Abstract
Epidermal growth factor receptor (EGFR) exemplifies the family of receptor tyrosine kinases that mediate numerous cellular processes including growth, proliferation and differentiation. Moreover, gene amplification and EGFR mutations have been identified in a number of human malignancies, making this receptor an important target for the development of anticancer drugs. In addition to ligand-dependent activation and concomitant tyrosine phosphorylation, EGFR stimulation results in the localized generation of H2O2 by NADPH-dependent oxidases. In turn, H2O2 functions as a secondary messenger to regulate intracellular signaling cascades, largely through the modification of specific cysteine residues within redox-sensitive protein targets, including Cys797 in the EGFR active site. In this review, we highlight recent advances in our understanding of the mechanisms that underlie redox regulation of EGFR signaling and how these discoveries may form the basis for development of new therapeutic strategies to target this and other H2O2-modulated pathways.
Activation of receptor tyrosine kinases (RTKs) by their respective extracellular ligands (e.g. growth factors) initiates signaling cascades that regulate cellular proliferation, differentiation, migration, and survival (1). Among the 58 RTKs identified in the human genome, epidermal growth factor (EGF) receptor (EGFR) has served as the quintessential model for understanding RTK biology in physiological signaling and cancer. EGFR, also known as HER1 (or erbB1), is a transmembrane protein grouped into a subfamily that consists of three additional, closely related receptors: HER2 (erbB2), HER3 (erbB3), and HER4 (erbB4). EGFR is comprised of a glycosylated extracellular ligand-binding domain, a transmembrane domain, and an intracellular domain containing its tyrosine kinase core. In response to ligand (e.g. EGF) binding, EGFR forms homo- or heterodimers with other HER family members, followed by autophosphorylation of key tyrosine residues located within the tyrosine kinase domain (2). Once activated, EGFR relays the signal through a variety of downstream intracellular signaling cascades, including the Ras/mitogen activated protein kinase (MAPK) pathway or the phosphatidylinositol 3′ kinase (PI3K)/Akt pathway. Since its discovery (3-6), EGFR has been widely studied in regards to its physiologic and pathological settings. In particular, EGFR and related family members have been found to be mutated or amplified in a number of human lung and breast cancers, rendering them an attractive target for the development of therapeutics (7-9).
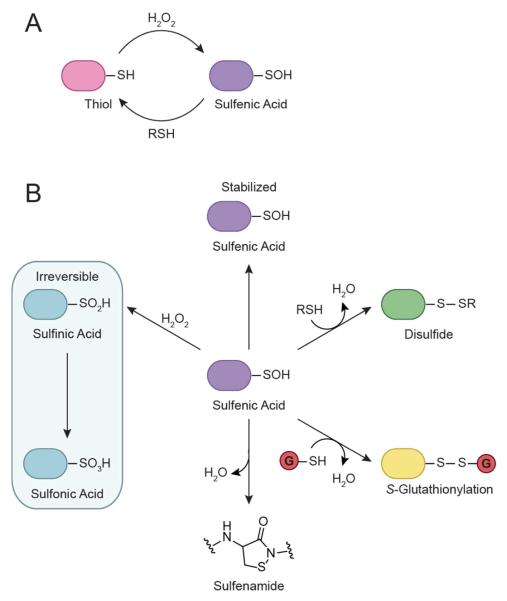
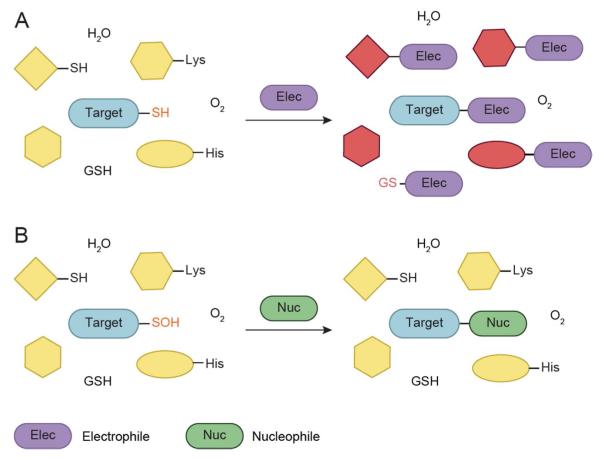
Beginning in the 1990s, data from several research groups demonstrated that EGF binding to EGFR triggered the production of endogenous hydrogen peroxide (H2O2) in cells (Fig. 1) (10, 11). Moreover, it was also established that other growth factors [FGF (12), PDGF (13), VEGF (14), insulin (15)], cytokines [TNF-α (12, 16), interleukin-1 (16)], and angiotensin II (17) stimulate H2O2 generation. Although chronic exposure to high concentrations of H2O2 can lead to a cellular condition known as oxidative stress, cells can also utilize H2O2 as a secondary messenger to regulate physiological signal transduction (18-21). H2O2 modulates signaling pathways largely through the modification of specific cysteine residues located within redox-sensitive protein targets (Fig. 2A). The direct product of the reaction between H2O2 and a protein thiolate (RS−) is sulfenic acid (RSOH). Such protein “sulfenylation” is reversible by thiol-disulfide