Abstract
We have constructed vectors from bacteriophage lambda and from plasmid pBR322 having a single EcoRI restriction site which is immediately downstream from the lac UV5 promotor. Each vector allows the fusion of a cloned gene to the lac Z gene in a different phase relative to the translation initiation codon of the lac Z gene. These vectors were constructed through modification of the initial EcoRI restriction site by S1 endonuclease treatment and then addition of octadeoxyribonucleotides (EcoRI linkers), which shifted the restriction site by 2 or 4 nucleotides. Used in combination these vectors should allow translation of a cloned gene in any one of the three coding phases. The bacteriophages vectors are certified as B2 (EK2) safety level vectors by the French "recombinaison génétique in vitro" committee (D.G.R.S.T.).
Full text
PDF




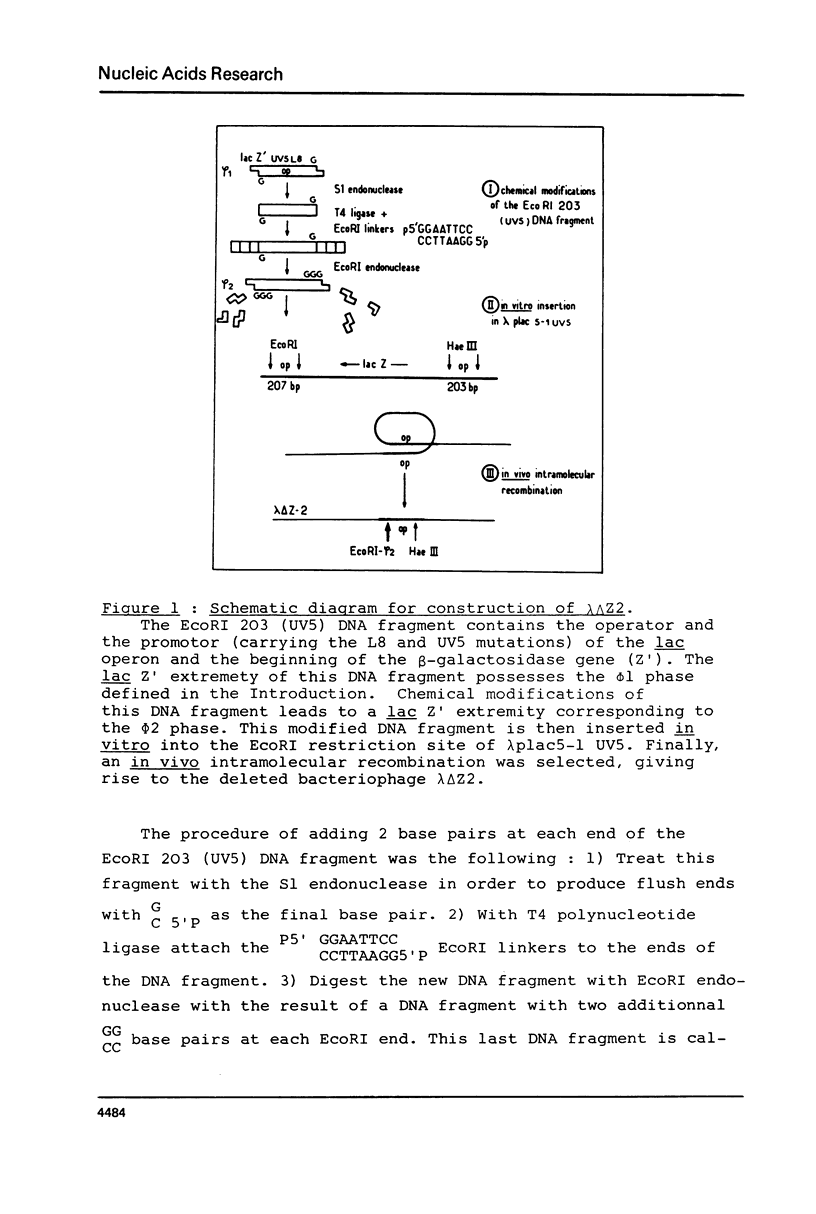





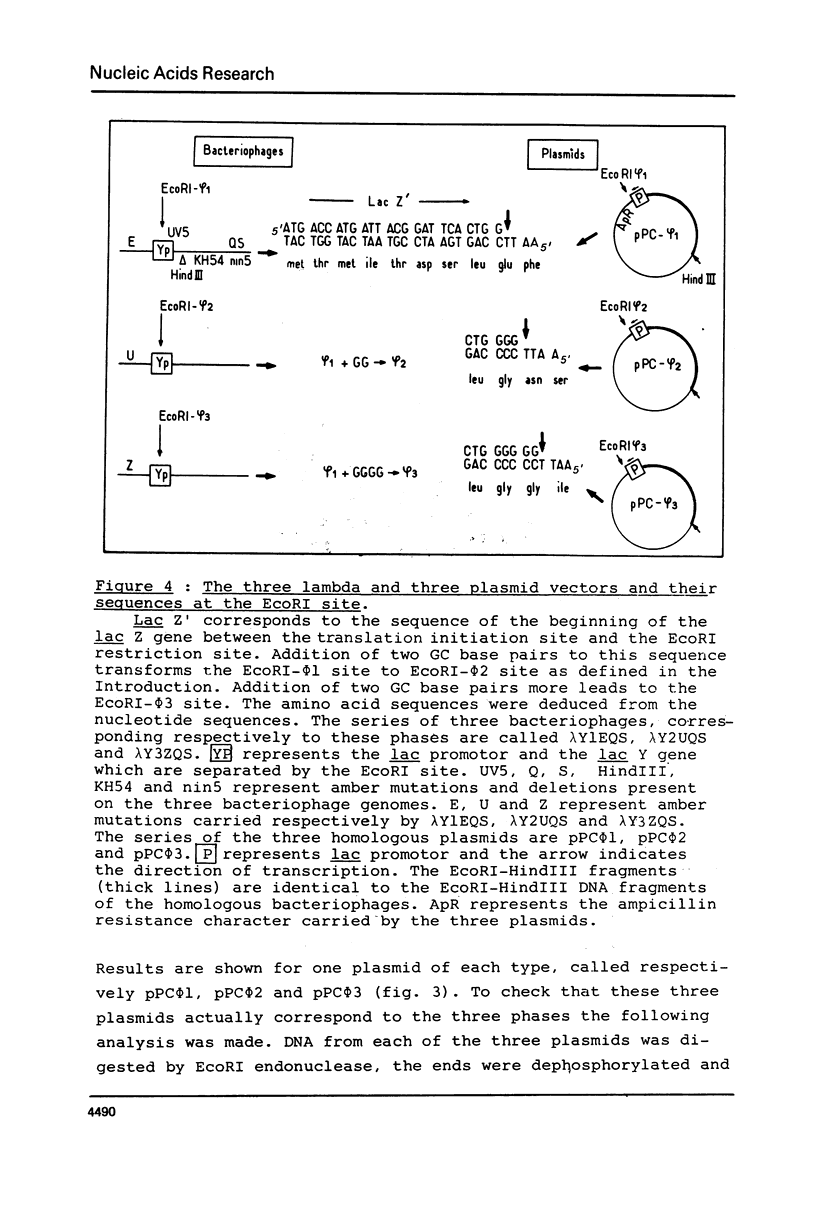




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Adams J. M., Jeppesen P. G., Sanger F., Barrell B. G. Nucleotide sequence from the coat protein cistron of R17 bacteriophage RNA. Nature. 1969 Sep 6;223(5210):1009–1014. doi: 10.1038/2231009a0. [DOI] [PubMed] [Google Scholar]
- Aloni Y., Dhar R., Laub O., Horowitz M., Khoury G. Novel mechanism for RNA maturation: the leader sequences of simian virus 40 mRNA are not transcribed adjacent to the coding sequences. Proc Natl Acad Sci U S A. 1977 Sep;74(9):3686–3690. doi: 10.1073/pnas.74.9.3686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bolivar F., Rodriguez R. L., Greene P. J., Betlach M. C., Heyneker H. L., Boyer H. W., Crosa J. H., Falkow S. Construction and characterization of new cloning vehicles. II. A multipurpose cloning system. Gene. 1977;2(2):95–113. [PubMed] [Google Scholar]
