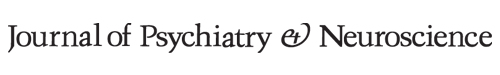Abstract
Background
Long-term administration of the dopamine (DA) D2-like (D3/2) receptor agonist pramipexole (PPX) has been previously found to desensitize D2 autoreceptors, thereby allowing a normalization of the firing of DA neurons and serotonin (5-HT)1A autoreceptors, permitting an enhancement of the spontaneous firing of 5-HT neurons. We hypothesized that PPX would increase overall DA and 5-HT neurotransmission in the forebrain as a result of these changes at the presynaptic level.
Methods
Osmotic minipumps were implanted subcutaneously in male Sprague-Dawley rats, delivering PPX at a dose of 1 mg/kg/d for 14 days. The in vivo electrophysiologic microiontophoretic experiments were carried out in anesthetized rats.
Results
The sensitivity of postsynaptic D2 receptors in the prefrontal cortex (PFC) remained unaltered following PPX administration, as indicated by the unchanged responsiveness to the microiontophoretic application of DA. Their tonic activation was, however, significantly increased by 104% compared with the control level. The sensitivity of postsynaptic 5-HT1A receptors was not altered, as indicated by the unchanged responsiveness to the microiontophoretic application of 5-HT. Similar to other antidepressant treatments, long-term PPX administration enhanced the tonic activation of 5-HT1A receptors on CA3 pyramidal neurons by 142% compared with the control level.
Limitations
The assessment of DA and 5-HT neuronal tone was restricted to the PFC and the hippocampus, respectively.
Conclusion
Chronic PPX administration led to a net enhancement in DA and 5-HT neurotransmission, as indicated by the increased tonic activation of postsynaptic D2 and 5-HT1A receptors in forebrain structures.
Introduction
Pramipexole (PPX) is a selective D2-like (D3/2) receptor agonist approved for the treatment of Parkinson disease and restless legs syndrome.1–3 Aside from its use for these neurologic conditions, PPX has also been shown to be efficacious in the treatment of major depressive disorder (MDD), both as a monotherapy4,5 and as an augmenting agent in patients with treatment-resistant depression.6–8 The efficacy of PPX against depressive symptoms was first noted in patients with Parkinson disease.1,9 This illness, characterized by a critical loss of the dopamine (DA) neurons, has a high incidence of comorbidity with MDD: up to 50%.10 These observations fall in line with research suggesting an important role of the DA system in both the pathophysiology and treatment of depression.11 Furthermore, not only PPX, but also other D2 receptor agonists with unrelated chemical structures, such as pergolide, piribedil and bromocriptine, have also been shown to possess antidepressant-like properties in animal studies and a therapeutic action in depressed patients.12–15 Imaging studies provided evidence that in depressed patients who achieve remission using PPX, the metabolic activity in brain areas affected by MDD was normalized.16 Moreover, prolonged PPX treatment not only brought brain metabolism to the control level, it was also found to restore cortical plasticity in patients with restless legs syndrome.17
Interestingly, chronic but not short-term stimulation of D2 receptors was found to promote neuronal proliferation in the rat hippocampus.18,19 This finding is of crucial importance, as enhanced neurogenesis appears to be one of the common changes occurring with drugs endowed with antidepressant properties. Despite their proven efficacy, the mechanisms responsible for the therapeutic actions of DA D2 agonists have not been fully elucidated.
The hippocampus and prefrontal cortex (PFC), structures manifesting volume decreases in depressed individuals, are also affected in rodents with chronic stress.20–24 It is not surprising that one of the common pathways for antidepressant response is an increase in the gene expression of neurotrophic/neuroprotective factors in the PFC and hippocampus.25,26 Previous work documented that prolonged administration of PPX in rats induced a desensitization of somatodendritic D2 autoreceptors in the ventral tegmental area (VTA), allowing the firing of DA neurons to normalize, and of 5-HT1A receptors in the dorsal raphe (DR) that enabled the spontaneous firing rate of 5-HT neurons to increase above control levels.27 Considering the effectiveness of PPX in the treatment of MDD, the importance of both DA and 5-HT systems in the pathophysiology of depression, and the DA innervation of the PFC and the 5-HT innervation of the hippocampus, the assessment of the net effect of chronic PPX administration on DA and 5-HT neuronal tone in the PFC and hippocampus, respectively, was deemed relevant to understand its antidepressant action.
Methods
Animals
Male Sprague-Dawley rats weighing 270–320 g at the time of recording were used for the experiments. They were kept under standard laboratory conditions (12:12 hour light/dark cycle with free access to food and water). All animal handling and procedures were carried out according to the guidelines of the Canadian Council on Animal Care, and protocols of this study were approved by the local Animal Care Committee (University of Ottawa Institute of Mental Health Research, Ottawa, Ont.).
Treatments
Rats were anesthetized with isoflurane for the subcutaneous implantation of osmotic minipumps (Alza), delivering PPX at a daily dose of 1 mg/kg for 14 days. Control rats were implanted with minipumps delivering physiologic saline. The electrophysiologic experiments were carried out with the minipumps in place.
In vivo electrophysiologic recordings
Rats were anesthetized with chloral hydrate (400 mg/kg, intraperioneal) and placed in a stereotaxic frame (David Kopf Instruments). To maintain a full anesthetic state, chloral hydrate supplements of 100 mg/kg were given intraperitoneally as needed to prevent any nociceptive reaction to paw pinching. Throughout the experiments, body temperature was maintained at 37°C using a thermistor-controlled heating pad. Extracellular recordings of pyramidal neurons in the hippocampal CA3 region and in the PFC were obtained using 5-barreled glass micropipettes. Their tips were of 3–5 μm in diameter, and impedance ranged between 4 and 7 M. Using this approach, during all recordings, signal-to-noise ratio was between 2 and 10; therefore, spike amplitude discrimination was reliable. Prior to electrophysiologic experiments, a catheter was inserted in the lateral tail vein for systemic intravenous injection of appropriate pharmacologic agents as necessary.
Extracellular recordings and microiontophoresis of pyramidal neurons in the PFC
The central barrel of the recording electrode was filled with 2 M of NaCl solution, and the 4 side barrels were filled with the following solutions: DA hydrochloride (5 mM in 200 mM of NaCl, pH 4) and 2 M of NaCl solution for automatic current balancing. The micropipettes were descended into the PFC using the following coordinates: 2.5 mm anterior and 1 mm lateral to the bregma.28 Pyramidal neurons were found at a depth of 2–4 mm below the surface of the brain and were characterized by firing at a range of 0.5–20 spikes/s, biphasic waveform with initial negative faze deflection and long-duration (0.8–1.2 ms) simple action potentials, alternating with complex spike discharges.29 The duration of microiontophoretic application of DA was 50 seconds. The 50-second duration of microiontophoretic application of the pharmacologic agents and the ejection currents (nA) were kept constant before and after each intravenous injection throughout the experiments. We assessed neuronal responsiveness to the microiontophoretic application of DA before and after intravenous injections by determining the number of spikes suppressed per nA, which was calculated by dividing the difference between the average number of spikes 50 seconds before and the average number of spikes during 50 seconds of ejection by the current of ejected DA in nA.