oxidoreductases of the thioredoxin (Trx) superfamily throughout the intracellular milieu and thus, constitutes a facile switch for modulating protein function, akin to phosphorylation. Associated regulatory redox modifications of cysteine include sulfenamides, nitrosothiols, disulfides, sulfinic, and sulfonic acids (Fig. 2B). Over the last several years, an increasing number of studies have demonstrated important functional roles for protein sulfenic acids in cell signaling (22-26). In particular, kinases and phosphatases are both known to undergo cysteine-based redox regulation (27, 28) and the collective efforts of many researchers have established that these enzymes are regulated by endogenous H2O2 produced during EGF-mediated cell signaling (29).
Figure 1.
Timeline outlining key events and discoveries relating to redox regulation of EGFR signaling through cysteine oxidation.
Figure 2.
Oxidative modification of cysteine residues by hydrogen peroxide (H2O2). (A) The initial reaction product of a thiolate with H2O2 yields sulfenic acid (RSOH). This modification, also known as sulfenylation, is reversible and can be directly reduced back to the thiol form or indirectly through disulfide bond formation. (B) Sulfenic acids can be stabilized by the protein microenvironment and/or undergo subsequent modification. For example, they can condense with a second cysteine in the same or different protein to generate a disulfide bond. Alternatively, reaction with the low molecular weight thiol glutathione (GSH, red circle) affords a mixed disulfide through a process known as S-glutathionylation. In a few proteins, such as PTP1B, nucleophilic attack of a backbone amide on RSOH results in sulfenamide formation. Sulfenyl groups can also oxidize further to the sulfinic (RSO2H) and/or sulfonic (RSO3H) acid form under conditions of high oxidative stress.
In this review, we present a historical overview of EGFR and ligand-mediated production of H2O2 as well as the molecular mechanisms involved in redox regulation of this signaling pathway (Fig. 1). We begin by highlighting early studies linking EGFR activation to EGF-induced H2O2 production. Subsequently, we address the effects of redox modulation on two major pathways downstream of EGFR, Ras/MAPK and PI3K/Akt. Additionally, we discuss the interplay between H2O2-mediated inactivation of protein tyrosine phosphatases (PTPs) and kinase activation on net cellular levels of tyrosine (Tyr) phosphorylation. Key enzymes involved in the generation and metabolism of H2O2 within EGFR signaling pathways will also be covered. Finally, we focus on recent examples from the literature demonstrating direct oxidation and regulation of EGFR by H2O2 and how these discoveries may form the basis for development of new therapeutic strategies to target this and other H2O2-modulated pathways.
Early Evidence for EGF-Mediated H2O2 Production and Redox Regulation of EGFR Signaling
In 1995, Gamou and Shimizu published the first report to suggest a connection between H2O2 and EGFR. In this study, the authors examined the effect of exogenously added H2O2 on EGFR phosphorylation (30). EGFR-hyper producing human squamous carcinoma NA cells treated with H2O2 (0 – 1 mM) exhibited an increase in the incorporation of [32P]-phosphate, albeit at half the signal observed for EGF-stimulated cells. On the basis of data obtained from tryptic phosphopeptide mapping, this discrepancy was attributed to the fact that H2O2 might preferentially enhance EGFR Tyr phosphorylation, whereas EGF stimulation would trigger both serine/threonine (Ser/Thr) as well as Tyr receptor phosphorylation.
