Abstract
Cardiac magnetic resonance imaging (CMRI) has emerged as a useful tertiary imaging tool in the investigation of patients suspected of many different types of cardiomyopathies. CMRI sequences are now of a sufficiently robust quality to enable high spatial and temporal resolution image acquisition. This has led to CMRI becoming an effective non-invasive imaging gold standard for many cardiomyopathies. In this 2-part review, we outline the typical sequences used to image cardiomyopathy, and present the imaging spectrum of cardiomyopathy. Part 1 focuses on the current classification of cardiomyopathy, basic CMRI sequences used in evaluating cardiomyopathy and the imaging spectrum of common phenotypes.
Keywords: Cardiac magnetic resonance imaging, Cardiomyopathies
INTRODUCTION
A cardiomyopathy has been described as a myocardial disorder in which the heart muscle is structurally and functionally abnormal, in the absence of coronary artery disease, hypertension, valvular disease and congenital heart disease sufficient to cause the observed myocardial abnormality, with the exception of ischemic cardiomyopathy (Table 1)[1]. Cardiomyopathy follows myocardial infarction as the commonest cause of sudden cardiac death[2].
Table 1.
Examples of common and rare cardiomyopathies
| HCM | DCM | ARVC | RCM | Unclassified | |
| Familial | Familial, unknown gene | Familial, unknown gene | Familial, unknown gene | Familial, unknown gene | Familial, unknown gene |
| Sarcomeric protein mutations | Sarcomeric protein mutations (see HCM) | Intercalated disc protein mutations | Sarcomeric protein mutations | Left ventricular Non-compaction | |
| β myosin heavy chain | Z-band | Plakoglobin | Troponin I (RCM +/- HCM) | Barth syndrome | |
| Cardiac myosin binding protein C | Muscle LIM protein | Desmoplakin | Essential light chain of myosin | Lamin A/C | |
| Cardiac troponin 1 | TCAP | Plakophilin 2 | Familial amyloidosis | ZASP | |
| Troponin T | Cytoskeletal genes | Desmoglein 2 | Transthyretin (RCM + neuropathy) | α-dystrophin | |
| α-tropomyosin | Dystrophin | Desmocollin 2 | Apolipoprotein (RCM + neuropathy) | ||
| Essential myosin light chain | Desmin | Cardiac RyR2 | Desminopathy | ||
| Regulatory myosin light chain | Metavinculin | TGFβ3 | Pseuxanthoma elasticum | ||
| Cardiac actin | Sarcoglycan complex | Haemochromatosis | |||
| α-myosin heavy chain | CRYAB | Anderson-Fabry disease | |||
| Titin | Epicardin | Glycogen storage disease | |||
| Troponin C | Nuclear membrane | ||||
| Muscle LIM protein | Lamin A/C | ||||
| Glycogen storage disease (e.g., Pompe; PRKAG2, Forbes’, Danon) | Emerin | ||||
| Lysosomal storage disease (e.g., Anderson-Fabry, Hurler’s) | Mildly dilated cardiomyopahy | ||||
| Disorders of fatty metabolism | Intercalated disc protein mutations (see ARVC) | ||||
| Carnitine deficiency | Mitochondrial myopathy | ||||
| Phosphorylase B kinase deficiencz | Dystrophies | ||||
| Mitochondrial cytopathies | |||||
| Syndromic HCM | |||||
| Noonan syndrome | |||||
| LEOPARD syndrome | |||||
| Friedriech’s ataxia | |||||
| Beckwith-Wiedermann syndrome | |||||
| Swyer’s syndrome | |||||
| Other | |||||
| Phospholamban promotor | |||||
| Familal amyloid | |||||
| Non familial | Obesity | Myocarditis (infective/toxic/autoimmune) | Inflammation? | Amyloid (AL/prealbumin) | Tako Tsubo cardiomyopathy |
| Infants of diabetic mothers | Kawasaki disease | Scleroderma | |||
| Athletic training | Eosinophilic (Churg Strauss syndrome) | Endomyocardial fibrosis | |||
| Amyloid (AL/prealbumin) | Viral persistence | Hypereosinophilic syndrome | |||
| Drugs | Idiopathic | ||||
| Pregnancy | Chromosomal cause | ||||
| Endocrine | Drugs (serotonin, methysergide, ergotamine) | ||||
| Nutritional - thiamine, carnitine, selenium, hypophosphatemia, hypocalcemia | |||||
| Alcohol | Carcinoid heart disease | ||||
| Tachycardiomyopathy | Metastatic cancers | ||||
| Radiation | |||||
| Drugs (anthracyclines) |
HCM: Hypertrophic cardiomyopathy; DCM: Dilated cardiomyopathy; ARVC: Arrhythmogenic right ventricular dysplasia; RCM: Restricted cardiomyopathy; RyR2: Ryanodine receptor; TGF: Transforming growth factor.
Cardiac magnetic resonance imaging (CMRI) has emerged as a useful non-invasive imaging modality capable of producing high-resolution images of the heart in any desired image plane and without ionizing radiation. As a result, it has become a primary imaging modality for many cardiomyopathies[3,4]. There are many different sequences that can be performed in various combinations, although a basic generic set of sequences is common to most protocols[5].
CLASSIFICATION OF CARDIOMYOPATHY
A recent statement from the European Society of Cardiology working group on myocardial and pericardial diseases[1] has grouped cardiomyopathies into specific morphological and functional phenotypes; each phenotype is then sub-classified into familial and non-familial forms. Familial refers to the occurrence, in more than one family member, of either the same disorder or a phenotype that is (or could be) caused by the same genetic mutation and not from acquired cardiac or systemic diseases in which the clinical phenotype is influenced by genetic polymorphism. Most familial cardiomyopathies are monogenic disorders. Non-familial cardiomyopathies are subdivided into idiopathic and acquired cardiomyopathies in which ventricular dysfunction is a complication of the disorder rather than an intrinsic feature of the disease. Left ventricular (LV) dysfunction secondary to coronary artery occlusion, hypertension, valve disease, and congenital heart disease are excluded because the diagnosis and treatment of these disorders are quite different from those encountered in most cardiomyopathies. The division of cardiomyopathies into familial and non-familial forms is useful as it raises awareness of genetic disease as a cause of heart muscle dysfunction.
BASIC CMRI PROTOCOLS FOR CARDIOMYOPATHY ASSESSMENT
The CMRI protocol used in imaging cardiomyopathy should be tailored specifically to the suspected type of cardiomyopathy. There are many different sequences, but all follow a basic generic protocol[5,6]: (1) scouting images - axial, coronal and sagittal; (2) stacking of axial slices of the thorax [half-fourier acquisition single-shot turbo spin-echo or steady-state free precession (SSFP)]; (3) vertical long-axis or 2-chamber steady-state free precession (SSFP) - this imaging plane typically depicts the left atrium and ventricle. If the right ventricle is the chamber of interest, the plane can be placed along the right ventricle; (4) horizontal long-axis or 4-chamber SSFP - depicts all 4 cardiac chambers; (5) short-axis SSFP - from the annulus to the apex. These are used to allow quantification of ventricular volumes and function; (6) T2-weighted typically short-axis sequence - to assess for acute myocardial edema; and (7) late gadolinium enhancement (LGE) - appears as high signal enhancement within the myocardium following a double-inversion fast spin echo sequence. Images are typically acquired 10 to 30 min after contrast injection. Many studies have now shown that myocardial enhancement using this sequence enables the detection of myocardial infarction, inflammation, infiltration or fibrosis. Late gadolinium enhancement sequences have added a tremendous additional dimension to CMRI in detecting, localizing and quantifying myocardial disease[7].
Specific additional sequences may be added depending on the particular cardiomyopathy being investigated[5]. When describing the location of myocardial pathology it is important to use standardized nomenclature[8]. Typically, the ventricle is divided into 3 levels (basal, mid and apical) and each level can be divided into 6 (basal), 6 (mid) and 4 (apical) segments with the 17th segment being represented by the apex. Note that there is normally a thin apical thin point, and it is important not to confuse this normal appearance with an apical aneurysm[9].
