Abstract
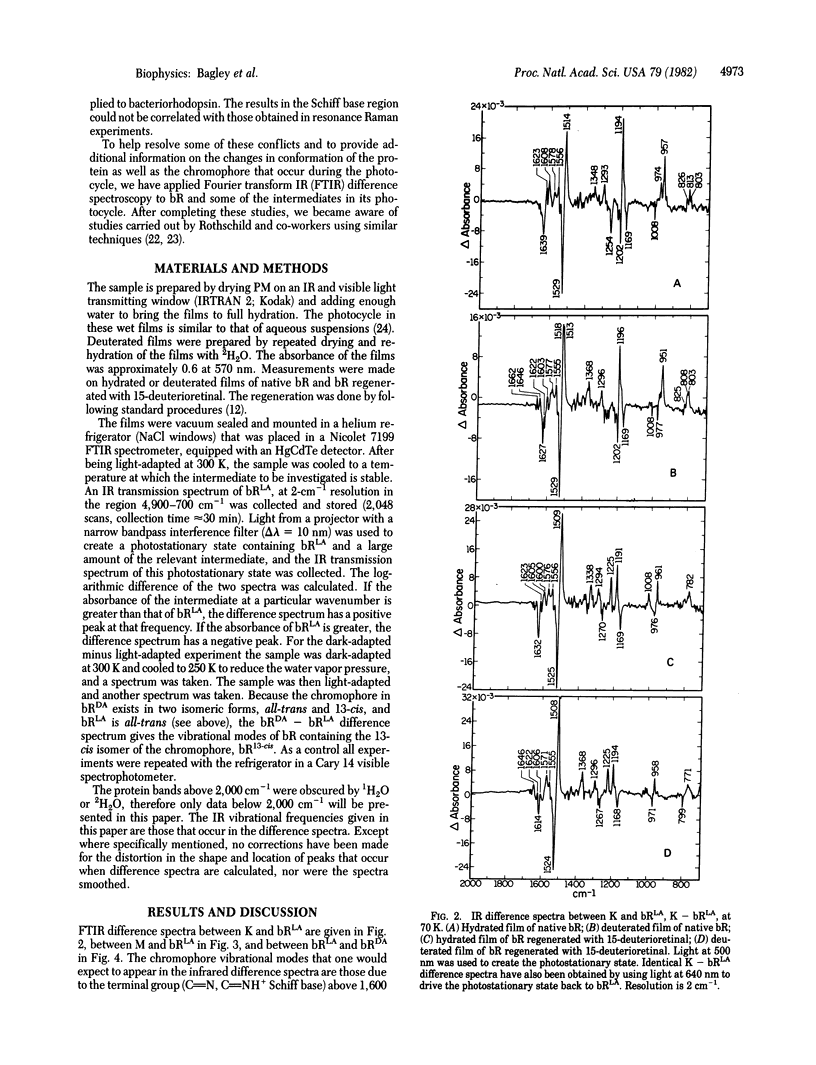
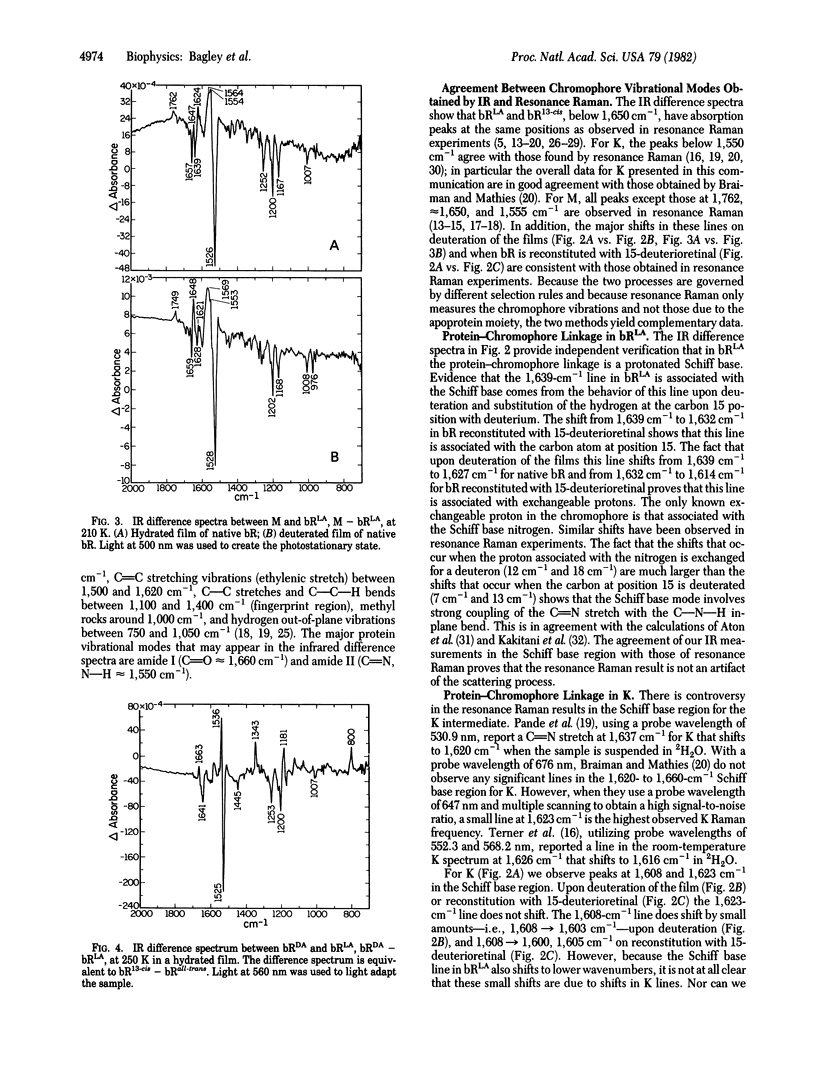
Fourier transform infrared difference spectroscopy has been used to obtain the vibrational modes in the chromophore and apoprotein that change in intensity or position between light-adapted bacteriorhodopsin and the K and M intermediates in its photocycle and between dark-adapted and light-adapted bacteriorhodopsin. Our infrared measurements provide independent verification of resonance Raman results that in light-adapted bacteriorhodopsin the protein-chromophore linkage is a protonated Schiff base and in the M state the Schiff base is unprotonated. Although we cannot unambiguously identify the Schiff base stretching frequency in the K state, the most likely interpretation of deuterium shifts of the chromophore hydrogen out-of-plane vibrations is that the Schiff base in K is protonated. The intensity of the hydrogen out-of-plane vibrations in the K state compared with the intensities of those in light-adapted and dark-adapted bacteriorhodopsin shows that the conformation of the chromophore in K is considerably distorted. In addition, we find evidence that the conformation of the protein changes during the photocycle.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aton B., Doukas A. G., Callender R. H., Becher B., Ebrey T. G. Resonance Raman studies of the purple membrane. Biochemistry. 1977 Jun 28;16(13):2995–2999. doi: 10.1021/bi00632a029. [DOI] [PubMed] [Google Scholar]
- Aton B., Doukas A. G., Callender R. H., Becher B., Ebrey T. G. Resonance Raman study of the dark-adapted form of the purple membrane protein. Biochim Biophys Acta. 1979 Feb 26;576(2):424–428. doi: 10.1016/0005-2795(79)90417-3. [DOI] [PubMed] [Google Scholar]
- Aton B., Doukas A. G., Narva D., Callender R. H., Dinur U., Honig B. Resonance Raman studies of the primary photochemical event in visual pigments. Biophys J. 1980 Jan;29(1):79–94. doi: 10.1016/S0006-3495(80)85119-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Becher B., Cassim J. Y. Effects of light adaptation on the purple membrane structure of Halobacterium halobium. Biophys J. 1976 Oct;16(10):1183–1200. doi: 10.1016/S0006-3495(76)85767-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Becher B., Tokunaga F., Ebrey T. G. Ultraviolet and visible absorption spectra of the purple membrane protein and the photocycle intermediates. Biochemistry. 1978 Jun 13;17(12):2293–2300. doi: 10.1021/bi00605a006. [DOI] [PubMed] [Google Scholar]
