Abstract
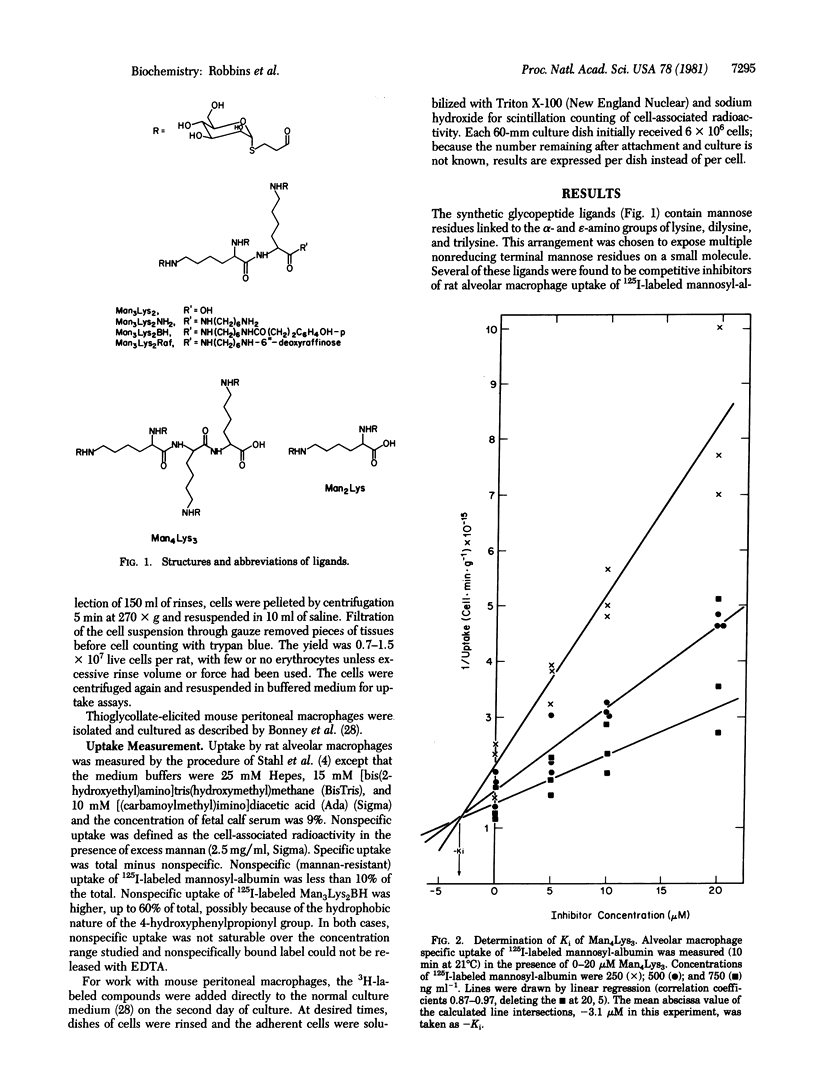
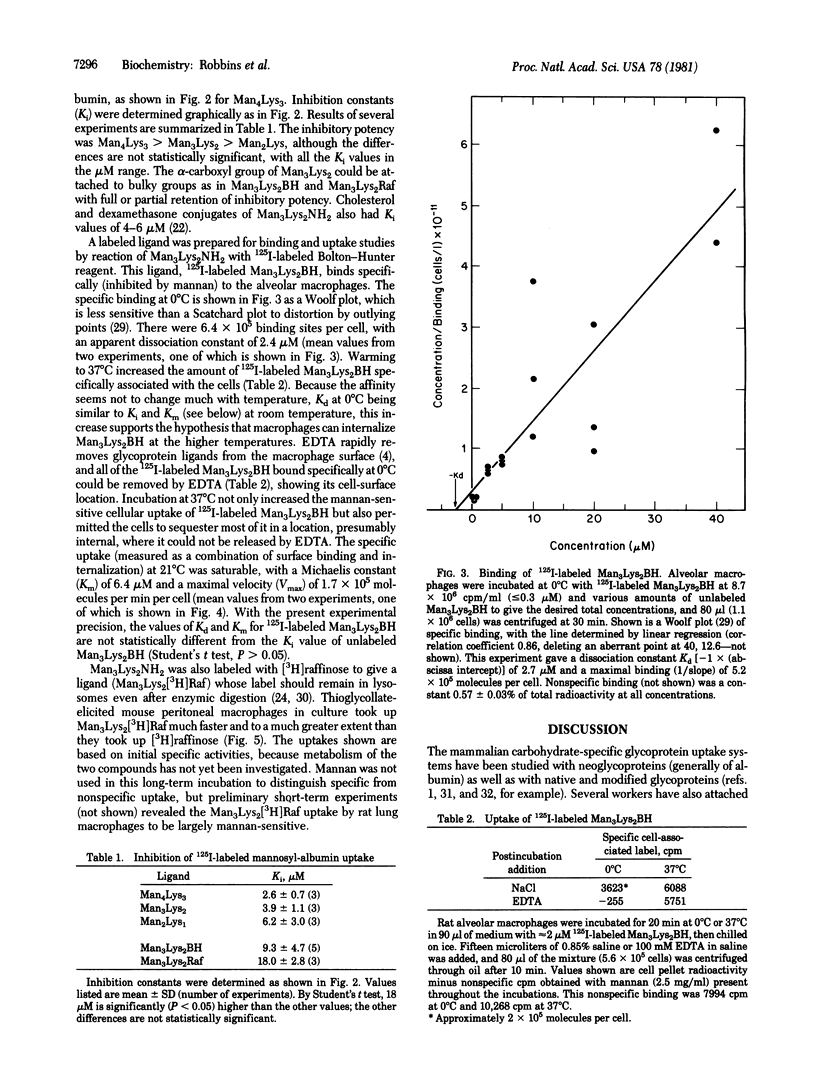
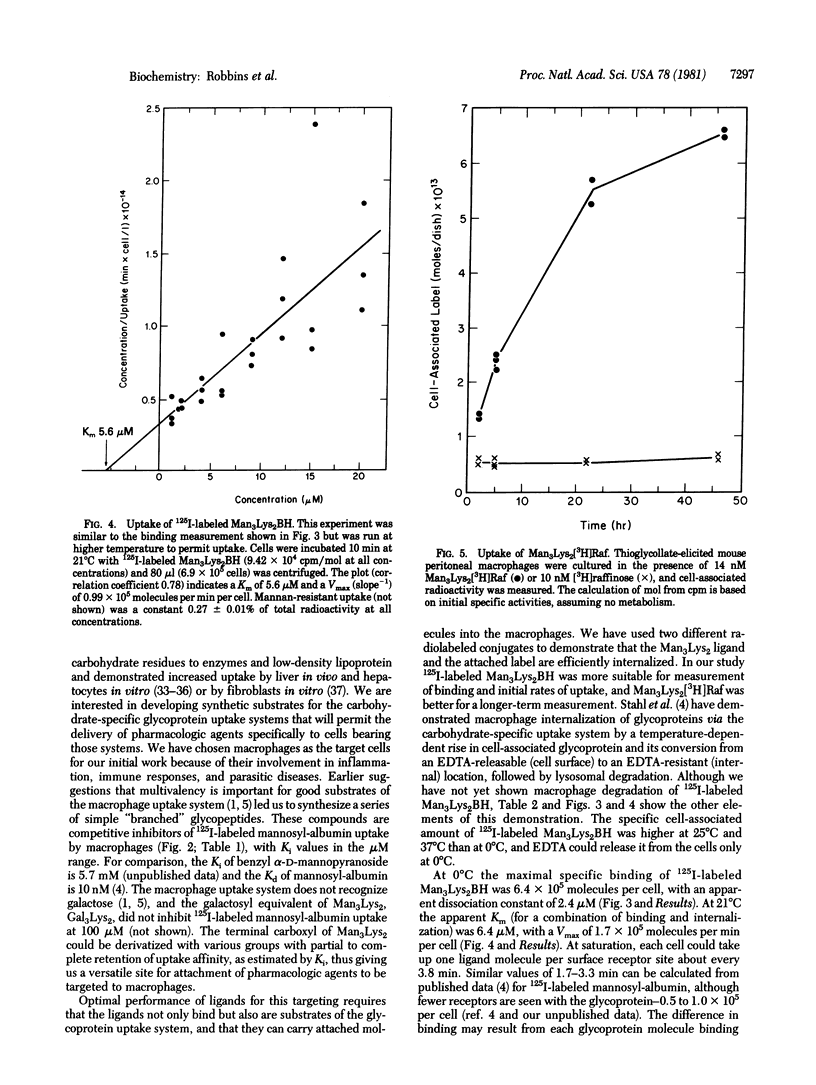
Mammalian macrophages contain a transport system that binds and internalizes glycoproteins with exposed mannose residues. This system and analogous systems on other types of cells require substrates to bear multiple nonreducing terminal residues of the appropriate sugar for effective uptake. Small multivalent synthetic glycopeptides with mannose residues covalently linked through a spacer arm to the alpha- and epsilon-amino groups of lysine, dilysine, and trilysine are competitive inhibitors of rat alveolar macrophage uptake of the neoglycoprotein mannosyl-bovine serum albumin with inhibition constants in the microM range. Various compounds could be covalently attached to the alpha-carboxyl group of these glycopeptides with substantial retention of inhibitory potency. This uptake system does not recognize galactose residues, and the galactosyl analog of an inhibitory mannosylpeptide did not inhibit uptake of mannosyl-bovine serum albumin. The trimannosyldilysine ligand is not only an inhibitor but also a substrate for specific uptake by macrophage, as shown with an 125I-labeled derivative. Macrophages bound 6.4 x 10(5) molecules per cell at 0 degrees C with a dissociation constant of 2 microM. At 21 degrees C the cells could internalize the labeled conjugate with an apparent Michaelis constant of 6 microM and a maximal velocity of 1.7 x 10(5) molecules per min per cel. The dissociation constant and Michaelis constant are similar to the inhibition constant of 9 microM determined at 21 degrees C for inhibition by this conjugate at mannosyl-bovine serum albumin uptake. These synthetic substrates may be useful in targeting pharmacologic agents to macrophages, and analogous compounds may target such agents to other types of cell.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Achord D. T., Brot F. E., Bell C. E., Sly W. S. Human beta-glucuronidase: in vivo clearance and in vitro uptake by a glycoprotein recognition system on reticuloendothelial cells. Cell. 1978 Sep;15(1):269–278. doi: 10.1016/0092-8674(78)90102-2. [DOI] [PubMed] [Google Scholar]
