Abstract
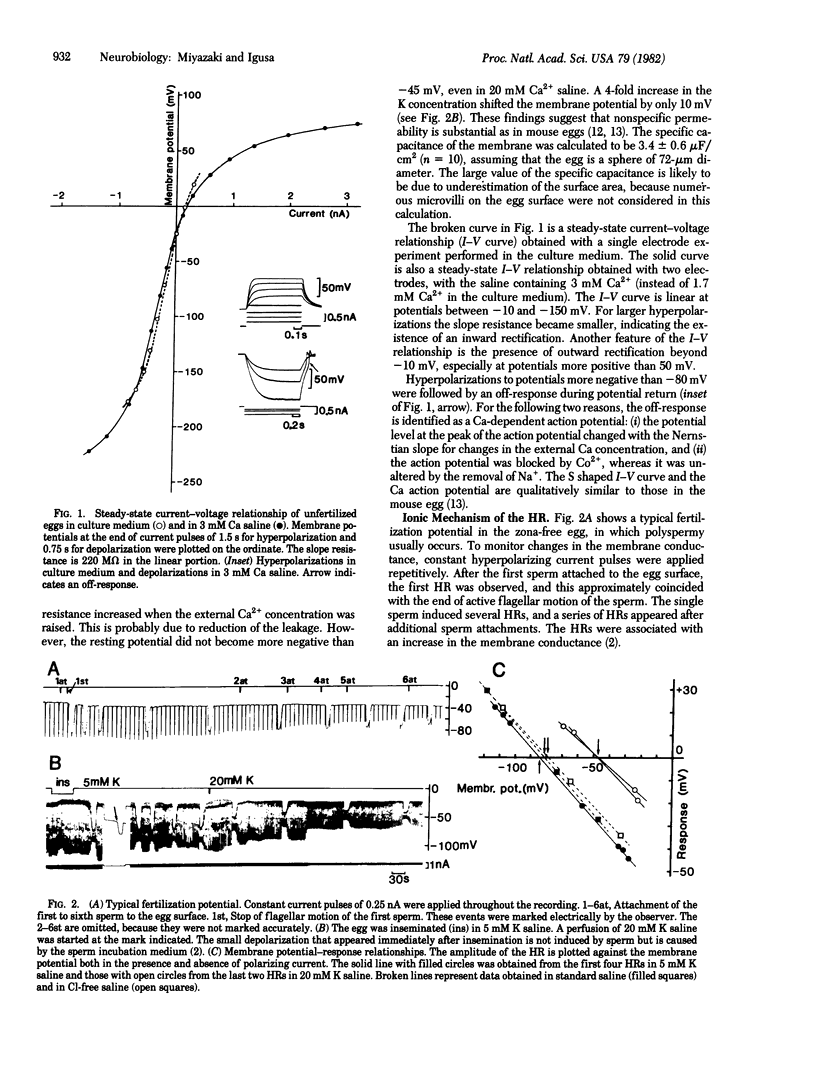
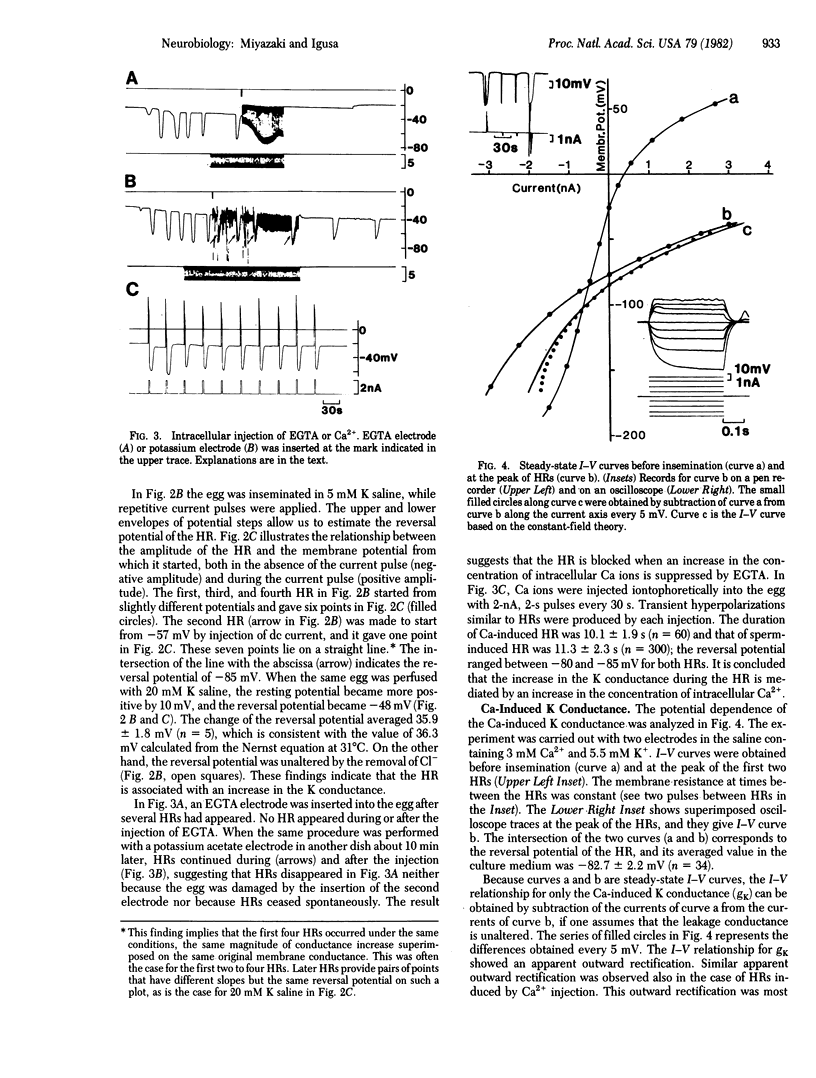
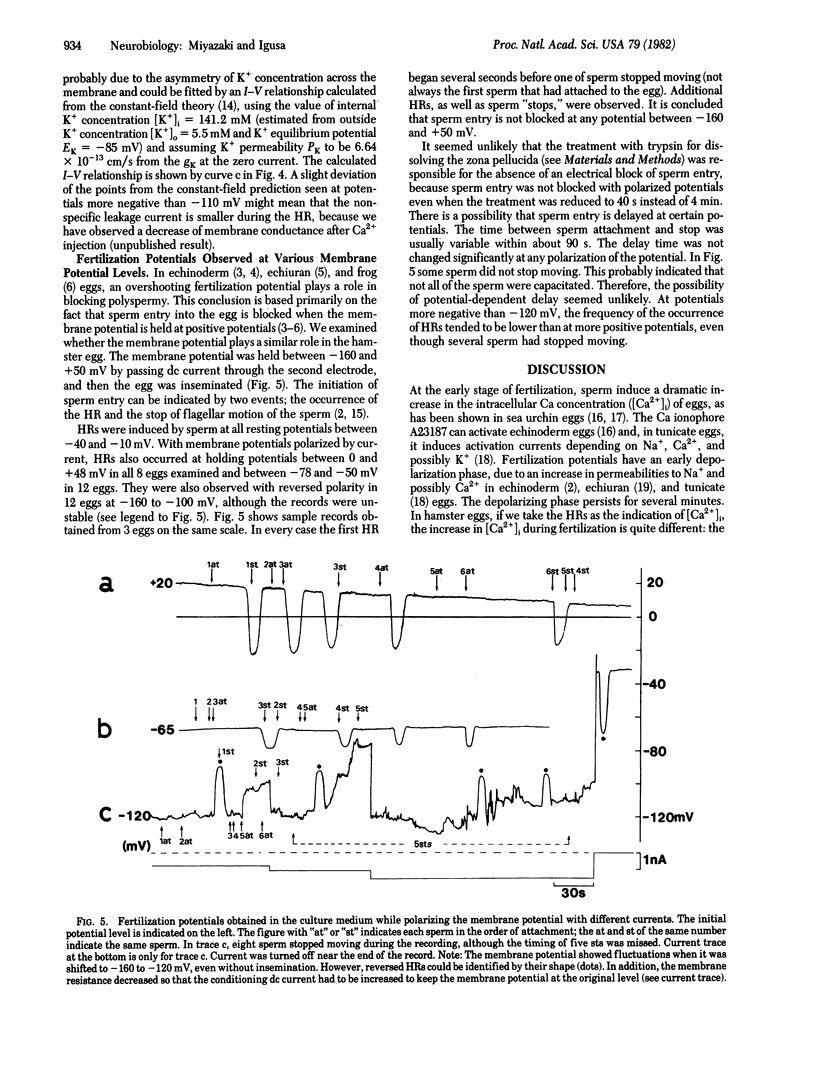
Golden hamster eggs respond to fertilization with recurring hyperpolarizations [Miyazaki, S. & Igusa, Y. (1981) Nature (London) 290, 703-705]. We analyzed the ionic mechanism of the fertilization potential and examined whether the fertilization potential plays a role in polyspermy block. Each hyperpolarizing response (HR) during fertilization is found to be caused by an increase in the K conductance activated by an increase in the intracellular Ca2+ concentration. This conclusion is based on the following: (i) The reversal potential of the HR shifted with the Nernstian slope for K ions when the external K concentration was changed, whereas it was unaltered by the removal of Cl ions. (ii) The HR was blocked by the intracellular injection of EGTA. (iii) Injection of Ca2+ into an egg induced a hyperpolarization of the membrane similar to the HR. The Ca-activated K conductance shows an apparent outward rectification, which could be explained by an asymmetric distribution of K ions across the membrane. The HR associated with sperm entry into the egg occurred at any membrane potential between -160 and +50 mV. Therefore, a potential-dependent block of sperm entry does not occur in the hamster egg.
Full text
PDF

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- AUSTIN C. R. The capacitation of the mammalian sperm. Nature. 1952 Aug 23;170(4321):326–326. doi: 10.1038/170326a0. [DOI] [PubMed] [Google Scholar]
- Cross N. L., Elinson R. P. A fast block to polyspermy in frogs mediated by changes in the membrane potential. Dev Biol. 1980 Mar;75(1):187–198. doi: 10.1016/0012-1606(80)90154-2. [DOI] [PubMed] [Google Scholar]