- Birge R. R. Photophysics of light transduction in rhodopsin and bacteriorhodopsin. Annu Rev Biophys Bioeng. 1981;10:315–354. doi: 10.1146/annurev.bb.10.060181.001531. [DOI] [PubMed] [Google Scholar]
- Braiman M., Mathies R. Resonance Raman evidence for an all-trans to 13-cis isomerization in the proton-pumping cycle of bacteriorhodopsin. Biochemistry. 1980 Nov 11;19(23):5421–5428. doi: 10.1021/bi00564a042. [DOI] [PubMed] [Google Scholar]
- Braiman M., Mathies R. Resonance Raman spectra of bacteriorhodopsin's primary photoproduct: evidence for a distorted 13-cis retinal chromophore. Proc Natl Acad Sci U S A. 1982 Jan;79(2):403–407. doi: 10.1073/pnas.79.2.403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dupuis P., Hárosi F. I., Sándorfy C., Leclercq J. M., Vocelle D. First step in vision: proton transfer or isomerization? Rev Can Biol. 1980 Dec;39(4):247–258. [PubMed] [Google Scholar]
- Korenstein R., Hess B. Hydration effects on the photocycle of bacteriorhodopsin in thin layers of purple membrane. Nature. 1977 Nov 10;270(5633):184–186. doi: 10.1038/270184a0. [DOI] [PubMed] [Google Scholar]
- Lazarev Y. A., Terpugov E. L. Effect of water on the structure of bacteriorhodopsin and photochemical processes in purple membranes. Biochim Biophys Acta. 1980 May 9;590(3):324–338. doi: 10.1016/0005-2728(80)90203-0. [DOI] [PubMed] [Google Scholar]
- Lewis A., Spoonhower J., Bogomolni R. A., Lozier R. H., Stoeckenius W. Tunable laser resonance raman spectroscopy of bacteriorhodopsin. Proc Natl Acad Sci U S A. 1974 Nov;71(11):4462–4466. doi: 10.1073/pnas.71.11.4462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marcus M. A., Lewis A. Resonance Raman spectroscopy of the retinylidene chromophore in bacteriorhodopsin (bR570), bR560, M421, and other intermediates: structural conclusions based on kinetics, analogues, models, and isotopically labeled membranes. Biochemistry. 1978 Oct 31;17(22):4722–4735. doi: 10.1021/bi00615a019. [DOI] [PubMed] [Google Scholar]
