Abstract
Activation of the cellular DNA damage response (DDR) is an important determinant of cell sensitivity to cisplatin and other chemotherapeutic drugs that eliminate tumor cells through induction of DNA damage. It is therefore important to investigate whether alterations of the DNA damage signaling pathway confer chemoresistance in cancer cells, and whether pharmacological manipulation of the DDR pathway can re-sensitize these cells to cancer therapy. In a panel of oral/laryngeal squamous cell carcinoma (SCC) cell lines, we observed deficiencies in DNA damage signaling in correlation with cisplatin-resistance, but not with DNA repair. These deficiencies are consistent with reduced expression of components of the ATM-dependent signaling pathway and, in particular, strong up-regulation of Wip1, a negative regulator of the ATM pathway. Wip1 knockdown or inhibition enhanced DNA damage signaling and re-sensitized oral SCC cells to cisplatin. In contrast to the previously reported involvement of Wip1 in cancer, Wip1 up-regulation and function in these SCC cells is independent of p53. Finally, using xenograft tumor models, we demonstrated that Wip1 up-regulation promotes tumorigenesis and its inhibition improves the tumor response to cisplatin. Thus, this study reveals that chemoresistance in oral SCCs is partially attributed to deficiencies in DNA damage signaling, and Wip1 is an effective drug target for enhanced cancer therapy.
Keywords: DNA damage signaling, chemotherapy, cisplatin, ATM, Wip1
Introduction
Radiation and most chemotherapeutic drugs exert their toxicity to tumor cells primarily through induction of DNA damage, which halts cell proliferation and induces cell death. One such genotoxic drug commonly used for chemotherapy is cisplatin, or cis-diamminedichloroplatinum(II). Cisplatin was first approved by the U.S. Food and Drug Administration in 1978 for the use in testicular and ovarian cancers. Over decades, cisplatin and other platinum-based analogs have become important weapons battling against a wide variety of cancers, including testicular, breast, ovarian, lung and oral cancers. Cisplatin delivers initial success with partial responses and disease stabilization in many patients, but unfortunately, its clinical usefulness is ultimately limited by the fact that a majority of cancer patients will eventually develop resistance to cisplatin, leading to tumor recurrence (1, 2). Upon cellular absorption and processing, cisplatin binds DNA and causes DNA strand cross-linking. This form of DNA lesion is then sensed by a cellular surveillance mechanism, generally termed “the DNA damage response (DDR)”, which ultimately determines cell fate through coordinated activation of various downstream pathways. It has been therefore widely anticipated that future delineation of the DDR pathway holds a key to better understanding of chemoresistance and development of more efficacious cancer treatments (3).
As a conserved mechanism evolved by eukaryotic cells to govern genomic integrity, the DDR encompasses various lesion-specific DNA repair pathways and a sophisticated signaling network. DNA damage signaling subsequently activates either the DNA damage checkpoint to arrest cell cycle progression, or apoptosis to eliminate the damaged cell. At the center of DNA damage signaling are the phosphoinositide 3-kinase-related kinases (PIKK) ATM and ATR. Activation of ATM/ATR by DNA damage in turn results in phosphorylation of dozens of physiological substrates that control various pathways of DNA repair, checkpoint control, apoptosis and transcription (4). For example, ATM and ATR activate the checkpoint kinases, Chk1 and Chk2, which then phosphorylate and inactivate Cdc25, a regulator of cyclin-dependent kinases (Cdks) required for Cdk activation and cell cycle progression. Moreover, ATM and ATR phosphorylate a histone H2A variant, H2AX, at the site of DNA damage. Phosphorylated H2AX (γ-H2AX) then recruits a number of DDR factors onto damaged chromatin to facilitate DNA repair and checkpoint signaling. Despite functional similarities, ATM and ATR are activated through distinct mechanisms. ATM activation is promoted by the heterotrimeric complex Mre11/Rad50/Nbs1 that may directly recognize DNA double strand breaks (5, 6), whereas ATR is activated through binding to RPA-coated ssDNA and TopBP1 (7, 8). Both ATM and ATR have been implicated in the cellular response to cisplatin (9–11).
As DDR signaling relies largely on kinase cascades and protein phosphorylation, it is not surprising that recent studies started to reveal important involvement of Ser/Thr phosphatases in this pathway (12). One of the best-studied examples is the wild-type p53-induced phosphatase 1 (Wip1). It has been shown that expression of Wip1 is induced after DNA damage in a p53-dependent manner. Upon induction, Wip1 functions as a homeostatic regulator of the DDR by dephosphorylating multiple S/TQ sites of ATM, Chk1, Chk2, p53, γ-H2AX that are targeted by ATM/ATR. Wip1-dependent dephosphorylation leads to deactivation of these DDR factors, thus allowing cells to turn off the DDR signaling pathway and resume cell proliferation (reviewed in (12, 13)). Interestingly, Wip1 has been shown to be amplified or overexpressed in a number of human cancers, including breast cancer, neuroblastoma and ovarian cancer. The oncogenic role of Wip1 has been well-demonstrated both in vitro and in vivo, particularly for breast cancer. Overexpression of Wip1 promotes cell growth and transformation induced by Ras or other oncogenes, whereas Wip1 depletion suppresses mammary tumorigenesis (reviewed in (13, 14)).
The ultimate outcome of the DDR could be either cell survival or death—if the cell successfully repairs DNA damage, it would turn off the DDR and return to cell proliferation, whereas failure of DNA repair leads to programmed cell death. Under normal circumstances, such a well-balanced mechanism ensures genomic integrity, yet at the same time, avoids excessive cell loss. However, in tumor cells treated with radio- or chemotherapy, DNA repair and cell survival attenuate the killing effect of the treatment and lead to undesired therapeutic outcomes. Cisplatin-induced DNA damage is repaired by nucleotide-excision repair (NER) and other repair pathways, and up-regulation of genes involved in these pathways, including ERCC1, POLH, BRCA1 and BRCA2, has been shown to render tumor cells resistant to cisplatin treatment (1).
Notably, the relationship between the DNA damage checkpoint pathway and tumor-resistance to cisplatin is less understood, and perhaps also more complicated in nature. On one hand, checkpoint activation allows time for DNA repair and may thereby contribute to cell survival, but on the other hand, the same signaling pathway may induce cell death or senescence to confer a better treatment outcome. In this study, we sought to investigate the potential connection between the DDR signaling pathway and cisplatin response in a panel of well-characterized oral/laryngeal squamous cell carcinoma (SCC) cell lines (15). We observed deficient DNA damage signaling in some SCC cell lines, resulting from both reduced expression of genes in the ATM-dependent checkpoint pathway, and up-regulation of Wip1, the negative regulator of DNA damage signaling. The deficiency in DNA damage signaling is correlated with chemoresistance to cisplatin, but not with DNA repair. Importantly, knockdown or inhibition of Wip1 re-sensitizes the cisplatin-resistant cells in vitro and in vivo, suggesting exciting opportunities for combinatorial therapy.