Extracellular recordings and microiontophoresis of pyramidal neurons in the CA3 dorsal hippocampus
Extracellular recording and microiontophoresis of CA3 pyramidal neurons were carried out with 5-barreled glass micropipettes. The central barrel used for the unitary recording was filled with 2 M of NaCl solution, and the 4 side barrels were filled with the following solutions: 5-HT creatinine sulfate (10 mM in 200 mM of NaCl, pH 4), quisqualic acid (1.5 mM in 200 mM of NaCl, pH 8) and 2 M of NaCl solution for automatic current balancing. The micropipettes were descended into the dorsal CA3 region of the hippocampus using the following coordinates: 4 mm anterior and 4.2 mm lateral to the lambda.28 Pyramidal neurons were found at a mean depth of 4.0 ± 0.5 mm below the surface of the brain. Since the pyramidal neurons do not discharge spontaneously in rats anesthetized with chloral hydrate, a small current of quisqualate (+1 to −6 nA) was used to activate them to fire at their physiologic rate (10–15 Hz).30 Pyramidal neurons were identified by their large amplitude (0.5–1.2 mV) and long-duration (0.8–1.2 ms) simple action potentials, alternating with complex spike discharges.31 The duration of microiontophoretic application of 5-HT was 50 seconds. The 50-second duration of microiontophoretic application of the pharmacologic agents and the ejection currents (nA) were kept constant before and after each intravenous injection throughout the experiments. We assessed neuronal responsiveness to the microiontophoretic application of 5-HT before and after intravenous injections by determining the number of spikes suppressed per nA, which was calculated by dividing the difference between the average number of spikes 50 seconds before and the average number of spikes during 50 seconds of ejection by the current of ejected 5-HT in nA.
Assessment of the tonic activation of postsynaptic D2 receptors
The degree of tonic activation of postsynaptic D2 receptors was assessed after 14 days of PPX administration. After a stable firing baseline was obtained, the D2-like receptor antagonist haloperidol was administered systemically at a dose of 200 μg/kg. The change in the discharge rate of pyramidal neurons was expressed as a percentage of baseline firing. This value was compared with that of the control group. Control rats were subjected to the same testing paradigm. To avoid drug residual effects, we tested only 1 neuron in each rat.
Assessment of the tonic activation of postsynaptic 5-HT1A receptors
The degree of tonic activation of postsynaptic 5-HT1A receptors was assessed after 14 days of PPX administration. The assessment of the tonic activation of postsynaptic 5-HT1A receptors is more accurate when the firing rate of the recorded neuron is low. Therefore, the firing rate of pyramidal neurons was reduced by lowering the ejection current of quisqualate. After a stable firing baseline was obtained, the selective 5-HT1A receptor antagonist WAY 100 635 was administered systemically in 4 incremental doses of 25 μg/kg each at 2-minute intervals. Neuronal response at each dose point was obtained for construction of the dose–response curve. Such curves represent stable changes in the firing rate of pyramidal neurons as percentages of baseline firing following each systemic drug administration. To avoid residual drug effects, we tested only 1 neuron in each rat.
Stimulation of the ascending DA pathway
The VTA was electrically stimulated using a bipolar electrode (NE-100; David Kopf Instruments). The electrode was implanted at 5.2 ± 0.6 mm posterior and 1.0 ± 0.5 mm lateral to the bregma, 7.4 ± 1.0 mm from the surface of the brain. The VTA was stimulated in a burst mode (train rate = 0.5 Hz, train duration = 30 ms, 6 pulses per train) via stimulator (S48; Grass Instruments) at an intensity of 500 μA. These stimulation parameters led to durations of suppression of firing of PFC neurons similar to those obtained in previous studies.32,33 The inhibition of the spontaneous activity of the PFC pyramidal neurons takes place owing to activation of postsynaptic inhibitory D2 receptors by DA, endogenously released as a result of the stimulation of DA afferents.34 We did not use different frequencies of stimulation because DA terminals in this brain region are devoid of D2 autoreceptors.35 The firing activity in relation to stimulation trains were analyzed by computer using Spike 2 (Cambridge Electronic Design Limited). Peristimulus time histograms of PFC pyramidal neurons were generated to determine the suppression of firing measured in absolute silence value (SIL) in milliseconds.
Stimulation of the ascending 5-HT pathway
The ascending 5-HT pathway was electrically stimulated using a bipolar electrode. The electrode was implanted 1 mm anterior to the lambda on the midline with a 10° backward angle in the ventromedial tegmentum and 8.0 ± 0.2 mm below the surface of the brain. Two hundred square pulses of 0.5 milliseconds were delivered by a stimulator at an intensity of 300 μA and frequencies of 1 Hz and 5 Hz. The inhibition of the spontaneous activity of the hippocampus pyramidal neuron takes place, at least in part, owing to activation of the postsynaptic inhibitory 5-HT1A receptors by 5-HT, endogenously released as a result of stimulation of the 5-HT afferents.36 The different frequencies were used to determine the function of terminal 5-HT1B autoreceptors.37 This approach is based on the evidence that when the frequency is increased to 5 Hz, more 5-HT is released in the extracellular cleft, which consequently exerts a greater negative feedback on the 5-HT release via the terminal 5-HT1B autoreceptors.37 Therefore, the release of 5-HT is inhibited quickly during the stimulation at 5 Hz, leading to a smaller release of 5-HT in the synapse for each action potential reaching the terminals. The stimulation pulses and the firing activity were analyzed by computer using Spike 2. Peristimulus time histograms of hippocampus pyramidal neurons were generated to determine the suppression of firing measured in SIL in miliseconds. The SIL represents the duration of a total suppression of the hippocampal neuron by endogenously released 5-HT.
Drugs
Pramipexole was generously provided by Boehringer Ingelheim Pharmaceuticals (Ingelheim); haloperidol, 5-HT creatinine sulfate, DA hydrochloride, quisqualic acid and WAY 100 635 were purchased from Sigma. All drugs except haloperidol were dissolved in distilled water. Haloperidol was dissolved in distilled water acidified with lactic acid, followed by pH control and normalization as needed.
