Abstract
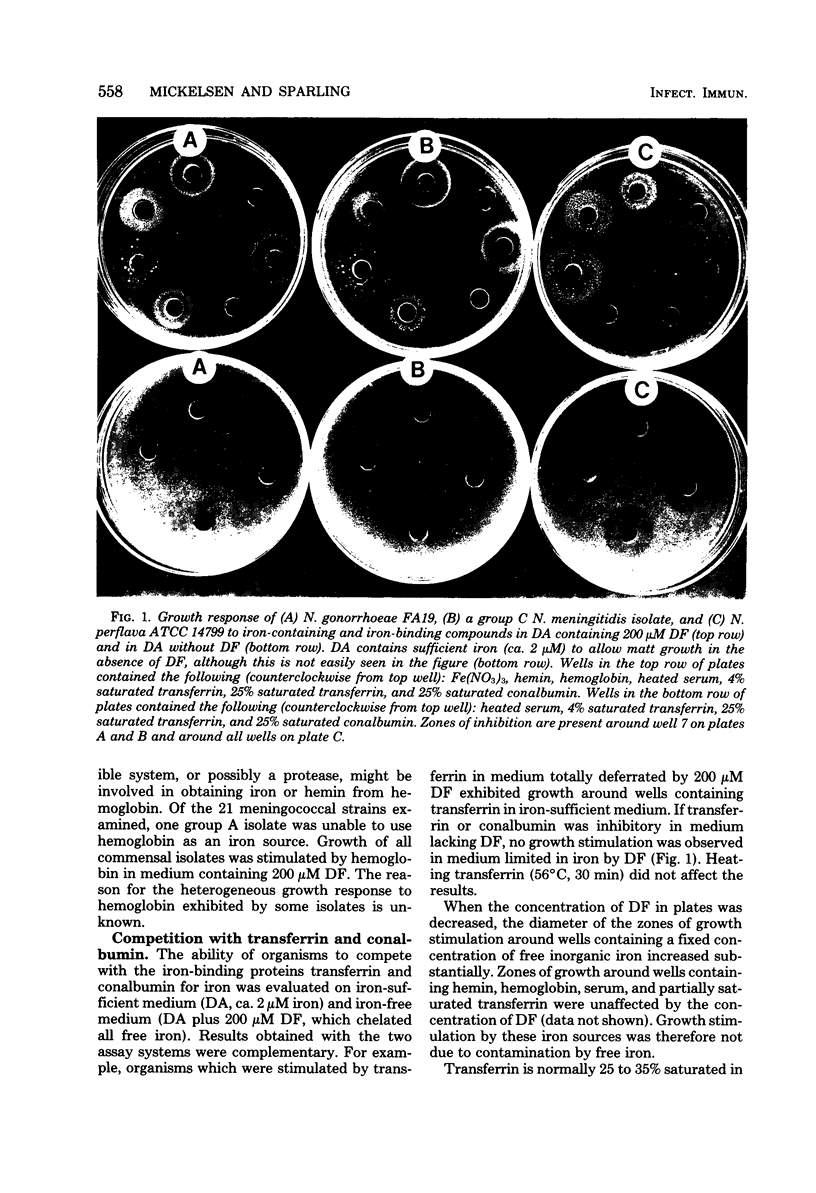
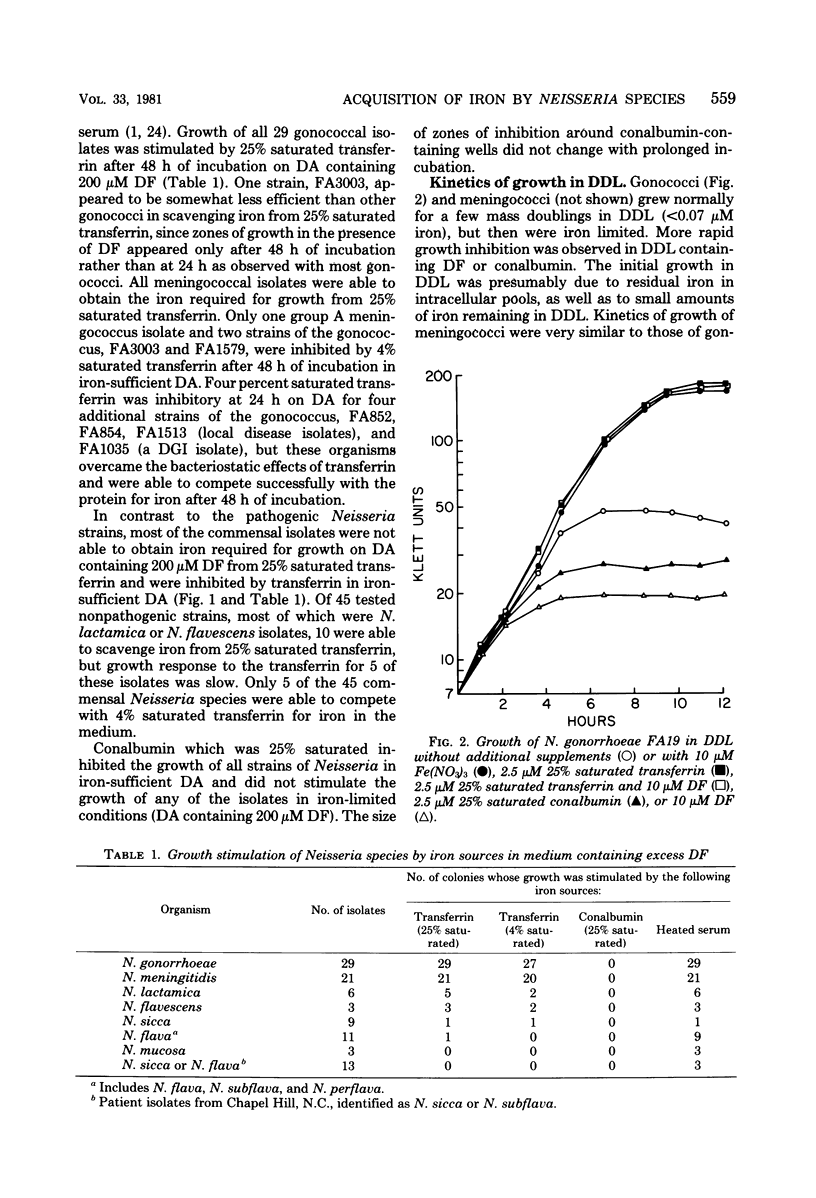
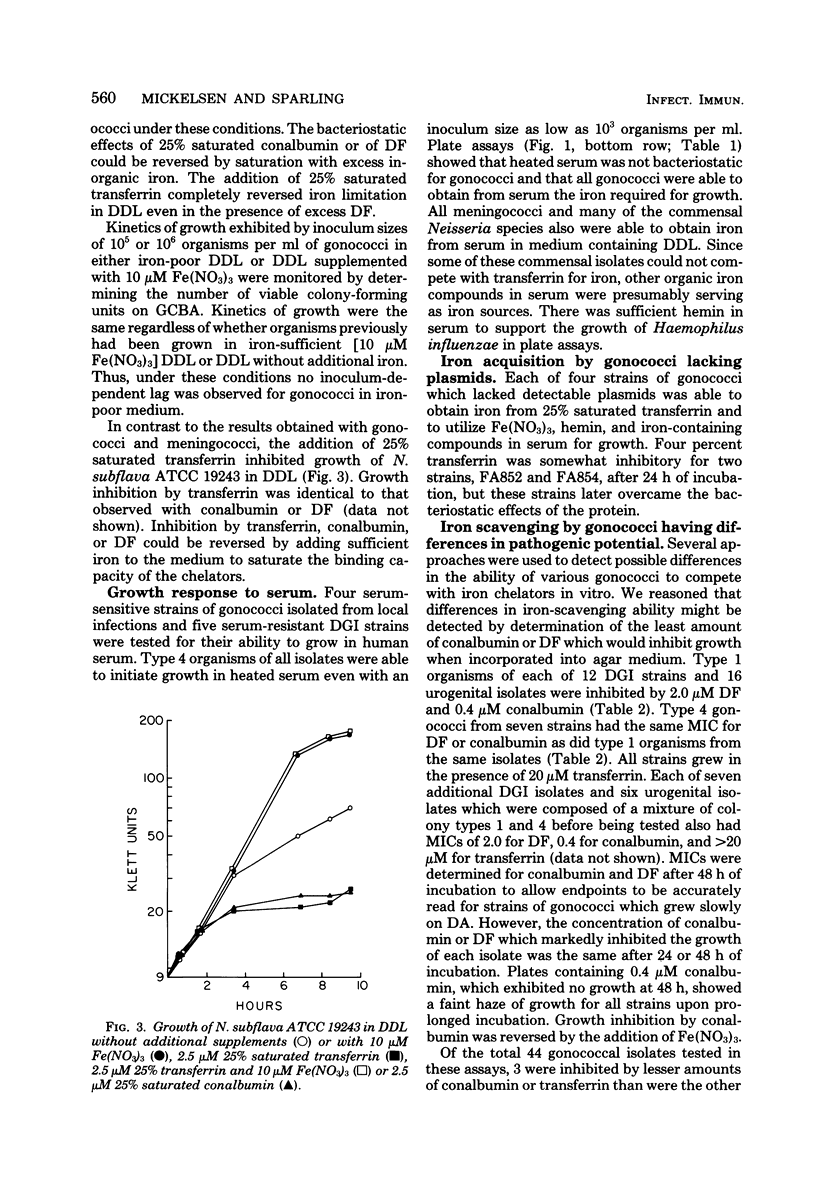
The ability of Neisseria species to use iron compounds and to compete with iron-binding proteins was examined with deferrated defined medium and the iron chelator deferoxamine. All Neisseria species were able to assimilate a variety of ferric and ferrous iron salts. They were not able to efficiently solubilize an inorganic iron salt such as ferric nitrate, but were able to use iron chelated by citrate, oxalacetate, pyrophosphate, or nitrilotriacetate. Each of the 95 Neisseria isolates examined was able to use hemin as a sole source of iron, and most, but not all, of the isolates were able to obtain iron from hemoglobin. Heated human serum stimulated growth of all gonococci, meningococci, and some commensal Neisseria species in iron-deficient medium. All gonococci and meningococci were able to scavenge iron from 25% saturated transferrin, whereas most commensal organisms were inhibited by this iron-binding protein. The ability to compete with transferrin was specific, since partially saturated conalbumin was bacteriostatic for all Neisseria species. Although the pathogenic Neisseria species were able to compete more efficiently with transferrin for iron than were the nonpathogenic Neisseria species, no correlation was observed between the virulence of different strains or colony types of gonococci and the ability to scavenge iron in vitro from transferrin or other chelators.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aisen P., Listowsky I. Iron transport and storage proteins. Annu Rev Biochem. 1980;49:357–393. doi: 10.1146/annurev.bi.49.070180.002041. [DOI] [PubMed] [Google Scholar]