- Chow L. T., Gelinas R. E., Broker T. R., Roberts R. J. An amazing sequence arrangement at the 5' ends of adenovirus 2 messenger RNA. Cell. 1977 Sep;12(1):1–8. doi: 10.1016/0092-8674(77)90180-5. [DOI] [PubMed] [Google Scholar]
- Donoghue D. J., Sharp P. A. An improved bacteriophage lambda vector: construction of model recombinants coding for kanamycin resistance. Gene. 1977 May;1(3-4):209–227. doi: 10.1016/0378-1119(77)90046-4. [DOI] [PubMed] [Google Scholar]
- Itakura K., Hirose T., Crea R., Riggs A. D., Heyneker H. L., Bolivar F., Boyer H. W. Expression in Escherichia coli of a chemically synthesized gene for the hormone somatostatin. Science. 1977 Dec 9;198(4321):1056–1063. doi: 10.1126/science.412251. [DOI] [PubMed] [Google Scholar]
- Jeffreys A. J., Flavell R. A. The rabbit beta-globin gene contains a large large insert in the coding sequence. Cell. 1977 Dec;12(4):1097–1108. doi: 10.1016/0092-8674(77)90172-6. [DOI] [PubMed] [Google Scholar]
- Marx J. L. Viral messenger structure: some surprising new developments. Science. 1977 Aug 26;197(4306):853–923. doi: 10.1126/science.197.4306.853. [DOI] [PubMed] [Google Scholar]
- Maxam A. M., Gilbert W. A new method for sequencing DNA. Proc Natl Acad Sci U S A. 1977 Feb;74(2):560–564. doi: 10.1073/pnas.74.2.560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mercereau-Puijalon O., Royal A., Cami B., Garapin A., Krust A., Gannon F., Kourilsky P. Synthesis of an ovalbumin-like protein by Escherichia coli K12 harbouring a recombinant plasmid. Nature. 1978 Oct 12;275(5680):505–510. doi: 10.1038/275505a0. [DOI] [PubMed] [Google Scholar]