- Dos Reis G. A., Oliveira-Castro G. M. Electrophysiology of phagocytic membranes. I. Potassium-dependent slow membrane hyperpolarizations in mice macrophages. Biochim Biophys Acta. 1977 Sep 19;469(3):257–263. doi: 10.1016/0005-2736(77)90161-4. [DOI] [PubMed] [Google Scholar]
- Gallin E. K., Gallin J. I. Interaction of chemotactic factors with human macrophages. Induction of transmembrane potential changes. J Cell Biol. 1977 Oct;75(1):277–289. doi: 10.1083/jcb.75.1.277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gallin E. K., Wiederhold M. L., Lipsky P. E., Rosenthal A. S. Spontaneous and induced membrane hyperpolarizations in macrophages. J Cell Physiol. 1975 Dec;86 (Suppl 2)(3 Pt 2):653–661. doi: 10.1002/jcp.1040860510. [DOI] [PubMed] [Google Scholar]
- Gould-Somero M., Jaffe L. A., Holland L. Z. Electrically mediated fast polyspermy block in eggs of the marine worm, Urechis caupo. J Cell Biol. 1979 Aug;82(2):426–440. doi: 10.1083/jcb.82.2.426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HODGKIN A. L., KATZ B. The effect of sodium ions on the electrical activity of giant axon of the squid. J Physiol. 1949 Mar 1;108(1):37–77. doi: 10.1113/jphysiol.1949.sp004310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hagiwara S., Jaffe L. A. Electrical properties of egg cell membranes. Annu Rev Biophys Bioeng. 1979;8:385–416. doi: 10.1146/annurev.bb.08.060179.002125. [DOI] [PubMed] [Google Scholar]
- Jaffe L. A. Fast block to polyspermy in sea urchin eggs is electrically mediated. Nature. 1976 May 6;261(5555):68–71. doi: 10.1038/261068a0. [DOI] [PubMed] [Google Scholar]