Ischemic cardiomyopathy
In ischemic disease, myocardial ischemia may result in 3 functionally altered states commonly referred to as stunning, hibernation, and true infarction[10]. The stunned myocardium typically occurs in the setting of an acute ischemic insult and results in reversible contractile dysfunction whereas hibernation is the term used to describe chronic contractile impairment secondary to obstructive coronary stenosis (Figure 1). CMRI allows the detection, location and quantification of the extent of acute and chronic myocardial infarction. Chronic myocardial infarction is defined as new pathological Q waves with or without symptoms, imaging evidence of a loss of myocardial viability with wall thinning that fails to contract, in the absence of a non-ischemic cause or pathological findings of a healed or healing myocardial infarction[11]. Cardiac MRI has become the clinical non-invasive gold standard for the assessment of myocardial viability. It has superseded cardiac positron emission tomography and single photon emission computed tomography scanning for the detection of subendocardial infarction[12]. Revascularization of viable tissue should lead to an improvement in myocardial contractility, as long as the myocardium is non-transmurally infarcted[13]. In full-thickness infarcted (scarred) tissue revascularization does not improve myocardial function[14]. Despite the small size of such infarcts, detection is critical, as even patients with small infarcts have a relatively poor prognosis compared to non-infarcted patients[15]. More recently, T2-weighted sequences (sensitive to myocardial edema) have shown the area-at-risk. This shows an area of higher signal, larger than the actual infarcted ‘dead-zone’ of the myocardium, and thus indicates the area at risk of further ischemia[16]. Regional wall motion abnormalities are detected on CMRI in the areas of abnormal enhancement.
Figure 1.
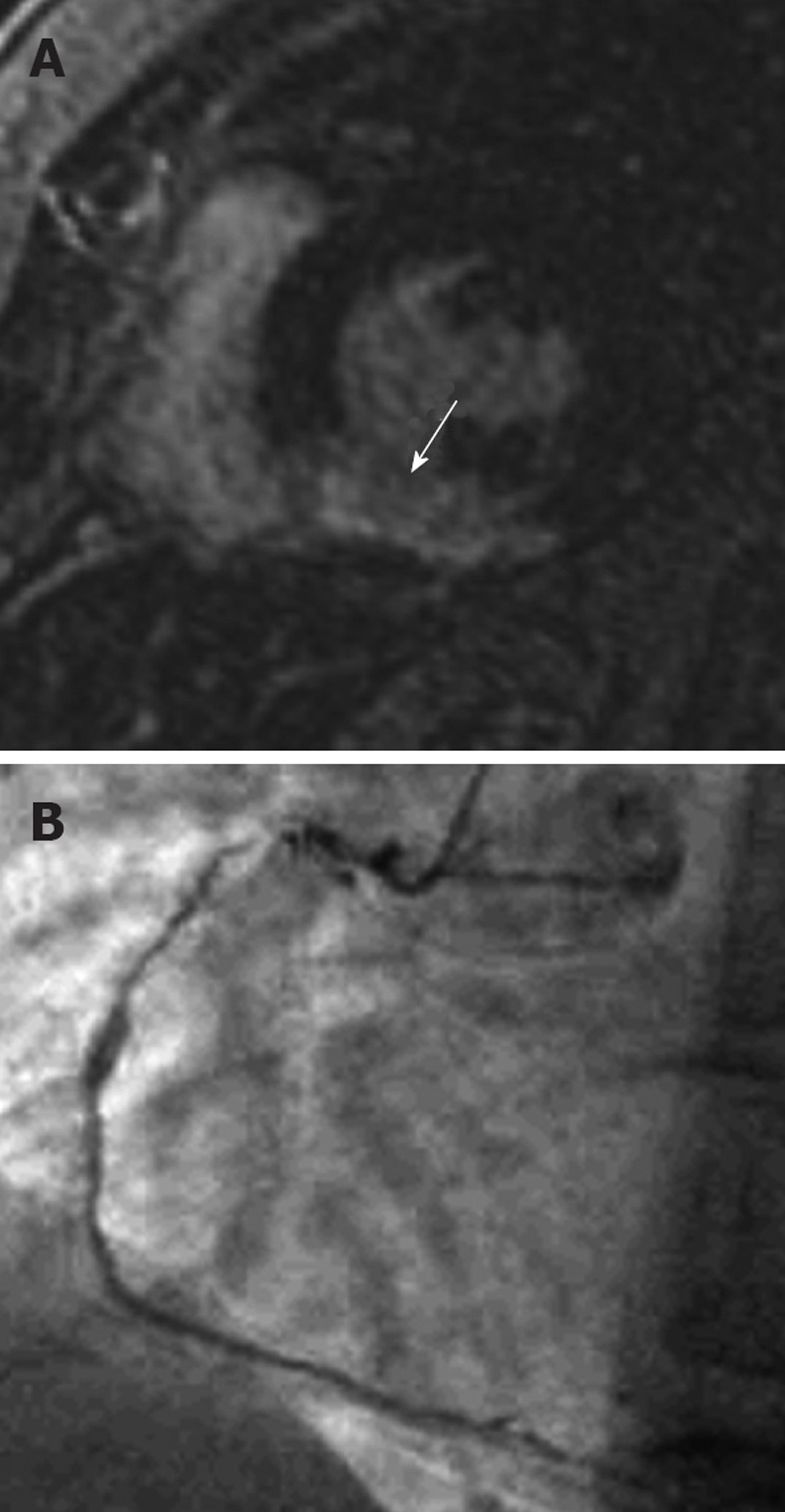
A 42-year-old female who presented with acute chest pain to the emergency department. She had no risk factors for coronary artery disease and the clinical suspicion was of myocarditis. A: Late-enhancement short axis sequence shows a transmural area (arrow) of high signal involving the inferior segment consistent with an acute myocardial infarction involving the right coronary artery territory. Note that it is an acute rather than chronic infarct, because there is no wall thinning; B: An invasive angiogram confirmed diffuse coronary artery disease throughout the right coronary artery. Note that the likelihood of recovery of this segment with revascularization is extremely low because it is a transmural infarct.
Hypertrophic cardiomyopathy
Hypertrophic cardiomyopathy (HCM) is characterized by ventricular muscle hypertrophy and impaired diastolic function associated with a non-dilated cavity, in the absence of another cardiac or systemic disease that could produce the magnitude of hypertrophy[17,18]. It is the commonest cause of sudden cardiac death in young people. It is genetically transmitted in an autosomal dominant pattern with variable penetrance and expression[19]. Symptoms are variable but can include dyspnea, orthopnea, paroxysmal nocturnal dyspnea and sudden death. Patients may also be asymptomatic. CMRI is the gold-standard non-invasive imaging modality for estimating LV volumetrics, ventricular mass and systolic function. As such, it has become a valuable imaging tool in patients suspected of HCM (Figure 2). The role of CMRI in providing prognostic information in HCM is evolving. An important initial study found LGE in the majority of patients with HCM[20]. The enhancement typically occurred in the hypertrophied regions, predominantly involving the middle third of the wall in a patchy, multifocal distribution. Moon et al[21] found that the extent of LGE was strongly associated with other risk factors for sudden death in HCM. Another study showed that even in mild HCM or asymptomatic HCM, the presence of LGE was associated with ventricular tachycardia[22]. More recently, O’Hanlon et al[23] demonstrated that in 217 patients with HCM followed for 3.1 years, the risk of unplanned heart failure admissions, deterioration to New York Heart Association functional class III or IV, or heart failure-related death was greater in the fibrosis group on CMRI [hazard ratio (HR): 2.5, P = 0.021], and this risk increased as the extent of fibrosis increased (HR: 1.16; 5% increase, P = 0.017). Thus, the presence and extent of late-enhancement on CMRI appears to hold prognostic information in patients with HCM.
Figure 2.
Hypertrophic cardiomyopathy. A: 28-year-old man who presented with progressive heart failure and palpitations. The horizontal long-axis steady-state free precession sequence demonstrates a hypertrophic interventricular septum measuring 23 mm (normal ≤ 11 mm) consistent with hypertrophic cardiomyopathy (HCM); B: Late-enhancement short-axis image shows late-enhancement in the hypertrophied septum (arrow). Note that there are 2 abnormal areas of enhancement corresponding to the right superior and inferior ventricular insertion points. This is a characteristic pattern in HCM. Such late-enhancement has prognostic implications for patients with HCM, being associated with an increased prevalence of heart failure admissions, deterioration to New York Heart Association functional class III or IV, or heart failure-related death.