- Archibald F. S., DeVoe I. W. Iron acquisition by Neisseria meningitidis in vitro. Infect Immun. 1980 Feb;27(2):322–334. doi: 10.1128/iai.27.2.322-334.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bullen J. J., Rogers H. J., Griffiths E. Role of iron in bacterial infection. Curr Top Microbiol Immunol. 1978;80:1–35. doi: 10.1007/978-3-642-66956-9_1. [DOI] [PubMed] [Google Scholar]
- Bullen J. J., Rogers H. J., Leigh L. Iron-binding proteins in milk and resistance to Escherichia coli infection in infants. Br Med J. 1972 Jan 8;1(5792):69–75. doi: 10.1136/bmj.1.5792.69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Catlin B. W. Nutritional profiles of Neisseria gonorrhoeae, Neisseria meningitidis, and Neisseria lactamica in chemically defined media and the use of growth requirements for gonococcal typing. J Infect Dis. 1973 Aug;128(2):178–194. doi: 10.1093/infdis/128.2.178. [DOI] [PubMed] [Google Scholar]
- Crosa J. H. A plasmid associated with virulence in the marine fish pathogen Vibrio anguillarum specifies an iron-sequestering system. Nature. 1980 Apr 10;284(5756):566–568. doi: 10.1038/284566a0. [DOI] [PubMed] [Google Scholar]
- Eisenstein B. I., Lee T. J., Sparling P. F. Penicillin sensitivity and serum resistance are independent attributes of strains of Neisseria gonorrhoeae causing disseminated gonococcal infection. Infect Immun. 1977 Mar;15(3):834–841. doi: 10.1128/iai.15.3.834-841.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grahm G., Bates G. W. Approaches to the standardization of serum unsaturated iron-binding capacity. J Lab Clin Med. 1976 Sep;88(3):477–486. [PubMed] [Google Scholar]
