Abstract
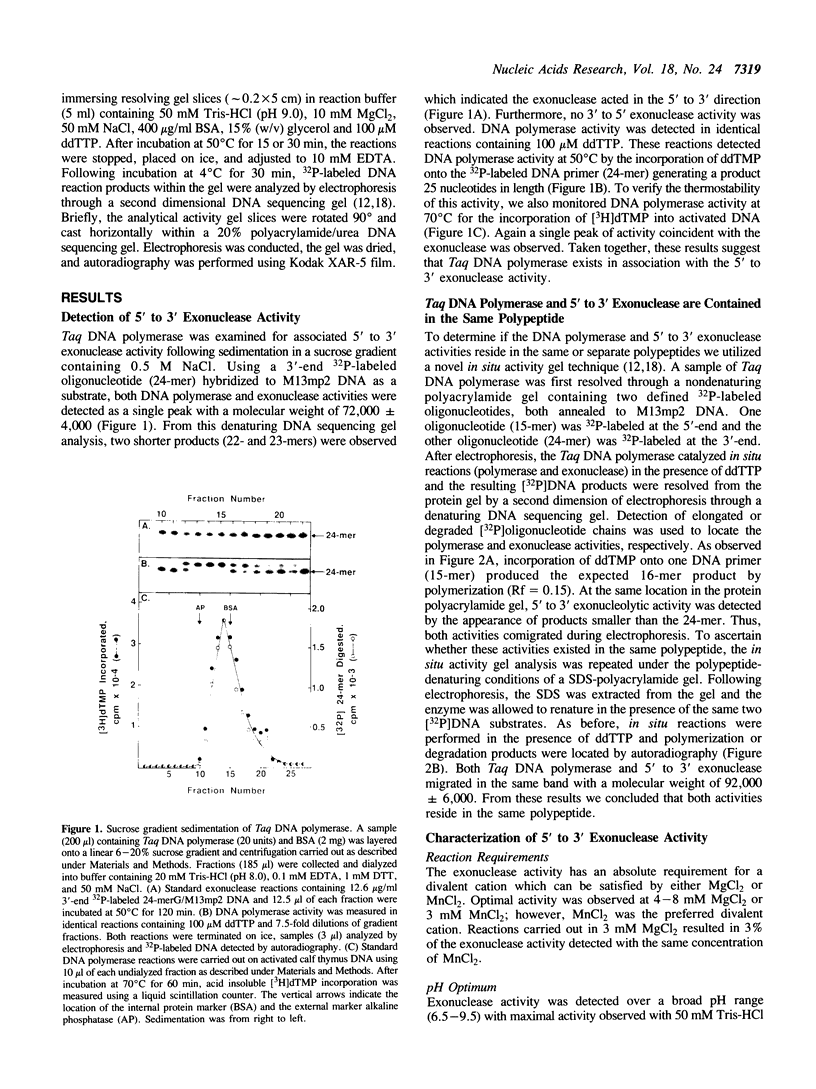
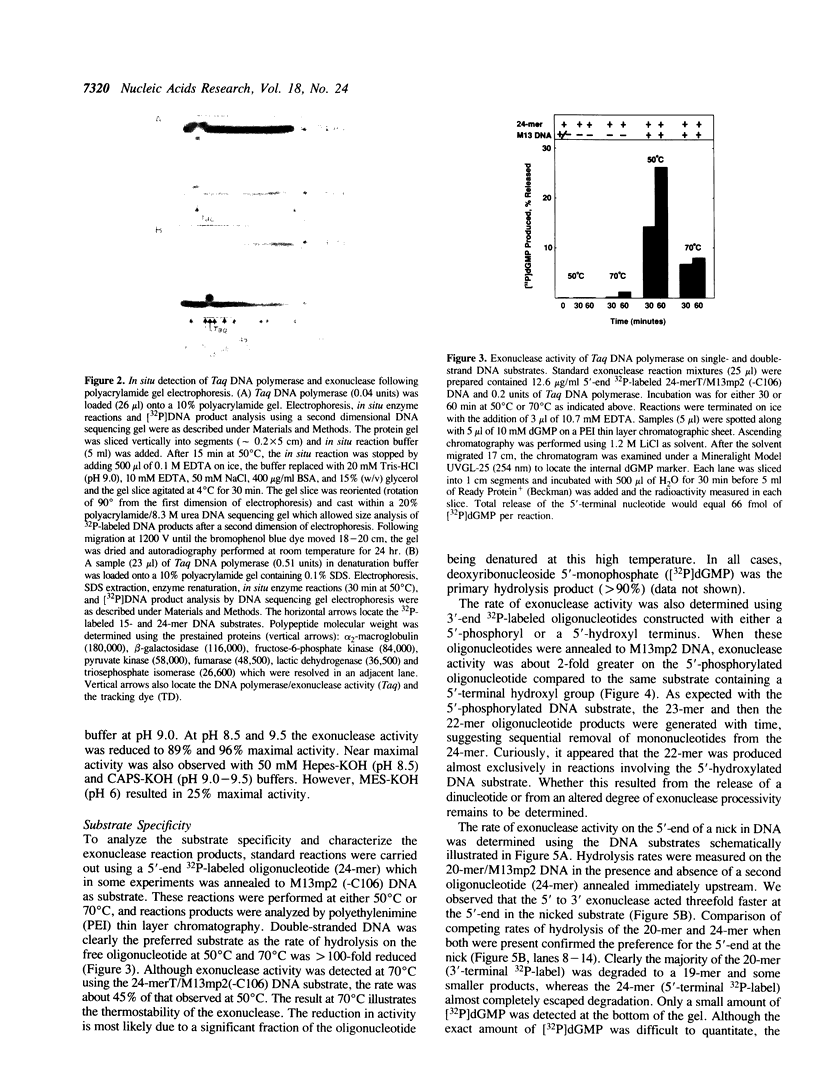
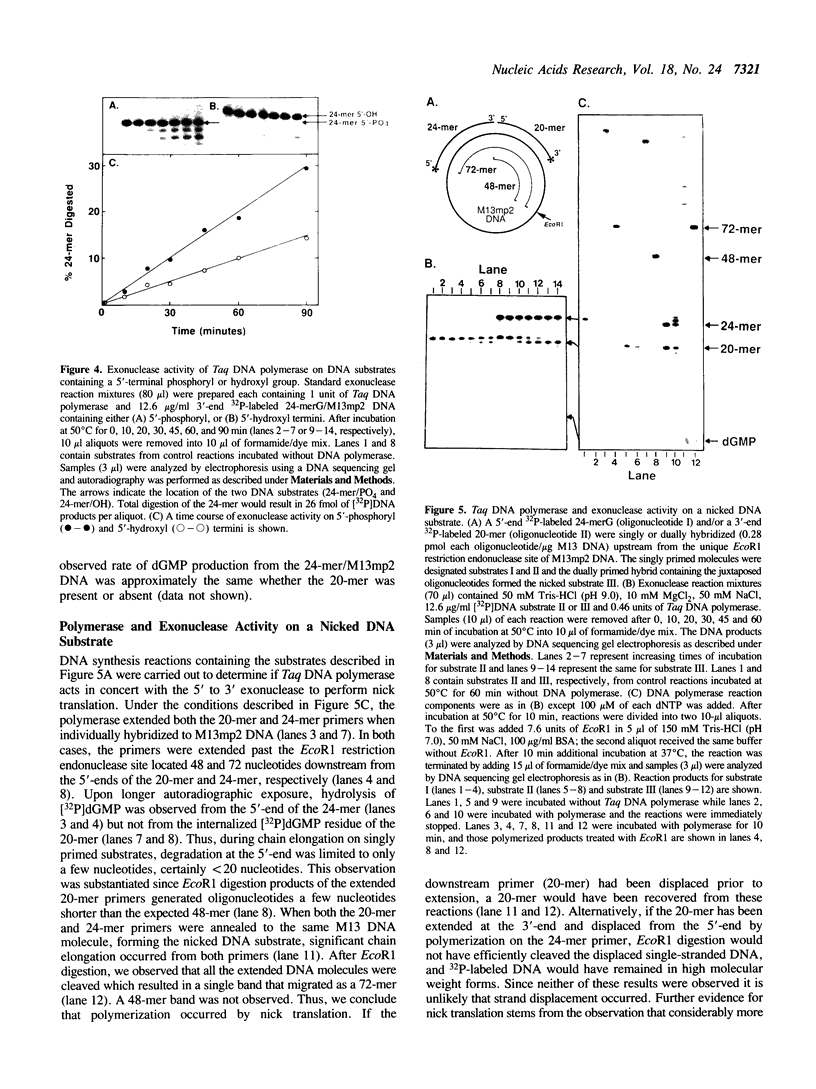
Thermus aquaticus DNA polymerase was shown to contain an associated 5' to 3' exonuclease activity. Both polymerase and exonuclease activities cosedimented with a molecular weight of 72,000 during sucrose gradient centrifugation. Using a novel in situ activity gel procedure to simultaneously detect these two activities, we observed both DNA polymerase and exonuclease in a single band following either nondenaturing or denaturing polyacrylamide gel electrophoresis: therefore, DNA polymerase and exonuclease activities reside in the same polypeptide. As determined by SDS-polyacrylamide gel electrophoresis this enzyme has an apparent molecular weight of 92,000. The exonuclease requires a divalent cation (MgCl2 or MnCl2), has a pH optimum of 9.0 and excises primarily deoxyribonucleoside 5'-monophosphate from double-stranded DNA. Neither heat denatured DNA nor the free oligonucleotide (24-mer) were efficient substrates for exonuclease activity. The rate of hydrolysis of a 5'-phosphorylated oligonucleotide (24-mer) annealed to M13mp2 DNA was about twofold faster than the same substrate containing a 5'-hydroxylated residue. Hydrolysis of a 5'-terminal residue from a nick was preferred threefold over the same 5'-end of duplex DNA. The 5' to 3' exonuclease activity appeared to function coordinately with the DNA polymerase to facilitate a nick translational DNA synthesis reaction.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1006/abio.1976.9999. [DOI] [PubMed] [Google Scholar]