Dilated cardiomyopathy
Dilated cardiomyopathy (DCM) is characterized by dilation of the cardiac chambers coupled with impaired contraction of the ventricles. The ventricular chambers exhibit increased diastolic and systolic volumes and a low ejection fraction <45%[24]. Although 50% of cases remain idiopathic[25], DCM may be a common end-pathway in many disease processes, such as chronic myocarditis and burnt-out HCM[26]. Several studies have shown that endomyocardial biopsy of a subgroup of patients with idiopathic dilated cardiomyopathy (IDC) reveals a viral genome or HCM genotype[27]. The presenting symptoms and signs are progressive dyspnea and orthopnea in the majority of patients. Arrhythmias and sudden death may also occur.
Chamber enlargement and decreased function are hallmarks of the pathological process in DCM (Figure 3). CMRI is the gold-standard non-invasive imaging modality for detecting such changes[28,29]. In addition, late contrast enhancement has been reported, most frequently in the mid-interventricular septum, indicating myocardial fibrosis. The presence of LGE has prognostic implications for patients with DCM[30]. Those patients with macroscopically detectable fibrosis on CMRI have a higher rate of repeat hospitalizations with cardiac failure, worsening symptoms, ventricular arrhythmias and cardiac related death. Thus, CMRI offers a useful non-invasive diagnostic method of diagnosing IDC whilst excluding many other potential causes of DCM, including ischemia[29]. CMRI offers the most accurate non-invasive method for assessing LV ejection fraction, can provide baseline volumetric measurements in order to monitor response to therapy and provides prognostic information.
Figure 3.
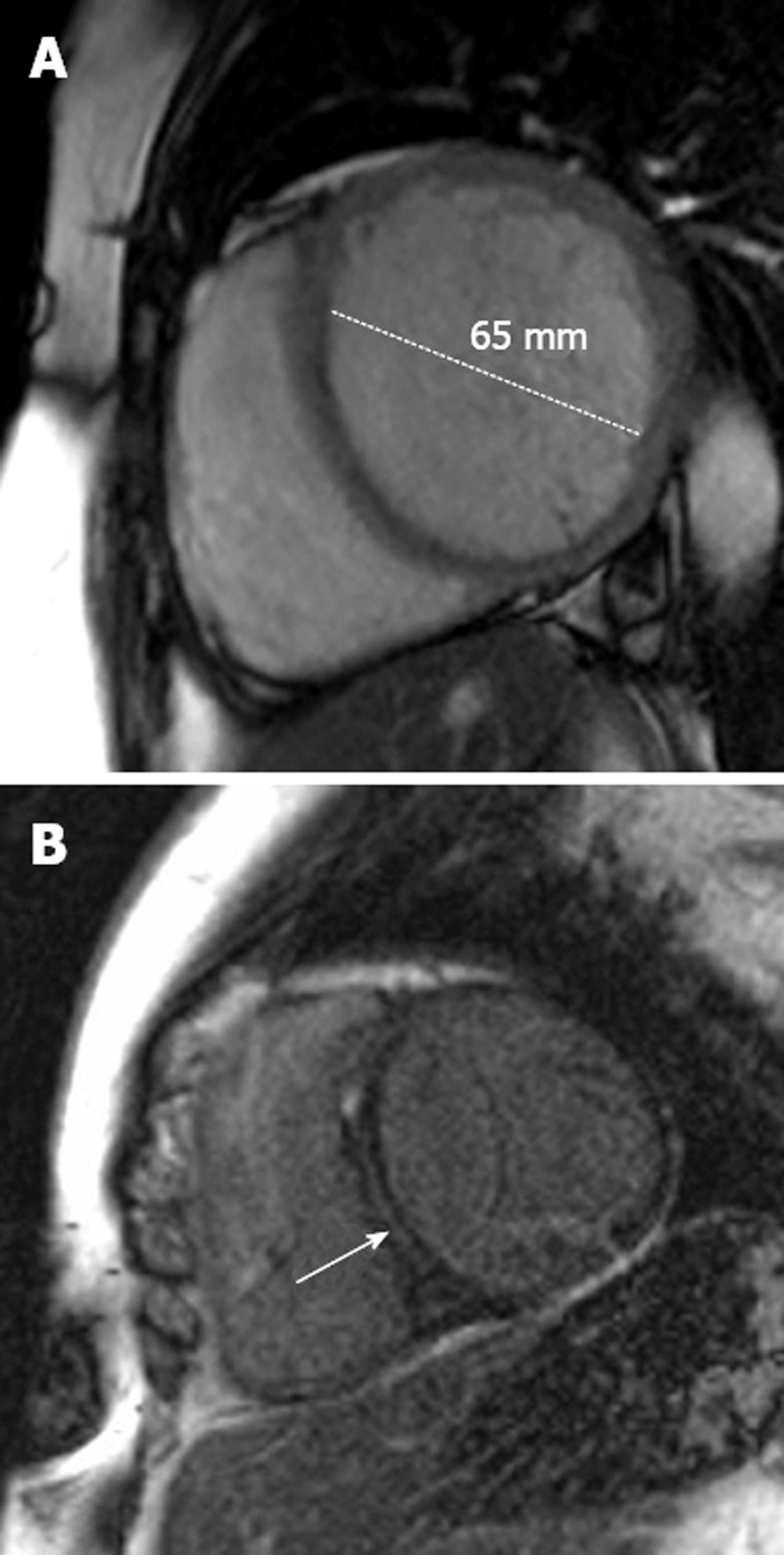
Dilated cardiomyopathy. A: A 48-year-old man with progressive shortness of breath. The short-axis steady-state free precession sequence demonstrates a dilated left ventricle with a thin wall characteristic of dilated cardiomyopathy (DCM); B: Late-enhancement short-axis image shows late-enhancement in the interventricular septum (arrow). This is a characteristic location for fibrosis detection in idiopathic DCM and effectively excludes an ischemic etiology. Such late-enhancement has prognostic implications for patients with idiopathic DCM, being associated with an increased prevalence of all-cause death, hospitalization, sudden cardiac death and ventricular tachycardia.
Cardiac sarcoid
Cardiac abnormalities are caused by myocardial infiltration by sarcoid granulomas[31]. The classical clinical presentation is heart block; however, other clinical features of sarcoid heart disease include congestive heart failure, cor pulmonale, supraventricular and ventricular arrhythmias, conductive disturbances, ventricular aneurysms, pericardial effusion, and sudden death. About 7% of patients with sarcoidosis develop cardiac symptoms but postmortem studies have revealed cardiac involvement in 20%-50% of patients[32].
The CMRI appearances of cardiac sarcoidosis depend on the acuteness of the process (Figure 4). In cases of acute inflammation, enhancement is typically in a subepicardial or mid-myocardial pattern[33]. In the chronic setting, granulomas cause focal areas of myocardial thinning resulting from scar formation. Late contrast enhancement can be seen in areas of granulomatous infiltration and is frequently patchy and nodular[34]. One study found that cardiac sarcoidosis predominantly affects the basal myocardium and the subepicardial layer[35]. They also found that hyperenhancement may be related to LV dysfunction[35]. CMRI provides an accurate estimation of the extent of cardiac involvement and may reveal signs of early infiltration that are not detected by standard echocardiographic assessment[36]. In a study of 81 consecutive patients with biopsy-proven extra-cardiac sarcoidosis, patients underwent CMRI and Japanese Ministry of Health (JMH) assessment[37]. Patients were followed for 21 ± 8 mo for major adverse events (death, defibrillator shock, or pacemaker requirement). Late gadolinium enhancement on CMRI identified cardiac involvement in 21 patients (26%) and JMH criteria in 10 (12%, 8 overlapping), a 2-fold higher rate for delayed enhancement-CMRI (P < 0.005). Pathology evaluation in 15 patients (19%) identified 4 with cardiac sarcoidosis and all 4 were positive by LGE, whereas 2 were JMH positive. On follow-up, 8 had adverse events, including 5 cardiac deaths. Patients with myocardial damage and LGE had a 9-fold higher rate of adverse events and an 11.5-fold higher rate of cardiac death than patients without enhancement.
