TCRζ as loading control. The results shown in A and B represent mean values ± SEM of triplicates determinations and are representative for three separate experiments. The results shown in C and D are representative for two separate experiments.
To further study the need for Cys during T cell activation we stimulated the cells in X-VIVO 15 medium supplemented with increasing quantities of Glu and measured the expression of the activation markers CD25, CD62L, CD69 and CD98 by FACS. We found that even Glu concentrations that completely abolished T cell proliferation did not affect the expression of the activation markers (Fig. 3C). Neither did Glu inhibit expression of the activation-induced xCT, ASCT1 and ASCT2 transporters (Fig. 3D). These experiments confirmed that early T cell activation, including the up-regulation of the Cys2 and Cys transporters, is independent of Cys, whereas T cell proliferation is strictly dependent on Cys.
Discussion
In this study we demonstrate that human naïve CD4+ T cells express no or very low levels of the Cys2 transporter xc− and the Cys transporters ASCT1 and ASCT2, and that the expression of these transporters is strongly up-regulated during T cell activation. This is the first study that directly measures the expression of ASCT1 and ASCT2 on human T cells. Concerning expression of ASCT1 and ASCT2, our results are in good agreement with previous studies, which found that the potential of resting T cells to take up Cys was very low and that it increased significantly following T cell activation14,15,24. Concerning expression of xc− on T cells, conflicting results have been reported. Based on measurements of Cys2 uptake and the potential of Glu to inhibit Cys2 uptake, some studies indirectly found evidence for the lack of expression of xc− on both resting and activated T cells14,15. However, the Glu concentrations used in these studies to inhibit xc− were quite low (0.5 – 2.5 mM), and in one of the studies uptake of Cys2 was actually observed in resting T cells and it increased by a factor three in activated T cells14. Another study found indirect evidence for xc− expression on activated T cells by observing Glu-mediated inhibition of T cell proliferation25. In agreement with our findings, recent studies have found that xc− is not expressed on resting T cells but becomes expressed following T cell activation6,18. Based on these and our study we believe that it can be concluded that naïve T cell express no or very low levels of xc−, ASCT1 and ASCT2, and that the expression of these transporters is strongly induced during T cell activation. In agreement with this, we found that early T cell activation is independent of Cys and thereby of Cys2 and Cys uptake via xc−, ASCT1 and ASCT2 transporters that the T cells do not express at this early stage of activation. In contrast to early T cell activation, we found that T cell proliferation is strictly dependent on Cys. This is in good agreement with previous studies, which found that T cell proliferation, specifically the progression from G1 of the cell cycle to the S phase, is dependent on an increased GSH synthesis in which Cys is the rate-limiting amino acid2,3,4,5. The increased production and translocation of GSH into the cell nucleus probably provides the reducing power required for the generation of dNTPs for DNA synthesis26,27.
At first sight it might seem puzzling that activated T cells can synthesize proteins like the activation markers, the Cys and Cys2 transporters and IL-2 reported in this study in the absence of exogenous Met, Cys and Cys2. However, the amino acids required for protein synthesis is probably provided from a combination of (i) the intracellular amino acid pool, (ii) protein catabolism, and (iii) degradation of GSH. It has been reported that resting T cells contain a pool of GSH that is rapidly degraded2,14 and rebuild probably via the γ-glutamyl cycle28,29. Degraded GSH might be an important source of Cys. Fundamentally different pathways are used by cells for protein versus GSH synthesis, and it is possible that during Cys scarcity GSH rebuilding is minimized in favor of protein synthesis.
In theory, T cells can also obtain Cys through the conversion of Met to Cys via the transsulfuration pathway6. However, we found that Cys could not be substituted by Met in T cell proliferation, and we conclude that if the transsulfuration pathway actually exists in T cells as previously suggested6,7, it cannot supply the T cells with the amount of Cys needed for T cell proliferation to take place.