During this same time frame (i.e. the mid-90s), the role of H2O2 expanded beyond the traditional view as “toxic byproducts of aerobic metabolism” and began to emerge as secondary messengers in physiological cell signaling. In order for H2O2 to serve as a signaling molecule, its concentration must increase rapidly above the steady-state threshold (i.e., high nanomolar to low millimolar) and remain elevated long enough for it to oxidize protein effectors (31). In one of the earliest examples, platelet-derived growth factor (PDGF) was shown to induce endogenous H2O2 generation, which is correlated with enhanced Tyr phosphorylation, activation of MAPK pathways, DNA synthesis, and chemotaxis (13). These results suggested that H2O2 might act as a signaling molecule generated in response to growth factor stimulation. Soon thereafter, Rhee and colleagues reported that addition of EGF to EGFR-overexpressing human epidermoid carcinoma A431 cells significantly elevated levels of intracellular reactive oxygen species (ROS) (10) as measured by 2′,7′-dichlorofluorescein diacetate (DCFH-DA) (32). Enzymes such as catalase, peroxiredoxins (Prxs), and glutathione peroxidases (Gpxs) scavenge endogenous H2O2 by catalyzing the dismutation (catalase) or reduction (Prxs, Gpxs) of H2O2 (33, 34). Introduction of exogenous catalase in EGF-stimulated A431 cells by electroporation attenuated the intracellular build-up of ROS, suggesting that H2O2 was the major ROS involved in EGF-dependent signal transduction (10). EGF-induced increases in Tyr phosphorylation levels of PLC-γ1, a well-characterized physiological substrate of EGFR, were also blunted by catalase incorporation. Although the precise role and/or target(s) of H2O2 generated for EGFR signaling were not directly addressed in this study, the authors proposed that inhibition of cysteine-dependent PTPs by H2O2 may be required to increase the steady-state level of protein Tyr phosphorylation. This landmark contribution by Rhee and coworkers set the stage for delineating the molecular details underlying redox regulation of EGFR signaling.
In a separate study, Goldkorn et al. reported that exogenous H2O2 stimulated EGFR tyrosine kinase activity and increased receptor half-life (35). Experiments performed in A549 human lung adenocarcinoma epithelial cells and isolated membrane fractions showed an increase in EGFR Tyr autophosphorylation levels when treated with H2O2 (0 – 200 PM), and also markedly enhanced receptor activation in conjunction with the native EGF ligand. Pulse-chase experiments with [35S]-methionine revealed that EGFR half-life was ~8 h when treated with EGF, whereas H2O2 treatment extended receptor half-life to 18 h (combined treatment with EGF and H2O2 yielded a half-life of 12 h). Additionally, two-dimensional phosphoamino acid analysis corroborated earlier findings (30) that the phosphorylation site distribution was shifted predominantly towards Tyr after exposure to H2O2. On the basis of these data, the authors of this study postulated that EGF- and H2O2-induced receptor activation may have separate functions and represent an alternate mechanism by which EGFR signaling can be tuned in parallel to treatment with its native ligand.
The Effect of H2O2 on Signaling Networks Downstream of EGFR
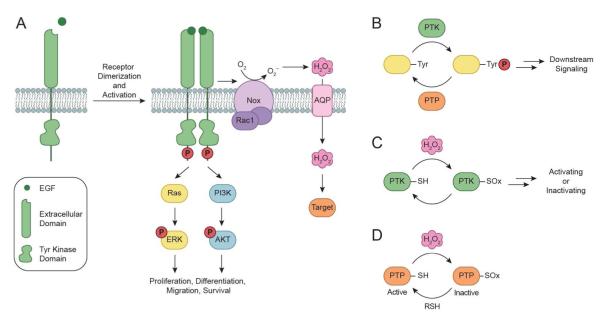
After ligand binding, EGFR transmits activation signals to prominent downstream cascades, including the Ras/MAPK and PI3K/Akt pathways (Fig. 3A). In addition to the overall increase in Tyr autophosphorylation of EGFR, the endogenous generation of H2O2 also appears to modulate the activity of these two signaling routes. Receptor activation initiates the Ras-Raf-MEK-Erk1/2 signaling module through recruitment of Src homology 2 domain-containing (SHC) adaptor protein, growth factor receptor-bound protein 2 (Grb2), and a guanine nucleotide exchange protein (SOS) to form the SHC-Grb2-SOS complex, a process stimulated by elevated intracellular H2O2 (36). Once associated with the receptor, SOS facilitates guanine nucleotide exchange to activate Ras, which subsequently activates a kinase cascade including Raf (MAP3K), MEK (MAP2K), and Erk1/2 (MAPK). Oxidative stress is known to influence the MAPK signaling pathways, but little is known about the molecular mechanisms responsible for such effects (37). For example, Erk1/2 is activated in response to exogenous H2O2, which enhances cell survival after oxidant injury (38, 39). On the other hand, it is not known whether the activity of Erk1/2 kinase is directly modulated by ROS.
