Abstract
Asthma may be triggered by multiple mediators, including allergen-IgE cross-linking and non-IgE mechanisms. Several clinical studies have shown acute ethanol consumption exacerbates asthma, yet no animal model exists to study this process. We developed a model of ethanol-triggered asthma in allergen-sensitized mice to evaluate the mechanisms of ethanol inducing asthma-like responses. Outbred mice were exposed to cockroach allergens on Days 0 and 14; and on Day 21, mice received ethanol by oral gavage. Tracer studies confirmed alcohol aspiration did not occur. Within 30 minutes, alcohol induced degranulation of over 74% of mast cells, and multiple parameters of asthma-like pulmonary inflammation were triggered. Ethanol-gavaged mice had a fivefold increased production of eotaxin-2 (534 pg/mL) and a sevenfold increase in bronchoalveolar eosinophils (70,080 cells). Ethanol induced a 10-fold increase in IL-13, from 84 pg/mL in sensitized mice to 845 pg/mL in ethanol-gavaged sensitized mice. In cockroach allergen–sensitized mice, ethanol triggered asthma-like changes in respiratory physiology and a significant fivefold increase in airway mucin production. Importantly, none of these asthmatic exacerbations were observed in normal mice gavaged with ethanol. Cromolyn sodium effectively stabilized mast cells, yet increased mucin production and bronchoalveolar eosinophil recruitment. Together, these data show a single oral alcohol exposure will trigger asthma-like pulmonary inflammation in allergen-sensitized mice, providing a novel asthma model.
Asthma is one of the most common chronic diseases, with increasing prevalence, affecting nearly 25 million people in the United States alone and 300 million worldwide.1 One in 10 children and nearly 1 in 12 adults have asthma.2 There are multiple triggers for asthma, including airway irritants, allergens, respiratory infections, aspirin, cold, and exercise.3 Despite recognition of asthma triggers and availability of a myriad of therapeutic options, in many patients, the etiology is undefined, suggesting other triggers and disease mechanisms have yet to be identified.
Recent increases in other health issues have been noted in teens and young adults, the age group also highly affected by asthma, including an increase in binge drinking.4,5 Currently, over half of American adults are regular drinkers, with even higher prevalence rates in high school and college-aged students.5,6 In the young adults that do drink, the majority of the drinking is in the form of binge drinking.7,8 Although the incidences of asthma and binge drinking are both on the rise, their interactions remain relatively unexplored. Multiple clinical studies have clearly shown that ethanol exacerbates asthma.9–12 Although a recent publication showed that chronic alcohol exposure would reduce inflammation in ovalbumin-induced murine asthma,13 our studies focused on whether acute alcohol exposure can trigger atopic asthma in a sensitized animal.
The allergen dose administered was carefully chosen to produce sensitized mice with the capacity to exhibit asthmatic responses following cockroach allergen (CRA) exposure, but that do not display chronic inflammation in the absence of a stimulus. This “sensitized, but asymptomatic” model was designed to mimic the majority of human asthmatics, who are allergen sensitized and primed to respond to provocation, but who do not display outright asthmatic inflammation and respiratory distress in the absence of a stimulus.14 Furthermore, this model of asthma allows the investigation of asthma triggers in a sensitized, but otherwise asymptomatic, host. The data presented here demonstrate a novel mechanism for triggering asthma by showing that a single instance of acute ethanol exposure can trigger multiple asthmatic symptoms in cockroach allergen–sensitized mice.
Materials and Methods
Animals
Female, outbred Hsd:ICR 18- to 20-g mice were used exclusively for these experiments (Harlan Laboratories, Indianapolis, IN). Mice were housed in a temperature-controlled room with 12-hour light/dark cycles and unrestricted access to food and water. All experiments were reviewed and approved by the Institutional Animal Care and Use Committee (Boston University).
Allergen Sensitization
Previous reports demonstrated that intratracheal sensitization with CRA, without the use of adjuvants, was sufficient to induce a robust asthmatic response.15–17 In the current study, the CRA dose was approximately 10-fold lower to develop a model of allergen sensitization. CRA was purchased from Greer Laboratories (Lenoir, NC) as a lyophilized extract and diluted in sterile Hanks' balanced salt solution to yield a combined dose of 100 ng of the allergens Blag1 and Blag2, and 30 ng of lipopolysaccharide per 50-μL dose. Allergen sensitization (Day 0) and challenge (Day 14) were given via intratracheal instillation as described.17
Mild Alcohol Intoxication Model
One week after the last CRA challenge, mice underwent food deprivation for 2 hours before water or ethanol administration by oral gavage. Absolute 200 proof ethanol (Sigma-Aldrich, St. Louis, MO) was diluted to 32%, 10.7%, or 3.2% (v/v) in water for blood ethanol concentration kinetics, and the 32% dose (approximately 3 g/kg) for all other experiments. Gavage was performed under light isoflurane anesthesia by administering 300 μL of water or ethanol to the stomach with an 18 ga × 51 mm gastric feeding tube (Instech Solomon, Plymouth Meeting, PA). Oral gavage was chosen as the physiologically relevant route for ethanol dosing,18,19 and mice were lightly anesthetized during the gavage procedure to reduce unnecessary stress-related immune modulation.20 In separate experiments, additional CRA-sensitized mice were given 200 μL of water or 32% ethanol by intraperitoneal (i.p.) injection to induce similar blood ethanol kinetics. To rule out potential aspiration during the gavage procedure, ethanol was measured in bronchoalveolar lavage (BAL) samples. In a separate study, naive food-deprived mice were gavaged with 300 μL of 32% ethanol containing 1 mg/mL methylene blue tracer dye.21 Immediately following gavage, the stomach and lungs were resected and homogenized in 0.1 mol/L sodium hydroxide, and the presence of dye assayed by optical density at 665 nm. For blood ethanol concentration kinetic measurements, naive mice were gavaged with 300 μL of 32%, 10.7%, or 3.2% ethanol, and 20-μL blood samples were obtained from the submandibular vein at designated time points. Plasma and BAL ethanol concentrations were determined using the alcohol dehydrogenase–based Ethanol-L3K Assay (Genzyme Diagnostics P.E.I., Charlottetown, PE Canada).
Mini Mitter Cross-Dose Movement Study
E-Mitter Transmitters (Mini Mitter Co., Bend, OR) were used as previously described.22 In brief, transmitters were placed subcutaneously 3 days before ethanol or water gavage. Mice were food deprived for 2 hours then gavaged with 300 μL of water or 32% ethanol. Movement was monitored continuously for 10 hours starting 3 hours before gavage. As a cross-over study, the day after the first gavage, mice were gavaged with water if gavaged with ethanol and vice versa, and the movement results combined. Movement counts (centimeters moved) were measured every 15 minutes.
Collection of Blood Cells, Plasma, and BAL Inflammatory Cells
Mice from 0.5 or 2 hours postgavage, or 0.5 hours after i.p. injection, were anesthetized with an intraperitoneal ketamine/xylazine injection at 87 and 13 μg/g body weight, respectively, bled retro-orbitally, and sacrificed by cervical dislocation. Twenty microliters of blood were analyzed with a Hemavet hematology system for blood cell counts (Drew Scientific, Oxford, CT), and the remaining blood sample was spun to collect plasma. BAL was performed as previously described.17 Total cell counts were performed on a Coulter particle counter. Cells were then spun onto a glass slide, stained with Diff-Quik Stain Set (Siemens, Newark, DE), and cell differentials were performed under a light microscope. Plasma and BAL cell methodology was repeated for naive (normal) mice and CRA-sensitized mice before gavage (sensitized).
Lung Histology and Processing
Sterile saline was used to perfuse heart and lungs followed by ethanol fixation and paraffin embedding of a single lobe. To test whether using ethanol as a fixative was having a confounding effect on the histology results, studies were performed that fixed paired lung sections in both 70% ethanol and 10% buffered formalin. The comparisons showed no significant differences in mucin staining and exhibited similar trends when comparing mast cell numbers between groups; thus 70% ethanol was used as fixative for all further experiments. Lung sections taken 0.5 or 2 hours postgavage were sectioned and stained with periodic acid-Schiff (PAS) for mucus analysis and quantified as previously described.16 Additional slides were also stained with toluidine blue for mast cell identification and quantified as previously described.23 The remainder of the unfixed lung was homogenized as described24 for tryptase, chemokine, and cytokine measurements. The lung processing protocol should not disrupt intact mast cell granules since this lung homogenization protocol did not include harsh detergents or sonication steps; therefore, lung homogenate tryptase levels represent tryptase released by the mast cells. Lung histology methodology was repeated for naive (normal) mice and CRA-sensitized mice before gavage (sensitized).
