Abstract

The ligand binding site of Cys-loop receptors is dominated by aromatic amino acids. In GABAC receptors, these are predominantly tyrosine residues, with a number of other aromatic residues located in or close to the binding pocket. Here we examine the roles of these residues using substitution with both natural and unnatural amino acids followed by functional characterization. Tyr198 (loop B) has previously been shown to form a cation−π interaction with GABA; the current data indicate that none of the other aromatic residues form such an interaction, although the data indicate that both Tyr102 and Phe138 may contribute to stabilization of the positively charged amine of GABA. Tyr247 (loop C) was very sensitive to substitution and, combined with data from a model of the receptor, suggest a π–π interaction with Tyr241 (loop C); here again functional data show aromaticity is important. In addition the hydroxyl group of Tyr241 is important, supporting the presence of a hydrogen bond with Arg104 suggested by the model. At position Tyr102 (loop D) size and aromaticity are important; this residue may play a role in receptor gating and/or ligand binding. The data also suggest that Tyr167, Tyr200, and Tyr208 have a structural role while Tyr106, Trp246, and Tyr251 are not critical. Comparison of the agonist binding site “aromatic box” across the superfamily of Cys-loop receptors reveals some interesting parallels and divergences.
Keywords: Cys-loop receptor, binding site, unnatural amino acid mutagenesis, cation−π interaction, π−π interaction
The agonist binding sites of all Cys-loop receptors are rich in aromatics, and the crystal structure of AChBP,1 a nACh receptor subunit,2 the prokaryotic homologues ELIC and GLIC,3−5 and the recently published GluCl structure6 all reveal a cluster of aromatic residues, the so-called “aromatic box”, which, in vertebrate receptors at least, encapsulates the agonist. One of these aromatic box residues has been shown to have a cation−π interaction with the natural agonist in most Cys-loop receptors studied to date, and the importance of such an interaction in this protein family is well-established.7 However, apart from this particular aromatic residue, information about the roles of the aromatic residues is scarce. This is of considerable interest as such studies provide essential information for verifying lower resolution structural data, and also for modeling and docking studies.
GABA, the major inhibitory neurotransmitter in the mammalian CNS, mediates its effects via ionotropic (GABAA or GABAC) and metabotropic (GABAB) receptors. GABAC receptors, which are constituted from ρ subunits,8 are a subclass of GABAA receptors, to which they have strong structural and functional similarities. GABAC receptors, however, have distinct pharmacological properties: GABAC receptor-mediated responses are not inhibited by bicuculline (the classic competitive GABAA receptor antagonist) nor induced by baclofen (the classic GABAB receptor agonist). Thus, there are clearly some significant differences in the microstructures of these different GABA binding sites.
A model of the GABAC receptor binding site, based on the structure of the acetylcholine binding protein AChBP, indicates that the site is dominated by aromatic amino acids.9 This is to be expected, as all Cys-loop receptor binding pockets contain a high proportion of aromatic amino acids.10 However, the GABAC receptor is unusual in that there are no tryptophan residues in the binding pocket; where Trp is positioned in related nACh, 5-HT3 and MOD-1 receptor binding sites, it has been replaced by Tyr in the GABAC receptor (Figure 1; Table 1). Experimental data to date indicate that the model is relatively accurate, but, as it is generated from a homologous protein, it has a number of limitations and some aspects will be inaccurate. Thus, it is important to use experimental evidence to prove or disprove specific details.
Figure 1.
Sequence alignments for the ligand binding domain of the GABAC and other Cys loop receptor subunits. Residues with similar chemical properties are shaded. The approximate locations of the loops that constitute the binding pocket are indicated with black bars. Those residues examined in this study are stared.
Table 1. Alignment of Residues Contributing to the Aromatic Box in Different Cys-Loop Receptorsa.
| loop C |
||||||
|---|---|---|---|---|---|---|
| receptor | loop A | loop B | C1 | C2 | loop D | ref |
| nACh | Y93 | W149 | Y190 | Y198 | W55 | (13) |
| 5-HT3 | F130 | W183 | F226 | F234 | W90 | (12) |
| MOD-1 | C120 | Y180 | Y221 | W226 | F83 | (14) |
| GABAA | Y97 | Y157 | F200 | Y205 | F65 | (19) |
| GABAC | F138 | Y198 | Y241 | Y247 | Y102 | (11) |
| RDL | F146 | F206 | L249 | Y254 | Y109 | (16) |
| Glycine | F99 | F159 | Y202 | F207 | F63 | (24) |
Residues in bold have been shown to be involved in cation−π interactions with agonists.
