Abstract
Eastern Africa has the world's highest cervical cancer incidence and mortality rates. We used epidemiologic data from Kenya, Mozambique, Tanzania, Uganda, and Zimbabwe to develop models of HPV-related infection and disease. For each country, we assessed HPV vaccination of girls before age 12 followed by screening with HPV DNA testing once, twice, or three times per lifetime (at ages 35, 40, 45). For women over age 30 we assessed only screening (with HPV DNA testing up to three times per lifetime or VIA at age 35). Assuming no waning immunity, mean reduction in lifetime cancer risk associated with vaccination ranged from 36-45%, and vaccination followed by screening once per lifetime at age 35 with HPV DNA testing ranged from 43-51%. For both younger and older women, the most effective screening strategy was HPV DNA testing three times per lifetime. Provided the cost per vaccinated girl was less than I$10 (I$2 per dose), vaccination had an incremental cost-effectiveness ratio (I$ [international dollars]/year of life saved [YLS]) less than the country-specific per capita GDP, a commonly cited heuristic for “very cost-effective” interventions. If the cost per vaccinated girl was between I$10 (I$2 per dose) and I$25 (I$5 per dose), vaccination followed by HPV DNA testing would save the most lives and would be considered good value for public health dollars. These results should be used to catalyze design and evaluation of HPV vaccine delivery and screening programs, and contribute to a dialogue on financing HPV vaccination in poor countries.
Keywords: human papillomavirus, cervical cancer, vaccination, screening, economic evaluation, Eastern Africa
INTRODUCTION
Cervical cancer, caused by infection with carcinogenic “high-risk” types of human papillomavirus (HPV), is a leading cause of cancer deaths among women in Africa.1 In Eastern Africa, which has the world's highest reported rates of cervical cancer incidence and mortality,1 HPV types 16 and 18 are associated with approximately 75% of cases.2
In countries with organized cervical cancer screening programs, incidence and mortality have decreased substantially.3 To date, screening in East African countries has been limited to demonstration projects or low levels of opportunistic screening in young women.4,i Barriers to secondary prevention in poor countries include lack of health delivery infrastructure, trained personnel, and equipment required for screening, diagnosis and treatment; limited health budgets; and competing healthcare priorities.5 Despite the difficulties of implementing and scaling up secondary prevention programs, economic evaluations and studies assessing test performance suggest that one- and two-visit screen-and-treat approaches could be feasible, beneficial, and cost-effective in resource-poor settings.6 A large randomized trial in India demonstrated that a single round of screening using HPV DNA testing in women over age 30 reduced advanced cervical cancer incidence and mortality in a developing country setting by 50%.7 A recently developed, lower-cost and less time-consuming HPV DNA test that is being piloted in several demonstration projects facilitates same-day testing and treatment, and may reduce costs and loss to follow-up in low-income countries.8,9
The potential for primary prevention has been realized with the availability of two HPV vaccines, both with high efficacy against infection with HPV 16/18, and recently prequalified by the World Health Organization.10,11,12 Challenges to vaccination of pre-adolescent girls include the high cost of the vaccines, the need for three doses at an age not routinely targeted for vaccination, and scant data on optimal delivery strategies for pre-adolescents.13
Acknowledging that country-specific data are limited, this analysis aims to synthesize available data to 1) inform policy makers and high-level decision makers of the potential value of alternative cervical cancer prevention strategies; and 2) explore the comparative performance of, and potential synergies between, primary and secondary prevention strategies. We estimate the reduction in lifetime risk of cervical cancer associated with HPV vaccination of pre-adolescent girls and screening of older women in five East African countries: Kenya, Mozambique, Tanzania, Uganda, and Zimbabwe. For selected countries, we assess the cost-effectiveness of prevention strategies, estimate the financial costs of pre-adolescent HPV vaccination, and explore alternative assumptions about vaccine efficacy, coverage, and duration of protection, as well as the impact of screening coverage, test performance, and loss to follow-up.
METHODS
Analytic overview
Using epidemiologic data for five East African countries, we adapted a previously described computer-based simulation model of cervical carcinogenesis14,15 to Kenya, Mozambique, Tanzania, Uganda, and Zimbabwe. Adopting a decision analytic approach, we estimated the health impact (e.g., reduction in lifetime cervical cancer risk) of: (1) HPV 16/18 vaccination of pre-adolescent girls; (2) screening of adult women over age 30 using HPV DNA testing or visual inspection with acetic acid (VIA); and (3) pre-adolescent vaccination followed by screening at older ages. Following standard guidelines for cost-effectiveness analysis,16,17,18 for four countriesii we estimated incremental cost-effectiveness ratios, defined as the additional cost of a particular strategy (per woman, in 2005 international dollars, I$) divided by its additional benefit (per woman life expectancy gain), compared to the next most costly strategy after eliminating strategies that are dominated (defined as more costly and less effective, or having higher incremental cost-effectiveness ratios than more effective options). We performed sensitivity analysis to evaluate the impact of uncertain parameters and assumptions.
Model
The model is described in previous publications.14,15,19 Briefly, the natural history of cervical carcinogenesis in an individual woman is represented as a sequence of monthly transitions between mutually-exclusive health states, including HPV infection status, grade of cervical intraepithelial neoplasia (CIN), and stage of cancer. Individual girls representative of a single birth cohort enter the model at age 9, prior to sexual debut, and are followed over their lifetimes. Transitions between health states depend upon HPV type, age, and history of prior type-specific infection (naturally-acquired immunity). HPV types are categorized hierarchically, with a woman classified according to her dominant type of infection: (1) HPV 16; (2) HPV 18; (3) other high-risk types (31, 33, 35, 39, 45, 51, 52, 56, 58, 59, and 68) and possibly high-risk types (26, 53, 66, 73, 82, and 82v); and (4) low-risk types.20 Strategies in the model can include age-specific vaccination, screening, or both, and the model differentiates the number of doses (in the case of vaccination) and test choice, frequency, and diagnostic protocol (in the case of screening). Women with cervical cancer can be detected via symptoms or screening, and are subject to stage-specific mortality rates in addition to all-cause age-specific mortality rates.
Epidemiologic Data and Calibration
Our model parameterization process has been described previously and is detailed in the Appendix.14,15,19
Briefly, we leveraged epidemiologic data (country-specific when possible) on age-specific prevalence of high-risk HPV in women with normal cytology, age-specific cervical cancer incidence, and prevalence of HPV 16/18 in cervical cancer. Data sources and summary statistics are listed in Table 1 and described further in the Appendix.
Table 1.