Figure 3.
EGFR signaling pathways and general mechanisms for thiol-based redox modulation of signaling proteins. (A) EGF binding to EGFR induces receptor dimerization, followed by autophosphorylation of tyrosine (Tyr) residues (red circles) within its cytoplasmic domain. In turn, these phosphorylated Tyr residues serve as docking sites for associating proteins to activate a number of downstream signaling cascades. Two such pathways, Ras/ERK and PI3K/AKT, are shown here for simplicity. The EGF-EGFR interaction also triggers the assembly and activation of NADPH oxidase (Nox) complexes, followed by subsequent production of H2O2 through spontaneous dismutation or action of SOD. Once formed, endogenous H2O2 may pass through specific aquaporin (AQP) channels and/or diffuse across the membrane to reach the intracellular cytosol. Transient increases in H2O2 leads to the oxidation of local redox targets. (B) Model for redox-dependent signal transduction. Protein tyrosine kinases (PTKs) catalyze the transfer of γ-phosphoryl groups from ATP to tyrosine hydroxyls of proteins, whereas protein tyrosine phosphatases (PTPs) remove phosphate groups from phosphorylated tyrosine residues. PTPs function in a coordinated manner with PTKs to control signaling pathways to regulate a diverse array of cellular processes. (C) Regulatory cysteines in protein kinases can undergo oxidation/reduction to modulate their function. Depending on the kinase, redox modifications can stimulate or inhibit function. (D) Oxidation of the conserved active site cysteine residue in PTPs inactivates these enzymes, and can be restored by reducing the oxidized residue to its thiol form. SOx: oxidized cysteine.
EGFR also transmits signals through the PI3K/Akt pathway (40). In response to EGF stimulation, PI3K increases the levels of phosphatidylinositol 3,4,5-trisphosphate (PIP3), which leads to recruitment and activation of serine/threonine kinase, Akt at the plasma membrane (41, 42). Activity of the PI3K/Akt pathway is balanced by action of the opposing lipid phosphatase, PTEN, which will be discussed in later sections of this review. In cells overexpressing the NADPH oxidase isoform Nox1, increased endogenous H2O2 production elevates intracellular PIP3 levels and consequently abrogates the ability of PTEN to promote downstream signaling (43). EGFR-dependent activation of Akt has also been shown to enhance cell survival during oxidative stress-induced apoptosis, analogous to Erk1/2 (44). Interestingly, structural analysis by x-ray crystallography has revealed that Akt2 can form an intramolecular disulfide bond between two cysteines in the activation loop, which inhibits the kinase (45). Overexpression of glutaredoxin (Grx) has been shown to protect Akt against H2O2-induced oxidation, resulting in sustained phosphorylation of Akt and inhibition of apoptosis (46). A more recent study is indicative of isoform-specific regulation of Akt by PDGF-induced H2O2 (47). In particular, the authors demonstrate that Akt2 Cys124 undergoes sulfenic acid modification during growth factor signaling, which inactivates the kinase.
EGFR activation can also occur through a process known as trans-activation. In this scenario, ligand-inaccessible RTKs may still initiate downstream signaling in lieu of ligand-receptor induced activation. Production of H2O2 induced by other ligands such as angiotensin II activates c-Src, a redox-regulated kinase (48). In turn, EGFR undergoes activation through c-Src initiated receptor Tyr phosphorylation to propagate downstream signaling (48-50). Neighboring EGFR kinases may then undergo activation in a lateral-based mechanism. Trans-activation represents another level by which EGFR activity can be modulated by H2O2. In addition, other studies have implicated oxidative inactivation of PTPs in promoting EGFR trans-activation (51).
