Abstract
The function of the brain depends on highly specific patterns of connections between populations of neurons. The establishment of these connections requires the targeting of axons and dendrites to defined zones or laminae, the recognition of individual target cells, the formation of synapses on particular regions of the dendritic tree, and the differentiation of pre- and postsynaptic specializations. Recent studies provide compelling evidence that transmembrane adhesion proteins of the immunoglobulin, cadherin, and leucine-rich repeat protein families, as well as secreted proteins such as semaphorins and FGFs, regulate distinct aspects of neuronal connectivity. These observations suggest that the coordinated actions of a number of molecular signals contribute to the specification and differentiation of synaptic connections in the developing brain.
Introduction
One of the remarkable features of the brain is the exquisite precision of synaptic connections that arises during development. The function of the brain is critically dependent on these highly specific patterns of connections between populations of neurons. The establishment of these connections requires the guidance of axons to correct target areas, their restriction to defined zones or laminae, the recognition of individual target cells, and the formation of synapses at particular regions on post-synaptic neurons. Much of our understanding of the molecular basis of synaptic specificity and synaptic target recognition comes from elegant experiments using the model organisms C. elegans, Drosophila, and Aplysia (Ackley and Jin, 2004; Colon-Ramos, 2009; Margeta et al., 2008; Mast et al., 2006; Rose and Chiba, 2000). These studies have led to the identification of individual genes involved in specificity and have laid an important conceptual framework for understanding connectivity. Whether similar mechanisms and molecules regulate synaptic specificity in the CNS of vertebrates is only beginning to be understood.
The establishment of synaptic specificity relies on the correct patterning of axons and dendrites. Here we discuss this process in the context of mechanisms that guide axons and dendrites to specific laminar, cellular, and subcellular structures. Many brain regions can be divided into anatomical laminae in which each lamina contains distinct populations of post-synaptic neurons. Laminar specificity develops when incoming axons are directed to specific laminae and form synapses only within these defined laminar borders (Fig. 1A). Cellular specificity occurs when different types of neurons, such as excitatory and inhibitory neurons, reside within a particular layer or brain region and different axons synapse specifically with one cell type over another (Fig. 1B). Subcellular specificity occurs when presynaptic axons synapse exclusively onto a precise domain of its target neuron (Fig. 1C). Although the molecular mechanisms of specificity can be discussed within the context of these three general processes, there is clearly overlap among them. For example, in many regions including cortex, retina, and hippocampus, dendrites from principal neurons extend through several different laminae and receive selective input at each level. From one perspective, this is an example of laminar specificity, but from another perspective, this is an example of subcellular specificity along the dendrite of the principal neuron.
Figure 1. Specificity of synaptic connections in the central nervous system.
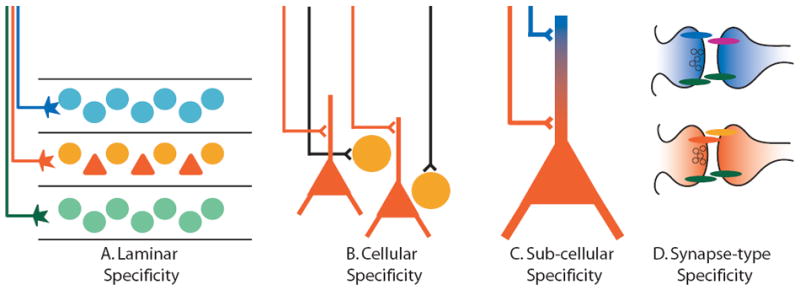
Diagramatic representation of specificity of neuronal connections at the laminar, cellular, subcellular, and synaptic levels. Precise regulation of connectivity at each of these levels contributes to the establishment of functional neural circuits.
The final step in the development of synaptic specificity involves the differentiation and maturation of distinct types of synapses. Once neurons find the correct synaptic partners, not all synapses are the same (Fig. 1D). Instead, synapses between different types of neurons have varying anatomical and functional properties. Recent work has identified several types of synaptogenic molecules that have the potential, through alternative splicing and interaction with multiple binding partners, to mediate the formation of distinct types of synapses.
Here we review our current understanding of the mechanisms mediating the development of synaptic specificity in vertebrates including mechanisms involving laminar, cellular, and subcellular target recognition and the subsequent differentiation of specific types of synapses. Mechanisms involved in synaptic targeting in invertebrates and in the olfactory system are not covered here as they have been recently reviewed elsewhere (Luo and Flanagan, 2007; Shen and Scheiffele, 2010).
Laminar Specificity
In many parts of the CNS, targeting afferents to specific laminae is a key feature of connectivity. This is particularly striking in the visual system, where there is evidence for laminar segregation at virtually every step of information processing from the retina to the visual cortex (Sanes and Zipursky, 2010). Within the retina, retinal ganglion cells (RGCs), the main output neurons of the retina, reside in the ganglion cell layer and extend their dendrites into the inner plexiform layer (IPL). The IPL can be further divided into several synaptic laminae and each lamina contains synapses between distinct populations of retinal interneurons and RGCs. This lamina-specific development of synapse formation is thought to be hardwired by restricted expression of extracellular molecular cues. Evidence in support for this hypothesis is increasing. First, development of lamina-specific targeting of one type of retinal interneuron, amacrine cells, has been observed in real time through time-lapse microscopy and found to occur correctly in the absence of neural activity (Godinho et al., 2005; Mumm et al., 2006; Nevin et al., 2008). These experiments rule out a major role of activity-dependent refinement in mediating laminar-specificity in the retina. Second, Sanes and colleagues analyzed cDNA libraries from subsets of RGC cells and used homology searches to identify several transmembrane molecules that direct laminar specificity (Yamagata and Sanes, 2008; Yamagata et al., 2002). Homophilic molecules of the Immunoglobulin Superfamily (IgSF), Dscam, DscamL, Sidekick-1, and Sidekick-2 are expressed in different combinations of IPL sub-laminae and their expression promotes layer-specific adhesion to corresponding populations of neurons that express the same IgSF molecule (Yamagata and Sanes, 2008; Yamagata et al., 2002). Despite this progress, the lamina-specific molecular code of the retina has not been fully deciphered. Not all interneurons express one of these four IgSF proteins and it is likely that other molecules and mechanisms contribute to lamina-specific connections in the IPL.
Just as RGC dendrites reside in a laminated structure, so do RGC axons. For example, in the chick, RGC axons project to 4 of 16 distinct laminae in the tectum known as the retino-recipient laminae. It is hypothesized that, in a mechanism similar to that in the IPL, differential expression of extracellular adhesion molecules in each retino-recipient layer directs RGC axons to their correct target layer. In support for this, co-cultures of chick optic tectum and retinal explants have been used to show that RGC axons target the correct retino-recipient laminae in vitro and this targeting is partially dependent on expression of the adhesion molecule N-cadherin since N-cadherin function blocking antibodies disrupt the laminar targeting of some RGCs (Inoue and Sanes, 1997; Yamagata and Sanes, 1995). More recently, several molecular markers have been identified that are expressed in target laminae and subsets of RGCs (Huberman et al., 2008; Huberman et al., 2009; Jiang et al., 2009; Yamagata et al., 1995; Yamagata et al., 2006). Now that more sophisticated markers and genetically encoded tools are available, this hypothesis can be more rigorously tested in both chick and mammalian systems.