- Holbein B. E. Iron-controlled infection with Neisseria meningitidis in mice. Infect Immun. 1980 Sep;29(3):886–891. doi: 10.1128/iai.29.3.886-891.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holbein B. E., Jericho K. W., Likes G. C. Neisseria meningitidis infection in mice: influence of iron, variations in virulence among strains, and pathology. Infect Immun. 1979 May;24(2):545–551. doi: 10.1128/iai.24.2.545-551.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Imsande J. Genetic regulation of penicillinase synthesis in Gram-positive bacteria. Microbiol Rev. 1978 Mar;42(1):67–83. doi: 10.1128/mr.42.1.67-83.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- KELLOGG D. S., Jr, PEACOCK W. L., Jr, DEACON W. E., BROWN L., PIRKLE D. I. NEISSERIA GONORRHOEAE. I. VIRULENCE GENETICALLY LINKED TO CLONAL VARIATION. J Bacteriol. 1963 Jun;85:1274–1279. doi: 10.1128/jb.85.6.1274-1279.1963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kojima N., Bates G. W. The reduction and release of iron from Fe3+ .transferrin.CO3(2-). J Biol Chem. 1979 Sep 25;254(18):8847–8854. [PubMed] [Google Scholar]
- Kvach J. T., Wiles T. I., Mellencamp M. W., Kochan I. Use of transferrin-iron enterobactin complexes as the source of iron by serum-exposed bacteria. Infect Immun. 1977 Nov;18(2):439–445. doi: 10.1128/iai.18.2.439-445.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meyers J. A., Sanchez D., Elwell L. P., Falkow S. Simple agarose gel electrophoretic method for the identification and characterization of plasmid deoxyribonucleic acid. J Bacteriol. 1976 Sep;127(3):1529–1537. doi: 10.1128/jb.127.3.1529-1537.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Norrod P., Williams R. P. Effects of iron and culture filtrates on killing of Neisseria gonorrhoeae by normal human serum. Infect Immun. 1978 Sep;21(3):918–924. doi: 10.1128/iai.21.3.918-924.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Payne S. M., Finkelstein R. A. Pathogenesis and immunology of experimental gonococcal infection: role of iron in virulence. Infect Immun. 