It has been postulated that naïve T cells are metabolically dependent on APC to meet their Cys requirements because Cys is the least abundant of the amino acids in the extracellular space and naïve T cells are unable to import Cys2 due to the absence of xc−6,16,17,18,19,20. However, we found that early activation of naïve T cells including expression of activation markers, Cys and Cys2 transporters and production of IL-2 is not dependent on Cys. Furthermore, we show that in addition to xc−, activated T cells express ASCT1 and ASCT2, and we present evidence that both the Cys2 and Cys transporters are functional transporters, which can provide T cells with the required amount of Cys needed for T cell proliferation. Thus, human naïve CD4+ T cells are fully equipped with mechanisms that allow T cell activation and proliferation independently of cysteine generated by antigen presenting cells.
Methods
Cells and reagents
Blood mononuclear cells were isolated by density gradient centrifugation using Lymphoprep (Nycomed, Roskilde, Denmark). Naïve CD4+ T cells were subsequently isolated by negative selection using the naïve CD4+ T cell isolation kit II (cat. no. 130-094-131, Miltenyi Biotec GmbH, Bergisch Gladbach, Germany) according to the manufacturer. The purified T cells were cultured in either Dulbecco's Modified Eagle Medium without L-methionine and L-cystine (cat. no. 21013, Invitrogen, Paisly, UK) or in X-VIVO 15 medium (cat. no. 1041, Lonza, Verviers, Belgium) that contains Cys2 and Met at 290 and 200 µM, respectively. The cells were stimulated with Dynabeads Human T-Activator CD3/CD28 (cat. no. 111.31D, Invitrogen) at 37°C in 5% CO2 at a cell concentration of 0.25 – 1.0×106 cells/ml for the times indicated at a bead to cell ratio of two to five. L-methionine (M5308), L-cystine dihydrochloride (C6727), L-glutamic acid (G5889), N-acetyl-L-cysteine (A7250) and L-glutathione (G4251) were from Sigma-Aldrich (Brondby, Denmark).
Flow cytometry and Western blot analysis
To determine the expression levels of various surface markers we incubated the cells with fluorochrome-conjugated anti-CD4 (clone SK3), anti-CD25 (clone 2A3), anti-CD45RA (clone HI100), anti-CD62L (clone Dreg 56), anti-CD69 (clone L78) or anti-CD98 (clone UM7F8) monoclonal antibodies all from BD Biosciences (San Diego, CA). The cells were subsequently run on a FACSCalibur (BD Biosciences). Data analysis was performed using FlowJo software (Tree Star, Ashland, OR, USA). For Western blot analyses cells were stimulated with Dynabeads Human T-Activator CD3/CD28 for the time indicated at 37°C, lysed in lysis buffer (50 mM Tris-Base pH 7.5, 150 mM NaCl, 1 mM Mg2Cl) supplemented with 1% Triton X-100, 1 mg/ml Pefabloc SC, 10 μg/ml Leupeptin, 10 μg/ml Pepstatin and 5 mM EDTA and run on 10% polyacrylamide gels. The proteins were transferred to Amersham Hybond ECL nitrocellulose sheets (GE Healthcare, Brondby, Denmark) and visualized by anti-xCT (cat. no NB300-318, Novus Biologicals, Littleton, CO, USA), anti-ASCT2 (clone G11, Cell Signaling, Technology, Danvers, MA, USA) or anti-TCRζ (clone 6B10.2, Santa Cruz Biotechnology, Santa Cruz, CA) antibodies followed by either HRP-swine anti-rabbit Ig or HRP-rabbit anti-mouse Ig (cat. no. P0399 and P0260, Dako, Glostrup, Denmark) using Amersham ECL technology (GE Healthcare).
Quantitative RT-PCR analysis
To quantify gene expression we used reversed transcription combined with the polymerase chain reaction (RT-PCR) method. Total RNA was isolated from purified naïve CD4+ T cells or stimulated CD4+ T cells. One μg of the total RNA from each sample was reverse-transcribed into cDNA using RevertAid™ First Strand cDNA Synthesis Kit (cat. no. K1621, Fermentas GmbH, St. Leon-Rot, Germany). The amount of xCT (SLC7A11), ASCT1 (SLC1A4), ASCT2 (SLC1A5) and GAPDH mRNA was determined by using primers from TAG Copenhagen and Stratagene Brilliant SYBR ® Green QPCR Master Mix (Agilent Technologies, Horsholm, Denmark). A PCR amplification profile was derived by recording the SYBR Green fluorescence intensity, which was in linear relation to the amount of formed PCR product as a function of PCR cycle number. The SLC7A11, SLC1A4 and SLC1A5 mRNAs were calculated relative to the GAPDH housekeeping gene and normalized to control conditions by using the 2−ΔΔCt method30. We used the following primers: xCT- forward 5′-GCT GTG ATA TCC CTG GCA TT-3′, xCT- reverse 5′-CAG AAT TGC TGT GAG CTT GC-3′, ASCT1 5′-ATT GGT CCT GTT TGC TCT GG-3′, ASCT1 reverse 5′-TGG GGA GAA TAA ACC TGC TG-3′, ASCT2 forward 5′-TAC ATT CTG TGC TGC CTG CT-3′, ASCT2 reverse 5′-ATG AAA CGG CTG ATG TGC TT -3′, GAPDH forward 5′-AAG GTG AAG GTC GGA GTC AA-3′ and GAPDH reverse 5′-AAT GAA GGG GTC ATT GAT GG-3′.