Materials and Methods
Cell culture
Human oral/laryngeal squamous-cell carcinoma cell lines were obtained from Dr. Thomas Carey (University of Michigan) in 2010 and 2011. These cell lines were previously characterized genetically and morphologically (15, 16), and have not been re-tested and authenticated in the present study. Cells were maintained in Dulbecco’s modified Eagle medium (DMEM, Sigma, St Louis, MO) supplemented with 10% fetal bovine serum (FBS, Sigma). To measure cell sensitivity to cisplatin, cells were treated with cisplatin at indicated concentrations, and incubated for 1–4 days. The numbers of viable cells were counted using a hemocytometer. Lentiviral vectors expressing control non-targeting or Wip1 shRNAs were purchased from Sigma and used to infect cells following the protocol recommended by the manufacturer.
Immunoblotting
Immunoblotting was performed as described previously (17, 18). Anti-ATM, Mre11, H2AX, γ-H2AX, Chk1, phospho-Chk2, phospho-ATR (Ser-428), p53 antibodies were purchased from Cell Signaling (Danvers, MA); anti-β-Actin antibody was obtained from Abcam (Cambridge, MA); anti-Smc1, Wip1, and phospho-Smc1 antibodies were purchased from Bethyl (Montgomery, TX). The intensity of band signals was measured using NIH Image-J software.
Comet assay
A modified comet assay was performed to measure DNA crosslink induced by cisplatin (19). Exponentially growing cells were treated with 16.7 µM cisplatin in DMEM medium for 2h at 37°C. Cells were washed with Phosphate buffered saline buffer and the medium replaced with fresh DMEM and incubated for 24h to allow repair. Cells were treated with H2O2 (250 µM) for 15 min before collection to expose DNA crosslinks. Cells were then trypsinized, washed, and plated in 0.65% low melting agarose. The agarose was allowed to solidify, and slides were incubated in lysis solution (2.5M NaCl, 100mM EDTA, 10mM Trizma base, pH 10.0) containing 1% Triton X-100 at 4°C for 2h. Slides were then washed with ice-cold water, and transferred to an electrophoresis tank containing room-temperature alkaline solution (50mM NaOH, 1mM disodium EDTA, pH >12.3) for 30 min incubation. Electrophoresis was carried out for 10 min at 1 V/cm. The slides were removed and incubated in neutralizing solution (0.4M Tris-HCl, pH 7.5) for 10 min. Slides were stained with propidum iodide (25 µg/ml) and washed 5 min in ice-cold water to remove excess stain. Images were captured at 10× magnification using a Carl Zeiss Axiovert 40 CFL inverted microscope equipped with a mercury lamp. Fifty cells per sample were analyzed for the level of DNA crosslink by comparing the tail moment of the cisplatin/H2O2 treated cells to the untreated samples. Percent decrease in tail moment (%DTM) was calculated with the formula , where TMc is the mean tail moment of the untreated control, TMh is the mean tail moment of the H2O2 control (representing no crosslink), and TMdh is the mean tail moment of the drug/H2O2 treated sample.
Xenograft tumor model
Athymic nude mice were purchased from NIH and housed at the animal facility at the UNMC College of Dentistry. SCC cells were implanted into 6-week old female mice by a single subcutaneous injection of tumor cells (2 – 6 × 105 cells in 100 microliters of sterile PBS). The growth rates of tumors were determined by daily monitoring of tumor volume with vernier calipers (tumor volume = 1/2(length × width2)). Upon completion of the study, the mice were euthanized, and their tumors were removed and weighed immediately. To test how tumors respond to chemotherapy, once the tumor size reached 50 mm3, cisplatin (5 mg/kg mouse), arsenic trioxide (10 mg/kg), and CCT007093 (2 mg/kg) were administered intraperitoneally every day for 5 days. Tumor size was monitored daily. Ten days after the initial treatment, the mice were euthanized, and tumors were removed and weighed. The volume of the tumor was compared among all experimental groups. Data were analyzed using an unpaired 2-tailed Student’s t test to determine the statistical significance.
Results
Altered DNA damage signaling in oral SCC cells
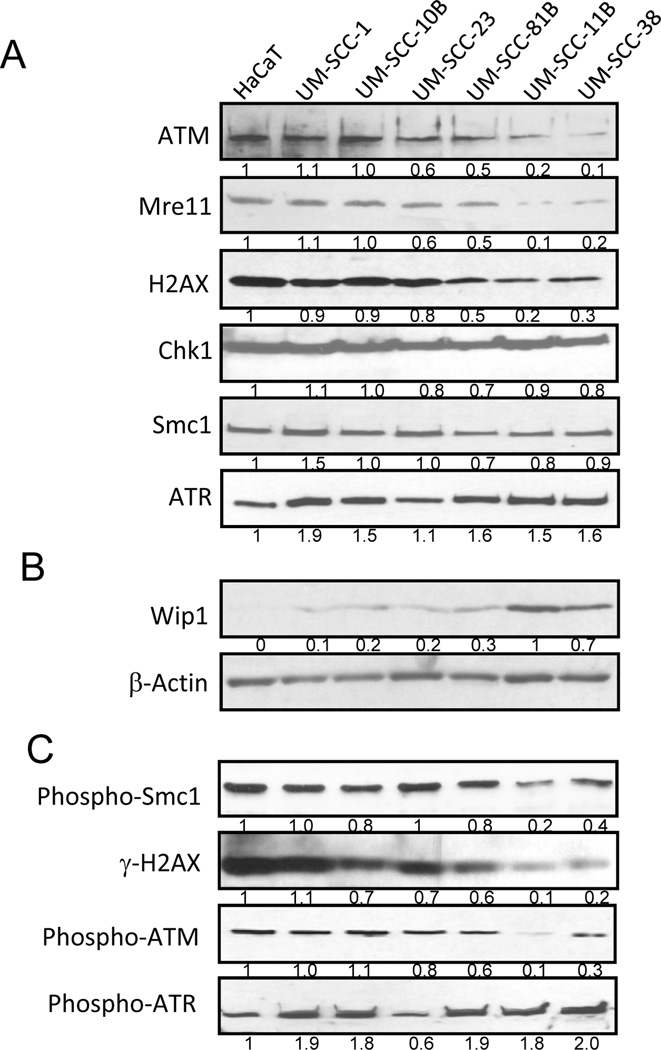
Activation of the DDR by cisplatin and other chemotherapeutic drugs involves complex signaling pathways composed of numerous proteins, such as ATM, Mre11, Chk1, and Smc1. We compared gene expression of multiple DDR factors in a panel of oral SCC cell lines in comparison to a non-tumorigenic control keratinocyte cell line HaCaT, and observed reduced expression of ATM, Mre11, and H2AX in UM-SCC-11B and -38 cells (Fig. 1A). By comparison, other DDR factors, including Chk1, Smc1, and ATR, are expressed to comparable levels in all cell lines (Fig. 1A). We further examined the expression of a negative regulator of the DNA damage checkpoint, Wip1, which has been shown to dephosphorylate and deactivate ATM, H2AX and other DDR factors. As shown in Fig. 1B, we detected strong up-regulation of Wip1 in UM-SCC-11B and -38 cells. Consistent with the reduced expression of ATM, Mre11, H2AX and enhanced expression of Wip1, activation of DNA damage signaling in UM-SCC-11B and -38 cells is significantly impaired in response to cisplatin, as judged by reduced phosphorylation of Smc1, H2AX, and ATM (Fig. 1C). In contrast, ATR is efficiently phosphorylated in SCC-11B and -38 cells (Fig. 1C).