Enzyme-Linked Immunosorbent Assay for Cytokines and Tryptase
Lung homogenate supernatants from lungs collected 30 minutes postgavage were diluted 1:10 for chemokine and cytokine measurements as described.25 Tryptase was measured by coating the BAL or lung homogenate on a 96-well plate overnight at 4°C. After blocking with casein, tryptase was detected with rat anti-mouse tryptase-β1 (R&D Systems, Minneapolis, MN) and developed with mouse anti-rat IgG coupled to horseradish peroxidase. Recombinant mouse tryptase β-1 was used as a standard (R&D Systems). Cytokine measurements were repeated for naive (normal) mice and CRA-sensitized mice before gavage (sensitized).
CRA-Specific IgE Measurements
Measurements were performed as previously described.24 A 96-well plate was coated overnight at 4°C with 50 μL per well of a CRA solution diluted to 5 μg/mL Blag2 allergen concentration in phosphate-buffered saline (PBS). Plates were blocked with Casein Blocker (Thermo Scientific, Waltham, MA) for 2.5 hours and then 50 μL of 1:10 diluted plasma, collected from each mouse 30 minutes postgavage, and was incubated overnight. IgE bound to CRA-coated plate was detected using goat-anti-mouse IgE–horseradish peroxide (Bethyl Laboratories, Montgomery, TX) and assay developed with tetramethylbenzidine. CRA-specific IgE measurements were repeated for naive (normal) mice and CRA-sensitized mice before gavage (sensitized).
Pulmonary Physiology
Respiratory function was evaluated using unrestrained whole-body plethysmography (Buxco Research Systems, Wilmington, NC). Select groups of mice receiving gavage (water or ethanol) were assayed either 30 minutes or 1 hour postgavage on Day 21 of immunization protocol. Other nongavaged mice (normal and sensitized) were assayed at arbitrary times on Day 21. All mice were acclimated 5 minutes in plethysmography chambers, then baseline respiratory physiology was recorded before aerosol exposures. Aerosol challenges of PBS, 25 mg/mL methacholine, and 50 mg/mL methacholine were given after baseline measurements were taken.
Heat-Map Data Presentation
Each parameter measured per mouse was z-score normalized using the average and SD of all available data for that parameter as previously described.16 Z-scores were averaged per group and loaded into MultiExperiment Viewer freeware (MeV; http://www.tm4.org/mev, last accessed October 16, 2011), and unsupervised hierarchical clustering was performed. Detailed descriptions of each parameter are listed in Supplemental Table S1, available at http://ajp.amjpathol.org.
Mast Cell Stabilization Experiments
The mast cell stabilizer, cromolyn sodium (Enzo Life Sciences, Exeter, UK), proven effective at stabilizing mast cells in other studies,26,27was used in ethanol-gavaged CRA-sensitized mice. Cromolyn sodium was given intratracheally once a day for 3 days before ethanol gavage (Days 18, 19, and 20) and once on Day 21, 1 hour before gavage. Each instillation was given as 30 mg/kg cromolyn in two 25-μL aliquots in saline (vehicle). Mice were sacrificed 30 minutes after ethanol gavage. Asthmatic responses for all mast cell stabilizer–treated mice were measured as described above.
Statistics
Statistical analysis was performed using Graphpad Prism 5.02 (GraphPad Software, La Jolla, CA). All data are presented as mean ± SEM. Significance was achieved if P < 0.05 using unpaired two-tailed Student's t-tests, one-way or two-way analysis of variance with a Bonferroni posttest with a confidence interval of 95%, or by χ2 analysis for comparisons with values below detection limits. Respiratory physiology was analyzed by two-way analysis of variance separately for normal (normal, normal + water, normal + ethanol) and for sensitized (sensitized, sensitized + water, sensitized + ethanol) mice.
Results
Development of Mild Alcohol Intoxication Model
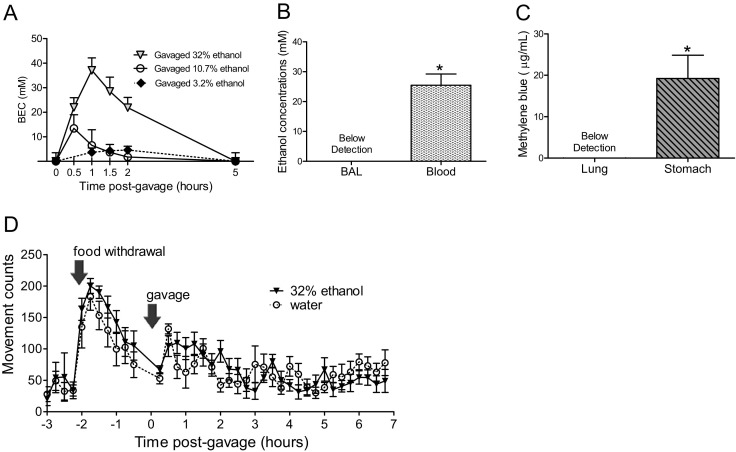
To assess the effects of alcohol consumption in our asthmatic mice, we first needed to design an acute ethanol exposure model to reproduce acute alcohol intoxication rather than chronic alcoholism. The model was developed because previous reports indicate that in humans, acute ethanol intoxication will induce clinical symptoms of asthma in sensitized individuals.11,28,29 Blood alcohol pharmacokinetics were determined by gavaging normal mice with various concentrations of ethanol and withdrawing serial blood samples from the same mouse. There was a clear dose-response effect on blood ethanol concentration (BEC) (Figure 1A), and the high-dose ethanol (32%; approximately 3g/kg) induced a peak BEC of 37 mmol/L (0.17%) at 1 hour postgavage, a level easily achieved by binge-drinking humans.30 To most closely correlate ethanol-induced behavioral changes and BEC to human intoxication, the 32% ethanol dose was selected for all subsequent studies. Additional experiments using i.p. injection of alcohol achieved similar blood ethanol kinetics with 200 μL administration (data not shown). To address the possibility of pulmonary aspiration of gavaged liquid, two separate methods were used. Ethanol was measured in the BAL obtained 30 minutes after gavage and found to be below detection limits (Figure 1B). Additional studies done by gavaging with a methylene blue tracer demonstrated that none of the tracer was detected in the lungs (Figure 1C). Both of these experiments demonstrated that no pulmonary aspiration occurred during the ethanol gavage procedure. Although these additional experiments were performed to confirm that substantial alcohol aspiration did not occur during gavage procedures, the presence of alcohol in the lung cannot be completely ruled out. Bronchial circulation and the volatile nature of alcohol allow for the vaporization of alcohol directly into the conducting airways to allow lung exposure to ethanol,31 hence the basis behind the breathalyzer test.32
Figure 1.
Binge-drinking model. A: Blood ethanol concentrations (BEC). BEC were measured from mice gavaged with 300 μL of the indicated concentrations of ethanol and serial blood samples collected. BEC peaked between 30 and 90 minutes and was undetectable by 5 hours. Values are mean ± SEM; n = 2 to 6 per group. B: Pulmonary aspiration ruled out by lung ethanol measurements. Ethanol concentrations were measured in BAL samples 30 minutes postgavage; all were below detection. Values are mean ± SEM; n = 7 to 8 per group. C: Pulmonary aspiration was additionally ruled out via gavaging mice with 300 μL of methylene blue in 32% ethanol solution and quantifying the dye in homogenized lung and stomach samples. Dye was undetectable in lung whereas it was measurable in stomach. Values are mean ± SEM; n = 4 per group. D: Locomotor activity monitored in gavaged mice. Subcutaneous Mini-Mitters were implanted, and 3 days later, the mice were gavaged with water or 32% ethanol to measure gross motor activity. Food withdrawal or gavage induced an increase in movement, but there was no difference between the water and ethanol groups. Values are mean ± SEM for total movement counts accumulated over 15 minutes, per mouse; n = 10 per group. *P < 0.05 BAL versus blood (B) or lung versus stomach (C), χ2 analysis.
Ethanol-gavaged mice showed mild ataxia similar to other murine binge drinking models.33 To rule out severe ataxia and a gross loss/gain of locomotor activity, we measured movement using a subcutaneously implanted radiotransmitter (Figure 1D). There were no differences in locomotor movements between the water- and ethanol-gavaged groups at any time point. Intestinal histology showed no ethanol-induced damage or induction of mucin (data not shown). These results indicate that our model is representative of mild acute alcohol intoxication.
Ethanol Triggers Mast Cell Degranulation in Allergen-Sensitized Mice
A murine asthma model previously described in our lab15 used 10-fold higher allergen concentrations and investigated asthmatic responses induced after a third allergen exposure on Day 21. The current study used a lower allergen concentration and only two pulmonary exposures to allergens (Days 0 and 14). The purpose of lowering the dose and number of exposures was to elicit sensitization without sustained asthma-like pulmonary inflammation, to allow the evaluation of whether ethanol could trigger asthma-like pulmonary inflammation in a symptomless, but sensitized, host. Even though we used a lower dose of allergen, there was still a clear asthmatic phenotype in our allergen-sensitized mice compared to normal mice, including increases in cockroach allergen–specific IgE, a 5-fold increase in circulating eosinophils, a 20-fold increase in BAL eosinophils, increased mucin production, and respiratory exacerbations as represented by increases in enhanced pause (PenH) in response to methacholine (Table 1). Allergen sensitization increased the numbers of lung mast cells approximately 10-fold compared to normal mice, consistent with previous publications showing increased lung mast cells in allergen-sensitized34 and asthmatic35 individuals.
