Abstract
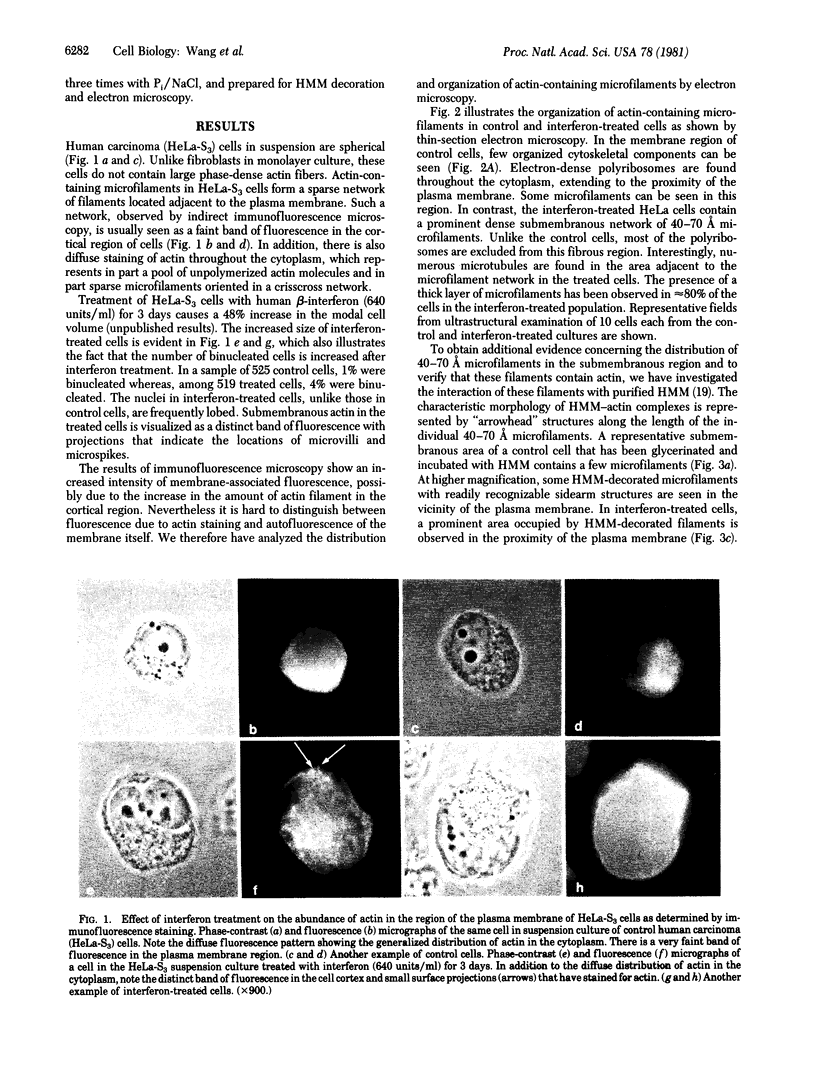
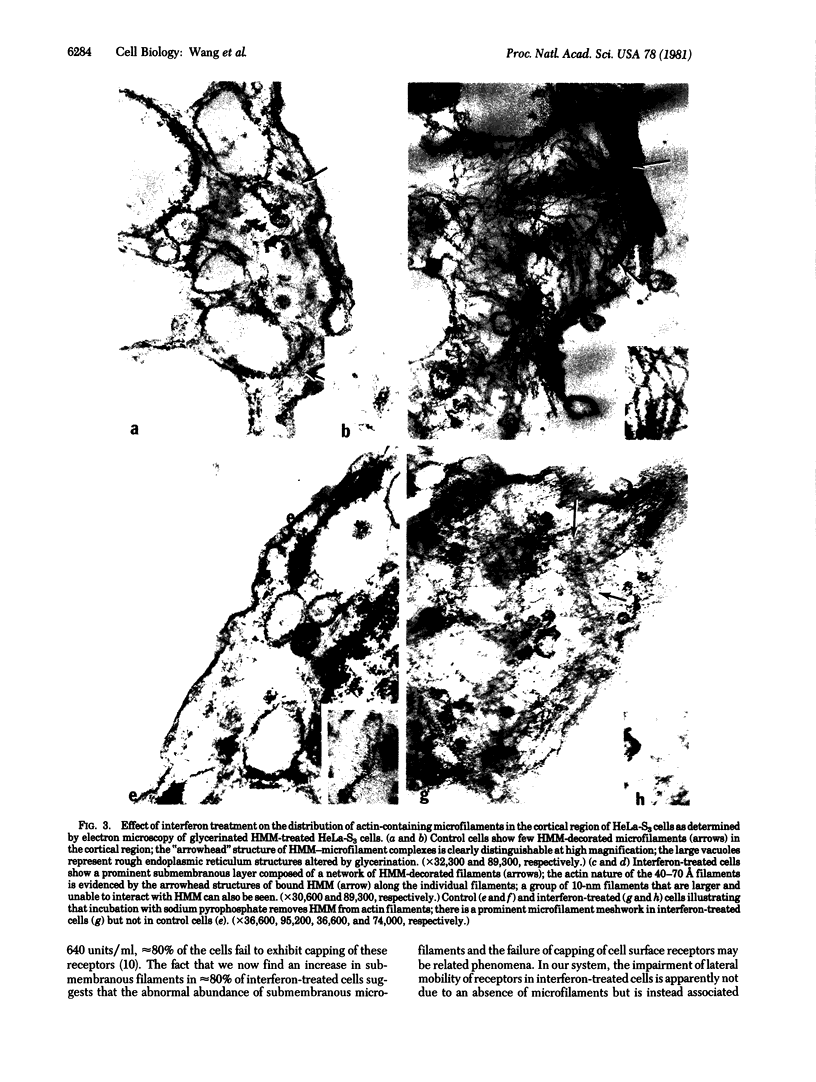
Human beta (fibroblast) interferon inhibits the proliferation of human HeLa-S3 carcinoma cells in suspension culture. Accompanying this effect, the lateral mobility of cell surface receptors for concanavalin A is decreased and the rigidity of the plasma membrane lipid bilayer is increased. The present findings show a marked increase in the number of polymerized actin-containing microfilaments 3 days after treatment of HeLa-S3 cells with beta-interferon (640 units/ml). The cortical region of the treated enlarged cells contains a thick and dense meshwork of 40-70 A microfilaments. The actin nature of the filaments was verified by their ability to bind heavy meromyosin. These results support the concept that beta-interferon induces a coordinated response in the plasma membrane and the underlying microfilaments in both tumor and normal cells.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Chudzio T. S., Inglot A. D. Platelet-derived growth factor (PDGF) inhibits priming in synchronous mouse or human fibroblasts producing interferon. J Interferon Res. 1980 Fall;1(1):31–36. doi: 10.1089/jir.1980.1.31. [DOI] [PubMed] [Google Scholar]
- Edelman G. M. Surface modulation in cell recognition and cell growth. Science. 1976 Apr 16;192(4236):218–226. doi: 10.1126/science.769162. [DOI] [PubMed] [Google Scholar]
- Forer A., Behnke O. An actin-like component in spermatocytes of a crane fly (Nephrotoma suturalis Loew). I. The spindle. Chromosoma. 1972;39(2):145–173. doi: 10.1007/BF00319840. [DOI] [PubMed] [Google Scholar]
- Geiger B. A 130K protein from chicken gizzard: its localization at the termini of microfilament bundles in cultured chicken cells. Cell. 1979 Sep;18(1):193–205. doi: 10.1016/0092-8674(79)90368-4. [DOI] [PubMed] [Google Scholar]
- Geiger B., Tokuyasu K. T., Dutton A. H., Singer S. J. Vinculin, an intracellular protein localized at specialized sites where microfilament bundles terminate at cell membranes. Proc Natl Acad Sci U S A. 1980 Jul;77(7):4127–4131. doi: 10.1073/pnas.77.7.4127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldman R. D., Chang C., Williams J. F. Properties and behavior of hamster embryo cells transformed by human adenovirus type 5. Cold Spring Harb Symp Quant Biol. 1975;39(Pt 1):601–614. doi: 10.1101/sqb.1974.039.01.074. [DOI] [PubMed] [Google Scholar]
