Abstract
The fetal semi-allograft can induce expansion and tolerance of antigen-specific maternal T and B cells through paternally inherited major histocompatibility complex and minor histocompatibility antigens (mHAgs). The effects of these antigens have important consequences on the maternal immune system both during and long after pregnancy. Herein, we investigate the possibility that the placental syncytiotrophoblast and deported trophoblastic debris serve as sources of fetal mHAgs. We mapped the expression of four mHAgs (human mHAg 1, pumilio domain-containing protein KIAA0020, B-cell lymphoma 2–related protein A1, and ribosomal protein S4, Y linked) in the placenta. Each of these proteins was expressed in several placental cell types, including the syncytiotrophoblast. These antigens and two additional Y chromosome–encoded antigens [DEAD box polypeptide 3, Y linked (DDX3Y), and lysine demethylase5D] were also identified by RT-PCR in the placenta, purified trophoblast cells, and cord blood cells. Finally, we used a proteomic approach to investigate the presence of mHAgs in the syncytiotrophoblast and trophoblast debris shed from first-trimester placenta. By this method, four antigens (DDX3Y; ribosomal protein S4, Y linked; solute carrier 1A5; and signal sequence receptor 1) were found in the syncytiotrophoblast, and one antigen (DDX3Y) was found in shed trophoblast debris. The finding of mHAgs in the placenta and in trophoblast debris provides the first direct evidence that fetal antigens are present in debris shed from the human placenta. The data, thus, suggest a mechanism by which the maternal immune system is exposed to fetal alloantigens, possibly explaining the relationship between parity and graft-versus-host disease.
During pregnancy, the maternal immune system must tolerate the semi-allograft of the fetus and its placenta. In human pregnancy, this is achieved through several means, including passively, through the highly restricted expression of class Ia and class II human leukocyte antigens (HLAs) in trophoblast cells. Active tolerance also plays an important role, for example, through trophoblast-associated immunoregulatory molecules.1 Studies2–5 in mice and women indicate that there is an important role for regulatory T cells in immune tolerance to fetal antigens and, furthermore, that this tolerance can be established early in pregnancy.
Minor histocompatibility antigens (mHAgs) represent a class of self-proteins encoded outside the major histocompatibility complex (MHC) that can elicit alloimmune responses across individuals. Many of these antigens are encoded by diallelic autosomal genes, others are encoded on the Y chromosome, and at least one gene is absent by deletion in some individuals.6 These antigens are HLA restricted: when broken down by antigen-presenting cells, one or more of the resulting peptides possess the correct structural properties to permit its presentation by class I or class II MHC molecules. Consequently, they are recognized by alloreactive CD4+or CD8+ T cells from individuals lacking and, therefore, untolerized to, these proteins. mHAgs were discovered as a cause of chronic graft rejection, graft-versus-host disease, and the graft-versus-leukemia effect in HLA-matched donor-recipient pairs.7–9
In addition to eliciting immune responses after transplantation, mHAgs can also induce immune responses in the physiological context of pregnancy, in both mice and women. In murine pregnancy, CD4+ and CD8+ T cells are stimulated and tolerized by paternally inherited natural mHAgs and transgenically expressed model minor antigens present in the seminal fluid and fetus.10–12 These fetal antigen-specific T cells can be isolated from blood in multiparous mice and possess functional activity.13 The expansion and persistence of fetal antigen-specific cohorts of T cells in women long after pregnancy have also been recognized.14–16 The exposure of maternal T and B cells to fetal mHAgs and the ensuing response are presumably tolerogenic in normal pregnancy but may have important clinical consequences. For example, multiparity has a negative impact on organ and hematopoietic stem cell transplantation, effectively increasing the risk of transplant rejection and graft-versus-host disease.17–19
Although the antigen-specific response of maternal lymphocytes to paternally inherited antigens during pregnancy is well documented, the route of exposure of these antigens remains uncertain. One possible route of exposure is fetal microchimerism, in which fetal and/or placental cells actively traffic across the placenta and lodge within maternal organs, where they remain long-lived.20–23 A second likely source of fetal antigen is the placenta. The human placenta, which possesses a hemochorial arrangement, is bathed by maternal blood during the latter two thirds of pregnancy. The syncytiotrophoblast forms the outer layer of the placental villi and is the major interface between fetal tissues and maternal blood. An abundance of placental material is shed into the maternal circulation, and this physiological property of the placenta is thought to have important consequences on maternal physiological and pathological responses in pregnancy.24,25 The terminally differentiated syncytiotrophoblast is continuously renewed by the fusion of underlying cytotrophoblast precursors, whereas aged or damaged portions of the syncytiotrophoblast layer are extruded into the maternal blood as large multinucleated structures called syncytial knots.25 Other trophoblast debris shed into the maternal circulation includes mononuclear cytotrophoblasts and subcellular microparticles and nanoparticles.26–28 Gram quantities of trophoblastic elements may be deported from the placenta each day in normal pregnancy,29–32 and the quantity of deported material may substantially increase during preeclamptic pregnancy.33,34 This trophoblast debris is rapidly cleared from the maternal circulation without apparently generating an inflammatory immune response in normal pregnancy.31
Researchers25,35–37 have proposed that the shedding of trophoblast debris from the placenta provides an important avenue for immunological exposure of the mother to paternally derived fetal antigens, serving to establish maternal immune tolerance to the fetus. Whether such tolerance of the maternal adaptive immune system is possible is unclear, however, because the syncytiotrophoblast and villous cytotrophoblasts lack polymorphic HLA. The objective of this study, therefore, was to investigate whether mHAgs are present in the human placenta and in trophoblast debris shed from the placenta. We chose to analyze the expression of six minor antigens in the placenta, whose properties and mRNA expression pattern are summarized in Table 1: the autosomal antigens [human mHAg 1 (HMHA1), KIAA0020, and B-cell lymphoma 2– related protein A1 (BCL2A1)]; and the Y chromosome–encoded antigens [lysine demethylase 5D (KDM5D), ribosomal protein S4, Y linked (RPS4Y1), and DEAD box polypeptide 3, Y linked (DDX3Y)]. The HMHA1 and KDM5D antigens elicit a maternal response in pregnancy,13,15,16 and, like KDM5D, RPS4Y1 and DDX3Y were of interest as male antigens. BCL2A1 expression was explored because it is a regulator of apoptosis, a property that may be of significance given the putative role of apoptosis in trophoblast shedding.28,30 Finally, KIAA0020 is of interest as a potentially novel inducer of the maternal immune system. By using molecular, immunohistochemical (IHC), and proteomic approaches, we provide the first evidence that the human placenta is a rich source of these and other fetally derived mHAgs.
Table 1.
Properties and Expression of Human mHAgs
| Minor antigen | Gene | Chromosome | HLA restriction | Function | Tissue expression (mRNA) | Reference no. |
|---|---|---|---|---|---|---|
| HA-1 | HMHA1 | 19p13.3 | HLA-A*0201, HLA-A*0206, HLA-B*60, and HLA-B*40012 | GTPase activator (putative) | Hematopoietic cells | 38–40 |
| HA-8 | KIAA0020 | 9p24.2 | HLA-A*0201 | RNA-binding protein (putative) | Broad | 41 |
| ACC-1 and ACC-2 | BCL2A1 | 15q24.3 | HLA-A*2402 and HLA-B*4403 | Negative regulator of apoptosis | Hematopoietic cells | 42 |
| SLC1A5 | SLC1A5 | 19q13.3 | HLA-B*4002 | Amino acid transporter | Some carcinomas, especially colorectal | 43 and 44 |
| LB-SSR1-1S | SSR1 | 6p24.3 | HLA-A*0201 | Signal sequence receptor in endoplasmic reticulum | Hematopoietic cells, small intestine | 44 and 45 |
| A2/HY and B7/HY | KDM5D | Yq11 | HLA-A*0201 and HLA-B*2705 | Protein- containing zinc finger domains | Broad; high in hematopoietic cells | 44 and 46 |
| B52/HY and DRB3/HY | RPS4Y1 | Yp11.3 | HLA-B*5201 and DRB3*0301 | RNA-binding protein | Broad | 47 and 48 |
| DQ5/HY and DRB1/HY | DDX3Y | Yq11a | DQB1*05 and DRB1*1501 | RNA helicase, involved in spermatogenesis | Broad; high in testis | 49 |
KIAA0020, pumilio domain-containing protein KIAA0020; SLC1A5, solute carrier 1A5; SSR1, signal sequence receptor 1.
Materials and Methods
Tissue Procurement
All tissues were obtained in accordance with protocols approved by the University of Kansas Medical Center Institutional Review Board (Kansas City, KS) and the Auckland Regional Ethics Committee (Auckland, New Zealand). First- and second-trimester placentas (8 to 12 and 13 to 21 weeks gestational age, respectively) were obtained from elective terminations of normal pregnancies from a Kansas City-area private office (Overland Park, KS), the University of Chicago Department of Obstetrics and Gynecology (Chicago, IL), the Epsom Day Unit, Greenlane Clinical Center (Auckland, New Zealand) and the Auckland Medical Aid Clinic (Auckland, New Zealand). Term placentas (≥37 weeks' gestational age) were obtained from the University of Kansas Hospital (Kansas City, KS) from uncomplicated pregnancies after elective cesarean sections in the absence of labor. Amniochorion membrane and placental sections were dissected from term placentas, and cord blood was collected after placental delivery.