Table 1.
Comparison of Asthmatic Parameters in Normal (Naive) and CRA-Sensitized Mice
| Parameter | Normal | Sensitized | P value |
|---|---|---|---|
| CRA-specific IgE (OD) | 0.223 ± 0.011 | 0.288 ± 0.015 | 0.007 |
| Eosinophils (per mL blood) | 5 ± 2 | 24 ± 10 | 0.126 |
| BAL eosinophils (per mouse) | 386 ± 96 | 7746 ± 2292 | 0.023 |
| Mast cells (per 10 lung slices) | 2.5 ± 2.5 | 22.5 ± 6.2 | 0.053 |
| Mucus (mean gray area × 1000) | 28 ± 9 | 125 ± 30 | 0.020 |
| PenH (25 mg/mL methacholine) | 0.436 ± 0.017 | 0.620 ± 0.051 | 0.028 |
Sensitized mice were immunized with cockroach allergens on Days 0 and 14 and then studied on Day 21, ie, 7 days after the last exposure to allergen. The final allergen exposure typically given on Day 21 was not administered.
OD, optical density.
Traditionally, allergic-type inflammatory events are potentiated by repeat antigen exposure, subsequent IgE cross-linking, and mast cell degranulation.36 Because antigens were not administered during the time of ethanol gavage in our model, we investigated whether mast cell degranulation was indeed involved, potentially through another mechanism.
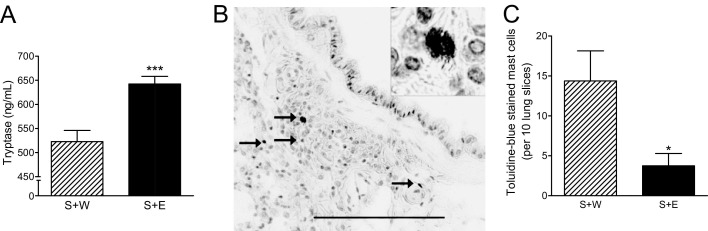
Acetaldehyde, the first metabolite of ethanol catabolism, is elevated immediately on ethanol ingestion in mice37 and will induce mast cell degranulation in vivo.38 To test whether ethanol exposure caused mast cell degranulation in our allergen-sensitized mice, we used two independent methods. First, we measured tryptase, one of the abundant proteins in mast cell granules, in lung homogenates 30 minutes postgavage (Figure 2A). Within 30 minutes, ethanol gavage [sensitized + ethanol gavage (S+E)] significantly increased tryptase release into the lung tissue in CRA-sensitized mice compared to water-gavaged controls [sensitized + water gavage (S+W)]. To confirm mast cell degranulation, lung sections from the same mice used for tryptase measurements were stained with toluidine blue (Figure 2B) and the number of intact mast cells counted in a blinded manner. We saw a statistically significant 74% reduction in toluidine blue–stained mast cells in the ethanol-gavaged group (Figure 2C) when compared to water-gavaged controls. Similar results were seen in mice administered ethanol by i.p. injection, which showed a statistically significant 91% reduction in toluidine blue–stained mast cells in ethanol-injected mice compared to sensitized mice, from 18.1 mast cells/10 lungs in sensitized mice to 1.3 mast cells/10 lungs, respectively (P < 0.05, data not shown). The immobile nature of tissue mast cells and the relatively short 30-minute time point signify a rapid degranulation occurred that resulted in the loss of granule staining. Cockroach allergen–specific IgE was measured and compared between the two groups, and was not significantly affected by ethanol gavage (data not shown), suggesting the cause of increased mast cell degranulation was not due to changes in allergen-specific IgE. Together, these data indicate acute ethanol exposure caused substantial mast cell degranulation in lungs of allergen-sensitized mice.
Figure 2.
Oral ethanol causes mast cell degranulation in CRA-sensitized mice. A: Mast cell degranulation measured by increased tryptase secretion. Tryptase present in lung homogenates was measured in CRA-sensitized mice gavaged with either water (S+W) or 32% ethanol (S+E) and sacrificed 30 minutes postgavage. Ethanol-gavaged mice had significantly higher tryptase release when compared to water-gavaged sensitized mice. Data are mean ± SEM; n = 8 per group. B: Photomicrograph of pulmonary mast cells visualized by toluidine blue staining of lung sections in sensitized mice. Arrows indicate mast cells; inset is a higher powered magnification (original magnification, ×100). C: Number of toluidine blue–stained mast cells. Granule-containing mast cells were counted per 4-μm lung slice. Sensitized mice gavaged with ethanol (S+E) had significantly fewer toluidine blue–stained mast cells than water-gavaged sensitized mice (S+W), indicating that rapid loss of granule contents occurs within 30 minutes of ethanol gavage. All data are mean ± SEM, representative of eight mice per group.*P < 0.05, ***P < 0.001 versus the indicated groups, two-tailed Student's t-test.
Ethanol Triggers Asthma-Like Pulmonary Inflammation in CRA-Sensitized Mice
As shown in Figure 2, ethanol induced mast cell degranulation in lungs of allergen-sensitized mice. Therefore, we hypothesized that other inflammatory events would be initiated in the lungs of these mice to result in asthma-like pulmonary inflammation. An array of asthmatic parameters was investigated, including pulmonary physiology, bronchoalveolar lavage inflammatory cell infiltrate, airway mucin, and cytokine production in the lung. Within 30 minutes of ethanol exposure, exacerbations in 45 of 76 asthmatic parameters were observed, including mucin production, eosinophil infiltration, increased production of eosinophil chemokines, and aggravations in respiratory physiology 1 hour after ethanol. By z-score normalization, we were able to transform these data into a heat map (Figure 3), allowing simultaneous comparison of all parameters and helping to visualize trends between the groups. A detailed listing and descriptions of all 76 parameters can be found in Supplemental Table S1, available at http://ajp.amjpathol.org. Six groups of mice were compared: normal (N), normal gavaged with water (N+W), normal gavaged with ethanol (N+E), CRA-sensitized, but not gavaged (S), sensitized gavaged with water (S+W), and sensitized gavaged with ethanol (S+E). Figure 3 clearly shows an ethanol-induced effect in the sensitized mice, with little to no effect in the water-gavaged controls. Importantly, ethanol did not induce exacerbations in normal mice, demonstrating that an atopic phenotype is required for ethanol-induced modulation of asthmatic parameters. The heat-map data were used to guide further detailed analysis.
Figure 3.

Heat-map representation of asthmatic parameters in normal or allergen-sensitized mice with or without water or ethanol gavage. Individual data points collected from pulmonary physiology, BAL inflammatory cell infiltrate, airway mucin production, and mast cell degranulation (tryptase) were z-score normalized and analyzed with MultiExperiment Viewer freeware. Normal mice (N) gavaged with water (N+W) or ethanol (N+E) displayed similar data patterns to nongavaged normal mice (N). Sensitized mice (S) gavaged with water (S+W) showed slight induction of asthmatic parameters. However, sensitized mice gavaged with ethanol (S+E) showed striking exacerbations in numerous inflammatory parameters as depicted in the far right column, with white bars showing greatest negative deviation from the mean, and black bars depicting greatest positive deviation. Hatched bars indicate data not collected for that parameter for that group. Complete listing and description of heat-map parameters are provided in Supplemental Table S1 (available at http://ajp.amjpathol.org).
