Abstract
A metabolism is a complex network of chemical reactions that converts sources of energy and chemical elements into biomass and other molecules. To design a metabolism from scratch and to implement it in a synthetic genome is almost within technological reach. Ideally, a synthetic metabolism should be able to synthesize a desired spectrum of molecules at a high rate, from multiple different nutrients, while using few chemical reactions, and producing little or no waste. Not all of these properties are achievable simultaneously. We here use a recently developed technique to create random metabolic networks with pre-specified properties to quantify trade-offs between these and other properties. We find that for every additional molecule to be synthesized a network needs on average three additional reactions. For every additional carbon source to be utilized, it needs on average two additional reactions. Networks able to synthesize 20 biomass molecules from each of 20 alternative sole carbon sources need to have at least 260 reactions. This number increases to 518 reactions for networks that can synthesize more than 60 molecules from each of 80 carbon sources. The maximally achievable rate of biosynthesis decreases by approximately 5 percent for every additional molecule to be synthesized. Biochemically related molecules can be synthesized at higher rates, because their synthesis produces less waste. Overall, the variables we study can explain 87 percent of variation in network size and 84 percent of the variation in synthesis rate. The constraints we identify prescribe broad boundary conditions that can help to guide synthetic metabolism design.
Introduction
Among the most important goals of synthetic biology and biotechnology is to engineer organisms with novel properties [1], [2]. Most current efforts focus on designing subsystems of organisms, such as regulatory circuits [1], [3] or metabolic pathways [4], [5]. However, recent advances in genomics and genome synthesis have allowed synthetic biology to make great strides towards the ultimate goal of designing new organisms from scratch [6]–[10].
To be able to synthesize new life, one needs to understand life’s minimal needs. Considerable effort has thus focused on understanding and creating minimal organisms [9]–[15]. One line of research studies organisms with very small genomes that comprise only a few hundred genes, such as the gamma proteobacteria [14], Blochannia floridanus [16] and Carsonella ruddii [17]. Such organisms are typically endosymbionts or endoparasites, and receive substantial resources from their host [15]. Although valuable knowledge has been gained by studying these organisms, this property renders them of limited use in understanding minimal requirements for a free-living organism.
A second, complementary line of research starts from a complex genome, successfully eliminates genes from it without affecting viability, and thus creates a genome that is (close to) minimal. This is possible because free-living organisms have many genes that are dispensable in any one environment [9]–[11], [13], [18]. Such systematic gene deletion efforts not only provide insight into minimal genomes, they can also help to eliminate the synthesis of undesired molecules, avoid excessive waste production, and thus increase the efficiency with which an organism synthesizes desired molecules [19]. Based on both computational and experimental approaches of genome reduction, several proposals for the gene complements of minimal organisms have been made. They range in size from 100 genes to more than 300 genes in organisms such as Mycoplasmas [9], [11], [13].
One indispensable feature of any living organisms is its metabolism. A metabolism is a complex network of chemical reactions, catalyzed by enzymes that are encoded in genes. It uses sources of energy and chemical elements – nutrients – to synthesize molecules that an organism needs, including precursors of biomass and various secondary products, such as molecules for defense and communication [14]. The manipulation of metabolism for technological purposes is known as metabolic engineering [19]–[22].
Metabolic engineering has multiple applications. They include the large-scale, fast, and efficient synthesis of pharmaceuticals, chemical reagents, and biofuels [2], [19], [23]–[26]. The latter class of molecules is especially important given their importance in energy security and in the reduction of greenhouse gas emissions [20], [21], [27], [28]. Another application is bioremediation, where microbes with properly engineered metabolic pathways may be able to clean hazardous waste in inaccessible places [28], [29].
Current metabolic engineering approaches typically manipulate one or a few enzyme-coding genes [1]. Because of the highly interconnected nature of metabolism, and because of the complexity of enzyme regulation, such manipulation faces several challenges. The first is to ensure a high level of expression of the genes and the enzymes they encode. A second challenge is to manipulate cells into selectively producing desired molecules at high rates and yield. Cells can be quite recalcitrant to such manipulations [30]. A third challenge is to ensure that a desired product can be produced from one source of chemical elements and energy, but from multiple sources, to ensure efficient production. For example, yeast species are good candidate organisms to synthesize ethanol, with the drawback that they are not highly efficient at fermenting cellulosic biomass. (In addition to glucose, which yeast strains can catabolize normally, cellulosic biomass contains five carbon sugars, such as arabinose and xylose, which yeast strains cannot catabolize.) Metabolic engineering can create yeast strains that ferment not only glucose but also mixtures of other sugars. [31]. A fourth challenge is to overcome the toxicity of some desired products when they accumulate at high concentrations in a cell. This holds especially for biofuels that are produced in large amounts [27]. Finally, metabolic engineering needs to control ratios of metabolites such as ATP/ADP or NAD+/NADH, which can influence product yields and lead to synthesis of undesired byproducts through their global effects on physiology [21].
While contemporary metabolic engineering focuses on altering existing pathways, future engineering will design metabolisms and minimal organisms de novo [1], [21], [32], [33]. Ongoing technological advances in sequencing and de-novo synthesis and declining prices in these technologies [2], [20], [34], [35] suggest that de-novo synthesis of minimal organisms for biomass production will be feasible soon. A small (synthetic) metabolism may also allow better control of metabolic properties than a large metabolism [1], [18].
To be able to design a metabolism, one needs to be able to predict system-wide metabolic properties. In recent years great strides have been made towards such prediction. Especially noteworthy are constraint-based modeling approaches, which can predict the spectrum of biosynthetic properties of a metabolism from knowledge about the reactions that its enzymes catalyze, and from the nutrients available in its environment [36]–[38]. One such approach, flux balance analysis (FBA) [36], [37], [39], [40], uses information about the stoichiometry of reactions in a metabolic network to predict the rate at which a network can synthesize a given spectrum of molecules, which we refer to as the network’s biosynthetic flux. FBA makes two main assumptions. The first is that a metabolism is in a steady state with a constant nutrient supply. The second is that it maximizes some property, such as biosynthetic flux [41]. While FBA faces challenges caused by regulatory constraints [42], [43], it is well suited to answer simple qualitative and quantitative questions about important properties of a metabolic system [44]. An especially important property is the minimal number of reactions R needed to synthesize a given number B of (biomass) molecules from a given spectrum N of nutrients. The ideal network has few reactions and can synthesize many molecules using a broad spectrum of nutrients. However trade-offs between these properties exist, which do not allow all these requirements to be met.
We here take a first step towards a quantitative understanding of these and other trade-offs using constraint-based methods. To this end, we study the properties of not just one metabolic network, but of multiple networks that differ in these properties. Experimental techniques are not yet suitable to do that, but computational approaches are [45]. The approach we use starts with the observation that any one metabolic network exists in a vast space of possible metabolic networks. The approach uses recently developed techniques [46], [47] to create unbiased arbitrary large samples of networks from this space, where each network of the sample has a specific property, such as a given number of reactions, a given number of carbon sources it can use, and a given set of molecules that it can synthesize. The underlying sampling technique, Markov Chain Monte Carlo sampling [48], [49] is a widely used approach with a well-developed statistical theory [50]–[54]. We use it to quantify the trade-offs and thus the design constraints imposed by important metabolic network properties. Specifically, a first part of our analysis focuses on three main properties. The first is nutrient flexibility N, that is, the number of different carbon sources a metabolic network can utilize as sole carbon sources. The second is a network’s biosynthetic ability B, that is, the number of biomass molecules that it can synthesize. The third is the number of reactions R in a network. We then extend our analysis to further properties, such as the biosynthetic flux S, the rate at which biomass molecules are synthesized, and the amount W of waste produced.
Results
Each Additional Nutrient Requires on Average Two Additional Reactions
Due to the presence of alternative metabolic routes that connect many pairs of molecules, most metabolic systems are able to tolerate genotypic changes, such as the deletion of enzyme coding genes. For instance, 80% of single gene deletions in budding yeast have no detectable phenotypic effect in standard laboratory environments [55]. More specifically, metabolic systems are to some extent robust against deletions of enzyme coding genes, because the resulting elimination of metabolic reactions from a metabolic network does not necessarily affect cell viability [9], [11], [13], [23]–[25], [56].
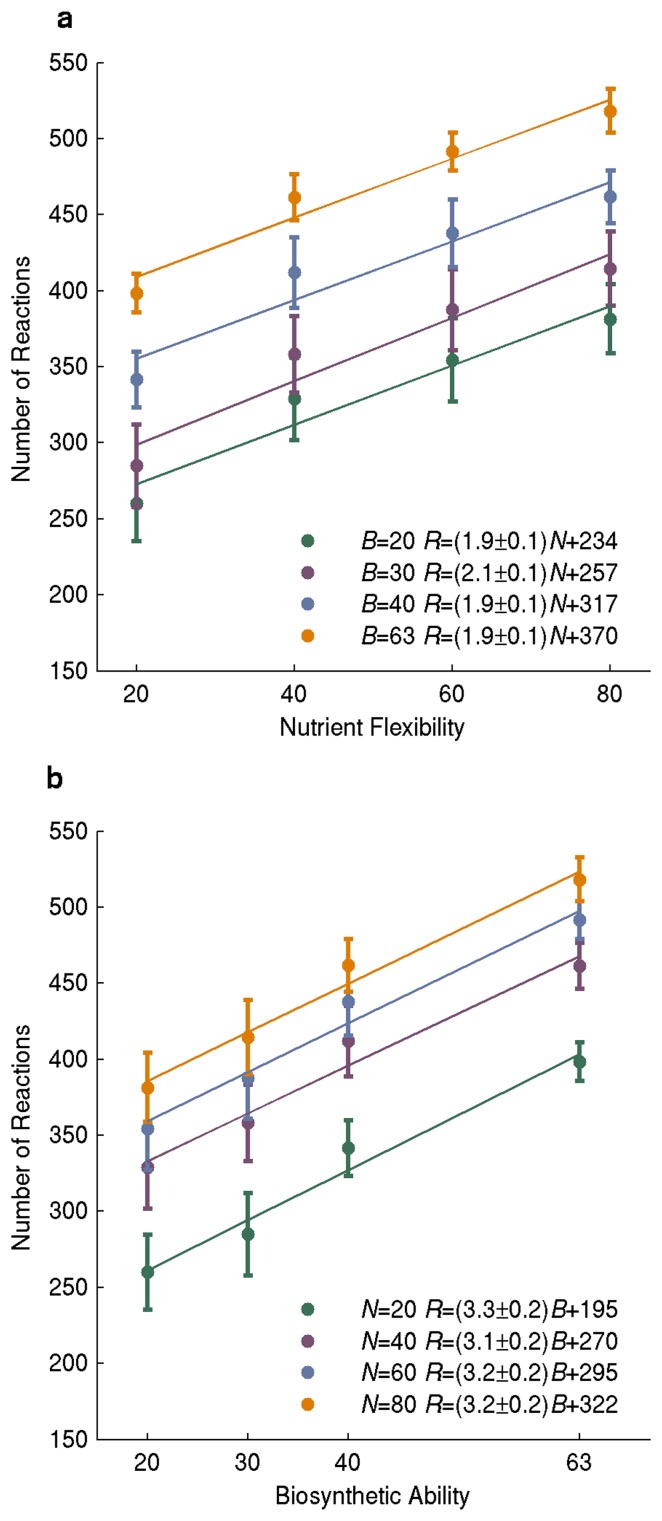
We first wanted to explore how the minimally needed number of reactions in a network depends on the network’s nutrient flexibility and its biosynthetic abilities. For example, a network that can synthesize biomass on an increasing number of different carbon sources will require more metabolic reactions. But how many more? To answer this question we created random minimal networks (see Methods) with a given nutrient flexibility N, that is, networks that can use N sole carbon sources to synthesize all biomass components, but in which every single reaction is essential, such that no reaction can be removed without abolishing viability of the network on at least one of the carbon sources. More specifically, we created 50 random viable networks that can use N = 20, 40, 60, and 80 carbon sources as sole carbon sources (for a total of 200 networks), and that could synthesize all B = 63 E. coli biomass components. We then studied the relationship between N and the number of reactions in these minimal networks (Figure 1a, orange data points). Linear regression analysis showed that the number of needed reactions increases by a number that is statistically indistinguishable from two for every additional carbon source that a network is required to be viable on. Specifically, we found R to depend on N as R = (1.9±0.1)N +234, where the number 0.1 indicates the 95 percent confidence interval of the regression coefficient (see Text S1 for details). We then asked whether the same relationship between N and R also holds if we vary biosynthetic ability. To this end, we repeated the analysis just described, but for networks that are able to synthesize B = 20, B = 30, and B = 40 biomass components. The slope of the regression line was indistinguishable from that of B = 63 in all three cases (Figure 1a, green, purple, and blue data; see Figure S3 for distributions of R). In other words, regardless of a network’s biosynthetic abilities, every additional carbon source that a network is required to be viable on requires on average two additional metabolic reactions.
