Abstract
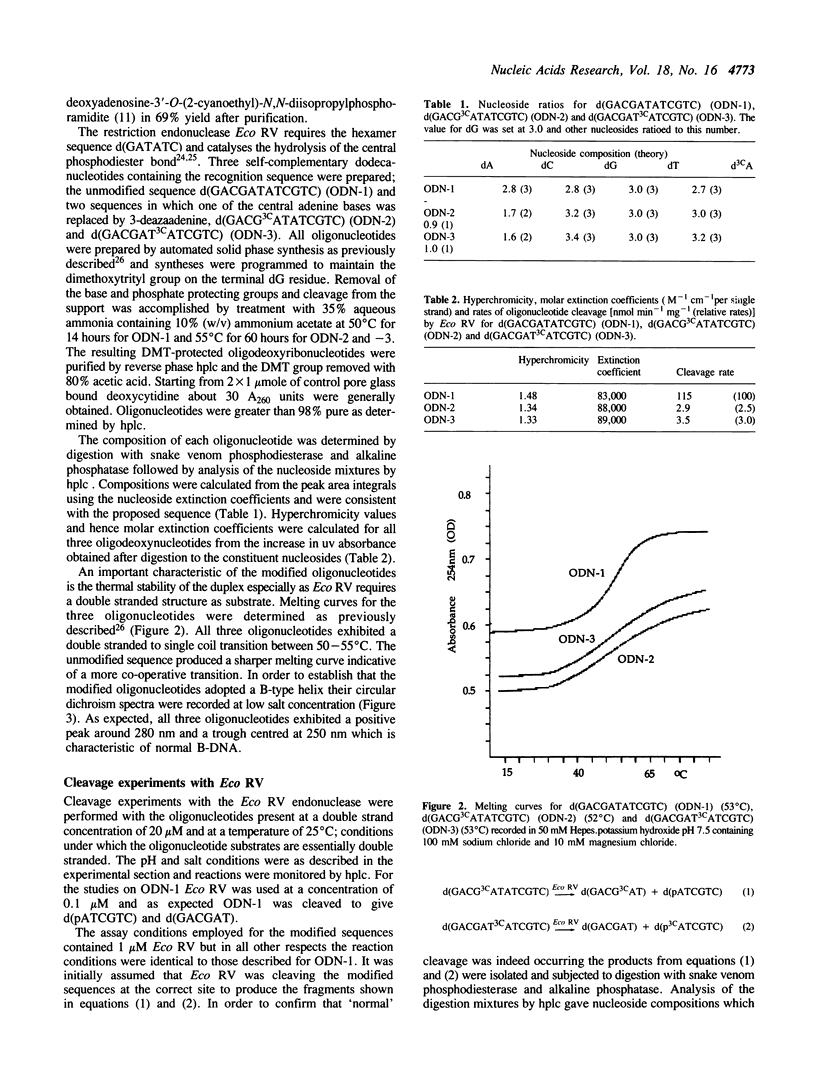
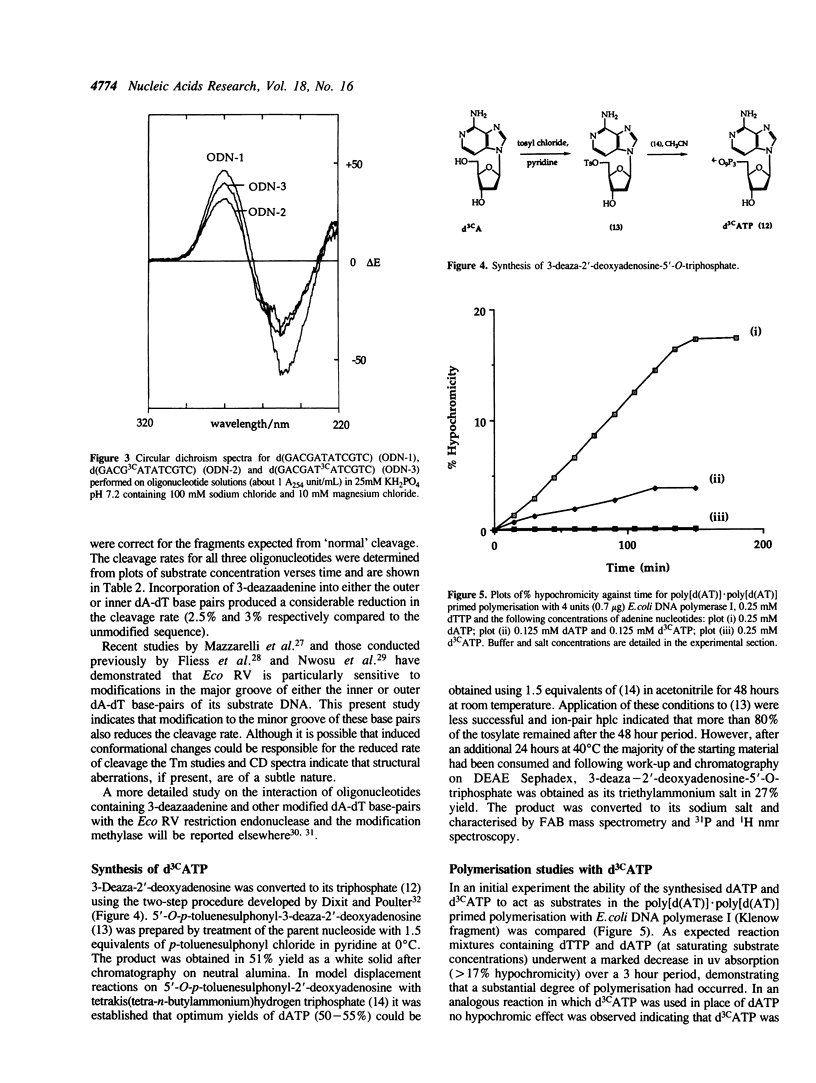
An improved procedure for the preparation of 3-deaza-2'-deoxyadenosine (d3CA) is described which is suitable for the synthesis of gram quantities of this analogue. Using phosphoramidite chemistry d3CA has been incorporated into the Eco RV restiction endonuclease recognition sequence (underlined) present in the self-complementary dodecamer d(GACGATATCGTC). The modified oligonucleotides have been thoroughly characterised by nucleoside composition analysis, circular dichroism and thermal melting studies. Studies with Eco RV show that incorporation of d3CA into either the central or outer dA-dT base-pair results in a substantial reduction in the rate of cleavage. The two-step conversion of d3CA to 3-deaza-2'-deoxyadenosine-5'-O-triphosphate (d3CATP) via the 5'-O-tosylate is also described. d3CATP is not a substrate in the poly[d(AT)].poly[d(AT)] primed polymerisation for either E. coli DNA polymerase I or Micrococcus luteus DNA polymerase. In a more detailed kinetic analysis d3CATP was shown to be a competitive inhibitor of E. coli DNA polymerase I with respect to dATP.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aggarwal A. K., Rodgers D. W., Drottar M., Ptashne M., Harrison S. C. Recognition of a DNA operator by the repressor of phage 434: a view at high resolution. Science. 1988 Nov 11;242(4880):899–907. doi: 10.1126/science.3187531. [DOI] [PubMed] [Google Scholar]
- Anderson J. E., Ptashne M., Harrison S. C. Structure of the repressor-operator complex of bacteriophage 434. 1987 Apr 30-May 6Nature. 326(6116):846–852. doi: 10.1038/326846a0. [DOI] [PubMed] [Google Scholar]
- Connolly B. A., Newman P. C. Synthesis and properties of oligonucleotides containing 4-thiothymidine, 5-methyl-2-pyrimidinone-1-beta-D(2'-deoxyriboside) and 2-thiothymidine. Nucleic Acids Res. 1989 Jul 11;17(13):4957–4974. doi: 10.1093/nar/17.13.4957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- D'Arcy A., Brown R. S., Zabeau M., van Resandt R. W., Winkler F. K. Purification and crystallization of the EcoRV restriction endonuclease. J Biol Chem. 1985 Feb 25;260(4):1987–1990. [PubMed] [Google Scholar]
- Dale R. M., Livingston D. C., Ward D. C. The synthesis and enzymatic polymerization of nucleotides containing mercury: potential tools for nucleic acid sequencing and structural analysis. Proc Natl Acad Sci U S A. 1973 Aug;70(8):2238–2242. doi: 10.1073/pnas.70.8.2238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Drew H. R., Dickerson R. E. Structure of a B-DNA dodecamer. III. Geometry of hydration. J Mol Biol. 1981 Sep 25;151(3):535–556. doi: 10.1016/0022-2836(81)90009-7. [DOI] [PubMed] [Google Scholar]