- Chien A., Edgar D. B., Trela J. M. Deoxyribonucleic acid polymerase from the extreme thermophile Thermus aquaticus. J Bacteriol. 1976 Sep;127(3):1550–1557. doi: 10.1128/jb.127.3.1550-1557.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Innis M. A., Myambo K. B., Gelfand D. H., Brow M. A. DNA sequencing with Thermus aquaticus DNA polymerase and direct sequencing of polymerase chain reaction-amplified DNA. Proc Natl Acad Sci U S A. 1988 Dec;85(24):9436–9440. doi: 10.1073/pnas.85.24.9436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacobsen H., Klenow H., Overgaard-Hansen K. The N-terminal amino-acid sequences of DNA polymerase I from Escherichia coli and of the large and the small fragments obtained by a limited proteolysis. Eur J Biochem. 1974 Jun 15;45(2):623–627. doi: 10.1111/j.1432-1033.1974.tb03588.x. [DOI] [PubMed] [Google Scholar]
- Joyce C. M., Fujii D. M., Laks H. S., Hughes C. M., Grindley N. D. Genetic mapping and DNA sequence analysis of mutations in the polA gene of Escherichia coli. J Mol Biol. 1985 Nov 20;186(2):283–293. doi: 10.1016/0022-2836(85)90105-6. [DOI] [PubMed] [Google Scholar]
- Joyce C. M., Kelley W. S., Grindley N. D. Nucleotide sequence of the Escherichia coli polA gene and primary structure of DNA polymerase I. J Biol Chem. 1982 Feb 25;257(4):1958–1964. [PubMed] [Google Scholar]