Figure 1. The number of required reactions increases with nutrient flexibility and biosynthetic ability.
The vertical axis shows the number of reactions in minimal networks as a function of a) nutrient flexibility and b) biosynthetic ability. Dots and length of error bars correspond to means and one standard deviation based on a sample of n = 50 minimal networks. Solid lines indicate linear regression lines for different values of B in a) and N in b). Numerical estimates of regression coefficients with 95% confidence intervals are given in the inset, in the format y = (a±e)x+b, where a is the regression coefficient, e the confidence interval, and b is the intercept of the regression line with the vertical axis.
Each Additional Biomass Molecule Requires Three Additional Reactions on Average
We next analyzed the relationship between biosynthetic ability and the numbers of reactions in greater detail. To this end, we used the same set of random viable minimal networks that we used in the previous analysis. First, we analyzed the number of reactions in 50 random viable minimal networks with the ability to synthesize B = 20, 30, 40, or 63 randomly chosen biomass components, that is, we analyzed a total of 200 networks. Each of these networks was required to be viable on N = 80 different sole carbon sources. We found that the number of reactions needed for viability under these conditions increased approximately linearly with biosynthetic ability (Figure 1b, orange), such that every additional biomass molecule required approximately 3 additional reactions Specifically, R = (3.3±0.2)B +195. We then asked again how this relationship between biosynthetic ability and number of reactions depends on N, and thus repeated this analysis for networks viable on N = 20, N = 40, and N = 60 sole carbon sources. The slope of the regression line was indistinguishable for the different values of N (Figure 1b, green, purple, and blue data), but the intercept differed, as one might expect from the analysis of the previous section.
The results of pairwise regression analysis are easy to display graphically and to interpret intuitively, which is why we use it here. However, where more than two quantities vary, pairwise regression analysis is insufficient to study dependencies among all of them. We thus also carried out a multiple regression analysis using all 800 minimal networks, where nutrient flexibility and biosynthetic ability were independent variables, and where the reaction number was the dependent variable. Not unexpectedly, the pairwise regression coefficients estimated from the multiple regression analysis were statistically indistinguishable from those estimated from the pairwise analysis above (R = 2.0N+3.2B+171). Overall, variation in the two independent variables explains 87 percent of the variation in the number of reactions of a metabolic network.
Our analysis thus far was only concerned with the qualitative question whether networks can synthesize a given number B of molecules, not with the rates at which these molecules can be synthesized. One would therefore expect that our observations are not sensitive to variation in the proportions in which biomass molecules are to be synthesized. An additional analysis (Figure S1) confirms this expectation.
Network Size does Not Significantly Influence Biosynthetic Flux
Engineered metabolic networks that produce one or more desired products at a high rate are a key goal of biotechnology. For our purpose, it is therefore important to ask whether the maximal rate at which biomass can be synthesized by a network depends on B and N. We here used the ability of FBA to predict the maximal biosynthetic flux (S) of viable networks to answer this question (see Methods).
Our analysis thus far was based on minimal networks, but it is possible that biosynthetic flux depends on the number of reactions in networks that are larger than minimal networks. We therefore first analyzed 1.6×104 random viable networks that are not minimal and that differ in size between R = 400 and R = 800 reactions. (From here on, we will use the term random networks to refer to those networks that are not minimal, unless stated otherwise.) We found that network size does not influence biosynthetic flux for networks able to synthesize all B = 63 E. coli biomass molecules. That is, the regression coefficient describing their relationship is statistically indistinguishable from zero (f = rR, with r = 3×10−5±7×10−5, n = 1000 networks). The same holds for networks that can synthesize B = 20, B = 30, and B = 40 biomass molecules. (See Figure S2 for flux distributions.).
High Biosynthetic Flux is Usually Achieved by a Modest Number of Active Reactions
We just showed that the number of reactions in a network does not affect its biosynthetic flux. However, it is well-known that only a subset of a network’s metabolic reactions are usually active in any one environment, that is, they have nonzero flux [12]. We thus wanted to know whether a relationship exists between biosynthetic flux and the number of active reactions, RA. This is indeed the case, based on an analysis of all our 1.6×104 random networks viable on glucose as a sole carbon source. More specifically, this relationship is strongly negative (Pearson's r = -0.65 for biosynthetic flux on glucose). That is, the greater the number of active reactions, the lower the biosynthetic flux of a network.
We next used linear regression to ask whether network size R itself influences the number of active reactions RA. The answer is no. Network size has no statistically significant influence on the number of active reactions for any combination of values of N and B. This observation is in agreement with earlier results by Nishikawa et al. [12], which showed that the number of active reactions in a network is not sensitive to the size of the network.
We subsequently asked whether synthesis of each additional biomass molecule also needs more active reactions. The answer is yes, as shown by linear regression analysis (3.6–4 additional reactions, on average, per biomass molecule).
The More Biomass Molecules a Network Synthesizes, the Smaller is its Biosynthetic Flux
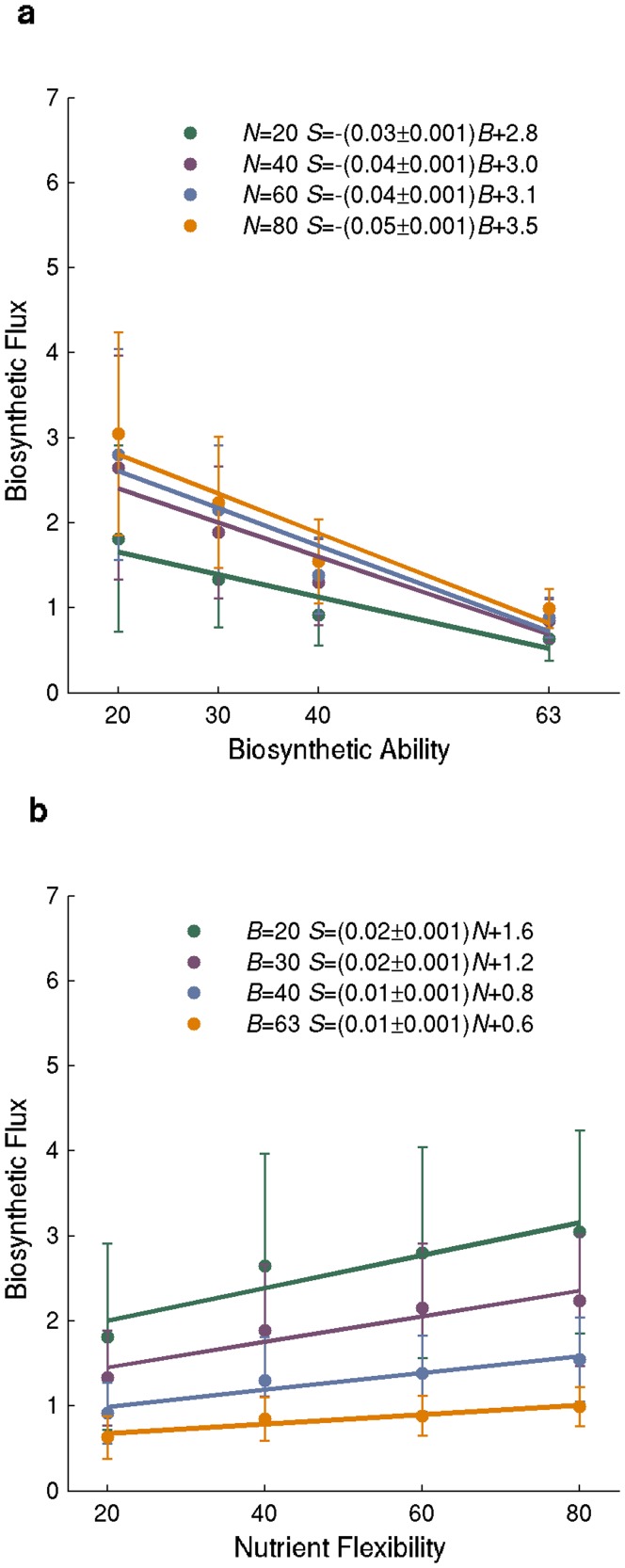
We next wanted to explore why the number of active reactions is negatively correlated with biosynthetic flux. Our observations so far show that the number of active reactions increases with biosynthetic ability. We thus hypothesized that increased biosynthetic ability entails smaller biosynthetic flux, because biosynthetic flux should decrease as the number of biomass molecules to be synthesized grows, given a constant nutrient supply. To test this hypothesis, we computed the correlation between the number of molecules synthesized and biosynthetic flux in multiple environments for all 16000 networks we had generated (see Methods). This correlation is significantly negative (Pearson’s r = -0.60; P<10−300). Each molecule to be synthesized decreases biosynthetic flux by 0.05 mmoles per g DW per hour, equivalent to a 6 percent decline relative to E.coli’s computed biosynthetic flux. Next, we analyzed networks viable on 80 different sole carbon sources that were able to synthesize B = 20, 30, 40, or 63 randomly chosen biomass components (1000 networks each, for a total of 4000 networks). Biosynthetic flux under these conditions also decreased approximately linearly with biosynthetic ability (Figure 2a, orange data). Specifically, every additional biomass molecule that a network needs to synthesize decreases the biosynthetic flux by approximately 0.05 mmoles of biomass per g DW per hour (6 percent; S = -(0.05±0.001)B +3.5). Similar relationships also hold for networks viable on N = 20, N = 40, and N = 60 sole carbon sources (Figure 2a, green, purple, and blue data). They show that, every additional biomass molecule to be synthesized reduces biosynthetic flux by 3–6 percent. Taken together, these observations help explain the negative relation between the number of active reactions and biosynthetic flux. Networks that can synthesize more biomass molecules need more active reactions. Given a constant nutrient supply, the total biosynthetic flux that can be realized by these networks must decrease as the number of molecules that they can synthesize increases. The negative correlation between the number of active reactions and biosynthetic flux is a by-product of the latter two correlations.
Figure 2. Biosynthetic flux decreases with biosynthetic ability and increases with nutrient flexibility.
The vertical axis shows biosynthetic flux in mmoles per g DW per hour in random networks as a function of a) biosynthetic ability and b) nutrient flexibility. Dots and lengths of error bars correspond to means and one standard deviations based on a sample of n = 1000 minimal networks. Solid lines indicate linear regression lines for different values of B. Numerical estimates of regression coefficients with 95% confidence intervals are given in the inset, in the format y = (a±e)x+b, where a is the regression coefficient, e the confidence interval, and b is the intercept of the regression line with the vertical axis.
Biosynthetic Flux Increases Weakly with Nutrient Flexibility
We next explored how the nutrient flexibility of a network affects its biosynthetic flux. To this end, we analyzed 1000 networks each that can use N = 20, 40, 60, and 80 carbon sources (a total of 4000 networks) and that can synthesize B = 63 biomass molecules. (Figure 2b, orange data points). We found that biosynthetic flux increases weakly but significantly with nutrient flexibility (S = (0.01±0.0001)N +0.6 mmoles of biomass per g DW hour, n = 1000 networks) for every additional carbon source that a network is viable on. This is equivalent to 1 percent of increase relative to E. coli's biosynthetic flux on glucose. Quantitatively similar relationships hold for networks able to synthesize B = 20, B = 30, and B = 40 biomass components (Figure 2b, green, purple, and blue data). We next turn to a variable that can help explain this association: waste production.
High Biosynthetic Flux Means Less Waste
Some 300 transport reactions are described in E. coli [57], many of which transport waste products either from the periplasm or the cytoplasm to the extracellular space. Metabolic networks may differ in the extent to which they produce waste products that are excreted from cells. Waste products may include molecules that are not biomass molecules (such as carbon dioxide or acetate), as well as excess biomass molecules [12], [57]. Waste production consumes resources, such as organic carbon, and will therefore reduce biosynthetic flux. With these considerations in mind, we next asked whether lower waste production might be responsible for the differences in biosynthetic flux we observed in networks with different nutrient flexibility.