- Fliess A., Wolfes H., Rosenthal A., Schwellnus K., Blöcker H., Frank R., Pingoud A. Role of thymidine residues in DNA recognition by the EcoRI and EcoRV restriction endonucleases. Nucleic Acids Res. 1986 Apr 25;14(8):3463–3474. doi: 10.1093/nar/14.8.3463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jordan S. R., Pabo C. O. Structure of the lambda complex at 2.5 A resolution: details of the repressor-operator interactions. Science. 1988 Nov 11;242(4880):893–899. doi: 10.1126/science.3187530. [DOI] [PubMed] [Google Scholar]
- Kopka M. L., Yoon C., Goodsell D., Pjura P., Dickerson R. E. Binding of an antitumor drug to DNA, Netropsin and C-G-C-G-A-A-T-T-BrC-G-C-G. J Mol Biol. 1985 Jun 25;183(4):553–563. doi: 10.1016/0022-2836(85)90171-8. [DOI] [PubMed] [Google Scholar]
- Leslie A. G., Arnott S., Chandrasekaran R., Ratliff R. L. Polymorphism of DNA double helices. J Mol Biol. 1980 Oct 15;143(1):49–72. doi: 10.1016/0022-2836(80)90124-2. [DOI] [PubMed] [Google Scholar]
- May J. A., Townsend L. B. A general synthesis of 4-substituted 1-(beta-D-ribofuranosyl) imidazo-[4, 5-c] pyridines. J Chem Soc Perkin 1. 1975;(2):125–129. [PubMed] [Google Scholar]
- Mazzarelli J., Scholtissek S., McLaughlin L. W. Effects of functional group changes in the EcoRV recognition site on the cleavage reaction catalyzed by the endonuclease. Biochemistry. 1989 May 30;28(11):4616–4622. doi: 10.1021/bi00437a016. [DOI] [PubMed] [Google Scholar]
- McClure W. R., Jovin T. M. The steady state kinetic parameters and non-processivity of Escherichia coli deoxyribonucleic acid polymerase I. J Biol Chem. 1975 Jun 10;250(11):4073–4080. [PubMed] [Google Scholar]
- Mizuno Y., Kitano S., Nomura A. Nucleotides. VI. Syntheses and spectral properties of some deazaadenylyl-deazaadenosines (dinucleoside monophosphates with unusual CD-spectrum) and closely related dinucleoside monophosphates. Nucleic Acids Res. 1975 Dec;2(12):2193–2207. doi: 10.1093/nar/2.12.2193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mizusawa S., Nishimura S., Seela F. Improvement of the dideoxy chain termination method of DNA sequencing by use of deoxy-7-deazaguanosine triphosphate in place of dGTP. Nucleic Acids Res. 1986 Feb 11;14(3):1319–1324. doi: 10.1093/nar/14.3.1319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nwosu V. U., Connolly B. A., Halford S. E., Garnett J. The cloning, purification and characterization of the Eco RV modification methylase. Nucleic Acids Res. 1988 May 11;16(9):3705–3720. doi: 10.1093/nar/16.9.3705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ono A., Ueda T. Minor-groove-modified oligonucleotides: synthesis of decadeoxynucleotides containing