Abstract
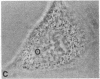
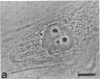
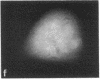
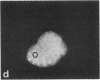
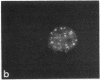
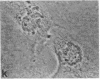
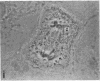
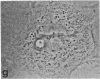
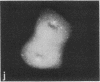
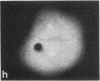
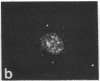
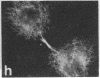
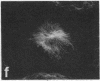
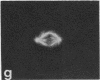
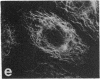
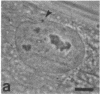
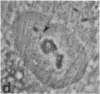
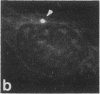
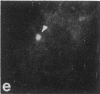
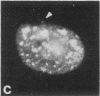
Protein phosphorylation has been suggested as an important control mechanism for the events leading toward the initiation and completion of mitosis. Using a monoclonal antibody recognizing a class of phosphoproteins abundant in mitotic cells, we demonstrated the localization of a subset of these phosphoproteins to several discrete mitotic structures. Patchy immunofluorescence was present in the interphase nuclei, but a significant increase in nuclear immunofluorescence was apparent at prophase. Subsequent mitotic stages demonstrated that immunoreactive material was particularly apparent at microtubule organizing centers, namely, centrosomes, kinetochores, and midbodies. Intense centrosomal localization occurred at the prophase-prometaphase transition and persisted until the reformation of the nuclear