Figure 4.
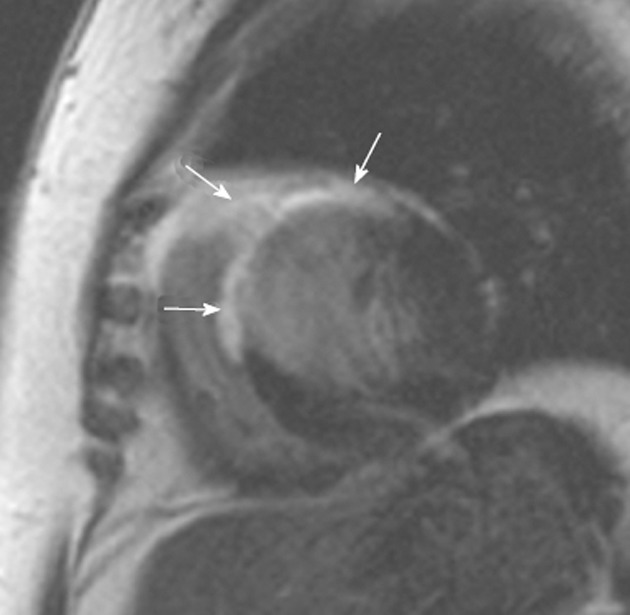
Cardiac sarcoidosis. A 54-year-old woman who presented with recurring palpitations and progressive shortness of breath for several months before suddenly collapsing. Cardiac magnetic resonance imaging demonstrates extensive scarring throughout the anterior and anteroseptal segments of the left ventricle (arrows). However, the involvement of the anterior segment of the right ventricle is unusual for ischemia and suggests another cause for the images. An endomyocardial biopsy revealed cardiac sarcoidosis. There was no previous history of lung or mediastinal sarcoid.
Cardiac amyloidosis
Cardiac amyloidosis describes amyloid deposition in the heart, which may occur as part of systemic amyloidosis or as a localized process.
Systemic amyloidosis is a complication of chronic inflammatory conditions with renal disease being the predominant feature, presenting with proteinuria and renal failure. Cardiac involvement is rare[38]. Systemic amyloidosis is the most commonly diagnosed form of clinical amyloid. Multiorgan involvement is common and the heart is affected approximately 90% of the time. Diastolic heart failure with right heart failure is the most common mode of presentation[38]. Hereditary systemic amyloidosis is due to deposition of amyloid fibrils derived from transthyretin, lysosome or apolipoprotein A-1. Clinical syndromes include cardiomyopathy, nephropathy or neuropathy. Senile systemic amyloidosis is caused by deposition of amyloid fibrils derived from normal wild-type transthyretin and presents as a slowly progressive infiltrative amyloid cardiomyopathy[38].
Cardiac amyloid typically demonstrates a diffuse decrease in signal intensity on T1-weighted fast spin echo images[39,40]. It generally causes diffuse hypertrophy of both the left and right ventricles, in contrast to HCM, which typically causes more focal hypertrophy (Figure 5). Thickening of the interatrial septum and posterior right atrial wall > 6 mm is also seen in cardiac amyloidosis[41]. Late gadolinium enhancement is another hallmark of cardiac amyloidosis on CMRI. In a study by Vogelsberg et al[42], LGE was demonstrated in 79% of patients with cardiac amyloidosis. Several different patterns of LGE were seen; LGE of the entire subendocardial circumference extending in various degrees into neighboring myocardium, circumferentially in the left ventricle with sparing of the subepicardial myocardium, and in the papillary muscles. Ejection fractions, LV end-diastolic volume and myocardial mass were not significantly different between the cardiac amyloid group and the other group of patients with various cardiac disorders. The average interventricular septum was 17 ± 4 mm in the amyloid group compared with 13 ± 3 mm in the non-amyloid group[42]. A more recent study by Syed et al[43] of CMRI in 120 patients with cardiac amyloidosis demonstrated LGE in 97% of patients and increased LV wall thickness in 91%. Global transmural or subendocardial LGE was the most common pattern seen in 83% of patients and this was associated with greater interstitial amyloid deposition.
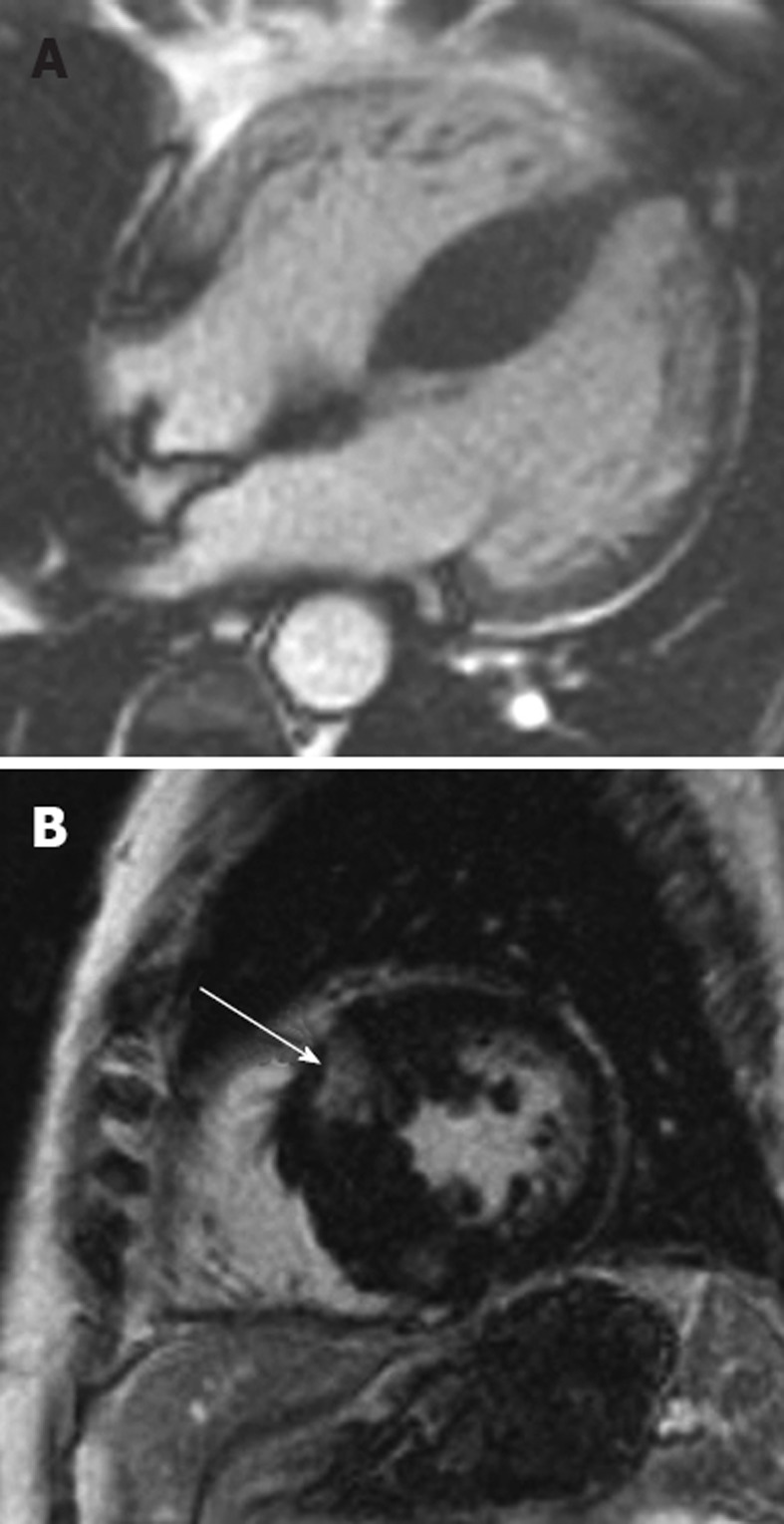
Figure 5.