We have previously established that GABA makes a potent cation−π interaction to Tyr198 on binding to the GABAC receptor.11 This is the residue that aligns with a Trp that has a similar interaction in nACh and 5-HT3 receptors,12,13 and was the first case in which a tyrosine was shown to play this role. Cation−π interactions are increasingly being appreciated as important for stabilizing both protein–protein and protein–ligand interactions, and, as more proteins structures are being revealed at atomic resolution, we have an ever increasing understanding of how and where these interactions are formed.7 It has long been known that Trp will form an energetically more favorable cation-π interaction than Tyr, and indeed it is clear from earlier studies on Cys-loop receptors that a Trp is preferred over a Tyr when there is a choice between them: In the MOD-1 receptor, where the Trp that forms a cation−π interaction with 5-HT has been replaced with a Tyr, and a Tyr that is located 8–10 Å distant on the other side of the binding pocket has been replaced with a Trp, 5-HT will now form a cation−π interaction with the Trp.14 There is no Trp in the GABAC binding pocket, but the recent model of the binding pocket of the GABAC receptor shows that four Tyr residues are within 5 Å of the amino group of GABA (Figure 2). Since it is well-established that multiple π systems can make cation−π interactions to a single cation,7,15 and recently two aromatic residues have been shown to contribute to such an interaction in an insect GABA receptor,16 one of the aims of this study was to probe this possibility in the GABAC receptor. We also explore the roles of other aromatics in or close to the binding site, and compare and contrast the roles these residues play with analogous residues in other Cys-loop receptors.
Figure 2.

(A) Model of the GABAC receptor binding site showing GABA docked into the binding pocket. For ease of viewing, only two of the five subunits are shown. Inset: Enlargement of the binding pocket showing GABA surrounded by aromatic binding site residues. (B) The location of GABA docked into the GABAC receptor binding pocket is consistent with a cation-π interaction at Y198 as previously shown.11 The model reveals there is also the potential for aromatic stabilization of GABA by Tyr102 and Phe138, as suggested by the data. The location of Arg104, which has previously been shown to be critical for GABA binding, is also consistent with the presence of a hydrogen bond with Tyr247 as suggested by the data.
Results and Discussion
Functional Studies of GABAC Receptors
Xenopus oocytes injected with WT GABAC cRNA showed robust responses to GABA application, and dose–response curves revealed an EC50 of 1.4 ± 0.2 μM, and Hill coefficient of 1.6 ± 0.3 (n = 4). These data are in good agreement with those previously published.17 For validation of the nonsense suppression technique, tyrosine was incorporated via acylated tRNA at each of the four positions being examined. Thus, mRNAs synthesized from TAG-containing mutants, when coinjected into Xenopus oocytes with tRNA-Tyr molecules, produced functional receptors that responded to application of GABA with similar EC50 values and Hill coefficients to WT receptors. Thus, the wild type phenotype was successfully “rescued” by the delivery of tRNA-Tyr molecules. For each receptor, maximal GABA-induced currents (Imax) were typically 0.4–1 μA at a holding potential of −60 mV (see examples in Figure 3). Since conventional expression of wild type GABAC receptors typically gives 3–5 μA currents, this suggests that the efficiency of nonsense suppression is probably close to 10–20%, similar to the value estimated for this technique from suppression of nACh receptors.18
Figure 3.
(A) Typical responses to GABA application to mutant GABAC receptors expressed in oocytes. (B) Typical concentration response data from which the parameters in Tables 2 and 3 are determined.
The Role of the Loop A Phenylalanine Residue
The canonical aromatic box in Cys-loop receptors is formed from five residues on loops A, B, C (principal subunit), and D (complementary subunit) (Table 1). The alignment of loop A across the Cys-loop family is challenging (e.g., see ref (19)), but we considered Phe138 to align with the aromatic in loop A. The data indicate that an aromatic is important but not critical at this location. The change in EC50 when this is substituted with Ala (∼30-fold) is small compared to the same substitution in the Tyr residues that contribute to the aromatic box in loops B, C, and D (>100 fold EC50 increases; Table 2), indicating that aromaticity is less critical here. Nevertheless, we propose that an aromatic here may contribute to stabilization of the positively charged amine.