Selected model variables from the baseline comparative analysis, and summary calibration data and sources.*
| Model variable | Baseline value | ||||
|---|---|---|---|---|---|
| Characteristics of screening tests† | |||||
| HPV DNA testing | |||||
| Sensitivity | 90% | ||||
| Specificity | 77% | ||||
| Visual inspection with acetic acid | |||||
| Sensitivity | 41% | ||||
| Specificity | 82% | ||||
| Characteristics of screening program‡ | |||||
| Population coverage | 70% | ||||
| Loss to follow-up (per visit) | 15% | ||||
| Characteristics of vaccination program | |||||
| Population coverage of pre-adolescent girls with at least one dose | 70% | ||||
| Attrition rate (per dose)§ | 15% | ||||
| Efficacy against HPV 16/18 (1 dose, 2 doses, 3 doses) | 30%, 90%, 100% | ||||
| Duration of vaccine protection | Lifelong | ||||
| Kenya | Mozambique | Tanzania | Uganda | Zimbabwe | |
| Costs (2005 I$) | |||||
| Visual inspection with acetic acid∥ | 1.78 | 1.55 | 1.70 | 1.63 | NA |
| Woman's time and transport (1-visit strategy; 2-visit strategy)¶ | 7.67; 15.20 | 6.59; 13.04 | 7.50; 14.92 | 7.19; 14.23 | NA |
| HPV DNA test∥ | 10.68 | 9.99 | 10.43 | 10.22 | NA |
| Woman's time and transport (1-visit strategy; 2-visit strategy)¶ | 9.36; 15.20 | 8.27; 13.04 | 8.44; 14.92 | 8.99; 14.23 | NA |
| Cryosurgery∥ | 23.78 | 20.44 | 22.56 | 21.53 | NA |
| Summary of data used for model calibration | |||||
| Prevalence of high-risk HPV among women with normal cytology, % (95% CI)** | 25 (21-30) (N=369) | 23 (17-29) (N=195) | 23 (19-28) (N=381) | ††[23 (17-29)] [N=195] | ††[23 (17-29)] [N=195] |
| Data source: | De Vuyst 2003 | Castellsague 2001 | Mayaud 2001 | [Castellsague 2001] | [Castellsague 2001] |
| Prevalence of HPV 16 in cervical cancer, % (95% CI) | 43 (37-51)‡‡ (N=204) | 50 (42-58)§§ (N=302) | 41 (32-51)§§ (N=102) | 50 (43-58)§§ (N=157) | 61 (51-71) (N=98) |
| Prevalence of HPV 18 in cervical cancer, % (95% CI)∥∥ | 17 (12-23)‡‡ (N=204) | 25 (20-30)§§ (N=302) | 31 (23-41)§§ (N=102) | 26 (20-34)§§ (N=43) | 14 (8-23) (N=98) |
| Data source: | De Vuyst 2008 | Castellsague 2008; Naucler 2004 | Bosch 1995; ter Meulen 1992 | Bosch 1995; Odida 2008 | Stanczuk 2003 |
| Cancer incidence (crude rate per 100,000 women)** | 10.18 | 32.1 | 40.6 | 16.22 | 18.62 |
| Data source (registry): | Eldoret, 1998-2000 (Cancer in Africa) | Globocan 2008 | Globocan, 2002; Dar es Salaam 1990-1991; Kilimanjaro, 1998-2000 | Kyadondo County, 1998-2002 (CI5C) | Harare (African), 1998-2002 (CI5C) |
HPV = human papillomavirus; I$ = international dollars; CI = confidence interval; CI5C: Cancer incidence in five continents; NA = not available.
Sensitivity is defined as the probability of a positive test given the presence of cervical intraepithelial neoplasia grade 2 or higher. Specificity is defined as the probability of a negative test given the presence of no lesion. Because the prevalence of high-risk HPV (and thus the probability of testing positive on HPV DNA testing) varies slightly by country, HPV DNA test sensitivity and specificity vary slightly by country; we present HPV DNA test performance from Kenya here.
For screening strategies that relied on a single visit, we assumed that women who were screen positive and eligible for cryosurgery were treated in the same day; for those not eligible for cryosurgery (e.g., those with lesions covering more than 75% of the cervix or extending to the vaginal wall), we assumed referral to a secondary facility for further diagnostic testing and treatment. For two-visit screening strategies, we assumed women were screened during the first visit and returned for a second visit to obtain results (in the case of HPV DNA testing) and, if they screened positive and were eligible, receive cryosurgery. Loss to follow-up between visits, as well as additional time and transport costs for the second visit, distinguishes the two-visit strategies from the one-visit strategies.
Of the 70% who received at least one dose, 15% received one dose, 12.75% received two doses, and 72.25% received all three doses.
Cost estimate includes direct medical costs (disposable supplies, staff time, equipment, laboratory transport, staff, facilities). For cryotherapy, cost estimate includes direct medical costs for follow-up care and complications.
Cost estimate includes woman's time and transportation (roundtrip time and transportation to a primary health clinic, waiting time at the clinic, screening procedure time).
The model was calibrated to age-specific high-risk HPV prevalence and age-specific cancer incidence, but we report overall prevalence and crude incidence rates per 100,000 women here to summarize the data. Age-specific data are presented in the Appendix.
Country-specific data unavailable. Data from Mozambique (Castellsague 2001) were used as a proxy.
Data were drawn from a frequency-matched study of HIV-positive and HIV-negative women. Here we weighted the prevalence statistics for HPV 16 and 18 in each group by the prevalence of HIV in the study population prior to matching. Further details are available in the Appendix.
Prevalence and confidence intervals were derived using fixed or random effects model to pool data from more than one available in-country study (see Appendix for details).
Because our model categorized women hierarchically according to the dominant type of infection, we counted study subjects with both HPV 16 and 18 infections as having cancer attributable to HPV 16 only. Thus the prevalence of HPV 18 in cervical cancer presented here represents women without accompanying HPV 16 infections.
Based on data from the published literature, a plausible range was established for each natural history parameter, and uniform distributions were sampled jointly. Each round of sampling generated a different set of candidate values to input into the model. For each of the greater than 2 million input parameter sets, outcomes generated by model simulations were compared to the country-specific epidemiologic data. A composite goodness-of-fit score for each parameter set was computed based on the sum of the log-likelihoods of each model outcome. We selected the top 50 sets for each country to use as model inputs for our analysis. Graphs depicting model fit to each country's epidemiologic data may be found in the Appendix. To incorporate the effects of parameter uncertainty, we report results as a mean and range of outcomes. Incremental cost-effectiveness ratios are reported as the ratio of the mean costs divided by the mean effects of one strategy versus another across the top 50 parameter sets.21
Strategies
To compare the potential benefits of any cervical cancer intervention with other public health interventions evaluated under optimistic delivery assumptions,22 our baseline ‘comparative’ analysis assumed 70% of the target population received the first step of the intervention (first dose of vaccination or screening), with an attrition rate of 15% for each subsequent intervention step (Table 1).
We assumed vaccination occurred before age 12 (prior to sexual debut for most women). For girls receiving all three doses, we assumed the vaccine provided full lifelong protection against HPV 16/18, while two doses conferred 90% and one dose conferred 30% lifelong protection (Table 1). Because of uncertainty in achievable coverage and real-world clinical effectiveness of the vaccine, we varied coverage and per-dose attrition rates, overall effectiveness (a function of per-dose efficacy and the per-dose attrition rate), and duration of immunity in additional analyses.
Screening strategies primarily employed HPV DNA testing, and we varied screening frequency (once, twice, or three times in a lifetime, at ages 35, 40, and 45, respectively) and the number of required clinical visits for screening and treatment. Only one VIA strategy was considered — a single test at age 35 — due to concerns about low sensitivity for incident lesions and declining test performance in older women.23,24,25 For screening strategies that relied on a single visit, we assumed that women who were screen positive and eligible for cryosurgery were treated the same day; for those not eligible for cryosurgery, we assumed referral to a secondary facility for further diagnostic testing and treatment. For two-visit screening strategies, we assumed women were screened during the first visit and returned for a second visit to obtain results (in the case of HPV DNA testing) and, if they screened positive and were eligible, receive cryosurgery (Table 1; Appendix). We assessed the impact of varying screening coverage rates and loss to follow-up after each clinic visit. To allow decision makers to contextualize results based on likelihood of uptake and coverage in a specific population, we also varied coverage for vaccination and screening differentially.
Cost data
Selected costs are presented in Table 1, and complete documentation of cost assumptions is provided in the Appendix. Costs are presented in 2005 international dollars (I$), a currency that provides a means of translating and comparing costs among countries, taking into account differences in purchasing power. When country-specific data were unavailable, we adapted cost data from other countries using previously published methods.6,14,19,26 Because the price of the HPV vaccineiii and programmatic costs of delivering an adolescent vaccine in Eastern Africa are not known, we express vaccine costs as an approximate composite value referred to as the ‘cost per vaccinated girl’, which we varied from I$5 to I$200; this was categorized into vaccine costs, wastage, freight and supplies, administration, immunization support and programmatic costs. For example, a cost of I$10 per vaccinated girl approximated three doses of vaccine at I$2.00 each, with the remainder allocated to the other component costs. For screening strategies, direct medical costs (e.g., staff, supplies, equipment, specimen transport) and women's time and transportation costs were included.