- Oesterhelt D., Meentzen M., Schuhmann L. Reversible dissociation of the purple complex in bacteriorhodopsin and identification of 13-cis and all-trans-retinal as its chromophores. Eur J Biochem. 1973 Dec 17;40(2):453–463. doi: 10.1111/j.1432-1033.1973.tb03214.x. [DOI] [PubMed] [Google Scholar]
- Oesterhelt D., Schuhmann L., Gruber H. Light-dependent reaction of bacteriorhodopsin with hydroxylamine in cell suspensions of Halobacterium halobium: demonstration of an apo-membrane. FEBS Lett. 1974 Aug 30;44(3):257–261. doi: 10.1016/0014-5793(74)81152-x. [DOI] [PubMed] [Google Scholar]
- Ohno K., Takeuchi Y., Yoshida M. Light-induced formation of the 410 nm intermediate from reconstituted bacteriorhodopsin. J Biochem. 1977 Oct;82(4):1177–1180. doi: 10.1093/oxfordjournals.jbchem.a131792. [DOI] [PubMed] [Google Scholar]
- Pande J., Callender R. H., Ebrey T. G. Resonance Raman study of the primary photochemistry of bacteriorhodopsin. Proc Natl Acad Sci U S A. 1981 Dec;78(12):7379–7382. doi: 10.1073/pnas.78.12.7379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pettei M. J., Yudd A. P., Nakanishi K., Henselman R., Stoeckenius W. Identification of retinal isomers isolated from bacteriorhodopsin. Biochemistry. 1977 May 3;16(9):1955–1959. doi: 10.1021/bi00628a031. [DOI] [PubMed] [Google Scholar]
- Rimai L., Heyde M. E., Gill D. Vibrational spectra of some carotenoids and related linear polyenes. A Raman spectroscopic study. J Am Chem Soc. 1973 Jul 11;95(14):4493–4501. doi: 10.1021/ja00795a005. [DOI] [PubMed] [Google Scholar]
- Rothschild K. J., Clark N. A. Anomalous amide I infrared absorption of purple membrane. Science. 1979 Apr 20;204(4390):311–312. doi: 10.1126/science.432645. [DOI] [PubMed] [Google Scholar]
- Rothschild K. J., Clark N. A. Polarized infrared spectroscopy of oriented purple membrane. Biophys J. 1979 Mar;25(3):473–487. doi: 10.1016/S0006-3495(79)85317-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rothschild K. J., Zagaeski M., Cantore W. A. Conformational changes of bacteriorhodopsin detected by Fourier transform infrared difference spectroscopy. Biochem Biophys Res Commun. 1981 Nov 30;103(2):483–489. doi: 10.1016/0006-291x(81)90478-2. [DOI] [PubMed] [Google Scholar]