Statistical analyses
All results are expressed as means and standard errors of the mean (SEM). The n values represent the number of neurons tested. In the experiments where pharmacologic agents were systemically administered, only the last neuron in each rat was used to avoid residual drug effects. Data were obtained from 5–7 rats per experimental group. Statistical comparisons were carried out using the 2-tailed Student t test when a parameter was studied in control and treated rats. We used the paired Student t test to assess the statistical significance of the variation of the measured parameter from the same neurons under 2 conditions, such as the SIL at 1 Hz and 5 Hz (for 5-HT). We performed analysis of covariance to assess statistical significance of the difference in the degree of reduction in the response of hippocampus neurons when the frequency of stimulation was increased from 1 to 5 Hz in control and PPX-treated rats. Statistical significance was set at p < 0.05.
Results
Effect of 14-day PPX administration on the responsiveness of PFC pyramidal neurons to exogenous DA
In line with previous data, DA applied microiontophoretically to the cell body of the neuron resulted in suppression of 31 of 36 recorded PFC pyramidal neurons. Such variability is normal for the given type of neurons in the PFC and has been documented in previous studies.38–40 Dopamine-induced inhibition of spontaneous firing in PFC pyramidal neurons is believed to be mediated by the D2 receptors.29,41 Therefore, to determine the responsiveness of postsynaptic D2 receptors, only the neurons responding with inhibition were analyzed. Chronic PPX treatment left the responsiveness of these receptors at the control level, as indicated by the unchanged number of spikes suppressed per nA (control: mean 14 [SEM 5], n = 31, baseline firing rate mean 0.7 [SEM 0.4] Hz v. PPX 14 days: mean 18 [SEM 8], n = 38, baseline firing rate mean 0.9 [SEM 0.5] Hz, p = 0.09 for SIL and p = 0.56 for firing; Fig. 1A and B).
Fig. 1.
Assessment of tonic activation of D2 receptors in the prefrontal cortex (PFC). Integrated firing rate histograms of PFC pyramidal neurons illustrating systemic administration of the D2 receptor antagonist haloperidol (HAL; 0.2 mg/kg) in (A) rats subjected to saline (controls) and (B) rats receiving a 14-day regimen of pramipexole (PPX; 1 mg/kg/d). Each bar corresponds to 50-second application of dopamine (DA), and the number above each bar corresponds to the ejection current in nA. The arrow indicates a point of injection of 200 μg/kg of haloperidol. (C) The overall effect of systemic administration of haloperidol on baseline firing of PFC pyramidal neurons in controls and rats receiving PPX (expressed as % of change in basal firing). **p = 0.002.
Effect of 14-day PPX administration on the degree of tonic activation of D2 receptors in the PFC
In the control group the blockade of inhibitory D2 receptors located on the cell body of PFC pyramidal neurons, achieved with the systemic administration of the selective D2 antagonist haloperidol, led to the decrease of their firing rate (Fig. 1A). However, after sustained PPX administration, this blockade led to a significant 104% disinhibition in the firing rate of pyramidal neurons compared with the control value (control n = 6, PPX 14d n = 7; t12 = 4.01, p = 0.002; Fig. 1B and C). The increase in firing following haloperidol administration indicates that the overall DA tone is increased by prolonged PPX administration.
Effect of 14-day PPX administration on the DA release potential in the PFC
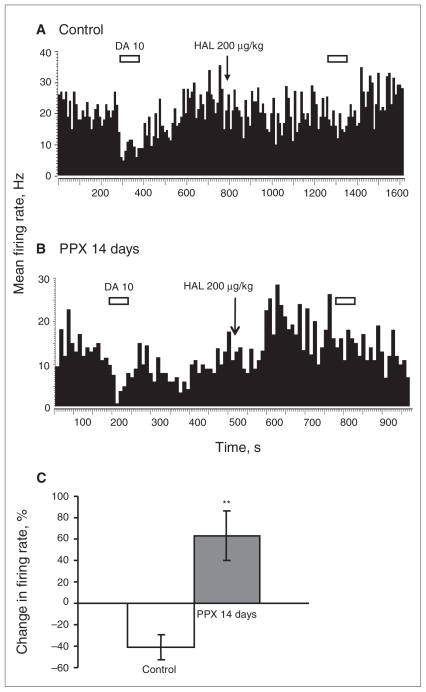
To assess the ability of PPX to modify the endogenous release of DA, the VTA bundle sending afferents to the PFC via the mesocortical pathway was electrically stimulated after DA neurons recovered their normal firing rate following sustained administration of PPX.27 Dopamine released as a result of stimulation produced a suppressant effect on PFC neuronal firing, which was quantified as SIL. In rats treated with PPX for 14 days, SIL remained at the level of the control group under both stimulation conditions (control: SIL = mean 130 [SEM 9], n = 20 v. PPX 14d: SIL = mean 115 [SEM 6], n = 34; p = 0.25; Fig. 2), indicating that the release of DA per impulse was not altered by prolonged administration of PPX.
Fig. 2.
Assessment of dopamine (DA) release in the prefrontal cortex (PFC). Peristimulus time histograms illustrating effects of stimulation of the ascending DA pathway on the firing activity of PFC pyramidal neurons in (A) controls and (B) rats administered pramipexole (PPX) for 14 days. The 2 10-ms bins at time 0 represent the stimulation artifacts. SIL = absolute silence value.
Effect of 14-day PPX administration on the responsiveness of dorsal hippocampus pyramidal neurons to exogenous 5-HT
The firing rate of hippocampus pyramidal neurons in control rats was decreased by 5-HT applied microiontophoretically in a current-dependent fashion. The sensitivity of the postsynaptic 5-HT1A receptors located on the cell body of CA3 pyramidal neurons was found to be unaltered by PPX, as indicated by the lack of change in the number of spikes suppressed per nA in comparison to the control group (control: mean 18 [SEM 1], n = 19 v. PPX 14 d: mean 18 [SEM 1], n = 24, p = 0.44; Fig. 3A and B).
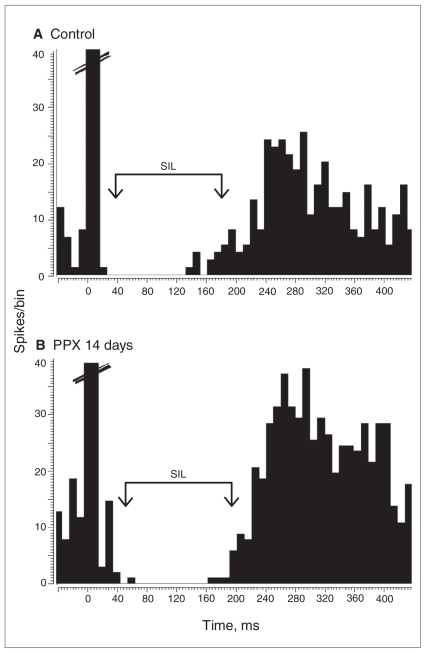
Fig. 3.