Table 2. Conventional Mutations of Other Aromatic Residues in or close to the Binding Pocketa.
| Y106 |
F138 |
Y167 |
Y200 |
Y208 |
W246 |
Y251 |
||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EC50 (μM) | nH | EC50 (μM) | nH | EC50 (μM) | nH | EC50 (μM) | nH | EC50 (μM) | nH | EC50 (μM) | nH | EC50 (μM) | nH | |
| F | 1.9 ± 0.4 | 1.5 ± 0.3 | 1.4 ± 0.2 | 1.6 ± 0.3 | 1.1 ± 0.2 | 1.1 ± 0.2 | 110 ± 34b | 1.2 ± 0.3 | 1.5 ± 0.1 | 1.7 ± 0.2 | 2.9 ± 0.6 | 1.4 ± 0.2 | 1.4 ± 0.3 | 1.6 ± 0.2 |
| A | 8.1 ± 1.2b | 1.4 ± 0.2 | 62 ± 12b | 1.1 ± 0.2 | 23 ± 2b | 1.4 ± 0.2 | NR | NR | 0.95 ± 0.09 | 1.1 ± 0.2 | 1.2 ± 0.3 | 1.7 ± 0.3 | ||
| S | 5.7 ± 0.9b | 1.7 ± 0.4 | 54 ± 4b | 1.7 ± 0.1 | 4.3 ± 0.5b | 1.5 ± 0.2 | NR | 2.0 ± 0.1 | 2.2 ± 0.3 | 1.4 ± 0.04 | 2.4 ± 0.1 | 3.9 ± 0.2b | 2.4 ± 0.2 | |
Data = mean ± SEM, n = 3–5.
Significantly different, p < 0.05.
The Role of the Loop B Tyrosine Residue
We have previously shown that Tyr198, the loop B residue of the aromatic box, makes a strong cation−π interaction to GABA.11 Consistent with this role for Tyr198, we found that substitution of Tyr198 with Phe or 4-MeO-Phe was reasonably well tolerated (<10 fold increase in EC50) while changing this residue to Ala or Ser was not, causing ∼1400- or ∼4000-fold increases in EC50, respectively (Table 3). Incorporation of Trp showed a ∼180 fold increase in EC50. Replacements of Tyr198 with 4-F-Phe, 3,5-F2-Phe, and 3,4,5-F3-Phe have already been reported and show substantial, incremental increases in EC50 with the addition of each F, demonstrating that this residue forms a strong cation−π interaction with GABA.11
Table 3. Mutations at Tyr102, Tyr198, Tyr241, and Tyr247a.
| Y102 |
Y198 |
Y241 |
Y247 |
|||||
|---|---|---|---|---|---|---|---|---|
| residues | EC50 (μM) | nH | EC50 (μM) | nH | EC50 (μM) | nH | EC50 (μM) | nH |
| Y(wild type) | 1.4 ± 0.2 | 1.6 ± 0.3 | 1.4 ± 0.2 | 1.6 ± 0.3 | 1.4 ± 0.2 | 1.6 ± 0.3 | 1.4 ± 0.2 | 1.6 ± 0.3 |
| F | 1.9 ± 0.6 | 1.1 ± 0.3 | 12 ± 1b | 1.6 ± 0.1 | 21 ± 1b | 1.3 ± 0.1 | 42 ± 4b | 1.5 ± 0.2 |
| A | 110 ± 20b | 1.2 ± 0.2 | 2000 ± 200b | 1.1 ± 0.1 | 4900 ± 480b | 1.1 ± 0.1 | NR | |
| S | 135 ± 25b | 1.3 ± 0.3 | 5600 ± 600b | 1.6 ± 0.2 | 1500 ± 96b | 1.2 ± 0.1 | NR | |
| W | 860 ± 60b | 1.4 ± 0.1 | 250 ± 30b | 1.1 ± 0.1 | 8.2 ± 1.4b | 1.3 ± 0.3 | 99 ± 8b | 1.3 ± 0.1 |
| 4-F-Phe | 3.1 ± 0.3 | 1.5 ± 0.2 | 34 ± 7c | 1.9 ± 0.3 | 1100 ± 370b | 0.6 ± 0.1b | 90 ± 3b | 1.9 ± 0.1 |
| 3,5-F2–Phe | 6.1 ± 0.9b | 1.6 ± 0.3 | 1700 ± 400c | 1.4 ± 0.2 | 490 ± 190b | 1.5 ± 0.3 | NR | |
| 3,4,5-F3-Phe | 3.4 ± 0.5 | 1.3 ± 0.2 | 8400 ± 1000c | 1.5 ± 0.1 | 950 ± 140b | 1.3 ± 0.3 | NR | |
| 4-Br-Phe | 1.6 ± 0.8 | 1.3 ± 0.4 | 620 ± 30b | 1.8 ± 0.2 | 2900 ± 170b | 1.1 ± 0.1 | ||
| 4-Me-Phe | 4.6 ± 2.1 | 1.4 ± 0.3 | 140 ± 100b | 1.4 ± 0.4 | NR | |||
| 4-MeO-Phe | 180 ± 20b | 1.4 ± 0.2 | 6.6 ± 2c | 1.6 ± 0.2 | 530 ± 50b | 1.7 ± 0.2 | NR | |
| 3-OH-Phe | 66 ± 9b | 1.7 ± 0.3 | 350 ± 70b | 1.5 ± 0.4 | NR | |||