hypoxanthine, N2-methylguanine and 3-deazaadenine, and their interactions with restriction endonucleases Bgl II, Sau, 3AI, and Mbo I (Nucleosides and Nucleotides Part 75). Nucleic Acids Res. 1987 Apr 10;15(7):3059–3072. doi: 10.1093/nar/15.7.3059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Otwinowski Z., Schevitz R. W., Zhang R. G., Lawson C. L., Joachimiak A., Marmorstein R. Q., Luisi B. F., Sigler P. B. Crystal structure of trp repressor/operator complex at atomic resolution. Nature. 1988 Sep 22;335(6188):321–329. doi: 10.1038/335321a0. [DOI] [PubMed] [Google Scholar]
- Pjura P. E., Grzeskowiak K., Dickerson R. E. Binding of Hoechst 33258 to the minor groove of B-DNA. J Mol Biol. 1987 Sep 20;197(2):257–271. doi: 10.1016/0022-2836(87)90123-9. [DOI] [PubMed] [Google Scholar]
- Prober J. M., Trainor G. L., Dam R. J., Hobbs F. W., Robertson C. W., Zagursky R. J., Cocuzza A. J., Jensen M. A., Baumeister K. A system for rapid DNA sequencing with fluorescent chain-terminating dideoxynucleotides. Science. 1987 Oct 16;238(4825):336–341. doi: 10.1126/science.2443975. [DOI] [PubMed] [Google Scholar]
- Schildkraut I., Banner C. D., Rhodes C. S., Parekh S. The cleavage site for the restriction endonuclease EcoRV is 5'-GAT/ATC-3'. Gene. 1984 Mar;27(3):327–329. doi: 10.1016/0378-1119(84)90078-7. [DOI] [PubMed] [Google Scholar]
- Sinha N. D., Biernat J., McManus J., Köster H. Polymer support oligonucleotide synthesis XVIII: use of beta-cyanoethyl-N,N-dialkylamino-/N-morpholino phosphoramidite of deoxynucleosides for the synthesis of DNA fragments simplifying deprotection and isolation of the final product. Nucleic Acids Res. 1984 Jun 11;12(11):4539–4557. doi: 10.1093/nar/12.11.4539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suck D., Lahm A., Oefner C. Structure refined to 2A of a nicked DNA octanucleotide complex with DNase I. Nature. 1988 Mar 31;332(6163):464–468. doi: 10.1038/332464a0. [DOI] [PubMed] [Google Scholar]
- Sági J. T., Szabolcs A., Szemzö A., Otvös L. Modified polynucleotides. I. Investigation of the enzymatic polymerization of 5-alkyl-dUTP-s. Nucleic Acids Res. 1977 Aug;4(8):2767–2777. doi: 10.1093/nar/4.8.2767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vorlícková M., Sági J., Szabolcs A., Szemzö A., Otvös L., Kypr J. Conformation of the synthetic DNA poly(amino2dA-dT) duplex in high-salt and aqueous alcohol solutions. Nucleic Acids Res. 1988 Jan 11;16(1):279–289. doi: 10.1093/nar/16.1.279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolberger C., Dong Y. C., Ptashne M., Harrison S. C. Structure of a phage 434 Cro/DNA complex. Nature. 1988 Oct 27;335(6193):789–795. doi: 10.1038/335789a0. [DOI] [PubMed] [Google Scholar]