- Attie A. D., Pittman R. C., Steinberg D. Metabolism of native and of lactosylated human low density lipoprotein: evidence for two pathways for catabolism of exogenous proteins in rat hepatocytes. Proc Natl Acad Sci U S A. 1980 Oct;77(10):5923–5927. doi: 10.1073/pnas.77.10.5923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baenziger J. U., Fiete D. Galactose and N-acetylgalactosamine-specific endocytosis of glycopeptides by isolated rat hepatocytes. Cell. 1980 Nov;22(2 Pt 2):611–620. doi: 10.1016/0092-8674(80)90371-2. [DOI] [PubMed] [Google Scholar]
- Bolton A. E., Hunter W. M. The labelling of proteins to high specific radioactivities by conjugation to a 125I-containing acylating agent. Biochem J. 1973 Jul;133(3):529–539. doi: 10.1042/bj1330529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bonney R. J., Wightman P. D., Davies P., Sadowski S. J., Kuehl F. A., Jr, Humes J. L. Regulation of prostaglandin synthesis and of the selective release of lysosomal hydrolases by mouse peritoneal macrophages. Biochem J. 1978 Nov 15;176(2):433–442. doi: 10.1042/bj1760433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brain J. D., Frank R. Alveolar macrophage adhesion: wash electrolyte composition and free cell yield. J Appl Physiol. 1973 Jan;34(1):75–80. doi: 10.1152/jappl.1973.34.1.75. [DOI] [PubMed] [Google Scholar]
- Brown T. L., Henderson L. A., Thorpe S. R., Baynes J. W. The effect of alpha-mannose-terminal oligosaccharides on the survival of glycoproteins in the circulation. Rapid uptake and catabolism of bovine pancreatic ribonuclease B by nonparenchymal cells of rat liver. Arch Biochem Biophys. 1978 Jun;188(2):418–428. doi: 10.1016/s0003-9861(78)80026-5. [DOI] [PubMed] [Google Scholar]
- Day J. F., Thornburg R. W., Thorpe S. R., Baynes J. W. Carbohydrate-mediated clearance of antibody . antigen complexes from the circulation. The role of high mannose oligosaccharides in the hepatic uptake of IgM . antigen complexes. J Biol Chem. 1980 Mar 25;255(6):2360–2365. [PubMed] [Google Scholar]
- GREENWOOD F. C., HUNTER W. M., GLOVER J. S. THE PREPARATION OF I-131-LABELLED HUMAN GROWTH HORMONE OF HIGH SPECIFIC RADIOACTIVITY. Biochem J. 1963 Oct;89:114–123. doi: 10.1042/bj0890114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hubbard A. L., Wilson G., Ashwell G., Stukenbrok H. An electron microscope autoradiographic study of the carbohydrate recognition systems in rat liver. I. Distribution of 125I-ligands among the liver cell types. J Cell Biol. 1979 