Data = mean ± SEM, n ≥ 3.
Significantly different to wild type, p < 0.05.
Previously published (ref (11)).
The Role of the Loop C Tyrosine Residues
Tyr241 is equivalent to αTyr190 in Torpedo nACh receptors, and both an aromatic and a hydroxyl residue here are critical for efficient binding and gating.20 The data obtained from insertion of alternative natural amino acids at this position suggest both of these groups are also important in the GABAC receptor: Ala and Ser substitutions cause large increases in EC50 (∼3600- and ∼1100-fold) while Phe (∼20-fold) and Trp (∼8-fold) have less effect. Trp causes a smaller change to the EC50 than Phe, suggesting that the role of the hydroxyl can perhaps be partially fulfilled by Trp, although not by Br, F, Me, or MeO at the 4 position or hydroxyl at the 3 position. These data support the model, which suggests a role for this hydroxyl: a hydrogen bond with Arg104, a residue which has been previously shown to be critical for GABA binding in this receptor.21
Tyr247 was the most sensitive to substitution of the Tyr residues in the GABAC receptor binding pocket, indicating its importance. Our data show that aromaticity is critical: functional receptors were only obtained when it was replaced by Phe or Trp. The model suggests it may form a π–π interaction with Tyr241, which could be important for defining the structure of the C loop. In addition, the hydroxyl is important at this location: replacement with Phe caused a large increase (∼30-fold) in EC50. A main chain hydrogen bond with Ala 199, as predicted by the model, can explain these data. The increase in EC50 for Trp was ∼70-fold, suggesting that size is also important at this position, a hypothesis that is again supported by the model. This residue is conserved as an aromatic residue throughout the Cys-loop family, and studies on GABAA and 5-HT3 receptors have shown the equivalent residue is critical for binding and/or gating (indeed in MOD-1 receptors, the equivalent residue Trp226 forms a cation−π interaction with the agonist). We propose that its crucial role is due to its specific interaction with surrounding residues that control the location and structure of the C loop.
Tyr241 and Tyr247 are both on the C loop, and in these locations are referred to as C1 and C2 respectively. In the extensively investigated nAChRs, C1 is highly sensitive to substitution, but C2 is quite permissive. Just the opposite appears to be happening in the GABAC receptor, where a large number of substitutions at C2 give nonfunctional receptors, while many substituents are tolerated at C1. The C loop is the most variable segment of the binding pocket between Cys loop receptors, and these data further emphasize that this region is not only constituted of different residues and is of different lengths, but also that different sections play different roles in the different receptors.