RESULTS
Population-level health benefits
First we present results for our baseline analysis, in order to compare the potential value of HPV vaccination and cervical cancer screening, analyzed under optimistic assumptions (e.g., 70% coverage, lifelong immunity), to other public health interventions.22 Results are then presented for more conservative scenarios.
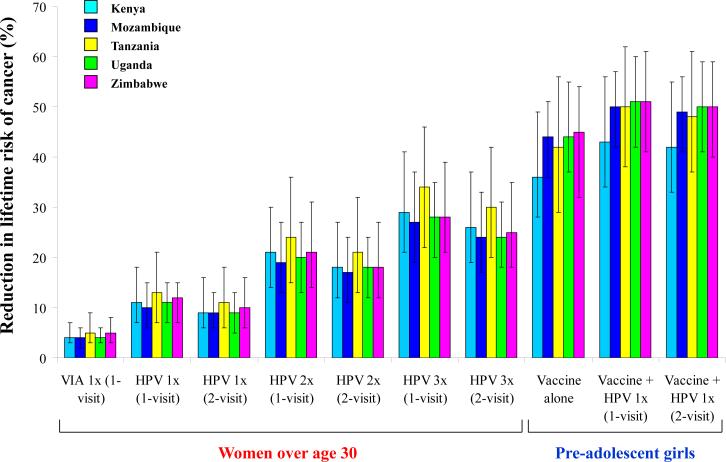
For pre-adolescent girls eligible for vaccination, the mean reduction in the lifetime risk of cancer with vaccination ranged from 36% (Kenya, range: 28-49%) to 45% (Zimbabwe, range: 32-54%) (Figure 1). The most effective strategy was a combined approach of adolescent vaccination followed by screening once per lifetime at age 35 using one-visit HPV DNA testing; this strategy was associated with a mean cancer reduction ranging from 43% in Kenya (range: 34-56%) to 51% in Uganda (range: 42-60%) and Zimbabwe (range: 41-61%). Results for additional strategies may be found in the Appendix.
Figure 1. Reduction in lifetime risk of cancer attributable to selected vaccination and screening strategies.
Colored vertical bars represent the mean reduction in lifetime risk of cancer (on the y-axis) for selected strategies (on the x-axis) in each country (light blue: Kenya; dark blue: Mozambique; yellow: Tanzania; green: Uganda; pink: Zimbabwe). Error bars represent the range of uncertainty in cancer reduction based on the 50 top-fitting parameter sets in each country. VIA = visual inspection with acetic acid; HPV = HPV DNA testing; 1x = screening once per lifetime at age 35; 2x = screening twice per lifetime at ages 35 and 40; 3x = screening three times per lifetime at ages 35, 40, and 45.
For women older than age 30, the most effective strategy was one-visit HPV DNA testing and the least effective once per lifetime screening strategy was VIA. Screening three times per lifetime with one-visit HPV DNA testing reduced cancer risk from 27% (Mozambique, range: 19-37%) to 34% (Tanzania, range: 22-46%).
Cost-effectiveness analysis
For pre-adolescent girls eligible for vaccination, results of analyses in which we vary the cost per vaccinated girl from I$5 (I$0.55 per dose) to I$200 (I$54.25 per dose) are shown in Table 2. Two independent analyses are shown — one assuming screening with one-visit HPV DNA testing, and one assuming screening with two-visit HPV DNA testing.
Table 2.
Cost-effectiveness results for pre-adolescent girls.*
| Kenya† | Mozambique† | Tanzania† | Uganda† | |
|---|---|---|---|---|
| I$/YLS | I$/YLS | I$/YLS | I$/YLS | |
| Adolescent vaccination followed by a single lifetime HPV DNA testing at age 35 (1-visit)‡ | ||||
| Cost per vaccinated girl: I$5 (I$0.55 per dose) | ||||
| Vaccine | 160 | 80 | CS | 20 |
| Screening with HPV, age 35 | § | § | § | § |
| Vaccine + screening with HPV, age 35 | 2090 | 1260 | 740 | 1000 |
| Cost per vaccinated girl: I$10 (I$2.00 per dose) | ||||
| Vaccine | 470 | 250 | 90 | 130 |
| Screening with HPV, age 35 | § | § | § | § |
| Vaccine + screening with HPV, age 35 | 2090 | 1260 | 740 | 1000 |
| Cost per vaccinated girl: I$25 (I$5.00 per dose) | ||||
| Screening with HPV, age 35 | 1400 | § | § | § |
| Vaccine | 1440 | 750 | 440 | 490 |
| Vaccine + screening with HPV, age 35 | 2090 | 1260 | 740 | 1000 |
| Cost per vaccinated girl: I$50 (I$12.25 per dose) | ||||
| Screening with HPV, age 35 | 1400 | 770 | 450 | 570 |
| Vaccine | § | § | § | § |
| Vaccine + screening with HPV, age 35 | 3580 | 1870 | 1260 | 1300 |
| Cost per vaccinated girl: I$200 (I$54.25 per dose) | ||||
| Screening with HPV, age 35 | 1400 | 770 | 450 | 570 |
| Vaccine | § | § | § | § |
| Vaccine + screening with HPV, age 35 | 15,000 | 7850 | 5610 | 5610 |
| Adolescent vaccination followed by a single lifetime HPV DNA testing at age 35 (2-visit)‡ | ||||
| Cost per vaccinated girl: I$5 (I$0.55 per dose) | ||||
| Vaccine | 160 | 80 | CS | 20 |
| Screening with HPV, age 35 | § | § | § | § |
| Vaccine + screening with HPV, age 35 | 2810 | 1690 | 1060 | 1370 |
| Cost per vaccinated girl: I$10 (I$2.00 per dose) | ||||
| Vaccine | 470 | 250 | 90 | 130 |
| Screening with HPV, age 35 | § | § | § | § |
| Vaccine + screening with HPV, age 35 | 2810 | 1690 | 1060 | 1370 |
| Cost per vaccinated girl: I$25 (I$5.00 per dose) | ||||
| Screening with HPV, age 35 | § | § | § | § |
| Vaccine | 1420 | 750 | 440 | 490 |
| Vaccine + screening with HPV, age 35 | 2810 | 1690 | 1060 | 1370 |
| Cost per vaccinated girl: I$50 (I$12.25 per dose) | ||||
| Screening with HPV, age 35 | 1860 | 1010 | 630 | 770 |
| Vaccine | § | § | § | 1240 |
| Vaccine + screening with HPV, age 35 | 3480 | 1830 | 1220 | 1370 |
| Cost per vaccinated girl: I$200 (I$54.25 per dose) | ||||
| Screening with HPV, age 35 | 1860 | 1010 | 630 | 770 |
| Vaccine | § | § | § | § |
| Vaccine + screening with HPV, age 35 | 14,570 | 7650 | 5420 | 5450 |
YLS = years of life saved; HPV = human papillomavirus DNA testing; CS = cost-saving. All currencies are reported in 2005 international dollars (I$).
Gross domestic product (GDP) per capita, 2005 I$ for each country is as follows: Kenya (I$1470); Mozambique (I$791); Tanzania (I$1167); Uganda (I$1077).28
Analyses assume either 1-visit HPV testing or 2-visit HPV testing. The results should be interpreted assuming that a country has already decided to utilize a 1-visit or 2-visit strategy. Analyses that rely on alternative assumptions are provided in the Appendix.
These strategies are either more costly and less effective, or have higher incremental cost-effectiveness ratios than more effective options, and are thus considered dominated.