Author Contributions
Conceived and designed the experiments: CG, AKH, TBL, MRvE, CMB, AW, NØ. Performed the experiments: TBL, BLN, AKH, MK. Analyzed the data: CG, TBL, AKH, CMB, AW, NØ. Wrote the paper: CG, TBL.
Supplementary Material
Supp info
Acknowledgments
This work was supported by grants from The Danish Medical Research Coun-cil, The Novo Nordisk Foundation, The Lundbeck Foundation, The A.P. Møller Foundation for the Advancement of Medical Sciences, and Lægeforeningens Forskningsfond.
References
- Hamilos D. L. & Wedner H. J. The role of glutathione in lymphocyte activation. I. Comparison of inhibitory effects of buthionine sulfoximine and 2-cyclohexene-1-one by nuclear size transformation. J. Immunol. 135, 2740–2747 (1985). [PubMed] [Google Scholar]
- Hamilos D. L., Zelarney P. & Mascali J. J. Lymphocyte proliferation in glutathione-depleted lymphocytes: direct relationship between glutathione availability and the proliferative response. Immunopharmacology 18, 223–235 (1989). [DOI] [PubMed] [Google Scholar]
- Messina J. P. & Lawrence D. A. Cell cycle progression of glutathione-depleted human peripheral blood mononuclear cells is inhibited at S phase. J. Immunol. 143, 1974–1981 (1989). [PubMed] [Google Scholar]
- Suthanthiran M., Anderson M. E., Sharma V. K. & Meister A. Glutathione regulates activation-dependent DNA synthesis in highly purified normal human T lymphocytes stimulated via the CD2 and CD3 antigens. Proc. Natl. Acad. Sci. U. S. A 87, 3343–3347 (1990). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu S. C. Regulation of glutathione synthesis. Mol. Aspects Med. 30, 42–59 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garg S. K., Yan Z., Vitvitsky V. & Banerjee R. Differential dependence on cysteine from transsulfuration versus transport during T cell activation. Antioxid. Redox. Signal. 15, 39–47 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sturman J. A., Beratis N. G., Guarini L. & Gaull G. E. Transsulfuration by human long term lymphoid lines. Normal and cystathionase-deficient cells. J. Biol. Chem. 255, 4763–4765 (1980). [PubMed] [Google Scholar]
- Eagle H., Washington C. & Friedman S. M. The synthesis of homocystine, cystathionine, and cystine by cultured diploid and heteroploid human cells. Proc. Natl. Acad. Sci. U. S. A 56, 156–163 (1966). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Srivastava M. K., Sinha P., Clements V. K., Rodriguez P. & Ostrand-Rosenberg S. Myeloid-derived suppressor cells inhibit T-cell activation by depleting cystine and cysteine. Cancer Res. 70, 68–77 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saetre R. & Rabenstein D. L. Determination of cysteine in plasma and urine and homocysteine in plasma by high-pressure liquid chromatography. Anal. Biochem. 90, 684–692 (1978). [DOI] [PubMed] [Google Scholar]
- Hack V. et al. Cystine levels, cystine flux, and protein catabolism in cancer cachexia, HIV/SIV infection, and senescence. FASEB J. 11, 84–92 (1997). [DOI] [PubMed] [Google Scholar]
- Kanai Y. & Hediger M. A. The glutamate/neutral amino acid transporter family SLC1: molecular, physiological and pharmacological aspects. Pflugers Arch. 447, 469–479 (2004). [DOI] [PubMed] [Google Scholar]
- Lo M., Wang Y. Z. & Gout P. W. The x(c)- cystine/glutamate antiporter: a potential target for therapy of cancer and other diseases. J. Cell Physiol 215, 593–602 (2008). [DOI] [PubMed] [Google Scholar]