The Role of the Loop D Tyrosine Residue
Tyr102 is contributed by the complementary subunit, and in the model of GABA docked into the GABAC binding pocket it is ∼4 Å from the primary amine, which would be close enough to provide some aromatic character to help stabilize this group. Our data, however, show no evidence for a cation−π interaction, although it is clear that a small aromatic residue here is strongly preferred: Phe, 4-F-Phe, 3,4,5-F3-Phe, 4-Me-Phe, and 4-Br-Phe can substitute effectively for Tyr, while Ala or Ser substitution result in ∼80- and ∼100-fold increases in EC50. Relocation of the hydroxyl group, as in 3-OH-Phe, however, is deleterious, as is an increase in size, such as with 4-MeO-Phe or Trp, which result in ∼130- or ∼630-fold increases in EC50, respectively. These data confirm the previous suggestion that aromaticity and size are important at this position,17 and these authors also suggested that the residue at this location should be rigid: substitution with Gly resulted in a ∼9000-fold increase in EC50. A subsequent study22 reported unusually high oocyte resting membrane conductance in Y102S mutant receptors, which could be blocked by picrotoxin, indicating spontaneously open channels. This residue is conserved as an aromatic across the Cys-loop family, indicating an important role, and mutagenesis in the GABAA receptor has demonstrated the homologous residue (α1Phe64) both forms part of the core of the ligand binding site, and is in a region (Asp162-Ser168) that appears to move during gating.23 Thus we conclude that an aromatic residue at position 102 is important both for binding and/or gating in GABAC receptors, but there is no direct contribution to a cation-π interaction.
The Role of Other Aromatic Residues Located in or Close to the Binding Site
Other aromatic residues which lie in or close to the binding pocket are Tyr106, Tyr167, Tyr200, Tyr208, Tyr251, and Trp246, (Figure 4). Tyr167 and Tyr208 are some distance from the binding pocket, but both were sensitive to substitution by Ala; EC50's were increased ∼20-fold (Tyr167) or resulted in nonfunctional receptors (Tyr208), suggesting a structural role. We propose this involves interactions with both the aromatic and the hydroxyl groups at both locations, as retaining one of these, that is replacement with Ser or Phe, caused only small or no changes in EC50. Tyr200 faces away from the ligand, yet was very sensitive to substitution: the only mutation that yielded functional receptors was Y200F, which resulted in an ∼100-fold increase in EC50, similar to data previously reported.17 Thus, Tyr200 may have an important role in binding or gating, although this may be secondary, such as ensuring Tyr198 is in the correct orientation. Alternatively, this residue may have a purely structural role, perhaps in receptor folding and/or for stabilizing the structure of the B loop, which is a rigid structure relative to which other regions of the binding pocket are proposed to move during receptor activation.24 Substituting Tyr106, and also Tyr251, with Ala or Phe resulted in <6 fold changes in EC50 compared to wild type receptors, indicating that these residues also do not play important roles in the function of the receptor; these residues are facing away from the binding pocket in the model, supporting this hypothesis.
Figure 4.
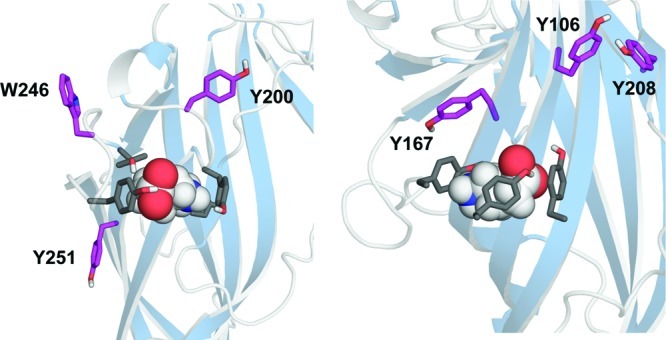
Location of aromatic residues tested in this study that do not directly contribute to the binding pocket on the principle (LHS) and complementary (RHS) faces. The data show that Tyr167, Tyr200, and Tyr208 are important for correct receptor function suggesting a role in the structure and/or gating of the receptor.
Conclusions
It has long been known that the binding site of all Cys-loop receptors is dominated by aromatic amino acids. In the nACh, 5-HT3, MOD-1, GABAA/C glycine, and GluCl receptors one of these amino acids has been shown to form a cation−π interaction with agonist,12−14,19,25 while two aromatics contribute to this interaction in an insect GABA receptor.16 Here we evaluate the roles of other aromatic residues in the GABAC receptor binding pocket. We find evidence for a range of interactions, but no other residue contributes to a cation−π interaction. Interesting parallels and divergences are evident when comparing the agonist binding site “aromatic box” across the superfamily of Cys-loop receptors.