Different metabolic networks may excrete different kinds of molecules, but to compare their waste production, it is useful to establish a common waste ‘currency’. Since our analysis is focused on carbon metabolism, we use the moles of carbon per g DW per hour as our unit of waste production. (Note that these moles of carbon may well come from a broad spectrum of different molecules.).
We first wanted to know if waste has any influence on biosynthetic flux. The answer is yes, based on an analysis of all our 1.6×104 random viable networks on glucose as the sole carbon source. This relationship is strongly negative (Pearson's r = -0.66) That is, the greater waste production is, the lower the biosynthetic flux.
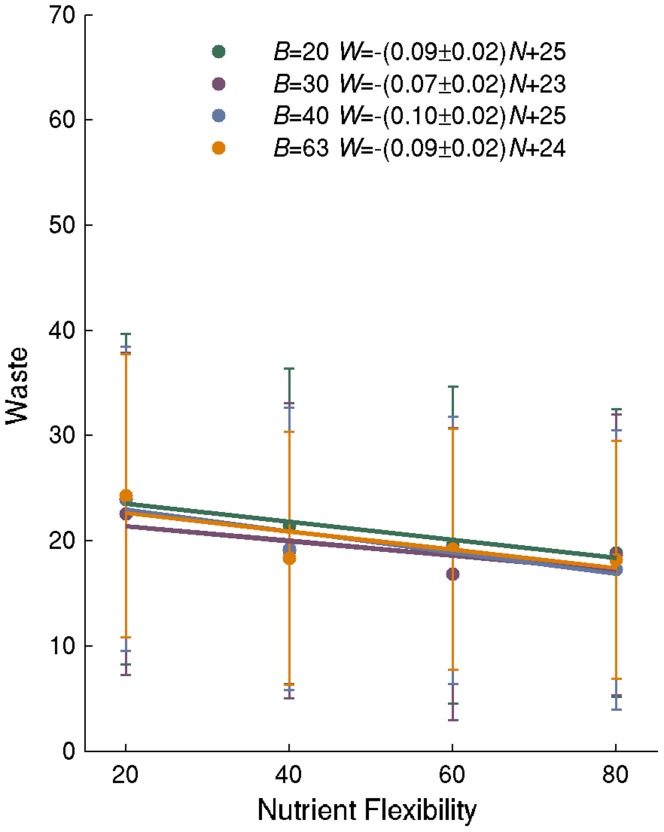
In the last section, we showed that for each additional carbon source a network was required to be viable on, biosynthetic flux increased by 0.01–0.02 mmoles of biomass per g DW and hour (Figure 3B). If we express biomass synthesis instead as moles of carbon (in synthesized biomass) per g DW and per hour, biosynthetic flux increases on average by 0.1 mmoles of carbon per g DW hour with increasing nutrient flexibility. We hypothesized that this increase can be explained through the influence of nutrient flexibility N on waste production W. To this end, we first analyzed random viable networks that vary in N and that can synthesize all B = 63 molecules E. coli biomass molecules (Figure 3, orange data points). We found that waste production decreased by a number that is statistically indistinguishable from 0.1 mmol of carbon per g DW hour (W = (-0.09±0.02)N +24, n = 1000 networks) for every additional carbon source that a network is viable on. Statistically indistinguishable relationships exist for networks with B = 20, B = 30, and B = 40 biomass components. (Figure 3, green, purple, and blue data). These observations suggest that the positive influence of nutrient flexibility on biosynthesis flux comes from reduced waste production. We later discuss experimental evidence supporting this observation.
Figure 3. Waste production decreases with nutrient flexibility.
The vertical axis shows waste that is excreted carbon in mmoles per g DW hour in random networks as a function of nutrient flexibility. Dots and lengths of error bars correspond to means and one standard deviation based on a sample of n = 1000 minimal networks. Solid lines indicate linear regression lines for different values of B. Numerical estimates of regression coefficients with 95% confidence intervals are given in the inset.
In a final analysis related to waste production, we studied the relationship between biosynthetic ability and waste production, and found no significant such relationship.
The Variables We Considered can Explain 84 Percent of the Variance in Biosynthetic Flux
Thus far, we have considered five variables and how they influence biosynthetic flux. These are network size R, nutrient flexibility N, biosynthetic ability B, waste production W, and numbers of active reaction RA. To understand how biosynthetic flux depends on not just one but all of these variables, we carried out a multiple regression analysis with flux as the dependent variable. This regression analysis showed that the variables we analyzed explain 84 percent of the variation in biosynthetic flux (R2 = 0.84).
Synthesis of Biochemically Related Molecules Requires Fewer Reactions, because it Produces Less Waste
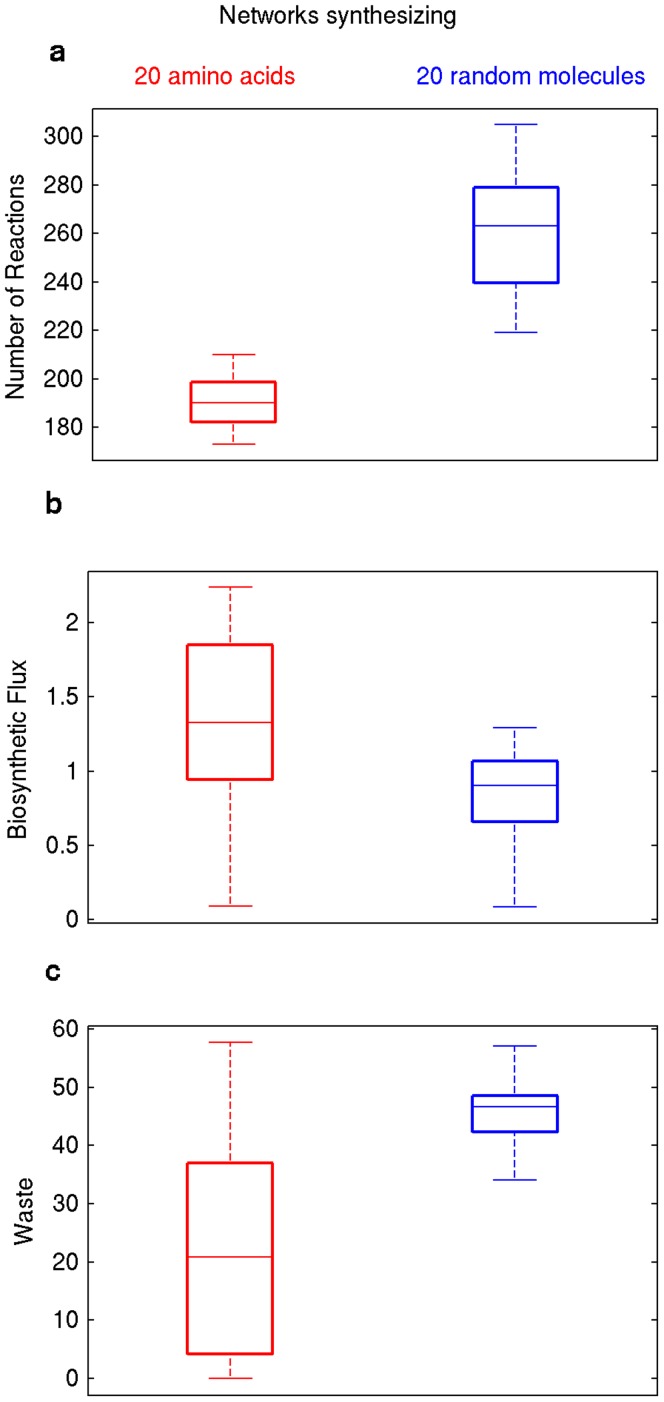
In our analysis of how required reaction numbers depend on a network’s biosynthetic abilities, we have purposely focused on randomly chosen biomass precursors, as they give us an unbiased view of this dependency. However, this relationship may change if one considers biochemically related biomass molecules. To obtain some insights how it changes, we next studied a group of molecules whose known biosynthesis pathways share several reactions. These are the 20 proteinaceous amino acids [58]. Figure 4a shows the number of reactions in minimal networks that can synthesize all 20 proteinaceous amino acids (left panel), as well as the number of reactions needed to synthesize 20 randomly chosen biomass molecules (right panel). Amino acid synthesizing networks need 191±10 (mean ± one standard deviation) reactions, 27 percent fewer than the 261±24 reactions needed to synthesize 20 arbitrary molecules. This difference is highly significant (P<10−10, Mann-Whitney U-test, n = 100) [59].
Figure 4. Synthesis of biochemically related compounds require less reactions, achieve higher biosynthetic flux with less waste.
Box-plot of a) number of reactions, b) biosynthetic flux in mmoles per g DW hour, and c) waste in mmoles per g DW per hour in minimal networks synthesizing twenty amino acids (left panel) and synthesizing twenty random biomass molecules (right panel). Horizontal lines in the middle of each box mark the median. The edges of the boxes correspond to the 25th and 75th percentiles. Data is based on a sample of n = 80 for each box.
In minimal networks every reaction is active under the conditions we study. Our observations in the last paragraph thus also show that amino acid biosynthesis needs fewer active reactions than biosynthesis of arbitrary biomass molecules. One of our analyses above showed that fewer active reactions also imply higher biosynthetic flux, which raises the question whether the 20 amino acids can be synthesized at higher rates. The answer is yes (Figure 5b). Minimal networks that synthesize 20 arbitrary biomass molecules synthesize them 35 percent more slowly than minimal networks that synthesize 20 amino acids (P<10−13, n = 80, Mann-Whitney U-test) [59].
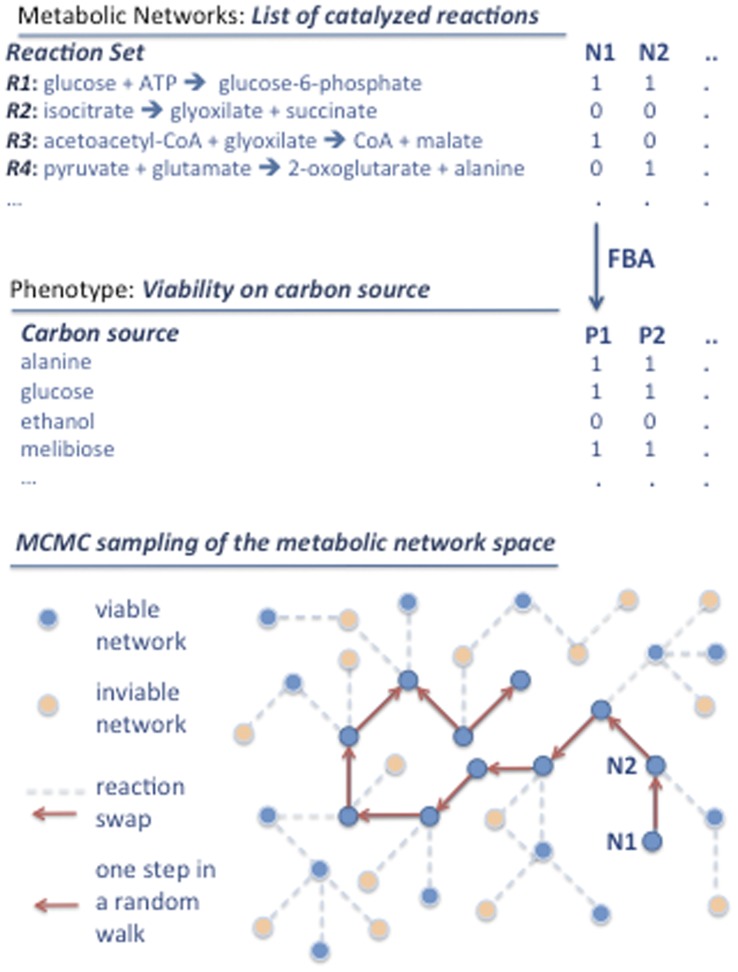
Figure 5. Exploration of a metabolic network space.