Assessment of tonic activation of 5-HT1A receptors in the dorsal hippocampus. Integrated firing rate histograms of dorsal hippocampus CA3 pyramidal neurons illustrating systemic administration of the 5-HT1A receptor antagonist WAY 100 635 in 4 incremental doses of (A) controls treated with 25 μg/kg of saline and (B) rats treated with 14 days of pramipexole (PPX; 1 mg/kg/d). Each bar corresponds to a 50-second application of 5-HT, and the number above each bar corresponds to the ejection current in nA. Each arrow indicates a single injection of 25 μg/kg of WAY 100 635. (C) The overall effect of cumulative systemic administration of WAY 100 635 on baseline firing of CA3 pyramidal neurons in control and PPX-treated rats (expressed as % of change in basal firing). **p = 0.004.
Effect of 14-day PPX administration on the degree of tonic activation of hippocampal 5-HT1A receptors
In the control group, blockade of inhibitory 5-HT1A receptors located on CA3 pyramidal neurons, achieved with the systemic administration of the selective 5-HT1A receptor antagonist WAY 100 635, did not modify their firing rate (Fig. 3A). Following 14 days of PPX administration this blockade led to the significant 142% disinhibition in the firing rate of pyramidal neurons in the dorsal hippocampus compared with the control value (control n = 6, PPX 14 d n = 7; t11 = 3.57, p = 0.044; Fig. 3B and C). This increase, also observed with all effective antidepressant treatments,42 indicates that the overall 5-HT tone is enhanced by long-term PPX administration.
Effect of 14-day PPX administration on the responsiveness of dorsal hippocampal pyramidal neurons to endogenous 5-HT
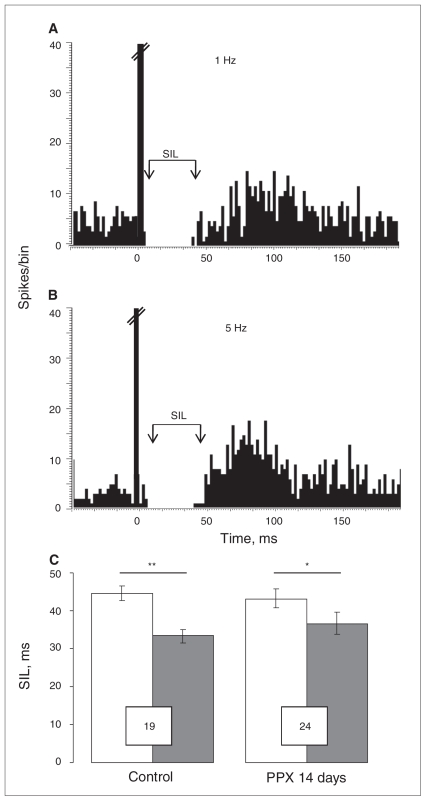
To assess the ability of PPX to modify the endogenous release of 5-HT, the 5-HT bundle containing most of the brain 5-HT afferents was electrically stimulated at a physiologic (1 Hz) and a maximal (5 Hz) rate.37 Serotonin released as a result of stimulation produced a suppressant effect on hippocampal neuronal firing, which was quantified as SIL. In rats exposed to PPX for 14 days, SIL remained at the level of the control group (Fig. 4), indicating that the sensitivity of terminal 5-HT1B receptors controlling the release of 5-HT remained unchanged.
Fig. 4.
Assessment of serotonin (5-HT) release and sensitivity of terminal 5-HT1B receptors. Peristimulus time histograms illustrating effects of stimulation of the ascending 5-HT pathway, with (A) 1 Hz and (B) 5 Hz, on the firing activity of CA3 pyramidal neurons in controls and rats treated with 14 days of pramipexole (PPX). (C) The suppressing effect of the 5-HT pathway stimulations (1 Hz and 5 Hz) on the firing activity of hippocampal neurons in rats administered either NaCl (control) or PPX. Numbers in the boxes indicate the number of assessed neurons. The 10 msec bin at time 0 represents the stimulation artifact. Stimulation at a frequency of 1 Hz is indicated in white and that at 5 Hz in black. *p = 0.023, **p = 0.006. SIL = absolute silence value.
Discussion
The present electrophysiologic study showed that long-term administration of the D2-like agonist PPX increased overall DA neurotransmission, as indicated by the disinhibition of spontaneous neuronal firing of PFC pyramidal neurons by systemic administration of the D2-like antagonist haloperidol. This enhancement of DA tone was not attributable to alterations of the release of DA or to an enhanced responsiveness of postsynaptic D2 receptors. We therefore conclude that it resulted from a summation of the normalized DA firing, presumably restoring DA release, and the presence of PPX in the synapse. The present study also showed that prolonged PPX administration increased the overall 5-HT tone without changing the release of 5-HT per action potential reaching hippocampus terminals or the sensitivity of terminal 5-HT1B autoreceptors. It can thus be concluded that the increase in 5-HT neuronal transmission resulted from the enhanced firing of 5-HT neurons.27
The PFC is believed to be under tonic inhibitory influence from endogenous DA.29 Microiontophoretic DA administration has an inhibitory effect on the spontaneous firing rate of PFC pyramidal neurons.38 The same effect can be produced by endogenous DA, as evidenced by the suppression of PFC discharge in response to the VTA stimulation.39,40 Although both D1-like and D2-like receptors are present in the PFC,43,44 the suppressant effect of DA was shown to be mediated by the latter, as selective blockade of D2-like but not D1-like receptors reversed this action.29,41
It was previously documented that the PPX-induced activation of somatodendritic D2 autoreceptors in the VTA led to the decrease in the firing rate of DA neurons, driven by the negative feedback mechanism exerted by the cell body D2 autoreceptors.27 With ongoing administration of PPX over 14 days, these receptors desensitize, allowing the firing to return to baseline. Conversely, the degree of inhibition of PFC pyramidal neurons by both exogenous (iontophoretically applied) and endogenous (stimulation-induced) DA was equal in control and PPX-treated rats. This lack of change indicated an unaltered responsiveness of PFC postsynaptic D2 receptors after 14 days of sustained exposure to PPX. Nevertheless, in rats receiving PPX on a long-term basis, but not in the control group, blockade of the inhibitory D2 receptors by intravenous administration of the selective antagonist haloperidol led to significant disinhibition of the spontaneous firing of pyramidal neurons (Fig. 1). As the sensitivity of the receptors mediating this response was found to be unchanged, the observed PPX-induced increase in the tonic activation of D2 receptors in the PFC was most likely attributable to the direct effect of this D2 receptor agonist, present at the time of the experiment, on the target receptor summating with a normalized DA release resulting from a recovered DA firing activity.27 Yet, it needs to be mentioned that both DA modulation and its effects in the PFC are complex and multisided and that no unified view has yet been established.45 For instance, depending on the dose, the state of the system and the predominance of direct versus indirect activation, the same drug may produce opposing effects on the elicited responses.45–47 Terminal D2 receptors, which play a prominent role in the control of DA release in limbic structures, are fewer in number in the PFC.35,48 Thus, under physiologic conditions, their stimulation by endogenous DA and/or exogenous D2 receptor agonists plays a negligible role in the amount of DA released.49
The maintenance of proper mesocortical DA levels plays an important role in different aspects of attention and learning and in behavioural and physiologic mechanisms of the stress response.50–52 These functions are often perturbed in patients with depression and may be related to the decrease in DA levels. The decrease in function of the frontal lobe is one of the most constant findings in the depressive state.53–55 The normalization of the frontocortical metabolism is consistently seen in patients who achieve remission following pharmacologic antidepressant treatment.56–58 The VTA provides a dense DA projection to the PFC. The PPX-induced increase in DA function, known to be dampened in patients with depression, potentially leads to the normalization of the modulatory DA tone in the PFC and a consequent restoration of the functions controlled by this brain region.