membrane in early G1. The cytoplasm of mitotic cells also contained immunoreactive material in sharp contrast to interphase cells that exhibited no cytoplasmic fluorescent staining. Much of the diffuse immunofluorescent cytoplasmic material was removed by a brief lysis of the cells with 0.15% Triton X-100 prior to fixation. The localization of the remaining immunoreactive material after detergent lysis to mitotic microtubule organizing centers suggests that they contain phosphoprotein structural components important, perhaps, in the mitotic phase-interphase transition.
Full text
PDF
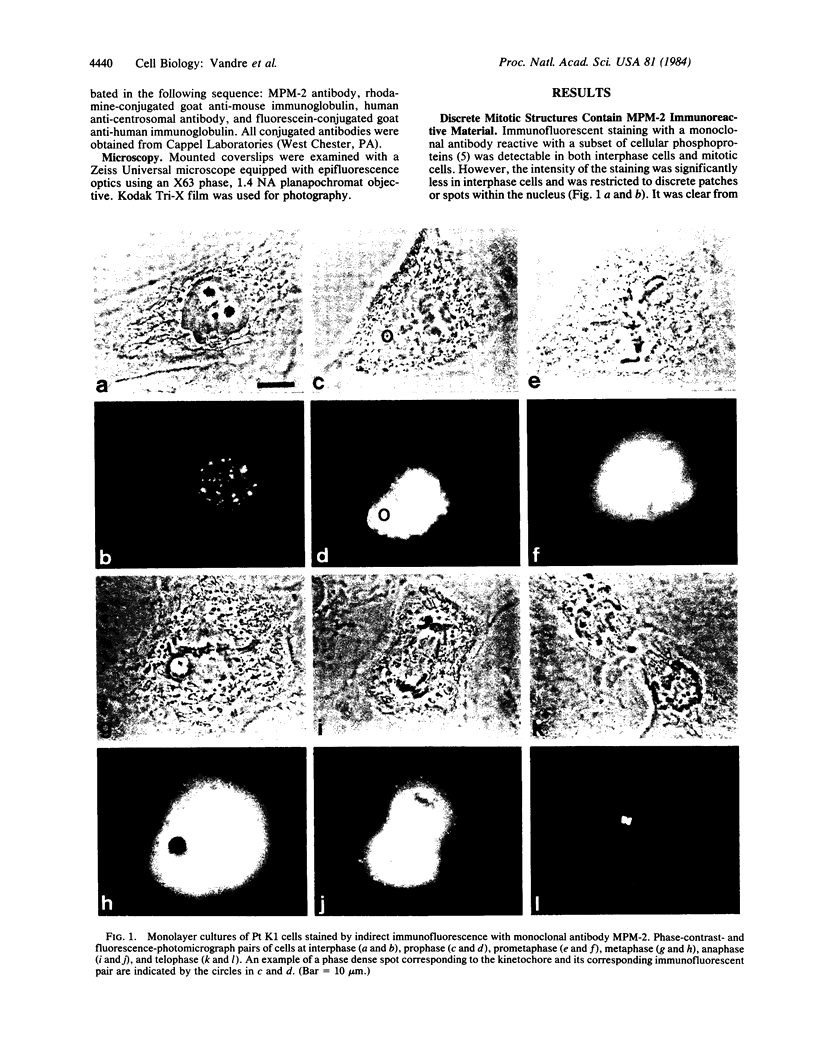
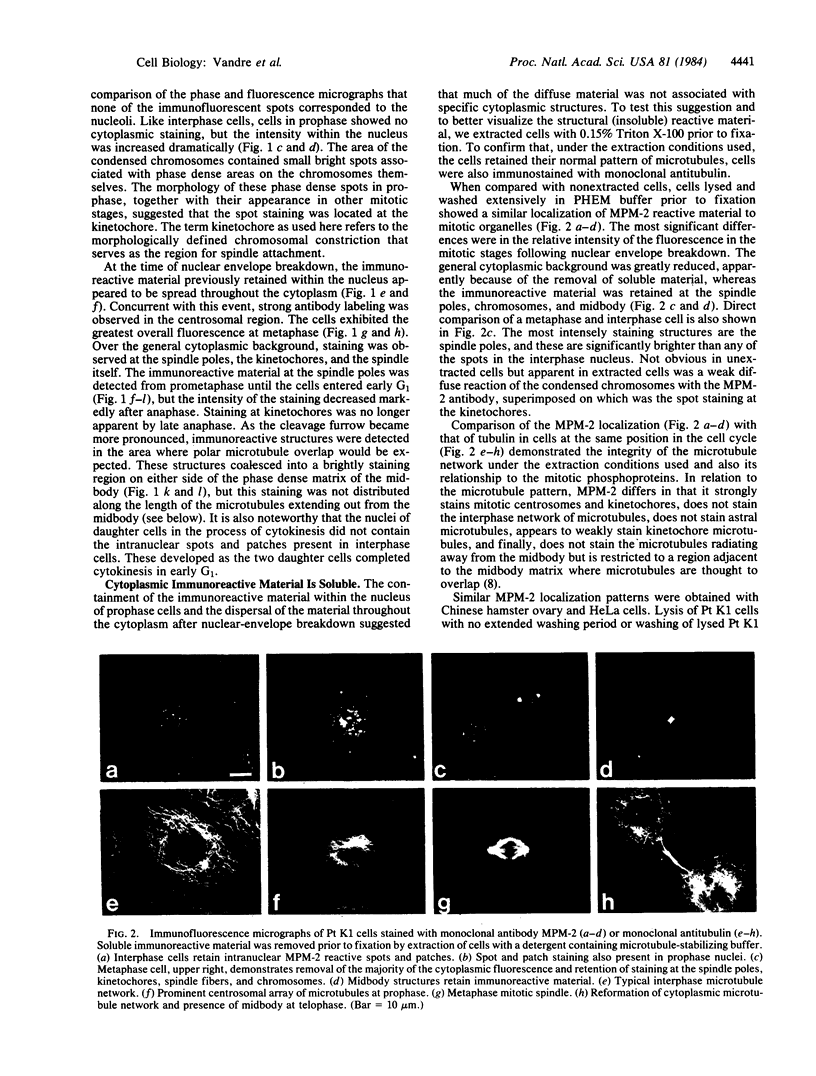
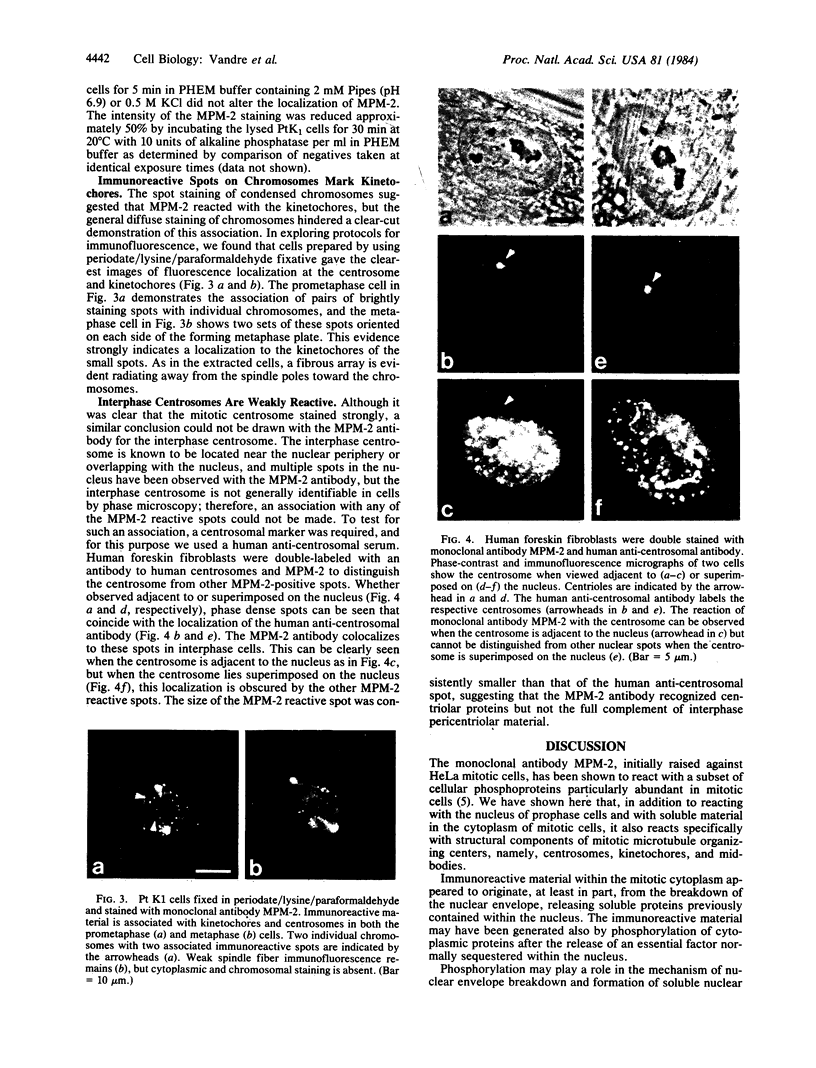

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Adlakha R. C., Sahasrabuddhe C. G., Wright D. A., Lindsey W. F., Rao P. N. Localization of mitotic factors on metaphase chromosomes. J Cell Sci. 1982 Apr;54:193–206. doi: 10.1242/jcs.54.1.193. [DOI] [PubMed] [Google Scholar]
- Brenner S., Pepper D., Berns M. W., Tan E., Brinkley B. R. Kinetochore structure, duplication, and distribution in mammalian cells: analysis by human autoantibodies from scleroderma patients. J Cell Biol. 1981 Oct;91(1):95–102. doi: 10.1083/jcb.91.1.95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cox J. V., Schenk E. A., Olmsted J. B. Human anticentromere antibodies: distribution, characterization of antigens, and effect on microtubule organization. Cell. 1983 Nov;35(1):331–339. doi: 10.1016/0092-8674(83)90236-2. [DOI] [PubMed] [Google Scholar]