Provided the cost per vaccinated girl was equal to, or below I$10 (I$2 per dose), vaccination was less than I$500 per year of life saved (YLS), and was more effective and had lower (more attractive) cost-effectiveness ratios than screening alone. For vaccine costs at or below I$25 per vaccinated girl (I$5 per dose), pre-adolescent vaccination followed by screening with one-visit HPV DNA testing at age 35 was associated with a cost per YLS ranging from I$740 (Tanzania) to I$2090 (Kenya). As the cost per vaccinated girl approached I$50 (I$12.25 per dose), vaccination alone was more costly and less cost-effective than screening alone, with the exception of two-visit HPV DNA testing in Uganda (I$1240 per YLS). At I$200 per vaccinated girl (I$54.25 per dose), adolescent vaccination followed by screening with one-visit HPV DNA testing at age 35 was associated with a cost per YLS ranging from I$5610 (Tanzania, Uganda) to I$15,000 (Kenya).
For women older than age 30, analyses shown in Table 3 assumed either one-visit VIA or HPV testing or two-visit VIA or HPV testing. We assumed countries would choose screening modality, frequency, and number of visits based on a number of factors — including existing pilot programs, available infrastructure and human resources, operational feasibility, and patient and cultural preferences — and thus we present scenarios based on alternative choices a country might make for reasons other than cost-effectiveness. Additional scenarios are presented in the Appendix.
Table 3.
Cost-effectiveness results for screening in women over age 30.*
| Kenya† | Mozambique† | Tanzania† | Uganda† | |
|---|---|---|---|---|
| $/YLS | $/YLS | $/YLS | $/YLS | |
| Screening with one-visit VIA once per lifetime‡ | ||||
| VIA at age 35 | 2010 | 1080 | 700 | 840 |
| Screening with either one-visit VIA or HPV DNA testing once per lifetime‡ | ||||
| VIA at age 35 | § | § | § | § |
| HPV at age 35 | 1400 | 770 | 450 | 570 |
| Screening with either two-visit VIA or HPV DNA testing once per lifetime‡ | ||||
| VIA at age 35 | § | § | § | § |
| HPV at age 35 | 1860 | 1010 | 630 | 770 |
| Screening with one-visit HPV DNA testing once or three times per lifetimeঠ| ||||
| HPV at age 35 | § | § | § | 570 |
| HPV at ages 35, 40, 45 | 1370 | 720 | 450 | 720 |
| Screening with two-visit HPV DNA testing once or three times per lifetimeঠ| ||||
| HPV at age 35 | § | § | § | 770 |
| HPV at ages 35, 40, 45 | 1770 | 920 | 610 | 930 |
YLS = years of life saved; VIA = visual inspection with acetic acid; HPV = human papillomavirus DNA testing.
Gross domestic product (GDP) per capita, 2005 I$ for each country is as follows: Kenya (I$1470); Mozambique (I$791); Tanzania (I$1167); Uganda (I$1077).28
Analyses assume either 1-visit VIA or HPV testing or 2-visit VIA or HPV testing. The results should be interpreted assuming that a country has already decided to utilize the strategy shown if a single strategy is presented; when two strategies are shown as options, this assumption is not made.
These strategies are either more costly and less effective, or have higher incremental cost-effectiveness ratios than more effective options, and are thus considered dominated.
HPV DNA testing three times per lifetime is compared to HPV DNA testing twice per lifetime, unless the strategy was dominated, in which case HPV DNA testing was compared to testing once per lifetime. Results for screening twice per lifetime are presented in the Appendix.
Provided that HPV DNA testing is available, HPV DNA testing strategies were more effective with lower cost-effectiveness ratios than VIA, ranging from I$450 (one-visit HPV testing once per lifetime, Tanzania) to I$1860 (two-visit HPV testing once per lifetime, Kenya) per YLS. When we assumed VIA was the only available option, screening once per lifetime yielded cost-effectiveness ratios ranging from I$700 (Tanzania) to I$2010 (Kenya) per YLS, reflecting both the low cost and low effectiveness of VIA.
In each of the four countries considered, screening three times per lifetime with one-visit HPV DNA testing was less than I$1400 per YLS, and with two-visit HPV DNA testing less than I$1800 per YLS. Additional results, including other screening ages and frequencies, are presented in the Appendix.
Additional analyses
Results from sensitivity analyses are shown below and in the Appendix using examples from Kenya and Uganda. The performance of vaccination depended upon vaccine efficacy, level of population coverage with at least one dose, attrition rate per dose, and duration of protection. As shown previously, the comparative performance of screening strategies depends on test performance, population coverage, and loss to follow-up.6,14,19
Varying efficacy, population coverage, and attrition rate per dose of the vaccine in Kenya
Figure 2 shows the reduction in the lifetime risk of cancer in Kenya as per-dose efficacy and coverage were varied. Overall effectiveness of the vaccine is a function of per-dose efficacy — for which we considered a scenario with the same protection as in the baseline analysis (one dose: 30%; two doses: 90%; three doses: 100% lifelong protection), as well as a more conservative scenario (one dose: 0%; two doses: 50%; three doses 100% lifelong protection) and a more optimistic scenario (any doses: 100% lifelong protection)— and the attrition rate following administration of each vaccine dose, which we varied from 0% to 40%. When 75% of girls received at least one dose of vaccine, the mean reduction in the lifetime risk of cancer was 22%, even with the more conservative vaccine protection scenario and an attrition rate of 40% for each subsequent dose. When 75% of girls received a vaccine conferring 100% protection with just one dose, cancer risk was reduced by 45%.
Figure 2. Impact of vaccine effectiveness and coverage level on reduction in lifetime risk of cancer in Kenya.
Colored vertical bars represent the mean reduction in cancer risk (on the y-axis) at different levels of overall vaccine effectiveness (60-100%, on the x-axis) for varying levels of vaccination coverage (i.e., proportion of the target population that receives at least one dose of vaccine) (blue, 25%; purple, 50%; pink, 75%). Error bars represent the range of uncertainty in cancer reduction based on the 50 top-fitting parameter sets. Overall vaccine effectiveness is a function of per-dose efficacy and the attrition rate following each dose (e.g., for an attrition rate of 40%: of the girls who received at least one dose, 40% received only the first dose, 24% received two doses, and 36% received three doses). For example, an overall vaccine effectiveness of approximately 60% would be realized with a vaccine conferring 100% efficacy with administration of 3 doses, 50% for 2 doses, and no benefit for 1 dose, and an attrition rate of 40% between each dose. The table at the top of the graph describes scenarios that yield costs, benefits, and reduction in cancer risk similar to each corresponding level of overall effectiveness shown.
Assuming an initial coverage rate with the first dose of vaccine of 75%, as overall vaccine effectiveness was varied from 60 to 100%, the corresponding cost-effectiveness ratios associated with vaccination were reduced by approximately 60% (Appendix).
Varying vaccination and screening coverage in Uganda
Figure 3 shows the reduction in the lifetime risk of cancer as screening and vaccination coverage were varied as part of a strategy that followed pre-adolescent vaccination with a single-lifetime screen with one-visit HPV DNA testing at age 35.
Figure 3. Impact of vaccination coverage and screening coverage on reduction in the lifetime risk of cancer and the incremental cost-effectiveness ratio of vaccination followed by one screening per lifetime with one-visit HPV DNA testing (comparator: vaccine alone; cost per vaccinated girl: I$10), Uganda.
Colored vertical bars represent mean reduction in lifetime risk of cancer (on the primary y-axis) at different levels of vaccination coverage (25-75%, on the x-axis) and screening coverage (green, 25%; blue, 50%; pink, 75%). Error bars represent the range of uncertainty in cancer reduction based on the 50 top-fitting parameter sets. Lines with colors corresponding to level of screening coverage represent the incremental cost-effectiveness ratio (on the secondary y-axis) at different levels of vaccination coverage. The dashed black line represents a threshold of 30% cancer reduction. ICER=incremental cost-effectiveness ratio; YLS = years of life saved.