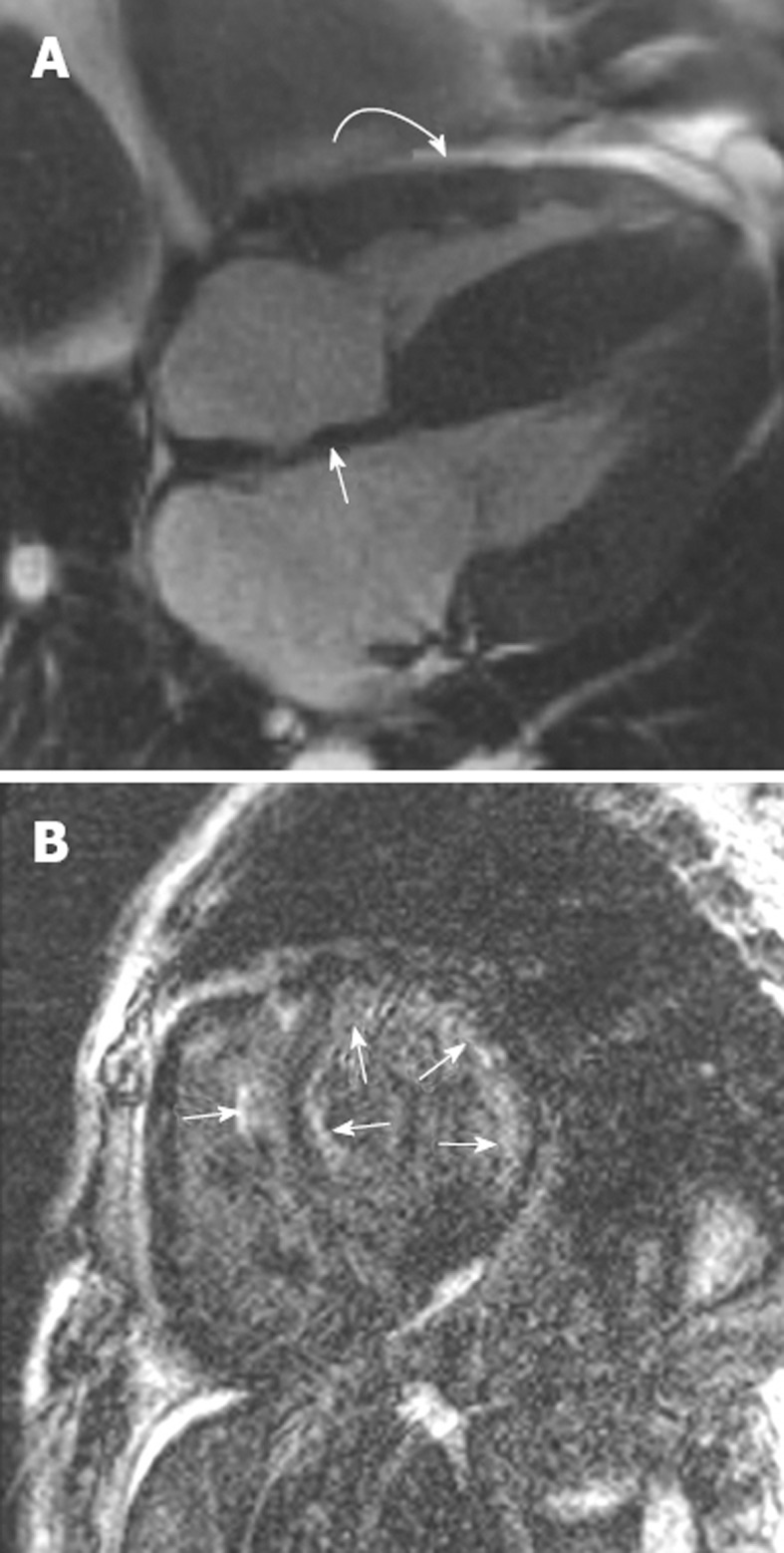
Right ventricular endomyocardial biopsy showed cardiac amyloid. A: Horizontal long axis steady-state free precession sequence showing hypertrophy of the basal segments of the left ventricle (straight arrow), and bi-atrial enlargement and thickening of the interatrial septum (curved arrow). Note the small pericardial effusion; B: Late-enhanced sequence shows a circumferential subendocardial high signal. Note the high signal on the right ventricular side of the interventricular septum resulting in the tram track sign (arrow). Note also the small pericardial effusion.
Iron overload cardiomyopathy
Iron overload occurs either due to excess gastrointestinal absorption or secondary to repeated blood transfusion. Iron overload cardiomyopathy (IOC) is the leading cause of death in patients receiving chronic blood transfusion therapy[44]. IOC is reversible, if chelation is started in time, but the diagnosis is often delayed due to the late appearance of symptoms and the absence of echocardiographic abnormalities[45]. IOC has been defined as the presence of systolic or diastolic cardiac dysfunction secondary to increased deposition of iron in the heart independent of other concomitant processes[46]. Serum ferritin or liver iron may be normal in the context of extensive myocardial iron overload. Similarly, echocardiography may be normal until extensive myocyte iron deposition has occurred.
The T2* sequence is the optimal sequence for detecting increased iron overload in the myocardium. Iron works as a paramagnetic substance, decreasing the T2* relaxation time of precessing protons in the x-y image plane. This reduces the signal from these protons, and thus leads to a darker appearance of the myocardium on grey-scale imaging (Figure 6). The decay in T2 signal can be semi-quantitatively graphed. In patients with moderate-to-severe iron deposition T2* values are substantially reduced - from the normal value of approximately 50 ms or greater to less than 20 ms. When T2* is less than 20 ms, LV systolic function is seen to decline progressively, accompanied by an increase in LV end-systolic volume index and LV mass[47]. In order to assist with clinically grading the severity of IOC, patients at risk of IOC may be divided into 3 categories based on cardiac T2* values[48]: (1) those with T2* > 20 ms (green zone) are at low risk for the imminent development of congestive heart failure; (2) those with T2* between 10 and 20 ms (yellow zone) in whom cardiac deposition has probably occurred are at intermediate risk of cardiac decompensation; and (3) those with T2* < 10 ms (red zone) are in the high-risk category of cardiac decompensation and need intensification of chelation therapy.
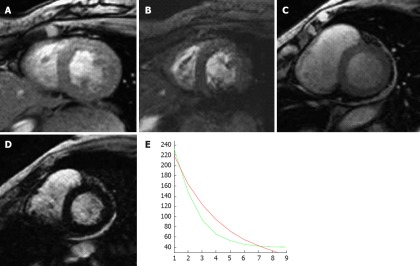
Figure 6.
A 79-year-old woman with myelodysplasia treated with blood transfusions over many years. A (echo time 10 ms) and B (echo time 20 ms) are two short axis T2star sequences from a normal patient with no evidence of myocardial iron overload; C (echo time 10 ms) and D (echo time 20 ms) are from the patient with myelodysplasia showing a progressive loss of signal with increasing T2 echo time indicating shortened T1 relation secondary to iron infiltration; E: Graph of decreasing T1 relaxation times (green line) compared with a normal patient (red line).
Metastatic disease
Metastatic disease to the heart and pericardium is uncommon but far more frequent than primary cardiac tumors[49]. Cardiac metastases are associated with a poor prognosis. The most common tumors to metastasize to the cardiac structures are lung, lymphoma, breast, renal and melanoma[50,51]. Bronchogenic carcinoma is the most common malignancy to spread to the cardiac structures and adenocarcinoma is the most common histological type[51]. Metastatic disease from bronchogenic carcinoma to the heart may be via direct spread, lymphatic or hematogenous spread[52]. Involvement of the heart and pericardium is usually a late manifestation of lymphoma, occurring in approximately 18% of cases. Primary cardiac lymphoma is diagnosed when there is no identifiable disease outside of the heart[53]. Metastases typically have the appearance of multiple focal nodules within the myocardium, and can also be found in the pericardium[54]. An interesting sub-type of cardiac metastases is “charcoal heart”, which has been described in melanoma spreading to the heart, and is related to the excessive pigment visualized in these metastases[55]. Breast cancer may spread via direct invasion (usually via the internal mammary lymph node chain) or hematogenous dissemination. Generally, metastases have irregular borders, are bulky, infiltrative, more commonly involve the right heart chambers and may be associated with a pericardial effusion[6].