Figure 1.
Deficient DNA damage signaling in oral cancer cells. (A, B) A panel of oral/laryngeal squamous cell carcinoma (SCC) cell lines and a control non-tumorigenic HaCaT cell line were maintained as described in Materials and Methods. These cells were examined by immunoblotting and compared for expression of ATM, Mre11, H2AX, Chk1, Smc1 and ATR (A), Wip1 and β-Actin (B). The intensity of band signals was measured using ImageJ software, and normalized to β-Actin. (C) HaCaT and SCC cells were treated with cisplatin (16.7 µM) for 4 hours, and harvested for immunoblotting using phospho-Smc1, ATM, ATR, and γ-H2AX antibodies.
Deficient DDR correlates with cisplatin-resistance but not with DNA repair
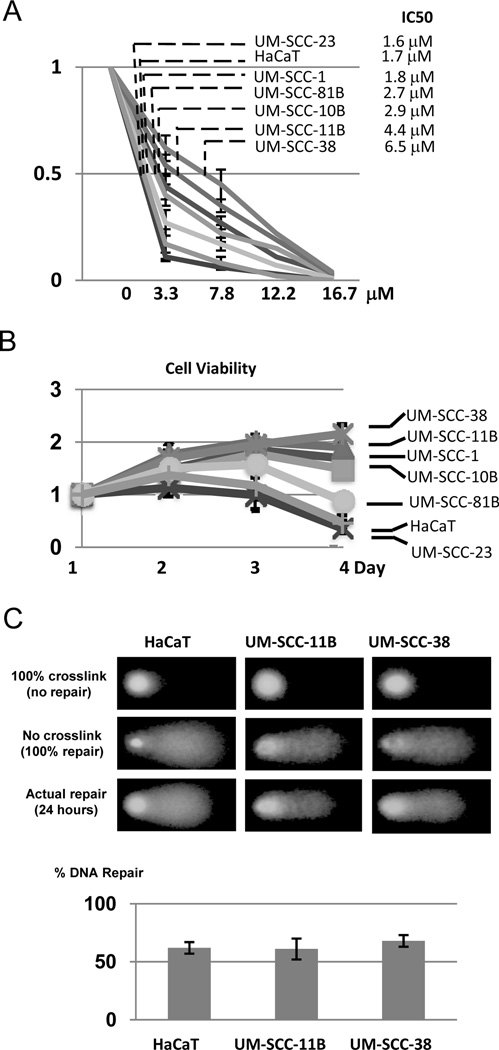
Interestingly, UM-SCC-11B and -38 cells that harbor impaired DDR signaling are more resistant to cisplatin. As shown in Fig. 2A & 2B, HaCaT and oral SCC cells exhibit varying sensitivities to cisplatin. UM-SCC-23 and -81B cells are generally as sensitive to cisplatin as the control HaCaT cells; UM-SCC-1 and -10B cells are considerably more resistant than HaCaT cells; whereas UM-SCC-11B and -38 cells are the most resistant to cisplatin. We then sought to determine the capacity of DNA repair in UM-SCC-11B and -38 cells. As shown in previous studies, DNA strand crosslink induced by cisplatin can be measured using the comet assay (19). Surprisingly, no repair deficiency was found in UM-SCC-11B and -38 cells when compared to the control HaCaT cell (Fig. 2D).
Figure 2.
Cisplatin-resistance and DNA repair capacity in oral cancer cells. (A) SCC and HaCaT cells were incubated with cisplatin at various concentrations for 4 days. Cell viability was determined as described in Materials and Methods, and normalized to that without cisplatin treatment. The concentration of cisplatin that inhibits 50% cell growth (IC50) was calculated for each cell line. (B) SCC and HaCaT cells were incubated with cisplatin at 3.3 µM and incubated for 1–4 days. Cell viability was determined and normalized to that of the first day. (C) HaCaT, UM-SCC-11B, and UM-SCC-38 cells were treated with cisplatin (16.7 µM) for 2 hours to induce DNA crosslink and then incubated for 24 hours for repair. The DNA repair capacity in these cells was assessed using a modified comet assay as described in Materials and Methods. Representative images of the comet assay are shown. The graph is based on three independent experiments with at least 50 cells per sample in each experiment.
P53-independent up-regulation of Wip1
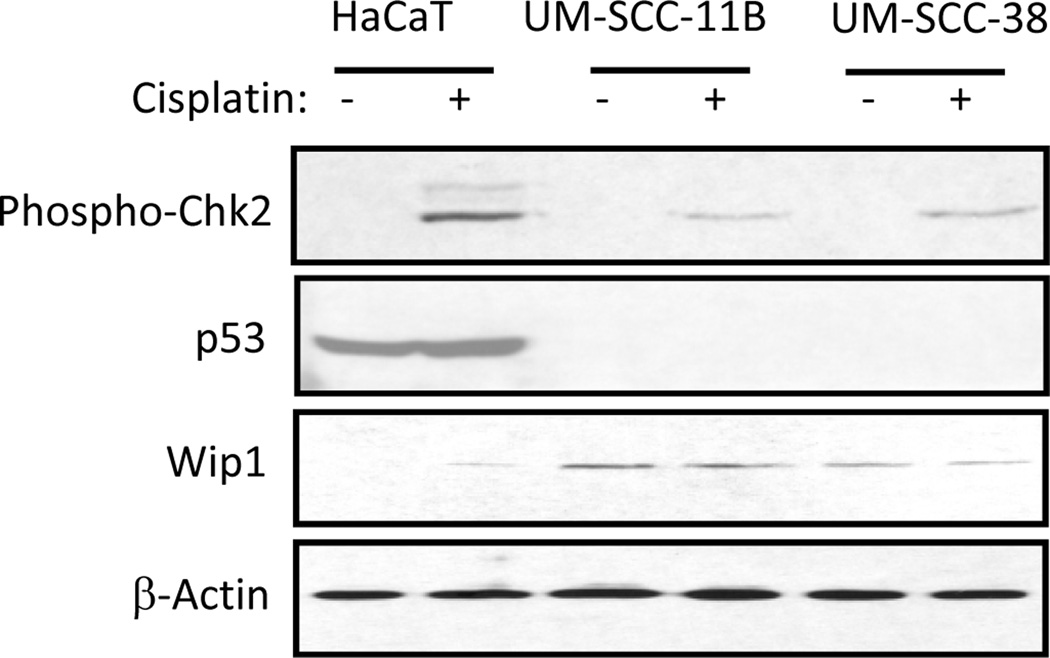
It has been shown that expression of Wip1 is induced by wild-type p53 after DNA damage (20). However, despite that previous studies discovered homozygous mutations of p53 in UM-SCC-11B and -38 cells (21), we observed strong up-regulation of Wip1 in these cells (Fig. 1B). Upon further examination of p53 status in UM-SCC-11B and -38 cells, we found that no p53 expression can be detected with or without cisplatin treatment (Fig. 3). Thus, the constitutive expression of Wip1 in these cells indicates Wip1 up-regulation in a p53- and DNA damage-independent manner.