IHC Data
Samples of first-trimester (n = 5 to 8 per antibody), second-trimester (n = 2 to 3 per antibody), and term (n = 8 to 10 per antibody) placentas, the latter with an attached basal plate (BP), and extraplacental membranes (n = 6 to 8) were fixed in 4% paraformaldehyde overnight, dehydrated through a graded series of ethanol, and embedded in paraffin. Sections (10 μm thick) were placed onto slides, rehydrated, and stained as previously described,50 with modifications dependent on each antibody, as described later. For all antibodies, tissues were subjected to hot citrate antigen retrieval using Reveal buffer (BioCare Medical, Walnut Creek, CA). Nonspecific antibody binding was blocked in 10% goat serum (Sigma-Aldrich, St. Louis, MO). The rabbit polyclonal primary antibodies targeting HMHA1 (12 μg/mL), KIAA0020 (2 μg/mL), and RPS4Y1 (12 μg/mL), all obtained from Sigma-Aldrich, were added to tissue sections, which were then incubated overnight at 4°C. For slides incubated with anti-KIAA0020 antibody, tissue sections were permeabilized in 0.5% Tween 20 (Sigma-Aldrich) in PBS for 30 minutes at room temperature before blocking, and 0.05% Tween 20 was added to PBS for all wash steps. For slides incubated with rabbit anti-BCL2A1 (6 μg/mL; Epitomics, Burlingame, CA), antibody was added for 1 hour at room temperature. After the addition of biotinylated goat anti-rabbit secondary antibodies and the depletion of endogenous peroxidases, binding of primary antibody was detected using streptavidin-peroxidase and aminoethyl carbazole (Invitrogen, Carlsbad, CA).
Purification of Cord Blood Mononuclear Cells and Cytotrophoblast Cells
For isolation of cord blood mononuclear cells, blood was diluted 1:2 in PBS and layered over Histopaque 1077 (Sigma-Aldrich). The blood was centrifuged for 30 minutes at 400 × g, and cells at the serum-Histopaque interface were collected, washed, and counted by trypan dye exclusion, as previously described.51
Cytotrophoblast cells were isolated as previously described.51 Approximately 40 g of villous material was dissected from term placentas, rinsed with 0.9% saline, and subjected to enzymatic digestion using trypsin (Invitrogen) and DNase (Sigma-Aldrich). Dispersed cells were layered over fetal bovine serum and centrifuged at 1000 × g, and after washing, the pellet was resuspended in HBSS, layered over a 5% to 70% stepwise Percoll gradient (Sigma-Aldrich), and centrifuged at 1200 × g. The band corresponding to trophoblast cells was aspirated, and the cells were subjected to a second stage of purification using immunomagnetic beads (Miltenyi Biotec, Boston, MA) coated with anti-class I MHC (W6/32), as previously described.51 Cell purity was assessed using anti-cytokeratin 7 by IHC or flow cytometry and was determined to be >90%.
RNA Isolation and RT-PCR
RNA was extracted from whole placenta (n = 3 female and 5 male), purified cytotrophoblast cells (n = 2 female and 3 male), and cord blood mononuclear cells (n = 2 female and 2 male) using TRI Reagent (Applied Biosystems, Foster City, CA). RNA concentrations were quantified using a BioPhotometer (Eppendorf, Hauppauge, NY), and 1 μg of RNA was reverse transcribed using Superscript III Reverse Transcriptase and random primers (Invitrogen) in a reaction volume of 20 μL. Parallel reactions containing RNA but no reverse transcriptase were included to verify the absence of genomic DNA in samples in the subsequent PCR steps (Fermentas, Glen Burnie, MD). Conventional RT-PCR was performed for DDX3Y, RPS4Y1, and glyceraldehyde-3-phosphate dehydrogenase, with primers designed using PrimerBLAST (National Center for Biotechnology Information, Bethesda, MD) and synthesized by Integrated DNA Technologies (Coralville, IA). Primer sequences were as follows: DDX3Y forward, 5′-TGCTGTCGTATAGCTGTGGG-3′; DDX3Y reverse, 5′-TCTCGAGACCCAAAACTGCT-3′; RPS4Y1 forward, 5′-AGATTCTCTTCCGTCGCAGA-3′; RPS4Y1 reverse, 5′-CATAGACCAGGCGGAAATGT-3′; glyceraldehyde-3-phosphate dehydrogenase forward, 5′-ACCACACTCCATGCCATCAC-3′; and glyceraldehyde-3-phosphate dehydrogenase reverse, 5′-TCCACCACCCTGTTGCTGTA-3′. The RT reaction sample, 2 μL, was subjected to PCR (Invitrogen) using one times PCR buffer, 0.2 mmol/L deoxyribonucleotide triphosphate, 1.5 mmol/L MgCl2, 1.5 U Taq polymerase, and 5 mmol/L of each primer in a volume of 20 μL. The PCR was performed with the following protocol: 94°C for 3 minutes; 35 cycles of 94°C for 45 seconds, 63°C for 30 seconds, and 72°C for 90 seconds; and 72°C for 5 minutes. TaqMan Gene Expression Assay kits for real-time RT-PCR were obtained from Applied Biosystems (Foster City, CA) (HMHA1, Hs00299628_m1; KIAA0020, Hs00972109_m1; BCL2A1, Hs00187845_m1; KDM5D, Hs01104415_m1; and β-actin, Hs99999903) and run according to the manufacturer's protocol. All RT-PCR samples were analyzed in triplicate, and a mean was generated from the three CT values for each sample. For each individual placenta, purified trophoblast or cord blood sample, the mean for each mHAg was subtracted from the mean for β-actin to provide an mHAg CT value that had been normalized to β-actin to account for discrepancies in RNA concentration or quality. These means were then pooled to generate an average CT value for each antigen in each tissue type.
Protein Extraction
For analysis of protein content of shed trophoblast debris, an established in vitro model of trophoblast shedding52 was used, together with a proteomic approach. Trophoblast debris was harvested from 12 first-trimester placentas (8 to 12 weeks' gestational age). Placental tissue was washed with PBS (pH 7.4), dissected into approximately 400 mg of wet-weight explants, and transferred into Net-well inserts (Corning, Lowell, MA) placed in a 12-well culture plate (Becton Dickinson, Franklin Lakes, NJ). The explants were cultured in 3 mL of Dulbecco's modified Eagle's medium/F-12 medium (Invitrogen) containing 5% fetal bovine serum and 1% penicillin-streptomycin for 12 hours at 37°C, in a humidified atmosphere containing 5% CO2 and 95% ambient oxygen. Trophoblast debris shed from the explants was collected from the bottom of the culture plate, and red blood cells were depleted by hypotonic lysis, while contaminating leukocytes were removed using anti-CD45–coated magnetic beads, as previously described.52 The trophoblast debris was subjected to two independent protein extraction methods to enrich the plasma membrane components in the extracts: Ludox CL cationic colloidal silica (Sigma-Aldrich) and direct trypsinization. The colloidal silica extraction protocol followed the method of Robinson et al,53 with minor modifications. For direct trypsinization, briefly, the trophoblast debris was suspended in 30 μL of prewarmed 50 mmol/L NH4HCO3 containing 1 μL of benzonase nuclease (Merck, Darmstadt, Germany). Sequencing-grade trypsin (Promega Corporation, Madison, WI), 1 μL, was added and incubated at 37°C for 80 minutes, with 1 μL of trypsin added at 20-minute intervals. The sample was centrifuged at 17,000 × g at 4°C for 15 minutes, and the supernatant was collected.
To characterize proteins from the syncytiotrophoblast, protein extraction was performed directly on six placentas after extensive washing in PBS. Syncytiotrophoblast proteins were isolated from the surface of placental explants using extraction methods to enrich the plasma membrane component in the extract. Two independent methods were used to extract proteins: Ludox CL cationic colloidal silica (previously described) and Triton X-114 detergent (Sigma-Aldrich). The Triton X-114 extraction protocol followed the method of Dickerson et al,54 with minor modifications. To confirm enrichment of plasma membrane components in the trophoblast debris, the relative proportion of proteins found primarily in the plasma membrane was compared between a non–plasma membrane enrichment technique (radioimmunoprecipitation assay lysis) and the two plasma membrane enrichment techniques, Ludox CL cationic colloidal silica and direct trypsinization. Data were analyzed through the use of Ingenuity Pathways Analysis (Ingenuity Systems, Redwood City, CA) and showed a relative enrichment of 12% (O.J.H. and L.W.C., unpublished data).
MS Data
Liquid chromatography/tandem mass spectrometry (MS/MS) was used to sequence tryptic peptides in the extracts from which protein identities were determined. Samples were electrophoresed on a 4% to 15% SDS polyacrylamide gel, and either the entire lane was excised as a whole or the lane was excised in sections for MS analysis. The excised protein lanes were subjected to in-gel digestion with trypsin using a robotic workstation for automated protein digestion (DigestPro Msi; Intavis AG, Cologne, Germany), following the method of Shevchenko et al.55 The MS measurements were made on the LTQ-Orbitrap hybrid MS (Thermo Scientific, Lafayette, CO). Extracted MS/MS spectra were automatically assigned to the best-matching peptide sequence using the SEQUEST algorithm and the Sequest Browser software package (ThermoFinnigan, Lafayette, CO). The false-discovery rate for the data was calculated by running a decoy search against the reversed Human Reference Sequence database (National Center for Biotechnology Information) using the same search parameters as the main analysis. The reversed database was built from the same copy of the Human Reference Sequence database that has been used for the main searches using the Decoy Database Tool (Trans-Proteomic Pipeline; Seattle Proteome Center, Institute for Systems Biology, Seattle, WA).