- Sperling W., Carl P., Rafferty Ch, Dencher N. A. Photochemistry and dark equilibrium of retinal isomers and bacteriorhodopsin isomers. Biophys Struct Mech. 1977 Jun 29;3(2):79–94. doi: 10.1007/BF00535798. [DOI] [PubMed] [Google Scholar]
- Stockburger M., Klusmann W., Gattermann H., Massig G., Peters R. Photochemical cycle of bacteriorhodopsin studied by resonance Raman spectroscopy. Biochemistry. 1979 Oct 30;18(22):4886–4900. doi: 10.1021/bi00589a017. [DOI] [PubMed] [Google Scholar]
- Stoeckenius W., Bogomolni R. A. Bacteriorhodopsin and related pigments of halobacteria. Annu Rev Biochem. 1982;51:587–616. doi: 10.1146/annurev.bi.51.070182.003103. [DOI] [PubMed] [Google Scholar]
- Stoeckenius W., Lozier R. H., Bogomolni R. A. Bacteriorhodopsin and the purple membrane of halobacteria. Biochim Biophys Acta. 1979 Mar 14;505(3-4):215–278. doi: 10.1016/0304-4173(79)90006-5. [DOI] [PubMed] [Google Scholar]
- Terner J., Campion A., El-Sayed M. A. Time-resolved resonance Raman spectroscopy of bacteriorhodopsin on the millisecond timescale. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5212–5216. doi: 10.1073/pnas.74.12.5212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Terner J., Hsieh C. L., Burns A. R., El-Sayed M. A. Time-resolved resonance Raman spectroscopy of intermediates of bacteriorhodopsin: The bK(590) intermediate. Proc Natl Acad Sci U S A. 1979 Jul;76(7):3046–3050. doi: 10.1073/pnas.76.7.3046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Terner J., Hsieh C. L., El-Sayed M. A. Time-resolved resonance Raman characterization of the bL550 intermediate and the two dark-adapted bRDA/560 forms of bacteriorhodopsin. Biophys J. 1979 Jun;26(3):527–541. doi: 10.1016/S0006-3495(79)85269-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsuda M., Ebrey T. G. Effect of high pressure on the absorption spectrum and isomeric composition of bacteriorhodopsin. Biophys J. 1980 Apr;30(1):149–157. doi: 10.1016/S0006-3495(80)85083-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsuda M., Glaccum M., Nelson B., Ebrey T. G. Light isomerizes the chromophore of bacteriorhodopsin. Nature. 1980 Sep 25;287(5780):351–353. doi: 10.1038/287351a0. [DOI] [PubMed] [Google Scholar]