Studies of thalamocortical (TC) projections also support the hypothesis that an extracellular molecular code mediates laminar-specificity. Thalamic axons of the visual system grow into the cortex and extend collaterals in layer 6, grow straight through layer 5, and terminate in layer 4 where they extensively branch and synapse with cortical neurons. What prevents TC axons from forming branches and synapses in layer 5 and what stops the axons in layer 4 so that they do not grow into layer 2/3? The TC projection between the lateral geniculate nucleus (LGN) and visual cortex (VC) can be recapitulated in vitro by co-culturing explants of LGN and VC adjacent to each other (Yamamoto et al., 1989). Axon labeling and electrophysiological recordings revealed that TC axons primarily grow into their appropriate laminar target, the layer 4 cortex, when thalamic tissue is placed at either the ventral, pial, or lateral side of a living or fixed cortical explant. (Bolz et al., 1992; Molnar and Blakemore, 1999; Yamamoto et al., 1997; Yamamoto et al., 1989). These experiments suggest that there are extracellular molecular programs within the cortex directing the laminar targeting of TC axons. It also suggests that there are both positive cues in the targeted layers and negative cues in the non-targeted layers that function together to accurately direct TC axon growth and synapse formation (Bolz et al., 1992; Molnar and Blakemore, 1999; Yamamoto et al., 1997; Yamamoto et al., 1989). Although the precise molecular mechanism that directs this laminar specificity of TC neurons remains unknown, N-Cadherin has been implicated in this process. When TC co-cultures are grown in the presence of N-Cadherin function blocking reagents, TC axons are no longer properly targeted to layer 4 (Poskanzer et al., 2003). Because of potential off-target effects of the blocking reagents it will be important to determine if genetic manipulations lead to similar defects. The cadherin family members, including protocadherins, have long been speculated to play an important role in mediating synaptic specificity. Cadherins are homophilic adhesion molecules with differential expression throughout the brain (Redies and Takeichi, 1996). Interestingly, despite extensive knowledge of cadherin structure and function in non-neuronal cells, the role of most cadherin family members in synaptic specificity, target recognition, and synapse formation in the CNS remains unknown.
Cellular Specificity
Once an axon reaches its final target or laminar region, it must form synapses with correct post-synaptic cells and not with incorrect cells. Different cell types within a target area may include excitatory and inhibitory neurons, local and projection neurons, and those that release different types of neurotransmitters. Selecting correct targets from a heterogeneous cell population is a challenging task since only a small fraction of axo-dendritic contacts correspond to appropriate synaptic partners. One excellent example of cellular specificity occurs in the hippocampus by the GABAergic neurons of the septohippocampal projection. These septal neurons project and synapse exclusively onto inhibitory neurons throughout the hippocampus while avoiding nearby excitatory neurons (Freund and Antal, 1988; Gulyas et al., 1991; Pascual et al., 2004). This projection leads to disinhibition of the principal hippocampal circuit and therefore may play an important role in hippocampal-dependent learning and memory, yet there is still little known about the mechanisms that mediate this highly specific connection.
A study from Lohmann and Bonhoeffer suggests that differences in intracellular signaling could contribute to cell type-specificity of synaptic connections. They compared intracellular changes in dendritic filopodia after contact with axons from excitatory versus inhibitory neurons in the CA3 region of the hippocampus and found that local dendritic calcium transients differ depending on the identity of the presynaptic axon (Lohmann and Bonhoeffer, 2008). Filopodia from labeled CA3 dendrites were imaged using time-lapse microscopy while simultaneously recording calcium signals in CA3 dendrites and nearby axons. Strong calcium transients occurred more frequently when CA3 filopodia contacted excitatory axons as compared to inhibitory axons (Lohmann and Bonhoeffer, 2008). The contacts with excitatory axons were also more likely to be stabilized, suggesting that there is a relationship between local calcium signals and target recognition. Since calcium is a well-known intracellular messenger, increasing calcium locally at sites of contact with a correct target cell may be important for initiating synapse formation at that location. It will be interesting to determine which contact-dependent molecules are required to induce these intracellular calcium changes and whether or not the calcium transients are required for subsequent synapse formation.
Studies in the spinal cord have also provided important insight into molecular mechanisms that contribute to cellular specificity. Spinal cord motor neurons cluster into spatially and genetically related groups of neurons called pools and each motor neuron pool sends and receives synaptic signals from a specific set of muscles and neurons (Jessell, 2000; Shirasaki and Pfaff, 2002). Recent experiments from Arber and colleagues suggest that genetic programs partially regulate synaptic specificity by regulating dendritic patterning (Vrieseling and Arber, 2006). Different classes of motor neurons extend dendrites in distinct spatial patterns within the spinal cord and this facilitates contact and synapse formation only with specific axons that grow near the dendrites and prevents synapse formation with axons that do not grow in the area. In mice lacking the transcription factor Pea3 the dendritic patterns of some types of motor neurons, including those of the cutaneous maximus (CM) motor pool, are significantly altered (Vrieseling and Arber, 2006) (Fig. 2A). In Pea3 -/- mice, the dendritic arbor of CM neurons extends into the central grey matter, a region that is normally devoid of CM dendrites in wildtype mice (Vrieseling and Arber, 2006). Axon growth is unaffected in Pea3 -/- mice and, therefore, tricep brachii (tri) afferents that normally grow into the central grey matter now incorrectly encounter dendrites from CM motor neurons (Fig. 2A). Consequently, tri afferents develop monosynaptic inputs to CM motor neurons in Pea3 -/- mice but never in wildtype mice (Vrieseling and Arber, 2006). Since Pea3 is a transcription factor, it likely regulates the expression of genes involved with axonal and dendritic patterning. In support of this, Pea3 expression is required for generating correct expression patterns of the recognition molecules, Cadherin-7 and Sema3E (Livet et al., 2002).
Figure 2. Cellular specificity in the spinal cord is disrupted in Pea3 and Sema3E knockout mice.
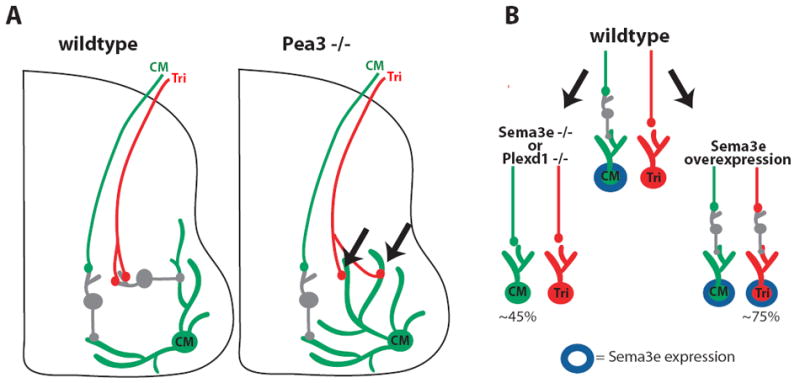
A) Cutaneous maximus (CM) and triceps brachii (Tri) afferents make polysynaptic connections to CM motor neurons in wildtype mice. However, in Pea3 -/- mice, CM dendrites grow toward the center of the spinal cord allowing Tri afferents to synapse directly onto CM dendrites and altering this motor circuit. B) Expression of Sema3E causes motor neurons to make polysynaptic connections with their propriceptive afferents.
Subsequently, a related study showed that Sema3E is expressed in a subset of motor neurons and expression of Sema3E is required to establish correct connectivity patterns (Pecho-Vrieseling et al., 2009) (Fig. 2B). In wildtype mice, CM motor neurons that express Sema3E develop polysynaptic connections with their propriceptive afferents and Tri motor neurons that do not express Sema3E develop monosynaptic connections with their propriceptive afferents. In mice lacking Sema3E or its receptor PlexD1, the monosynaptic Tri connections were unaltered but CM motor neurons converted from poly to monosynaptic (Pecho-Vrieseling et al., 2009). Conversely, in mice over-expressing Sema3E, Tri motor neurons became polysynaptic to their afferents while CM connections were unaltered (Pecho-Vrieseling et al., 2009) (Fig. 2B). These results suggest that Sema3E signaling prevents monosynaptic innervation and thus contributes to the development of synaptic specificity in the spinal cord. Furthermore, only axonal populations that express the Sema3E receptor plexinD1 are sensitive to this avoidance signal. Taken together, these studies suggest that intrinsic transcriptional programs can control synaptic specificity by regulating the expression of signals that mediate cell-cell interactions.
Subcellular Specificity
Proper function of neural circuits not only requires that synapses be made with the correct target cell but also at the correct subcellular location. Subcellular location is important since the relative strength and position of a synapse affects how much influence it has on the generation of action potentials. GABAergic inhibitory neurons show amazing specificity in targeting subcellular compartments. In fact, inhibitory neurons are often classified based on their affinity for a particular region of a target cell. For example, basket cells are so called because their axons elaborate and synapse on or near the soma of neurons, forming a basket-like structure around the target neuron. How do axons recognize the correct subcellular compartment to form a synapse? Similar to an axon’s ability to identify a particular cell type by an extracellular molecular code, it is thought that some cell adhesion molecules are preferentially localized to subcellular compartments and that these molecules can serve as a guide for axons and synapse formation.