As the 5-HT tone in the hippocampus was significantly increased after prolonged PPX administration, it was important to determine the mechanism of such an enhancement. The 5-HT ascending bundle was electrically stimulated, first, to assess the amount of 5-HT released per electrical pulse reaching the terminals and, second, to assess the sensitivity of terminal 5-HT1B autoreceptors that control 5-HT release. The stimulation at a physiologic rate of 1 Hz led to the same degree of suppression of the firing activity of CA3 pyramidal neurons in rats that received PPX for 14 days compared with controls. When the frequency of stimulation was increased from 1 Hz to 5 Hz, the suppression of the firing was reduced to a similar extent in treated and control rats, indicating an unaltered responsiveness of terminal 5-HT1B autoreceptors. This result stands in contrast with the decreased responsiveness of the terminal 5-HT1B autoreceptors occurring with long-term administration of selective serotonin reuptake inhibitors, such as citalopram, paroxetine, fluvoxamine and fluoxetine.59 The sensitivity of postsynaptic 5-HT1A receptors was also unaltered, as shown by the unchanged inhibitory potency of 5-HT applied on the CA3 pyramidal neurons by iontophoresis. Since 5-HT activating the postsynaptic 5-HT1A receptor equally suppressed the firing in rats exposed to PPX and controls, it can be concluded that in rats receiving PPX the disinhibition in response to 5-HT1A receptor blockade (Fig. 3) was due to an overall increase of the 5-HT tone, not the modified sensitivity of the receptor mediating this response.
This elevation of the 5-HT neuronal tone likely stemmed from the PPX-induced amplification in the firing rate of DR 5-HT neurons that occurred after the same 14-day, but not the 2-day, PPX regimen.27 Enhancement of the tonic activation of postsynaptic 5-HT1A receptors resulting from the increase in the firing rate of 5-HT neurons is not unique to PPX. The catecholamine-releasing agent bupropion and prolonged vagus nerve stimulation have been shown to produce an analogous change.60,61 Similarly to PPX, the increase in the spontaneous discharge of DR 5-HT neurons produced by subchronic administration of the atypical antipsychotic aripiprazole has been found to be due to activation of the D2-like receptors and desensitization of 5-HT1A autoreceptors.62 Such a phenomenon is in line with previous in vivo and in vitro studies documenting the enhancement of the 5-HT tone in response to the stimulation of DR D2-like receptors by prodopaminergic agents.63–65
Limitations
The present study has some limitations. First, unlike for the 5-HT sensitivity in the hippocampus, the sensitivity of postsynaptic DA in the frontal cortex was not assessed using a range of ejection currents of DA; therefore, it is possible that we may have missed a subtle difference in sensitivity. Nevertheless, the 10 nA current did not produce a maximal inhibition of firing, which would not place that value at the extremes of a current-effect curve. Second, the neuronal tone was assessed within the mesocortical system, but not within the mesolimbic system. Nevertheless, similar changes combining activation of postsynaptic D2 receptors with both endogenous DA and the exogenous agonist PPX are likely taking place within the mesolimbic system as well because VTA gives rise to the DA innervation in both circuits. Stress response and cognitive functions, regulated by the mesocortical DA, as well as hedonia, regulated by the mesolimbic DA, are impaired in patients with depression.66–68 Major depression is characterized by abnormalities in activity and/or functional connectivity within both these systems,69,70 thus changes in their function produced by prolonged PPX administration likely contribute to the clinical benefits of this drug in patients with MDD. Indeed, a recent clinical study documented that depressed patients responding to long-term treatment with PPX showed normalization of the regional blood flow in the orbitofrontal cortex, anteromedial and ventrolateral PFC, posterior cingulate, hippocampus and accumbens.16 Importantly, activity of these brain regions is known to be altered in the depressed state.23,24 It is noteworthy that the metabolic changes produced by sustained PPX closely follow those of antidepressants and deep-brain stimulation.71–73
Conclusion
Despite the lack of affinity toward any component of the 5-HT system, PPX produces a significant increase in 5-HT neurotransmission in an indirect manner. These observations with PPX therefore add to the large body of data showing the commonality of this change by all effective antidepressants tested thus far.42
The current study thus put into evidence that chronic treatment with the D2 agonist PPX increased DA neurotransmission in the PFC and 5-HT neurotransmission in the hippocampus in rats. Given the documented normalization of the brain function within the same regions in depressed patients treated with PPX,16 it is likely that the observed changes in the function of the above-mentioned modulatory mono-aminergic systems may underlie to some degree the clinical effectiveness of PPX in the treatment of depression.
Acknowledgments
This study was supported by the Canadian Institutes of Health Research grant to P. Blier. The authors thank Boehringer Ingelheim Pharmaceuticals for providing pramipexole. P. Blier received support for investigator initiated grants, and/or honoraria for advisory boards and/or speaking engagements from Astra-Zeneca, Biovail, Bristol Myers Squibbs, Eli Lilly & Company, Janssen Pharmaceuticals, Labopharm, Lundbeck, Pfizer, Schering-Plough/Merck, Servier, Takeda and Wyeth.
Footnotes
Competing interests: As above for P. Blier. None declared for O. Chernoloz and M. El Mansari.
Contributors: P. Blier designed the study. O. Chernoloz acquired the data, which all authors analyzed. O. Chernoloz wrote the article, which M. El Mansari and P. Blier reviewed. All authors approved its publication.
