Abstract
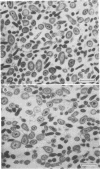
Coxiella burnetii, phase I and II, cells cultivated in the yolk sac of chicken embryos were separated from host cell components by two cycles of isopycnic Renografin gradient centrifugation. Initial steps in the purification of viable C. burnetii involved differential centrifugation and sedimentation through an aqueous solution of 30% sucrose and 7.6% Renografin. After the first, but not the second, cycle of Renografin gradient centrifugation, the cells were passed through microfilter glass filters which facilitated the removal of host components. The integrity of morphologically different cell variants was maintained during purification procedures by suspending highly purified C. burnetii in phosphate-buffered saline-sucrose solutions. C. burnetii, phases I and II, obtained by these methods appeared to be free from host cell components by serological methods while retaining morphological integrity and infectivity for yolk sacs and experimental animals. Average yields of C. burnetii were 2.83, 1.5, and 0.84 mg (dry weight) per yolk sac of the Ohio strain (phase I), 9 Mile strain (phase I), and 9 Mile strain (phase II), respectively. Recovery of phase I cells averaged about 70%, whereas the recovery of phage II cells was approximately 40%. The temporal sequence of phase I and II antibody response was demonstrated in infected and vaccinated animals. Also, no antibody response in mice and guinea pigs to yolk sac antigens was detectable after two injections of vaccine or viable cells. Importantly, this is the first report of the separation of viable phase II cells of C. burnetii free of host components.
Full text
PDF









Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Anacker R. L., McCaul T. F., Burgdorfer W., Gerloff R. K. Properties of selected rickettsiae of the spotted fever group. Infect Immun. 1980 Feb;27(2):468–474. doi: 10.1128/iai.27.2.468-474.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burton P. R., Stueckemann J., Welsh R. M., Paretsky D. Some ultrastructural effects of persistent infections by the rickettsia Coxiella burnetii in mouse L cells and green monkey kidney (Vero) cells. Infect Immun. 1978 Aug;21(2):556–566. doi: 10.1128/iai.21.2.556-566.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- COLTER J. S., BROWN R. A., BIRD H. H., COX H. R. The preparation of a soluble immunizing antigen from Q-fever rickettsiae. J Immunol. 1956 Apr;76(4):270–274. [PubMed] [Google Scholar]
- Canonico P. G., Van Zwieten M. J., Christmas W. A. Purification of large quantities of coxiella burnetii rickettsia by density gradient zonal centrifugation. Appl Microbiol. 1972 May;23(5):1015–1022. doi: 10.1128/am.23.5.1015-1022.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coolbaugh J. C., Progar J. J., Weiss E. Enzymatic activities of cell-free extracts of Rickettsia typhi. Infect Immun. 1976 Jul;14(1):298–305. doi: 10.1128/iai.14.1.298-305.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DAVIS J. L., PATRICK W. C., 3rd NEW METHOD FOR PURIFYING COXIELLA BURNETII BY DEXTRAN SULFATE PRECIPITATION FOLLOWED BY CENTRIFUGATION. Appl Microbiol. 1965 Jan;13:99–102. doi: 10.1128/am.13.1.99-102.1965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dasch G. A., Weiss E. Characterization of the Madrid E strain of Rickettsia prowazekii purified by renografin density gradient centrifugation. Infect Immun. 1977 Jan;15(1):280–286. doi: 10.1128/iai.15.1.280-286.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fiset P., Ormsbee R. A., Silberman R., Peacock M., Spielman S. H. A microagglutination technique for detection and measurement of rickettsial antibodies. Acta Virol. 1969 Jan;13(1):60–66. [PubMed] [Google Scholar]
- Genig V. A. Heterogenous populations of C. burneti and selection analysis of them. Bull Soc Pathol Exot Filiales. 1969 May-Jun;62(3):476–493. [PubMed] [Google Scholar]
- Giménez D. F. Gram Staining of Coxiella burnetii. J Bacteriol. 1965 Sep;90(3):834–835. doi: 10.1128/jb.90.3.834-835.1965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gowans E. J. An improved method of agar pelleted cells for electron microscopy. Med Lab Technol. 1973 Apr;30(2):113–115. [PubMed] [Google Scholar]