In the cerebellum, specialized inhibitory basket neurons form synapses, called pinceau synapses, specifically with the axon initial segment (AIS) of Purkinje neurons. During development, basket axons travel first to Purkinje somata and then are guided to the AIS where they eventually develop synapses (Ango et al., 2004) (Fig. 3A). Because basket axons show clear directionality for the AIS, it is thought that surface molecules specific to this region guide the axons. Moreover, several proteins accumulate at the Purkinje AIS including the intracellular, membrane associated adaptor protein ankyrin-G and one of its binding partners, the L1 cell adhesion molecule neurofascin (Ango et al., 2004; Davis et al., 1996; Jenkins and Bennett, 2001; Zhou et al., 1998). In a series of experiments examining ankyrin-G knockout mice, Huang and colleagues found that ankyrin-G is required for basket axon targeting and pinceau synapse formation at the Purkinje AIS (Ango et al., 2004). In these mice, basket axons are no longer restricted to the AIS but instead localize to slightly more distal Purkinje axon segments (Ango et al., 2004) (Fig. 3A,B). Since ankyrin-G is required to restrict neurofascin to the AIS, it is possible that neurofascin is a key surface molecule required for communication with the basket axons. In support of this idea, expression of dominant negative neurofascin in Purkinje cells decreases pinceau synapse formation although it does not affect axon guidance to the AIS (Ango et al., 2004). Therefore, either enough endogenous neurofascin remains to direct the axons to the AIS or neurofascin may be specifically required for synapse formation or stabilization while other ankyrin-dependent molecules guide the basket axons.
Figure 3. Subcellular specificity of cerebellar Purkinje neurons is dependent on the cell adhesion molecules Neurofascin and CHL1.
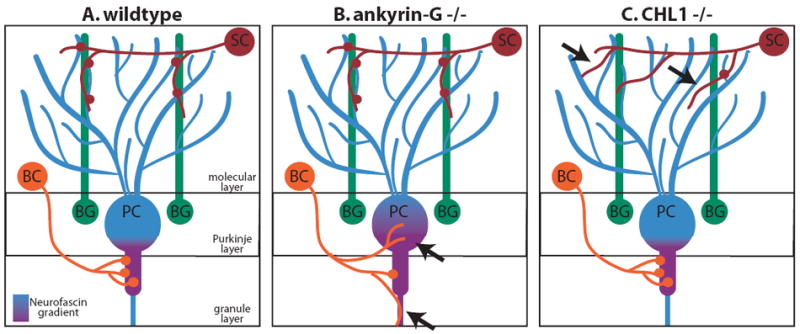
Schematics showing the wildtype (A) connectivity patterns for Purkinje neurons and those of ankyrin-G (B) and CHL1 (C) knockout mice. In Ankyrin-G -/- mice, a gradient of Neurofascin is no longer restricted to the axon initial segment (AIS) and, consequently, basket cell axons are not properly targeted to the AIS and synapse formation is decreased. In CHL1 -/- mice, stellate cell axons are not properly targeted along Bergman glia fibers and synapse formation is decreased. SC - stellate cell, BC – basket cell, PC – Purkinje cell, BG – Bergman glia.
Another example of subcellular targeting by interneuron axons is the innervation of Purkinje cell dendrites by stellate interneurons. In this case, proper targeting of stellate axons to Purkinje dendrites and synapse formation appears to be mediated by a scaffold of Bergman glial fibers (Fig. 3A). Bergman glial fibers grow among the Purkinje cell dendrites and express the IgSF family member CHL1 on their surface. In the absence of CHL1, stellate axons are misguided and fail to properly innervate Purkinje cell dendrites (Ango et al., 2008) (Fig. 3A,C). These studies on GABAergic innervation of Purkinje cells indicate that cell surface molecules regulate subcellular synaptic specificity both by mediating direct interaction between axon and dendrite as well as by providing an intermediate target to facilitate local connectivity.
Not all instances of subcellular specificity involve inhibitory connections. The segregation of different classes of excitatory inputs is particularly striking in the hippocampus, and recent studies suggest that two families of molecules, semaphorins and netrin-Gs, play important roles in regulating the subcellular specificity of excitatory synapses in hippocampal neurons.
In the CA3 region, the main bundle of dentate gyrus (DG) axons projects and synapses only with the most proximal dendrites of CA3 neurons. In mice lacking the semaphorin co-receptors Plexin-A2 or Plexin-A4, the mossy fibers grow and synapse in inappropriate synaptic zones. In Plexin-A2 mutant mice the mossy fibers grow in the pyramidal zone containing CA3 cell bodies and in Plexin-A4 mutant mice mossy fibers grow too far into distal layers of the CA3 (Suto et al., 2007). Interestingly, despite their hippocampal expression, neither Sema3F nor Sema6A knockout mice have any defects in the main mossy fiber projection (Sahay et al., 2003; Suto et al., 2007). Nonetheless, Sema6A repels mossy fibers in vitro and Sema6A interacts genetically with Plexin-A2 to rescue the mossy fiber defect in those mice. The authors propose that mossy fibers expressing Plexin-A4 are repelled from the distal CA3 layers by Sema6A and that Plexin-A2 in the proximal mossy fiber layer attenuates the local Sema6A repulsive signal to allow mossy fibers into to this zone (Suto et al., 2007).
In the dentate gyrus, DG neurons receive synaptic input from the lateral and medial perforant pathways onto non-overlapping dendritic zones. Netrin-Gs are glycosylphosphatidylinositol (GPI)-anchored proteins expressed on the surface of distinct axonal subsets (Nakashiba et al., 2000; Nakashiba et al., 2002; Yin et al., 2002). Netrin-G1 specifically binds the postsynaptic receptor NGL-1 while Netrin-G2 specifically binds the postsynaptic receptor NGL-2 (Kim et al., 2006; Lin et al., 2003; Nishimura-Akiyoshi et al., 2007). Intriguingly, this ligand/receptor specificity matches the segregated targeting of perforant path axons onto DG neurons. NGL1 protein localizes to distal dendritic compartments that recieve input from lateral perforant path axons expressing Netrin-G1. Conversely, NGL2 localizes to dendritic compartments that recieve input from medial perforant path axons expressing Netrin-G2 (Nishimura-Akiyoshi et al., 2007). Furthermore, loss of Netrin-G1 or Netrin-G2 disrupts the specific dendritic targeting of their respective ligands (Nishimura-Akiyoshi et al., 2007). Since Netrin-Gs and NGLs function during synaptogenesis (Kim et al., 2006; Woo et al., 2009), it will be interesting to investigate whether the specific localization of NGLs to different subcellular zones is required for the establishment of subcellular synaptic specificity.
Synapse Specific Differentiation
The development of synaptic specificity does not end once a neuron has identified its correct synaptic partner. The initial axo-dendritic contact is transformed into a functional synapse by the recruitment of pre- and post-synaptic components. Much of the early work in identifying synaptogenic molecules focused on general synapse formation and the recruitment of basic building blocks, such as synaptic vesicles, that are common to most synapses. However, synapses are extremely diverse and there are many different anatomically and functionally distinct types of synapses that form between different populations of neurons. The development of different classes of synapses may be facilitated by the specific axon guidance and target recognition mechanisms discussed above but there is evidence that other specificity molecules function primarily during synaptogenesis.
One of the best-characterized trans-synaptic adhesion complexes is the neurexin/neuroligin complex in which presynaptic neurexins bind postsynaptic neuroligins to bi-directionally organize synapses. Both molecules are well known for their remarkable ability to induce functional synaptic specializations when presented to neurons on the surface of non-neuronal cells or beads (Graf et al., 2004; Kang et al., 2008; Nam and Chen, 2005; Scheiffele et al., 2000). Almost all neurons express neurexins and neuroligins, which would suggest a general role in synapse formation rather than specificity. However, the three mammalian neurexin genes undergo extensive alternative splicing in their extracellular domain. This potentially generates thousands of different isoforms and has been hypothesized to play a role in mediating synaptic specificity (Rowen et al., 2002; Tabuchi and Sudhof, 2002; Ullrich et al., 1995; Ushkaryov et al., 1992).