Ethanol Consumption Increases BAL Eosinophil Recruitment in Asthmatic Mice
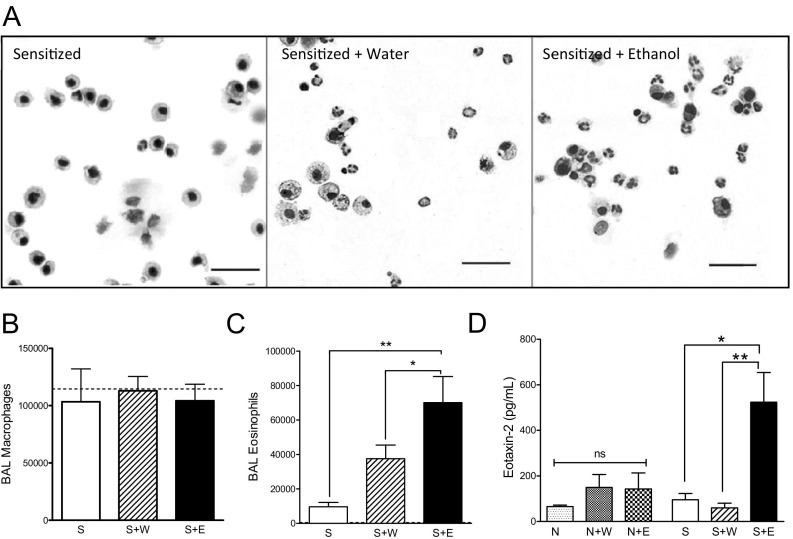
Pulmonary eosinophil recruitment represents a hallmark of asthma and is a marker of disease severity.36,39 We investigated whether ethanol consumption would trigger an influx of eosinophils by quantifying the number of cells in the BAL fluid at 30 minutes postgavage. BAL cells collected from sensitized mice were almost exclusively alveolar macrophages, with scant eosinophils (Figure 4A, left panel). The water-gavaged, sensitized mice also showed primarily macrophages, few lymphocytes, and increased eosinophils (Figure 4A, center panel). Acute ethanol exposure showed a substantial increase in eosinophils recovered from the BAL (Figure 4A, right panel). Neither water nor ethanol gavage increased numbers of BAL macrophages in allergen-sensitized mice (Figure 4B). However, at this time point, either water or ethanol gavage caused greater BAL eosinophil recruitment when compared to nongavaged sensitized mice (Figure 4C). Although the water gavage did increase eosinophil recruitment, this change was not significant and most likely due to stress from the gavage procedure. By contrast, ethanol-gavaged mice showed a significant sevenfold eosinophil increase when compared to sensitized mice and significant twofold increase over BAL eosinophil numbers from water-gavaged CRA-sensitized mice. Additional studies administering water or alcohol by i.p. injection showed similar trends. Injection of water did not induce significant eosinophil recruitment, whereas i.p. ethanol resulted in significant increases over sensitized mice (3.95 × 105 eosinophils per mouse; P < 0.05 vs sensitized).
Figure 4.
Eosinophil recruitment induced by ethanol. A: Representative images of BAL cytospins of an allergen-sensitized mouse and of sensitized mice gavaged with water or ethanol and sacrificed 30 minutes postgavage. Scale bar = 50 μm. B: BAL macrophages. Ethanol gavage in sensitized (S+E) mice did not induce increases in all inflammatory cells, as BAL macrophages were unaffected 30 minutes postgavage. Dotted line depicts average BAL macrophages in normal, naive mice. Data are mean ± SEM for 7 to 15 mice. C: BAL eosinophils. Water gavage in sensitized mice (S+W) increased BAL eosinophil recruitment when compared to sensitized levels immediately postgavage (30 minutes), although the increase was not significant. However, ethanol gavage in sensitized mice (S+E) shows a significant sevenfold increase in BAL eosinophils at this time point when compared to nongavaged sensitized mice, and a significant doubling of BAL eosinophils when compared to water-gavaged control mice. Data are mean ± SEM for seven to eight mice. D: Eosinophil chemokine production. Eotaxin-2 was increased by fivefold in the lungs of ethanol-gavaged sensitized mice 30 minutes postgavage as compared to both sensitized (S) and water-gavaged sensitized mice (S+W). No significant increases in eotaxin-2 were seen for any other group. Data are mean ± SEM for four to eight mice. ns, not significant. ***P < 0.001, **P < 0.01, and *P < 0.05 using one-way analysis of variance with Bonferroni posttests.
To determine potential mechanisms responsible for greater eosinophil recruitment in ethanol-gavaged mice, we measured eosinophil chemotactic factors in lung homogenates obtained from the 30-minute postgavage groups. No differences between water and ethanol groups were detected for eotaxin-1 and regulated on activation normal T cell expressed and secreted (RANTES) (data not shown). However, pulmonary eotaxin-2 levels in ethanol-gavaged sensitized mice were significantly increased approximately fivefold over sensitized mice or sensitized mice with water gavage (Figure 4D). No increases in eotaxin-2 were observed in normal mice gavaged with ethanol. These data suggest acute oral ethanol triggers increases in lung eotaxin-2 and results in significant eosinophil infiltration into the bronchoalveolar spaces.
Increased Th-2 Cytokine Production in Lungs of Ethanol-Gavaged Mice
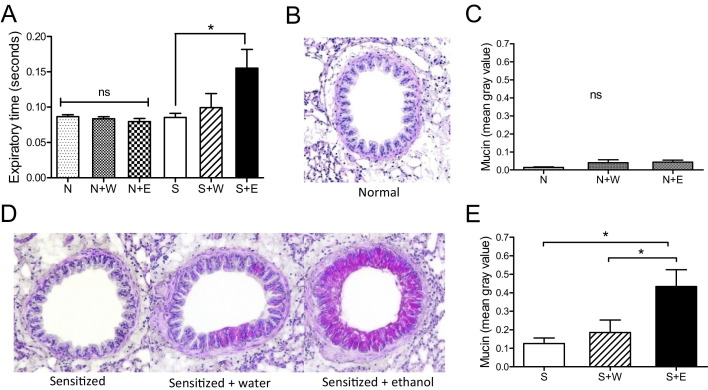
Ethanol increased eotaxin-2 in lungs of sensitized mice, raising the question of whether ethanol augmented other inflammatory mediators. To test this hypothesis, we measured the Th2-type cytokines IL-4, IL-5, and IL-13 at 30 minutes postgavage in lungs of normal mice gavaged with water or ethanol, and CRA-sensitized mice gavaged with water or ethanol. These cytokines were selected since they are associated with allergic diseases36 and are increased on allergen exposure in a CRA asthma model.15,17 Ethanol failed to elicit increases in any of the cytokines in normal mice (data not shown). By contrast, ethanol gavage increased all three cytokines in asthmatic mice (Figure 5). IL-5 was below detection (<6 pg/mL) in water-gavaged mice but increased to 175 pg/mL in lungs of ethanol-gavaged mice. Ethanol induced a modest increase in IL-4. Importantly, a significant 10-fold increase in IL-13 was observed in lungs of ethanol-gavaged sensitized mice over water-gavaged controls.
Figure 5.
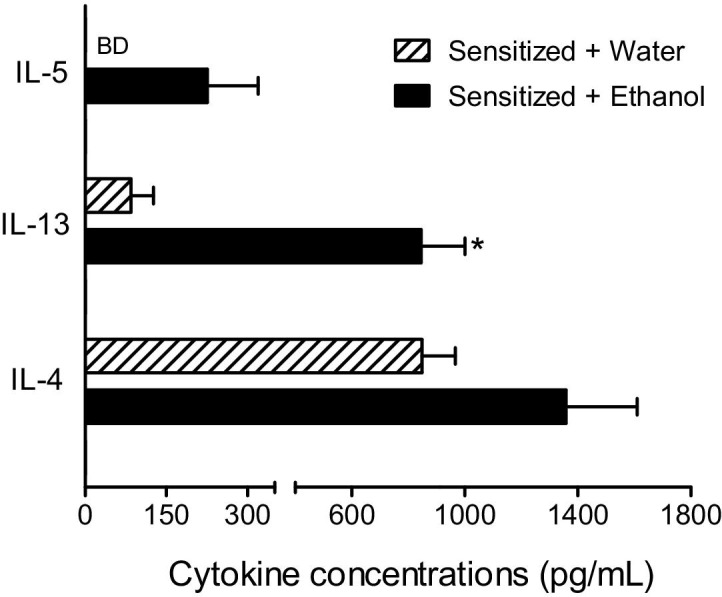
Th-2 type cytokine production. Ethanol gavage resulted in increased levels of Th-2 type cytokines IL-4, IL-5, and IL-13 in lungs of sensitized mice 30 minutes postgavage, with a significant 10-fold increase in IL-13. Data are mean ± SEM, n = 4 per group. BD, below detection. *P < 0.01 using two-tailed Student's t-test.