References
- 1.Guttman M, Jaskolka J. The use of pramipexole in Parkinson’s disease: Are its actions D3 mediated? Parkinsonism Relat Disord. 2001;7:231–4. doi: 10.1016/s1353-8020(00)00062-6. [DOI] [PubMed] [Google Scholar]
- 2.Piercey MF. Pharmacology of pramipexole, a dopamine D3-preferring agonist useful in treating Parkinson’s disease. Clin Neuropharmacol. 1998;21:141–51. [PubMed] [Google Scholar]
- 3.Reichmann H, Odin P, Brecht HM, et al. Changing dopamine agonist treatment in Parkinson’s disease: experiences with switching to pramipexole. J Neural Transm Suppl. 2006;71:17–25. doi: 10.1007/978-3-211-33328-0_2. [DOI] [PubMed] [Google Scholar]
- 4.Corrigan MH, Denahan AQ, Wright EC, et al. Comparison of pramipexole, fluoxetine, and placebo in patients with major depression. Depress Anxiety. 2000;11:58–65. doi: 10.1002/(sici)1520-6394(2000)11:2<58::aid-da2>3.0.co;2-h. [DOI] [PubMed] [Google Scholar]
- 5.Lattanzi L, Dell’Osso L, Cassano P, et al. Pramipexole in treatment-resistant depression: a 16-week naturalistic study. Bipolar Disord. 2002;4:307–314. doi: 10.1034/j.1399-5618.2002.01171.x. [DOI] [PubMed] [Google Scholar]
- 6.Sporn J, Ghaemi SN, Sambur MR, et al. Pramipexole augmentation in the treatment of unipolar and bipolar depression: a retrospective chart review. Ann Clin Psychiatry. 2000;12:137–40. doi: 10.1023/a:1009060800999. [DOI] [PubMed] [Google Scholar]
- 7.Cassano P, Lattanzi L, Soldani F, et al. Pramipexole in treatment-resistant depression: an extended follow-up. Depress Anxiety. 2004;20:131–8. doi: 10.1002/da.20038. [DOI] [PubMed] [Google Scholar]
- 8.Goldberg JF, Burdick KE, Endick CJ. Preliminary randomized, double-blind, placebo-controlled trial of pramipexole added to mood stabilizers for treatment-resistant bipolar depression. Am J Psychiatry. 2004;161:564–6. doi: 10.1176/appi.ajp.161.3.564. [DOI] [PubMed] [Google Scholar]
- 9.Bennett JP, Jr, Piercey MF. Pramipexole — a new dopamine agonist for the treatment of Parkinson’s disease. J Neurol Sci. 1999;163:25–31. doi: 10.1016/s0022-510x(98)00307-4. [DOI] [PubMed] [Google Scholar]
- 10.Tandberg E, Larsen JP, Aarsland D, et al. The occurrence of depression in Parkinson’s disease: a community-based study. Arch Neurol. 1996;53:175–9. doi: 10.1001/archneur.1996.00550020087019. [DOI] [PubMed] [Google Scholar]
- 11.Dunlop BW, Nemeroff CB. The role of dopamine in the pathophysiology of depression. Arch Gen Psychiatry. 2007;64:327–37. doi: 10.1001/archpsyc.64.3.327. [DOI] [PubMed] [Google Scholar]
- 12.Brocco M, Dekeyne A, Papp M, et al. Antidepressant-like properties of the anti-Parkinson agent, piribedil, in rodents: mediation by dopamine D2 receptors. Behav Pharmacol. 2006;17:559–72. doi: 10.1097/01.fbp.0000236267.41806.5b. [DOI] [PubMed] [Google Scholar]
- 13.Izumi T, Inoue T, Kitagawa N, et al. Open pergolide treatment of tricyclic and heterocyclic antidepressant-resistant depression. J Affect Disord. 2000;61:127–32. doi: 10.1016/s0165-0327(99)00199-8. [DOI] [PubMed] [Google Scholar]
- 14.Muscat R, Papp M, Willner P. Antidepressant-like effects of dopamine agonists in an animal model of depression. Biol Psychiatry. 1992;31:937–46. doi: 10.1016/0006-3223(92)90119-k. [DOI] [PubMed] [Google Scholar]
- 15.Waehrens J, Gerlach J. Bromocriptine and imipramine in endogenous depression. A double-blind controlled trial in outpatients. J Affect Disord. 1981;3:193–202. doi: 10.1016/0165-0327(81)90044-6. [DOI] [PubMed] [Google Scholar]
- 16.Mah L, Zarate CA, Nugent AC, et al. Neural mechanisms of antidepressant efficacy of the dopamine receptor agonist pramipexole in treatment of bipolar depression. Int J Neuropsychopharmacol. 2011;14:545–51. doi: 10.1017/S1461145710001203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Rizzo V, Aricò I, Mastroeni C, et al. Dopamine agonists restore cortical plasticity in patients with idiopathic restless legs syndrome. Mov Disord. 2009;24:710–5. doi: 10.1002/mds.22436. [DOI] [PubMed] [Google Scholar]
- 18.Hiramoto T, Kanda Y, Satoh Y, et al. Dopamine D2 receptor stimulation promotes the proliferation of neural progenitor cells in adult mouse hippocampus. Neuroreport. 2007;18:659–64. doi: 10.1097/WNR.0b013e3280bef9d3. [DOI] [PubMed] [Google Scholar]
- 19.Höglinger GU, Rizk P, Muriel MP, et al. Dopamine depletion impairs precursor cell proliferation in Parkinson’s disease. Nat Neurosci. 2004;7:726–35. doi: 10.1038/nn1265. [DOI] [PubMed] [Google Scholar]
- 20.Czéh B, Simon M, Schmelting B, et al. Astroglial plasticity in the hippocampus is affected by chronic psychosocial stress and concomitant fluoxetine treatment. Neuropsychopharmacology. 2006;31:1616–26. doi: 10.1038/sj.npp.1300982. [DOI] [PubMed] [Google Scholar]
- 21.McEwen BS, Magarinos AM. Stress and hippocampal plasticity: implications for the pathophysiology of affective disorders. Hum Psychopharmacol. 2001;16(Suppl 1):S7–19. doi: 10.1002/hup.266. [DOI] [PubMed] [Google Scholar]
- 22.Radley JJ, Rocher AB, Rodriguez A, et al. Repeated stress alters dendritic spine morphology in the rat medial prefrontal cortex. J Comp Neurol. 2008;507:1141–50. doi: 10.1002/cne.21588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Drevets WC. Prefrontal cortical-amygdalar metabolism in major depression. Ann N Y Acad Sci. 1999;877:614–37. doi: 10.1111/j.1749-6632.1999.tb09292.x. [DOI] [PubMed] [Google Scholar]
- 24.Drevets WC, Videen TO, Price JL, et al. A functional anatomical study of unipolar depression. J Neurosci. 1992;12:3628–41. doi: 10.1523/JNEUROSCI.12-09-03628.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Manji HK, Drevets WC, Charney DS. The cellular neurobiology of depression. Nat Med. 2001;7:541–7. doi: 10.1038/87865. [DOI] [PubMed] [Google Scholar]