Results
RNA for mHAgs in the Placenta and Cord Blood
To determine whether the placenta or fetal blood cells could serve as sources of mHAgs, whole placental lysates, purified cytotrophoblast cells, and fetal cord blood RNA were examined by real-time RT-PCR for the presence of four genes known to encode mHAg proteins. The expression of mRNA for HMHA1, KIAA0020, BCL2A1, and KDM5D was found in placental lysates, purified cytotrophoblast cells, and fetal cord blood (Figure 1A). No expression of the Y chromosome–encoded mHAg KDM5D was found in samples from pregnancies with a female fetus. Because of lack of availability of TaqMan real-time PCR gene expression assay kits that distinguish between the male and female isoforms of the DDX3Y (DDX3X) and RPS4Y1 (RPS4X) genes, we designed primers for conventional RT-PCR that would distinguish these transcripts. Like KDM5D, DDX3Y and RPS4Y1 were identified in whole placenta, cytotrophoblast cells, and cord blood from male, but not female, fetuses (Figure 1, B and C).
Figure 1.
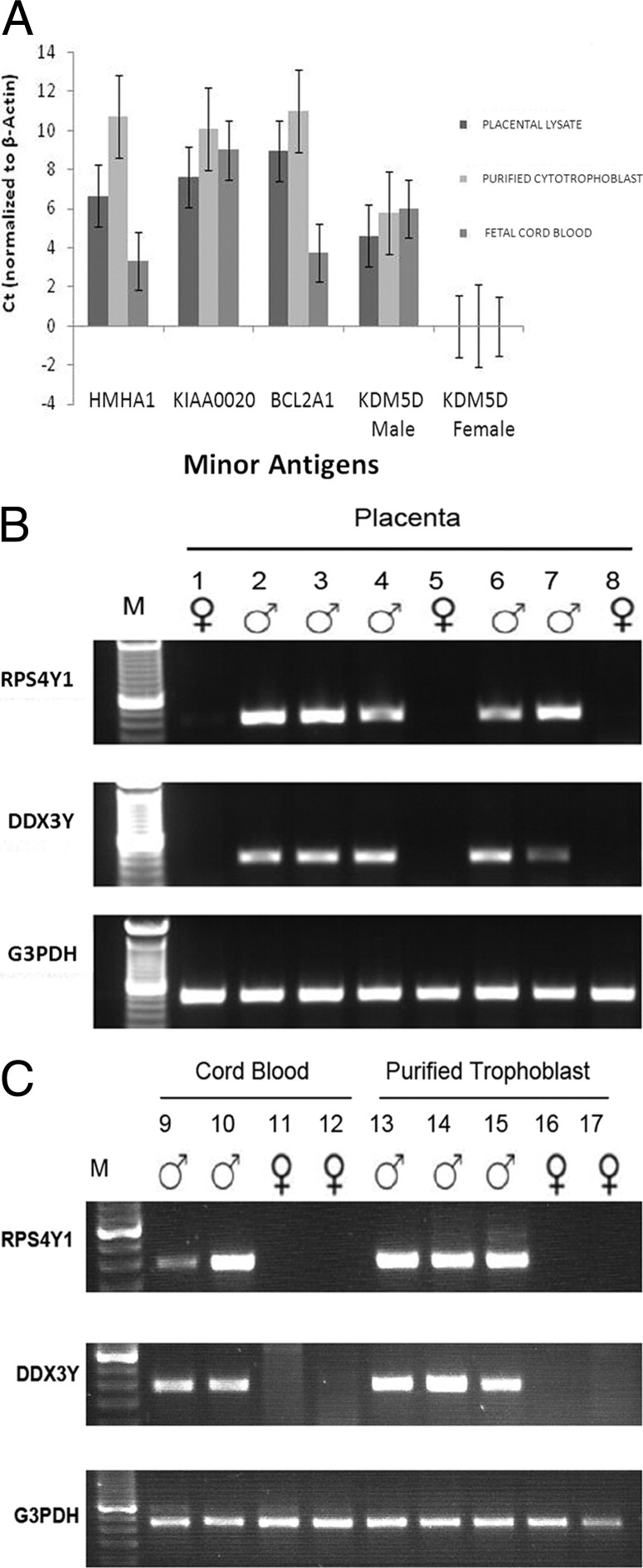
Minor antigen mRNA expression in placenta, cytotrophoblast cells, and fetal cord blood. A: Real-time RT-PCR for autosomally encoded mHAgs (HMHA1, KIAA0020, and BCL2A1) and the Y chromosome–encoded mHAg (KDM5D), for placental lysate and purified cell populations. Data are expressed as the mean ± SEM difference in CT values between the gene of interest and β-actin for the same samples, calculated as described in Materials and Methods. The average ± SEM β-actin values in each sample were as follows: 24.9 ± 0.59 (placenta), 21.4 ± 0.24 (cytotrophoblast), and 26.53 ± 1.56 (cord blood mononuclear cells). B and C: Conventional RT-PCR data for placental lysate and purified cells, respectively. Glyceraldehyde 3-phosphate dehydrogenase (G3PDH) was used as a control; the sex of the infant is indicated by symbols above each lane. CB, cord blood mononuclear cells; CT, cytotrophoblast cells; M, mol. wt. marker.
Analysis of mHAg Proteins in the Placenta
General Properties of mHAg Immunoreactivities in the Placenta
First-trimester, second-trimester, and term placental villi, as well as term extraplacental membranes, were examined by IHC using antibodies targeting the mHAg proteins HMHA1, KIAA0020, BCL2A1, and RPS4Y1. All four minor antigens could be observed in both villous and extravillous trophoblast (EVT) populations, with variation occurring within and between placental samples, across gestation, and according to antigen and cell type (Figure 2 and Figure 3). IHC for DDX3Y and KDM5D was attempted using commercially available antibodies, but failed to yield any positive staining under a variety of conditions, including positive control testis tissue.
Figure 2.
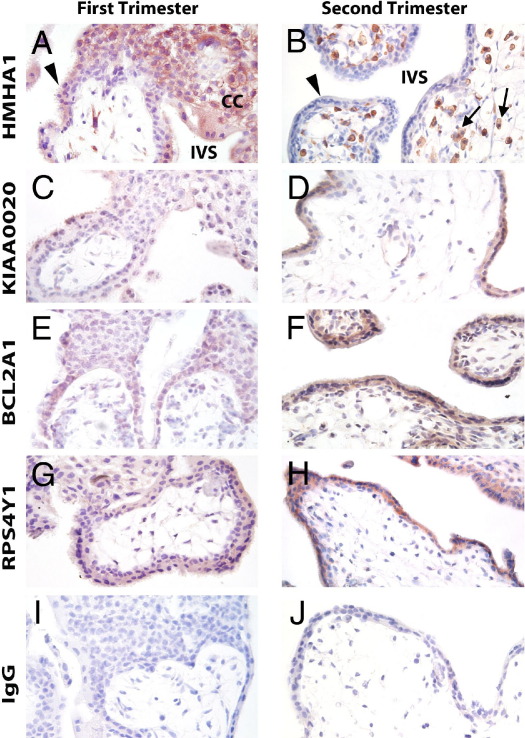
mHAg expression in first- and second-trimester placentas. A, C, E, G, and I: First-trimester placenta (gestational age, 8 to 12 weeks). B, D, F, H, and J: Second-trimester placenta (gestational age, 13 to 21 weeks). Reddish-brown staining represents specific immunoreactivity of mHAg-specific antibodies with cells; bluish-purple staining, hematoxylin counterstain. Arrowheads indicate syncytiotrophoblast; arrows, Hofbauer cells. IVS, intervillous space; CC, trophoblast cell column.
Figure 3.
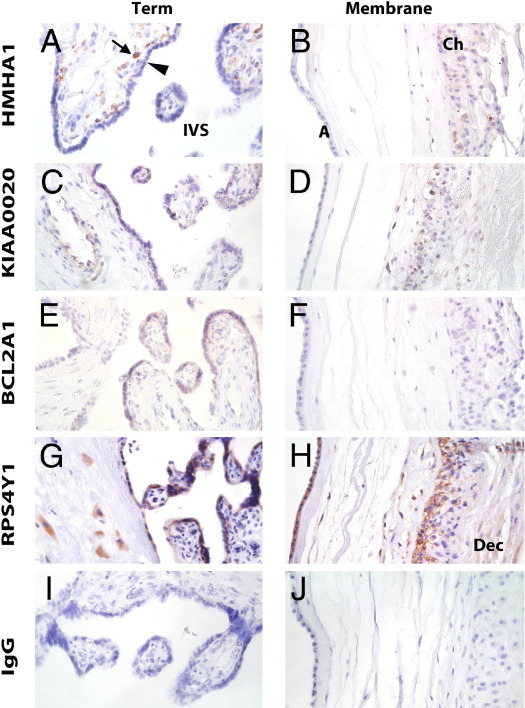
mHAg expression in term placentas and extraplacental membranes. A, C, E, G, and I: Term placenta. B, D, F, H, and J: Extraplacental membranes. Arrowhead indicates syncytiotrophoblast; arrow, Hofbauer cells. A, amnion membrane; Ch, chorion membrane; Dec, decidua; IVS, intervillous space.