Methods
Modeling and Ligand Docking
This was performed as previously described.9 Briefly the extracellular domain of the human GABA ρ1 subunit sequence was aligned with the sequence of a monomer of the AChBP using FUGUE,26 and 3D models created using MODELLER v 8.127 based on the crystal structure of AChBP (PDB: 1UV6). The models were analyzed using RAMPAGE (de Bakker and Lovell, http://raven.bioc.cam.ac.uk/rampage.php), and the best model was considered to be the one with fewest amino acids in unfavorable regions of the Ramachandran plot. A pentamer was generated by superimposing the modified monomer onto each monomer of the pentameric AChBP. The GABAC pentamer was then energy minimized in SYBYL using the AMBER force field.28 GABA was docked into the binding region of the GABAC receptor using GOLD v 3.0 (Cambridge Crystallographic Data Centre).
Site-Directed Mutagenesis
Mutagenesis reactions were performed using the method developed by Kunkel.29 For nonsense suppression, each of the tyrosine codons was replaced by TAG as previously described.11 A silent restriction site was incorporated in each to assist rapid identification.
Oocyte Preparation
Harvested stage V–VI Xenopus oocytes were washed in four changes of OR2 (82.5 mM NaCl, 2 mM KCl, 1 mM MgCl2, 5 mM HEPES, pH 7.5), defolliculated in 1 mg/mL collagenase for approximately 1 h, washed again in four changes of OR2 and transferred to 70% Leibovitz media (Gibco) buffered with 10 mM HEPES, pH 7.5. The following day they were injected with 5 ng of mRNA produced by in vitro transcription using the mMESSAGE mMACHINE kit (Ambion) from DNA subcloned into pGEMHE as previously described.9 Electrophysiological measurements were performed 24–72 h postinjection.
Synthesis of tRNA and dCA-Amino Acids
Unnatural amino acids were chemically synthesized as nitroveratryloxycarbonyl protected cyanomethyl esters and coupled to the dinucleotide dCA, which was then enzymatically ligated to 74-mer THG73 tRNACUA as detailed previously.13 Immediately prior to coinjection with mRNA, tRNA-aa was deprotected by photolysis. Typically 5 ng of mRNA and 25 ng of tRNA-aa were injected into Stage V–VI oocytes in a total volume of 50 nL. For control experiments, mRNA was injected (1) in the absence of tRNA and (2) with the THG73 74-mer tRNA. Experiments were performed 18–72 h postinjection.
Electrophysiological Recordings
Two-electrode voltage clamping of Xenopus oocytes was performed using standard electrophysiological procedures, using either a GeneClamp 500 amplifier or an OpusXpress system (Axon Instruments, Inc., Union City, CA). All experiments were performed at 22–25 °C. Glass microelectrodes were backfilled with 3 M KCl and had a resistance of approximately 1 MΩ. The holding potential was −60 mV. Oocytes were perfused with saline at a rate of 4 mL/min. Extracellular saline contained (mM), 96 NaCl, 2 KCl, 1.8 mM CaCl2, 1 MgCl2, and 5 mM HEPES, pH 7.4. Concentration–response curves and parameters were obtained using Prism software (GraphPad, Prism v 4.0, San Diego, CA).
Values are shown for a series of experiments and presented as the mean ± SEM. Statistical analysis was performed in Prism v4.03 using a one-way ANOVA or Student’s t test.
Glossary
Abbreviations
- AChBP
acetylcholine binding protein
- ACh
acetylcholine
- EC50
effective concentration that activates 50% of the receptors
- GABA
γ-aminobutyric acid
- HEPES
4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid
- LGIC
ligand-gated ion channel
- nACh
nicotinic acetylcholine
- 5-HT
5-hydroxytryptamine
Author Contributions
Participated in research design: S.C.R.L. and D.A.D. Conducted experiments: S.C.R.L., N.J.H., J.W., J.A.A., K.S.M., and D.L.B. Contributed new reagents or analytic tools: D.A.D. Performed data analysis: S.C.R.L., N.J.H., J.W., J.A.A., K.S.M., and D.L.B. Wrote or contributed to the writing of the manuscript: S.C.R.L. and D.A.D.