- 26.Santarelli L, Saxe M, Gross C, et al. Requirement of hippocampal neurogenesis for the behavioral effects of antidepressants. Science. 2003;301:805–9. doi: 10.1126/science.1083328. [DOI] [PubMed] [Google Scholar]
- 27.Chernoloz O, El Mansari M, Blier P. Sustained administration of pramipexole modifies the spontaneous firing of dopamine, norepinephrine, and serotonin neurons in the rat brain. Neuropsychopharmacology. 2009;34:651–61. doi: 10.1038/npp.2008.114. [DOI] [PubMed] [Google Scholar]
- 28.Paxinos G, Watson C. The rat brain in stereotaxic coordinates. 2nd ed. San Diego (CA): Academic Press; 1986. [Google Scholar]
- 29.Sesack SR, Bunney BS. Pharmacology of dopamine-induced electrophysiological responses in the rat prefrontal cortex: D1- or D2-mediated? Ann N Y Acad Sci. 1988;537:529–30. [Google Scholar]
- 30.Ranck JB. Behavioral correlates and firing repertoires of neurons in the dorsal hippocampal formation and septum of unrestrained rats. In: Isaacson RC, Pribram KH, editors. The hippocampus. New York (NY): Plenum; 1975. pp. 207–44. [Google Scholar]
- 31.Kandel ER, Spencer WA. Electrophysiology of hippocampal neurons. II. After-potentials and repetitive firing. J Neurophysiol. 1961;24:243–59. doi: 10.1152/jn.1961.24.3.243. [DOI] [PubMed] [Google Scholar]
- 32.Au-Young SMW, Shen H, Yang CR. Medial prefrontal cortical output neurons to the ventral tegmental area (VTA) and their responses to burst-patterned stimulation of the VTA: neuroanatomical and in vivo electrophysiological analyses. Synapse. 1999;34:245–55. doi: 10.1002/(SICI)1098-2396(19991215)34:4<245::AID-SYN1>3.0.CO;2-D. [DOI] [PubMed] [Google Scholar]
- 33.Godbout R, Mantz J, Pirot S, et al. Inhibitory influence of the mesocortical dopaminergic neurons on their target cells: electrophysiological and pharmacological characterization. J Pharmacol Exp Ther. 1991;258:728–38. [PubMed] [Google Scholar]
- 34.Floresco SB, Grace AA. Gating of hippocampal-evoked activity in prefrontal cortical neurons by inputs from the mediodorsal thalamus and ventral tegmental area. J Neurosci. 2003;23:3930–43. doi: 10.1523/JNEUROSCI.23-09-03930.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Bannon MJ, Wolf ME, Roth RH. Pharmacology of dopamine neurons innervating the prefrontal cingulate and piriform cortices. Eur J Pharmacol. 1983;92:119–25. doi: 10.1016/0014-2999(83)90116-4. [DOI] [PubMed] [Google Scholar]
- 36.Chaput Y, Blier P, De Montigny C. In vivo electrophysiological evidence for the regulatory role of autoreceptors on serotonergic terminals. J Neurosci. 1986;6:2796–801. doi: 10.1523/JNEUROSCI.06-10-02796.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Chaput Y, Blier P, De Montigny C. In vivo electrophysiological evidence for the regulatory role of autoreceptors on serotonergic terminals. J Neurosci. 1986;6:2796–801. doi: 10.1523/JNEUROSCI.06-10-02796.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Bunney BS, Aghajanian GK. Dopamine and norepinephrine innervated cells in the rat prefrontal cortex: pharmacological differentiation using microiontophoretic techniques. Life Sci. 1976;19:1783–9. doi: 10.1016/0024-3205(76)90087-4. [DOI] [PubMed] [Google Scholar]
- 39.Ferron A, Thierry AM, Le Douarin C, et al. Inhibitory influence of the mesocortical dopaminergic system on spontaneous activity or excitatory response induced from the thalamic mediodorsal nucleus in the rat medial prefrontal cortex. Brain Res. 1984;302:257–65. doi: 10.1016/0006-8993(84)90238-5. [DOI] [PubMed] [Google Scholar]
- 40.Thierry AM, Le Douarin C, Penit J. Variation in the ability of neuroleptics to block the inhibitory influence of dopaminergic neurons on the activity of cells in the rat prefrontal cortex. Brain Res Bull. 1986;16:155–60. doi: 10.1016/0361-9230(86)90027-4. [DOI] [PubMed] [Google Scholar]
- 41.Mantz J, Godbout R, Pirot S, et al. Inhibitory effects of mesocortical dopaminergic neurons on their target cells: electrophysiological and pharmacological characterization. Neurochem Int. 1992;20(Suppl):251S–254S. doi: 10.1016/0197-0186(92)90247-o. [DOI] [PubMed] [Google Scholar]
- 42.Haddjeri N, Blier P, De Montigny C. Long-term antidepressant treatments result in a tonic activation of forebrain 5-HT(1A) receptors. J Neurosci. 1998;18:10150–6. doi: 10.1523/JNEUROSCI.18-23-10150.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Marchais D, Tassin JP, Bockaert J. Dopaminergic component of [3H]spiroperidol binding in the rat anterior cerebral cortex. Brain Res. 1980;183:235–40. doi: 10.1016/0006-8993(80)90136-5. [DOI] [PubMed] [Google Scholar]
- 44.Bockaert J, Tassin JP, Thierry AM. Characteristics of dopamine and β adrenergic sensitive adenylate cyclases in the frontal cerebral cortex of the rat. Comparative effects of neuroleptics on frontal cortex and striatal dopamine sensitive adenylate cyclases. Brain Res. 1977;122:71–86. doi: 10.1016/0006-8993(77)90663-1. [DOI] [PubMed] [Google Scholar]
- 45.Seamans JK, Yang CR. The principal features and mechanisms of dopamine modulation in the prefrontal cortex. Prog Neurobiol. 2004;74:1–58. doi: 10.1016/j.pneurobio.2004.05.006. [DOI] [PubMed] [Google Scholar]
- 46.Gorelova N, Seamans JK, Yang CR. Mechanisms of dopamine activation of fast-spiking interneurons that exert inhibition in rat prefrontal cortex. J Neurophysiol. 2002;88:3150–66. doi: 10.1152/jn.00335.2002. [DOI] [PubMed] [Google Scholar]
- 47.Gulledge AT, Jaffe DB. Dopamine decreases the excitability of layer V pyramidal cells in the rat prefrontal cortex. J Neurosci. 1998;18:9139–51. doi: 10.1523/JNEUROSCI.18-21-09139.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Neve KA, Seamans JK, Trantham-Davidson H. Dopamine receptor signaling. J Recept Signal Transduct Res. 2004;24:165–205. doi: 10.1081/rrs-200029981. [DOI] [PubMed] [Google Scholar]