HMHA1 and RPS4Y1 expression was cytoplasmic (Figure 2, A, B, G, and H, and Figure 3, A, B, G, and H). KIAA0020 and BCL2A1 were also usually seen in the cytoplasm, but this was frequently accompanied by nuclear expression (Figure 2, C–F, and Figure 3, C–F). The RPS4Y1 antibody stained trophoblast cells (all subpopulations) of both male and female placentas, because this antibody does not distinguish between the Y- and X-encoded isoforms of this protein. Expression observed using this antibody is, therefore, described as RPS4X/Y1 hereafter.
Villous Trophoblast Cells
In the syncytiotrophoblast, immunoreactivity for all four mHAgs examined was observed, although staining intensity for each antigen always varied, both within and between placentas. Immunoreactivity for BCL2A1 was generally stronger and more uniform in first- and second-trimester villous cytotrophoblasts and syncytiotrophoblasts than at term (Figure 2, E and F, and Figure 3E). However, in three of the eight term placentas examined, BCL2A1 was often concentrated or restricted to areas of aggregated syncytiotrophoblast nuclei, possibly representing syncytial knots before their shedding from the placenta.
KIAA0020 and RPS4X/Y1 proteins were observed in villous trophoblasts of all placentas throughout gestation. Immunoreactivity varied within and between placentas (Figure 2, C, D, G, and H, and Figure 3, C, D, and G). KIAA0020 was always found in the cytoplasm, and some first-trimester samples exhibited nuclear staining as well. This protein was often concentrated at the microvillous surface and displayed stronger immunolocalization in the syncytiotrophoblast than in the cytotrophoblast.
HMHA1 immunoreactivity in the villous trophoblast was only observed consistently in first-trimester samples. One of five second-trimester samples, and none of the term samples, expressed this protein in the syncytiotrophoblast (Figure 2, A and B, and Figure 3A). Within each individual first-trimester sample, trophoblast expression of HMHA1 was variable. In the first- and second-trimester samples, syncytial immunoreactivity was always stronger than cytotrophoblast staining.
EVT Data
The expression of all four mHAg proteins was frequently observed in EVTs. BCL2A1 expression was moderate in first-trimester (Figure 2E) and second-trimester (data not shown) trophoblastic columns, whereas at term, staining for BCL2A1 was weak or absent in the EVTs, basal plate, and chorion membrane (Figure 3F). KIAA0020, HMHA1, and RPS4Y1 antibodies consistently reacted with EVT cells throughout gestation; in the term chorion membrane, trophoblast cells proximal to the amnion exhibited stronger immunoreactivity for HMHA1 and RPS4Y1 than those distal to the amnion (Figure 3, B and H).
Fetal Leukocytes
Fetal leukocytes contained within the vasculature of the term placentas were generally negative for KIAA0020, BCL2A1, and RPS4X/Y1; however, a few weakly positive leukocytes were occasionally present in these samples. Fetal leukocytes in term placentas consistently and strongly expressed HMHA1 (data not shown).
Hofbauer and Mesenchymal Cells
Hofbauer cells, which are found within the villous mesenchyme and share lineage with macrophages, showed strong expression of HMHA1 throughout gestation (Figure 2, A and B, and Figure 3A). In the term placenta, stromal fibroblasts could also be seen to express HMHA1. BCL2A1, KIAA0020, and RPS4X/Y1 antibodies generally did not react with placental fibroblasts or Hofbauer cells, with the exception that most second-trimester placentas exhibited weak staining of both cell types.
Endothelial cells were consistently immunoreactive with the KIAA0020 antibody throughout gestation (Figure 2D and Figure 3C); with the exception of sporadic staining of these cells for RPS4X/Y1, endothelial cells generally lacked the other mHAg proteins investigated in this study.
Maternal Tissues
The expression of mHAgs in the decidua was determined by examining term basal plate and extraplacental membranes with their associated decidual tissue. HMHA1, RPS4X/Y1, and KIAA0020 were expressed by decidual cells, whereas BCL2A1 expression was weak or absent (Figure 3, B, D, F, H, and J). KIAA0020, BCL2A1, and RPS4X/Y1 were generally negative in maternal leukocytes associated with decidual tissue. Maternal leukocytes consistently expressed HMHA1 in both first- and third-trimester tissues. These cells could be observed both in peripheral blood located within the intervillous space and within decidual tissue associated with the chorion and basal plate.
Proteomic Analysis of the Syncytiotrophoblast and Trophoblast Debris
Proteomic analysis was performed on the syncytiotrophoblast and on the trophoblast debris shed from cultured first-trimester placental explants. A total of 1590 proteins were identified as being present in the syncytiotrophoblast, and 375 proteins were identified as being expressed in trophoblast debris, with a false-discovery rate of 0.0087. The identity of these proteins was confirmed by the characterization of at least two peptides from the protein sequences. Two autosomally encoded mHAgs (solute carrier 1A5 and signal sequence receptor 1) and two Y chromosome–encoded mHAgs (RPS4Y1 and DDX3Y) were found in the proteomic analysis. RPS4Y1, solute carrier 1A5, and signal sequence receptor 1 were identified in the syncytiotrophoblast, whereas DDX3Y was present in both the syncytiotrophoblast and the shed trophoblast debris (Table 2).
Table 2.
Antigenic Minor Histocompatibility Proteins Detected by MS in Shed Trophoblast Debris and in the Syncytiotrophoblast
| mHAg | Protein name | Gene | Trophoblast debris | Syncytiotrophoblast |
|---|---|---|---|---|
| DRB1/HY and DQ5/HY | ATP-dependent RNA helicase DDX3Y | DDX3Y | + | + |
| B52/HY and DRB3/HY | 40S ribosomal protein S4, Y isoform 1 | RPS4Y1 | − | + |
| SLC1A5 | Neutral amino acid transporter B(0) isoform 1 | SLC1A5 | − | + |
| LB-SSR1-1S | Translocon-associated protein subunit α | SSR1 | − | + |
| HA-1 | Minor histocompatibility protein HA-1 | HMHA1 | − | − |
| HA-8 | Pumilio domain-containing protein KIAA0020 | KIAA0020 | − | − |
| ACC-1 and ACC-2 | Bcl-2–related protein A1 | BCL2A1 | − | − |
| A2/HY and B7/HY | Lysine-specific demethylase 5D | KDM5D | − | − |
The protein name and gene symbol of each minor antigen are given.
KIAA0020, pumilio domain-containing protein KIAA0020; SLC1A5, solute carrier 1A5; SSR1, signal sequence receptor 1; +, found; −, not found.
Discussion
Multiparous women have previously been shown to possess expanded cohorts of T cells specific for fetal mHAgs.13–15 However, the source of the antigens that trigger this T-cell expansion has not been definitively identified. In this study, we identified the placenta, a fetal organ, as a probable source of foreign mHAgs to which the mother is exposed during pregnancy. By using RT-PCR, we demonstrated that mRNAs for the autosomal mHAgs (HMHA1, KIAA0020, and BCL2A1) and the Y chromosome–encoded mHAgs (KDM5D, DDX3Y, and RPS4Y1) are found in placental tissue and purified trophoblast cells. These results are consistent with those of Warren et al56 who, by tissue array, identified mRNA for another male mHAg, UTY, in human placental homogenate. IHC analysis revealed that four of the six mHAgs studied were expressed by several different placental cell types, including trophoblast subpopulations, mesenchymal cells, placental macrophages, and cord blood leukocytes. Importantly, all four mHAgs examined by IHC were expressed within the syncytiotrophoblast, which, for most of the pregnancy, is bathed directly in maternal blood. This then provides an open avenue for release of mHAg-containing shed material from the placenta into the blood and subsequent trafficking to maternal lymphoid organs, where an immune response might be generated. Indeed, we show that at least one antigen, DDX3Y, is present in trophoblast debris shed from first-trimester placenta.
Our findings of robust expression of fetal mHAgs in the placenta align with earlier work13–15,57 demonstrating that pregnancy results in the expansion of maternal minor antigen–specific CD8+ T cells. One antigen that elicits antigen-specific maternal immunoreactivity, HMHA1,16 was strongly expressed in the syncytiotrophoblast and EVTs, both of which are fetal cells to which the maternal immune system has direct contact. Protein expression studies of other Y chromosome–encoded antigens known to elicit maternal T-cell reactivity, such as KDM5D,13,15 are limited by antibody availability and specificity. Indeed, caution in interpretation of RPS4Y1 IHC studies is warranted because the peptide immunogen for the RPS4X/Y1 antibody derives from regions conserved between the male and female isoforms of this protein, thus rendering the antibody undiscerning between male and female placentas. Nonetheless, we have shown the presence of the male-specific isoform, RPS4Y1, by both RT-PCR and proteomic analyses in the trophoblast of male placentas.
