Abstract
Human platelets contain a mitogen, the platelet-derived growth factor (PDGF), that stimulates the proliferation of a variety of cell types in culture and that may play a role in atherogenesis. Studies were conducted to explore the effects of PDGF on low density lipoprotein (LDL) receptor activity of cultured human fibroblasts. The PDGF utilized in these studies was partially purified from human platelet-rich plama by ion exchange chromatography and gel filtration. LDL receptor activity was assessed by both specific binding of 125I-labeled LDL to the fibroblast's surface at 4 degrees C, and the incorporation of [14C]oleate into cholesteryl esters. Exposure of normal human fibroblasts to increasing amounts of PDGF (0.1-10 microgram/ml) for 48 hr caused a dose-related increase in 125I-labeled LDL binding to a maximum of approximately 300%. In the presence of added LDL, this increase in LDL binding was not seen. Cholesterol esterifiction was also stimulated following a 48-hr exposure to PDGF. Following a conditioning period in LDL- and PDGF-depleted medium, cholesterol esterification was greatly increased during a 48-hr exposure to LDL alone; a smaller but significant increase occurred with PDGF alone. However, both PDGF and LDL were required to return the level of esterification to that observed with whole human serum. Fibroblasts from a patient with homozygous familial hypercholesterolemia, which lack the LDL receptor, also showed a significant increase in cholesteol esterification with PDGF alone, whereas LDL had no effect. These studies demonstrate that PDGF can stimulate the LDL receptor activity in cultured human fibroblasts. The effect on other related activities of the LDL receptor system and the mechanism involved remain to be defined.
Full text
PDF
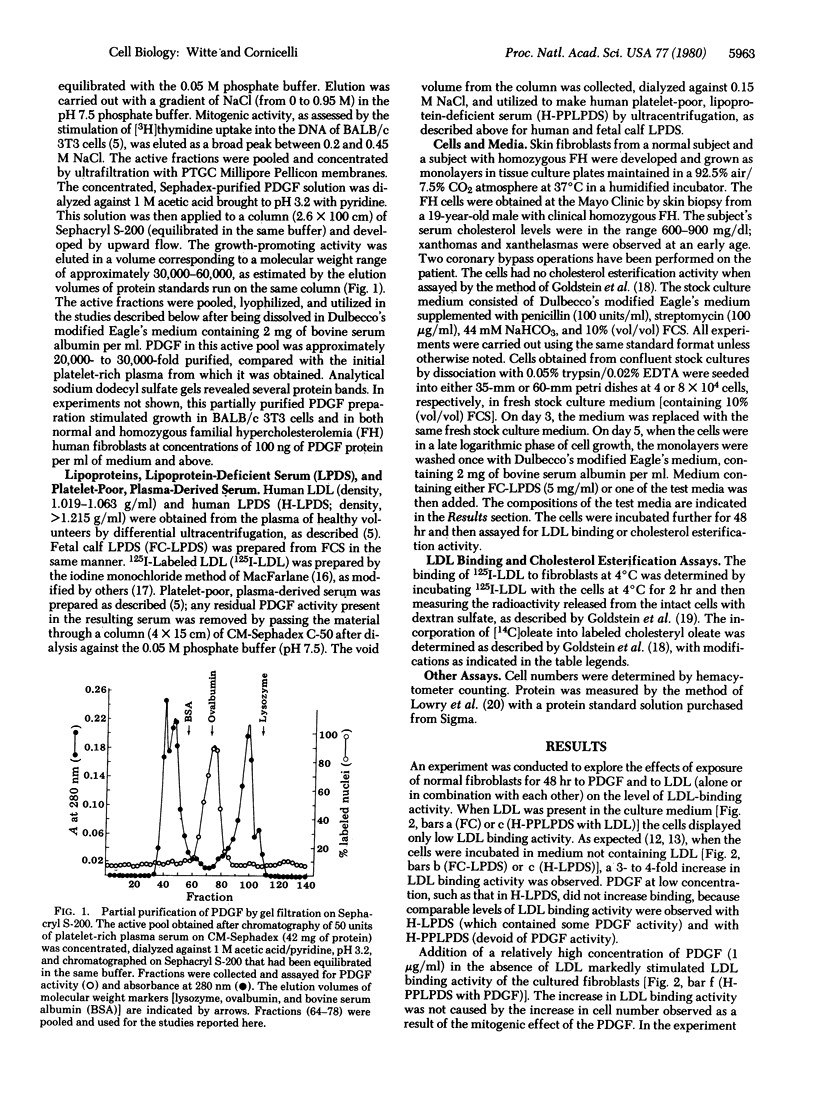
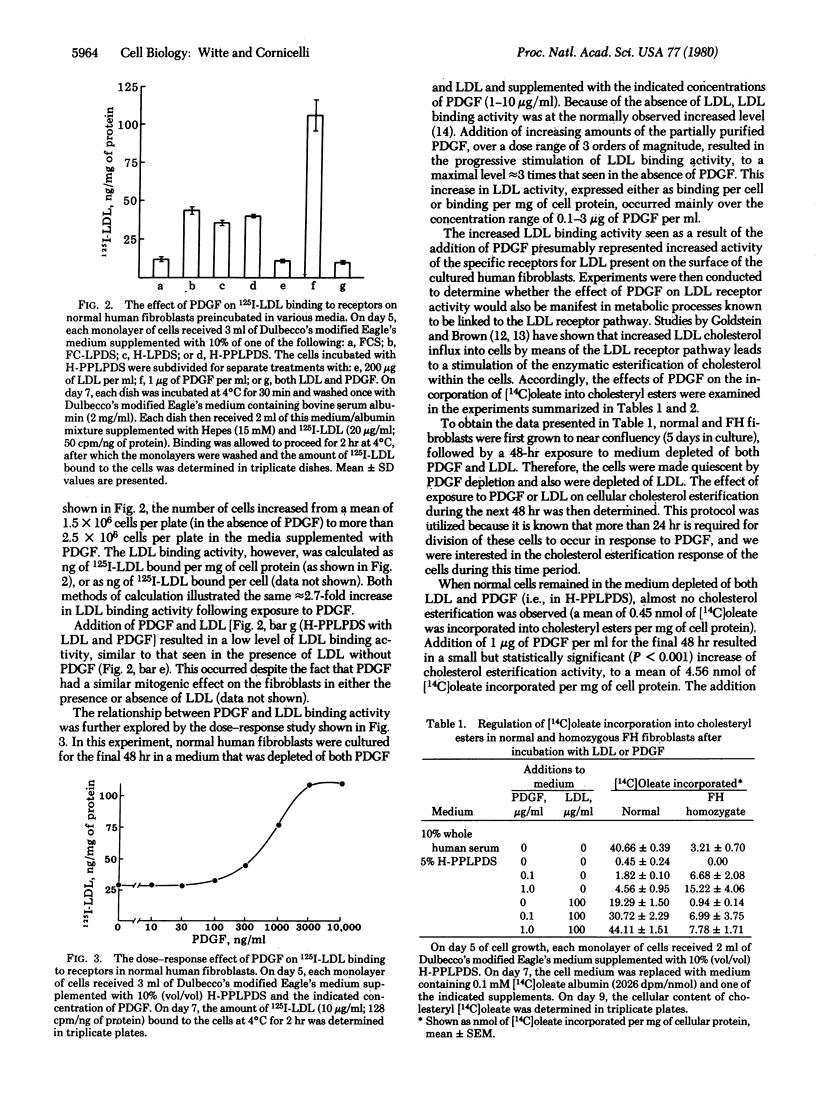


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Antoniades H. N., Scher C. D., Stiles C. D. Purification of human platelet-derived growth factor. Proc Natl Acad Sci U S A. 1979 Apr;76(4):1809–1813. doi: 10.1073/pnas.76.4.1809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bilheimer D. W., Eisenberg S., Levy R. I. The metabolism of very low density lipoprotein proteins. I. Preliminary in vitro and in vivo observations. Biochim Biophys Acta. 1972 Feb 21;260(2):212–221. doi: 10.1016/0005-2760(72)90034-3. [DOI] [PubMed] [Google Scholar]