Each level of vaccination coverage displayed represents a potential scenario in Uganda, depending on which proxy indicators for achievable HPV vaccination coverage prove to be most realistic. For example, a modeled vaccination coverage level of 25% resembles the coverage achieved with the hepatitis B vaccine in its first year of roll-out in Uganda (29%).27 Under this assumption, combined HPV vaccination and screening reduced cancer risk by 19% to 26%, depending on screening coverage, and cost less than I$700 per YLS. If girls’ likelihood of continuation to school grade 5 is a better indicator of achievable HPV vaccination coverage, coverage could be closer to 50%.28 Under this assumption, the strategy reduced cancer risk by 35 to 40% and cost less than I$850 per YLS. If HPV vaccination coverage were to exceed current childhood vaccination coverage with three doses each of DTP, Hepatitis B, and Hib vaccines (64-68%), a reasonable proxy for projections might be a modeled coverage level of 75%. Under this assumption, the strategy reduced cancer risk by 50 to 54% and cost less than I$1100 per YLS.
Varying the performance and cost of HPV DNA testing in women over age 30 in Kenya
As we varied HPV DNA test sensitivity to detect cervical intraepithelial neoplasia grade 2 or higher (CIN2+) from approximately 90% to 63%, the expected reduction in lifetime risk of cancer associated with a single lifetime screening (one-visit) fell from 11% to 8% and the cost-effectiveness ratio increased from I$1400 to I$1840 per YLS.
If the cost of HPV DNA testing was reduced from the baseline assumption of I$10.68 to I$5.34, the cost-effectiveness ratio for a single lifetime screening (one-visit) decreased from I$1400 to I$1120 per YLS. If instead the HPV DNA testing cost is doubled to I$21.36, the cost-effectiveness ratio for a single lifetime screening increases to I$1940 per YLS (Appendix).
We describe the dramatic impact on the population-level benefits and the cost-effectiveness of two-visit HPV DNA testing, as loss to follow-up associated with each clinical visit was varied, in the Appendix.
DISCUSSION
The vaccine-preventable burden of cervical cancer in Eastern Africa is a function of cervical cancer incidence, the proportion of disease attributable to HPV 16/18, long-term vaccine efficacy, and the ability to achieve widespread coverage in girls prior to sexual debut. In our analysis — intended to provide estimates of the potential value of vaccination and screening if implemented under optimistic assumptions — we found that HPV 16/18 vaccination at 70% coverage of girls between ages 9 and 12 is expected to reduce the lifetime risk of cancer by approximately 40%, even when considering attrition rates of 15% between doses. For girls vaccinated as pre-adolescents, subsequent screening with HPV DNA testing at least once per lifetime between ages 35 and 40 is expected to cut the lifetime risk of cancer nearly in half. For women over age 30 today, screening three times per lifetime with one-visit HPV DNA testing reduced cancer risk from 27% in Mozambique to 34% in Tanzania.
We may have over- or underestimated vaccine performance. Clinical benefits associated with the vaccine may be greater than predicted if there are herd immunity benefits to nonvaccinated individuals, or if the vaccine provides long-term cross-protection against high-risk HPV types other than HPV 16/18. Analysis of clinical trial data suggests that the quadrivalent HPV vaccine may provide some degree of cross-protection against HPV 31 and HPV 59, 29 while the bivalent vaccine appears to provide cross-protection against persistent infection at 6 months with HPV 31, 45, and 52.30 While HPV types 31, 45, 52, and 59 are detected in 2.5%, 6.3%, 1.2%, and 0.4%, respectively, of cancers in Eastern Africa,2 nearly 17% of cancers in the region are associated with multiple HPV types, and thus any improved efficacy due to cross-protection may not be fully additive.
The effectiveness of the vaccine may be lower than our baseline analysis suggests if vaccine-induced immunity is low due to malnutrition, severe anemia, or co-morbidities such as HIV. Effectiveness of vaccination programs may also be less than predicted if the attrition rate between doses is high and administration of less than three doses confers little protection against HPV16/18. At this time, published data on the efficacy of one- and two-dose regimens are limited. A bivalent vaccine trial in Costa Rica found comparable efficacy for one, two, or three doses against persistent HPV infection over a three-year period.31 Vaccination may be less effective if the vaccine wanes while women are still at high risk of new HPV infections. Because clinical studies of vaccine efficacy only extend to 5-7 years of follow-up,10,11 the actual duration of protection is uncertain.
There is no universal criterion that defines a threshold cost-effectiveness ratio, below which an intervention would be considered cost-effective. One heuristic has evolved from the Commission on Macroeconomics and Health, suggesting interventions with a cost-effectiveness ratio less than the Gross Domestic Product (GDP) per capita would be “very cost-effective” and less than three times the GDP per capita, “cost-effective”.32 Provided the cost per vaccinated girl was less than I$10 (I$2 per dose), vaccination had an incremental cost-effectiveness ratio less than the country-specific per capita GDP. If the cost per vaccinated girl was between I$10 (I$2 per dose) and I$25 (I$5 per dose), vaccination followed by screening at age 35 with one-visit HPV DNA testing would also be considered good value for public health dollars. These results are similar to those reported previously in a different analysis.33
The single most influential factor on the cost-effectiveness of vaccination is the cost of vaccinating adolescents with a three-dose vaccine. There is uncertainty both in the price of the vaccine for countries of different income levels and in the programmatic costs associated with an adolescent vaccine. By expressing a composite cost per vaccinated girl, we capture the potential cost-effectiveness of vaccination under a wide range of vaccine price and program cost scenarios. Should future studies indicate that one- or two-dose regimens are noninferior, the cost per vaccinated girl may be closer to the lower range we consider. The HPV vaccine will be competing for the same resources as other new vaccines, such as the Haemophilus influenzae type B (Hib) and hepatitis B vaccines, which have been introduced in 80% and 96% (respectively) of African countries, and vaccines against rotavirus and pneumococcal diseases, which are eligible for GAVI Alliance support (roll-out of pneumococcal vaccination has begun in Kenya).34 The estimated cost per disability-adjusted life year (DALY) averted for the Hib vaccine in Kenya was $38, and for the hepatitis B vaccine in The Gambia was $28.35,36 Estimated cost-effectiveness of rotavirus vaccination in Kenya and Malawi ranges from $75 to $227 per DALY averted when the vaccine course (including two doses and programmatic costs) cost $9.26 to $11.70.37,38 Given these comparative cost-effectiveness estimates, it will be difficult for the HPV vaccine to compete for dollars earmarked for existing programs or dollars considered for new programs if the cost per vaccinated girl exceeds I$10 (I$2 per dose). That being said, provided the price and cost of programmatic delivery can be lowered, the benefits are comparable to those of other new vaccines.33
While cost-effectiveness analysis provides information on value for money, this is not equivalent to affordability, or the financial impact of a cervical cancer prevention program on a payer's budget. The estimated financial costs of vaccine roll-out scenarios in four countries at an estimated cost of I$10 per vaccinated girl, adapted from Goldie (2008),33 are displayed in the Appendix. Both the cost-effectiveness profile and financial costs of rolling out a vaccine program will need to be favorable in order to implement a sustainable vaccination program.