The finding that mHAgs are expressed in the syncytiotrophoblast and in trophoblast debris shed from the placenta provides the first direct evidence that trophoblast and deported trophoblast debris contain proteins that are responsible for triggering cell-mediated immune responses. A single mHAg was found in proteomic analysis of trophoblast debris shed from the placenta. It seems likely that other mHAgs are also present in the debris, because MS proteomic analysis is only able to detect proteins that are present in relatively high abundance; some of the mHAgs may be expressed at a level too low to detect by this method. Current efforts are focused on identifying these antigens in trophoblast debris using more sensitive methods.
Much trophoblast debris is thought to be shed into the maternal blood daily in a normal pregnancy.29,30 This enormous amount of trophoblast debris is rapidly cleared from the maternal blood by an unknown mechanism, but it is likely that maternal phagocytes, such as macrophages, are at least partially responsible. Phagocytosis of mHAg-containing trophoblast debris by maternal antigen-presenting cells would likely produce some form of maternal adaptive immune response. Additionally, the context in which the adaptive immune system detects antigen is critical in determining the nature of the ensuing response. Antigens produce tolerogenic immune responses when they are presented from apoptotic cells.58 Because trophoblast debris is thought to be shed from the placenta via apoptosis,28,52 it seems likely that DDX3Y and other mHAgs and/or HLAs36 in deported trophoblast debris would lead to a tolerogenic immune response that may be important in promoting the survival of placental allografts that express this protein. On the other hand, an excessive antigenic load and the association of antigen with necrotic cellular debris can incite inflammatory responses. The quantity of shed trophoblast cells and microparticles is increased in preeclamptic pregnancies,33,34 and reduced/fluctuating oxygen concentrations associated with preeclampsia may drive syncytial fragments in the process of deportation toward a necrotic, rather than an apoptotic, phenotype.59 It follows, then, that the presence of mHAgs in high amounts of necrotic trophoblast debris may stimulate an active immune response to the placenta/fetus, which might occur in some cases of recurrent miscarriage or other pathological conditions of pregnancy.
An important consideration is the mechanism by which antigens derived from trafficking placental cells or debris might be presented by antigen-presenting cells. Little is known about the form in which fetal antigen may promote maternal lymphocyte expansion. In a mouse model of paternally inherited fetal antigen, the indirect pathway of antigen presentation occurs whereby maternal (but not fetal) antigen-presenting cells cross present fetal antigen to MHC class I–restricted maternal CD8+ T cells.10,11 Cross presentation can result in tolerance or priming of T cells, and occurs as a result of phagocytosis of apoptotic or necrotic cellular material by dendritic cells, with antigen processing occurring either before or after transfer to the recipient antigen-presenting cell.60 Phagocytosis of apoptotic and necrotic human-deported trophoblasts by macrophages has been recapitulated in vitro.25 Intriguingly, the physiological state of the phagocytosed trophoblast material had a strong influence on the phenotype of the antigen-presenting cell; this may, in turn, translate into different functional outcomes of cognate interaction with lymphocytes. Such outcomes and their consequences in human pregnancy remain to be investigated.
The lymphocyte responses arising from fetal mHAgs may include generation of fetal antigen-specific regulatory T cells. van Halteren et al16 demonstrated the presence of putative regulatory CD8+ T cells in parous women, including those specific for SMCY and HMHA1, antigens that were identified in trophoblasts in this study. Interestingly, results using the abortion model of mating CBA/J female mice to DBA/2J males suggested that paternal minor antigen-specific CD4+ regulatory T cells could be protective against maternal anti-fetal responses.4 Thus, it is possible that antigen-specific regulatory-type cells are critical for conferring tolerance to these antigens.
It is likely that tolerance to paternally inherited antigens is induced at the time of first exposure to that antigen. From this study, however, it is not possible to discern the point at which women are first introduced to each mHAg. Studies5,11,61 in mice using model antigens suggest that if antigen is present, tolerance to that antigen can be established as early as insemination and, furthermore, that this early exposure is critical to pregnancy success. KDM5D protein on approximately 50% of ejaculated sperm has been reported,62 raising the possibility that this antigen is introduced to the maternal immune system at copulation and that tolerance to KDM5D could be established at that point. On the other hand, neither DDX3Y nor RPS4Y1 could be found on spermatozoa, despite their expression at the level of mRNA62; it remains possible that these antigens are present in seminal fluid or, alternatively, that tolerance to these antigens occurs at some later time point. Other work63–65 suggested that undefined Y-encoded mHAgs are expressed on male preimplantation embryos of several species, although expression decreased at the blastocyst stage and was absent on trophectoderm. However, because of the lack of lymphatic drainage of the human decidua,66 mHAg expression on preimplantation or early postimplantation embryos may go undetected. Taken together, the initial events of antigenic exposure and tolerance induction likely vary according to the spatial and temporal regulation of individual antigens. The route by which paternally inherited antigen is exposed to maternal lymphoid organs probably also depends on when and where the antigen is expressed. If first expression is on the placenta itself, the route is likely hematogenous; if onset of expression occurs in seminal fluid or on spermatozoa, exposure likely occurs via lymphatic drainage.
Another mechanism distinct from trophoblast shedding that could lead to maternal alloimmunization to fetal antigen is microchimerism. Herein, fetal progenitor cells actively traffic into the maternal blood and tissues, where they can be harbored for decades after childbirth.23 Blood cells from the fetus may be the source of these cells,20,21,67,68 although the mechanism by which fetal cells traffic into the maternal circulation is unknown. The presence of detectable mHAgs within fetal leukocytes is suggestive that microchimerism can be a mechanism of maternal alloimmunization to these antigens, as has been suggested by others.16 Indeed, some mothers possessing T cells directed against their children's mHAgs have possessed significant fetal microchimerism.14,21 Yet, another potential mechanism that could result in maternal exposure to fetal blood antigens is leakage into the maternal circulation during placental hemorrhage before or during parturition. Indeed, this occurs with isoimmunization of rhesus antigen–negative women carrying rhesus-positive fetuses.
Maternal alloimmunization by fetal antigens during pregnancy is thought to have important clinical implications for organ and stem cell transplantation, subsequent pregnancies, and autoimmunity. Donor parity has been identified as a risk factor for acute and chronic graft-versus-host disease in patients undergoing hematopoietic stem cell transplantation,17,18 and recipients of parous female hematopoietic stem cell transplantation have both higher rates of graft-versus-host disease and graft-versus-leukemia effects.6,69–71 Researchers hypothesize that mHAg-specific T cells, which had resulted from exposure of fetal mHAgs to the maternal compartment during pregnancy, are provided by the female donor. The long-term impact of pregnancy-induced maternal sensitization is demonstrated by the findings that mHAg-specific T cells may persist up to 22 years after pregnancy.6,15,72
Secondary recurrent miscarriage is defined as three or more consecutive miscarriages after a birth at 24 weeks' gestation or later. Two independent research groups have found a significant association between the development of secondary recurrent miscarriage and prior birth of a male infant. This may be due, at least in part, to an aberrant maternal immune response to fetal Y chromosome–encoded antigens.73,74 In addition, maternal carriage of HY-restricting MHC class II alleles is associated with poor pregnancy outcomes in women with a prior live birth of a male infant and a diagnosis of secondary recurrent miscarriage.75 Our data indicate that maternal exposure to Y chromosome–encoded fetal antigens is likely to occur during pregnancy, at least in part as a result of shedding and deportation of trophoblast debris into the maternal blood. Therefore, aberrant responses to mHAgs may contribute to inappropriate adaptive maternal immune responses to the placenta/fetus.
Finally, our results have implications for the relationship between fetal microchimerism and maternal autoimmune disease, in that they further the possibility of differential antigen recognition by the maternal immune system and/or engrafted fetal cells. Nelson et al76 and other researchers77–79 noticed an increase in the prevalence of male DNA of fetal origin within blood and skin lesions of women with systemic sclerosis. The relationship between microchimerism and systemic sclerosis has not been definitively elucidated, but possibilities include mutual recognition of minor and major histocompatibility antigens by maternal cells or engrafted fetal cells.80,81 Because placental deportation may be a mechanism of priming toward fetal antigen, the environmental context during pregnancy may determine future reactivity toward antigen on re-exposure in various states, including reactivity to microchimeric fetal cells, future pregnancy, and transplanted organs and cells.
In conclusion, we have demonstrated that paternally inherited fetal mHAgs are expressed in the fetal blood, the syncytiotrophoblast of the placenta, and trophoblast debris shed from the placenta. This finding provides support for the hypothesis that mHAg-specific T cells are generated during pregnancy as the result of both trophoblast deportation from the placenta into the maternal blood and fetal microchimerism. Importantly, these results refute the concept that the trophoblast is antigenically inert. Rather, the trophoblast is a source of fetal antigen that is likely to tolerize the maternal adaptive immune system in normal pregnancy, and may stimulate adverse responses by maternal immune cells in diseases of pregnancy and complications of transplantation.
Acknowledgments
We thank Sarika Kshirsagar, Sheikh Alam Khorshed, and Antoine Perchellet for assistance with this project; Kristina Adams Waldorf for critically reading the manuscript; the staff and patients of the Center for Women's Health (Overland Park, KS); the University of Chicago Department of Obstetrics and Gynecology; the Epsom Day Unit, Greenlane Clinical Centre (Auckland, New Zealand); and the Auckland Medical Aid Clinic (Auckland, New Zealand).