- Brown M. S., Faust J. R., Goldstein J. L., Kaneko I., Endo A. Induction of 3-hydroxy-3-methylglutaryl coenzyme A reductase activity in human fibroblasts incubated with compactin (ML-236B), a competitive inhibitor of the reductase. J Biol Chem. 1978 Feb 25;253(4):1121–1128. [PubMed] [Google Scholar]
- Brown M. S., Goldstein J. L. Suppression of 3-hydroxy-3-methylglutaryl coenzyme A reductase activity and inhibition of growth of human fibroblasts by 7-ketocholesterol. J Biol Chem. 1974 Nov 25;249(22):7306–7314. [PubMed] [Google Scholar]
- Burke J. M., Ross R. Collagen synthesis by monkey arterial smooth muscle cells during proliferation and quiescence in culture. Exp Cell Res. 1977 Jul;107(2):387–395. doi: 10.1016/0014-4827(77)90360-3. [DOI] [PubMed] [Google Scholar]
- Castelli W. P., Doyle J. T., Gordon T., Hames C. G., Hjortland M. C., Hulley S. B., Kagan A., Zukel W. J. HDL cholesterol and other lipids in coronary heart disease. The cooperative lipoprotein phenotyping study. Circulation. 1977 May;55(5):767–772. doi: 10.1161/01.cir.55.5.767. [DOI] [PubMed] [Google Scholar]
- Davies P. F., Ross R. Mediation of pinocytosis in cultured arterial smooth muscle and endothelial cells by platelet-derived growth factor. J Cell Biol. 1978 Dec;79(3):663–671. doi: 10.1083/jcb.79.3.663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Faust J. R., Goldstein J. L., Brown M. S. Squalene synthetase activity in human fibroblasts: regulation via the low density lipoprotein receptor. Proc Natl Acad Sci U S A. 1979 Oct;76(10):5018–5022. doi: 10.1073/pnas.76.10.5018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Faust J. R., Goldstein J. L., Brown M. S. Squalene synthetase activity in human fibroblasts: regulation via the low density lipoprotein receptor. Proc Natl Acad Sci U S A. 1979 Oct;76(10):5018–5022. doi: 10.1073/pnas.76.10.5018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Friedman R. J., Stemerman M. B., Wenz B., Moore S., Gauldie J., Gent M., Tiell M. L., Spaet H. The effect of thrombocytopenia on experimental arteriosclerotic lesion formation in rabbits. Smooth muscle cell proliferation and re-endothelialization. J Clin Invest. 1977 Nov;60(5):1191–1201. doi: 10.1172/JCI108872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldstein J. L., Basu S. K., Brunschede G. Y., Brown M. S. Release of low density lipoprotein from its cell surface receptor by sulfated glycosaminoglycans. Cell. 1976 Jan;7(1):85–95. doi: 10.1016/0092-8674(76)90258-0. [DOI] [PubMed] [Google Scholar]