Funding for this work was received from The Wellcome Trust (S.C.R.L.: WT 81925; S.C.R.L. is a Wellcome Trust Senior Research Fellow in Basic Biomedical Science), the U.S. National Institutes of Health (D.A.D.: NS34407), the Medical Research Council (N.J.H. and K.S.M. held MRC studentships); the European Union (J.A.A.; S.C.R.L.: NeuroCypres FP7).
The authors declare no competing financial interest.
Funding Statement
National Institutes of Health, United States
References
- Brejc K.; van Dijk W. J.; Klaassen R. V.; Schuurmans M.; van Der Oost J.; Smit A. B.; Sixma T. K. (2001) Crystal structure of an ACh-binding protein reveals the ligand-binding domain of nicotinic receptors. Nature 411, 269–276. [DOI] [PubMed] [Google Scholar]
- Dellisanti C. D.; Yao Y.; Stroud J. C.; Wang Z. Z.; Chen L. (2007) Crystal structure of the extracellular domain of nAChR alpha1 bound to alpha-bungarotoxin at 1.94 A resolution. Nat. Neurosci. 10, 953–962. [DOI] [PubMed] [Google Scholar]
- Hilf R. J.; Dutzler R. (2008) X-ray structure of a prokaryotic pentameric ligand-gated ion channel. Nature 452, 375–379. [DOI] [PubMed] [Google Scholar]
- Hilf R. J.; Dutzler R. (2009) Structure of a potentially open state of a proton-activated pentameric ligand-gated ion channel. Nature 457, 115–118. [DOI] [PubMed] [Google Scholar]
- Bocquet N.; Nury H.; Baaden M.; Le Poupon C.; Changeux J. P.; Delarue M.; Corringer P. J. (2009) X-ray structure of a pentameric ligand-gated ion channel in an apparently open conformation. Nature 457, 111–114. [DOI] [PubMed] [Google Scholar]
- Hibbs R. E.; Gouaux E. (2011) Principles of activation and permeation in an anion-selective Cys-loop receptor. Nature 474, 54–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gallivan J. P.; Dougherty D. A.. (1999) Cation-π interactions in structural biology. Proc. Natl. Acad. Sci. U.S.A. 96, 9459–9464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cutting G. R.; Lu L.; O’Hara B. F.; Kasch L. M.; Montrose-Rafizadeh C.; Donovan D. M.; Shimada S.; Antonarakis S. E.; Guggino W. B.; Uhl G. R.; Kazazian, H. H. (1991) Cloning of the gamma-aminobutyric acid (GABA) rho1 cDNA: a GABA receptor subunit highly expressed in the retina. Proc. Natl. Acad. Sci. U.S.A. 88, 2673–2677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harrison N. J.; Lummis S. C. (2006) Molecular modeling of the GABAC receptor ligand-binding domain. J. Mol. Model. 12, 317–324. [DOI] [PubMed] [Google Scholar]
- Dougherty D. A. (2008) Cys-loop neuroreceptors: structure to the rescue?. Chem. Rev. 108, 1642–1653. [DOI] [PubMed] [Google Scholar]
- Lummis S. C.; Beene D. L.; Harrison N. J.; Lester H. A.; Dougherty D. A. (2005) A cation-π binding interaction with a tyrosine in the binding site of the GABAC receptor. Chem. Biol 12, 993–997. [DOI] [PubMed] [Google Scholar]
- Beene D. L.; Brandt G. S.; Zhong W.; Zacharias N. M.; Lester H. A.; Dougherty D. A. (2002) Cation-π interactions in ligand recognition by serotonergic (5-HT3A) and nicotinic acetylcholine receptors: the anomalous binding properties of nicotine. Biochemistry 41, 10262–10269. [DOI] [PubMed] [Google Scholar]
- Zhong W.; Gallivan J. P.; Zhang Y.; Li L.; Lester H. A.; Dougherty D. A. (1998) From ab initio quantum mechanics to molecular neurobiology: a cation-π binding site in the nicotinic receptor. Proc. Natl. Acad. Sci. U.S.A. 95, 12088–12093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mu T. W.; Lester H. A.; Dougherty D. A. (2003) Different binding orientations for the same agonist at homologous receptors: a lock and key or a simple wedge?. J. Am. Chem. Soc. 125, 6850–6851. [DOI] [PubMed] [Google Scholar]