- Davis F. M., Tsao T. Y., Fowler S. K., Rao P. N. Monoclonal antibodies to mitotic cells. Proc Natl Acad Sci U S A. 1983 May;80(10):2926–2930. doi: 10.1073/pnas.80.10.2926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Franke W. W., Schmid E., Wellsteed J., Grund C., Gigi O., Geiger B. Change of cytokeratin filament organization during the cell cycle: selective masking of an immunologic determinant in interphase PtK2 cells. J Cell Biol. 1983 Oct;97(4):1255–1260. doi: 10.1083/jcb.97.4.1255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gerace L., Blobel G. The nuclear envelope lamina is reversibly depolymerized during mitosis. Cell. 1980 Jan;19(1):277–287. doi: 10.1016/0092-8674(80)90409-2. [DOI] [PubMed] [Google Scholar]
- Gerace L., Blum A., Blobel G. Immunocytochemical localization of the major polypeptides of the nuclear pore complex-lamina fraction. Interphase and mitotic distribution. J Cell Biol. 1978 Nov;79(2 Pt 1):546–566. doi: 10.1083/jcb.79.2.546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henry S. M., Hodge L. D. Nuclear matrix: a cell-cycle-dependent site of increased intranuclear protein phosphorylation. Eur J Biochem. 1983 Jun 1;133(1):23–29. doi: 10.1111/j.1432-1033.1983.tb07425.x. [DOI] [PubMed] [Google Scholar]
- Izant J. G., Weatherbee J. A., McIntosh J. R. A microtubule-associated protein antigen unique to mitotic spindle microtubules in PtK1 cells. J Cell Biol. 1983 Feb;96(2):424–434. doi: 10.1083/jcb.96.2.424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kletzien R. F., Miller M. R., Pardee A. B. Unique cytoplasmic phosphoproteins are associated with cell growth arrest. Nature. 1977 Nov 3;270(5632):57–59. doi: 10.1038/270057a0. [DOI] [PubMed] [Google Scholar]