- Gresser I. On the varied biologic effects of interferon. Cell Immunol. 1977 Dec;34(2):406–415. doi: 10.1016/0008-8749(77)90262-3. [DOI] [PubMed] [Google Scholar]
- Havell E. A., Vilcek J. Production of high-titered interferon in cultures of human diploid cells. Antimicrob Agents Chemother. 1972 Dec;2(6):476–484. doi: 10.1128/aac.2.6.476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Locke M., Krishnan N. Hot alcoholic phosphotungstic acid and uranyl acetate as routine stains for thick and thin sections. J Cell Biol. 1971 Aug;50(2):550–557. doi: 10.1083/jcb.50.2.550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Norberg R., Thorstensson R., Utter G., Fagraeus A. F-Actin-depolymerizing activity of human serum. Eur J Biochem. 1979 Oct 15;100(2):575–583. doi: 10.1111/j.1432-1033.1979.tb04204.x. [DOI] [PubMed] [Google Scholar]
- Oleszak E., Inglot A. D. Platelet-derived growth factor (PDGF) inhibits antiviral and anticellular actin of interferon in synchronized mouse or human cells. J Interferon Res. 1980 Fall;1(1):37–48. doi: 10.1089/jir.1980.1.37. [DOI] [PubMed] [Google Scholar]
- PAUCKER K., CANTELL K., HENLE W. Quantitative studies on viral interference in suspended L cells. III. Effect of interfering viruses and interferon on the growth rate of cells. Virology. 1962 Jun;17:324–334. doi: 10.1016/0042-6822(62)90123-x. [DOI] [PubMed] [Google Scholar]
- Pfeffer L. M., Murphy J. S., Tamm I. Interferon effects on the growth and division of human fibroblasts. Exp Cell Res. 1979 Jun;121(1):111–120. doi: 10.1016/0014-4827(79)90450-6. [DOI] [PubMed] [Google Scholar]
- Pfeffer L. M., Wang E., Tamm I. Interferon effects on microfilament organization, cellular fibronectin distribution, and cell motility in human fibroblasts. J Cell Biol. 1980 Apr;85(1):9–17. doi: 10.1083/jcb.85.1.9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pfeffer L. M., Wang E., Tamm I. Interferon inhibits the redistribution of cell surface components. J Exp Med. 1980 Aug 1;152(2):469–474. doi: 10.1084/jem.152.2.469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pollard T. D., Weihing R. R. Actin and myosin and cell movement. CRC Crit Rev Biochem. 1974 Jan;2(1):1–65. doi: 10.3109/10409237409105443. [DOI] [PubMed] [Google Scholar]
- SZENT-GYORGYI A. G. Meromyosins, the subunits of myosin. Arch Biochem Biophys. 1953 Feb;42(2):305–320. doi: 10.1016/0003-9861(53)90360-9. [DOI] [PubMed] [Google Scholar]
