Abstract
The first complete amino acid sequence of a H1 histone of a monocotyledonous plant was deduced from a cDNA isolated from a maize library. The encoded H1 protein is 245 amino acid-long and shows the classical tripartite organization of this class of histones. The central globular region of 76 residues shows 60% sequence homology with H1 proteins from dicots but only 20% with the animal H1 proteins. However, several of the amino acids considered as being important in the structure of the nucleosome are conserved between this protein and its animal counterparts. The N-terminal region contains an equal number of acidic and basic residues which appears as a general feature of plant H1 proteins. The 124 residue long and highly basic C-terminal region contains a 7-fold repeated element KA/PKXA/PAKA/PK. Southern-blot hybridization showed that the H1 protein is encoded by a small multigene family. Highly homologous H1 gene families were also detected in the genomes of several more or less closely related plant species. The general expression pattern of these genes was not significantly different from that of these genes encoding the core-histones neither during germination nor in the different tissues of adult maize.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Allan J., Mitchell T., Harborne N., Bohm L., Crane-Robinson C. Roles of H1 domains in determining higher order chromatin structure and H1 location. J Mol Biol. 1986 Feb 20;187(4):591–601. doi: 10.1016/0022-2836(86)90337-2. [DOI] [PubMed] [Google Scholar]
- Allan J., Smith B. J., Dunn B., Bustin M. Antibodies against the folding domain of histone H5 cross-react with H1(0) but not with H1. J Biol Chem. 1982 Sep 25;257(18):10533–10535. [PubMed] [Google Scholar]
- Busslinger M., Schümperli D., Birnstiel M. L. Regulation of histone gene expression. Cold Spring Harb Symp Quant Biol. 1985;50:665–670. doi: 10.1101/sqb.1985.050.01.081. [DOI] [PubMed] [Google Scholar]
- Cary P. D., Hines M. L., Bradbury E. M., Smith B. J., Johns E. W. Conformation studies of histone H1(0) in comparison with histones H1 and H5. Eur J Biochem. 1981 Nov;120(2):371–377. doi: 10.1111/j.1432-1033.1981.tb05714.x. [DOI] [PubMed] [Google Scholar]
- Chaboute M. E., Chaubet N., Clement B., Gigot C., Philipps G. Polyadenylation of histone H3 and H4 mRNAs in dicotyledonous plants. Gene. 1988 Nov 15;71(1):217–223. doi: 10.1016/0378-1119(88)90095-9. [DOI] [PubMed] [Google Scholar]
- Chaubet N., Chaboute M. E., Clément B., Ehling M., Philipps G., Gigot C. The histone H3 and H4 mRNAs are polyadenylated in maize. Nucleic Acids Res. 1988 Feb 25;16(4):1295–1304. doi: 10.1093/nar/16.4.1295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clore G. M., Gronenborn A. M., Nilges M., Sukumaran D. K., Zarbock J. The polypeptide fold of the globular domain of histone H5 in solution. A study using nuclear magnetic resonance, distance geometry and restrained molecular dynamics. EMBO J. 1987 Jun;6(6):1833–1842. doi: 10.1002/j.1460-2075.1987.tb02438.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cole R. D. A minireview of microheterogeneity in H1 histone and its possible significance. Anal Biochem. 1984 Jan;136(1):24–30. doi: 10.1016/0003-2697(84)90303-8. [DOI] [PubMed] [Google Scholar]
- Crane-Robinson C., Ptitsyn O. B. Binding of the globular domain of linker histones H5/H1 to the nucleosome: a hypothesis. Protein Eng. 1989 Aug;2(8):577–582. doi: 10.1093/protein/2.8.577. [DOI] [PubMed] [Google Scholar]
- Gantt J. S., Key J. L. Molecular cloning of a pea H1 histone cDNA. Eur J Biochem. 1987 Jul 1;166(1):119–125. doi: 10.1111/j.1432-1033.1987.tb13490.x. [DOI] [PubMed] [Google Scholar]