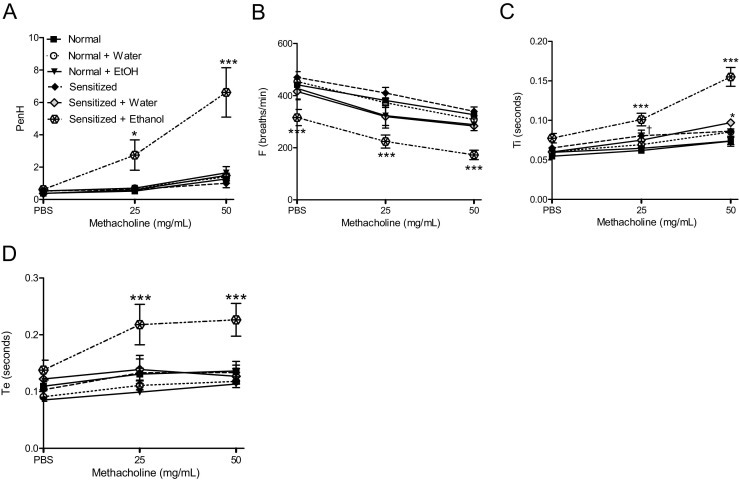
Ethanol Exacerbates Respiratory Function in Allergen-Sensitized Mice
As shown in Figure 3, 14 of the 18 measured respiratory parameters altered in ethanol-gavaged sensitized mice were not changed in ethanol-gavaged normal mice. Among these are several parameters previously shown to be exacerbated in CRA-triggered murine asthma, including increases in PenH, inspiratory time, and expiratory time and decreases in breathing frequency, all associated with worsening respiratory function.16 PenH was measured 1 hour postgavage in six groups of mice: normal; normal gavaged with water; normal gavaged with ethanol; CRA-sensitized, but not gavaged; sensitized gavaged with water; and sensitized gavaged with ethanol (Figure 6A). Sensitized mice gavaged with ethanol showed striking increases in PenH when exposed to increasing methacholine challenges, whereas all other groups challenged with methacholine displayed PenH values similar to or only slightly elevated from baseline measurements. In addition to the increases in PenH, the ethanol-gavaged sensitized group showed substantial depressions in breathing frequency (Figure 6B), as well as lengthened times of inspiration (Figure 6C) and expiration (Figure 6D), all suggesting acute oral ethanol was contributing to pulmonary distress in CRA-sensitized mice. Normal mice gavaged with ethanol did not show these respiratory exacerbations, again suggesting ethanol-triggered asthmatic responses require a sensitized host.
Figure 6.
Pulmonary physiology exacerbated in ethanol-gavaged mice. A: Enhanced pause (PenH). A significant increase in PenH was observed in sensitized mice gavaged with ethanol. No changes in PenH were noted for any other group. B: Breathing frequency (F). Breathing frequency was significantly depressed in ethanol-gavaged sensitized mice at all doses challenged. C: Time of inspiration (Ti). Inspiratory time was significantly increased in sensitized mice gavaged with ethanol. Normal mice gavaged with ethanol and sensitized mice gavaged with water also show slight Ti increases in response to methacholine. D: Time of expiration (Te). Expiratory time was significantly lengthened in ethanol-gavaged sensitized mice. Data are mean ± SEM, n = 3 to 7 per group. *P < 0.05, ***P < 0.001 versus sensitized mice at that dose; †P < 0.05 normal mice versus normal + ethanol, two-way analysis of variance with Bonferroni posttests.
The 1-hour time point was an ideal time to measure early respiratory physiology responses, as it lessened the possibility of gavage-related anesthetic having confounding effects on the airway physiology. However, additional experiments were performed at an earlier (30-minute) time point that confirmed the 1-hour whole-body plethysmography data. Specifically, ethanol-gavaged mice had significant increases in PenH over sensitized mice (PenH = 2.5 vs PenH = 1.0; P < 0.01), whereas water-gavaged mice did not (PenH = 1.5 vs PenH = 1.0; not significant). The other respiratory parameters followed similar trends at the 30-minute time point (data not shown).
Ethanol Gavage Increases Mucin Production and Respiratory Obstruction in Sensitized Mice
Close examination of the PBS data in Figure 6 shows that even without the methacholine challenge, there were differences in respiratory parameters in the sensitized mice gavaged with ethanol. This was examined more closely by evaluating the baseline expiratory time (Te) before nebulization of even PBS. Of the six groups of mice, Te was only increased in sensitized mice gavaged with ethanol (Figure 7A). One hallmark of asthma is bronchial mucus production, which may account for airway luminal occlusion40 and lead to intrapleural airway obstruction and increased expiratory time.41,42 Mucin production was investigated in PAS-stained lungs of normal mice and normal mice gavaged with water or ethanol and sacrificed 30 minutes postgavage. None of the lungs examined (normal, water-gavaged normal, or ethanol-gavaged normal) displayed mucin production. Representative histology is given (Figure 7B), and morphometric analysis confirmed these findings (Figure 7C). It is important to note that because fresh lung tissue was needed for lung homogenate cytokine and tryptase measurements, insufflation with the fixative could not be performed. As a result, the airway epithelium has a “serrated” appearance.
Figure 7.
Ethanol induces airway obstruction and mucin accumulation in allergen-sensitized mice. A: Baseline expiratory times. Time of expiration, before exposure to PBS or methacholine, was found to be significantly elevated in sensitized mice receiving oral alcohol 30 minutes postgavage. Normal mice gavaged with alcohol did not exhibit increased baseline Te. Data are mean ± SEM, n = 3 to 7 per group. B: Representative PAS-stained histology for normal mouse lungs with or without water or ethanol gavage, 30 minutes postgavage. No mucin was detectable in any normal mouse lung with either gavage. C: Morphometric analysis of histology at ×2 magnification from B showing mucin production is not induced in normal mice gavaged with water or ethanol. Data are mean ± SEM, n = 4 per group. D: Representative PAS-stained histology of sensitized mouse lungs 30 minutes after water or ethanol gavage. Bright pink depicts massive mucin production in ethanol-gavaged sensitized mice. E: Morphometric analysis of histology at ×2 magnification from D showing large, significant increases in mucin production induced in ethanol-gavaged sensitized mice over both sensitized and water-gavaged sensitized controls. Data are mean ± SEM for eight mice, representative of two separate experiments. All photomicrographs are the same magnification. N, normal; N+E, normal gavaged with ethanol; N+W, normal gavaged with water; ns, not significant; S, CRA-sensitized, but not gavaged; S+E, sensitized gavaged with ethanol; S+W, sensitized gavaged with water. *P < 0.05, **P < 0.01 versus the respective group, one-way analysis of variance with Bonferroni posttests.
Next, PAS-stained lungs were examined from sensitized mice and sensitized mice gavaged with water or ethanol and sacrificed 30 minutes postgavage. Representative histology (Figure 7D) and morphometric analysis (Figure 7E) are shown. The majority of CRA-sensitized mice did not show bronchial mucin production (Figure 7D, left panel). Water-gavaged sensitized mice showed a small amount of mucin production (Figure 7D, center panel); however, this was not significantly elevated over that of sensitized mice. Ethanol gavage caused a massive induction of bronchial mucin as demonstrated by the bright magenta staining (Figure 7D, right panel). Computer-aided morphometry demonstrated that this increased staining was significantly elevated over both sensitized and water-gavaged sensitized mucin production (Figure 7E). The ethanol-induced mucin production follows the same trends as the expiratory time exacerbations, suggesting that in a sensitized host, ethanol will induce mucin overproduction, leading to airway obstruction, which can be measured through analysis of whole-body plethysmography. Importantly, mucin induction was not observed in normal mice gavaged with ethanol, suggesting lungs needed to be allergen sensitized or “primed” in order for ethanol-induced mucin production to occur.
Mast Cell Stabilizers Exacerbate Pulmonary Eosinophilia and Mucin Overproduction
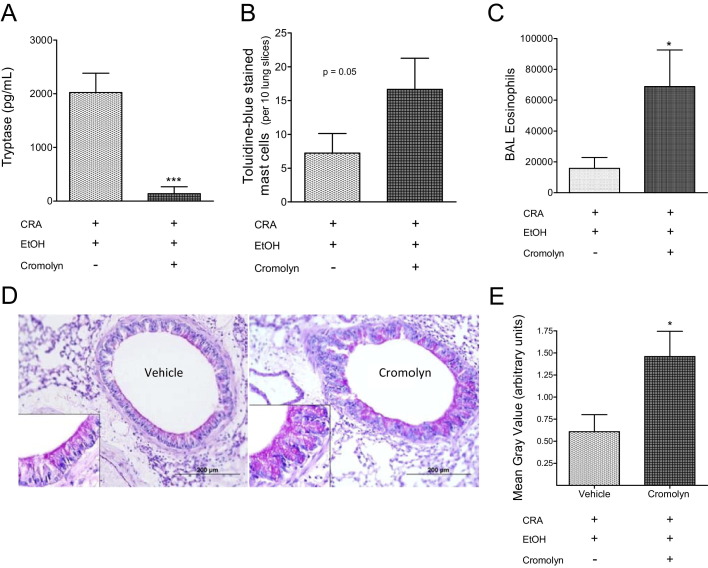
Figure 2 demonstrated rapid mast cell degranulation in CRA-sensitized mice on ethanol administration. The soluble mediators released by mast cells43,44may provide the mechanism(s) for the rapid increase in asthmatic responses observed in ethanol-gavaged mice. To address this possibility, mice were treated with the mast cell stabilizer cromolyn sodium or saline vehicle as described. Cromolyn sodium–treated mice exhibited mast cell stabilization, with significant reduction of tryptase secretion into the BAL (Figure 8A) and increased detection of intact, toluidine blue–staining mast cells in the lung tissue (Figure 8B). Despite the mast cell stabilization, cromolyn-treated mice exhibited no differences in lung or BAL levels of eotaxin-2, IL-4, IL-5, or IL-13 (data not shown). Interestingly, lungs of cromolyn-treated mice displayed higher numbers of BAL eosinophils (Figure 8C) and increased amounts of bronchial mucin staining (Figure 8, D and E).