Footnotes
Supported by the Foundation for Research, Science and Technology, New Zealand (L.W.C.) and grants from the NIH (R01HD045611 to M.G.P. and P01049480 to J.S. Hunt/C. Ober, directors of Tissue Collection Core). Further resources and services were provided by the University of Kansas Medical Center (KUMC) Center for Reproductive Sciences (Eunice Kennedy Shriver National Institute of Child Health & Human Development), the KUMC Center of Biomedical Research Excellence (COBRE) program in Cell Development and Differentiation (P20RR024214), the Kansas State University COBRE program in Epithelial Function (P20RR017686), and the Kansas IDeA Network of Biomedical Research Infrastructure (P20 RR016475).
O.J.H. and C.L. contributed equally to this work.
References
- 1.Petroff M.G. Immune interactions at the maternal-fetal interface. J Reprod Immunol. 2005;68:1–13. doi: 10.1016/j.jri.2005.08.003. [DOI] [PubMed] [Google Scholar]
- 2.Aluvihare V.R., Kallikourdis M., Betz A.G. Regulatory T cells mediate maternal tolerance to the fetus. Nat Immunol. 2004;5:266–271. doi: 10.1038/ni1037. [DOI] [PubMed] [Google Scholar]
- 3.Darrasse-Jeze G., Klatzmann D., Charlotte F., Salomon B.L., Cohen J.L. CD4+CD25+ regulatory/suppressor T cells prevent allogeneic fetus rejection in mice. Immunol Lett. 2006;102:106–109. doi: 10.1016/j.imlet.2005.07.002. [DOI] [PubMed] [Google Scholar]
- 4.Schumacher A., Wafula P.O., Bertoja A.Z., Sollwedel A., Thuere C., Wollenberg I., Yagita H., Volk H.-D., Zenclussen A.C. Mechanisms of action of regulatory T cells specific for paternal antigens during pregnancy. Obstet Gynecol. 2007;110:1137–1145. doi: 10.1097/01.AOG.0000284625.10175.31. [DOI] [PubMed] [Google Scholar]
- 5.Robertson S.A., Guerin L.R., Bromfield J.J., Branson K.M., Ahlström A.C., Care A.S. Seminal fluid drives expansion of the CD4+CD25+ T regulatory cell pool and induces tolerance to paternal alloantigens in mice. Biol Reprod. 2009;80:1036–1045. doi: 10.1095/biolreprod.108.074658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Goulmy E. Minor histocompatibility antigens: from transplantation problems to therapy of cancer. Human Immunol. 2006;67:433–438. doi: 10.1016/j.humimm.2006.03.012. [DOI] [PubMed] [Google Scholar]
- 7.Goulmy E., Gratama J.W., Blokland E., Zwaan F.E., van Rood J.J. A minor transplantation antigen detected by MHC-restricted cytotoxic T lymphocytes during graft-versus-host disease. Nature. 1983;302:159–161. doi: 10.1038/302159a0. [DOI] [PubMed] [Google Scholar]
- 8.Goulmy E., Schipper R., Pool J., Blokland E., Falkenburg J.H., Vossen J., Gratwohl A., Vogelsang G.B., van Houwelingen H.C., van Rood J.J. Mismatches of minor histocompatibility antigens between HLA-identical donors and recipients and the development of graft-versus-host disease after bone marrow transplantation. N Engl J Med. 1996;334:281–285. doi: 10.1056/NEJM199602013340501. [DOI] [PubMed] [Google Scholar]
- 9.Goulmy E., Termijtelen A., Bradley B.A., van Rood J.J. Alloimmunity to human H-Y. Lancet. 1976;2:1206. doi: 10.1016/s0140-6736(76)91727-x. [DOI] [PubMed] [Google Scholar]
- 10.Erlebacher A., Vencato D., Price K.A., Zhang D., Glimcher L.H. Constraints in antigen presentation severely restrict T cell recognition of the allogeneic fetus. J Clin Invest. 2007;117:1399–1411. doi: 10.1172/JCI28214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Moldenhauer L.M., Diener K.R., Thring D.M., Brown M.P., Hayball J.D., Robertson S.A. Cross-presentation of male seminal fluid antigens elicits T cell activation to initiate the female immune response to pregnancy. J Immunol. 2009;182:8080–8093. doi: 10.4049/jimmunol.0804018. [DOI] [PubMed] [Google Scholar]
- 12.Tagalauer E.S., Yankee T.M., Petroff M.G. Maternal PD-1 regulates accumulation of fetal antigen-specific CD8+ T cells in pregnancy. J Reprod Immunol. 2009;80:12–21. doi: 10.1016/j.jri.2008.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.James E., Chai J.G., Dewchand H., Macchiarulo E., Dazzi F., Simpson E. Multiparity induces priming to male-specific minor histocompatibility antigen, HY, in mice and humans. Blood. 2003;102:388–393. doi: 10.1182/blood-2002-10-3170. [DOI] [PubMed] [Google Scholar]
- 14.Verdijk R.M., Kloosterman A., Pool J., van de Keur M., Naipal A.M., van Halteren A.G., Brand A., Mutis T., Goulmy E. Pregnancy induces minor histocompatibility antigen-specific cytotoxic T cells: implications for stem cell transplantation and immunotherapy. Blood. 2004;103:1961–1964. doi: 10.1182/blood-2003-05-1625. [DOI] [PubMed] [Google Scholar]
- 15.Piper K.P., McLarnon A., Arrazi J., Horlock C., Ainsworth J., Kilby M.D., Martin W.L., Moss P.A. Functional HY-specific CD8+ T cells are found in a high proportion of women following pregnancy with a male fetus. Biol Reprod. 2007;76:96–101. doi: 10.1095/biolreprod.106.055426. [DOI] [PubMed] [Google Scholar]
- 16.van Halteren A.G., Jankowska-Gan E., Joosten A., Blokland E., Pool J., Brand A., Burlingham W.J., Goulmy E. Naturally acquired tolerance and sensitization to minor histocompatibility antigens in healthy family members. Blood. 2009;114:2263–2272. doi: 10.1182/blood-2009-01-200410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Flowers M.E., Pepe M.S., Longton G., Doney K.C., Monroe D., Wietherspoon R.P., Sullivan K.M., Storb R. Previous donor pregnancy as a risk factor for acute graft-versus-host disease in patients with aplastic anaemia treated by allogeneic marrow transplantation. Br J Haematol. 1990;74:492–496. doi: 10.1111/j.1365-2141.1990.tb06340.x. [DOI] [PubMed] [Google Scholar]
- 18.Kollman C., Howe C.W., Anasetti C., Antin J.H., Davies S.M., Filipovich A.H., Hegland J., Kamani N., Kernan N.A., King R., Ratanatharathorn V., Weisdorf D., Confer D.L. Donor characteristics as risk factors in recipients after transplantation of bone marrow from unrelated donors: the effect of donor age. Blood. 2001;98:2043–2051. doi: 10.1182/blood.v98.7.2043. [DOI] [PubMed] [Google Scholar]
- 19.Adams K.M., Holmberg L.A., Leisenring W., Guthrie K.A., Tylee T.S., Nelson J.L. Risk factors for syngeneic graft-versus-host disease in adult hematopoietic stem cell transplants. Blood. 2004;104:1894–1897. doi: 10.1182/blood-2004-02-0508. [DOI] [PubMed] [Google Scholar]
- 20.Adams K.M., Lambert N.C., Heimfeld S., Tylee T.S., Pang J.M., Erickson T.D., Nelson J.L. Male DNA in female donor apheresis and CD34-enriched products. Blood. 2003;102:3845–3847. doi: 10.1182/blood-2003-05-1570. [DOI] [PubMed] [Google Scholar]
- 21.Evans P.C., Lambert N., Maloney S., Furst D.E., Moore J.M., Nelson J.L. Long-term fetal microchimerism in peripheral blood mononuclear cell subsets in healthy women and women with scleroderma. Blood. 1999;93:2033–2037. [PubMed] [Google Scholar]
- 22.Khosrotehrani K., Bianchi D.W. Mult-lineage potential of fetal cells in maternal tissue: a legacy in reverse. J Cell Sci. 2005;118(Pt 8):1558–1563. doi: 10.1242/jcs.02332. [DOI] [PubMed] [Google Scholar]
- 23.Nelson J.L. Your cells are my cells. Sci Am. 2008;298:72–79. [Google Scholar]
- 24.Petroff M.G. Review: fetal antigens–identity, origins, and influences on the maternal immune system. Placenta. 2011;32(Suppl 2):S176–S181. doi: 10.1016/j.placenta.2010.12.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Chamley L.W., Chen Q., Ding J., Stone P.R., Abumaree M. Trophoblast deportation: just a waste disposal system or antigen sharing? J Reprod Immunol. 2011;88:99–105. doi: 10.1016/j.jri.2011.01.002. [DOI] [PubMed] [Google Scholar]