Each neurexin gene can be transcribed from two alternative promoters to generate long α-neurexins or short β-neurexins and it has been suggested that α- and β-neurexins may differentially regulate glutamatergic versus GABAergic synapse formation. α-Neurexins expressed in non-neuronal cells selectively induce inhibitory postsynaptic differentiation (Chih et al., 2006; Kang et al., 2008). Consistent with this, α-neurexin triple knockout mice have fewer symmetric (presumptive inhibitory) synapses but normal numbers of asymmetric (presumptive excitatory) synapses, although functionally both inhibitory and excitatory synaptic transmission is reduced (Dudanova et al., 2007; Missler et al., 2003). In contrast to α-neurexins, β-neurexins expressed in non-neuronal cells induce both excitatory and inhibitory postsynaptic differentiation (Chih et al., 2006; Kang et al., 2008).
In addition to the use of alternative promoters, each neurexin gene has several other sites that undergo alternative splicing. One key site, known as splice site #4 (SS#4), has an important role in mediating the interaction between neurexins and its postsynaptic ligands. SS#4 is a 30 amino acid region in the extracellular domain of both α– and β-neurexins. Inclusion of SS#4 reduces the affinity of neurexin for neuroligin (Boucard et al., 2005; Comoletti et al., 2006; Graf et al., 2006). In addition to neuroligins, neurexins bind a structurally unrelated, leucine-rich repeat containing postsynaptic cell adhesion molecule called LRRTM2 (de Wit et al., 2009; Ko et al., 2009). Neurexin-LRRTM2 binding is also regulated by alternative splicing since only neurexins lacking the SS#4 insert bind LRRTM2 (Ko et al., 2009; Siddiqui et al., 2010) (Fig. 4). This differs from the binding of neuroligins to neurexins, which is modulated by SS#4 but not in an all-or-none fashion (Boucard et al., 2005; Comoletti et al., 2006). Furthermore, binding of LRRTMs or neuroligins to neurexin is mutually exclusive and suggests that these ligands compete for neurexin binding (Ko et al., 2009; Siddiqui et al., 2010).
Figure 4. Alternative splicing of neurexin regulates selectivity in neurexin-ligand interactions.
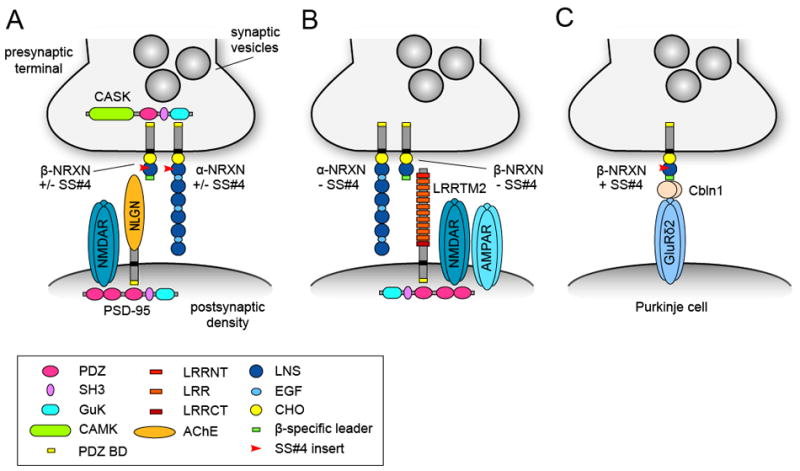
(A) Schematic drawing summarizing the trans-synaptic interaction between presynaptic α (long) and β (short) neurexins (NRXN) and its postsynaptic binding partner neuroligin (NLGN). Neurexins with or without a 30 amino acid insert at splice site #4 (SS#4) can bind neuroligins. Neurexins interact with the scaffolding molecule CASK and neuroligins interact with the scaffolding molecule PSD-95, which binds NMDAR receptors (NMDARs) via its PDZ domain. (B) Only neurexins lacking the SS#4 insert bind the postsynaptic adhesion molecule LRRTM2, which can recruit NMDARs and AMPARs. (C) SS#4 containing neurexins in cerebellar granule cells form a synapse-specific trans-synaptic adhesion complex with the secreted cerebellin precursor protein 1 (Cbln1) and the postsynaptic GluRδ2 receptor on Purkinje cell dendritic spines. GuK, guanylate kinase domain; CaMK, Ca2+/calmodulin-dependent kinase domain; LRR, leucine-rich repeat; LRRNT and LRRCT, N-terminal and C-terminal LRR flanking domains; PDZ BD, PDZ binding domain; AChE, acetylcholinesterase homology domain; LNS, laminin/neurexin/sex-hormone-binding protein domain; EGF, epidermal growth factor-like domain; CHO, carbohydrate attachment sequence.
Further evidence for neurexin SS#4 in regulating selective ligand binding comes from recent work showing that the secreted cerebellin1 precursor protein (Cbln1), which is essential for specific synapse formation between parallel fibers and Purkinje cells in the cerebellum, exclusively binds to β-neurexins containing the SS#4 insert (Uemura et al., 2010). Cbln1 is secreted from the presynaptic terminals of granule cells and binds the synaptogenic postsynaptic glutamate receptor GluRδ2 on Purkinje cell dendritic spines (Matsuda et al., 2010), forming a trans-synaptic complex consisting of SS#4-containing neurexins, Cbln1 and GluRδ2.
In addition to neuroligin, LRRTMs, and Cbln1/GluRδ2, splice forms of neurexin interact with other ligands, including dystroglycan, neurexophilin and the GABA(A) receptor (Petrenko et al., 1996; Sugita et al., 2001; Zhang et al., 2010). Given that neurexins functionally interact with so many distinct ligands, an interesting possibility is that different neurexin ligands organize different types of synapses. The localization and distribution of neurexin ligands is consistent with such a possibility. Neuroligin-1 localizes to excitatory synapses whereas neuroligin-2 localizes exclusively to inhibitory synapses (Song et al., 1999; Varoqueaux et al., 2004). Neuroligin-2 knockout mice have impairments in inhibitory synaptic transmission at specific subtypes of inhibitory synapses, suggesting a synapse-specific role for neurexin-neuroligin-2 interactions (Chubykin et al., 2007; Gibson et al., 2009; Poulopoulos et al., 2009). The LRRTM genes show striking differences in expression pattern in the cortex and hippocampus, and loss of LRRTM1 alters the distribution of presynaptic VGlut1 puncta in a lamina-specific manner (de Wit et al., 2009; Linhoff et al., 2009). Determining whether there is an interaction code between neurexin isoforms and specific neuroligin and LRRTM proteins should provide important insight into the potential role of neurexin isoforms in synaptic specificity.
The differential interaction of neurexins with various postsynaptic partners could also contribute to the diversity of functional properties of excitatory synapses. Neuroligins for instance bind the third PDZ domain of PSD-95, whereas NMDA receptors (NMDARs) and K+ channels bind the first and second PDZ domains of PSD-95, respectively (Irie et al., 1997). Therefore, by recruiting PSD-95, neuroligins indirectly recruit ion channels and neurotransmitter receptors to the synapse. Indeed, β-neurexin-induced clustering of neuroligins results in co-clustering of NMDARs but not AMPARs (Graf et al., 2004; Nam and Chen, 2005), and neuroligin-1 knockout mice have decreased NMDAR but not AMPAR-dependent synaptic transmission (Chubykin et al., 2007) (Fig. 4). LRRTM2 also interacts with PSD-95 through its C-terminal PDZ domain binding motif and binds NMDAR and AMPAR subunits in heterologous cells (de Wit et al., 2009; Linhoff et al., 2009). Artificial aggregation of transfected LRRTM2 on the dendritic surface induces co-clustering of the NMDA receptor subunit NR1 (Linhoff et al., 2009), suggesting that LRRTM2 can organize postsynaptic differentiation through recruitment of postsynaptic scaffolding proteins and neurotransmitter receptors (Fig. 4). Consistent with a role in regulating postsynaptic differentiation, lentiviral-mediated knockdown of LRRTM2 in hippocampal granule cells in vivo strongly reduces evoked AMPAR and NMDAR-mediated transmission compared to neighboring uninfected cells (de Wit et al., 2009). The differential effects of neuroligin and LRRTM2 on recruitment of glutamate receptors suggests that neurexin isoforms could influence subtype-specific synaptic properties by preferential interaction with specific neuroligin and LRRTM proteins.