- Goldstein J. L., Brown M. S. Binding and degradation of low density lipoproteins by cultured human fibroblasts. Comparison of cells from a normal subject and from a patient with homozygous familial hypercholesterolemia. J Biol Chem. 1974 Aug 25;249(16):5153–5162. [PubMed] [Google Scholar]
- Goldstein J. L., Brown M. S. The low-density lipoprotein pathway and its relation to atherosclerosis. Annu Rev Biochem. 1977;46:897–930. doi: 10.1146/annurev.bi.46.070177.004341. [DOI] [PubMed] [Google Scholar]
- Goldstein J. L., Dana S. E., Brown M. S. Esterification of low density lipoprotein cholesterol in human fibroblasts and its absence in homozygous familial hypercholesterolemia. Proc Natl Acad Sci U S A. 1974 Nov;71(11):4288–4292. doi: 10.1073/pnas.71.11.4288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldstein J. L., Helgeson J. A., Brown M. S. Inhibition of cholesterol synthesis with compactin renders growth of cultured cells dependent on the low density lipoprotein receptor. J Biol Chem. 1979 Jun 25;254(12):5403–5409. [PubMed] [Google Scholar]
- Harker L. A., Ross R., Slichter S. J., Scott C. R. Homocystine-induced arteriosclerosis. The role of endothelial cell injury and platelet response in its genesis. J Clin Invest. 1976 Sep;58(3):731–741. doi: 10.1172/JCI108520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heldin C. H., Westermark B., Wasteson A. Platelet-derived growth factor: purification and partial characterization. Proc Natl Acad Sci U S A. 1979 Aug;76(8):3722–3726. doi: 10.1073/pnas.76.8.3722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kandutsch A. A., Chen H. W. Consequences of blocked sterol synthesis in cultured cells. DNA synthesis and membrane composition. J Biol Chem. 1977 Jan 25;252(2):409–415. [PubMed] [Google Scholar]