The Interplay of Reversible Tyrosine Phosphorylation and Redox-Dependent Signaling
Regulation of tyrosine phosphorylation depends on the delicate balance between the action of protein kinases and phosphatase (Fig. 3B, and C). In EGFR signaling, the coordinated regulation of this equilibrium allows for rapid response to changing growth factor levels, whereas dysregulated kinase and phosphatase activity can have severe pathological consequences such as cancer, diabetes, and inflammation (52, 53). Because the balance of these two opposing forces is a central event in cell signaling, it is not surprising that phosphatase as well as kinase activities are tightly regulated at several levels, including cysteine-based redox modulation. In large part, owing to early studies carried out by Denu and Tanner (54), attention rapidly focused on PTPs as the direct targets of H2O2 (Fig. 3D). The PTP superfamily contains a signature motif, (I/V)HCXXGXXR(S/T), which includes an invariant cysteine residue that functions as a nucleophile in catalysis. The catalytic cysteine of PTPs is characterized by a low pKa value ranging from 4.6 – 5.5 due to the unique electrostatic environment of the active site, which also renders the enzyme susceptible to inactivation by reversible oxidation (55, 56). Second-order rate constants for oxidation of the PTP cysteine thiolate by H2O2 have been measured in vitro and range from 10 to 160 M−1 s−1 (54, 57, 58).
Protein tyrosine phosphatase 1B (PTP1B) functions as a negative regulator of the EGFR signaling pathway by directly targeting phosphorylated tyrosine residues that control signaling output (59-62). Given that PTP1B is localized exclusively to the cytoplasmic face of the endoplasmic reticulum (ER), it has been proposed to dephosphorylate activated EGFR at sites of contact between the ER and plasma membranes or upon trafficking of internalized EGFR in close proximity to the ER (63, 64). Studies reported in 1998 provided the first indication that EGF-mediated H2O2 production is correlated with oxidation and inactivation of PTP1B (65). Incorporation of radiolabeled iodoacetic acid (IAA) – a sulfhydryl-modifying reagent that reacts with active site Cys215 of PTP1B (54) – was decreased in PTP1B after EGF stimulation of A431 cells, consistent with oxidation of this essential residue. Conversely, treatment with dithiothreitol (DTT), thioredoxin (Trx), or glutaredoxin (Grx) as reductants readily reversed PTP1B inhibition. Interestingly, assays with recombinant protein indicated that the Trx system functioned more efficiently as an electron donor for PTP1B reactivation, as compared to Grx or glutathione (GSH), suggesting that Trx may function as the physiological reductant (54).
The phosphatase and tensin homologue (PTEN) is another PTP known to maintain a closely intertwined relationship with EGFR. PTEN exhibits dual protein and lipid phosphatase activity and functions as a negative regulator of the PI3K/Akt signaling pathway (66), one of the two major signaling routes downstream of EGFR. PTEN contains five cysteine residues in its catalytic domain and undergoes reversible inactivation by H2O2 (67). Site-directed mutagenesis and mass spectrometry indicated that Cys124 was the primary target of H2O2, yielding a sulfenic acid intermediate that condenses with Cys71 to form an intramolecular disulfide bond (68). Cellular studies also point to a connection between growth factor-induced generation of H2O2 and reversible inactivation of PTEN. For example, EGF stimulation of cells results in elevated levels of oxidized PTEN in lysates, as indicated by an electrophoretic mobility shift assay that reports on disulfide formation (69). In an alternative approach, protein thiols are alkylated by N-ethylmaleimide (NEM) in lysates generated from growth factor stimulated cells. Reversibly oxidized protein thiols are then reduced with DTT and alkylated with biotin-conjugated maleimide. This method was used to examine reversible oxidation of PTEN in response to EGFR activation (69). Together, these studies indicate that EGF-induced activation of PI3K correlates with inactivation of the opposing phosphatase, PTEN. In this way, PTEN inactivation by endogenous H2O2 serves as a positive feedback loop to enhance PIP3 accumulation/Akt activation during EGFR signaling.