Secreted factors are also important organizers of presynaptic differentiation. Recent work has revealed that FGF22 and FGF7 function as secreted organizers of excitatory and inhibitory presynaptic terminals, respectively (Terauchi et al., 2010). FGF22 and FGF7 are both expressed by CA3 pyramidal neurons but are differentially localized to excitatory (FGF22) or inhibitory synapses (FGF7) where they recruit neurotransmitter-specific synaptic vesicles to presynaptic terminals. Another class of secreted molecules that has been implicated in the development of synaptic specificity is the semaphorins. Mice lacking the secreted semaphorin3F (Sema3F) or its receptor neuropilin-2 show increased spine numbers in proximal but not distal dendrites of hippocampal granule cells and layer 5 pyramidal neurons (Tran et al., 2009). This suggests that Sema3F signaling through neuropilin-2 normally restricts spine formation from specific dendritic segments (Tran et al., 2009).
A number of other molecules including Eph/Ephrins, SALMs, Cadherins, and SynCAMs localize to synapses and can affect synapse formation and function (Arikkath and Reichardt, 2008; Biederer et al., 2002; Dalva et al., 2000; Kayser et al., 2006; Ko et al., 2006; Tai et al., 2008; Takeichi, 2007; Wang et al., 2006). Although direct evidence for a role for most of these molecules in synaptic specificity is still scarce, nearly all display differential expression patterns in the brain, which suggests that they may function in a synapse or cell-specific manner in vivo.
The mechanisms discussed here, including cell type-specific expression and alternative splicing, differential ligand binding, and pro- and anti-synaptogenic activity, are all likely to contribute to the specificity and identity of synaptic connections. The large number and variety of these intercellular signaling molecules suggests that each type of synapse is probably not specified by a unique ligand-receptor interaction. Instead, different types of synapses are likely regulated by overlapping but distinct combinations of membrane bound and secreted trans-synaptic signaling molecules that bias the association of pre- and post-synaptic elements and confer functional synaptic identity through differential recruitment of neurotransmitter vesicles, receptors, and modulators.
Conclusions and Future directions
Studies investigating synaptic specificity in the CNS strongly support a role for cell surface molecular interactions in regulating connectivity at all levels from laminar targeting to the development of distinct classes of synapses. Transmembrane molecules allow neurons to identify one another and then send intracellular signals pre- and post-synaptically to regulate the recruitment or dispersal of synaptic components. Recognition molecules are most likely used in combinations to ensure that the brain is wired with very high fidelity. Additionally, the link between synapse formation and disease is becoming clearer. Patients with many types of neurological and psychiatric disorders do not have obvious anatomical defects in the brain but instead appear to have defects in synapse formation and function. Several synaptic recognition molecules, including members of the neurexin, neuroligin and cadherin families, have been genetically linked to autism spectrum disorders (Jamain et al., 2003; Wang et al., 2009) and understanding how they direct synapse formation could eventually lead to more effective treatments.
While there has been significant progress in the identification of genes that contribute to synaptic specificity, in most cases our understanding of their role in the organization of neural circuits is still very limited. One of the major challenges in addressing molecular mechanisms of synaptic specificity in vertebrates is that it is technically difficult to determine the entire synaptic connectivity of a neuron. This problem is being addressed by new approaches to assess anatomical and functional connectivity. Rabies virus-mediated retrograde labeling methods, which can be used to identify neurons that form synapses onto a target cell, show promising results in slice culture and are now being explored in vivo (Wickersham et al., 2007). Another effort to describe connectivity at the electron microscopic level is being led by Denk and colleagues who have developed a serial block-face scanning electron microscope that can be used to do volume serial reconstructions (Denk and Horstmann, 2004). These advances should enhance our knowledge of connectivity patterns, which in turn will facilitate analysis of defects of connectivity in response to molecular perturbations.
Along with anatomical advances, new opto-genetic tools are facilitating functional analysis of connectivity at a level that is difficult to achieve with classical electrophysiological techniques. The applications of using channelrhodopsin to examine functional connectivity have been particularly impressive (Nagel et al., 2003; Petreanu et al., 2007). GFP fusions of channelrhodopsin can be delivered into the brain using viruses and labeled cells and axons can be stimulated with light to induce local depolarizations. Svoboda and colleagues have used this strategy to reveal previously unknown inputs that display subcellular specificity onto cortical neurons (Petreanu et al., 2009). Since then, several channelrhodopsin-related proteins have been developed that depolarize or activate neurons in response to different wavelengths of light and halorhodopsins that hyperpolarize or silence neurons in response to light (Gradinaru et al., 2010). Combining optogenetic approaches with selective manipulation of synaptogenic proteins will likely prove be a powerful approach for understanding the molecular basis of circuit organization.
Finally, the search for genes that regulate neuronal connectivity is being aided by large-scale expression analysis projects such as the GENSAT project, which has generated a large number of BAC reporter lines (www.gensat.org) and the Allen Brain Atlas project (www.brain-map.org), which has provided high-resolution genome-wide expression data for neural tissue at various developmental stages (Lein et al., 2007). This will greatly facilitate the identification of differentially expressed genes that may contribute to synaptic specificity. Functional analysis of these genes should be facilitated by the International Knockout Mouse Consortium, which aims to generate mutant embryonic stem cells for all protein encoding genes in the mouse (www.knockoutmouse.org). These parallel advances in technology and genetic information have and are likely to continue to accelerate the pace of discovery in this field.
Acknowledgments
This work was supported by NIH grants NS067216 and NS064124.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Ackley BD, Jin Y. Genetic analysis of synaptic target recognition and assembly. Trends Neurosci. 2004;27:540–547. doi: 10.1016/j.tins.2004.07.003. [DOI] [PubMed] [Google Scholar]
- Ango F, di Cristo G, Higashiyama H, Bennett V, Wu P, Huang ZJ. Ankyrin-based subcellular gradient of neurofascin, an immunoglobulin family protein, directs GABAergic innervation at purkinje axon initial segment. Cell. 2004;119:257–272. doi: 10.1016/j.cell.2004.10.004. [DOI] [PubMed] [Google Scholar]
- Ango F, Wu C, Van der Want JJ, Wu P, Schachner M, Huang ZJ. Bergmann glia and the recognition molecule CHL1 organize GABAergic axons and direct innervation of Purkinje cell dendrites. PLoS Biol. 2008;6:e103. doi: 10.1371/journal.pbio.0060103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arikkath J, Reichardt LF. Cadherins and catenins at synapses: roles in synaptogenesis and synaptic plasticity. Trends Neurosci. 2008;31:487–494. doi: 10.1016/j.tins.2008.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Biederer T, Sara Y, Mozhayeva M, Atasoy D, Liu X, Kavalali ET, Sudhof TC. SynCAM, a synaptic adhesion molecule that drives synapse assembly. Science. 2002;297:1525–1531. doi: 10.1126/science.1072356. [DOI] [PubMed] [Google Scholar]