Metabolic networks can be viewed as subsets of enzyme-catalyzed metabolic reactions in a global reaction set. Formally, they can be represented as binary vectors listing the reactions catalyzed by enzymes in an organism, as indicated for two hypothetical metabolic networks (N1, N2) in the figure. Metabolic phenotypes are computed from metabolic networks using FBA. They can be represented as binary vectors indicating the carbon sources (i.e.: alanine, glucose, melibiose,…) on which a network is viable, that is, on which it can synthesize a given set of (biomass) molecules. Neighboring networks (blue circles linked by edges) differ by a single reaction swap (edges between circles) that leaves the metabolic phenotype unchanged. A reaction swap consists of two changes: one random reaction addition (R4 in the example) and one random reaction deletion (R3 in the example). A series of successful reaction swaps is called a random walk (indicated by red arrows). The Markov Chain Monte Carlo (MCMC) technique allows one to randomly sample networks with a given phenotype by generating long random walks through genotype space, where each step in a walk consists of a reaction swap. The advantage of using reaction swaps is that they leave the number of reactions constant.
We next hypothesized that these dependencies might be explicable as a by-product of a lower cost of amino acid synthesis. If amino acids have fewer carbon molecules than the average biomass molecule, both fewer steps would be needed to synthesize them and a higher synthesis rate could be achieved. To test this hypothesis, we computed the average carbon content of all 20 amino acids, weighted by each amino acid’s stoichiometry in biomass, as 1.28±0.60 mmoles of carbon per mmole biomass. This molecular weight turned out not to be smaller but significantly larger than the carbon content of 20 random biomass molecules, also weighted by their stoichiometry in biomass (0.87±0.15 mmoles of carbon, P<2.2×10−16, one sample t-test, for n = 105 sets of 20 random biomass molecules). This means that low amino acid cost cannot explain a higher rate of amino acid production.
We next asked whether lower waste production might be responsible for the lower biosynthetic flux we observed. We therefore analyzed the quantity of secreted waste products in 80 networks that synthesize 20 amino acids, and in 80 networks that synthesize 20 random biomass compounds. Waste production is indeed significantly lower in networks that synthesize amino acids (P<10−17, Mann-Whitney U-test) [59]. Specifically, amino acid synthesizing networks produce 47 percent less waste than networks that synthesize arbitrary molecules. They excrete on average 22.8 mmoles of carbon waste per g DW hour (Figure 5c).
To provide a concrete example of a prominent waste molecule, consider the amount of secreted carbon dioxide. For each mole of carbon entering a network in the form of glucose, amino acid synthesizing networks excrete 0.18±0.12 (mean ± one standard deviation) moles of carbon per g DW hour as carbon dioxide, whereas networks synthesizing 20 arbitrary molecules excrete 0.31±0.06 mmoles of carbon per g DW hour as carbon dioxide (P<10−25, Mann-Whitney U-test) [59]. In sum, waste production is an important factor in explaining the smaller costs of amino acid biosynthesis compared to arbitrary biomass molecules.
Discussion
We here studied typical properties of metabolic networks with recently developed techniques to create random and unbiased samples of metabolic networks from a vast space of such networks [46], [47]. Specifically, we studied the quantitative relationships between 6 different properties. These are the number of alternative sole carbon sources N that a network can use – its nutrient flexibility –, the number of (biomass) molecules B it can synthesize, the rate S at which it can synthesize these molecules, the number of reactions R in the network, the number of active reactions RA, that is, reactions that have nonzero metabolic flux, and the amount of waste W the network produces.
We focused first on how the number of minimally needed reactions R depends on N and B, because this number of reactions is an important design variable. Smaller networks would be easier to design and smaller genomes would be easier to synthesize [35]. More than that, biosynthetic processes are more controllable and predictable in a small metabolism [1], [18]. We found that for every additional molecule to be synthesized, a network needs on average three additional reactions.
In a related analysis, we focused on nutrient flexibility. The ability of a metabolic network to sustain life on multiple sources of chemical elements and energy is highly desirable in industrial production processes [31]. Examples include biofuel production from cellulosic biomass. Cellulosic biomass contains both hexoses (e.g., glucose) and pentoses, whose major constituent is xylose [31], [60]. Many yeast species, for instance, do not consume pentoses, and need to be engineered to have greater nutrient flexibility to make biofuel production more efficient [60]. In our analysis, we found that for every additional carbon source to be utilized, a network needs on average two additional reactions. Thus, it is more expensive, in terms of the number of needed reactions, to synthesize additional molecules than to synthesize the same molecules but from a larger number of alternative carbon sources.
Anecdotal evidence for the tendency that increased nutrient flexibility requires larger networks is provided by two networks for which nutrient flexibility is experimentally known [57], [61]. These are E. coli where N = 54 and R = 1396 and Bacillus Subtilis where N is smaller (N = 41) and so is R (R = 769). We also compared the computationally predicted number of carbon sources that can be utilized by metabolic models of seven organisms (Column 4 of Table 1). Although based on few data points, this analysis suggests that there is a significant correlation (Pearson’s r = 0.9, P = 0.005) between the in silico nutrient flexibilities and networks sizes of these model organisms.
Table 1. Biosynthetic ability B, number R of metabolic reactions (size of metabolic networks), number RA of number of active reactions, and number N of carbon sources predicted to be catabolized in computational metabolic models of various organisms.
| Organism | B | R | RA | N | References |
| Buchnera Aphidicola | 43 | 205 | 183 | 2 | [97] |
| Helicobacter pylori | 52 | 394 | 298 | 35 | [98] |
| Staphlococcus aureaus | 58 | 534 | 286 | 41 | [99] |
| Bacillus Subtilis | 59 | 769 | 327 | 97 | [61] |
| Methanosarcina barkeri | 63 | 531 | 352 | 6 | [100] |
| Escherichia coli | 67 | 1396 | 388 | 175 | [57] |
| Mycobacterium tubeculosis | 93 | 836 | 408 | 39 | [101] |
We took the number of synthesized molecules B, and the number of metabolic reactions R from the genome scale metabolic network reconstructions of these organisms, as listed in the column ‘References’. Active reactions are reactions that have non-zero flux as determined by FBA. We computed nutrient flexibilities by testing the viability of metabolic models on all known carbon containing metabolites that are educts or products of reactions in the global network by FBA, and assumed a metabolite to be a carbon source if the metabolic model can utilize it.
As a result of dependencies between N and R, the minimally needed number of reactions varies widely among the networks we study. For example, for networks that can use 20 alternative carbon sources, it ranges from an average of 260 reactions (which would correspond to, for comparison, 19% of the whole E. coli network) needed to synthesize B = 20 molecules to an average of 381 reactions (29% of the E. coli network) to synthesize all B = 63 biomass molecules of E. coli. For networks that can use 80 alternative carbon sources, the largest number we studied, it increases from 398 (27%) to 518 reactions (37% of the E. coli network) as B increases from 20 to 63.
N and B can explain the vast majority (87%) of the variance in R, in a relationship that is close to linear. Note that the proportions in which biomass molecules are to be synthesized are immaterial, as long as the metabolic reactions to synthesize each required molecule are present. This is why the stoichiometry of biomass composition would not influence the relationships we observe. The remaining 13% of variance is unexplained by linear relationships among the variables we considered. Such unexplained variance could have multiple sources, and especially nonlinear relationships among the variables. For example, as the number of biomass molecules to be synthesized increases, fewer and fewer additional reactions might be required, because most precursors of the additional biomass molecules may already be synthesized by existing reactions.
We showed that the reactions needed to utilize an additional carbon-containing molecule or to synthesize an additional biomass molecule typically involve the additional molecule. For example, the utilization of an additional carbon source requires a reaction that catabolizes that molecule. The synthesis of an additional molecule requires a reaction that produces that molecule. We emphasize that the numbers of reactions required for an additional carbon source or biomass molecule reflect statistical patterns, averages over multiple networks. Some additional carbon sources or biomass molecules need more reactions than the average we identified, whereas others need fewer. (For some examples see Table S3, Table S4, Text S2 and Text S3.).
We also studied the number of active reactions – reactions with nonzero flux on a given carbon source. Quantitatively, we found that the number of active reactions increases by 3.6–4 reactions for each additional biomass molecule to be synthesized. Qualitatively, this tendency – increasing synthetic ability requires more active reactions – is not surprising, it is also corroborated by empirical data. Table 1 shows several well-known organisms with known metabolic network sizes R and biosynthetic abilities B. Note that, with exceptions, the number of active reactions RA tends to become higher as B increases for these organisms. Albeit based on few species, a regression analysis of the data in Table 1 shows that the relationship between RA and B is similar to what we find in our much larger samples of random networks (RA = 3.4B+120). Our approach has the advantage of allowing us to make quantitative statements about the number of active reactions required to synthesize additional molecules that are not just based on few, well studied organisms, as in Table 1, but on arbitrarily large and random samples of metabolic networks.
The identity of active metabolic reactions can depend strongly on the nutrient environment [12], [46], [62]. A reaction active in one environment may be inactive in another. Organisms would cope with such variation by regulating the activity of reactions, for example by regulating the expression of enzyme-coding genes or by regulating the enzymes themselves [63]. The importance of reaction activity in our analysis thus points towards the importance of regulating metabolic flux in response to the environment. Optimal regulation of enzymes is a key challenge in metabolic engineering [64], [65]. It will be an even greater challenge in a synthetic metabolism, especially if such a metabolism is to function efficiently in multiple chemical environments.
A second major set of analyses focused on how metabolic network properties influence biosynthetic flux S. These analyses are motivated by the fact that high synthesis rates of one or more target molecules are important goals of metabolic engineering [19]–[21]. Our first major observation in this regard concerns the number of reactions in a metabolic network. This number has virtually no influence on the attainable biosynthetic flux S. In stark contrast, the number of active reactions – reactions with nonzero flux – shows a strong negative association with biosynthetic flux. That is, the higher the biosynthetic flux is, the smaller is the number of active reactions. (This analysis is based on networks that have more than the minimally necessary number of reactions in a given environment. In minimal networks, all reactions must be active.) This observation is consistent with earlier work based on four microbial metabolic networks [12].
A second important influence on biosynthetic flux S is the number of biomass molecules to be synthesized. Specifically, every additional such molecule reduces S by approximately 5 percent. This influence is stronger than the influence of nutrient flexibility, where each additional carbon source changes biomass synthesis flux by 1 percent. That nutrient flexibility influences biosynthesis rates is known from experiment [31], [66]–[69]. For example, engineered yeast strains capable of growing on more carbon sources achieve higher product yields [31].
The amount of waste W that a network produces is a third, and the most important influence on biosynthetic flux S. Metabolic networks generally excrete waste, for example, in the form of carbon dioxide [12], acetate [12], [70], pyruvate [70] or glycerol [60], [66]. Not only does this mean wasted resources, some waste products may also be toxic and interfere with goals of metabolic engineering [27]. We found that biosynthetic flux S shows a strong negative correlation with waste production W in the form of excreted carbon. This observation is in qualitative agreement with experimental observations [60], [66], [70]. For example in E. coli, Weber et al [70] demonstrated that excretion of methylglyoxal, D- and L-lactate, pyruvate, and acetate decreases growth rates. We also observed that networks with higher nutrient flexibility produce less waste, such that for each additional carbon source, networks produce 0.1 mmoles less in excreted carbon waste. This relationship can help explain the positive influence of N on flux S. Experimental support for this observation exists as well. For example, Wisselink et al. [31] observed that increased ethanol production in engineered yeast with higher nutrient flexibility is caused by reduced production of the waste products xylitol and arabinitol. Relatedly, the elimination of glycerol formation increases the yield of ethanol up to 10 percent in yeast [66]. Ho et al. [60] report similar observations.
We also found that an analogous association does not exist for B. Networks seem to produce indistinguishable amounts of waste products regardless of how many biomass molecules they synthesize.
Overall, the quantities we examined can explain 84 percent of the variance in biosynthetic flux S we observed. This means, that these quantities account for most of the variation in the biosynthetic flux, and are thus important factors in the design of a synthetic metabolism.