Most cardiac metastases are low signal intensity on T1 sequences and brighter on T2 sequences[56] with the exception of melanoma which appears as a high signal on T1 and T2 weighted sequences due to the paramagnetic properties of melanin. Malignant disease typically enhances post contrast administration (Figure 7). An issue that is a common problem on CMRI is the blood-pool artifact on immediate post-gadolinium T1 weighted sequences. An adaption of a double-inversion fast spin echo sequence using a tissue nulling time of 600 ms is a useful additional sequence to confirm a tumor and exclude a thrombus[57]. Osteogenic sarcoma involving the heart is rare but merits mention as the metastasis contains bone. These calcific areas of increased opacity may be visible on a chest X-ray but are better characterized on computed tomography. Calcification is shown as a low signal on CMRI.
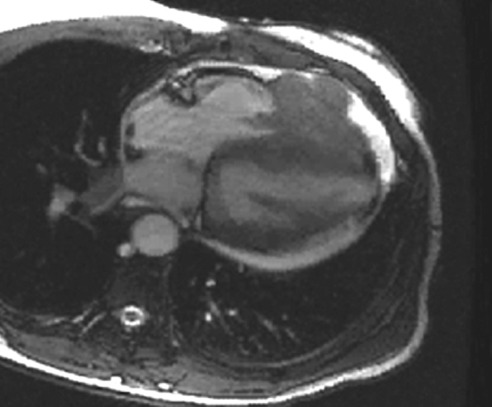
Figure 7.
A 72-year-old woman with metastatic breast cancer to the heart. Note the location of the metastatic nodules in the right heart chambers, the infiltration through the right ventricular free wall, the pericardial effusion and the pericardial metastasis. Any infiltrative appearing cardiac mass involving the right heart chambers in the presence of a pericardial effusion should be considered suspicious for malignancy.
CONCLUSION
CMRI has established itself as an important diagnostic investigation tool for assessing the morphological and functional characteristics of cardiomyopathy. Particular strengths of CMRI are the ability to overcome anatomical limitations, suboptimal acoustic windows, a multi-sequencing approach to uniquely characterize the myocardium, and the absence of ionizing radiation. The use of LGE sequences has had an important impact on the ability to characterize the myocardium, and also aids in improving clinical risk stratification for many cardiomyopathies.
Footnotes
Peer reviewers: Jalal K Ghali, MD, Chief of Cardiology, Detroit Receiving Hospital, Director, Heart Failure Program Medical Director, Cardiovascular, Clinical Trials Program, DMC Cardiovascular Institute, Professor of Medicine, Wayne State University, 3990 John R, 9 Webber Suite 9370, Detroit, MI 48201, United States; Antigone Lazou, Professor of Physiology, Lab of Animal Physiology, Sch of Biology, Aristotle University of Thessaloniki, 54124 Thessaloniki, Greece; Dr. Steven J Haas, National Coroners Information System, Victorian Institute of Forensic Medicine, 57-83 Kavanagh Street, Southbank 3006, Australia
S- Editor Cheng JX L- Editor Cant MR E- Editor Li JY
References
- 1.Elliott P, Andersson B, Arbustini E, Bilinska Z, Cecchi F, Charron P, Dubourg O, Kühl U, Maisch B, McKenna WJ, et al. Classification of the cardiomyopathies: a position statement from the European Society Of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur Heart J. 2008;29:270–276. doi: 10.1093/eurheartj/ehm342. [DOI] [PubMed] [Google Scholar]
- 2.Rosamond W, Flegal K, Friday G, Furie K, Go A, Greenlund K, Haase N, Ho M, Howard V, Kissela B, et al. Heart disease and stroke statistics--2007 update: a report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation. 2007;115:e69–171. doi: 10.1161/CIRCULATIONAHA.106.179918. [DOI] [PubMed] [Google Scholar]
- 3.Bluemke DA, Achenbach S, Budoff M, Gerber TC, Gersh B, Hillis LD, Hundley WG, Manning WJ, Printz BF, Stuber M, et al. Noninvasive coronary artery imaging: magnetic resonance angiography and multidetector computed tomography angiography: a scientific statement from the american heart association committee on cardiovascular imaging and intervention of the council on cardiovascular radiology and intervention, and the councils on clinical cardiology and cardiovascular disease in the young. Circulation. 2008;118:586–606. doi: 10.1161/CIRCULATIONAHA.108.189695. [DOI] [PubMed] [Google Scholar]
- 4.Pennell DJ, Sechtem UP, Higgins CB, Manning WJ, Pohost GM, Rademakers FE, van Rossum AC, Shaw LJ, Yucel EK. Clinical indications for cardiovascular magnetic resonance (CMR): Consensus Panel report. J Cardiovasc Magn Reson. 2004;6:727–765. doi: 10.1081/jcmr-200038581. [DOI] [PubMed] [Google Scholar]
- 5.Kramer CM, Barkhausen J, Flamm SD, Kim RJ, Nagel E. Standardized cardiovascular magnetic resonance imaging (CMR) protocols, society for cardiovascular magnetic resonance: board of trustees task force on standardized protocols. J Cardiovasc Magn Reson. 2008;10:35. doi: 10.1186/1532-429X-10-35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.O'Donnell DH, Abbara S, Chaithiraphan V, Yared K, Killeen RP, Cury RC, Dodd JD. Cardiac tumors: optimal cardiac MR sequences and spectrum of imaging appearances. AJR Am J Roentgenol. 2009;193:377–387. doi: 10.2214/AJR.08.1895. [DOI] [PubMed] [Google Scholar]
- 7.Kim RJ, Shah DJ, Judd RM. How we perform delayed enhancement imaging. J Cardiovasc Magn Reson. 2003;5:505–514. doi: 10.1081/JCMR-120022267. [DOI] [PubMed] [Google Scholar]
- 8.Cerqueira MD, Weissman NJ, Dilsizian V, Jacobs AK, Kaul S, Laskey WK, Pennell DJ, Rumberger JA, Ryan T, Verani MS. Standardized myocardial segmentation and nomenclature for tomographic imaging of the heart: a statement for healthcare professionals from the Cardiac Imaging Committee of the Council on Clinical Cardiology of the American Heart Association. Circulation. 2002;105:539–542. doi: 10.1161/hc0402.102975. [DOI] [PubMed] [Google Scholar]
- 9.Ferencik M, Abbara S, Hoffmann U, Cury RC, Brady TJ, Achenbach S. Left ventricular thin-point detection using multidetector spiral computed tomography. Am J Cardiol. 2004;93:949–951. doi: 10.1016/j.amjcard.2003.12.033. [DOI] [PubMed] [Google Scholar]
- 10.Kaandorp TA, Lamb HJ, van der Wall EE, de Roos A, Bax JJ. Cardiovascular MR to access myocardial viability in chronic ischaemic LV dysfunction. Heart. 2005;91:1359–1365. doi: 10.1136/hrt.2003.025353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Thygesen K, Alpert JS, White HD. Universal definition of myocardial infarction. Eur Heart J. 2007;28:2525–2538. doi: 10.1093/eurheartj/ehm355. [DOI] [PubMed] [Google Scholar]
- 12.Wagner A, Mahrholdt H, Holly TA, Elliott MD, Regenfus M, Parker M, Klocke FJ, Bonow RO, Kim RJ, Judd RM. Contrast-enhanced MRI and routine single photon emission computed tomography (SPECT) perfusion imaging for detection of subendocardial myocardial infarcts: an imaging study. Lancet. 2003;361:374–379. doi: 10.1016/S0140-6736(03)12389-6. [DOI] [PubMed] [Google Scholar]