Figure 3.
P53-independent expression of Wip1. HaCaT, UM-SCC-11B, and UM-SCC-38 cells were treated with cisplatin (16.7 µM) for 12 hours and then analyzed by immunoblotting for expression of p53, Wip1, β-Actin, and phosphorylation of Chk2.
Wip1 suppression sensitizes UM-SCC-11B cells to cisplatin
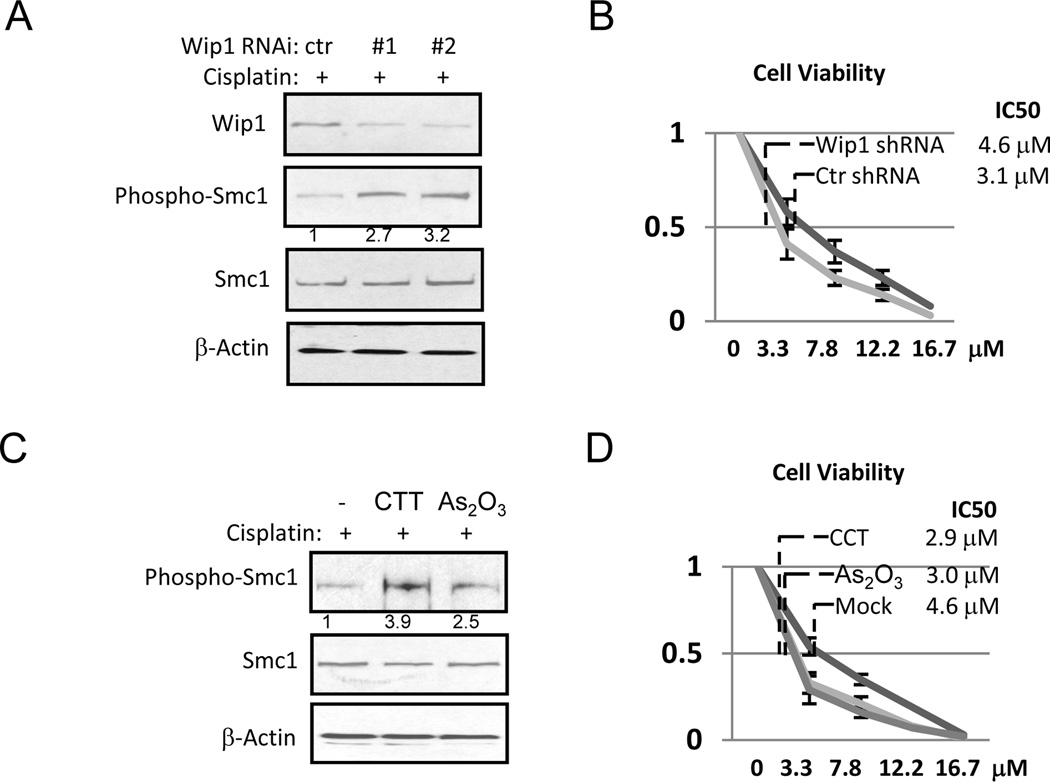
To investigate whether Wip1 up-regulation contributes to chemoresistance in UM-SCC-11B cells through DDR regulation, we sought to knockdown Wip1 expression using two lentiviral shRNA vectors. Both shRNA vectors partially reduced the level of Wip1 expression, and consequently, enhanced DDR signaling, as measured by Smc1 phosphorylation in response to cisplatin (Fig. 4A). Importantly, cell viability after cisplatin treatment was significantly reduced by Wip1 knockdown, indicating that chemoresistance in these cells is partially caused by Wip1 overexpression (Fig. 4B).
Figure 4.
Wip1 knockdown or inhibition enhances cisplatin-sensitivity. (A) UM-SCC-11B cells with control non-targeting shRNA (ctr) or Wip1 shRNA (#1 and #2) were treated with cisplatin (16.7 µM), and analyzed by immunoblotting. (B) UM-SCC-11B cells with ctr or Wip1 shRNA were incubated with cisplatin at various concentrations for 4 days (as in Fig. 2A). Cell viability was determined as described in Materials and Methods. (C) UM-SCC-11B cells were treated with cisplatin, CTT, and As2O3 as indicated, and analyzed by immunoblotting. (D) Cells in panel C were incubated with cisplatin at various concentrations for 4 days (as in Fig. 2A). Cell viability was determined as described in Materials and Methods.
Recent studies have shown that the phosphatase activity of Wip1 can be suppressed by specific inhibitors, including arsenic trioxide (As2O3) and CCT007093 (CCT) (22, 23). Like Wip1 knockdown, inhibition of Wip1 by arsenic trioxide or CCT augmented DDR signaling, as judged by cisplatin-induced phosphorylation of Smc1 (Fig. 4C). Moreover, the treatment with either arsenic trioxide or CCT led to a significant increase in cell sensitivity to cisplatin, as measured by cell viability (Fig. 4D).
In vivo tumor growth of UM-SCC-11B cells is impaired by Wip1 knockdown
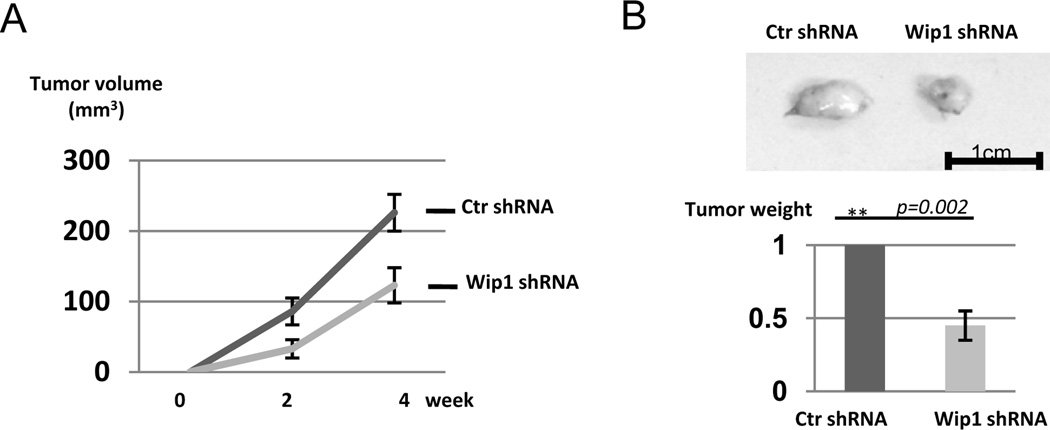
Implantation of SCC cells, including UM-SCC-11B cells, leads to formation of subcutaneous tumors in nude mice. Interestingly, UM-SCC-11B cells harboring knockdown of Wip1 expression exhibited reduced potential of tumor formation compared to the parental UM-SCC-11B cells (Fig. 5A). Tumors were excised and weighted at the end of the experiment, and Wip1 knockdown consistently reduced the tumor size by more than one-half (Fig. 5B). Our results suggest a partial addiction of these cells to Wip1 up-regulation during tumorigenesis.