SHP2 and DEP-1 represent two additional phosphatases that have been shown to interact with EGFR. SHP2 directly associates with EGFR through its SH2 domains and regulates receptor interactions with downstream signaling components such as Ras (70). Co-localization of SHP2 with EGFR occurs at low nanomolar concentrations (4 ng/ml) of EGF, and was identified as the most sensitive PTP in response to signal-derived H2O2 (71). It is unknown if DEP-1 co-localizes with EGFR intracellularly, albeit evidence implicates in vitro oxidation of DEP-1 (72).
Generation and Metabolism of H2O2 During EGFR Signaling
The family of NADPH oxidase (Nox) enzymes and their dual oxidase counterparts (Duox) generate superoxide by transferring electrons from cytosolic nicotinamide adenine dinucleotide phosphate (NADPH) to molecular oxygen (73). Once it is generated, superoxide is dismutated spontaneously (~105 M−1 s−1 at pH 7) or enzymatically by superoxide dismutase (SOD; ~109 M−1 s−1) to H2O2 and molecular O2 (74, 75). The prototypical Nox isoform, Nox2 (also known as gp91phox) was originally identified and characterized in macrophages and neutrophils, where it functions as an integral part of the innate immune system (76). The active form of Nox2 exists as a multi-subunit complex, consisting of the membrane-bound cytochrome b558 (gp91phox and p22phox), several cytosolic proteins (p47phox, p40phox, p67phox), and the small GTPase Rac1. Since the initial discovery of Nox2, other Nox homologues (Nox 1-5 and Duox 1-2) have been identified in almost every cell type, localized both to the plasma membrane (where they produce superoxide extracellularly) as well as intracellular organelles, and serve as major sources of H2O2 for signaling (77-79).
Several Nox isoforms play a critical role in EGFR-mediated signaling cascades. For example, Park et al. demonstrated that the PI3K pathway constitutes a positive feedback loop for Nox1 activation in growth factor-stimulated cells (43). This study demonstrated that the Rac-guanine nucleotide exchange factor (GEF) βPix is required for and also augments EGF-induced generation of H2O2. In this loop, β-Pix and activated Rac1 bind to the C-terminal region of Nox1, relieving auto-inhibitory constraints. Independent studies have also shown that phosphorylation of Nox activator 1 (NOXA1) on Ser282 by Erk1/2 kinases and on Ser172 by protein kinases C and A decreases Rac1 binding to down-regulate Nox1 (80, 81).
Nox4 is an isoform that regulates the activity of internalized EGFR. Keaney and colleagues report that Nox4 is localized to the ER in vascular endothelial cells where it appears to regulate the activity of PTP1B in a spatially dependent manner (82). Nox4-dependent oxidation of PTP1B required co-localization of both proteins in the ER, as shown by targeting PTP1B to the cytoplasm. Importantly, the study also demonstrated that Nox4-dependent oxidation and inactivation of PTP1B is correlated with reduced phosphorylation of EGFR in proximity to the ER. The significance of co-localization was also verified by ER-targeting of the antioxidant enzyme, catalase. Lastly, EGF-stimulation of A431 cells leads to specific complex formation between Nox2 and EGFR, as demonstrated by co-immunoprecipitation experiments (71).
Superoxide dismutase 1 (SOD1) is another enzyme involved in redox mediation of growth factor signaling. SOD1 is an abundant copper/zinc enzyme located in the cytoplasm and belongs to the SOD family of enzymes. Other members of this family include mitochondrial SOD2 (manganese) and extracellular SOD3 (copper/zinc). Although superoxide can spontaneously dismutate to form H2O2, the second-order rate constant of the reaction is enhanced over 10,000-fold by SOD (74, 75). Indeed, SODs catalyze the conversion of superoxide into H2O2 and molecular O2 to maintain superoxide at low steady-state concentration (~10−10 M) (83, 84). Inhibition of SOD1 by the tetrathiomolybdate inhibitor, ATN-224, increases the intracellular concentration of superoxide at the expense of H2O2 production, thereby attenuating EGFR and Erk1/2 phosphorylation (85).
Membrane transport represents another important mechanism to modulate endogenous H2O2 produced during EGFR signaling. Early studies demonstrated that EGF stimulation of cells leads to a rapid increase in the concentration of extracellular H2O2 and that the addition of catalase to culture medium was sufficient to inhibit EGFR autophosphorylation (86, 87). These findings raise the question of how extracellularly generated H2O2 could mediate intracellular signaling pathways. Although it had been largely assumed that H2O2 could diffuse across cellular membranes, more recent evidence indicates that H2O2 may preferentially enter the cell through specific plasma membrane aquaporin channels (88-90). Collectively, these studies suggest that the number or type of aquaporins expressed on the cell surface might modulate the level of intracellular H2O2 available for signaling.