- Kruth H. S., Avigan J., Gamble W., Vaughan M. Effect of cell density on binding and uptake of low density lipoprotein by human fibroblasts. J Cell Biol. 1979 Dec;83(3):588–594. doi: 10.1083/jcb.83.3.588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Linder B. L., Chernoff A., Kaplan K. L., Goodman D. S. Release of platelet-derived growth factor from human platelets by arachidonic acid. Proc Natl Acad Sci U S A. 1979 Aug;76(8):4107–4111. doi: 10.1073/pnas.76.8.4107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quesney-Huneeus V., Wiley M. H., Siperstein M. D. Essential role for mevalonate synthesis in DNA replication. Proc Natl Acad Sci U S A. 1979 Oct;76(10):5056–5060. doi: 10.1073/pnas.76.10.5056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ross R., Glomset J. A. The pathogenesis of atherosclerosis (second of two parts). N Engl J Med. 1976 Aug 19;295(8):420–425. doi: 10.1056/NEJM197608192950805. [DOI] [PubMed] [Google Scholar]
- Ross R., Glomset J., Kariya B., Harker L. A platelet-dependent serum factor that stimulates the proliferation of arterial smooth muscle cells in vitro. Proc Natl Acad Sci U S A. 1974 Apr;71(4):1207–1210. doi: 10.1073/pnas.71.4.1207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ross R., Harker L. Hyperlipidemia and atherosclerosis. Science. 1976 Sep 17;193(4258):1094–1100. doi: 10.1126/science.822515. [DOI] [PubMed] [Google Scholar]
- Ross R., Vogel A. The platelet-derived growth factor. Cell. 1978 Jun;14(2):203–210. doi: 10.1016/0092-8674(78)90107-1. [DOI] [PubMed] [Google Scholar]
- Stein O., Stein Y. Surface binding and interiorization of homologous and heterologous serum lipoproteins by rat aortic smooth muscle cells in culture. Biochim Biophys Acta. 1975 Sep 19;398(3):377–384. doi: 10.1016/0005-2760(75)90188-5. [DOI] [PubMed] [Google Scholar]
- Weiss H. J., Witte L. D., Kaplan K. L., Lages B. A., Chernoff A., Nossel H. L., Goodman D. S., Baumgartner H. R. Heterogeneity in storage pool deficiency: studies on granule-bound substances in 18 patients including variants deficient in alpha-granules, platelet factor 4, beta-thromboglobulin, and platelet-derived growth factor. Blood. 1979 Dec;54(6):1296–1319. [PubMed] [Google Scholar]
- Witte L. D., Kaplan K. L., Nossel H. L., Lages B. A., Weiss H. J., Goodman D. S. Studies of the release from human platelets of the growth factor for cultured human arterial smooth muscle cells. Circ Res. 1978 Mar;42(3):402–409. doi: 10.1161/01.res.42.3.402. [DOI] [PubMed] [Google Scholar]