- Bolz J, Novak N, Staiger V. Formation of specific afferent connections in organotypic slice cultures from rat visual cortex cocultured with lateral geniculate nucleus. J Neurosci. 1992;12:3054–3070. doi: 10.1523/JNEUROSCI.12-08-03054.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boucard AA, Chubykin AA, Comoletti D, Taylor P, Sudhof TC. A splice code for trans-synaptic cell adhesion mediated by binding of neuroligin 1 to alpha- and beta-neurexins. Neuron. 2005;48:229–236. doi: 10.1016/j.neuron.2005.08.026. [DOI] [PubMed] [Google Scholar]
- Chih B, Gollan L, Scheiffele P. Alternative splicing controls selective trans-synaptic interactions of the neuroligin-neurexin complex. Neuron. 2006;51:171–178. doi: 10.1016/j.neuron.2006.06.005. [DOI] [PubMed] [Google Scholar]
- Chubykin AA, Atasoy D, Etherton MR, Brose N, Kavalali ET, Gibson JR, Sudhof TC. Activity-dependent validation of excitatory versus inhibitory synapses by neuroligin-1 versus neuroligin-2. Neuron. 2007;54:919–931. doi: 10.1016/j.neuron.2007.05.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colon-Ramos DA. Synapse formation in developing neural circuits. Curr Top Dev Biol. 2009;87:53–79. doi: 10.1016/S0070-2153(09)01202-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Comoletti D, Flynn RE, Boucard AA, Demeler B, Schirf V, Shi J, Jennings LL, Newlin HR, Sudhof TC, Taylor P. Gene selection, alternative splicing, and post-translational processing regulate neuroligin selectivity for beta-neurexins. Biochemistry. 2006;45:12816–12827. doi: 10.1021/bi0614131. [DOI] [PubMed] [Google Scholar]
- Dalva MB, Takasu MA, Lin MZ, Shamah SM, Hu L, Gale NW, Greenberg ME. EphB receptors interact with NMDA receptors and regulate excitatory synapse formation. Cell. 2000;103:945–956. doi: 10.1016/s0092-8674(00)00197-5. [DOI] [PubMed] [Google Scholar]
- Davis JQ, Lambert S, Bennett V. Molecular composition of the node of Ranvier: identification of ankyrin-binding cell adhesion molecules neurofascin (mucin+/third FNIII domain-) and NrCAM at nodal axon segments. J Cell Biol. 1996;135:1355–1367. doi: 10.1083/jcb.135.5.1355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Wit J, Sylwestrak E, O’Sullivan ML, Otto S, Tiglio K, Savas JN, Yates JR, 3rd, Comoletti D, Taylor P, Ghosh A. LRRTM2 interacts with Neurexin1 and regulates excitatory synapse formation. Neuron. 2009;64:799–806. doi: 10.1016/j.neuron.2009.12.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Denk W, Horstmann H. Serial block-face scanning electron microscopy to reconstruct three-dimensional tissue nanostructure. PLoS Biol. 2004;2:e329. doi: 10.1371/journal.pbio.0020329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dudanova I, Tabuchi K, Rohlmann A, Sudhof TC, Missler M. Deletion of alpha-neurexins does not cause a major impairment of axonal pathfinding or synapse formation. J Comp Neurol. 2007;502:261–274. doi: 10.1002/cne.21305. [DOI] [PubMed] [Google Scholar]
- Freund TF, Antal M. GABA-containing neurons in the septum control inhibitory interneurons in the hippocampus. Nature. 1988;336:170–173. doi: 10.1038/336170a0. [DOI] [PubMed] [Google Scholar]
- Gibson JR, Huber KM, Sudhof TC. Neuroligin-2 deletion selectively decreases inhibitory synaptic transmission originating from fast-spiking but not from somatostatin-positive interneurons. J Neurosci. 2009;29:13883–13897. doi: 10.1523/JNEUROSCI.2457-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Godinho L, Mumm JS, Williams PR, Schroeter EH, Koerber A, Park SW, Leach SD, Wong RO. Targeting of amacrine cell neurites to appropriate synaptic laminae in the developing zebrafish retina. Development. 2005;132:5069–5079. doi: 10.1242/dev.02075. [DOI] [PubMed] [Google Scholar]
- Gradinaru V, Zhang F, Ramakrishnan C, Mattis J, Prakash R, Diester I, Goshen I, Thompson KR, Deisseroth K. Molecular and cellular approaches for diversifying and extending optogenetics. Cell. 2010;141:154–165. doi: 10.1016/j.cell.2010.02.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Graf ER, Kang Y, Hauner AM, Craig AM. Structure function and splice site analysis of the synaptogenic activity of the neurexin-1 beta LNS domain. J Neurosci. 2006;26:4256–4265. doi: 10.1523/JNEUROSCI.1253-05.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Graf ER, Zhang X, Jin SX, Linhoff MW, Craig AM. Neurexins induce differentiation of GABA and glutamate postsynaptic specializations via neuroligins. Cell. 2004;119:1013–1026. doi: 10.1016/j.cell.2004.11.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gulyas AI, Seress L, Toth K, Acsady L, Antal M, Freund TF. Septal GABAergic neurons innervate inhibitory interneurons in the hippocampus of the macaque monkey. Neuroscience. 1991;41:381–390. doi: 10.1016/0306-4522(91)90334-k. [DOI] [PubMed] [Google Scholar]
- Huberman AD, Manu M, Koch SM, Susman MW, Lutz AB, Ullian EM, Baccus SA, Barres BA. Architecture and activity-mediated refinement of axonal projections from a mosaic of genetically identified retinal ganglion cells. Neuron. 2008;59:425–438. doi: 10.1016/j.neuron.2008.07.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huberman AD, Wei W, Elstrott J, Stafford BK, Feller MB, Barres BA. Genetic identification of an On-Off direction-selective retinal ganglion cell subtype reveals a layer-specific subcortical map of posterior motion. Neuron. 2009;62:327–334. doi: 10.1016/j.neuron.2009.04.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inoue A, Sanes JR. Lamina-specific connectivity in the brain: regulation by N-cadherin, neurotrophins, and glycoconjugates. Science. 1997;276:1428–1431. doi: 10.1126/science.276.5317.1428. [DOI] [PubMed] [Google Scholar]
- Irie M, Hata Y, Takeuchi M, Ichtchenko K, Toyoda A, Hirao K, Takai Y, Rosahl TW, Sudhof TC. Binding of neuroligins to PSD-95. Science. 1997;277:1511–1515. doi: 10.1126/science.277.5331.1511. [DOI] [PubMed] [Google Scholar]
- Jamain S, Quach H, Betancur C, Rastam M, Colineaux C, Gillberg IC, Soderstrom H, Giros B, Leboyer M, Gillberg C, Bourgeron T. Mutations of the X-linked genes encoding neuroligins NLGN3 and NLGN4 are associated with autism. Nat Genet. 2003;34:27–29. doi: 10.1038/ng1136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jenkins SM, Bennett V. Ankyrin-G coordinates assembly of the spectrin-based membrane skeleton, voltage-gated sodium channels, and L1 CAMs at Purkinje neuron initial segments. J Cell Biol. 2001;155:739–746. doi: 10.1083/jcb.200109026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jessell TM. Neuronal specification in the spinal cord: inductive signals and transcriptional codes. Nat Rev Genet. 2000;1:20–29. doi: 10.1038/35049541. [DOI] [PubMed] [Google Scholar]
- Jiang Y, Obama H, Kuan SL, Nakamura R, Nakamoto C, Ouyang Z, Nakamoto M. In vitro guidance of retinal axons by a tectal lamina-specific glycoprotein Nel. Mol Cell Neurosci. 2009;41:113–119. doi: 10.1016/j.mcn.2009.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kang Y, Zhang X, Dobie F, Wu H, Craig AM. Induction of GABAergic postsynaptic differentiation by alpha-neurexins. J Biol Chem. 2008;283:2323–2334. doi: 10.1074/jbc.M703957200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kayser MS, McClelland AC, Hughes EG, Dalva MB. Intracellular and trans-synaptic regulation of glutamatergic synaptogenesis by EphB receptors. J Neurosci. 2006;26:12152–12164. doi: 10.1523/JNEUROSCI.3072-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim S, Burette A, Chung HS, Kwon SK, Woo J, Lee HW, Kim K, Kim H, Weinberg RJ, Kim E. NGL family PSD-95-interacting adhesion molecules regulate excitatory synapse formation. Nat Neurosci. 2006;9:1294–1301. doi: 10.1038/nn1763. [DOI] [PubMed] [Google Scholar]