The effectiveness of a screening program depends upon population coverage, test performance, and the ability to screen and treat in as few visits as possible. The impact of multiple screenings may be less than our analysis indicates if attendance at one screening correlates with attendance at subsequent sessions. Based on the most recently available data, VIA was less effective and cost-effective than the strategy appeared in older analyses.6 While data on the performance of both VIA and lower-cost HPV DNA testing, used in a single-visit screening strategy, are limited in Eastern Africa, a recent trial in South Africa found that conventional HPV DNA testing reduced CIN2+ over three years by 70-80%; reductions in the VIA arm were less evident.39 We used recent data from a Chinese study comparing VIA with conventional (Hybrid Capture 2 [HC2]; Qiagen, Gaithersburg, MD, USA) and lower-cost HPV DNA (careHPV; Qiagen) testing to inform test performance;8 HC2 and careHPV were found to have comparable sensitivity. In China, careHPV had a sensitivity of 81% on self-collected vaginal specimens (95% confidence interval: 72-91%), and we examined the impact of an HPV test with approximately 63% sensitivity to capture the effectiveness of this strategy if patient preferences and operational constraints necessitate self-sampling.
The cost of the careHPV test has not been established, and may be different than the values assumed in our baseline comparative analysis. Like vaccination, HPV testing costs strongly influence the cost-effectiveness of screening with the rapid test. If self-sampling is accepted and facilitates greater screening coverage at lower costs, the cost-effectiveness of HPV testing will become even more attractive. Some have advocated VIA as an alternative for very low-resource settings until HPV DNA testing becomes more economical,13,40 arguing that training health workers to visualize the transformation zone of the cervix will be an essential component of screen-and-treat strategies involving HPV DNA testing in the future.
We have previously discussed inherent limitations in any model-based decision analytic approach,14,19 but we reiterate key points here. In addition to model structure and parameter uncertainty, there are uncertainties with respect to the natural history of HPV (particularly in older women), the nature of type-specific immunity following natural infection, and the relationship between HIV and the course of HPV infection. We summarize limitations related to the availability and quality of country-specific data used for model calibration in the Appendix.
In the countries considered here, where adult HIV prevalence ranges from 5.4% (Uganda) to 15.3% (Zimbabwe), we are mindful of how current data limitations regarding the interaction between HPV and HIV may affect results. While several studies from developed countries report increased incidence of invasive cervical among those infected with HIV,41,42 cancer registries in African countries have not generally reflected increased incidence rates corresponding to time trends in the HIV epidemic.43 Our assumption of steady time trends for cervical cancer incidence over the lifetime of a cohort of pre-adolescent girls may over- or underestimate health benefits, depending on changes in HIV prevalence in women, the future availability of antiretroviral therapy, and the extent to which antiretroviral therapy leads to increased (due to greater life expectancy) or decreased (due to immune reconsititution) cervical cancer incidence. (We note, however, that for women with HIV whose lives are prolonged by antiretroviral therapy, cervical cancer is a preventable cause of mortality.) Adding to the uncertain dynamic between the two viruses, early data suggest a potential beneficial impact of HPV vaccination on acquisition of HIV.44,45,46 If these findings are valid and substantial, the benefits of the HPV vaccine in Eastern Africa will be greater.
We did not consider the impact of vaccination on other HPV-related diseases that are attributable to HPV 16/18 — including anal cancer, vulvar and vaginal cancer, and oropharyngeal and oral cancer — and thus may have underestimated potential benefits of the vaccine. We assumed there was no correlation between the probability a woman received the vaccine as a pre-adolescent girl and the probability she subsequently received screening as an adult. The validity of this assumption in a resource-poor setting is unclear. Additionally, country-specific cost data are lacking and many of our estimates were derived using indirect estimation techniques as summarized in the Appendix.
Given the above limitations, our estimates of the benefits and cost-effectiveness of cervical cancer prevention strategies should be interpreted in the context of our analytic purpose — namely, to provide initial insight to policy makers in Africa, financing alliances (e.g., GAVI), and other potential payers by leveraging the best available data. Country implementation will require a second series of decisions and corresponding new analyses that consider the likelihood of uptake and acceptability with country-contextualized strategies. Forthcoming evidence from vaccine demonstration projects in Uganda and Tanzania47,48 and implementation in Rwanda49 will provide valuable information regarding the effectiveness and costs of delivery strategies, as well as the role of specific communication and educational efforts. Following implementation, decisions regarding whether and how to monitor vaccine impact through investment in monitoring systems and cancer registries in target areas will also be necessary. Vaccination and screening are applied to different age groups, rely on different components of existing infrastructure, and require the mobilization of financial resources that are likely to come from different payers. Programs are synergistic in that vaccination prevents infection with HPV 16/18, while screening allows for treatment of precancerous lesions caused by any high-risk HPV type before progression to invasive cancer. Screening adult women once has been shown to decrease cancer incidence and mortality in a resource-poor setting in less than ten years.7,24 We will not see cancer reduction from a vaccination program for many years to come. Screening programs will reduce cancer risk among those who do not receive the vaccine, those who are infected with non-vaccine targeted HPV types, and those who may experience reduced vaccine efficacy (as a result of immunosuppression or missed doses), and will provide insurance at the population level, given the uncertainties surrounding the long-term vaccine performance. Furthermore, screening with HPV DNA tests may eventually enable surveillance of HPV infection (with HPV typing in a subset who are HPV positive), and thus assessment of vaccine impact, within a population.50 Finally, screening remains the only cervical cancer prevention for the millions of women in Eastern Africa over age 20, who are beyond the targeted age for vaccination.
In 2008, an estimated 53,000 women on the African continent died of cervical cancer. By 2030, this number will nearly double.1 Most of these women will not have access to curative treatment and will die from this preventable disease at an age when they are vital to social and economic stability. In societies already ravaged by HIV, the loss of these women will be felt acutely. Pre-adolescent vaccines to prevent infection with HPV 16/18 and a lower-cost HPV DNA test offer opportunity to prevent these deaths. Provided vaccine and screening test costs are low, these interventions are of great promise. Yet even those strategies we have identified as cost-effective will likely be unaffordable without assistance from the global community. We hope this analysis will catalyze the current dialogue about how to 1) expediently secure these necessary resources, 2) develop delivery programs and evaluate alternative implementation strategies for primary and secondary prevention, and 3) begin the discourse at the country level over preferences for prevention modality, prioritization relative to other health problems, timing of introduction, and mechanisms for evaluation.
Supplementary Material
ACKNOWLEDGEMENTS
This work was funded by the Bill & Melinda Gates Foundation (30505) and the National Cancer Institute (R01 CA93435). This work was presented at the 26th International Papillomavirus Conference, July 2010, Montreal, Quebec, Canada (P-806). P.E.C. has received donations of HPV tests from Qiagen and serves on a data safety and monitoring committee for Merck, the manufacturer of Gardasil, for which he receives compensation.
Abbreviations
- HPV
human papillomavirus
- YLS
year of life saved
- I$
international dollars
- VIA
visual inspection with acetic acid
- CIN
cervical intraepithelial neoplasia
- HIV
human immunodeficiency virus
Footnotes
In April 2011, the Rwandan government announced the launch of a comprehensive cervical cancer prevention program including screening with HPV DNA testing and HPV vaccination.
Because the Zimbabwe dollar was discontinued in 2009 due to severe hyperinflation, we do not report cost-effectiveness results for Zimbabwe. The reliability of international dollars to convey the purchasing power of local currency units within a country's borders is limited when the value of local currency changes dramatically within a short period.
In June 2011, Merck announced that it will offer its HPV vaccine to the GAVI Alliance at US$5 per dose (http://www.gavialliance.org/media_centre/press_releases/vaccine_prices.php).
REFERENCES
- 1.Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM. GLOBOCAN 2008, Cancer incidence and mortality worldwide: IARC CancerBase No. 10. International Agency for Research on Cancer; Lyon, France: 2010. [September 22, 2010]. Available online at http://globocan.iarc.fr. [Google Scholar]
- 2.Smith JS, Lindsay L, Hoots B, Keys J, Franceschi S, Winer R, Clifford GM. Human papillomavirus type distribution in invasive cervical cancer and high-grade cervical lesions: a meta-analysis update. Int J Cancer. 2007;121:621–32. doi: 10.1002/ijc.22527. [DOI] [PubMed] [Google Scholar]
- 3.World Health Organization (WHO)/Institute Català d'Oncologia (ICO) Information Centre on HPV and Cervical Cancer (HPV Information Centre) [September 22, 2010];Summary report on HPV and cervical cancer statistics in Africa. 2010 Available online at www.who.int/hpvcentre.