In our final analysis, we showed that the nature of the molecules to be synthesized can influence biosynthetic flux. For example, in biotechnological applications, a metabolic network may need to synthesize molecules that are closely related, and whose biosynthetic pathways are therefore similar, in the sense that they share many reactions. Examples include vitamins and coenzymes [71], taxols and related taxoids [72], hydrocarbons and related ether lipids [73], as well as amino acids [74]. We examined the influence of biochemical relatedness by studying metabolic networks that synthesize all 20 amino acids, and by comparing them with networks that synthesize 20 arbitrary biomass molecules. In this analysis, we found that amino acids could be synthesized with fewer reactions, which reflects their biosynthetic relatedness. They can also be synthesized at a rate that is 35 percent higher, because their biosynthesis produces 47 percent less waste as excreted carbon. These differences are substantial, given that metabolic engineering in biotechnological processes typically increases synthesis rates of desired products by 5–10 percent [66], [75]–[77].
We conclude with some caveats and limitations of our analysis. First, we are well aware that there is still a gap between current metabolic engineering and synthetic biology experiments on the one hand, and theoretical studies such as ours on the other. We see the value of studies like ours as providing quantitative reference points for future experimental work in this area.
Second, flux balance analysis, on which our approach rests, assumes a metabolic system that is in a steady-state, such as might be achieved by a microbial population growing under a constant nutrient supply. This assumption ignores possible additional constraints that regulation and enzyme kinetics exert on a metabolism [78], [79]. While achieving proper regulation remains a big challenge in synthetic biology, we note that some of our observations are unlikely to be affected by this assumption. For example, relaxing this assumption would not reduce the minimal number of reactions needed for a given biosynthetic task.
Third, we focus on typical network properties, that is, properties of metabolic networks sampled at random from a large space of such networks. Optimization procedures that search metabolic networks space systematically may be able to identify individual networks whose properties deviate from those we identified as typical. For example, they may identify networks that are able to synthesize biomass with even fewer reactions or at higher rates. Such procedures have been successful in other combinatorial optimization problems. They include simulated annealing [80], [81], bi-level optimization [82], [83], OptFlux [84], convex optimization [85] and evolutionary optimization [86], [87]. The extent to which they can identify networks with superior design remains an important subject of future work.
Fourth, the expression of enzymes itself has a metabolic cost. This cost may be reduced by regulating enzyme expression depending on the nutrient environment (through a regulatory machinery whose expression may itself be costly.) We did not consider costs like these here, because they are poorly understood on a quantitative level. Their analysis also remains an important goal for future work.
Finally, we note that an organism’s genome encodes more than metabolism. Nonmetabolic genes, even in small genomes serve roles in systems that allow cell motility, signaling, secretion, and defense [14]. The heterogeneity of these systems will make general, quantitative statements about design constraints for entire genomes more difficult to obtain. In this regard, we note that the proportion of a gene’s genome devoted to metabolism increases in small genomes. For example, in the free-living E. coli with a large genome, fewer than 75 percent of genes exert metabolic functions [88]. In contrast, in some endosymbionts including Buchnera, Wiggleswothia glossinidia and Thiomicrospira crunogena more than 80 percent of genes are devoted to metabolic functions, and in yet others, such as the gamma proteobacteria, Blochmanni floridanus and Wolbachia pipientis more than 95 percent have metabolic functions [14]. Thus, the constraints we identified here would affect the majority and perhaps the vast majority of a synthetic minimal organism’s genome.
Methods
The metabolic genotype of an organism comprises all genes that encode metabolic enzymes. This genotype can be compactly represented through a list of reactions that can take place in the organism and that are catalyzed by enzymes [36], [37], [89]–[92]. Each metabolic genotype exists in a vast metabolic genotype space that contains all possible metabolic networks – all possible combinations of reactions drawn from a set of biochemically feasible reactions – that could take place in a living system. According to our current knowledge, there are more than 5000 such reactions, which means that metabolic genotype space comprises 25000 possible metabolic networks. The metabolic genotype or metabolic network of any one organism can be viewed as a point in this space. Two genotypes differing from each other in a single reaction are neighbors in this space. We refer to a network’s metabolic phenotype as the spectrum of chemical environments – defined by nutrients these environments contain – on which the network can synthesize a predetermined spectrum of molecules, as well as the rate at which it can synthesize these molecules. We call a network viable in a given environment if it can synthesize all these molecules in the environment.
Flux Balance Analysis
Flux balance analysis (FBA) is a constraint-based modeling approach that predicts steady state metabolic fluxes – rates of substrate to product conversion – for all metabolic reactions in a metabolic network. It can thus also predict the biomass yield and other complex metabolic attributes of a metabolic network [78], [79]. Because FBA does not need kinetic information, but only stoichiometric information about the reactions involved, it is a widely used approach for analyzing the metabolism of well-studied organisms such as E. coli and S. cerevisiae [40], [78], [79]. The needed stoichiometric information comes from experimental biochemical analysis, as well as from comparative analyses of enzyme-coding genes in different, completely sequenced genomes. This information is encapsulated in a stoichiometric matrix S of dimensions m×n, where m denotes the number of metabolites, and n is the number of reactions in a network [40], [79]. FBA assumes that a metabolic network is in a metabolic steady-state, such as may occur in a microbial population that proliferates in an unchanging environment. Because mass needs to be conserved under these conditions, any vector v of metabolic fluxes – the rates at which a network’s metabolic reactions convert substrates into products – must satisfy the equation.
This equation typically has a large solution space of allowable fluxes. The size of this space can be reduced somewhat by placing biochemically motivated constraints on the irreversibility of some reactions and on maximal flux magnitudes [93]. In the (reduced) solution space, FBA then uses linear programming to identify regions in the space that maximize a quantity of interest, which can be represented by a linear objective function Z [40], [79]. More specifically, the linear programming formulation of an FBA problem can be written as:
| (1) |
where the vector c T stands for a transposed (T) array c of scalar coefficients that define the objective function. Vectors a and b contain lower and upper limits of reaction fluxes in the flux vector v, respectively. A particularly important quantity to be maximized is the rate of synthesis of a given set biomass molecules. We refer to this quantity as the biosynthetic flux (in units of mmol per g DW per hour) of a metabolic network. We used the software packages CPLEX (11.0, ILOG; http://www.ilog.com/) and CLP (1.4, Coin-OR; https://projects/coinor.org/Clp) to solve all linear programming problems that arise in this study.
Biomass Molecules
The starting point of our analysis was a set of 67 biomass molecules from E. coli [57]. We used these molecules, because E. coli’s biomass composition is well studied, and many of its components – amino acids, nucleotides, etc. – also occur in most other free-living organisms. In addition, E. coli and its biomass molecules are highly relevant to biotechnological applications [18], [19], [21]. For some of our analyses, we needed to vary the spectrum of metabolites that a metabolic network needs to synthesize. Among the 67 E. coli biomass molecules, we allowed all but four molecules to vary. These four molecules are the “currency metabolites” adenosine 5′-diphosphate (ADP), phosphoric acid (Pi), pyrophosphoric acid (PPi), and hydrogen ions. A complete list of the biomass molecules we used can be found in Table S1.
Carbon Sources
Any one metabolic network can only synthesize biomass if its chemical environment contains specific nutrients [94]. In FBA, these nutrients are represented by special exchange reactions that reflect transport of nutrients into the cell. The environments we study are minimal chemical environments that contain a single molecular source for each essential chemical element. These sources are oxygen, ammonium, inorganic phosphate, sulfate, sodium, potassium, cobalt, iron, protons, water, molybdate, copper, calcium, chloride, magnesium, manganese and zinc, as well as a single source of carbon. We study how variation in carbon sources constrains the composition of metabolic networks that needs to synthesize a given spectrum of molecules. We chose to vary carbon sources for this purpose, because of carbon’s centrality as a chemical element in biomass. A complete list of the carbon sources we used is given in the Table S2.
We computed the biomass growth of a network by taking the average of its biomass growth rates on each carbon source that the network is required to be viable on, except where mentioned otherwise.
Global Network
Some of our analyses use a “global reaction network”. This global network contains a comprehensive set of known biochemical reactions from multiple organisms, and it has universal biosynthetic abilities. We are fully aware that no such network is likely to exist in any one organism. We use this global network merely as a starting point for successive elimination of reactions, as described in the next section. We generated this global network by merging reactions from two sources, as described in more detail in [47]. The first is the LIGAND reaction database of the Kyoto Encyclopedia of Genes and Genomes (KEGG) [36], [37], [89]–[92]. The second is the complete metabolic reaction set of E. coli (iAF1260), which contains 1397 non-transport reactions [57]. We excluded from this network (i) all reactions involving polymer metabolites of unspecified numbers of monomer units, (ii) general polymerization reactions with uncertain stoichiometry, (ii) reactions involving glycans, due to their complex structure, (iv) reactions with unbalanced stoichiometry, and (v) reactions involving complex metabolites without chemical information about their structure [47]. After these procedures the global network contained 5906 internal (non-transport reactions) and 5030 metabolites.
Essential Reactions and Minimal Networks
A gene whose deletion abolishes the viability of an organism is called an essential gene. Analogously, an essential reaction in a metabolic network is a reaction that cannot be removed without abolishing the organism’s viability in a given environment. A minimal metabolic network is a network from which not a single reaction can be removed without abolishing viability in a given environment. In other words, all reactions of a minimal network are essential in that environment. We emphasize that a minimal network is not the smallest possible viable network, which would be difficult to find in a vast space of metabolic networks. We note that there may be multiple minimal networks, which contain different pathways among a set of possible alternate pathway from a nutrient to a biomass precursor. These networks need not have the same size. We also note that the number of essential reactions in any one non-minimal network may be smaller than the size of a minimal network, because non-minimal networks may contain alternate pathways able to by-pass any one reaction.
Generation of Minimal Networks
To analyze the smallest number of reactions that a network needs to have in order to (i) synthesize a given number B of biomass molecules on (ii) each of N different carbon sources, we created and analyzed many minimal networks with different values of B and N. Because there are astronomically many combinations of different values of B and N, and because the FBA approach we use is computationally expensive, we focused on subsets of such combinations, which we created as follows.
First, we created 10 different sets each of B = 40, B = 30, and B = 20 randomly chosen biomass molecules from the set of 63 E. coli biomass molecules (excluding the four ‘currency’ metabolites mentioned above in the Section ‘Biomass Molecules’, which are found in all of the sets) [57]. These sets served as the basis for our analysis of networks that vary in their biosynthetic ability B. To arrive at identical sample sizes for subsequent analyses, we also included 10 “sets” with B = 63, that is, each of these sets contained all 63 biomass molecules. In total, we thus created a total of 40 (10×4) sets of biomass molecules.
Second, for each of these 40 sets we determined 4 different sets of nutrients, where each set contained a different number of randomly chosen carbon sources from the list of carbon sources we used (see Section Carbon Sources). Specifically, these sets of nutrients contained N = 20, N = 40, N = 60, and N = 80 carbon sources. Thus, up to this second step we had generated a total of 160 (40×4) combinations of sets of biomass molecules and nutrients.
Finally, for each of these 160 combinations, we created 5 minimal metabolic networks. To create each minimal network, we used the following procedure. We started from the global network and sequentially removed individual randomly chosen reactions from it. After each reaction removal we verified that the network was still viable – able to synthesize all B biomass molecules in the set – when each of the N nutrients was provided as the sole carbon source, that is, the network was required to be viable on each carbon source. If that was not the case, we reversed the reaction elimination and removed a different, randomly chosen reaction, until the resulting network was viable. We continued this procedure until no further reactions could be removed from the network without abolishing viability. In this fashion, we generated 800 (160×5) minimal networks. Note that carrying out this procedure repeatedly may not arrive at the same minimal network, because reactions are removed at random. That is, different minimal networks may contain different numbers and different sets of reactions. What unites them is that all their reactions are essential.
Minimal Networks with Isostoichiometric Biomass Composition
We asked whether the stoichiometric composition of biomass, that is, the relative amounts of different biomass molecule in biomass, influences the relationships we explore here. To this end, we generated minimal networks synthesizing given biomass compounds, as described in the previous section, with the difference that these networks synthesized these compounds in equal molarities, that is isostoichiometrically. In other words, for the purpose of this analysis we changed the stoichiometric coefficients c T in the biomass growth function of Equation 1 to a value of one. We used the same combinations of sets nutrients and biomass molecules as described above, except that we generated only 2 minimal networks (instead of 5) for each combination, in order to reduce computational cost. In total, we thus analyzed 320 minimal networks with isostoichiometric biomass composition.