- 13.Pegg TJ, Selvanayagam JB, Jennifer J, Francis JM, Karamitsos TD, Dall'Armellina E, Smith KL, Taggart DP, Neubauer S. Prediction of global left ventricular functional recovery in patients with heart failure undergoing surgical revascularisation, based on late gadolinium enhancement cardiovascular magnetic resonance. J Cardiovasc Magn Reson. 2010;12:56. doi: 10.1186/1532-429X-12-56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kim RJ, Wu E, Rafael A, Chen EL, Parker MA, Simonetti O, Klocke FJ, Bonow RO, Judd RM. The use of contrast-enhanced magnetic resonance imaging to identify reversible myocardial dysfunction. N Engl J Med. 2000;343:1445–1453. doi: 10.1056/NEJM200011163432003. [DOI] [PubMed] [Google Scholar]
- 15.Kwong RY, Chan AK, Brown KA, Chan CW, Reynolds HG, Tsang S, Davis RB. Impact of unrecognized myocardial scar detected by cardiac magnetic resonance imaging on event-free survival in patients presenting with signs or symptoms of coronary artery disease. Circulation. 2006;113:2733–2743. doi: 10.1161/CIRCULATIONAHA.105.570648. [DOI] [PubMed] [Google Scholar]
- 16.Raman SV, Simonetti OP, Winner MW, Dickerson JA, He X, Mazzaferri EL, Ambrosio G. Cardiac magnetic resonance with edema imaging identifies myocardium at risk and predicts worse outcome in patients with non-ST-segment elevation acute coronary syndrome. J Am Coll Cardiol. 2010;55:2480–2488. doi: 10.1016/j.jacc.2010.01.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hansen MW, Merchant N. MRI of hypertrophic cardiomyopathy: part 2, Differential diagnosis, risk stratification, and posttreatment MRI appearances. AJR Am J Roentgenol. 2007;189:1344–1352. doi: 10.2214/AJR.07.2287. [DOI] [PubMed] [Google Scholar]
- 18.Hansen MW, Merchant N. MRI of hypertrophic cardiomyopathy: part I, MRI appearances. AJR Am J Roentgenol. 2007;189:1335–1343. doi: 10.2214/AJR.07.2286. [DOI] [PubMed] [Google Scholar]
- 19.Maron BJ, Towbin JA, Thiene G, Antzelevitch C, Corrado D, Arnett D, Moss AJ, Seidman CE, Young JB. Contemporary definitions and classification of the cardiomyopathies: an American Heart Association Scientific Statement from the Council on Clinical Cardiology, Heart Failure and Transplantation Committee; Quality of Care and Outcomes Research and Functional Genomics and Translational Biology Interdisciplinary Working Groups; and Council on Epidemiology and Prevention. Circulation. 2006;113:1807–1816. doi: 10.1161/CIRCULATIONAHA.106.174287. [DOI] [PubMed] [Google Scholar]
- 20.Choudhury L, Mahrholdt H, Wagner A, Choi KM, Elliott MD, Klocke FJ, Bonow RO, Judd RM, Kim RJ. Myocardial scarring in asymptomatic or mildly symptomatic patients with hypertrophic cardiomyopathy. J Am Coll Cardiol. 2002;40:2156–2164. doi: 10.1016/S0735-1097(02)02602-5. [DOI] [PubMed] [Google Scholar]
- 21.Moon JC, McKenna WJ, McCrohon JA, Elliott PM, Smith GC, Pennell DJ. Toward clinical risk assessment in hypertrophic cardiomyopathy with gadolinium cardiovascular magnetic resonance. J Am Coll Cardiol. 2003;41:1561–1567. doi: 10.1016/S0735-1097(03)00189-X. [DOI] [PubMed] [Google Scholar]
- 22.Adabag AS, Maron BJ, Appelbaum E, Harrigan CJ, Buros JL, Gibson CM, Lesser JR, Hanna CA, Udelson JE, Manning WJ, et al. Occurrence and frequency of arrhythmias in hypertrophic cardiomyopathy in relation to delayed enhancement on cardiovascular magnetic resonance. J Am Coll Cardiol. 2008;51:1369–1374. doi: 10.1016/j.jacc.2007.11.071. [DOI] [PubMed] [Google Scholar]
- 23.O'Hanlon R, Grasso A, Roughton M, Moon JC, Clark S, Wage R, Webb J, Kulkarni M, Dawson D, Sulaibeekh L, et al. Prognostic significance of myocardial fibrosis in hypertrophic cardiomyopathy. J Am Coll Cardiol. 2010;56:867–874. doi: 10.1016/j.jacc.2010.05.010. [DOI] [PubMed] [Google Scholar]
- 24.Richardson P, McKenna W, Bristow M, Maisch B, Mautner B, O'Connell J, Olsen E, Thiene G, Goodwin J, Gyarfas I, et al. Report of the 1995 World Health Organization/International Society and Federation of Cardiology Task Force on the Definition and Classification of cardiomyopathies. Circulation. 1996;93:841–842. doi: 10.1161/01.CIR.93.5.841. [DOI] [PubMed] [Google Scholar]
- 25.Felker GM, Hu W, Hare JM, Hruban RH, Baughman KL, Kasper EK. The spectrum of dilated cardiomyopathy. The Johns Hopkins experience with 1,278 patients. Medicine (Baltimore) 1999;78:270–283. doi: 10.1097/00005792-199907000-00005. [DOI] [PubMed] [Google Scholar]
- 26.Harris KM, Spirito P, Maron MS, Zenovich AG, Formisano F, Lesser JR, Mackey-Bojack S, Manning WJ, Udelson JE, Maron BJ. Prevalence, Clinical Profile, and Significance of Left Ventricular Remodeling in the End-Stage Phase of Hypertrophic Cardiomyopathy. Circulation. 2006;114:216–225. doi: 10.1161/CIRCULATIONAHA.105.583500. [DOI] [PubMed] [Google Scholar]
- 27.Kamisago M, Sharma SD, DePalma SR, Solomon S, Sharma P, McDonough B, Smoot L, Mullen MP, Woolf PK, Wigle ED, et al. Mutations in sarcomere protein genes as a cause of dilated cardiomyopathy. N Engl J Med. 2000;343:1688–1696. doi: 10.1056/NEJM200012073432304. [DOI] [PubMed] [Google Scholar]
- 28.Bellenger NG, Burgess MI, Ray SG, Lahiri A, Coats AJ, Cleland JG, Pennell DJ. Comparison of left ventricular ejection fraction and volumes in heart failure by echocardiography, radionuclide ventriculography and cardiovascular magnetic resonance; are they interchangeable? Eur Heart J. 2000;21:1387–1396. doi: 10.1053/euhj.2000.2011. [DOI] [PubMed] [Google Scholar]
- 29.McCrohon JA, Moon JC, Prasad SK, McKenna WJ, Lorenz CH, Coats AJ, Pennell DJ. Differentiation of heart failure related to dilated cardiomyopathy and coronary artery disease using gadolinium-enhanced cardiovascular magnetic resonance. Circulation. 2003;108:54–59. doi: 10.1161/01.CIR.0000078641.19365.4C. [DOI] [PubMed] [Google Scholar]
- 30.Assomull RG, Prasad SK, Lyne J, Smith G, Burman ED, Khan M, Sheppard MN, Poole-Wilson PA, Pennell DJ. Cardiovascular magnetic resonance, fibrosis, and prognosis in dilated cardiomyopathy. J Am Coll Cardiol. 2006;48:1977–1985. doi: 10.1016/j.jacc.2006.07.049. [DOI] [PubMed] [Google Scholar]
- 31.Smedema JP, Snoep G, van Kroonenburgh MPG, van Geuns RJ, Cheriex EC, Gorgels APM, Crigns HJGM. The additional value of gadolinium-enhanced MRI to standard assessment for cardiac involvement in patients with pulmonary sarcoidosis. Chest. 2005;128:1629–1637. doi: 10.1378/chest.128.3.1629. [DOI] [PubMed] [Google Scholar]