- Maller J., Wu M., Gerhart J. C. Changes in protein phosphorylation accompanying maturation of Xenopus laevis oocytes. Dev Biol. 1977 Jul 15;58(2):295–312. doi: 10.1016/0012-1606(77)90093-8. [DOI] [PubMed] [Google Scholar]
- McLean I. W., Nakane P. K. Periodate-lysine-paraformaldehyde fixative. A new fixation for immunoelectron microscopy. J Histochem Cytochem. 1974 Dec;22(12):1077–1083. doi: 10.1177/22.12.1077. [DOI] [PubMed] [Google Scholar]
- Miake-Lye R., Newport J., Kirschner M. Maturation-promoting factor induces nuclear envelope breakdown in cycloheximide-arrested embryos of Xenopus laevis. J Cell Biol. 1983 Jul;97(1):81–91. doi: 10.1083/jcb.97.1.81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mullins J. M., McIntosh J. R. Isolation and initial characterization of the mammalian midbody. J Cell Biol. 1982 Sep;94(3):654–661. doi: 10.1083/jcb.94.3.654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nelkin B., Nichols C., Vogelstein B. Protein factor(s) from mitotic CHO cells induce meiotic maturation in Xenopus laevis oocytes. FEBS Lett. 1980 Jan 14;109(2):233–238. doi: 10.1016/0014-5793(80)81094-5. [DOI] [PubMed] [Google Scholar]
- Schliwa M., van Blerkom J. Structural interaction of cytoskeletal components. J Cell Biol. 1981 Jul;90(1):222–235. doi: 10.1083/jcb.90.1.222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sunkara P. S., Wright D. A., Rao P. N. Mitotic factors from mammalian cells induce germinal vesicle breakdown and chromosome condensation in amphibian oocytes. Proc Natl Acad Sci U S A. 1979 Jun;76(6):2799–2802. doi: 10.1073/pnas.76.6.2799. [DOI] [PMC free article] [PubMed] [Google Scholar]