Minimal Networks Synthesizing 20 Amino Acids
To study examples of networks that synthesize biochemically related biomass molecules, we studied minimal networks synthesizing only the 20 proteinaceous amino acids. We generated these networks as described in ‘Generation of Minimal Networks’, except that we did not choose the biomass molecules to be synthesized at random. More specifically, we created 10 sets each of N = 20, N = 40, N = 60, and N = 80 randomly chosen carbon sources, and analyzed 8 minimal networks for each set, for a total of 320 networks.
MCMC Sampling and Random Networks
As we mentioned earlier, changing the genotype of a network does not necessarily cause a change in its phenotype. One can take advantage of this property to generate arbitrarily large and unbiased random samples of metabolic networks with any desired property (see Figure 5), such that viable networks with a given number of reactions are sampled uniformly from the space of such networks. Such samples are central to our analysis. To create them we used a procedure built on Markov Chain Monte Carlo (MCMC) sampling described previously [46], [47]. This procedure fulfills the important detailed balance condition for MCMC sampling. Briefly, the procedure constructs a sequence of metabolic networks, where the next network in the sequence is created from the previous network through a reaction swap. Such a reaction swap consists of the removal of a randomly chosen reaction from the network, followed by the addition of a randomly chosen reaction taken from the global network. We used such reaction swaps, because they preserve the exact number of reactions in a metabolic network, which is important for our analysis. If a reaction swap preserves viability of the network in a given environment, then the swap is accepted, otherwise it is rejected, and a new swap is attempted until a viable genotype is found. We note that subsequent networks in a sequence show autocorrelation in their properties, and are thus not suitable for random sampling. Past work has shown that after 5×103 swaps, the autocorrelation of two genotypes becomes negligible [46]. We therefore started the random walk from E. coli metabolic network and after 2.5×106 reaction swaps, we sampled networks every 5×103 steps in a sequence. Overall, the network samples we created comprise 1000 networks for each condition we study. We did not subject transport reactions to the reaction swap procedure.
To compare our observations from randomly sampled metabolic networks to a reference from biology, it is useful to use a network from a well-studied organism. For this purpose, we used the metabolic network of E. coli, because E. coli is able to survive in multiple different environments [95], [96].
Generation of Starting Networks for MCMC Sampling
To initiate the MCMC procedure that generates random samples of networks with a given set of properties, we needed starting networks that have these properties. Specifically, we needed to create networks with a given number of reactions R, nutrient flexibility N, and biosynthetic ability B. To this end, we started with the same 160 sets of N nutrients and B biomass molecules described in ‘Generation of Minimal Networks’. We created for each set 5 viable networks that differ in their number of reactions, that is, they had 400, 500, 600, 700 and 800 reactions. To create these networks, we used the same procedure as for the production of minimal networks, except that we stopped removing reactions when a network with the desired number of reactions had been reached. At the end of this procedure, we had 800 networks (160×5) with various combinations of the 3 network properties. We used each of these networks as starting networks for MCMC sampling, and generated 20 random viable networks from each of the 800 starting networks, for a total of 16000 random networks. This amounts to 200 networks for each different combination of N, B, and R.
We performed all our analyses using MATLAB (7.10.0, The MathWorks Inc., Natick, MA, R2010a) and R (R Development Core Team, 2008).
Supporting Information
Biomass stoichiometry does not affect the number of reactions in a minimal network. The vertical axis shows the number of reactions in minimal networks as a function of a) biosynthetic ability and b) nutrient flexibility. Dots and lengths of error bars correspond to means and one standard deviation. Each blue dot of size 200 indicates networks with the biomass stoichiometry of E. coli [57]. Each red dot of size 80 indicates networks with isostoichiometric biomass (see Methods). Red and blue means do not differ from each other significantly (P>0.40, Mann-Whitney U-test).
(TIF)
Biosynthetic flux distribution. The flux distributions in units of mmoles per g DW per hour are shown for each combination of biosynthetic ability (B = 20 first row, B = 30 second row, B = 40 third row, B = 63 last row) and nutrient flexibility (N = 20 first column, N = 30 second column, N = 40 third column, N = 63 last column) that we examined. Data are based on 16,000 random viable networks, as described in methods (1000 networks per panel).
(TIF)
Distribution of number R of reactions. The distributions of R are shown for each combination of biosynthetic ability (B = 20 first row, B = 30 second row, B = 40 third row, B = 63 last row) and nutrient flexibility (N = 20 first column, N = 30 second column, N = 40 third column, N = 63 last column) that we examined, and for in total 800 minimal viable networks (50 Networks per panel).
(TIF)
List of carbon sources used in this study.
(DOC)
Examples of reactions required to synthesize additional biomass molecules. The table contains 20 arbitrary biomass molecules (left), and a list of reactions that are required to synthesize the molecule in a random minimal network (in addition to the reactions that the network needed to synthesize other biomass molecules). The analysis is based on minimal networks that are required (i) to synthesize 62 E. coli biomass molecules and (ii) to be viable on glucose. The Table illustrates that the number of additional reactions needed depends on the biomass molecule. (It may also depend on other reactions in a network, but for each biomass molecule results from only one network are shown.)
(DOC)
Examples of reactions required to utilize additional carbon sources. The table contains arbitrary carbon sources (left) and a list of reactions that are required to utilize the carbon source in a random minimal network (in addition to the reactions that the network needs to utilize other carbon sources). The analysis is based on minimal networks that were required (i) to synthesize all E. coli biomass molecules and (ii) to be viable on 30 other carbon sources. The Table illustrates that the number of additional reactions needed depends on the carbon source. (It may also depend on other reactions in a network, but for each carbon source results for only one network are shown.)
(DOC)
Confidence Interval Calculation.
(DOC)
Examples of reactions needed to metabolize new carbon sources.
(DOC)
Examples of reactions needed to synthesize additional biomass molecules.
(DOC)
Acknowledgments
We thank Olivier Martin, João F. Matias Rodrigues, Vardan Andriasyan and Aditya Barve for helpful discussions.
Footnotes
Competing Interests: The authors have declared that no competing interests exist.
Funding: We acknowledge support through Swiss National Science Foundation grant 315230-129708, as well as through the YeastX project of SystemsX.ch, and the University Priority Research Program in Systems Biology at the University of Zurich. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Purnick PEM, Weiss R. The second wave of synthetic biology: from modules to systems. Nature reviews Molecular cell biology 10: 410–422. 2009. Available: http://www.ncbi.nlm.nih.gov/pubmed/19461664. Accessed 2011 Jun 10. [DOI] [PubMed]
- 2.Smolke CD, Silver PA. Informing biological design by integration of systems and synthetic biology. Cell 144: 855–859. 2011. Available: http://www.ncbi.nlm.nih.gov/pubmed/21414477. Accessed 2011 Jun 10. [DOI] [PMC free article] [PubMed]
- 3.Sprinzak D, Elowitz MB. Reconstruction of genetic circuits. Nature 438: 443–448. 2005. Available: http://www.ncbi.nlm.nih.gov/pubmed/16306982. Accessed 2011 Jul 5. [DOI] [PubMed]
- 4.Benner SA, Sismour AM. Synthetic biology. Nature Reviews Genetics 6: 533–543. 2005. Available: http://www.ncbi.nlm.nih.gov/pubmed/15995697. Accessed 2012 Jun 11. [DOI] [PMC free article] [PubMed]
- 5.Yarmush ML, Banta S. Metabolic engineering: advances in modeling and intervention in health and disease. Annual review of biomedical engineering 5: 349–381. 2003. Available: http://www.ncbi.nlm.nih.gov/pubmed/14527316. Accessed 2012 Jun 11. [DOI] [PubMed]
- 6.Smith HO, Hutchison CA, Pfannkoch C, Venter JC. Generating a synthetic genome by whole genome assembly: phiX174 bacteriophage from synthetic oligonucleotides. Proceedings of the National Academy of Sciences of the United States of America 100: 15440–15445. 2003. Available: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=307586&tool=pmcentrez&rendertype=abstract. Accessed 2012 Jun 11. [DOI] [PMC free article] [PubMed]
- 7.Gibson DG, Benders GA, Andrews-Pfannkoch C, Denisova EA, Baden-Tillson H, et al. Complete chemical synthesis, assembly, and cloning of a Mycoplasma genitalium genome. Science (New York, NY) 319: 1215–1220. 2008. Available: http://www.ncbi.nlm.nih.gov/pubmed/18218864. Accessed 2011 Jul 11. [DOI] [PubMed]
- 8.Gibson DG, Glass JI, Lartigue C, Noskov VN, Chuang R-Y, et al. Creation of a bacterial cell controlled by a chemically synthesized genome. Science (New York, NY) 329: 52–56. 2010. Available: http://www.ncbi.nlm.nih.gov/pubmed/20488990. Accessed 2011 Jul 17. [DOI] [PubMed]
- 9.Murtas G. Question 7: construction of a semi-synthetic minimal cell: a model for early living cells. Origins of life and evolution of the biosphere?: the journal of the International Society for the Study of the Origin of Life 37: 419–422. 2007. Available: http://www.ncbi.nlm.nih.gov/pubmed/17668286. Accessed 2011 Aug 10. [DOI] [PubMed]
- 10.Rasmussen S, Chen L, Deamer D, Krakauer DC, Packard NH, et al. Evolution. Transitions from nonliving to living matter. Science (New York, NY) 303: 963–965. 2004. Available: http://www.ncbi.nlm.nih.gov/pubmed/14963315. Accessed 2011 Jul 27. [DOI] [PubMed]
- 11.Glass JI, Assad-Garcia N, Alperovich N, Yooseph S, Lewis MR, et al. Essential genes of a minimal bacterium. Proceedings of the National Academy of Sciences of the United States of America 103: 425–430. 2006. Available: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1324956&tool=pmcentrez&rendertype=abstract. Accessed 2012 Jun 11. [DOI] [PMC free article] [PubMed]
- 12.Nishikawa T, Gulbahce N, Motter AE. Spontaneous reaction silencing in metabolic optimization. PLoS computational biology 4: e1000236. 2008. Available: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2582435&tool=pmcentrez&rendertype=abstract. Accessed 2011 Jul 21. [DOI] [PMC free article] [PubMed]
- 13.Forster AC, Church GM. Towards synthesis of a minimal cell. Molecular systems biology 2: 45. 2006. Available: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1681520&tool=pmcentrez&rendertype=abstract. Accessed 2011 Jun 12. [DOI] [PMC free article] [PubMed]
- 14.Kuwahara H, Yoshida T, Takaki Y, Shimamura S, Nishi S, et al. Reduced genome of the thioautotrophic intracellular symbiont in a deep-sea clam, Calyptogena okutanii. Current biology?: CB 17: 881–886. 2007. Available: http://www.ncbi.nlm.nih.gov/pubmed/17493812. Accessed 2011 Jul 18. [DOI] [PubMed]
- 15.Mira A, Ochman H, Moran NA. Deletional bias and the evolution of bacterial genomes. Trends in Genetics 17: 589–596. 2001. Available: http://www.ncbi.nlm.nih.gov/pubmed/11585665. Accessed 2012 Jun 11. [DOI] [PubMed]
- 16.Gil R, Silva FJ, Zientz E, Delmotte F, González-Candelas F, et al. The genome sequence of Blochmannia floridanus: comparative analysis of reduced genomes. Proceedings of the National Academy of Sciences of the United States of America 100: 9388–9393. 2003. Available: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=170928&tool=pmcentrez&rendertype=abstract. Accessed 2012 Jun 11. [DOI] [PMC free article] [PubMed]