- Ishii T., Sugita Y. & Bannai S. Regulation of glutathione levels in mouse spleen lymphocytes by transport of cysteine. J Cell Physiol 133, 330–336 (1987). [DOI] [PubMed] [Google Scholar]
- Gmunder H., Eck H. P. & Droge W. Low membrane transport activity for cystine in resting and mitogenically stimulated human lymphocyte preparations and human T cell clones. Eur. J. Biochem. 201, 113–117 (1991). [DOI] [PubMed] [Google Scholar]
- Angelini G. et al. Antigen-presenting dendritic cells provide the reducing extracellular microenvironment required for T lymphocyte activation. Proc. Natl. Acad. Sci. U. S. A 99, 1491–1496 (2002). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yan Z., Garg S. K., Kipnis J. & Banerjee R. Extracellular redox modulation by regulatory T cells. Nat. Chem. Biol. 5, 721–723 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Castellani P., Angelini G., Delfino L., Matucci A. & Rubartelli A. The thiol redox state of lymphoid organs is modified by immunization: role of different immune cell populations. Eur. J Immunol 38, 2419–2425 (2008). [DOI] [PubMed] [Google Scholar]
- Rubartelli A. & Sitia R. Chemo-metabolic regulation of immune responses by Tregs. Nat. Chem. Biol. 5, 709–710 (2009). [DOI] [PubMed] [Google Scholar]
- Yan Z. & Banerjee R. Redox remodeling as an immunoregulatory strategy. Biochemistry 49, 1059–1066 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- von Essen M. R. et al. Vitamin D controls T cell antigen receptor signaling and activation of human T cells. Nat. Immunol. 11, 344–349 (2010). [DOI] [PubMed] [Google Scholar]
- Broer S. Amino acid transport across mammalian intestinal and renal epithelia. Physiol Rev. 88, 249–286 (2008). [DOI] [PubMed] [Google Scholar]
- Kanai Y. & Hediger M. A. The glutamate and neutral amino acid transporter family: physiological and pharmacological implications. Eur. J. Pharmacol. 479, 237–247 (2003). [DOI] [PubMed] [Google Scholar]
- Borghetti A. F., Tramacere M., Ghiringhelli P., Severini A. & Kay J. E. Amino acid transport in pig lymphocytes. Enhanced activity of transport system asc following mitogenic stimulation. Biochim. Biophys. Acta 646, 218–230 (1981). [DOI] [PubMed] [Google Scholar]
- Lombardi G., Dianzani C., Miglio G., Canonico P. L. & Fantozzi R. Characterization of ionotropic glutamate receptors in human lymphocytes. Br. J. Pharmacol. 133, 936–944 (2001). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Markovic J. et al. Glutathione is recruited into the nucleus in early phases of cell proliferation. J. Biol. Chem. 282, 20416–20424 (2007). [DOI] [PubMed] [Google Scholar]
- Zahedi A. F. & Holmgren A. Molecular mechanisms of thioredoxin and glutaredoxin as hydrogen donors for Mammalian s phase ribonucleotide reductase. J. Biol. Chem. 284, 8233–8240 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Orlowski M. & Meister A. The gamma-glutamyl cycle: a possible transport system for amino acids. Proc. Natl. Acad. Sci. U. S. A 67, 1248–1255 (1970). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu G., Fang Y. Z., Yang S., Lupton J. R. & Turner N. D. Glutathione metabolism and its implications for health. J. Nutr. 134, 489–492 (2004). [DOI] [PubMed] [Google Scholar]
- Livak K. J. & Schmittgen T. D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCt method. Methods 25, 402–408 (2001). [DOI] [PubMed] [Google Scholar]
- Shih A. Y. et al. Cystine/glutamate exchange modulates glutathione supply for neuroprotection from oxidative stress and cell proliferation. J. Neurosci. 26, 10514–10523 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supp info