- Jaffe L. A., Gould-Somero M., Holland L. Ionic mechanism of the fertilization potential of the marine worm, Urechis caupo (Echiura). J Gen Physiol. 1979 Apr;73(4):469–492. doi: 10.1085/jgp.73.4.469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuba K., Nishi S. Rhythmic hyperpolarizations and depolarization of sympathetic ganglion cells induced by caffeine. J Neurophysiol. 1976 May;39(3):547–563. doi: 10.1152/jn.1976.39.3.547. [DOI] [PubMed] [Google Scholar]
- Meech R. W. Calcium-dependent potassium activation in nervous tissues. Annu Rev Biophys Bioeng. 1978;7:1–18. doi: 10.1146/annurev.bb.07.060178.000245. [DOI] [PubMed] [Google Scholar]
- Miyazaki S., Hirai S. Fast polyspermy block and activation potential. Correlated changes during oocyte maturation of a starfish. Dev Biol. 1979 Jun;70(2):327–340. doi: 10.1016/0012-1606(79)90031-9. [DOI] [PubMed] [Google Scholar]
- Nelson P. G., Peacock J., Minna J. An active electrical response in fibroblasts. J Gen Physiol. 1972 Jul;60(1):58–71. doi: 10.1085/jgp.60.1.58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okada Y., Doida Y., Roy G., Tsuchiya W., Inouye K., Inouye A. Oscillations of membrane potential in L cells. I. Basic characteristics. J Membr Biol. 1977 Aug 4;35(4):319–335. doi: 10.1007/BF01869957. [DOI] [PubMed] [Google Scholar]
- Okada Y., Tsuchiya W., Yada T., Yano J., Yawo H. Phagocytic activity and hyperpolarizing responses in L-strain mouse fibroblasts. J Physiol. 1981;313:101–119. doi: 10.1113/jphysiol.1981.sp013653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okamoto H., Takahashi K., Yamashita N. Ionic currents through the membrane of the mammalian oocyte and their comparison with those in the tunicate and sea urchin. J Physiol. 