Oct;83(1):47–64. doi: 10.1083/jcb.83.1.47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaplan A., Achord D. T., Sly W. S. Phosphohexosyl components of a lysosomal enzyme are recognized by pinocytosis receptors on human fibroblasts. Proc Natl Acad Sci U S A. 1977 May;74(5):2026–2030. doi: 10.1073/pnas.74.5.2026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karson E. M., Neufeld E. F., Sando G. N. p-Isothiocyanatophenyl 6-phospho-alpha-D-mannopyranoside coupled to albumin. A model compound recognized by the fibroblast lysosomal enzyme uptake system. 2. Biological properties. Biochemistry. 1980 Aug 5;19(16):3856–3860. doi: 10.1021/bi00557a033. [DOI] [PubMed] [Google Scholar]
- Kawasaki T., Etoh R., Yamashina I. Isolation and characterization of a mannan-binding protein from rabbit liver. Biochem Biophys Res Commun. 1978 Apr 14;81(3):1018–1024. doi: 10.1016/0006-291x(78)91452-3. [DOI] [PubMed] [Google Scholar]
- Kawasaki T., Mizuno Y., Masuda T., Yamashina I. Mannan-binding protein in lymphoid tissues of rats. J Biochem. 1980 Dec;88(6):1891–1894. [PubMed] [Google Scholar]
- Keightley D. D., Cressie N. A. The Woolf plot is more reliable than the Scatchard plot in analysing data from hormone receptor assays. J Steroid Biochem. 1980 Nov;13(11):1317–1323. doi: 10.1016/0022-4731(80)90092-8. [DOI] [PubMed] [Google Scholar]
- Kozutsumi Y., Kawasaki T., Yamashina I. Isolation and characterization of a mannan-binding protein from rabbit serum. Biochem Biophys Res Commun. 1980 Jul 31;95(2):658–664. doi: 10.1016/0006-291x(80)90836-0. [DOI] [PubMed] [Google Scholar]
- Krantz M. J., Holtzman N. A., Stowell C. P., Lee Y. C. Attachment of thioglycosides to proteins: enhancement of liver membrane binding. Biochemistry. 1976 Sep 7;15(18):3963–3968. doi: 10.1021/bi00663a009. [DOI] [PubMed] [Google Scholar]
- Lee Y. C., Stowell C. P., Krantz M. J. 2-Imino-2-methoxyethyl 1-thioglycosides: new reagents for attaching sugars to proteins. Biochemistry. 1976 Sep 7;15(18):3956–3963. doi: 10.1021/bi00663a008. [DOI] [PubMed] [Google Scholar]
- Lunney J., Ashwell G. A hepatic receptor of avian origin capable of binding specifically modified glycoproteins. Proc Natl Acad Sci U S A. 1976 Feb;73(2):341–343. doi: 10.1073/pnas.73.2.341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marsh J. W., Denis J., Wriston J. C., Jr Glycosylation of Escherichia coli L-asparaginase. J Biol Chem. 1977 Nov 10;252(21):7678–7684. [PubMed] [Google Scholar]