- Kaledin A. S., Sliusarenko A. G., Gorodetskii S. I. Vydelenie i svoistva DNK-polimerazy is ekstremal'no-termofil'noi bakterii Thermus Aquaticus YT1. Biokhimiia. 1980 Apr;45(4):644–651. [PubMed] [Google Scholar]
- Kunkel T. A., Mosbaugh D. W. Exonucleolytic proofreading by a mammalian DNA polymerase. Biochemistry. 1989 Feb 7;28(3):988–995. doi: 10.1021/bi00429a011. [DOI] [PubMed] [Google Scholar]
- Kunkel T. A., Roberts J. D., Zakour R. A. Rapid and efficient site-specific mutagenesis without phenotypic selection. Methods Enzymol. 1987;154:367–382. doi: 10.1016/0076-6879(87)54085-x. [DOI] [PubMed] [Google Scholar]
- Lawyer F. C., Stoffel S., Saiki R. K., Myambo K., Drummond R., Gelfand D. H. Isolation, characterization, and expression in Escherichia coli of the DNA polymerase gene from Thermus aquaticus. J Biol Chem. 1989 Apr 15;264(11):6427–6437. [PubMed] [Google Scholar]
- Lundquist R. C., Olivera B. M. Transient generation of displaced single-stranded DNA during nick translation. Cell. 1982 Nov;31(1):53–60. doi: 10.1016/0092-8674(82)90404-4. [DOI] [PubMed] [Google Scholar]
- MARTIN R. G., AMES B. N. A method for determining the sedimentation behavior of enzymes: application to protein mixtures. J Biol Chem. 1961 May;236:1372–1379. [PubMed] [Google Scholar]