- Singer S. J., Ash J. F., Bourguignon L. Y., Heggeness M. H., Louvard D. Transmembrane interactions and the mechanisms of transport of proteins across membranes. J Supramol Struct. 1978;9(3):373–389. doi: 10.1002/jss.400090308. [DOI] [PubMed] [Google Scholar]
- Unanue E. R., Perkins W. D., Karnovsky M. J. Ligand-induced movement of lymphocyte membrane macromolecules. I. Analysis by immunofluorescence and ultrastructural radioautography. J Exp Med. 1972 Oct 1;136(4):885–906. doi: 10.1084/jem.136.4.885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang E., Goldberg A. R. Binding of deoxyribonuclease I to actin: a new way to visualize microfilament bundles in nonmuscle cells. J Histochem Cytochem. 1978 Sep;26(9):745–749. doi: 10.1177/26.9.361884. [DOI] [PubMed] [Google Scholar]
- Wessells N. K., Spooner B. S., Ash J. F., Bradley M. O., Luduena M. A., Taylor E. L., Wrenn J. T., Yamada K. Microfilaments in cellular and developmental processes. Science. 1971 Jan 15;171(3967):135–143. doi: 10.1126/science.171.3967.135. [DOI] [PubMed] [Google Scholar]
- Willingham M. C., Yamada K. M., Yamada S. S., Pouysségur J., Pastan I. Microfilament bundles and cell shape are related to adhesiveness to substratum and are dissociable from growth control in cultured fibroblasts. Cell. 1977 Mar;10(3):375–380. doi: 10.1016/0092-8674(77)90024-1. [DOI] [PubMed] [Google Scholar]
- Yahara I., Edelman G. M. The effects of concanavalin A on the mobility of lymphocyte surface receptors. Exp Cell Res. 1973 Sep;81(1):143–155. doi: 10.1016/0014-4827(73)90121-3. [DOI] [PubMed] [Google Scholar]
- Yin H. L., Stossel T. P. Purification and structural properties of gelsolin, a Ca2+-activated regulatory protein of macrophages. J Biol Chem. 1980 Oct 10;255(19):9490–9493. [PubMed] [Google Scholar]
- Yin H. L., Zaner K. S., Stossel T. P. Ca2+ control of actin gelation. Interaction of gelsolin with actin filaments and regulation of actin gelation. J Biol Chem. 1980 Oct 10;255(19):9494–9500. [PubMed] [Google Scholar]
- de Petris S. Concanavalin A receptors, immunoglobulins, and theta antigen of the lymphocyte surface. Interactions with concanavalin A and with Cytoplasmic structures. J Cell Biol. 1975 Apr;65(1):123–146. doi: 10.1083/jcb.65.1.123. [DOI] [PMC free article] [PubMed] [Google Scholar]