- Maynard Y., Baenziger J. U. Oligosaccharide specific endocytosis by isolated rat hepatic reticuloendothelial cells. J Biol Chem. 1981 Aug 10;256(15):8063–8068. [PubMed] [Google Scholar]
- Morell A. G., Irvine R. A., Sternlieb I., Scheinberg I. H., Ashwell G. Physical and chemical studies on ceruloplasmin. V. Metabolic studies on sialic acid-free ceruloplasmin in vivo. J Biol Chem. 1968 Jan 10;243(1):155–159. [PubMed] [Google Scholar]
- Murray G. J., Neville D. M., Jr Mannose 6-phosphate receptor-mediated uptake of modified low density lipoprotein results in down regulation of hydroxymethylglutaryl-CoA reductase in normal and familial hypercholesterolemic fibroblasts. J Biol Chem. 1980 Dec 25;255(24):11942–11948. [PubMed] [Google Scholar]
- Pittman R. C., Attie A. D., Carew T. E., Steinberg D. Tissue sites of degradation of low density lipoprotein: application of a method for determining the fate of plasma proteins. Proc Natl Acad Sci U S A. 1979 Oct;76(10):5345–5349. doi: 10.1073/pnas.76.10.5345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prieels J. P., Pizzo S. V., Glasgow L. R., Paulson J. C., Hill R. L. Hepatic receptor that specifically binds oligosaccharides containing fucosyl alpha1 leads to 3 N-acetylglucosamine linkages. Proc Natl Acad Sci U S A. 1978 May;75(5):2215–2219. doi: 10.1073/pnas.75.5.2215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rogers J. C., Kornfeld S. Hepatic uptake of proteins coupled to fetuin glycopeptide. Biochem Biophys Res Commun. 1971 Nov 5;45(3):622–629. doi: 10.1016/0006-291x(71)90462-1. [DOI] [PubMed] [Google Scholar]
- Sando G. N., Neufeld E. F. Recognition and receptor-mediated uptake of a lysosomal enzyme, alpha-l-iduronidase, by cultured human fibroblasts. Cell. 1977 Nov;12(3):619–627. doi: 10.1016/0092-8674(77)90262-8. [DOI] [PubMed] [Google Scholar]
- Schlesinger P. H., Doebber T. W., Mandell B. F., White R., DeSchryver C., Rodman J. S., Miller M. J., Stahl P. Plasma clearance of glycoproteins with terminal mannose and N-acetylglucosamine by liver non-parenchymal cells. Studies with beta-glucuronidase, N-acetyl-beta-D-glucosaminidase, ribonuclease B and agalacto-orosomucoid. Biochem J. 1978 Oct 15;176(1):103–109. doi: 10.1042/bj1760103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shepherd