- Mosbaugh D. W. Purification and characterization of porcine liver DNA polymerase gamma: utilization of dUTP and dTTP during in vitro DNA synthesis. Nucleic Acids Res. 1988 Jun 24;16(12):5645–5659. doi: 10.1093/nar/16.12.5645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mosbaugh D. W., Stalker D. M., Probst G. S., Meyer R. R. Novikoff hepatoma deoxyribonucleic acid polymerase. Identification of a stimulatory protein bound to the beta-polymerase. Biochemistry. 1977 Apr 5;16(7):1512–1518. doi: 10.1021/bi00626a041. [DOI] [PubMed] [Google Scholar]
- Ollis D. L., Brick P., Hamlin R., Xuong N. G., Steitz T. A. Structure of large fragment of Escherichia coli DNA polymerase I complexed with dTMP. 1985 Feb 28-Mar 6Nature. 313(6005):762–766. doi: 10.1038/313762a0. [DOI] [PubMed] [Google Scholar]
- Saiki R. K., Gelfand D. H., Stoffel S., Scharf S. J., Higuchi R., Horn G. T., Mullis K. B., Erlich H. A. Primer-directed enzymatic amplification of DNA with a thermostable DNA polymerase. Science. 1988 Jan 29;239(4839):487–491. doi: 10.1126/science.2448875. [DOI] [PubMed] [Google Scholar]
- Saiki R. K., Scharf S., Faloona F., Mullis K. B., Horn G. T., Erlich H. A., Arnheim N. Enzymatic amplification of beta-globin genomic sequences and restriction site analysis for diagnosis of sickle cell anemia. Science. 1985 Dec 20;230(4732):1350–1354. doi: 10.1126/science.2999980. [DOI] [PubMed] [Google Scholar]
- Tindall K. R., Kunkel T. A. Fidelity of DNA synthesis by the Thermus aquaticus DNA polymerase. Biochemistry. 1988 Aug 9;27(16):6008–6013. doi: 10.1021/bi00416a027. [DOI] [PubMed] [Google Scholar]