- 26.Taylor D.D., Akyol S., Gercel-Taylor C. Pregnancy-associated exosomes and their modulation of T cell signaling. J Immunol. 2006;176:1534–1542. doi: 10.4049/jimmunol.176.3.1534. [DOI] [PubMed] [Google Scholar]
- 27.Redman C.W., Sargent I.L. Microparticles and immunomodulation in pregnancy and pre-eclampsia. J Reprod Immunol. 2007;76:61–67. doi: 10.1016/j.jri.2007.03.008. [DOI] [PubMed] [Google Scholar]
- 28.Burton G.J., Jones C.J. Syncytial knots, sprouts, apoptosis, and trophoblast deportation from the human placenta. Taiwan J Obstet Gynecol. 2009;48:28–37. doi: 10.1016/S1028-4559(09)60032-2. [DOI] [PubMed] [Google Scholar]
- 29.Huppertz B., Frank H.-G., Kingdom J.C.P., Reister F., Kaufmann P. Villous cytotrophoblast regulation of the syncytial apoptotic cascade in the human placenta. Histochem Cell Biol. 1998;110:495–508. doi: 10.1007/s004180050311. [DOI] [PubMed] [Google Scholar]
- 30.Huppertz B., Kaufmann P., Kingdom J. Trophoblast turnover in health and disease. Fetal Maternal Med Rev. 2002;13:103–118. [Google Scholar]
- 31.Attwood H.A., Park W.W. Embolism to the lungs by trophoblast. J Obstet Gynaecol Br Comm. 1961;68:611–617. doi: 10.1111/j.1471-0528.1961.tb02778.x. [DOI] [PubMed] [Google Scholar]
- 32.Ilke F.A. Dissemination of syncytiotrophoblastic cells in the maternal blood stream during pregnancy. Bull Schweiz Akad Med. 1964;20:62–72. [PubMed] [Google Scholar]
- 33.Chua S., Wilkins T., Sargent I., Redman C. Trophoblast deportation in pre-eclamptic pregnancy. Br J Obstet Gynaecol. 1991;98:973–979. doi: 10.1111/j.1471-0528.1991.tb15334.x. [DOI] [PubMed] [Google Scholar]
- 34.Knight M., Redman C.W., Linton E.A., Sargent I.L. Shedding of syncytiotrophoblast microvilli into the maternal circulation in pre-eclamptic pregnancies. Br J Obstet Gynaecol. 1998;105:632–640. doi: 10.1111/j.1471-0528.1998.tb10178.x. [DOI] [PubMed] [Google Scholar]
- 35.Jiang S.P., Vacchio M.S. Multiple mechanisms of peripheral T cell tolerance to the fetal “allograft.”. J Immunol. 1998;160:3086–3090. [PubMed] [Google Scholar]
- 36.Adams Waldorf K.M., Yan Z., Stevens A.M., Nelson J.L. The changing maternal “self” hypothesis: a mechanism for maternal tolerance of the fetus. Placenta. 2007;28:378–382. doi: 10.1016/j.placenta.2006.07.003. [DOI] [PubMed] [Google Scholar]
- 37.Taglauer E.S., Adams Waldorf K.M., Petroff M.G. The hidden maternal-fetal interface: events involving the lymphoid organs in maternal-fetal tolerance. Int J Dev Biol. 2010;54:421–430. doi: 10.1387/ijdb.082800et. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.den Haan J.M., Meadows L.M., Wang W., Pool J., Blokland E., Bishop T.L., Reinhardus C., Shabanowitz J., Offringa R., Hunt D.F., Engelhard V.H., Goulmy E. The minor histocompatibility antigen HA-1: a diallelic gene with a single amino acid polymorphism. Science. 1998;279:1054–1057. doi: 10.1126/science.279.5353.1054. [DOI] [PubMed] [Google Scholar]
- 39.Mommaas B., Kamp J., Drijfhout J.W., Beekman N., Ossendorp F., Van Veelen P., Den Haan J., Goulmy E., Mutis T. Identification of a novel HLA-B60-restricted T cell epitope of the minor histocompatibility antigen HA-1 locus. J Immunol. 2002;169:3131–3136. doi: 10.4049/jimmunol.169.6.3131. [DOI] [PubMed] [Google Scholar]
- 40.Wilke M., Pool J., den Haan J.M., Goulmy E. Genomic identification of the minor histocompatibility antigen HA-1 locus by allele-specific PCR. Tissue Antigens. 1998;52:312–317. doi: 10.1111/j.1399-0039.1998.tb03053.x. [DOI] [PubMed] [Google Scholar]
- 41.Brickner A.G., Warren E.H., Caldwell J.A., Akatsuka Y., Golovina T.N., Zarling A.L., Shabanowitz J., Eisenlohr L.C., Hunt D.F., Engelhard V.H., Riddell S.R. The immunogenicity of a new human minor histocompatibility antigen results from differential antigen processing. J Exp Med. 2001;193:195–206. doi: 10.1084/jem.193.2.195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Akatsuka Y., Nishida T., Kondo E., Miyazaki M., Taji H., Iida H., Tsujimura K., Yazaki M., Naoe T., Morishima Y., Kodera Y., Kuzushima K., Takahashi T. Identification of a polymorphic gene, BCL2A1, encoding two novel hematopoietic lineage-specific minor histocompatibility antigens. J Exp Med. 2003;197:1489–1500. doi: 10.1084/jem.20021925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kamei M., Nannya Y., Torikai H., Kawase T., Taura K., Inamoto Y., Takahashi T., Yazaki M., Morishima S., Tsujimura K., Miyamura K., Ito T., Togari H., Riddell S.R., Kodera Y., Morishima Y., Takahashi T., Kuzushima K., Ogawa S., Akatsuka Y. HapMap scanning of novel human minor histocompatibility antigens. Blood. 2009;113:5041–5048. doi: 10.1182/blood-2008-07-171678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Wu C., Orozco C., Boyer J., Leglise M., Goodale J., Batalov S., Hodge C.L., Haase J., Janes J., Huss J.W., Su A.I. BioGPS: an extensible and customizable portal for querying and organizing gene annotation resources. Genome Biol. 2009;10:R130. doi: 10.1186/gb-2009-10-11-r130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Van Bergen C.A., Rutten C.E., Van Der Meijden E.D., Van Luxemburg-Heijs S.A., Lurvink E.G., Houwing-Duistermaat J.J., Kester M.G., Mulder A., Willemze R., Falkenburg J.H., Griffioen M. High-throughput characterization of 10 new minor histocompatibility antigens by whole genome association scanning. Cancer Res. 2010;70:9073–9083. doi: 10.1158/0008-5472.CAN-10-1832. [DOI] [PubMed] [Google Scholar]
- 46.Wang W., Meadows L.R., den Haan J.M., Sherman N.E., Chen Y., Blokland E., Shabanowitz J., Agulnik A.I., Hendrickson R.C., Bishop C.E., Hunt D.F., Goulmy E., Engelhard V.H. Human H-Y: a male-specific histocompatibility antigen derived from the SMCY protein. Science. 1995;269:1588–1590. doi: 10.1126/science.7667640. [DOI] [PubMed] [Google Scholar]
- 47.Spierings E., Vermeulen C.J., Vogt M.H., Doerner L.E.E., Falkenburg J.H.F., Mutis T., Goulmy E. Identification of HLA class II-restricted H-Y-specific T-helper epitope evoking CD4+ T-helper cells in H-Y-mismatched transplantation. Lancet. 2003;362:610–615. doi: 10.1016/S0140-6736(03)14191-8. [DOI] [PubMed] [Google Scholar]
- 48.Ivanov R., Aarts T., Hol S., Doornenbal A., Hagenbeek A., Petersen E., Ebeling S. Identification of a 40S ribosomal protein S4–derived H-Y epitope able to elicit a lymphoblast-specific cytotoxic T lymphocyte response. Clin Cancer Res. 2005;11:1694–1703. doi: 10.1158/1078-0432.CCR-04-1772. [DOI] [PubMed] [Google Scholar]
- 49.Rosinski K.V., Fujii N., Mito J.K., Koo K.K., Xuereb S.M., Sala-Torra O., Gibbs J.S., Radich J.P., Akatsuka Y., Van den Eynde B.J., Riddell S.R., Warren E.H. DDX3Y encodes a class I MHC-restricted H-Y antigen that is expressed in leukemic stem cells. Blood. 2008;111:4817–4826. doi: 10.1182/blood-2007-06-096313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Holets L.M., Hunt J.S., Petroff M.G. Trophoblast CD274 (B7-H1) is differentially expressed across gestation: influence of oxygen concentration. Biol Reprod. 2006;74:352–358. doi: 10.1095/biolreprod.105.046581. [DOI] [PubMed] [Google Scholar]
- 51.Petroff M.G., Phillips T.A., Ka H., Pace J.L., Hunt J.S. Isolation and culture of term human trophoblast cells. Methods Mol Med. 2006;121:203–217. doi: 10.1385/1-59259-983-4:201. [DOI] [PubMed] [Google Scholar]
- 52.Abumaree M.H., Stone P.R., Chamley L.W. An in vitro model of human placental trophoblast deportation/shedding. Mol Hum Reprod. 2006;12:687–694. doi: 10.1093/molehr/gal073. [DOI] [PubMed] [Google Scholar]