V. L., Lee Y. C., Schlesinger P. H., Stahl P. D. L-Fucose-terminated glycoconjugates are recognized by pinocytosis receptors on macrophages. Proc Natl Acad Sci U S A. 1981 Feb;78(2):1019–1022. doi: 10.1073/pnas.78.2.1019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stahl P. D., Rodman J. S., Miller M. J., Schlesinger P. H. Evidence for receptor-mediated binding of glycoproteins, glycoconjugates, and lysosomal glycosidases by alveolar macrophages. Proc Natl Acad Sci U S A. 1978 Mar;75(3):1399–1403. doi: 10.1073/pnas.75.3.1399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stahl P., Schlesinger P. H., Sigardson E., Rodman J. S., Lee Y. C. Receptor-mediated pinocytosis of mannose glycoconjugates by macrophages: characterization and evidence for receptor recycling. Cell. 1980 Jan;19(1):207–215. doi: 10.1016/0092-8674(80)90402-x. [DOI] [PubMed] [Google Scholar]
- Steer C. J., Ashwell G. Studies on a mammalian hepatic binding protein specific for asialoglycoproteins. Evidence for receptor recycling in isolated rat hepatocytes. J Biol Chem. 1980 Apr 10;255(7):3008–3013. [PubMed] [Google Scholar]
- Steer C. J., Kusiak J. W., Brady R. O., Jones E. A. Selective hepatic uptake of human beta-hexosaminidase A by a specific glycoprotein recognition system on sinusoidal cells. Proc Natl Acad Sci U S A. 1979 Jun;76(6):2774–2778. doi: 10.1073/pnas.76.6.2774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Townsend R., Stahl P. Isolation and characterization of a mannose/N-acetylglucosamine/fucose-binding protein from rat liver. Biochem J. 1981 Jan 15;194(1):209–214. doi: 10.1042/bj1940209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Zile J., Henderson L. A., Baynes J. W., Thorpe S. R. [3H]Raffinose, a novel radioactive label for determining organ sites of catabolism of proteins in the circulation. J Biol Chem. 1979 May 10;254(9):3547–3553. [PubMed] [Google Scholar]
- Warr G. A. A macrophage receptor for (mannose/glucosamine)-glycoproteins of potential importance in phagocytic activity. Biochem Biophys Res Commun. 1980 Apr 14;93(3):737–745. doi: 10.1016/0006-291x(80)91139-0. [DOI] [PubMed] [Google Scholar]
- Wilson G. Effect of reductive lactosamination on the hepatic uptake of bovine pancreatic ribonuclease A dimer. J Biol Chem. 1978 Apr 10;253(7):2070–2072. [PubMed] [Google Scholar]