1977 May;267(2):465–495. doi: 10.1113/jphysiol.1977.sp011822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Powers R. D., Tupper J. T. Some electrophysiological and permeability properties of the mouse egg. Dev Biol. 1974 Jun;38(2):320–331. doi: 10.1016/0012-1606(74)90010-4. [DOI] [PubMed] [Google Scholar]
- Steinhardt R. A., Epel D. Activation of sea-urchin eggs by a calcium ionophore. Proc Natl Acad Sci U S A. 1974 May;71(5):1915–1919. doi: 10.1073/pnas.71.5.1915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinhardt R., Zucker R., Schatten G. Intracellular calcium release at fertilization in the sea urchin egg. Dev Biol. 1977 Jul 1;58(1):185–196. doi: 10.1016/0012-1606(77)90084-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toyoda Y., Chang M. C. Fertilization of rat eggs in vitro by epididymal spermatozoa and the development of eggs following transfer. J Reprod Fertil. 1974 Jan;36(1):9–22. doi: 10.1530/jrf.0.0360009. [DOI] [PubMed] [Google Scholar]
- Yanagimachi R. In vitro capacitation of golden hamster spermatozoa by homologous and heterologous blood sera. Biol Reprod. 1970 Oct;3(2):147–153. doi: 10.1093/biolreprod/3.2.147. [DOI] [PubMed] [Google Scholar]
- Yanagimachi R. In vitro capacitation of hamster spermatozoa by follicular fluid. J Reprod Fertil. 1969 Mar;18(2):275–286. doi: 10.1530/jrf.0.0180275. [DOI] [PubMed] [Google Scholar]
- Yanagimachi R., Noda Y. D. Electron microscope studies of sperm incorporation into the golden hamster egg. Am J Anat. 1970 Aug;128(4):429–462. doi: 10.1002/aja.1001280404. [DOI] [PubMed] [Google Scholar]