- 53.Robinson J.M., Ackerman W.E., 4th, Tewari A.K., Kniss D.A., Vandre D.D. Isolation of highly enriched apical plasma membranes of the placental syncytiotrophoblast. Anal Biochem. 2009;387:87–94. doi: 10.1016/j.ab.2009.01.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Dickerson H.M., Clark T.G., Findly R.C. Ichthyophthirius multifiliis has membrane-associated immobilization antigens. J Protozool. 1989;36:159–164. doi: 10.1111/j.1550-7408.1989.tb01065.x. [DOI] [PubMed] [Google Scholar]
- 55.Shevchenko A., Jensen O.N., Podtelejnikov A.V., Sagliocco F., Wilm M., Vorm O., Mortensen P., Shevchenko A., Boucherie H., Mann M. Linking genome and proteome by mass spectrometry: large-scale identification of yeast proteins from two dimensional gels. Proc Natl Acad Sci U S A. 1996;93:14440–14445. doi: 10.1073/pnas.93.25.14440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Warren E.H., Gavin M.A., Simpson E., Chandler P., Page D.C., Disteche C., Stankey K.A., Greenberg P.D., Riddell S.R. The human UTY gene encodes a novel HLA-B8-restricted H-Y antigen. J Immunol. 2000;164:2807–2814. doi: 10.4049/jimmunol.164.5.2807. [DOI] [PubMed] [Google Scholar]
- 57.Burlingham W.J., Goulmy E. Human CD8+ T-regulatory cells with low-avidity T-cell receptor specific for minor histocompatibility antigens. Hum Immunol. 2008;69:728–731. doi: 10.1016/j.humimm.2008.08.289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Gregory C.D., Pound J.D. Cell death in the neighbourhood: direct microenvironmental effects of apoptosis in normal and neoplastic tissues. J Pathol. 2011;223:178–195. doi: 10.1002/path.2792. [DOI] [PubMed] [Google Scholar]
- 59.Huppertz B., Kingdom J., Caniggia I., Desoye G., Black S., Korr H., Kaufman P. Hypoxia favors necrotic versus apoptotic shedding of placental syncytiotrophoblast into the maternal circulation. Placenta. 2003;24:181–190. doi: 10.1053/plac.2002.0903. [DOI] [PubMed] [Google Scholar]
- 60.Blachere N.E., Darnell R.B., Albert M.L. Apoptotic cells deliver antigen to dendritic cells for cross-presentation. PLoS Biol. 2005;6:1070–1078. doi: 10.1371/journal.pbio.0030185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Shima T., Sasaki Y., Itoh M., Nakashima A., Ishii N., Sugamura K., Saito S. Regulatory T cells are necessary for implantation and maintenance of early pregnancy but not late pregnancy in allogeneic mice. J Reprod Immunol. 2010;85:121–129. doi: 10.1016/j.jri.2010.02.006. [DOI] [PubMed] [Google Scholar]
- 62.Yao C., Wang Z., Zhou Y., Xu W., Li Q., Ma D., Wang L., Qiao Z. A study of Y chromosome gene mRNA in human ejaculated spermatozoa. Mol Reprod Dev. 2010;77:158–166. doi: 10.1002/mrd.21116. [DOI] [PubMed] [Google Scholar]
- 63.Krco C.J., Goldberg E.H. H-Y (male) antigen: detection on eight-cell mouse embryos. Science. 1976;193:1134–1135. doi: 10.1126/science.959826. [DOI] [PubMed] [Google Scholar]
- 64.White K.L., Anderson G.B., Pashen R.L., BonDurant R.H. Detection of histocompatibility-Y antigen: identification of sex of pre-implantation ovine embryos. J Reprod Immunol. 1987;10:27–32. doi: 10.1016/0165-0378(87)90047-7. [DOI] [PubMed] [Google Scholar]
- 65.Hossepian de Lima V.F.M., Moreira-Filho C.A., De Bem A.R., Jorge W. Sex determination of murine and bovine embryos using cytotoxicity and immunofluorescence assays. Theriogenology. 1993;39:1343–1352. [Google Scholar]
- 66.Volchek M., Girling J.E., Lash G.E., Cann L., Kumar B., Robson S.C., Bulmer J.N., Rogers P.A. Lymphatics in the human endometrium disappear during decidualization. Hum Reprod. 2010;25:2455–2464. doi: 10.1093/humrep/deq224. [DOI] [PubMed] [Google Scholar]
- 67.Bianchi D.W. Fetal cells in the maternal circulation: feasibility for prenatal diagnosis. Br J Haematol. 1999;105:574–583. doi: 10.1046/j.1365-2141.1999.01383.x. [DOI] [PubMed] [Google Scholar]
- 68.Osada H., Doi S., Fukushima T., Nakauchi H., Seki K., Sekiya S. Detection of fetal HPCs in maternal circulation after delivery. Transfusion. 2001;41:499–503. doi: 10.1046/j.1537-2995.2001.41040499.x. [DOI] [PubMed] [Google Scholar]
- 69.Randolph S.S., Gooley T.A., Warren E.H., Appelbaum F.R., Riddell S.R. Female donors contribute to a selective graft-versus-leukemia effect in male recipients of HLA-matched, related hematopoietic stem cell transplants. Blood. 2004;103:347–352. doi: 10.1182/blood-2003-07-2603. [DOI] [PubMed] [Google Scholar]
- 70.Loren A.W., Bunin G.R., Boudreau C., Champlin R.E., Cnaan A., Horowitz M.M., Loberiza F.R., Porter D.L. Impact of donor and recipient sex and parity on outcomes of HLA-identical sibling allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant. 2006;12:758–769. doi: 10.1016/j.bbmt.2006.03.015. [DOI] [PubMed] [Google Scholar]
- 71.Takami A., Sugimori C., Feng X., Yachie A., Kondo Y., Nishimura R., Kuzushima K., Kotani T., Asakura H., Shiobara S., Nakao S. Expansion and activation of minor histocompatibility antigen HY-specific T cells associated with graft-versus-leukemia response. Bone Marrow Transplant. 2004;34:703–709. doi: 10.1038/sj.bmt.1704583. [DOI] [PubMed] [Google Scholar]
- 72.Verdjik R.M., Kloosterman A., Pool J., van de Keur M., Naipal A.M., van Halteren A.G., Brand A., Mutis T., Goulmy E. Pregnancy induces minor histocompatibility antigen-specific cytotoxic T cells: implications for stem cell transplantation and immunotherapy. Blood. 2004;103:1961–1964. doi: 10.1182/blood-2003-05-1625. [DOI] [PubMed] [Google Scholar]
- 73.Christiansen O.B., Steffensen R., Nielsen H.S. The impact of anti-HY responses on outcome in current and subsequent pregnancies of patients with recurrent pregnancy losses. J Reprod Immunol. 2010;85:9–14. doi: 10.1016/j.jri.2009.12.008. [DOI] [PubMed] [Google Scholar]
- 74.Ooi P.V., Russell N., O'Donoghue K. Secondary recurrent miscarriage is associated with previous male birth. J Reprod Immunol. 2011;88:38–41. doi: 10.1016/j.jri.2010.10.004. [DOI] [PubMed] [Google Scholar]
- 75.Nielsen H.S., Steffensen R., Varming K., Van Halteren A.G., Spierings E., Ryder L.P., Goulmy E., Christiansen O.B. Association of HY-restricting HLA class II alleles with pregnancy outcome in patients with recurrent miscarriage subsequent to a firstborn boy. Hum Mol Genet. 2009;18:1684–1691. doi: 10.1093/hmg/ddp077. [DOI] [PubMed] [Google Scholar]
- 76.Nelson J.L., Furst D.E., Maloney S., Gooley T., Evans P.C., Smith A., Bean M.A., Ober C., Bianchi D.W. Microchimerism and HLA-compatible relationships of pregnancy in scleroderma. Lancet. 1998;351:559–562. doi: 10.1016/S0140-6736(97)08357-8. [DOI] [PubMed] [Google Scholar]
- 77.Artlett C.M., Smith J.B., Jimenez S.A. Identification of fetal DNA and cells in skin lesions from women with systemic sclerosis. N Engl J Med. 1998;338:1186–1191. doi: 10.1056/NEJM199804233381704. [DOI] [PubMed] [Google Scholar]
- 78.Ohtsuka T., Miyamoto Y., Yamakage A., Yamazaki S. Quantitative analysis of microchimerism in systemic sclerosis skin tissue. Arch Dermatol Res. 2001;293:387–391. doi: 10.1007/s004030100245. [DOI] [PubMed] [Google Scholar]
- 79.Sawaya H.H.B., Jimenez S.A., Artlett C.M. Quantification of fetal microchimeric cells in clinically affected and unaffected skin of patients with systemic sclerosis. Rheumatology. 2004;43:965–968. doi: 10.1093/rheumatology/keh211. [DOI] [PubMed] [Google Scholar]
- 80.Nelson J.L. Microchimerism and the pathogenesis of systemic sclerosis. Curr Opin Rheumatol. 1998;10:564–571. doi: 10.1097/00002281-199811000-00010. [DOI] [PubMed] [Google Scholar]
- 81.Fugazzola L., Cirello V., Beck-Peccoz P. Fetal microchimerism as an explanation of disease. Nat Rev Endocrinol. 2011;7:89–97. doi: 10.1038/nrendo.2010.216. [DOI] [PubMed] [Google Scholar]