- Moir A., Brammar W. J. The use of specialised transducing phages in the amplification of enzyme production. Mol Gen Genet. 1976 Nov 24;149(1):87–99. doi: 10.1007/BF00275963. [DOI] [PubMed] [Google Scholar]
- Ratzkin B., Carbon J. Functional expression of cloned yeast DNA in Escherichia coli. Proc Natl Acad Sci U S A. 1977 Feb;74(2):487–491. doi: 10.1073/pnas.74.2.487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanger F., Coulson A. R. A rapid method for determining sequences in DNA by primed synthesis with DNA polymerase. J Mol Biol. 1975 May 25;94(3):441–448. doi: 10.1016/0022-2836(75)90213-2. [DOI] [PubMed] [Google Scholar]
- Struhl K., Cameron J. R., Davis R. W. Functional genetic expression of eukaryotic DNA in Escherichia coli. Proc Natl Acad Sci U S A. 1976 May;73(5):1471–1475. doi: 10.1073/pnas.73.5.1471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tiollais P., Perricaudet M., Pettersson U., Philipson L. Propagation in E. coli of bacteriophage lambda with integrated fragments of adenovirus 2 DNA. Gene. 1976;1(1):49–63. doi: 10.1016/0378-1119(76)90006-8. [DOI] [PubMed] [Google Scholar]
- Vapnek D., Hautala J. A., Jacobson J. W., Giles N. H., Kushner S. R. Expression in Escherichia coli K-12 of the structural gene for catabolic dehydroquinase of Neurospora crassa. Proc Natl Acad Sci U S A. 1977 Aug;74(8):3508–3512. doi: 10.1073/pnas.74.8.3508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Villa-Komaroff L., Efstratiadis A., Broome S., Lomedico P., Tizard R., Naber S. P., Chick W. L., Gilbert W. A bacterial clone synthesizing proinsulin. Proc Natl Acad Sci U S A. 1978 Aug;75(8):3727–3731. doi: 10.1073/pnas.75.8.3727. [DOI] [PMC free article] [PubMed] [Google Scholar]