- 49.Santiago M, Machado A, Cano J. Regulation of prefrontal cortical dopamine release by dopamine receptor agonists and antagonists. Eur J Pharmacol. 1993;239:83–91. doi: 10.1016/0014-2999(93)90979-r. [DOI] [PubMed] [Google Scholar]
- 50.Robbins TW. From arousal to cognition: the integrative position of the prefrontal cortex. Prog Brain Res. 2000;126:469–83. doi: 10.1016/S0079-6123(00)26030-5. [DOI] [PubMed] [Google Scholar]
- 51.Berridge CW, Espana RA, Stalnaker TA. Stress and coping: asymmetry of dopamine efferents within the prefrontal cortex. In: Hugdahl K, Davidson RJ, editors. The asymmetryical brain. Cambridge (MA): MIT Press; 2003. pp. 69–104. [Google Scholar]
- 52.Deutch AY, Roth RH. The determinants of stress-induced activation of the prefrontal cortical dopamine system. Prog Brain Res. 1990;85:367–402. doi: 10.1016/s0079-6123(08)62691-6. discussion 402–3. [DOI] [PubMed] [Google Scholar]
- 53.Drevets WC, Price JL, Furey ML. Brain structural and functional abnormalities in mood disorders: implications for neurocircuitry models of depression. Brain Struct Funct. 2008;213:93–118. doi: 10.1007/s00429-008-0189-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Drevets WC. Prefrontal cortical-amygdalar metabolism in major depression. Ann N Y Acad Sci. 1999;877:614–37. doi: 10.1111/j.1749-6632.1999.tb09292.x. [DOI] [PubMed] [Google Scholar]
- 55.Mayberg HS, Liotti M, Brannan SK, et al. Reciprocal limbic-cortical function and negative mood: converging PET findings in depression and normal sadness. Am J Psychiatry. 1999;156:675–82. doi: 10.1176/ajp.156.5.675. [DOI] [PubMed] [Google Scholar]
- 56.Kennedy SH, Evans KR, Krüger S, et al. Changes in regional brain glucose metabolism measured with positron emission tomography after paroxetine treatment of major depression. Am J Psychiatry. 2001;158:899–905. doi: 10.1176/appi.ajp.158.6.899. [DOI] [PubMed] [Google Scholar]
- 57.Mayberg HS, Brannan SK, Tekell JL, et al. Regional metabolic effects of fluoxetine in major depression: serial changes and relationship to clinical response. Biol Psychiatry. 2000;48:830–43. doi: 10.1016/s0006-3223(00)01036-2. [DOI] [PubMed] [Google Scholar]
- 58.Stefurak T, Mahurin R, Soloman D. Response specific regional metabolism changes with fluoxetine treatment in depressed Parkinson’s patients. Mov Disord. 2001;16(Suppl 1):S39. [Google Scholar]
- 59.Piñeyro G, Blier P. Autoregulation of serotonin neurons: role in antidepressant drug action. Pharmacol Rev. 1999;51:533–91. [PubMed] [Google Scholar]
- 60.El Mansari M, Ghanbari R, Janssen S, et al. Sustained administration of bupropion alters the neuronal activity of serotonin, norepinephrine but not dopamine neurons in the rat brain. Neuropharmacology. 2008;55:1191–8. doi: 10.1016/j.neuropharm.2008.07.028. [DOI] [PubMed] [Google Scholar]
- 61.Manta S, Dong J, Debonnel G, et al. Enhancement of the function of rat serotonin and norepinephrine neurons by sustained vagus nerve stimulation. J Psychiatry Neurosci. 2009;34:272–80. [PMC free article] [PubMed] [Google Scholar]
- 62.Chernoloz O, El Mansari M, Blier P. Electrophysiological studies in the rat brain on the basis for aripiprazole augmentation of antidepressants in major depressive disorder. Psychopharmacology (Berl) 2009;206:335–44. doi: 10.1007/s00213-009-1611-7. [DOI] [PubMed] [Google Scholar]
- 63.Ferré S, Artigas F. Dopamine D2 receptor-mediated regulation of serotonin extracellular concentration in the dorsal raphe nucleus of freely moving rats. J Neurochem. 1993;61:772–5. doi: 10.1111/j.1471-4159.1993.tb02187.x. [DOI] [PubMed] [Google Scholar]
- 64.Haj-Dahmane S. D2-like dopamine receptor activation excites rat dorsal raphe 5-HT neurons in vitro. Eur J Neurosci. 2001;14:125–34. doi: 10.1046/j.0953-816x.2001.01616.x. [DOI] [PubMed] [Google Scholar]
- 65.Aman TK, Shen RY, Haj-Dahmane S. D2-like dopamine receptors depolarize dorsal raphe serotonin neurons through the activation of nonselective cationic conductance. J Pharmacol Exp Ther. 2007;320:376–85. doi: 10.1124/jpet.106.111690. [DOI] [PubMed] [Google Scholar]
- 66.Nestler EJ, Carlezon WA., Jr The mesolimbic dopamine reward circuit in depression. Biol Psychiatry. 2006;59:1151–9. doi: 10.1016/j.biopsych.2005.09.018. [DOI] [PubMed] [Google Scholar]
- 67.Swerdlow NR, Koob GF. Dopamine, schizophrenia, mania, and depression: toward a unified hypothesis of cortico-striato-pallido-thalamic function. Behav Brain Sci. 1987;10:197–245. [Google Scholar]
- 68.Mayberg HS. Modulating dysfunctional limbic-cortical circuits in depression: towards development of brain-based algorithms for diagnosis and optimised treatment. Br Med Bull. 2003;65:193–207. doi: 10.1093/bmb/65.1.193. [DOI] [PubMed] [Google Scholar]
- 69.Anand A, Li Y, Wang Y, et al. Activity and connectivity of brain mood regulating circuit in depression: a functional magnetic resonance study. Biol Psychiatry. 2005;57:1079–88. doi: 10.1016/j.biopsych.2005.02.021. [DOI] [PubMed] [Google Scholar]
- 70.Bench CJ, Friston LKJ, Brown RG, et al. Regional cerebral blood flow in depression measured by positron emission tomography: the relationship with clinical dimensions. Psychol Med. 1993;23:579–90. doi: 10.1017/s0033291700025368. [DOI] [PubMed] [Google Scholar]
- 71.Drevets WC. Orbitofrontal cortex function and structure in depression. Ann N Y Acad Sci. 2007;1121:499–527. doi: 10.1196/annals.1401.029. [DOI] [PubMed] [Google Scholar]
- 72.Drevets WC, Bogers W, Raichle ME. Functional anatomical correlates of antidepressant drug treatment assessed using PET measures of regional glucose metabolism. Eur Neuropsychopharmacol. 2002;12:527–44. doi: 10.1016/s0924-977x(02)00102-5. [DOI] [PubMed] [Google Scholar]
- 73.Lozano AM, Mayberg HS, Giacobbe P, et al. Subcallosal cingulate gyrus deep brain stimulation for treatment-resistant depression. Biol Psychiatry. 2008;64:461–7. doi: 10.1016/j.biopsych.2008.05.034. [DOI] [PubMed] [Google Scholar]