Figure 5.
Wip1 knockdown reduces tumor growth in vivo. As described in Materials and Methods, UM-SCC-11B cells with control (ctr) shRNA or Wip1 shRNA were implanted into nude mice to form xenograft tumors. The volume of tumors was determined as described in Materials and Methods (A). At the end of the experiment, tumors were excised and weighed (B). Statistical significance was determined by Student’s t-test (N=5 per group).
In vivo inhibition of Wip1 for cancer therapy
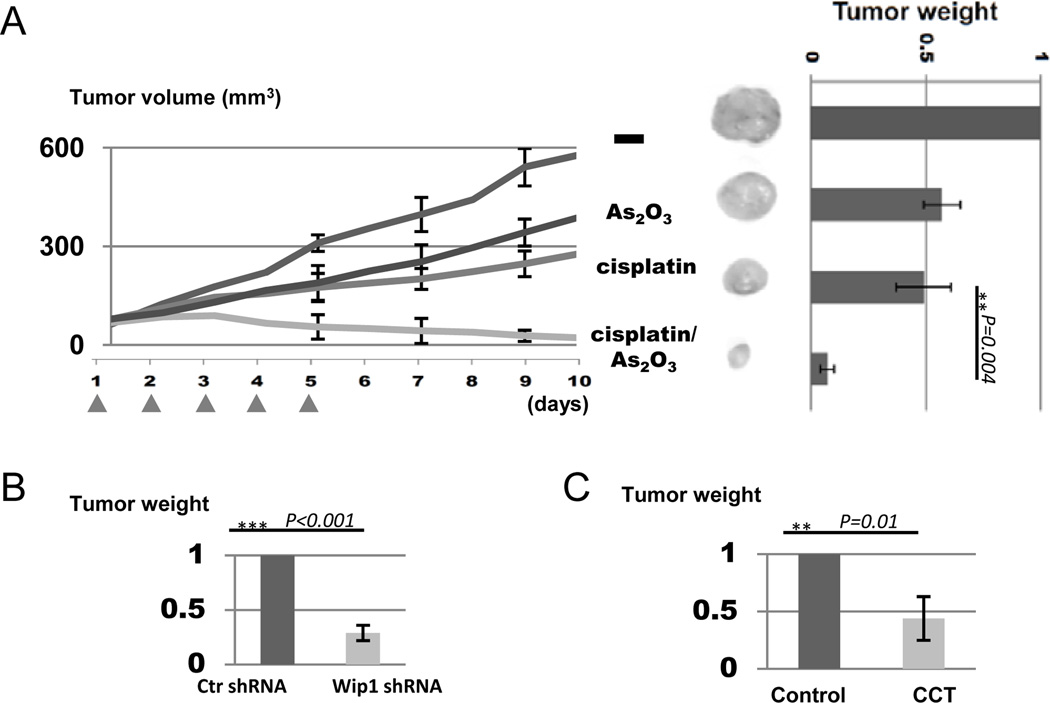
Prompted by our results showing that Wip1 knockdown or inhibition in cell culture sensitized UM-SCC-11B cells to cisplatin (Fig. 4), we sought to explore the potential of Wip1 inhibition for chemo-sensitization using the xenograft tumor model. We implanted UM-SCC-11B cells into nude mice to form tumors, and allowed tumors to reach 50 mm3 in volume. We then administered cisplatin and/or arsenic trioxide intraperitoneally every day for 5 days. As shown in Fig. 6A, while treatment with either cisplatin or arsenic trioxide as a single therapeutic agent moderately reduced the rate of tumor growth, the combination treatment using both cisplatin and arsenic trioxide exhibited strong synergy and yielded a greatly improved treatment outcome in shrinking tumors (Fig. 6A). Moreover, the combination treatment is well-tolerated as judged by the animal viability and weight (data not shown).
Figure 6.
Wip1 inhibition enhances tumor response to cisplatin in vivo. (A) UM-SCC-11B cells were implanted into nude mice to form tumors. As described in Materials and Methods, mice were then treated with As2O3 and cisplatin with five daily injections, as indicated. Tumor volume was measured, and tumors were excised and weighed. Statistical significance was determined by Student’s t-test (N≥4 per group) (B) UM-SCC-11B cells with control (ctr) shRNA or Wip1 shRNA were implanted into nude mice to form xenograft tumors. Once the tumor reached 50 mm3 in volume, the mouse was then treated with cisplatin as in panel A, and tumor weight was determined. Statistical significance was determined by Student’s t-test (N=4 per group) (C) As in panel A & B, UM-SCC-11B cells were implanted into nude mice, which were then treated with cisplatin alone (control), or cisplatin with CCT. Tumor weight was measured. Statistical significance was determined by Student’s t-test (N=5 per group).
To confirm that the synergistic effect seen with arsenic trioxide in chemotherapy is at least partially due to Wip1 inhibition, we implanted UM-SCC-11B cells with or without Wip1 knockdown into nude mice, and performed the above described therapeutic treatment using cisplatin. Interestingly, Wip1 knockdown also sensitized the cells to cisplatin as judged by decreased tumor weight (Fig. 6B). The enhanced tumor response to cisplatin observed with Wip1 knockdown is seemingly less profound than that seen with arsenic trioxide treatment, which may reflect the fact that only a partial knockdown of Wip1 expression was achieved. However, it should be noted that arsenic trioxide can simultaneously target other cellular pathways that contribute to cisplatin-sensitization (24). Finally, we examined the effect of CCT for chemosensitization using the xenograft tumor model. Unfortunately, in our experiments, the use of CCT was limited to low concentrations due to animal lethality, suggesting very limited therapeutic window using this inhibitor. Nevertheless, like arsenic trioxide or Wip1 knockdown, the use of CCT also improved the efficacy of cisplatin (Fig. 6C).
Discussion
Like other forms of radio- or chemotherapy, administration of cisplatin causes DNA damage and activates the cellular DDR pathway. In this study, we report that in some oral/laryngeal SCC cell lines, expression of several DDR factors, including ATM, Mre11 and H2AX, is significantly reduced. We showed that DDR signaling is partially defective in these cells, in correlation with chemoresistance to cisplatin. These findings are in line with several previous studies in head and neck cancer. For example, Parikh et. al. have previously shown that partial loss of the chromosome 11q21-23 region that harbors Mre11, ATM, and H2AX genes frequently occurs in oral SCCs (25). While the sensitivity of these cells to cisplatin was not examined, the authors reported that these cells exhibit decreased sensitivity to ionizing radiation in clonogenic survival assays (25). Moreover, independent studies discovered reduced ATM expression in a portion of head and neck cancer cases, in correlation with more aggressive clinicopathological features (26, 27). Thus, although ATM deficiency has been shown to cause hypersensitivity to DNA damage, as characterized in AT cells (4), our study is consistent with a number of others in patients and animal models indicating that loss of ATM correlates with resistance to chemotherapy and poor patient survival (25, 28–32). It is plausible that ATM can differentially affect how tumors respond to chemotherapy depending on their genetic background, such as p53 status (28).