- Diederich F. (2007) 40 years of supramolecular chemistry. Angew. Chem., Int. Ed. 46, 68–69. [DOI] [PubMed] [Google Scholar]
- Lummis S. C.; McGonigle I.; Ashby J. A.; Dougherty D. A. (2011) Two Amino Acid Residues Contribute to a Cation-π Binding Interaction in the Binding Site of an Insect GABA Receptor. J. Neurosci. 31, 12371–12376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amin J.; Weiss D. S. (1994) Homomeric rho 1 GABA channels: activation properties and domains. Recept. Channels 2, 227–236. [PubMed] [Google Scholar]
- Kearney P. C.; Nowak M. W.; Zhong W.; Silverman S. K.; Lester H. A.; Dougherty D. A. (1996) Dose-response relations for unnatural amino acids at the agonist binding site of the nicotinic acetylcholine receptor: tests with novel side chains and with several agonists. Mol. Pharmacol. 50, 1401–1412. [PubMed] [Google Scholar]
- Padgett C. L.; Hanek A. P.; Lester H. A.; Dougherty D. A.; Lummis S. C. (2007) Unnatural amino acid mutagenesis of the GABAA receptor binding site residues reveals a novel cation-π interaction between GABA and β2Tyr97. J. Neurosci. 27, 886–892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galzi J. L.; Bertrand D.; Devillers-Thiery A.; Revah F.; Bertrand S.; Changeux J. P. (1991) Functional significance of aromatic amino acids from three peptide loops of the alpha 7 neuronal nicotinic receptor site investigated by site-directed mutagenesis. FEBS Lett. 294, 198–202. [DOI] [PubMed] [Google Scholar]
- Melis C.; Lummis S. C.; Molteni C. (2008) Molecular dynamics simulations of GABA binding to the GABAC receptor: the role of Arg104. Biophys. J. 95, 4115–4123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Torres V. I.; Weiss D. S. (2002) Identification of a tyrosine in the agonist binding site of the homomeric rho1 gamma-aminobutyric acid (GABA) receptor that, when mutated, produces spontaneous opening. J. Biol. Chem. 277, 43741–43748. [DOI] [PubMed] [Google Scholar]
- Holden J. H.; Czajkowski C. (2002) Different residues in the GABAA receptor α1T60-α1K70 region mediate GABA and SR-95531 actions. J. Biol. Chem. 277, 18785–18792. [DOI] [PubMed] [Google Scholar]
- Thompson A. J.; Lochner M.; Lummis S. C. (2008) Loop B is a major structural component of the 5-HT3 receptor. Biophys. J. 95, 5728–5736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pless S. A.; Millen K. S.; Hanek A. P.; Lynch J. W.; Lester H. A.; Lummis S. C.; Dougherty. D. A. (2008) A cation-π interaction in the binding site of the glycine receptor is mediated by a phenylalanine residue. J. Neurosci. 28, 10937–10942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi J.; Blundell T. L.; Mizuguchi K. (2001) FUGUE: sequence-structure homology recognition using environment-specific substitution tables and structure-dependent gap penalties. J. Mol. Biol. 310, 243–257. [DOI] [PubMed] [Google Scholar]
- Sali A.; Blundell. T. L. (1993) Comparative protein modelling by satisfaction of spatial restraints. J. Mol. Biol. 234, 779–815. [DOI] [PubMed] [Google Scholar]
- Weiner S. J.; Kollman P. A.; Case D. A.; Singh U. C.; Ghio C.; Alagona G.; Profeta S.; Weiner P. (1984) A new force-field for molecular mechanical simulation of nucleic-acids and proteins. J. Am. Chem. Soc. 106, 765–784. [Google Scholar]
- Kunkel T. A. (1985) Rapid and efficient site-specific mutagenesis without phenotypic selection. Proc. Natl. Acad. Sci. U.S.A. 82, 488–492. [DOI] [PMC free article] [PubMed] [Google Scholar]