- 32.Smedema JP, Snoep G, van Kroonenburgh MP, van Geuns RJ, Dassen WR, Gorgels AP, Crijns HJ. Evaluation of the accuracy of gadolinium-enhanced cardiovascular magnetic resonance in the diagnosis of cardiac sarcoidosis. J Am Coll Cardiol. 2005;45:1683–1690. doi: 10.1016/j.jacc.2005.01.047. [DOI] [PubMed] [Google Scholar]
- 33.Matoh F, Satoh H, Shiraki K, Odagiri K, Saitoh T, Urushida T, Katoh H, Takehara Y, Sakahara H, Hayashi H. The usefulness of delayed enhancement magnetic resonance imaging for diagnosis and evaluation of cardiac function in patients with cardiac sarcoidosis. J Cardiol. 2008;51:179–188. doi: 10.1016/j.jjcc.2008.03.002. [DOI] [PubMed] [Google Scholar]
- 34.Vignaux O. Cardiac sarcoidosis: spectrum of MRI features. AJR Am J Roentgenol. 2005;184:249–254. doi: 10.2214/ajr.184.1.01840249. [DOI] [PubMed] [Google Scholar]
- 35.Ichinose A, Otani H, Oikawa M, Takase K, Saito H, Shimokawa H, Takahashi S. MRI of cardiac sarcoidosis: basal and subepicardial localization of myocardial lesions and their effect on left ventricular function. AJR Am J Roentgenol. 2008;191:862–869. doi: 10.2214/AJR.07.3089. [DOI] [PubMed] [Google Scholar]
- 36.Smedema JP, Snoep G, van Kroonenburgh MP, van Geuns RJ, Cheriex EC, Gorgels AP, Crijns HJ. The additional value of gadolinium-enhanced MRI to standard assessment for cardiac involvement in patients with pulmonary sarcoidosis. Chest. 2005;128:1629–1637. doi: 10.1378/chest.128.3.1629. [DOI] [PubMed] [Google Scholar]
- 37.Patel MR, Cawley PJ, Heitner JF, Klem I, Parker MA, Jaroudi WA, Meine TJ, White JB, Elliott MD, Kim HW, et al. Detection of myocardial damage in patients with sarcoidosis. Circulation. 2009;120:1969–1977. doi: 10.1161/CIRCULATIONAHA.109.851352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Selvanayagam JB, Hawkins PN, Paul B, Myerson SG, Neubauer S. Evaluation and management of the cardiac amyloidosis. J Am Coll Cardiol. 2007;50:2101–2110. doi: 10.1016/j.jacc.2007.08.028. [DOI] [PubMed] [Google Scholar]
- 39.vanden Driesen RI, Slaughter RE, Strugnell WE. MR findings in cardiac amyloidosis. AJR Am J Roentgenol. 2006;186:1682–1685. doi: 10.2214/AJR.04.0871. [DOI] [PubMed] [Google Scholar]
- 40.Krombach GA, Hahn C, Tomars M, Buecker A, Grawe A, Günther RW, Kühl HP. Cardiac amyloidosis: MR imaging findings and T1 quantification, comparison with control subjects. J Magn Reson Imaging. 2007;25:1283–1287. doi: 10.1002/jmri.20917. [DOI] [PubMed] [Google Scholar]
- 41.Rathi VK, Doyle M, Yamrozik J, Williams RB, Caruppannan K, Truman C, Vido D, Biederman RW. Routine evaluation of left ventricular diastolic function by cardiovascular magnetic resonance: a practical approach. J Cardiovasc Magn Reson. 2008;10:36. doi: 10.1186/1532-429X-10-36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Vogelsberg H, Mahrholdt H, Deluigi CC, Yilmaz A, Kispert EM, Greulich S, Klingel K, Kandolf R, Sechtem U. Cardiovascular magnetic resonance in clinically suspected cardiac amyloidosis: noninvasive imaging compared to endomyocardial biopsy. J Am Coll Cardiol. 2008;51:1022–1030. doi: 10.1016/j.jacc.2007.10.049. [DOI] [PubMed] [Google Scholar]
- 43.Syed IS, Glockner JF, Feng D, Araoz PA, Martinez MW, Edwards WD, Gertz MA, Dispenzieri A, Oh JK, Bellavia D, et al. Role of cardiac magnetic resonance imaging in the detection of cardiac amyloidosis. JACC Cardiovasc Imaging. 2010;3:155–164. doi: 10.1016/j.jcmg.2009.09.023. [DOI] [PubMed] [Google Scholar]
- 44.Olivieri NF, Nathan DG, MacMillan JH, Wayne AS, Liu PP, McGee A, Martin M, Koren G, Cohen AR. Survival in medically treated patients with homozygous beta-thalassemia. N Engl J Med. 1994;331:574–578. doi: 10.1056/NEJM199409013310903. [DOI] [PubMed] [Google Scholar]
- 45.Mavrogeni S. Evaluation of myocardial iron overload using magnetic resonance imaging. Blood Transfus. 2009;7:183–187. doi: 10.2450/2008.0063-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Liu P, Olivieri N. Iron overload cardiomyopathies: new insights into an old disease. Cardiovasc Drugs Ther. 1994;8:101–110. doi: 10.1007/BF00877096. [DOI] [PubMed] [Google Scholar]
- 47.Anderson LJ, Holden S, Davis B, Prescott E, Charrier CC, Bunce NH, Firmin DN, Wonke B, Porter J, Walker JM, et al. Cardiovascular T2-star (T2*) magnetic resonance for the early diagnosis of myocardial iron overload. Eur Heart J. 2001;22:2171–2179. doi: 10.1053/euhj.2001.2822. [DOI] [PubMed] [Google Scholar]
- 48.Wood JC. Magnetic resonance imaging measurement of iron overload. Curr Opin Hematol. 2007;14:183–190. doi: 10.1097/MOH.0b013e3280d2b76b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Lam KY, Dickens P, Chan AC. Tumors of the heart. A 20-year experience with a review of 12,485 consecutive autopsies. Arch Pathol Lab Med. 1993;117:1027–1031. [PubMed] [Google Scholar]
- 50.Mukai K, Shinkai T, Tominaga K, Shimosato Y. The incidence of secondary tumors of the heart and pericardium: a 10-year study. Jpn J Clin Oncol. 1988;18:195–201. [PubMed] [Google Scholar]
- 51.Klatt EC, Heitz DR. Cardiac metastases. Cancer. 1990;65:1456–1459. doi: 10.1002/1097-0142(19900315)65:6<1456::aid-cncr2820650634>3.0.co;2-5. [DOI] [PubMed] [Google Scholar]
- 52.Choe DH, Lee JH, Lee BH, Kim KH, Chin SY, Zo JI, Baek HJ, Park JH. Obliteration of the pulmonary vein in lung cancer: significance in assessing local extent with CT. J Comput Assist Tomogr. 1998;22:587–591. doi: 10.1097/00004728-199807000-00014. [DOI] [PubMed] [Google Scholar]
- 53.Bambury R, Gallagher F, Dodd JD, Fennelly D. Primary cardiac lymphoma: diagnostic tools and treatment challenges. Ir J Med Sci. 2011;180:271–273. doi: 10.1007/s11845-009-0446-x. [DOI] [PubMed] [Google Scholar]
- 54.Roberts WC, Glancy DL, DeVita VT. Heart in malignant lymphoma (Hodgkin's disease, lymphosarcoma, reticulum cell sarcoma and mycosis fungoides). A study of 196 autopsy cases. Am J Cardiol. 1968;22:85–107. doi: 10.1016/0002-9149(68)90250-6. [DOI] [PubMed] [Google Scholar]
- 55.Waller BF, Gottdiener JS, Virmani R, Roberts WC. The "charcoal heart; " melanoma to the cor. Chest. 1980;77:671–676. doi: 10.1378/chest.77.5.671. [DOI] [PubMed] [Google Scholar]
- 56.Fujita N, Caputo GR, Higgins CB. Diagnosis and characterization of intracardiac masses by magnetic resonance imaging. Am J Card Imaging. 1994;8:69–80. [PubMed] [Google Scholar]
- 57.O'Neill A, Ridge C, Cury RC, Quinn M, Keane D, Dodd JD. Cardiovascular flashlight. Detection of cardiac tumour-surface thrombus: utilization of cardiac magnetic resonance imaging avascular tissue nulling sequence. Eur Heart J. 2010;31:1421. doi: 10.1093/eurheartj/ehq029. [DOI] [PubMed] [Google Scholar]