In the study by Parikh et. al., it has also been suggested that oral SCC cells with reduced expression of ATM, Mre11 and H2AX are deficient in DNA repair, based on the observation that these cells, collectively, exhibit elevated levels of chromosomal abnormalities (25). In the present study, we directly measured the repair efficiency in SCC cells treated with cisplatin using a modified comet assay that quantifies the presence of DNA crosslink. Interestingly, no DNA repair deficiency was detected in cells with reduced ATM, Mre11, and H2AX expression. On one hand, this finding is consistent with the fact that these cells are more resistant to cisplatin; but on the other hand, it is surprising as ATM, Mre11, and H2AX are important DNA damage response proteins that contribute to DNA repair directly or indirectly through checkpoint activation. We speculate that this discrepancy could be due to two reasons: 1) redundant pathways, especially ATR-dependent signaling cascades, may compensate the loss of the ATM-dependent pathway and sustain the checkpoint and repair functions; and 2) key repair genes involved in nucleotide-excision repair or other repair mechanisms could be increased to allow cell survival after cisplatin treatment. Further clarifications of these aspects are potentially important as these efforts may reveal new drug targeting strategies that exploit the differential requirement of certain DDR pathways in normal and cancer cells to selectively sensitize the cancer cells to chemotherapy.
In addition to the reduced expression of ATM, Mre11 and H2AX, we discovered in the cisplatin-resistant SCC-11B and -38 cells simultaneous up-regulation of Wip1. Wip1 is a homeostatic regulator of the DDR that dephosphorylates and deactivates numerous DDR factors of the ATM-dependent signaling pathway (13). Therefore, Wip1 up-regulation in oral cancer cells indicates an effective mechanism to modulate DDR signaling. Wip1 was originally defined as a p53 substrate gene whose expression is induced by DNA damage in a p53-dependent manner (20). However, in UM-SCC-11B and -38 cells, we found that expression of Wip1 is neither induced by DNA damage nor dependent on p53. Thus the elevated and constitutive expression of Wip1 in oral SCCs is attributed to other mechanisms. Interestingly, several recent studies in breast and lung cancer cells also suggested p53-independent up-regulation of Wip1 expression (33–35). Clearly, the underlying mechanism of this regulation and its involvement in tumorigenesis should be investigated in the future.
We then sought to determine the functional relevance of Wip1 up-regulation in oral/laryngeal SCC cells. Wip1 knockdown reduced in vivo tumor growth of UM-SCC-11B cell, suggesting a partial addiction of these tumor cells to Wip1 up-regulation. To our knowledge, this is the first evidence that functionally characterizes an involvement of Wip1 in head and neck cancer tumorigenesis. Importantly, Wip1 knockdown enhanced ATM-dependent signaling pathway and rendered cells more sensitive to cisplatin treatment. These results suggest a therapeutic potential of Wip1 targeting in conjunction with cisplatin treatment in oral SCCs that are deficient in ATM-dependent DDR signaling pathway and resistant to cisplatin. Notably, the increased cisplatin-sensitivity resulted from Wip1 depletion or inhibition is clearly independent of p53, as UM-SCC-11B cells harbor a mutant p53 whose expression is completely suppressed. This is a surprising finding as the current understanding of the role of Wip1 in cancer progression and treatment is largely centered at its connection with p53. It has been shown in the normal cellular context that Wip1 expression is induced by p53, which is itself a key substrate of Wip1-mediated dephosphorylation and deactivation. Breast tumors with Wip1 overexpression rarely contain mutations in the p53 gene, suggesting that Wip1 may promote tumorigenesis largely through p53 inhibition (13). Moreover, several recent studies have suggested that Wip1 inactivation sensitized cancer cells to chemotherapy through p53-dependent apoptosis pathways (36–38). It has also been shown that down-regulation of Wip1 enhanced chemo-sensitivity only in tumor cells with wt-p53, whereas, by comparison, Wip1 down-regulation was completely ineffective in p53-deficient tumor cells (39). Thus our results extend the current knowledge about the role of Wip1 in cancer progression and therapy, and strongly argue for further examination into the therapeutic potential of Wip1 targeting in p53-negative tumors. Based on these results, we propose an involvement of a p53-independent cell death pathway(s) that accounts for the tumor response to cisplatin, whereas activation of this pathway is negatively regulated by Wip1. Our results suggest that p53 status does not adequately determine the therapeutic potential of Wip1 targeting, and p53-dependent apoptosis alone may not serve as an exclusive readout for the efficacy of chemotherapy. This notion is in line with earlier findings showing that the wild-type status of p53 is not positively correlated or even negatively correlated with chemosensitivity in oral SCCs (21, 40).
Recent studies have identified several chemical inhibitors of Wip1, including arsenic trioxide (As2O3) and CCT007093 (22, 23). Historically described in traditional Chinese medicine, arsenic trioxide has been shown to possess anti-cancer activity, and approved by the U.S. Food and Drug Administration for the treatment of acute myeloid leukemia (41). The potential use of arsenic trioxide in brain, breast, ovarian, prostate, and many other types of cancers, either as a monotherapeutic agent or in combination with other anti-cancer drugs, has been increasingly implicated (reviewed in (24, 42)). A recent study discovered that arsenic trioxide exerts its anti-proliferative activity at least partially through Wip1 inhibition (22). CCT007093 was recently identified as a potent inhibitor of Wip1 that selectively induces cell death in human breast cancer cells with Wip1 overexpression (23). In this study we directly evaluate the therapeutic potential of these Wip1 inhibitors as conjunctive treatments with cisplatin. Excitingly, our study demonstrated that both arsenic trioxide and CCT007093 exhibited strong synergy with cisplatin in oral SCCs: in cell culture, the combinatorial treatments led to elevated DDR signaling and decreased cell viability compared to treatment with cisplatin alone; and in xenograft tumor models, the combinatorial treatments more effectively shrunk the tumor. The use of CCT exhibited certain toxicity to the animal, which limits the therapeutic potential for this inhibitor in its current form. By comparison, the combination treatment using arsenic trioxide and cisplatin did not cause more animal lethality or weight-loss in host mice compared to treatment with cisplatin alone, suggesting good drug-tolerance. The effect of CCT and Arsenic oxide is conferred largely through Wip1 inhibition because: 1) CCT and Arsenic oxide were shown to inhibit Wip1 (22, 23); 2) both CCT and Arsenic oxide enhanced DNA damage signaling, consistent with their role in inhibiting Wip1; 3) CCT and Arsenic oxide-induced effects are similar to that observed with Wip1 knockdown. Our results thus validate Wip1 inhibition as an attractive strategy to confer cisplatin-sensitization in oral SCCs, especially those with deficient DDR to cisplatin. This notion shall be further explored in both pre-clinical and clinical arenas.