- 4.Chirenje ZM, Rusakaniko S, Kirumbi L, Ngwalle EW, Makuta-Tlebere P, Kaggwa S, Mpanju-Shumbusho W, Makoae L. Situation analysis for cervical cancer diagnosis and treatment in East, Central, and Southern African countries. Bull World Health Organ. 2001;79:127–32. [PMC free article] [PubMed] [Google Scholar]
- 5.Denny L, Quinn M, Sankaranarayanan R. Chapter 8: Screening for cervical cancer in developing countries. Vaccine. 2006;24:S71–7. doi: 10.1016/j.vaccine.2006.05.121. [DOI] [PubMed] [Google Scholar]
- 6.Goldie SJ, Gaffikin L, Goldhaber-Fiebert JD, Gordillo-Tobar A, Levin C, Mahé C, Wright TC, Alliance for Cervical Cancer Prevention Cost Working Group Cost-effectiveness of cervical cancer screening in five developing countries. N Engl J Med. 2005;353:2158–68. doi: 10.1056/NEJMsa044278. [DOI] [PubMed] [Google Scholar]
- 7.Sankaranarayanan R, Nene B, Shastri SS, Jayant K, Muwonge R, Budukh AM, Hingmire S, Malvi SG, Thorat R, Kothari A, Chinoy R, Kelkar R, et al. HPV screening for cervical cancer in rural India. N Engl J Med. 2009;360:1385–94. doi: 10.1056/NEJMoa0808516. [DOI] [PubMed] [Google Scholar]
- 8.Qiao YL, Sellors JW, Eder PS, Bao YP, Lim JM, Zhao FH, Weigl B, Zhang WH, Peck RB, Li L, Chen F, Pan QJ, et al. A new HPV-DNA test for cervical cancer screening in developing regions: a cross-sectional study of clinical accuracy in rural China. Lancet Oncol. 2008;9:929–36. doi: 10.1016/S1470-2045(08)70210-9. [DOI] [PubMed] [Google Scholar]
- 9.Qiagen [January 4, 2011];The careHPV test. Available at: http://www.qiagen.com/about/whoweare/qiagencares/the-carehpv-test.pdf.
- 10.De Carvalho N, Teixeira J, Roteli-Martins CM, Naud P, Zahaf T, Sanchez N, Schuind A. Sustained efficacy and immunogenicity of the HPV-16/18 AS04-adjuvanted vaccine up to 7.3 years in young adult women. Vaccine. 2010:6247–6255. doi: 10.1016/j.vaccine.2010.07.007. [DOI] [PubMed] [Google Scholar]
- 11.Villa LL, Costa RL, Petta CA, Andrade RP, Paavonen J, Iversen OE, Olsson SE, Høye J, Steinwall M, Riis-Johannessen G, Andersson-Ellstrom A, Elfgren K, et al. High sustained efficacy of a prophylactic quadrivalent human papillomavirus types 6/11/16/18 L1 virus-like particle vaccine through 5 years of follow-up. Br J Cancer. 2006;95(11):1459–66. doi: 10.1038/sj.bjc.6603469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.World Health Organization (WHO) Human papillomavirus (HPV) vaccine background paper. World Health Organization; Geneva: 2008. [Google Scholar]
- 13.Muñoz N, Franco EL, Herrero R, Andrus JK, de Quadros C, Goldie SJ, Bosch FX. Recommendations for cervical cancer prevention in Latin America and the Caribbean. Vaccine. 2008;26S:L96–107. doi: 10.1016/j.vaccine.2008.05.062. [DOI] [PubMed] [Google Scholar]
- 14.Goldie SJ, Kim JJ, Kobus K, Goldhaber-Fiebert JD, Salomon J, O'Shea MK, Bosch FX, de Sanjosé S, Franco EL. Cost-effectiveness of HPV 16, 18 vaccination in Brazil. Vaccine. 2007;25:6257–70. doi: 10.1016/j.vaccine.2007.05.058. [DOI] [PubMed] [Google Scholar]
- 15.Kim JJ, Kuntz KM, Stout NK, Mahmud S, Villa LL, Franco EL, Goldie SJ. Multiparameter calibration of a natural history model of cervical cancer. Am J Epi. 2007;166:137–50. doi: 10.1093/aje/kwm086. [DOI] [PubMed] [Google Scholar]
- 16.Disease Control Priorities Project (DCPP) [October 25, 2007]; Available at http://www.dcp2.org/main/Home.html.
- 17.Gold MR, Siegel JE, Russell LB, Weinstein MC, editors. Cost-effectiveness in health and medicine. Oxford University Press; New York, NY: 1996. [Google Scholar]
- 18.World Health Organization statistical information system: CHOICE (CHOosing Interventions that are Cost Effective) [October 2, 2007]; Available: http://www.who.int/choice/en/.
- 19.Kim JJ, Kobus KE, Diaz M, O'Shea M, Minh HV, Goldie SJ. Exploring the cost-effectiveness of HPV vaccination in Vietnam: Insights for evidence-based cervical cancer prevention policy. Vaccine. 2008;26:4015–24. doi: 10.1016/j.vaccine.2008.05.038. [DOI] [PubMed] [Google Scholar]
- 20.Bouvard V, Baan R, Straif K, Grosse Y, Secretan B, El Ghissassi F, Benbrahim-Tallaa L, Guha N, Freeman C, Galichet L, Cogliano V, et al. A review of human carcinogens – Part B: Biological agents. Lancet Oncol. 2009;19:321–22. doi: 10.1016/s1470-2045(09)70096-8. [DOI] [PubMed] [Google Scholar]
- 21.Stinnett AA, Paltiel DA. Estimating CE ratios under second-order uncertainty: the mean ratio versus the ratio of the means. Med Decis Making. 1997;17:483–9. doi: 10.1177/0272989X9701700414. [DOI] [PubMed] [Google Scholar]
- 22.Goldie SJ, Yazdanpanah Y, Losina E, Weinstein MC, Anglaret X, Walensky RP, Hsu HE, Kimmel A, Holmes C, Kaplan JE, Freedberg KA. Cost-effectiveness of HIV treatment in resource-poor settings: the case of Côte d'Ivoire. N Engl J Med. 2006;355:1141–53. doi: 10.1056/NEJMsa060247. [DOI] [PubMed] [Google Scholar]
- 23.Rodriguez AC, Morera LA, Bratti C, Herrero R, Cox JT, Morales J, Alfaro M, Hutchinson M, Castle PE, Hildesheim A, Schiffman M. Performance of direct visual inspection of the cerviz with acetic acid and magnification in a previously screened population. J Low Genit Tract Dis. 2004;8:132–8. doi: 10.1097/00128360-200404000-00009. [DOI] [PubMed] [Google Scholar]
- 24.Sankaranarayanan R, Esmy PO, Rajkumar R, Muwonge R, Swaminathan R, Shanthakumari S, Fayette JM, Cherian J. Effect of visual screening on cervical cancer incidence and mortality in Tamil Nadu, India: a cluster-randomised trial. Lancet. 2007;370:398–406. doi: 10.1016/S0140-6736(07)61195-7. [DOI] [PubMed] [Google Scholar]
- 25.Schneider DL, Burke L, Wright TC, Spitzer M, Chatterjee N, Wacholder S, Herrero R, Bratti MC, Greenberg MD, Hildesheim A, Sherman ME, Morales J, et al. Can cervicography be improved? An evaluation with arbitrated cervicography interpretations. Am J Obstet Gynecol. 2002;187:15–23. doi: 10.1067/mob.2002.122848. [DOI] [PubMed] [Google Scholar]
- 26.Goldhaber-Fiebert JD, Goldie SJ. Estimating the cost of cervical cancer screening in five developing countries. Cost Eff Resour Alloc. 2006;4:13. doi: 10.1186/1478-7547-4-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.World Health Organization Vaccine Preventable Diseases Monitoring System [February 2, 2010]; Available at http://www.who.int/immunization_monitoring/en/globalsummary/countryprofileselect.cfm.