Figure 8.
Mast cell stabilizer cromolyn sodium exacerbates BAL eosinophil recruitment and mucin overproduction. A: Tryptase secretion. Mast cell stabilization 30 minutes postgavage was monitored by measuring a significant decrease in tryptase secretion into the BAL in cromolyn-treated mice. B: Mast cell staining. Mast cell stabilization was confirmed by counting toluidine blue–stained mast cells in ethanol-fixed lungs. A significant increase in granule-intact toluidine blue–stained mast cells was observed in cromolyn-treated mice. C: BAL eosinophils. Eosinophil infiltration in the BAL was quantified 30 minutes postgavage. Cromolyn treated mice exhibited a significant fourfold increase in BAL eosinophils compared to saline-treated controls. D: Representative PAS-stained histology showing mucin production in lungs of vehicle-treated ethanol-gavaged sensitized mice (left) and cromolyn-treated ethanol-gavaged sensitized mice (right). E: Morphometric analysis of histology at ×4 magnification from D shows a significant increase in mucin production in cromolyn-treated mice. Data are mean ± SEM, n = 6 to 7 per group. *P < 0.05, ***P < 0.001, using a priori one-tailed Student's t-test (A and B) and two-tailed Student's t-test (C and E).
Discussion
The role of alcohol on asthma, particularly the ethanol-induced mechanism of asthma exacerbations, remains undefined.11,29 To our knowledge, this is the first study in a relevant animal model examining the mechanism(s) of alcohol-triggered asthma. Clinical studies have demonstrated that alcohol will trigger asthma in Caucasians9,11,12as well as in Asian populations10,29,45 with or without a genetic deficiency of acetaldehyde dehydrogenase.9,11,12,28 Therefore, this study addresses an important public health issue, and our findings suggest alcohol should be included on the list with other asthma triggers such as cigarette smoke, allergens, respiratory infections, and exercise.
Acetaldehyde, the first metabolite of ethanol, has been shown to cause mast cell degranulation.38 Interestingly, acetaldehyde, but not ethanol, will stimulate histamine release from peripheral blood leukocytes.29 Other well-known asthma triggers such as automobile exhaust and tobacco smoke46 also contain acetaldehyde47,48; it is entirely appropriate to hypothesize that the mast cell is the main effector cell for the ethanol-induced asthmatic exacerbations observed here. We did indeed observe mast cell degranulation in ethanol-gavaged mice; however, the extent of their role in inducing other asthmatic responses is unclear because mast cell stabilization actually increased pulmonary inflammation and mucin production in this model. Our observations can possibly be explained by the fact that the mast cell stabilizers provide an increased resistance to apoptosis to the immune cells,49,50or the selective production and storage of discrete granule contents,50 such that tryptase granules are stabilized by cromolyn, but other granule stores containing chemokines and cytokines may be unaffected by the stabilizers. Alternatively, cytokines may not be stored in granules, and ethanol or its metabolites induce secretion of the cytokines through a mechanism independent of mast cell granule stabilization.51 Another possibility is that ethanol triggers mediator production in other cell types, for example, resident alveolar macrophages52 or bronchoepithelial cells.53
Perhaps the most striking finding presented here is the rapid and massive ethanol-induced mucin production in the lung. Within minutes of gavage, ethanol stimulated mucin production in the lungs of sensitized mice, but not in those of normal mice, suggesting an exclusive mechanism of induction in sensitized lungs and ruling out a nonspecific irritant effect of alcohol. The rapidity of induction is not uncommon; for example, gastric mucin induction has been shown to be as rapid as 5 minutes after insult.54 Ethanol-triggered mucin production in the asthmatic lung has not yet been reported, and the role of ethanol on airway mucin remains understudied.31 Our novel finding could have implications affecting disease morbidity, because refractory, severe airway disease has been thought to arise from the coexistence of multiple inflammatory stimuli,55 and mucus plugging is a major contributor in fatal asthma.40 In essence, ethanol-induced mucin overproduction may provide the additional inflammatory stimulus and push a patient into refractory asthma and increase disease morbidity.
Our data demonstrate a rapid inflammatory response is initiated on ethanol gavage in allergen-sensitized mice. These results do not agree with the majority of clinical and laboratory studies on the effects of ethanol, which have shown that alcohol consumption is generally immunosuppressive.56–58 Alcohol intake is a well-known risk factor for the increased incidence and severity of lung infections31,59–61 and has been shown to decrease cytokine and chemokine production, inflammatory cell recruitment, and other inflammatory responses in the lung. By contrast, our studies show that acute oral ethanol will actually trigger asthma-like pulmonary inflammation, although this only occurs in allergen-sensitized mice. The explanation for our conflicting evidence may lie in the ethanol-induced polarization of the immune system. Ethanol abuse has been shown to have a differential effect on Th1 and Th2 responses,62 and it is possible that this polarized effect may occur as rapidly as 30 minutes after ethanol ingestion. This could represent a potential explanation for how ethanol induces exacerbation of the pre-existing Th2 state in the CRA-sensitized mice whereas normal, nonasthmatic mice have no asthmatic inflammatory exacerbations. By suggesting ethanol can stimulate an immune response, our data do not necessarily contradict the immunosuppressive effects others have shown but instead describe a distinct mechanism for the role of ethanol in allergen-sensitized individuals.
An interesting finding was observed in the control group, the water-gavaged sensitized mice, which exhibited some increased inflammation when compared to sensitized, but nongavaged, mice. Our results showed water gavage induced slight mucin production and eosinophil infiltration within 30 minutes of the gavage procedure. These findings are not entirely surprising because others have shown vehicle gavage to modulate immunity and attributed this to the stress of the procedure.43,63 A possible mechanism for this may be the close network of nerves distributed along the oral and pharyngeal cavities and along the esophagus, including the vagal nerve,52 and the possible manipulation of this nerve system by the physical stress of gavage. A relationship between neural activity and asthma has been suggested since mast cells have been demonstrated to be in close proximity with the vagal nerve endings. Furthermore, neurotransmitters play an important role in the modulation of asthma. The neurotransmitter acetylcholine has been shown to exacerbate asthma by stimulating bronchoconstriction and increasing mucus secretion.44 In addition, the literature shows another neurotransmitter, serotonin, mediates the secretion of chemokines and cytokines from alveolar epithelial cells.44 Interestingly, serotonin is found in rodent mast cell granules,64 and its release can be triggered by changes in pressure across the gut wall.53 This may have relevance to ethanol administration by gavage because we gave our mice a relatively large bolus (300 μL) of water or ethanol.
Multiple factors were considered in the decision to give ethanol via oral gavage. Gavage assures a rapid and accurate dose of ethanol is administered to each mouse, and is presented to the mice in the same route and rapid timeframe by which binge-drinking humans consume alcohol. It could be argued that systemic alcohol administration by intraperitoneal or intravenous injection would have generated equivalent results; however, these routes are not as physiologically relevant, and do not mimic the biology of drinking and processing of ethanol through the gut. Important interactions between alcohol and the stomach need to be considered; for example, on entering the gut, ethanol can stimulate production of glucocorticoids and immunoregulatory cytokines and chemokines,33 all important factors that may play a role in modulating the asthmatic response and which should be included in a binge-drinking model studying inflammation.
In conclusion, our data demonstrate that ethanol gavage caused significant, substantial inflammatory exacerbations over water gavage in the parameters measured in our mice. For this reason, and for the explanations given above, we believe the results presented in this paper are representative of true ethanol-elicited inflammatory exacerbations and were investigated in a model suitable to the human context of acute ethanol exposure. Our data clearly demonstrate oral ethanol rapidly triggers asthma-like pulmonary inflammation in allergen-sensitized mice. The rapidity of the exacerbations suggests that the events are initiated by secreted preformed mediators such as chemokines (eotaxin-2) and cytokines (IL-4, IL-5, and IL-13) to induce eosinophil infiltration and mucin production, and lead to respiratory obstruction. The role of mast cells in this response is complex, and experiments presented here suggest mast cell stabilization may actually contribute to exacerbations of pulmonary inflammation and mucin production. As ethanol exposure failed to elicit inflammatory responses in naive, nonasthmatic mice, the ethanol-induced exacerbations appear to be restricted to allergen-sensitized individuals.
Acknowledgments
We thank Timothy Elton for his substantial analytical contributions to the mast cell stabilization experiments.
Footnotes
Supported by NIH grant R01 ES013538 (D.G.R.).
Supplemental material for this article can be found at http://ajp.amjpathol.org or at http://dx.doi.org/10.1016/j.ajpath.2012.05.020.