Efforts have also focused on delineating mechanisms to regulate intracellular H2O2 levels during EGF-mediated signaling. In this regard, superoxide dismutases, catalase, and other peroxidases all function to protect cells against undue oxidative stress. Prxs are a family of thiol-based peroxidases that catalyze the dismutation of H2O2 into water and molecular oxygen (91). Prx possesses two cysteines that transiently oxidize to form a disulfide as it metabolizes H2O2. The disulfide in Prx is subsequently reduced by the protein disulfide reductase, Trx, to complete the catalytic cycle. Overexpression of the PrxII isoform reduces EGF-induced intracellular H2O2 levels (92). Another study demonstrated that PrxII overexpression is associated with a decrease in EGF-induced cellular PIP3, whereas a dominant negative (DN) form of PrxII increased PIP3 levels, presumably by H2O2 modulation of PTEN redox state (69). Similarly, overexpression of another antioxidant enzyme, glutathione peroxidase-1 (Gpx1), decreased Tyr phosphorylation of EGFR, activation of Akt, and cellular proliferation (93). In addition to SODs and peroxidases, cells rely on the glutaredoxin/glutathione (Grx/GSH), Trx/thioredoxin reductase (Trx/TrxR), and glutathione/glutathione reductase (GSH/GSR) buffering systems. For example, EGFR signaling is associated with subcellular compartmental oxidation of Trx1. Dean and colleagues have measured the redox states of cytosolic and nuclear Trx1 and mitochondrial Trx2 using redox Western blot methods during endogenous H2O2 production induced by EGF signaling (94). Interestingly, results from this study showed that only the cytoplasmic Trx1 pool undergoes significant oxidation in response to growth factor treatment. Furthermore, the GSH/GSSG redox couple, which was also examined in this study, did not undergo oxidation. This work suggests that physiological H2O2 generation in response to EGF signaling is specifically associated with oxidation of the Trx1 system and not the GSH system. However, whether Trx1 oxidation is part of the signaling mechanism itself or simply results from peroxidase-dependent termination of the redox signal remains an active area of investigation.
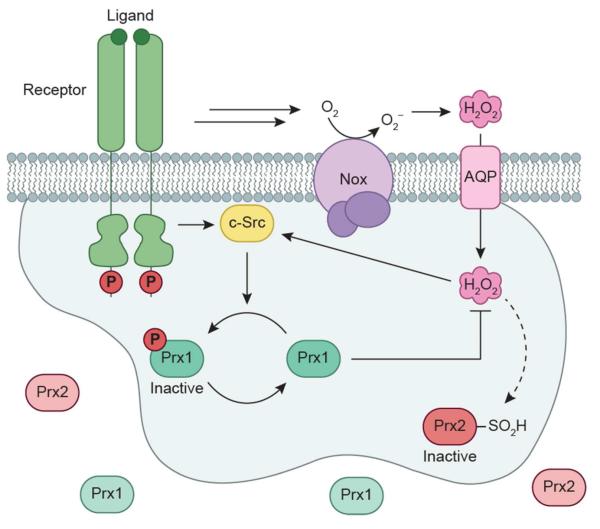
The estimated intracellular steady-state concentration of H2O2 hovers in the low-nanomolar to low-micromolar range (31). Then again, these estimates assume that H2O2 is uniformly distributed throughout the cell. Given that the source of H2O2 produced for EGF signaling (e.g. Nox enzymes) are localized to specific regions of the cell, it stands to reason that signal mediated changes in H2O2 concentration may not be homogenous throughout the cell. Rather, the oxidant concentration near a source of generation must achieve high local concentrations to function effectively as a second messenger. A growing body of research has focused on delineating the mechanisms that facilitate localized rises in intracellular H2O2 during growth factor signaling. Although the cell contains millimolar concentrations of GSH, it reacts too slowly with H2O2 to provide much buffering capacity (95). By contrast, Prxs are extremely efficient at H2O2 elimination, reducing H2O2 at second-order rate constants of 105-108 M−1s−1 (96, 97). Recent work reported by the Rhee laboratory has shown that membrane localized PrxI can be deactivated by phosphorylation in EGF-stimulated cells and in mice during wound healing (98). Knockdown experiments suggested that c-Src kinase is at least partially responsible for PrxI phosphorylation. RTK activation (e.g. PDGFR) can also lead to overoxidation of the PrxII isoform catalytic cysteine to sulfinic acid, resulting in a transiently inactivated protein (98). Collectively, these studies demonstrate that selective inactivation of PrxI and PrxII allows for transient H2O2 accumulation around plasma membranes where signaling components are concentrated, while simultaneously preventing toxic accumulation of ROS elsewhere in the cell during EGF signaling (Fig. 4). Other mechanisms to modulate localized redox buffering capacity surely await discovery.