- Ko J, Fuccillo MV, Malenka RC, Sudhof TC. LRRTM2 functions as a neurexin ligand in promoting excitatory synapse formation. Neuron. 2009;64:791–798. doi: 10.1016/j.neuron.2009.12.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ko J, Kim S, Chung HS, Kim K, Han K, Kim H, Jun H, Kaang BK, Kim E. SALM synaptic cell adhesion-like molecules regulate the differentiation of excitatory synapses. Neuron. 2006;50:233–245. doi: 10.1016/j.neuron.2006.04.005. [DOI] [PubMed] [Google Scholar]
- Lein ES, Hawrylycz MJ, Ao N, Ayres M, Bensinger A, Bernard A, Boe AF, Boguski MS, Brockway KS, Byrnes EJ, et al. Genome-wide atlas of gene expression in the adult mouse brain. Nature. 2007;445:168–176. doi: 10.1038/nature05453. [DOI] [PubMed] [Google Scholar]
- Lin JC, Ho WH, Gurney A, Rosenthal A. The netrin-G1 ligand NGL-1 promotes the outgrowth of thalamocortical axons. Nat Neurosci. 2003;6:1270–1276. doi: 10.1038/nn1148. [DOI] [PubMed] [Google Scholar]
- Linhoff MW, Lauren J, Cassidy RM, Dobie FA, Takahashi H, Nygaard HB, Airaksinen MS, Strittmatter SM, Craig AM. An unbiased expression screen for synaptogenic proteins identifies the LRRTM protein family as synaptic organizers. Neuron. 2009;61:734–749. doi: 10.1016/j.neuron.2009.01.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Livet J, Sigrist M, Stroebel S, De Paola V, Price SR, Henderson CE, Jessell TM, Arber S. ETS gene Pea3 controls the central position and terminal arborization of specific motor neuron pools. Neuron. 2002;35:877–892. doi: 10.1016/s0896-6273(02)00863-2. [DOI] [PubMed] [Google Scholar]
- Lohmann C, Bonhoeffer T. A role for local calcium signaling in rapid synaptic partner selection by dendritic filopodia. Neuron. 2008;59:253–260. doi: 10.1016/j.neuron.2008.05.025. [DOI] [PubMed] [Google Scholar]
- Luo L, Flanagan JG. Development of continuous and discrete neural maps. Neuron. 2007;56:284–300. doi: 10.1016/j.neuron.2007.10.014. [DOI] [PubMed] [Google Scholar]
- Margeta MA, Shen K, Grill B. Building a synapse: lessons on synaptic specificity and presynaptic assembly from the nematode C. elegans. Curr Opin Neurobiol. 2008;18:69–76. doi: 10.1016/j.conb.2008.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mast JD, Prakash S, Chen PL, Clandinin TR. The mechanisms and molecules that connect photoreceptor axons to their targets in Drosophila. Semin Cell Dev Biol. 2006;17:42–49. doi: 10.1016/j.semcdb.2005.11.004. [DOI] [PubMed] [Google Scholar]
- Matsuda K, Miura E, Miyazaki T, Kakegawa W, Emi K, Narumi S, Fukazawa Y, Ito-Ishida A, Kondo T, Shigemoto R, et al. Cbln1 is a ligand for an orphan glutamate receptor delta2, a bidirectional synapse organizer. Science. 2010;328:363–368. doi: 10.1126/science.1185152. [DOI] [PubMed] [Google Scholar]
- Missler M, Zhang W, Rohlmann A, Kattenstroth G, Hammer RE, Gottmann K, Sudhof TC. Alpha-neurexins couple Ca2+ channels to synaptic vesicle exocytosis. Nature. 2003;423:939–948. doi: 10.1038/nature01755. [DOI] [PubMed] [Google Scholar]
- Molnar Z, Blakemore C. Development of signals influencing the growth and termination of thalamocortical axons in organotypic culture. Exp Neurol. 1999;156:363–393. doi: 10.1006/exnr.1999.7032. [DOI] [PubMed] [Google Scholar]
- Mumm JS, Williams PR, Godinho L, Koerber A, Pittman AJ, Roeser T, Chien CB, Baier H, Wong RO. In vivo imaging reveals dendritic targeting of laminated afferents by zebrafish retinal ganglion cells. Neuron. 2006;52:609–621. doi: 10.1016/j.neuron.2006.10.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagel G, Szellas T, Huhn W, Kateriya S, Adeishvili N, Berthold P, Ollig D, Hegemann P, Bamberg E. Channelrhodopsin-2, a directly light-gated cation-selective membrane channel. Proc Natl Acad Sci U S A. 2003;100:13940–13945. doi: 10.1073/pnas.1936192100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakashiba T, Ikeda T, Nishimura S, Tashiro K, Honjo T, Culotti JG, Itohara S. Netrin-G1: a novel glycosyl phosphatidylinositol-linked mammalian netrin that is functionally divergent from classical netrins. J Neurosci. 2000;20:6540–6550. doi: 10.1523/JNEUROSCI.20-17-06540.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakashiba T, Nishimura S, Ikeda T, Itohara S. Complementary expression and neurite outgrowth activity of netrin-G subfamily members. Mech Dev. 2002;111:47–60. doi: 10.1016/s0925-4773(01)00600-1. [DOI] [PubMed] [Google Scholar]
- Nam CI, Chen L. Postsynaptic assembly induced by neurexin-neuroligin interaction and neurotransmitter. Proc Natl Acad Sci U S A. 2005;102:6137–6142. doi: 10.1073/pnas.0502038102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nevin LM, Taylor MR, Baier H. Hardwiring of fine synaptic layers in the zebrafish visual pathway. Neural Dev. 2008;3:36. doi: 10.1186/1749-8104-3-36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nishimura-Akiyoshi S, Niimi K, Nakashiba T, Itohara S. Axonal netrin-Gs transneuronally determine lamina-specific subdendritic segments. Proc Natl Acad Sci U S A. 2007;104:14801–14806. doi: 10.1073/pnas.0706919104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pascual M, Perez-Sust P, Soriano E. The GABAergic septohippocampal pathway in control and reeler mice: target specificity and termination onto Reelin-expressing interneurons. Mol Cell Neurosci. 2004;25:679–691. doi: 10.1016/j.mcn.2003.12.009. [DOI] [PubMed] [Google Scholar]
- Pecho-Vrieseling E, Sigrist M, Yoshida Y, Jessell TM, Arber S. Specificity of sensory-motor connections encoded by Sema3e-Plxnd1 recognition. Nature. 2009;459:842–846. doi: 10.1038/nature08000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Petreanu L, Huber D, Sobczyk A, Svoboda K. Channelrhodopsin-2-assisted circuit mapping of long-range callosal projections. Nat Neurosci. 2007;10:663–668. doi: 10.1038/nn1891. [DOI] [PubMed] [Google Scholar]
- Petreanu L, Mao T, Sternson SM, Svoboda K. The subcellular organization of neocortical excitatory connections. Nature. 2009;457:1142–1145. doi: 10.1038/nature07709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Petrenko AG, Ullrich B, Missler M, Krasnoperov V, Rosahl TW, Sudhof TC. Structure and evolution of neurexophilin. J Neurosci. 1996;16:4360–4369. doi: 10.1523/JNEUROSCI.16-14-04360.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poskanzer K, Needleman LA, Bozdagi O, Huntley GW. N-cadherin regulates ingrowth and laminar targeting of thalamocortical axons. J Neurosci. 2003;23:2294–2305. doi: 10.1523/JNEUROSCI.23-06-02294.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poulopoulos A, Aramuni G, Meyer G, Soykan T, Hoon M, Papadopoulos T, Zhang M, Paarmann I, Fuchs C, Harvey K, et al. Neuroligin 2 drives postsynaptic assembly at perisomatic inhibitory synapses through gephyrin and collybistin. Neuron. 2009;63:628–642. doi: 10.1016/j.neuron.2009.08.023. [DOI] [PubMed] [Google Scholar]
- Redies C, Takeichi M. Cadherins in the developing central nervous system: an adhesive code for segmental and functional subdivisions. Dev Biol. 1996;180:413–423. doi: 10.1006/dbio.1996.0315. [DOI] [PubMed] [Google Scholar]
- Rose D, Chiba A. Synaptic target recognition at Drosophila neuromuscular junctions. Microsc Res Tech. 2000;49:3–13. doi: 10.1002/(SICI)1097-0029(20000401)49:1<3::AID-JEMT2>3.0.CO;2-E. [DOI] [PubMed] [Google Scholar]
- Rowen L, Young J, Birditt B, Kaur A, Madan A, Philipps DL, Qin S, Minx P, Wilson RK, Hood L, Graveley BR. Analysis of the human neurexin genes: alternative splicing and the generation of protein diversity. Genomics. 2002;79:587–597. doi: 10.1006/geno.2002.6734. [DOI] [PubMed] [Google Scholar]