Supplementary data
References
- 1.Global Initiative for Asthma . Global Initiative for Asthma, National Heart, Lung, and Blood Institute; Bethesda, MD: 2010. Pocket Guide for Asthma Management and Prevention. [Google Scholar]
- 2.Akinbami L.J., Moorman J.E., Liu X. Asthma prevalence, health care use, and mortality: United States, 2005–2009. Natl Health Stat Report. 2011, Jan 12:1–14. [PubMed] [Google Scholar]
- 3.Goksel O., Celik G.E., Erkekol F.O., Gullu E., Mungan D., Misirligil Z. Triggers in adult asthma: are patients aware of triggers and doing right? Allergol Immunopathol (Madr) 2009;37:122–128. doi: 10.1016/S0301-0546(09)71723-9. [DOI] [PubMed] [Google Scholar]
- 4.Jackson J.E., Doescher M.P., Hart L.G. Problem drinking: rural and urban trends in America, 1995/1997 to 2003. Prev Med. 2006;43:122–124. doi: 10.1016/j.ypmed.2006.02.006. [DOI] [PubMed] [Google Scholar]
- 5.Serdula M.K., Brewer R.D., Gillespie C., Denny C.H., Mokdad A. Trends in alcohol use and binge drinking, 1985-1999: results of a multi-state survey. Am J Prev Med. 2004;26:294–298. doi: 10.1016/j.amepre.2003.12.017. [DOI] [PubMed] [Google Scholar]
- 6.Naimi T.S., Brewer R.D., Mokdad A., Denny C., Serdula M.K., Marks J.S. Binge drinking among US adults. JAMA. 2003;289:70–75. doi: 10.1001/jama.289.1.70. [DOI] [PubMed] [Google Scholar]
- 7.NIAAA Newsletter. 2004 Winter. NIAAA Council approves definition of binge drinking; p. 3. [Google Scholar]
- 8.Courtney K.E., Polich J. Binge drinking in young adults: data, definitions, and determinants. Psych Bul. 2009;135:142–156. doi: 10.1037/a0014414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ayres J.G. Alcohol-induced bronchial asthma. J Allergy Clin Immunol. 1997;99:860. doi: 10.1016/s0091-6749(97)80026-7. [DOI] [PubMed] [Google Scholar]
- 10.Harada S., Agarwal D.P., Goedde H.W. Mechanism of alcohol sensitivity and disulfiram-ethanol reaction. Subst Alcohol Actions Misuse. 1982;3:107–115. [PubMed] [Google Scholar]
- 11.Vally H., de Klerk N., Thompson P.J. Alcoholic drinks: important triggers for asthma. J Allergy Clin Immunol. 2000;105:462–467. doi: 10.1067/mai.2000.104548. [DOI] [PubMed] [Google Scholar]
- 12.Zellweger J.P. Alcohol-induced asthma: not only in Asians. J Allergy Clin Immunol. 1997;99:860. doi: 10.1016/s0091-6749(97)80027-9. [DOI] [PubMed] [Google Scholar]
- 13.Oldenburg P.J., Poole J.A., Sisson J.H. Alcohol reduces airway hyperresponsiveness (AHR) and allergic airway inflammation in mice. Am J Physiol Lung Cell Mol Physiol. 2012;302:L308–L315. doi: 10.1152/ajplung.00077.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Koh M.S., Irving L.B. Evidence-based pharmacologic treatment for mild asthma. Int J Clin Pract. 2007;61:1375–1379. doi: 10.1111/j.1742-1241.2007.01410.x. [DOI] [PubMed] [Google Scholar]
- 15.Vaickus L.J., Bouchard J., Kim J., Natarajan S., Remick D.G. Inbred and outbred mice have equivalent variability in a cockroach allergen-induced model of asthma. Comp Med. 2010;60:420–426. [PMC free article] [PubMed] [Google Scholar]
- 16.Vaickus L.J., Bouchard J., Kim J., Natarajan S., Remick D.G. Assessing pulmonary pathology by detailed examination of respiratory function. Am J Pathol. 2010;177:1861–1869. doi: 10.2353/ajpath.2010.100053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Vaickus L.J., Bouchard J., Kim J., Natarajan S., Remick D.G. Oral tolerance inhibits pulmonary eosinophilia in a cockroach allergen induced model of asthma: a randomized laboratory study. Respir Res. 2010;11:160. doi: 10.1186/1465-9921-11-160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lim R.T., Jr., Gentry R.T., Ito D., Yokoyama H., Baraona E., Lieber C.S. First-pass metabolism of ethanol is predominantly gastric. Alcohol Clin Exp Res. 1993;17:1337–1344. doi: 10.1111/j.1530-0277.1993.tb05250.x. [DOI] [PubMed] [Google Scholar]
- 19.Elgebaly S.A., Kozol R.A., Kreutzer D.L. Alcohol and immune system: role of gastric tissue. Prog Clin Biol Res. 1990;325:75–78. [PubMed] [Google Scholar]
- 20.Plackett T.P., Kovacs E.J. Acute models of ethanol exposure to mice. Methods Mol Biol. 2008;447:3–9. doi: 10.1007/978-1-59745-242-7_1. [DOI] [PubMed] [Google Scholar]
- 21.Fergani A., Oudart H., Gonzalez De Aguilar J.L., Fricker B., Rene F., Hocquette J.F., Meininger V., Dupuis L., Loeffler J.P. Increased peripheral lipid clearance in an animal model of amyotrophic lateral sclerosis. J Lipid Res. 2007;48:1571–1580. doi: 10.1194/jlr.M700017-JLR200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Remick D.G., Bolgos G., Copeland S., Siddiqui J. Role of interleukin-6 in mortality from and physiologic response to sepsis. Infect Immun. 2005;73:2751–2757. doi: 10.1128/IAI.73.5.2751-2757.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.de Waal Malefyt R., Figdor C.G., Huijbens R., Mohan-Peterson S., Bennett B., Culpepper J., Dang W., Zurawski G., de Vries J.E. Effects of IL-13 on phenotype, cytokine production, and cytotoxic function of human monocytes: Comparison with IL-4 and modulation by IFN-gamma or IL-10. J Immunol. 1993;151:6370–6381. [PubMed] [Google Scholar]
- 24.Natarajan S., Kim J., Bouchard J., Cruikshank W., Remick D.G. Reducing LPS content in cockroach allergens increases pulmonary cytokine production without increasing inflammation: a randomized laboratory study. BMC Pulm Med. 2011;11:12. doi: 10.1186/1471-2466-11-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Nemzek J.A., Siddiqui J., Remick D.G. Development and optimization of cytokine ELISAs using commercial antibody pairs. J Immunol Methods. 2001;255:149–157. doi: 10.1016/s0022-1759(01)00419-7. [DOI] [PubMed] [Google Scholar]
- 26.Santone D.J., Shahani R., Rubin B.B., Romaschin A.D., Lindsay T.F. Mast cell stabilization improves cardiac contractile function following hemorrhagic shock and resuscitation. Am J Physiol Heart Circ Physiol. 2008;294:H2456–H2464. doi: 10.1152/ajpheart.00925.2007. [DOI] [PubMed] [Google Scholar]
- 27.Holgate S.T., Peters-Golden M., Panettieri R.A., Henderson W.R., Jr. Roles of cysteinyl leukotrienes in airway inflammation, smooth muscle function, and remodeling. J Allergy Clin Immunol. 2003;111(Suppl):S18–S34. doi: 10.1067/mai.2003.25. discussion S34-S36. [DOI] [PubMed] [Google Scholar]
- 28.Ayres J.G., Clark T.J. Alcoholic drinks and asthma: a survey. Br J Dis Chest. 1983;77:370–375. [PubMed] [Google Scholar]
- 29.Shimoda T., Kohno S., Takao A., Fujiwara C., Matsuse H., Sakai H., Watanabe T., Hara K., Asai S. Investigation of the mechanism of alcohol-induced bronchial asthma. J Allergy Clin Immunol. 1996;97:74–84. doi: 10.1016/s0091-6749(96)70285-3. [DOI] [PubMed] [Google Scholar]
- 30.Urso T., Gavaler J.S., Van Thiel D.H. Blood ethanol levels in sober alcohol users seen in an emergency room. Life Sci. 1981;28:1053–1056. doi: 10.1016/0024-3205(81)90752-9. [DOI] [PubMed] [Google Scholar]
- 31.Sisson J.H. Alcohol and airways function in health and disease. Alcohol. 2007;41:293–307. doi: 10.1016/j.alcohol.2007.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Hlastala M.P. The alcohol breath test: a review. J Appl Physiol. 1998;84:401–408. doi: 10.1152/jappl.1998.84.2.401. [DOI] [PubMed] [Google Scholar]
- 33.Carson E.J., Pruett S.B. Development and characterization of a binge drinking model in mice for evaluation of the immunological effects of ethanol. Alcohol Clin Exp Res. 1996;20:132–138. doi: 10.1111/j.1530-0277.1996.tb01055.x. [DOI] [PubMed] [Google Scholar]
