Abstract
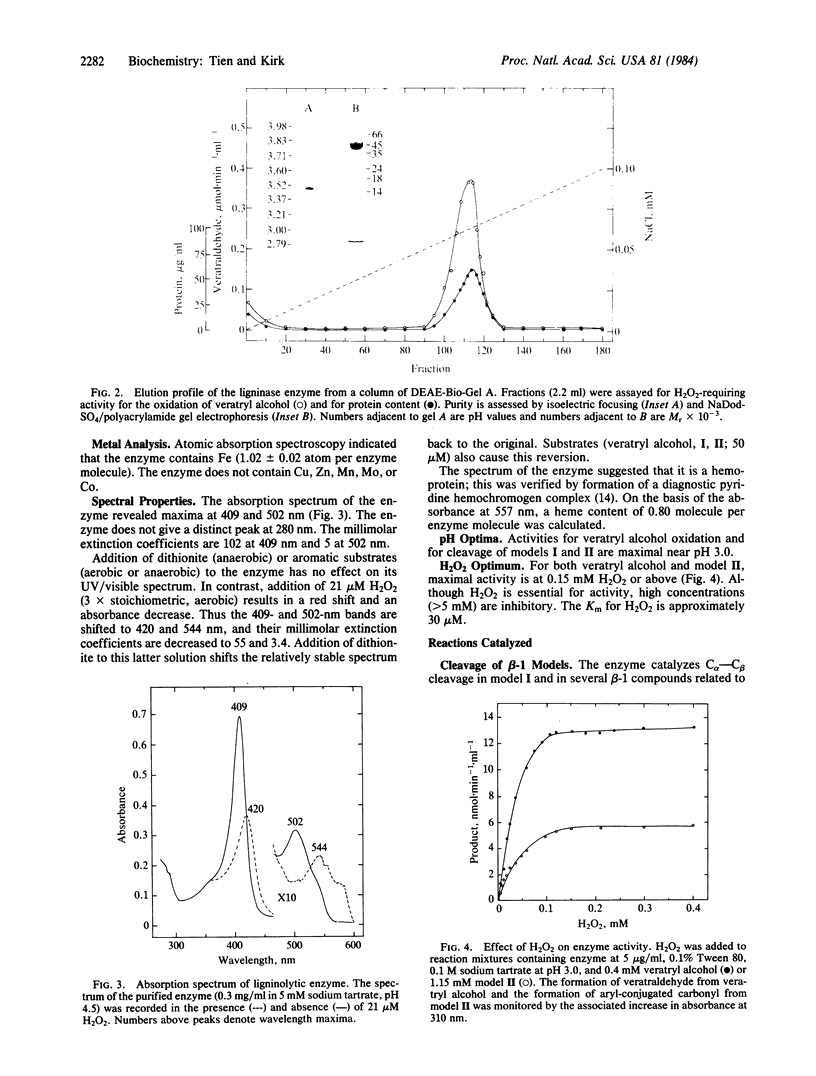
An extracellular lignin-degrading enzyme from the basidiomycete Phanerochaete chrysosporium Burdsall was purified to homogeneity by ion-exchange chromatography. The 42,000-dalton ligninase contains one protoheme IX per molecule. It catalyzes, nonstereospecifically, several oxidations in the alkyl side chains of lignin-related compounds: Cα—Cβ cleavage in lignin-related compounds of the type aryl—CαHOH—CβHR—CγH2OH (R = -aryl or -O-aryl), oxidation of benzyl alcohols to aldehydes or ketones, intradiol cleavage in phenylglycol structures, and hydroxylation of benzylic methylene groups. It also catalyzes oxidative coupling of phenols, perhaps explaining the long-recognized association between phenol oxidation and lignin degradation. All reactions require H2O2. The Cα—Cβ cleavage and methylene hydroxylation reactions involve substrate oxygenation; the oxygen atom is from O2 and not H2O2. Thus the enzyme is an oxygenase, unique in its requirement for H2O2.
Keywords: hemoprotein, stereospecificity, 18O2 incorporation, white-rot fungi, wood decay
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- CHANCE B. The spectra of the enzyme-substrate complexes of catalase and peroxidase. Arch Biochem Biophys. 1952 Dec;41(2):404–415. doi: 10.1016/0003-9861(52)90469-4. [DOI] [PubMed] [Google Scholar]
- Fairbanks G., Steck T. L., Wallach D. F. Electrophoretic analysis of the major polypeptides of the human erythrocyte membrane. Biochemistry. 1971 Jun 22;10(13):2606–2617. doi: 10.1021/bi00789a030. [DOI] [PubMed] [Google Scholar]
- Glenn J. K., Morgan M. A., Mayfield M. B., Kuwahara M., Gold M. H. An extracellular H2O2-requiring enzyme preparation involved in lignin biodegradation by the white rot basidiomycete Phanerochaete chrysosporium. Biochem Biophys Res Commun. 1983 Aug 12;114(3):1077–1083. doi: 10.1016/0006-291x(83)90672-1. [DOI] [PubMed] [Google Scholar]
- Guengerich F. P. Preparation and properties of highly purified cytochrome p-450 and NADPH-cytochrome P-450 reductase from pulmonary microsomes of untreated rabbits. Mol Pharmacol. 1977 Sep;13(5):911–923. [PubMed] [Google Scholar]
- Miller M. J., Gennis R. B. The purification and characterization of the cytochrome d terminal oxidase complex of the Escherichia coli aerobic respiratory chain. J Biol Chem. 1983 Aug 10;258(15):9159–9165. [PubMed] [Google Scholar]
- Pajot P., Groudinsky O. Molecular weight and quaternary structure of yeast L-lactate dehydrogenase (cytochrome b2). 2. Revised heme extinction coefficients and minimal molecular weight. Eur J Biochem. 1970 Jan;12(1):158–164. doi: 10.1111/j.1432-1033.1970.tb00833.x. [DOI] [PubMed] [Google Scholar]
- Tien M., Kirk T. K. Lignin-Degrading Enzyme from the Hymenomycete Phanerochaete chrysosporium Burds. Science. 1983 Aug 12;221(4611):661–663. doi: 10.1126/science.221.4611.661. [DOI] [PubMed] [Google Scholar]
- Weber K., Osborn M. The reliability of molecular weight determinations by dodecyl sulfate-polyacrylamide gel electrophoresis. J Biol Chem. 1969 Aug 25;244(16):4406–4412. [PubMed] [Google Scholar]