- 28.World Bank World Development Indicators [January 24, 2010]; WDI Online. Available online at http://www.worldbank.org/.
- 29.Wheeler CM, Kjaer SK, Sigurdsson K, Iversen OE, Hernandez-Avila M, Perez G, Brown DR, Koutsky LA, Tay EH, Garcia P, Ault KA, Garland SM, et al. The impact of quadrivalent human papillomavirus (HPV; types 6, 11, 16, and 18) L1 virus-like particle vaccine on infection and disease due to oncogenic nonvaccine HPV types in sexually active women aged 16-26 years. JID. 2009;199:936–44. doi: 10.1086/597309. [DOI] [PubMed] [Google Scholar]
- 30.Paavonen J, Jenkins D, Bosch FX, Naud P, Saleron J, Wheeler CM, Chow S, Apter DL, Kitchener HC, Castellsague X, de Carvalho NS, Skinner SR, et al. Efficacy of a prophylactic adjuvanted bivalent L1 virus-like-particle vaccine against infection with human papillomavirus types 16 and 18 in young women: an interim analysis of a phase III double-blind, randomized controlled trial. Lancet. 2007;369:2161–70. doi: 10.1016/S0140-6736(07)60946-5. [DOI] [PubMed] [Google Scholar]
- 31.Kreimer A, Rodriguez AC, Hildesheim A, Herrero R, Porras C, Schiffman M, Gonzalez P, Salomon D, Jimenez S, Schiller J. Proof-of-principle: efficacy of fewer than 3-doses of a bivalent HPV16/18 vaccine against incident persistent HPV infection in Guanacaste, Costa Rica.. Presented at the 26th International Human Papillomavirus Conference; Montreal, Canada. July 8, 2010. [Google Scholar]
- 32.World Health Organization (WHO) Macroeconomics and health: investing in health for economic development: report of the Commission on Macroeconomics and Health. World Health Organization; Geneva: 2001. [Google Scholar]
- 33.Goldie SJ, O'Shea M, Campos NG, Diaz M, Sweet S, Kim SY. Health and economic outcomes of HPV 16, 18 vaccination in 72 GAVI-eligible countries. Vaccine. 2008;26:4080–93. doi: 10.1016/j.vaccine.2008.04.053. [DOI] [PubMed] [Google Scholar]
- 34.GAVI Alliance [February 14, 2011];What you can apply for. Available at: http://www.gavialliance.org/support/what/index.php.
- 35.Kim SY, Salomon JA, Goldie SJ. Economic evaluation of hepatitis B vaccination in low-income countries: using cost-effectiveness affordability curves. Bull WHO. 2007;85:833–42. doi: 10.2471/BLT.06.038893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Akumu AO, English M, Scout JAG, Griffiths UK. Economic evaluation of delivering Haemophilus influenzae type b vaccine in routine immunization services in Kenya. Bull WHO. 2007;85:511–8. doi: 10.2471/BLT.06.034686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Tate JE, Rheingans RD, O'Reilly CE, Obonyo B, Burton DC, Tornheim JA, Adazu K, Jaron P, Ochieng B, Kerin T, Calhoun L, Hamel M, et al. Rotavirus disease burden and impact and cost-effectiveness of a rotavirus vaccination program in Kenya. JID. 2009;200:S76–84. doi: 10.1086/605058. [DOI] [PubMed] [Google Scholar]
- 38.Berry SA, Johns B, Shih C, Berry AA, Walker DG. The cost-effectiveness of rotavirus vaccination in Malawi. JID. 2010;202(Suppl. 1):S108–15. doi: 10.1086/653578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kuhn L, Wang C, Tsaie W- Y, Wright TC, Denny L. Efficacy of human papillomavirus-based screen-and-treat for cervical cancer prevention among HIV-infected women. AIDS. 2010;24:2253–61. doi: 10.1097/QAD.0b013e32833e163e. [DOI] [PubMed] [Google Scholar]
- 40.Herrero R, Ferreccio C, Salmeron J, Almonte M, Sanchez GI, Lazcano-Ponce E, Jeronimo J. New approaches to cervical cancer screening in Latin America and the Caribbean. Vaccine. 2008;26S:L49–58. doi: 10.1016/j.vaccine.2008.05.025. [DOI] [PubMed] [Google Scholar]
- 41.Chaturvedi AK, Madeleine MM, Biggar RJ, Engels EA. Risk of human papillomavirus-associated cancers among persons with AIDS. JNCI. 2009;101:1120–30. doi: 10.1093/jnci/djp205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Clifford GM, Polesel J, Rickenbach M, Dal Maso L, Keiser O, Kofler A, Rapiti E, Levi F, Jundt G, Fisch T, Bordoni A, De Weck D, et al. Cancer risk in the Swiss HIV Cohort Study: associations with immunodeficiency, smoking, and highly active antiretroviral therapy. JNCI. 2005;97:407–9. doi: 10.1093/jnci/dji072. [DOI] [PubMed] [Google Scholar]
- 43.Parkin DM, Sitas F, Chirenje M, Stein L, Abratt R, Wabinga H. Part I: Cancer in indigenous Africans – burden, distribution, and trends. Lancet Oncol. 2008;9:683–92. doi: 10.1016/S1470-2045(08)70175-X. [DOI] [PubMed] [Google Scholar]
- 44.Averbach SH, Gravitt PE, Nowak RG, Celentano DD, Dunbar MS, Morrison CS, Grimes B, Padian NS. The association between cervical human papillomavirus infection and HIV acquisition among women in Zimbabwe. AIDS. 2010;24:1035–42. doi: 10.1097/qad.0b013e3283377973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Smith-McCune KK, Shiboski S, Chirenje SE, Magure T, Tuveson J, Ma Y, Da Costa M, Moscicki A, Palefsky JM, Makuniki-Mutasa R, Chipato T, van der Straten A, et al. Type-specific cervico-vaginal human papillomavirus infection increases risk of HIV acquisition independent of other sexually transmitted infections. PLoS One. 2010 Apr 8;5:e10094. doi: 10.1371/journal.pone.0010094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Veldhuijzen NJ, Vyankandondera J, van de Wijgert JH. HIV acquisition is associated with prior high-risk human papillomavirus infection among high-risk women in Rwanda. AIDS. 2010;24:2289–92. doi: 10.1097/QAD.0b013e32833cbb71. [DOI] [PubMed] [Google Scholar]
- 47.PATH and Child Health Development Centre (CHDC) Shaping a strategy to introduce HPV vaccines in Uganda: formative research results from the HPV vaccines: evidence for impact project. PATH; Seattle: 2009. [Google Scholar]
- 48.Human Papillomavirus (HPV) Vaccine Pilot Project NCT01173900.
- 49. [June 23, 2011];Rwanda launches comprehensive cervical cancer prevention program. Government of the Republic of Rwanda News Release. Available at: http://www.gov.rw/Rwanda-launches-Comprehensive-Cervical-Cancer-Prevention-Program.
- 50.Franco EL, Tsu V, Herrero R, Lazcano-Ponce E, Hildesheim A, Muñoz N, Murillo R, Sànchez GI, Andrus JK. Integration of human papillomavirus vaccination and cervical cancer screening in Latin America and the Caribbean. Vaccine. 2008;26S:L88–95. doi: 10.1016/j.vaccine.2008.05.026. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.