Acknowledgements
We thank Dr. Thomas Carey (University of Michigan) for providing cell lines, and Drs. Richard MacDonald and Thomas Petro for critically reading the manuscript.
Grant support
This project was supported by Grant Number 5P20GM103489 from the National Center for Research Resources (NCRR), a component of the National Institutes of Health (NIH), and an American Cancer Society grant (RSG-10-031-01-CCG, to G.G.O).
Grant Support: Supported by the National Institute of Health grant (8P20GM103489) to A.P., and American Cancer Society grant (RSG-10-031-01-CCG) to G.G.O.
Footnotes
Potential Conflicts of Interest: None.
References
- 1.Galluzzi L, Senovilla L, Vitale I, Michels J, Martins I, Kepp O, et al. Molecular mechanisms of cisplatin resistance. Oncogene. 2012;31:1869–1883. doi: 10.1038/onc.2011.384. [DOI] [PubMed] [Google Scholar]
- 2.Caponigro F, Milano A, Basile M, Ionna F, Iaffaioli RV. Recent advances in head and neck cancer therapy: the role of new cytotoxic and molecular-targeted agents. Curr opin oncol. 2006;18:247–252. doi: 10.1097/01.cco.0000219253.53091.fb. [DOI] [PubMed] [Google Scholar]
- 3.Zhou BBS, Anderson HJ, Roberge M. Targeting DNA checkpoint kinases in cancer therapy. Cancer biol ther. 2003;2:S16–S22. [PubMed] [Google Scholar]
- 4.Shiloh Y. ATM and related protein kinases: Safeguarding genome integrity. Nat Rev Cancer. 2003;3:155–168. doi: 10.1038/nrc1011. [DOI] [PubMed] [Google Scholar]
- 5.Uziel T, Lerenthal Y, Moyal L, Andegeko Y, Mittelman L, Shiloh Y. Requirement of the MRN complex for ATM activation by DNA damage. EMBO J. 2003;22:5612–5621. doi: 10.1093/emboj/cdg541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Carson CT, Schwartz RA, Stracker TH, Lilley CE, Lee DV, Weitzman MD. The Mre11 complex is required for ATM activation and the G2/M checkpoint. EMBO J. 2003;22:6610–6620. doi: 10.1093/emboj/cdg630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zou L, Elledge SJ. Sensing DNA damage through ATRIP recognition of RPA-ssDNA complexes. Science. 2003;300:1542–1548. doi: 10.1126/science.1083430. [DOI] [PubMed] [Google Scholar]
- 8.Kumagai a, Lee J, Yoo HY, Dunphy WG. TopBP1 activates the ATR-ATRIP complex. Cell. 2006;124:943–955. doi: 10.1016/j.cell.2005.12.041. [DOI] [PubMed] [Google Scholar]
- 9.Prendergast AM, Cruet-Hennequart S, Shaw G, Barry FP, Carty MP. Activation of DNA damage response pathways in human mesenchymal stem cells exposed to cisplatin or gamma-irradiation. Cell Cycle. 2011;10:3768–3777. doi: 10.4161/cc.10.21.17972. [DOI] [PubMed] [Google Scholar]
- 10.Pabla N, Ma Z, McIlhatton MA, Fishel R, Dong Z. hMSH2 recruits ATR to DNA damage sites for activation during DNA damage-induced apoptosis. J Biol Chem. 2011;286:10411–10418. doi: 10.1074/jbc.M110.210989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Liang X, Guo Y, Figg WD, Fojo AT, Mueller MD, Yu JJ. The Role of Wild-Type p53 in Cisplatin-Induced Chk2 Phosphorylation and the Inhibition of Platinum Resistance with a Chk2 Inhibitor. chemother res pract. 2011;2011:715469. doi: 10.1155/2011/715469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Peng A, Maller JL. Serine/Threonine Protein Phosphatases in the DNA Damage Response and Cancer. Oncogene. 2010;29:5977–5988. doi: 10.1038/onc.2010.371. [DOI] [PubMed] [Google Scholar]
- 13.Lu XB, Nguyen TA, Moon SH, Darlington Y, Sommer M, Donehower LA. The type 2C phosphatase Wip1: An oncogenic regulator of tumor suppressor and DNA damage response pathways. Cancer Metastasis Rev. 2008;27:123–135. doi: 10.1007/s10555-008-9127-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Le Guezennec X, Bulavin DV. WIP1 phosphatase at the crossroads of cancer and aging. Trends biochem sci. 2010;35:109–114. doi: 10.1016/j.tibs.2009.09.005. [DOI] [PubMed] [Google Scholar]
- 15.Brenner JC, Graham MP, Kumar B, Saunders LM, Kupfer R, Lyons RH, et al. Genotyping of 73 UM-SCC head and neck squamous cell carcinoma cell lines. Head neck. 2010;32:417–426. doi: 10.1002/hed.21198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Manthey KC, Glanzer JG, Dimitrova DD, Oakley GG. Hyperphosphorylation of Replication Protein a in Cisplatin-Resistant and -Sensitive Head and Neck Squamous Cell Carcinoma Cell Lines. Head Neck. 2010;32:636–645. doi: 10.1002/hed.21234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Peng A, Wang L, Fisher LA. Greatwall and Polo-like kinase 1 coordinate to promote checkpoint recovery. J Biol Chem. 2011;286:28996–29004. doi: 10.1074/jbc.M111.257121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wang L, Fisher LA, Wahl JK, 3rd, Peng A. Monoclonal antibodies against Xenopus greatwall kinase. Hybridoma. 2011;30:469–474. doi: 10.1089/hyb.2011.0051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Spanswick VJ, Hartley JM, Ward TH, Hartley JA. Measurement of drug-induced DNA interstrand crosslinking using the single-cell gel electrophoresis (comet) assay. Methods mol med. 1999;28:143–154. doi: 10.1385/1-59259-687-8:143. [DOI] [PubMed] [Google Scholar]
- 20.Fiscella M, Zhang H, Fan S, Sakaguchi K, Shen S, Mercer WE, et al. Wip1, a novel human protein phosphatase that is induced in response to ionizing radiation in a p53-dependent manner. Proc Natl Acad Sci U S A. 1997;94:6048–6053. doi: 10.1073/pnas.94.12.6048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bradford CR, Zhu S, Ogawa H, Ogawa T, Ubell M, Narayan A, et al. P53 mutation correlates with cisplatin sensitivity in head and neck squamous cell carcinoma lines. Head neck. 2003;25:654–661. doi: 10.1002/hed.10274. [DOI] [PubMed] [Google Scholar]