- 17.Nakabachi A, Yamashita A, Toh H, Ishikawa H, Dunbar HE, et al. Bacterial Endosymbiont Carsonella. October. 2008. pp. 3–5. [DOI] [PubMed]
- 18.Mizoguchi H, Sawano Y, Kato J-ichi, Mori H. Superpositioning of deletions promotes growth of Escherichia coli with a reduced genome. DNA research?: an international journal for rapid publication of reports on genes and genomes 15: 277–284. 2008. Available: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2575892&tool=pmcentrez&rendertype=abstract. Accessed 2011 Aug 10. [DOI] [PMC free article] [PubMed]
- 19.Rude MA, Schirmer A. New microbial fuels: a biotech perspective. Current opinion in microbiology 12: 274–281. 2009. Available: http://www.ncbi.nlm.nih.gov/pubmed/19447673. Accessed 2011 Jul 12. [DOI] [PubMed]
- 20.Antoni D, Zverlov VV, Schwarz WH. Biofuels from microbes. Applied microbiology and biotechnology 77: 23–35. 2007. Available: http://www.ncbi.nlm.nih.gov/pubmed/17891391. Accessed 2011 Jul 17. [DOI] [PubMed]
- 21.Lee SK, Chou H, Ham TS, Lee TS, Keasling JD. Metabolic engineering of microorganisms for biofuels production: from bugs to synthetic biology to fuels. Current opinion in biotechnology 19: 556–563. 2008. Available: http://www.ncbi.nlm.nih.gov/pubmed/18996194. Accessed 2011 Jul 5. [DOI] [PubMed]
- 22.Bailey JE. Toward a science of metabolic engineering. Science 252: 1668–1675. 1991. Available: http://www.ncbi.nlm.nih.gov/pubmed/2047876. Accessed 2012 Jun 11. [DOI] [PubMed]
- 23.Steen EJ, Chan R, Prasad N, Myers S, Petzold CJ, et al. Metabolic engineering of Saccharomyces cerevisiae for the production of n-butanol. Microbial cell factories 7: 36. 2008. Available: http://www.ncbi.nlm.nih.gov/pubmed/19055772. Accessed 2012 Jun 11. [DOI] [PMC free article] [PubMed]
- 24.Steen EJ, Kang Y, Bokinsky G, Hu Z, Schirmer A, et al. Microbial production of fatty-acid-derived fuels and chemicals from plant biomass. Nature 463: 559–562. 2010. Available: http://www.ncbi.nlm.nih.gov/pubmed/20111002. Accessed 2012 Jun 11. [DOI] [PubMed]
- 25.Schirmer A, Rude MA, Li X, Popova E, del Cardayre SB. Microbial Biosynthesis of Alkanes. Science 329: 559–562. 2010. Available: http://www.sciencemag.org/cgi/doi/10.1126/science.1187936. Accessed 2010 Jul 30. [DOI] [PubMed]
- 26.Keasling JD. Manufacturing Molecules Through Metabolic Engineering. Science 330: 1355–1358. 2010. Available: http://www.sciencemag.org/cgi/doi/10.1126/science.1193990. Accessed 2010 Dec 2. [DOI] [PubMed]
- 27.Mukhopadhyay A, Redding AM, Rutherford BJ, Keasling JD. Importance of systems biology in engineering microbes for biofuel production. Current opinion in biotechnology 19: 228–234. 2008. Available: http://www.ncbi.nlm.nih.gov/pubmed/18515068. Accessed 2011 Jul 11 July. [DOI] [PubMed]
- 28.Schmidt CW. Synthetic Biology: Environmental Health Implications of a New Field. Environmental Health Perspectives. 2010;118:A118–A123. doi: 10.1289/ehp.118-a118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Cases I, Lorenzo V. Genetically modified organisms for the environment: stories of success and failure and what we have learned from them. International microbiology 8: 213–222. 2005. Available: http://scielo.isciii.es/scielo.php?pid=S1139-67092005000300009&script=sci_abstract&tlng=e. Accessed 2011 Nov 28. [PubMed]
- 30.Stephanopoulos G, Vallino JJ. Network rigidity and metabolic engineering in metabolite overproduction. Science 252: 1675–1681. 1991. Available: http://www.ncbi.nlm.nih.gov/pubmed/1904627. Accessed 2012 Jun 11. [DOI] [PubMed]
- 31.Wisselink HW, Toirkens MJ, Wu Q, Pronk JT, van Maris AJ a. Novel evolutionary engineering approach for accelerated utilization of glucose, xylose, and arabinose mixtures by engineered Saccharomyces cerevisiae strains. Applied and environmental microbiology 75: 907–914. 2009. Available: http://www.ncbi.nlm.nih.gov/pubmed/19074603. Accessed 2012 Jun 11. [DOI] [PMC free article] [PubMed]
- 32.Liang JC, Bloom RJ, Smolke CD. Engineering Biological Systems with Synthetic RNA Molecules. Molecular Cell 43: 915–926. 2011. Available: http://linkinghub.elsevier.com/retrieve/pii/S1097276511006447. Accessed 2011 Sep 19. [DOI] [PMC free article] [PubMed]
- 33.Savage DF, Way J, Silver PA. Defossiling fuel: how synthetic biology can transform biofuel production. ACS Chemical Biology. 2008;3:13–16. doi: 10.1021/cb700259j. [DOI] [PubMed] [Google Scholar]
- 34.Shendure J, Mitra RD, Varma C, Church GM. Advanced sequencing technologies: methods and goals. Nature reviews Genetics 5: 335–344. 2004. Available: http://www.ncbi.nlm.nih.gov/pubmed/15143316. Accessed 2011 Jul 16. [DOI] [PubMed]
- 35.Carr PA, Church GM. Genome engineering. Nature biotechnology 27: 1151–1162. 2009. Available: http://www.ncbi.nlm.nih.gov/pubmed/20010598. Accessed 2011 Jul 27. [DOI] [PubMed]
- 36.Edwards JS, Palsson BO. Robustness analysis of the Escherichia coli metabolic network. Biotechnology Progress. 2000;16:927–939. doi: 10.1021/bp0000712. [DOI] [PubMed] [Google Scholar]
- 37.Edwards JS, Palsson BO. Metabolic flux balance analysis and the in silico analysis of Escherichia coli K-12 gene deletions. BMC bioinformatics. 2000;1:1. doi: 10.1186/1471-2105-1-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Segrè D, Vitkup D, Church GM. Analysis of optimality in natural and perturbed metabolic networks. Proceedings of the National Academy of Sciences of the United States of America. 2002;99:15112–15117. doi: 10.1073/pnas.232349399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Smallbone K, Simeonidis E. Flux balance analysis: a geometric perspective. Journal of theoretical biology. 2009;258:311–315. doi: 10.1016/j.jtbi.2009.01.027. [DOI] [PubMed] [Google Scholar]
- 40.Orth JD, Thiele I, Palsson BØ. What is flux balance analysis? Nature biotechnology. 2010;28:245–248. doi: 10.1038/nbt.1614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Feist AM, Palsson BO. The biomass objective function. Current opinion in microbiology. 2010;13:344–349. doi: 10.1016/j.mib.2010.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Fong SS, Marciniak JY, Palsson BØ. Description and Interpretation of Adaptive Evolution of Escherichia coli K-12 MG1655 by Using a Genome-Scale In Silico Metabolic Model. Journal Of Bacteriology. 2003;185:6400–6408. doi: 10.1128/JB.185.21.6400-6408.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Fong S, Joyce A. Parallel adaptive evolution cultures of Escherichia coli lead to convergent growth phenotypes with different gene expression states. Genome research 15: 1365–1372. 2005. Available: http://genome.cshlp.org/content/15/10/1365.short. Accessed 2011 Nov 28. [DOI] [PMC free article] [PubMed]
- 44.Schellenberger J, Que R, Fleming RMT, Thiele I, Orth JD, et al. Quantitative prediction of cellular metabolism with constraint-based models: the COBRA Toolbox. Nature Protocols. 2007;2:1290–1307. doi: 10.1038/nprot.2011.308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Oberhardt MA, Palsson BØ, Papin JA. Applications of genome-scale metabolic reconstructions. Molecular Systems Biology. 2009;5:320. doi: 10.1038/msb.2009.77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Samal A, Matias Rodrigues JF, Jost J, Martin OC, Wagner A. Genotype networks in metabolic reaction spaces. BMC systems biology. 2010;4:30. doi: 10.1186/1752-0509-4-30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Matias Rodrigues JF, Wagner A. Evolutionary plasticity and innovations in complex metabolic reaction networks. PLoS computational biology. 2009;5:e1000613. doi: 10.1371/journal.pcbi.1000613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Paul G. Higgs TKA. Bioinformatics and molecular evolution. Oxford, UK: John Wiley and Sons, Ltd. 2005.
- 49.Robert CP, Casella G. Monte Carlo Statistical Methods. Berlin: Springer. 2004.
- 50.Neal RM. Probabilistic Inference Using Markov Chain Monte Carlo Methods. Intelligence. 1993;62:144. [Google Scholar]
- 51.Li XR. Generation of random points uniformly distributed in hyperellipsoids. Proc First IEEE Conference on Control Applications. p. 654––658 vol.2. 1992. doi: 10.1109/CCA.1992.269770.
- 52.Diaconis P. The Markov chain Monte Carlo revolution. Bulletin of the American Mathematical Society. 2008;46:179–205. [Google Scholar]
- 53.Brooks S. Markov chain Monte Carlo method and its application. Journal of the Royal Statistical Society Series D The Statistician. 1998;47:69–100. [Google Scholar]
- 54.Ciliberti S, Martin OC, Wagner A. Robustness Can Evolve Gradually in Complex Regulatory Gene Networks with Varying Topology. PLoS Computational Biology. 2007;3:10. doi: 10.1371/journal.pcbi.0030015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Hillenmeyer ME, Fung E, Wildenhain J, Pierce SE, Hoon S, et al. The chemical genomic portrait of yeast: uncovering a phenotype for all genes. Science (New York, NY) 320: 362–365. 2008. Available: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2794835&tool=pmcentrez&rendertype=abstract. Accessed 2011 Jul 18. [DOI] [PMC free article] [PubMed]
- 56.Beller HR, Goh E-B, Keasling JD. Genes involved in long-chain alkene biosynthesis in Micrococcus luteus. Applied and environmental microbiology 76: 1212–1223. 2010. Available: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2820947&tool=pmcentrez&rendertype=abstract. Accessed 2011 Apr 3. [DOI] [PMC free article] [PubMed]
- 57.Feist AM, Henry CS, Reed JL, Krummenacker M, Joyce AR, et al. A genome-scale metabolic reconstruction for Escherichia coli K-12 MG1655 that accounts for 1260 ORFs and thermodynamic information. Molecular systems biology. 2007;3:121. doi: 10.1038/msb4100155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Morot-Gaudry JF, Job D, Lea PJ. Amino acid metabolism. Medical Biochemistry. Springer Verlag, Vol. 2001;28:299–329. [Google Scholar]
- 59.Mann HB, Whitney DR. On a test of whether one of two random variables is stochastically larger than the other. The Annals of Mathematical Statistics. 1947;18:50–60. [Google Scholar]
- 60.Ho NWY, Chen Z, Brainard AP. Genetically engineered Saccharomyces yeast capable of effective cofermentation of glucose and xylose. Applied and Environmental Microbiology. 1998;64:1852–1859. doi: 10.1128/aem.64.5.1852-1859.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Oh Y-K, Palsson BO, Park SM, Schilling CH, Mahadevan R. Genome-scale reconstruction of metabolic network in Bacillus subtilis based on high-throughput phenotyping and gene essentiality data. The Journal of biological chemistry 282: 28791–28799. 2007. Available: http://www.ncbi.nlm.nih.gov/pubmed/17573341. Accessed 2011 Aug 3. [DOI] [PubMed]
- 62.Wang Z, Zhang J. Abundant Indispensable Redundancies in Cellular Metabolic Networks. Genome biology and evolution. 2009;2009:23–33. doi: 10.1093/gbe/evp002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Cooper GM. The Cell. Cell. 2008;77:67–109. [Google Scholar]
- 64.Wessely F, Bartl M, Guthke R, Li P, Schuster S, et al. Optimal regulatory strategies for metabolic pathways in Escherichia coli depending on protein costs. Molecular Systems Biology. 2011;7:515. doi: 10.1038/msb.2011.46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Kim J, Reed JL. OptORF: Optimal metabolic and regulatory perturbations for metabolic engineering of microbial strains. BMC systems biology. 2010;4:53. doi: 10.1186/1752-0509-4-53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Nevoigt E. Progress in Metabolic Engineering of Saccharomyces cerevisiae. Microbiology and molecular biology reviews MMBR. 2008;72:379–412. doi: 10.1128/MMBR.00025-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Madhavan A, Tamalampudi S, Srivastava A, Fukuda H, Bisaria VS, et al. Alcoholic fermentation of xylose and mixed sugars using recombinant Saccharomyces cerevisiae engineered for xylose utilization. Applied Microbiology and Biotechnology. 2009;82:1037–1047. doi: 10.1007/s00253-008-1818-2. [DOI] [PubMed] [Google Scholar]