- Hayashi H., Iwai K., Johnson J. D., Bonner J. Pea histones H2A and H2B. Variable and conserved regions in the sequences. J Biochem. 1977 Aug;82(2):503–510. [PubMed] [Google Scholar]
- Hentschel C. C., Birnstiel M. L. The organization and expression of histone gene families. Cell. 1981 Aug;25(2):301–313. doi: 10.1016/0092-8674(81)90048-9. [DOI] [PubMed] [Google Scholar]
- Hurley C. K., Stout J. T. Maize histone H1: a partial structural characterization. Biochemistry. 1980 Feb 5;19(3):410–416. doi: 10.1021/bi00544a002. [DOI] [PubMed] [Google Scholar]
- Lehrach H., Diamond D., Wozney J. M., Boedtker H. RNA molecular weight determinations by gel electrophoresis under denaturing conditions, a critical reexamination. Biochemistry. 1977 Oct 18;16(21):4743–4751. doi: 10.1021/bi00640a033. [DOI] [PubMed] [Google Scholar]
- Lewin B. Driving the cell cycle: M phase kinase, its partners, and substrates. Cell. 1990 Jun 1;61(5):743–752. doi: 10.1016/0092-8674(90)90181-d. [DOI] [PubMed] [Google Scholar]
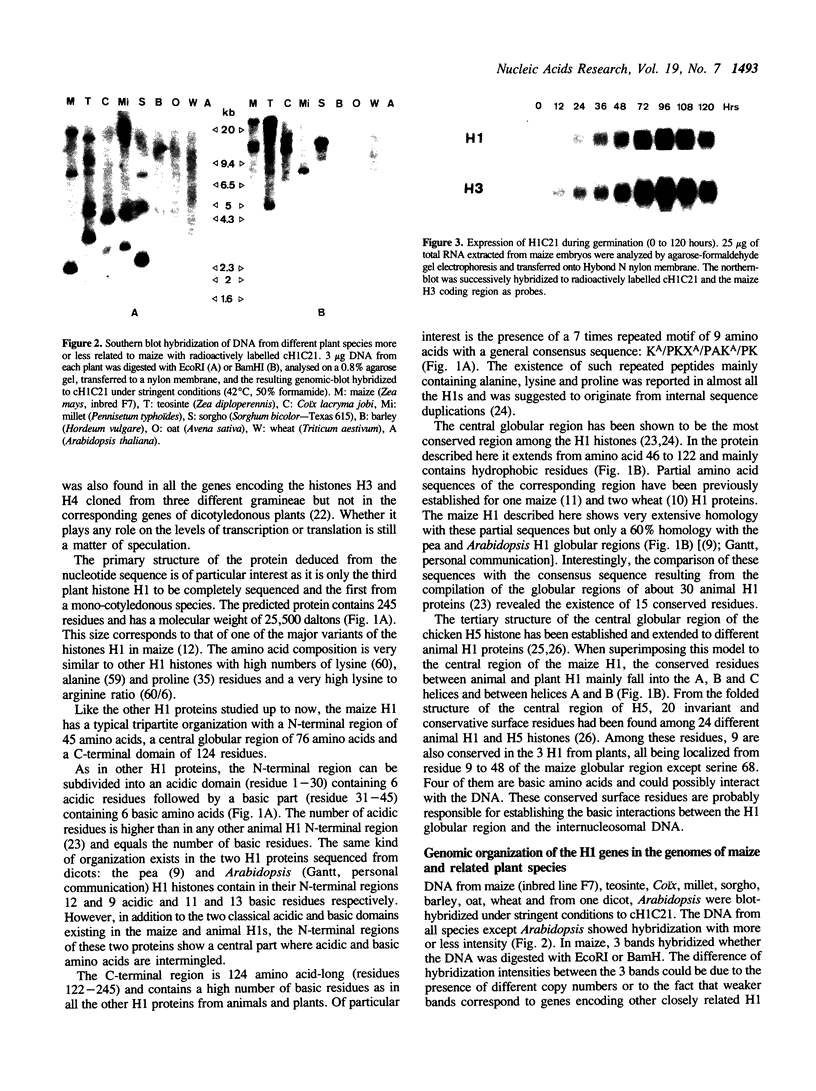
- Maxson R., Cohn R., Kedes L., Mohun T. Expression and organization of histone genes. Annu Rev Genet. 1983;17:239–277. doi: 10.1146/annurev.ge.17.120183.001323. [DOI] [PubMed] [Google Scholar]
- Mazzolini L., Vaeck M., van Montagu M. Conserved epitopes on plant H1 histones recognized by monoclonal antibodies. Eur J Biochem. 1989 Jan 2;178(3):779–787. doi: 10.1111/j.1432-1033.1989.tb14509.x. [DOI] [PubMed] [Google Scholar]
- Old R. W., Woodland H. R. Histone genes: not so simple after all. Cell. 1984 Oct;38(3):624–626. doi: 10.1016/0092-8674(84)90256-3. [DOI] [PubMed] [Google Scholar]
- Peng Z. G., Wu R. A simple and rapid nucleotide sequencing strategy and its application in analyzing a rice histone 3 gene. Gene. 1986;45(3):247–252. doi: 10.1016/0378-1119(86)90022-3. [DOI] [PubMed] [Google Scholar]
- Philipps G., Chaubet N., Chaboute M. E., Ehling M., Gigot C. Genomic organization and nucleotide sequences of two corn histone H4 genes. Gene. 1986;42(2):225–229. doi: 10.1016/0378-1119(86)90301-x. [DOI] [PubMed] [Google Scholar]
- Rodrigues J. de A., Brandt W. F., von Holt C. The amino acid sequence of wheat histone H2A(1). A core histone with a C-terminal extension. Eur J Biochem. 1985 Aug 1;150(3):499–505. doi: 10.1111/j.1432-1033.1985.tb09050.x. [DOI] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schümperli D. Cell-cycle regulation of histone gene expression. Cell. 1986 May 23;45(4):471–472. doi: 10.1016/0092-8674(86)90277-1. [DOI] [PubMed] [Google Scholar]
- Southern E. M. Detection of specific sequences among DNA fragments separated by gel electrophoresis. J Mol Biol. 1975 Nov 5;98(3):503–517. doi: 10.1016/s0022-2836(75)80083-0. [DOI] [PubMed] [Google Scholar]
- Tabata T., Sasaki K., Iwabuchi M. The structural organization and DNA sequence of a wheat histone H4 gene. Nucleic Acids Res. 1983 Sep 10;11(17):5865–5875. doi: 10.1093/nar/11.17.5865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thoma F., Losa R., Koller T. Involvement of the domains of histones H1 and H5 in the structural organization of soluble chromatin. J Mol Biol. 1983 Jul 5;167(3):619–640. doi: 10.1016/s0022-2836(83)80102-8. [DOI] [PubMed] [Google Scholar]
- Von Holt C., Strickland W. N., Brandt W. F., Strickland M. S. More histone structures. FEBS Lett. 1979 Apr 15;100(2):201–218. doi: 10.1016/0014-5793(79)80337-3. [DOI] [PubMed] [Google Scholar]
- Zlatanova J. Histone H1 and the regulation of transcription of eukaryotic genes. Trends Biochem Sci. 1990 Jul;15(7):273–276. doi: 10.1016/0968-0004(90)90053-e. [DOI] [PubMed] [Google Scholar]