- 22.Yoda A, Toyoshima K, Watanabe Y, Onishi N, Hazaka Y, Tsukuda Y, et al. Arsenic trioxide augments Chk2/p53-mediated apoptosis by inhibiting oncogenic Wip1 phosphatase. J Biol Chem. 2008;283:18969–18979. doi: 10.1074/jbc.M800560200. [DOI] [PubMed] [Google Scholar]
- 23.Rayter S, Elliott R, Travers J, Rowlands MG, Richardson TB, Boxall K, et al. A chemical inhibitor of PPM1D that selectively kills cells overexpressing PPM1D. Oncogene. 2008;27:1036–1044. doi: 10.1038/sj.onc.1210729. [DOI] [PubMed] [Google Scholar]
- 24.Platanias LC. Biological responses to arsenic compounds. J Biol Chem. 2009;284:18583–18587. doi: 10.1074/jbc.R900003200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Parikh RA, White JS, Huang X, Schoppy DW, Baysal BE, Baskaran R, et al. Loss of distal 11q is associated with DNA repair deficiency and reduced sensitivity to ionizing radiation in head and neck squamous cell carcinoma. Genes, chromosomes cancer. 2007;46:761–775. doi: 10.1002/gcc.20462. [DOI] [PubMed] [Google Scholar]
- 26.He Y, Chen Q, Li B. ATM in oral carcinogenesis: association with clinicopathological features. J Cancer Res Clin Oncol. 2008;134:1013–1020. doi: 10.1007/s00432-008-0365-7. [DOI] [PubMed] [Google Scholar]
- 27.Lee KW, Tsai YS, Chiang FY, Huang JL, Ho KY, Yang YH, et al. Lower ataxia telangiectasia mutated (ATM) mRNA expression is correlated with poor outcome of laryngeal and pharyngeal cancer patients. Annals oncol. 2011;22:1088–1093. doi: 10.1093/annonc/mdq569. [DOI] [PubMed] [Google Scholar]
- 28.Jiang H, Reinhardt HC, Bartkova J, Tommiska J, Blomqvist C, Nevanlinna H, et al. The combined status of ATM and p53 link tumor development with therapeutic response. Genes Dev. 2009;23:1895–1909. doi: 10.1101/gad.1815309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Gumy-Pause F, Wacker P, Sappino AP. ATM gene and lymphoid malignancies. Leukemia. 2004;18:238–242. doi: 10.1038/sj.leu.2403221. [DOI] [PubMed] [Google Scholar]
- 30.Haidar MA, Kantarjian H, Manshouri T, Chang CY, O'Brien S, Freireich E, et al. ATM gene deletion in patients with adult acute lymphoblastic leukemia. Cancer. 2000;88:1057–1062. doi: 10.1002/(sici)1097-0142(20000301)88:5<1057::aid-cncr16>3.0.co;2-6. [DOI] [PubMed] [Google Scholar]
- 31.Austen B, Skowronska A, Baker C, Powell JE, Gardiner A, Oscier D, et al. Mutation status of the residual ATM allele is an important determinant of the cellular response to chemotherapy and survival in patients with chronic lymphocytic leukemia containing an 11q deletion. J Clin Oncol. 2007;25:5448–5457. doi: 10.1200/JCO.2007.11.2649. [DOI] [PubMed] [Google Scholar]
- 32.Ripolles L, Ortega M, Ortuno F, Gonzalez A, Losada J, Ojanguren J, et al. Genetic abnormalities and clinical outcome in chronic lymphocytic leukemia. Cancer genet cytogenet. 2006;171:57–64. doi: 10.1016/j.cancergencyto.2006.07.006. [DOI] [PubMed] [Google Scholar]
- 33.Park JY, Song JY, Kim HM, Han HS, Seol HS, Jang SJ, et al. p53-Independent expression of wild-type p53-induced phosphatase 1 (Wip1) in methylmethane sulfonate-treated cancer cell lines and human tumors. Int J biochem cell biol. 2012;44:896–904. doi: 10.1016/j.biocel.2012.02.013. [DOI] [PubMed] [Google Scholar]
- 34.Lowe JM, Cha H, Yang Q, Fornace AJ., Jr Nuclear factor-kappaB (NF-kappaB) is a novel positive transcriptional regulator of the oncogenic Wip1 phosphatase. J Biol Chem. 2010;285:5249–5257. doi: 10.1074/jbc.M109.034579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Han HS, Yu E, Song JY, Park JY, Jang SJ, Choi J. The estrogen receptor alpha pathway induces oncogenic Wip1 phosphatase gene expression. Mol cancer res. 2009;7:713–723. doi: 10.1158/1541-7786.MCR-08-0247. [DOI] [PubMed] [Google Scholar]
- 36.Yagi H, Chuman Y, Kozakai Y, Imagawa T, Takahashi Y, Yoshimura F, et al. A small molecule inhibitor of p53-inducible protein phosphatase PPM1D. Bioorg med chem lett. 2012;22:729–732. doi: 10.1016/j.bmcl.2011.10.084. [DOI] [PubMed] [Google Scholar]
- 37.Ali AY, Abedini MR, Tsang BK. The oncogenic phosphatase PPM1D confers cisplatin resistance in ovarian carcinoma cells by attenuating checkpoint kinase 1 and p53 activation. Oncogene. 2011;31:2175–2186. doi: 10.1038/onc.2011.399. [DOI] [PubMed] [Google Scholar]
- 38.Kong W, Jiang X, Mercer WE. Downregulation of Wip-1 phosphatase expression in MCF-7 breast cancer cells enhances doxorubicin-induced apoptosis through p53-mediated transcriptional activation of Bax. Cancer biol ther. 2009;8:555–563. doi: 10.4161/cbt.8.6.7742. [DOI] [PubMed] [Google Scholar]
- 39.Goloudina AR, Tanoue K, Hammann A, Fourmaux E, Le Guezennec X, Bulavin DV, et al. Wip1 promotes RUNX2-dependent apoptosis in p53-negative tumors and protects normal tissues during treatment with anticancer agents. Proc Natl Acad Sci U S A. 2012;109:E68–E75. doi: 10.1073/pnas.1107017108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Bauer JA, Trask DK, Kumar B, Los G, Castro J, Lee JS, et al. Reversal of cisplatin resistance with a BH3 mimetic, (Ȣ)-gossypol, in head and neck cancer cells: role of wild-type p53 and Bcl-xL. Mol Cancer Ther. 2005;4:1096–1104. doi: 10.1158/1535-7163.MCT-05-0081. [DOI] [PubMed] [Google Scholar]
- 41.Verstovsek S, Giles F, Quintas-Cardama A, Perez N, Ravandi-Kashani F, Beran M, et al. Arsenic derivatives in hematologic malignancies: a role beyond acute promyelocytic leukemia? Hematol oncol. 2006;24:181–188. doi: 10.1002/hon.787. [DOI] [PubMed] [Google Scholar]
- 42.Emadi A, Gore SD. Arsenic trioxide - An old drug rediscovered. Blood rev. 2010;24:191–199. doi: 10.1016/j.blre.2010.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]