1975 Dec;12(6):1313–1318. doi: 10.1128/iai.12.6.1313-1318.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Payne S. M., Finkelstein R. A. The critical role of iron in host-bacterial interactions. J Clin Invest. 1978 Jun;61(6):1428–1440. doi: 10.1172/JCI109062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Payne S. M., Holmes K. K., Finkelstein R. A. Role of iron in disseminated gonococcal infections. Infect Immun. 1978 May;20(2):573–574. doi: 10.1128/iai.20.2.573-574.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salit I. E., Gotschlich E. C. Gonococcal color and opacity variants: virulence for chicken embryos. Infect Immun. 1978 Nov;22(2):359–364. doi: 10.1128/iai.22.2.359-364.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schoolnik G. K., Buchanan T. M., Holmes K. K. Gonococci causing disseminated gonococcal infection are resistant to the bactericidal action of normal human sera. J Clin Invest. 1976 Nov;58(5):1163–1173. doi: 10.1172/JCI108569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- WILLIAMS J. A comparison of conalbumin and transferrin in the domestic fowl. Biochem J. 1962 May;83:355–364. doi: 10.1042/bj0830355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams P. H., Warner P. J. ColV plasmid-mediated, colicin V-independent iron uptake system of invasive strains of Escherichia coli. Infect Immun. 1980 Aug;29(2):411–416. doi: 10.1128/iai.29.2.411-416.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yancey R. J., Breeding S. A., Lankford C. E. Enterochelin (enterobactin): virulence factor for Salmonella typhimurium. Infect Immun. 1979 Apr;24(1):174–180. doi: 10.1128/iai.24.1.174-180.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]