- HOYER B. H., BOLTON E. T., ORMSBEE R. A., LEBOUVIER G., RITTER D. B., LARSON C. L. Mammalian viruses and Rickettsiae; their purification and recovery by cellulose anion exchange columns has significant implications. Science. 1958 Apr 18;127(3303):859–863. doi: 10.1126/science.127.3303.859. [DOI] [PubMed] [Google Scholar]
- Howard L., Orenstein N. S., King N. W. Purification on renografin density gradients of Chlamydia trachomatis grown in the yolk sac of eggs. Appl Microbiol. 1974 Jan;27(1):102–106. doi: 10.1128/am.27.1.102-106.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- ORMSBEE R. A. A method of purifying Coxiella burnetii and other pathogenic Rickettsiae. J Immunol. 1962 Jan;88:100–108. [PubMed] [Google Scholar]
- ORMSBEE R. A., PEACOCK M. G. METABOLIC ACTIVITY IN COXIELLA BURNETII. J Bacteriol. 1964 Nov;88:1205–1210. doi: 10.1128/jb.88.5.1205-1210.1964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ormsbee R., Peacock M., Gerloff R., Tallent G., Wike D. Limits of rickettsial infectivity. Infect Immun. 1978 Jan;19(1):239–245. doi: 10.1128/iai.19.1.239-245.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- PARETSKY D., DOWNS C. M., CONSIGLI R. A., JOYCE B. K. Studies on the physiology of rickettsiae. I. Some enzyme systems of Coxiella burnetii. J Infect Dis. 1958 Jul-Aug;103(1):6–11. doi: 10.1093/infdis/103.1.6. [DOI] [PubMed] [Google Scholar]
- Peacock M. G., Ormsbee R. A., Johnson K. M. Rickettsioses of Central America. Am J Trop Med Hyg. 1971 Nov;20(6):941–949. doi: 10.4269/ajtmh.1971.20.941. [DOI] [PubMed] [Google Scholar]
- Philip R. N., Casper E. A., Burgdorfer W., Gerloff R. K., Hughes L. E., Bell E. J. Serologic typing of rickettsiae of the spotted fever group by microimmunofluorescence. J Immunol. 1978 Nov;121(5):1961–1968. [PubMed] [Google Scholar]
- REED C. F., WENTWORTH B. B. Q fever studies in Ohio. J Am Vet Med Assoc. 1957 May 15;130(10):458–461. [PubMed] [Google Scholar]
- REYNOLDS E. S. The use of lead citrate at high pH as an electron-opaque stain in electron microscopy. J Cell Biol. 1963 Apr;17:208–212. doi: 10.1083/jcb.17.1.208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silverman D. J., Fiset P., Wisseman C. L., Jr Simple, differential staining technique for enumerating rickettsiae in yolk sac, tissue culture extracts, or purified suspensions. J Clin Microbiol. 1979 Mar;9(3):437–440. doi: 10.1128/jcm.9.3.437-440.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spurr A. R. A low-viscosity epoxy resin embedding medium for electron microscopy. J Ultrastruct Res. 1969 Jan;26(1):31–43. doi: 10.1016/s0022-5320(69)90033-1. [DOI] [PubMed] [Google Scholar]
- WISSEMAN C. L., Jr, JACKSON E. B., HAHN F. E., LEY A. C., SMADEL J. E. Metabolic studies of rickettsiae. I. The effects of antimicrobial substances and enzyme inhibitors on the oxidation of glutamate by purified rickettsiae. J Immunol. 1951 Aug;67(2):123–136. [PubMed] [Google Scholar]
- Weiss E., Coolbaugh J. C., Williams J. C. Separation of viable Rickettsia typhi from yolk sac and L cell host components by renografin density gradient centrifugation. Appl Microbiol. 1975 Sep;30(3):456–463. doi: 10.1128/am.30.3.456-463.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weiss E. Growth and physiology of rickettsiae. Bacteriol Rev. 1973 Sep;37(3):259–283. doi: 10.1128/br.37.3.259-283.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wiebe M. E., Burton P. R., Shankel D. M. Isolation and characterization of two cell types of Coxiella burneti phase I. J Bacteriol. 1972 Apr;110(1):368–377. doi: 10.1128/jb.110.1.368-377.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams J. C. Adenine nucleotide degradation by the obligate intracellular bacterium Rickettsia typhi. Infect Immun. 1980 Apr;28(1):74–81. doi: 10.1128/iai.28.1.74-81.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams J. C., Peterson J. C. Enzymatic activities leading to pyrimidine nucleotide biosynthesis from cell-free extracts of Rickettsia typhi. Infect Immun. 1976 Aug;14(2):439–448. doi: 10.1128/iai.14.2.439-448.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams J. C., Weiss E. Energy metabolism of Rickettsia typhi: pools of adenine nucleotides and energy charge in the presence and absence of glutamate. J Bacteriol. 1978 Jun;134(3):884–892. doi: 10.1128/jb.134.3.884-892.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]