- Sahay A, Molliver ME, Ginty DD, Kolodkin AL. Semaphorin 3F is critical for development of limbic system circuitry and is required in neurons for selective CNS axon guidance events. J Neurosci. 2003;23:6671–6680. doi: 10.1523/JNEUROSCI.23-17-06671.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanes JR, Zipursky SL. Design principles of insect and vertebrate visual systems. Neuron. 2010;66:15–36. doi: 10.1016/j.neuron.2010.01.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scheiffele P, Fan J, Choih J, Fetter R, Serafini T. Neuroligin expressed in nonneuronal cells triggers presynaptic development in contacting axons. Cell. 2000;101:657–669. doi: 10.1016/s0092-8674(00)80877-6. [DOI] [PubMed] [Google Scholar]
- Shen K, Scheiffele P. Genetics and cell biology of building specific synaptic connectivity. Annu Rev Neurosci. 2010;33:473–507. doi: 10.1146/annurev.neuro.051508.135302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shirasaki R, Pfaff SL. Transcriptional codes and the control of neuronal identity. Annu Rev Neurosci. 2002;25:251–281. doi: 10.1146/annurev.neuro.25.112701.142916. [DOI] [PubMed] [Google Scholar]
- Siddiqui TJ, Pancaroglu R, Kang Y, Rooyakkers A, Craig AM. LRRTMs and neuroligins bind neurexins with a differential code to cooperate in glutamate synapse development. J Neurosci. 2010;30:7495–7506. doi: 10.1523/JNEUROSCI.0470-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song JY, Ichtchenko K, Sudhof TC, Brose N. Neuroligin 1 is a postsynaptic cell-adhesion molecule of excitatory synapses. Proc Natl Acad Sci U S A. 1999;96:1100–1105. doi: 10.1073/pnas.96.3.1100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sugita S, Saito F, Tang J, Satz J, Campbell K, Sudhof TC. A stoichiometric complex of neurexins and dystroglycan in brain. J Cell Biol. 2001;154:435–445. doi: 10.1083/jcb.200105003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suto F, Tsuboi M, Kamiya H, Mizuno H, Kiyama Y, Komai S, Shimizu M, Sanbo M, Yagi T, Hiromi Y, et al. Interactions between plexin-A2, plexin-A4, and semaphorin 6A control lamina-restricted projection of hippocampal mossy fibers. Neuron. 2007;53:535–547. doi: 10.1016/j.neuron.2007.01.028. [DOI] [PubMed] [Google Scholar]
- Tabuchi K, Sudhof TC. Structure and evolution of neurexin genes: insight into the mechanism of alternative splicing. Genomics. 2002;79:849–859. doi: 10.1006/geno.2002.6780. [DOI] [PubMed] [Google Scholar]
- Tai CY, Kim SA, Schuman EM. Cadherins and synaptic plasticity. Curr Opin Cell Biol. 2008;20:567–575. doi: 10.1016/j.ceb.2008.06.003. [DOI] [PubMed] [Google Scholar]
- Takeichi M. The cadherin superfamily in neuronal connections and interactions. Nat Rev Neurosci. 2007;8:11–20. doi: 10.1038/nrn2043. [DOI] [PubMed] [Google Scholar]
- Terauchi A, Johnson-Venkatesh EM, Toth AB, Javed D, Sutton MA, Umemori H. Distinct FGFs promote differentiation of excitatory and inhibitory synapses. Nature. 2010;465:783–787. doi: 10.1038/nature09041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tran TS, Rubio ME, Clem RL, Johnson D, Case L, Tessier-Lavigne M, Huganir RL, Ginty DD, Kolodkin AL. Secreted semaphorins control spine distribution and morphogenesis in the postnatal CNS. Nature. 2009;462:1065–1069. doi: 10.1038/nature08628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uemura T, Lee SJ, Yasumura M, Takeuchi T, Yoshida T, Ra M, Taguchi R, Sakimura K, Mishina M. Trans-synaptic interaction of GluRdelta2 and Neurexin through Cbln1 mediates synapse formation in the cerebellum. Cell. 2010;141:1068–1079. doi: 10.1016/j.cell.2010.04.035. [DOI] [PubMed] [Google Scholar]
- Ullrich B, Ushkaryov YA, Sudhof TC. Cartography of neurexins: more than 1000 isoforms generated by alternative splicing and expressed in distinct subsets of neurons. Neuron. 1995;14:497–507. doi: 10.1016/0896-6273(95)90306-2. [DOI] [PubMed] [Google Scholar]
- Ushkaryov YA, Petrenko AG, Geppert M, Sudhof TC. Neurexins: synaptic cell surface proteins related to the alpha-latrotoxin receptor and laminin. Science. 1992;257:50–56. doi: 10.1126/science.1621094. [DOI] [PubMed] [Google Scholar]
- Varoqueaux F, Jamain S, Brose N. Neuroligin 2 is exclusively localized to inhibitory synapses. Eur J Cell Biol. 2004;83:449–456. doi: 10.1078/0171-9335-00410. [DOI] [PubMed] [Google Scholar]
- Vrieseling E, Arber S. Target-induced transcriptional control of dendritic patterning and connectivity in motor neurons by the ETS gene Pea3. Cell. 2006;127:1439–1452. doi: 10.1016/j.cell.2006.10.042. [DOI] [PubMed] [Google Scholar]
- Wang CY, Chang K, Petralia RS, Wang YX, Seabold GK, Wenthold RJ. A novel family of adhesion-like molecules that interacts with the NMDA receptor. J Neurosci. 2006;26:2174–2183. doi: 10.1523/JNEUROSCI.3799-05.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang K, Zhang H, Ma D, Bucan M, Glessner JT, Abrahams BS, Salyakina D, Imielinski M, Bradfield JP, Sleiman PM, et al. Common genetic variants on 5p14.1 associate with autism spectrum disorders. Nature. 2009 doi: 10.1038/nature07999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wickersham IR, Lyon DC, Barnard RJ, Mori T, Finke S, Conzelmann KK, Young JA, Callaway EM. Monosynaptic restriction of transsynaptic tracing from single, genetically targeted neurons. Neuron. 2007;53:639–647. doi: 10.1016/j.neuron.2007.01.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woo J, Kwon SK, Choi S, Kim S, Lee JR, Dunah AW, Sheng M, Kim E. Trans-synaptic adhesion between NGL-3 and LAR regulates the formation of excitatory synapses. Nat Neurosci. 2009;12:428–437. doi: 10.1038/nn.2279. [DOI] [PubMed] [Google Scholar]
- Yamagata M, Herman JP, Sanes JR. Lamina-specific expression of adhesion molecules in developing chick optic tectum. J Neurosci. 1995;15:4556–4571. doi: 10.1523/JNEUROSCI.15-06-04556.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamagata M, Sanes JR. Lamina-specific cues guide outgrowth and arborization of retinal axons in the optic tectum. Development. 1995;121:189–200. doi: 10.1242/dev.121.1.189. [DOI] [PubMed] [Google Scholar]
- Yamagata M, Sanes JR. Dscam and Sidekick proteins direct lamina-specific synaptic connections in vertebrate retina. Nature. 2008;451:465–469. doi: 10.1038/nature06469. [DOI] [PubMed] [Google Scholar]
- Yamagata M, Weiner JA, Dulac C, Roth KA, Sanes JR. Labeled lines in the retinotectal system: markers for retinorecipient sublaminae and the retinal ganglion cell subsets that innervate them. Mol Cell Neurosci. 2006;33:296–310. doi: 10.1016/j.mcn.2006.08.001. [DOI] [PubMed] [Google Scholar]
- Yamagata M, Weiner JA, Sanes JR. Sidekicks: synaptic adhesion molecules that promote lamina-specific connectivity in the retina. Cell. 2002;110:649–660. doi: 10.1016/s0092-8674(02)00910-8. [DOI] [PubMed] [Google Scholar]
- Yamamoto N, Higashi S, Toyama K. Stop and branch behaviors of geniculocortical axons: a time-lapse study in organotypic cocultures. J Neurosci. 1997;17:3653–3663. doi: 10.1523/JNEUROSCI.17-10-03653.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamamoto N, Kurotani T, Toyama K. Neural connections between the lateral geniculate nucleus and visual cortex in vitro. Science. 1989;245:192–194. doi: 10.1126/science.2749258. [DOI] [PubMed] [Google Scholar]
- Yin Y, Miner JH, Sanes JR. Laminets: laminin- and netrin-related genes expressed in distinct neuronal subsets. Mol Cell Neurosci. 2002;19:344–358. doi: 10.1006/mcne.2001.1089. [DOI] [PubMed] [Google Scholar]
- Zhang C, Atasoy D, Arac D, Yang X, Fucillo MV, Robison AJ, Ko J, Brunger AT, Sudhof TC. Neurexins physically and functionally interact with GABA(A) receptors. Neuron. 2010;66:403–416. doi: 10.1016/j.neuron.2010.04.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou D, Lambert S, Malen PL, Carpenter S, Boland LM, Bennett V. AnkyrinG is required for clustering of voltage-gated Na channels at axon initial segments and for normal action potential firing. J Cell Biol. 1998;143:1295–1304. doi: 10.1083/jcb.143.5.1295. [DOI] [PMC free article] [PubMed] [Google Scholar]