- 34.Di Santo E., Meazza C., Sironi M., Fruscella P., Mantovani A., Sipe J.D., Ghezzi P. IL-13 inhibits TNF production but potentiates that of IL-6 in vivo and ex vivo in mice. J Immunol. 1997;159:379–382. [PubMed] [Google Scholar]
- 35.Muchamuel T., Menon S., Pisacane P., Howard M.C., Cockayne D.A. IL-13 protects mice from lipopolysaccharide-induced lethal endotoxemia: correlation with down-modulation of TNF-alpha, IFN-gamma, and IL-12 production. J Immunol. 1997;158:2898–2903. [PubMed] [Google Scholar]
- 36.Hamelmann E., Gelfand E.W. IL-5-induced airway eosinophilia: the key to asthma? Immunol Rev. 2001;179:182–191. doi: 10.1034/j.1600-065x.2001.790118.x. [DOI] [PubMed] [Google Scholar]
- 37.Park H.M., Eo Y.W., Cha K.S., Kim Y.M., Lee K.B. Determination of free acetaldehyde in total blood for investigating the effect of aspartate on metabolism of alcohol in mice. J Chromatogr B Biomed Sci Appl. 1998;719:217–221. doi: 10.1016/s0378-4347(98)00389-2. [DOI] [PubMed] [Google Scholar]
- 38.Kawano T., Matsuse H., Kondo Y., Machida I., Saeki S., Tomari S., Mitsuta K., Obase Y., Fukushima C., Shimoda T., Kohno S. Acetaldehyde induces histamine release from human airway mast cells to cause bronchoconstriction. Int Arch Allergy Immunol. 2004;134:233–239. doi: 10.1159/000078771. [DOI] [PubMed] [Google Scholar]
- 39.Reed C.E. The importance of eosinophils in the immunology of asthma and allergic disease. Ann Allergy. 1994;72:376–380. [PubMed] [Google Scholar]
- 40.Kuyper L.M., Pare P.D., Hogg J.C., Lambert R.K., Ionescu D., Woods R., Bai T.R. Characterization of airway plugging in fatal asthma. Am J Med. 2003;115:6–11. doi: 10.1016/s0002-9343(03)00241-9. [DOI] [PubMed] [Google Scholar]
- 41.Asti C., Melillo G., Caselli G.F., Daffonchio L., Hernandez A., Clavenna G., Omini C. Effectiveness of carbocysteine lysine salt monohydrate on models of airway inflammation and hyperresponsiveness. Pharmacol Res. 1995;31:387–392. doi: 10.1016/1043-6618(95)80094-8. [DOI] [PubMed] [Google Scholar]
- 42.Shimura S., Andoh Y., Haraguchi M., Shirato K. Continuity of airway goblet cells and intraluminal mucus in the airways of patients with bronchial asthma. Eur Respir J. 1996;9:1395–1401. doi: 10.1183/09031936.96.09071395. [DOI] [PubMed] [Google Scholar]
- 43.Roberts R.A., Soames A.R., James N.H., Gill J.H., Wheeldon E.B. Dosing-induced stress causes hepatocyte apoptosis in rats primed by the rodent nongenotoxic hepatocarcinogen cyproterone acetate. Toxicol Appl Pharmacol. 1995;135:192–199. doi: 10.1006/taap.1995.1223. [DOI] [PubMed] [Google Scholar]
- 44.Mirotti L., Castro J., Costa-Pinto F.A., Russo M. Neural pathways in allergic inflammation. J Allergy (Cairo) 2010;2010:491928. doi: 10.1155/2010/491928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Harada S., Agarwal D.P., Goedde H.W. Aldehyde dehydrogenase deficiency as cause of facial flushing reaction to alcohol in Japanese. Lancet. 1981;2:982. doi: 10.1016/s0140-6736(81)91172-7. [DOI] [PubMed] [Google Scholar]
- 46.Tatum A.J., Shapiro G.G. The effects of outdoor air pollution and tobacco smoke on asthma. Immunol Allergy Clin North Am. 2005;25:15–30. doi: 10.1016/j.iac.2004.09.003. [DOI] [PubMed] [Google Scholar]
- 47.Cao J., Belluzzi J.D., Loughlin S.E., Keyler D.E., Pentel P.R., Leslie F.M. Acetaldehyde, a major constituent of tobacco smoke, enhances behavioral, endocrine, and neuronal responses to nicotine in adolescent and adult rats. Neuropsychopharmacology. 2007;32:2025–2035. doi: 10.1038/sj.npp.1301327. [DOI] [PubMed] [Google Scholar]
- 48.Komazaki Y., Narita Y., Tanaka S. Development of an automated measurement system using a diffusion scrubber and high-performance liquid chromatography for the monitoring of formaldehyde and acetaldehyde in automotive exhaust gas. Analyst. 1998;123:2343–2349. doi: 10.1039/a805999c. [DOI] [PubMed] [Google Scholar]
- 49.Bloom J.W., Kaltenborn W.T., Quan S.F. Risk factors in a general population for snoring: Importance of cigarette smoking and obesity. Chest. 1988;93:678–683. doi: 10.1378/chest.93.4.678. [DOI] [PubMed] [Google Scholar]
- 50.Hoshino Y., Ayuse T., Kurata S., Schneider H., Kirkness J.P., Patil S.P., Schwartz A.R., Oi K. The compensatory responses to upper airway obstruction in normal subjects under propofol anesthesia. Respir Physiol Neurobiol. 2009;166:24–31. doi: 10.1016/j.resp.2009.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Giulian D., Ingeman J.E. Colony-stimulating factors as promoters of ameboid microglia. J Neurosci. 1988;8:4707–4717. doi: 10.1523/JNEUROSCI.08-12-04707.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Myers A.C. Electrophysiology of airway nerves. Curr Protoc Pharmacol. 2007 doi: 10.1002/0471141755.ph1110s39. [DOI] [PubMed] [Google Scholar]; Chapter 11:Unit11.10
- 53.Bearcroft C.P., Perrett D., Farthing M.J. Postprandial plasma 5-hydroxytryptamine in diarrhoea predominant irritable bowel syndrome: a pilot study. Gut. 1998;42:42–46. doi: 10.1136/gut.42.1.42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Miyake K., Tanaka T., McNeil P.L. Disruption-induced mucus secretion: repair and protection. PLoS Biol. 2006;4:e276. doi: 10.1371/journal.pbio.0040276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Pavord I.D., Birring S.S., Berry M., Green R.H., Brightling C.E., Wardlaw A.J. Multiple inflammatory hits and the pathogenesis of severe airway disease. Eur Respir J. 2006;27:884–888. doi: 10.1183/09031936.06.00128105. [DOI] [PubMed] [Google Scholar]
- 56.Cook R.T. Alcohol abuse, alcoholism, and damage to the immune system: a review. Alcohol Clin Exp Res. 1998;22:1927–1942. [PubMed] [Google Scholar]
- 57.Pavia C.S., La Mothe M., Kavanagh M. Influence of alcohol on antimicrobial immunity. Biomed Pharmacother. 2004;58:84–89. doi: 10.1016/j.biopha.2003.12.005. [DOI] [PubMed] [Google Scholar]
- 58.Szabo G. Consequences of alcohol consumption on host defence. Alcohol Alcohol. 1999;34:830–841. doi: 10.1093/alcalc/34.6.830. [DOI] [PubMed] [Google Scholar]
- 59.Goral J., Karavitis J., Kovacs E.J. Exposure-dependent effects of ethanol on the innate immune system. Alcohol. 2008;42:237–247. doi: 10.1016/j.alcohol.2008.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Raasch C.E., Zhang P., Siggins R.W., 2nd, LaMotte L.R., Nelson S., Bagby G.J. Acute alcohol intoxication impairs the hematopoietic precursor cell response to pneumococcal pneumonia. Alcohol Clin Exp Res. 2010;34:2035–2043. doi: 10.1111/j.1530-0277.2010.01291.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Gamble L., Mason C.M., Nelson S. The effects of alcohol on immunity and bacterial infection in the lung. Med Mal Infect. 2006;36:72–77. doi: 10.1016/j.medmal.2005.08.010. [DOI] [PubMed] [Google Scholar]
- 62.Peterson J.D., Vasquez K., Waltenbaugh C. Interleukin-12 therapy restores cell-mediated immunity in ethanol-consuming mice. Alcohol Clin Exp Res. 1998;22:245–251. [PubMed] [Google Scholar]
- 63.Dozier M.M., Ratajczak H.V., Sothern R.B., Thomas P.T. The influence of vehicle gavage on seasonality of immune system parameters in the B6C3F1 mouse. Fundam Appl Toxicol. 1997;38:116–122. doi: 10.1006/faat.1997.2335. [DOI] [PubMed] [Google Scholar]
- 64.Hay D.W., Torphy T.J., Undem B.J. Cysteinyl leukotrienes in asthma: old mediators up to new tricks. Trends Pharmacol Sci. 1995;16:304–309. doi: 10.1016/s0165-6147(00)89059-8. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.