- 68.Garcia Sanchez R, Karhumaa K, Fonseca C, Sànchez Nogué V, Almeida JR, et al. Improved xylose and arabinose utilization by an industrial recombinant Saccharomyces cerevisiae strain using evolutionary engineering. Biotechnology for biofuels. 2010;3:13. doi: 10.1186/1754-6834-3-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Liu E, Hu Y. Construction of a xylose-fermenting Saccharomyces cerevisiae strain by combined approaches of genetic engineering, chemical mutagenesis and evolutionary adaptation. Biochemical Engineering Journal. 2010;48:204–210. [Google Scholar]
- 70.Weber J, Kayser A, Rinas U. Metabolic flux analysis of Escherichia coli in glucose-limited continuous culture. II. Dynamic response to famine and feast, activation of the methylglyoxal pathway and oscillatory behaviour. Microbiology (Reading, England) 151: 707–716. 2005. Available: http://www.ncbi.nlm.nih.gov/pubmed/15758217. Accessed 2011 Aug 24. [DOI] [PubMed]
- 71.Vandamme EJ. Production of vitamins, coenzymes and related biochemicals by biotechnological processes. Journal of chemical technology and biotechnology Oxford Oxfordshire 1986. 1992;53:313–327. doi: 10.1002/jctb.280530402. Available: http://doi.wiley.com/10.1002/chin.199229310. [DOI] [PubMed] [Google Scholar]
- 72.Expósito O, Bonfill M, Moyano E, Onrubia M, Mirjalili MH, et al. Biotechnological production of taxol and related taxoids: current state and prospects. Anticancer agents in medicinal chemistry. 2009;9:109–121. doi: 10.2174/187152009787047761. [DOI] [PubMed] [Google Scholar]
- 73.Metzger P, Largeau C. Botryococcus braunii: a rich source for hydrocarbons and related ether lipids. Applied Microbiology and Biotechnology. 2005;66:486–496. doi: 10.1007/s00253-004-1779-z. [DOI] [PubMed] [Google Scholar]
- 74.Leuchtenberger W, Huthmacher K, Drauz K. Biotechnological production of amino acids and derivatives: current status and prospects. Applied Microbiology and Biotechnology. 2005;69:1–8. doi: 10.1007/s00253-005-0155-y. [DOI] [PubMed] [Google Scholar]
- 75.Raghevendran V, Patil KR, Olsson L, Nielsen J. Hap4 is not essential for activation of respiration at low specific growth rates in Saccharomyces cerevisiae. The Journal of Biological Chemistry. 2006;281:12308–12314. doi: 10.1074/jbc.M512972200. [DOI] [PubMed] [Google Scholar]
- 76.Dueñas-Sánchez R, Codón AC, Rincón AM, Benítez T. Increased biomass production of industrial bakers’ yeasts by overexpression of Hap4 gene. International Journal of Food Microbiology. 2010;143:150–160. doi: 10.1016/j.ijfoodmicro.2010.08.009. [DOI] [PubMed] [Google Scholar]
- 77.Raab AM, Hlavacek V, Bolotina N, Lang C. Shifting the Fermentative/Oxidative Balance in Saccharomyces cerevisiae by Transcriptional Deregulation of Snf1 via Overexpression of the Upstream Activating Kinase Sak1p. Applied and Environmental Microbiology. 2011;77:1981–1989. doi: 10.1128/AEM.02219-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Price ND, Reed JL, Palsson BØ. Genome-scale models of microbial cells: evaluating the consequences of constraints. Nature reviews Microbiology 2: 886–897. 2004. Available: http://www.ncbi.nlm.nih.gov/pubmed/15494745. Accessed 2010 Jul 16. [DOI] [PubMed]
- 79.Kauffman KJ, Prakash P, Edwards JS. Advances in flux balance analysis. Current Opinion in Biotechnology 14: 491–496. 2003. Available: http://linkinghub.elsevier.com/retrieve/pii/S0958166903001174. Accessed 2010 Jul 16. [DOI] [PubMed]
- 80.Gonzalez OR, Küper C, Jung K, Naval PC, Mendoza E. Parameter estimation using Simulated Annealing for S-system models of biochemical networks. Bioinformatics. 2007;23:480–486. doi: 10.1093/bioinformatics/btl522. [DOI] [PubMed] [Google Scholar]
- 81.Tomshine J, Kaznessis YN. Optimization of a Stochastically Simulated Gene Network Model via Simulated Annealing. Biophysical Journal. 2006;91:3196–3205. doi: 10.1529/biophysj.106.083485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Domingues A, Vinga S, Lemos JM. Optimization strategies for metabolic networks. BMC systems biology. 2010;4:113. doi: 10.1186/1752-0509-4-113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Yang L, Mahadevan R, Cluett W. A bilevel optimization algorithm to identify enzymatic capacity constraints in metabolic networks. Computers & Chemical Engineering. 2008;32:2072–2085. [Google Scholar]
- 84.Rocha I, Maia P, Evangelista P, Vilaça P, Soares S, et al. OptFlux: an open-source software platform for in silico metabolic engineering. BMC systems biology. 2010;4:45. doi: 10.1186/1752-0509-4-45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Julius AA, Imielinski M, Pappas GJ. Metabolic networks analysis using convex optimization. 2008 47th IEEE Conference on Decision and Control. 2008. pp. 762–767.
- 86.Patil KR, Rocha I, Förster J, Nielsen J. Evolutionary programming as a platform for in silico metabolic engineering. BMC Bioinformatics. 2005;6:308. doi: 10.1186/1471-2105-6-308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Ebenhöh O, Heinrich R. Evolutionary optimization of metabolic pathways. Theoretical reconstruction of the stoichiometry of ATP and NADH producing systems. Bulletin of Mathematical Biology. 2001;63:21–55. doi: 10.1006/bulm.2000.0197. [DOI] [PubMed] [Google Scholar]
- 88.Riley M. Genes and proteins of Escherichia coli K-12 (GenProtEC). Nucleic Acids Research. 1997;25:51–52. doi: 10.1093/nar/25.1.51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Goto S, Nishioka T, Kanehisa M. LIGAND: chemical database of enzyme reactions. Nucleic acids research. 2000;28:380–382. doi: 10.1093/nar/28.1.380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Kanehisa M, Goto S. KEGG: kyoto encyclopedia of genes and genomes. Nucleic acids research. 2000;28:27–30. doi: 10.1093/nar/28.1.27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Goto S, Okuno Y, Hattori M, Nishioka T, Kanehisa M. LIGAND: database of chemical compounds and reactions in biological pathways. Nucleic acids research. 2002;30:402–404. doi: 10.1093/nar/30.1.402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Klasson L. Evolution of minimal-gene-sets in host-dependent bacteria. Trends in Microbiology 12: 37–43. 2004. Available: http://linkinghub.elsevier.com/retrieve/pii/S0966842X03003111. Accessed 2011 Jul 19. [DOI] [PubMed]
- 93.Covert MW, Schilling CH, Palsson B. Regulation of gene expression in flux balance models of metabolism. Journal of theoretical biology. 2001;213:73–88. doi: 10.1006/jtbi.2001.2405. [DOI] [PubMed] [Google Scholar]
- 94.Handorf T, Christian N, Ebenhöh O, Kahn D. An environmental perspective on metabolism. Journal of Theoretical Biology. 2008;252:530–537. doi: 10.1016/j.jtbi.2007.10.036. [DOI] [PubMed] [Google Scholar]
- 95.Welch RA, Burland V, Plunkett G, Redford P, Roesch P, et al. Extensive mosaic structure revealed by the complete genome sequence of uropathogenic Escherichia coli. Proceedings of the National Academy of Sciences of the United States of America. 2002;99:17020–17024. doi: 10.1073/pnas.252529799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Touchon M, Hoede C, Tenaillon O, Barbe V, Baeriswyl S, et al. Organised genome dynamics in the Escherichia coli species results in highly diverse adaptive paths. PLoS genetics 5: e1000344. 2009. Available: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2617782&tool=pmcentrez&rendertype=abstract. Accessed 2011 Jul 21. [DOI] [PMC free article] [PubMed]
- 97.Thomas GH, Zucker J, Macdonald SJ, Sorokin A, Goryanin I, et al. A fragile metabolic network adapted for cooperation in the symbiotic bacterium Buchnera aphidicola. BMC systems biology 3: 24. 2009. Available: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2649895&tool=pmcentrez&rendertype=abstract. Accessed 2011 Jun 17. [DOI] [PMC free article] [PubMed]
- 98.Schilling CH, Covert MW, Famili I, Church GM, Edwards JS, et al. Genome-scale metabolic model of Helicobacter pylori 26695. Journal Of Bacteriology. 2002;184:4582–4593. doi: 10.1128/JB.184.16.4582-4593.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Becker SA, Palsson BØ. Genome-scale reconstruction of the metabolic network in Staphylococcus aureus N315: an initial draft to the two-dimensional annotation. BMC microbiology 5: 8. 2005. Available: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1079855&tool=pmcentrez&rendertype=abstract. Accessed 2011 Jul 18. [DOI] [PMC free article] [PubMed]
- 100.Feist AM, Scholten JCM, Palsson BØ, Brockman FJ, Ideker T. Modeling methanogenesis with a genome-scale metabolic reconstruction of Methanosarcina barkeri. Molecular systems biology 2: 2006.0004. 2006. Available: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1681478&tool=pmcentrez&rendertype=abstract. Accessed 2011 Jul 20. [DOI] [PMC free article] [PubMed]
- 101.Beste DJV, Hooper T, Stewart G, Bonde B, Avignone-Rossa C, et al. GSMN-TB: a web-based genome-scale network model of Mycobacterium tuberculosis metabolism. Genome biology 8: R89. 2007. Available: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1929162&tool=pmcentrez&rendertype=abstract. Accessed 2011 Jun 14. [DOI] [PMC free article] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Biomass stoichiometry does not affect the number of reactions in a minimal network. The vertical axis shows the number of reactions in minimal networks as a function of a) biosynthetic ability and b) nutrient flexibility. Dots and lengths of error bars correspond to means and one standard deviation. Each blue dot of size 200 indicates networks with the biomass stoichiometry of E. coli [57]. Each red dot of size 80 indicates networks with isostoichiometric biomass (see Methods). Red and blue means do not differ from each other significantly (P>0.40, Mann-Whitney U-test).
(TIF)
Biosynthetic flux distribution. The flux distributions in units of mmoles per g DW per hour are shown for each combination of biosynthetic ability (B = 20 first row, B = 30 second row, B = 40 third row, B = 63 last row) and nutrient flexibility (N = 20 first column, N = 30 second column, N = 40 third column, N = 63 last column) that we examined. Data are based on 16,000 random viable networks, as described in methods (1000 networks per panel).
(TIF)
Distribution of number R of reactions. The distributions of R are shown for each combination of biosynthetic ability (B = 20 first row, B = 30 second row, B = 40 third row, B = 63 last row) and nutrient flexibility (N = 20 first column, N = 30 second column, N = 40 third column, N = 63 last column) that we examined, and for in total 800 minimal viable networks (50 Networks per panel).
(TIF)
List of carbon sources used in this study.
(DOC)
Examples of reactions required to synthesize additional biomass molecules. The table contains 20 arbitrary biomass molecules (left), and a list of reactions that are required to synthesize the molecule in a random minimal network (in addition to the reactions that the network needed to synthesize other biomass molecules). The analysis is based on minimal networks that are required (i) to synthesize 62 E. coli biomass molecules and (ii) to be viable on glucose. The Table illustrates that the number of additional reactions needed depends on the biomass molecule. (It may also depend on other reactions in a network, but for each biomass molecule results from only one network are shown.)
(DOC)
Examples of reactions required to utilize additional carbon sources. The table contains arbitrary carbon sources (left) and a list of reactions that are required to utilize the carbon source in a random minimal network (in addition to the reactions that the network needs to utilize other carbon sources). The analysis is based on minimal networks that were required (i) to synthesize all E. coli biomass molecules and (ii) to be viable on 30 other carbon sources. The Table illustrates that the number of additional reactions needed depends on the carbon source. (It may also depend on other reactions in a network, but for each carbon source results for only one network are shown.)
(DOC)
Confidence Interval Calculation.
(DOC)
Examples of reactions needed to metabolize new carbon sources.
(DOC)
Examples of reactions needed to synthesize additional biomass molecules.
(DOC)