Figure 4.
Redox regulation of peroxiredoxins (Prxs) during EGFR signaling. Receptor activation results in localized phosphorylation and inactivation of peroxiredoxin I (PrxI) by PTKs, such as the redox-regulated cytoplasmic Src (c-Src). Deactivation of PrxI diminishes the redox-buffering capacity adjacent to the cell membrane, allowing for a transient and localized increase in H2O2 levels for signal transduction. Additionally, elevated H2O2 concentrations can inactivate Prx2 by oxidation of its catalytic cysteine to sulfinic acid.
Regulation of Intrinsic EGFR Tyrosine Kinase Activity Through Cysteine Oxidation
As outlined above, a growing body of evidence demonstrates that EGFR activity and downstream signaling pathways are regulated by redox-based mechanisms. Up until this point we have only considered downstream events that enhance the overall extent of EGFR activation, such as PTP inactivation. We have also highlighted key regulatory themes in redox signaling, including co-localization of sources/targets of H2O2 and modulation of local redox buffering capacity. Until recently, there was scant evidence to indicate that the intrinsic tyrosine kinase activity of EGFR itself might be regulated by endogenous H2O2 produced during cell signaling (99-101). In the section below, we recount recent work from our own laboratory, which has demonstrated that EGFR is directly modulated by endogenous H2O2, as well as studies from other groups that hint at the possibility of modification by reactive nitrogen species (RNS).
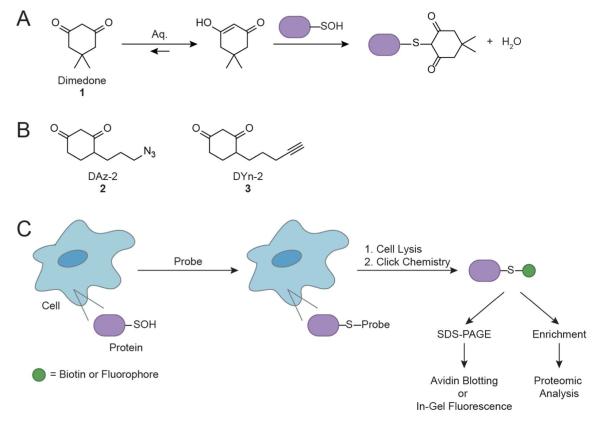
Previous data from our group demonstrated that breast cancer cells associated with increased expression of EGFR is correlated with a substantial increase in protein sulfenylation (102). Prompted by this observation, we conducted a more detailed investigation of EGF-induced protein sulfenylation in A431 cells (71). Facilitating these studies, we have recently described a new technology that allows for the detection and visualization of sulfenylation proteins within intact cells (103-106), thereby circumventing concerns associated with the analysis of lysates/homogenates including limited spatial-temporal resolution and oxidation artifacts inherent to the lysis procedure (107). Inspired by earlier work demonstrating the selective reaction of 5,5-dimethyl-1,3,-cyclohexanedione (commercially known as dimedone) with protein sulfenic acids (108) (Fig. 5A), we developed a suite of bi-functional probes that contain a membrane-permeable analog of dimedone coupled to an azide or alkyne chemical handle (Fig. 5B). An orthogonally functionalized biotin or fluorescent tag can be appended post-cell labeling for detection via the Staudinger ligation (109) or Huisgen [3+2] cycloaddition also known as click chemistry (110). Overall, this general approach provides a facile method for monitoring protein sulfenylation directly in living cells (Fig. 5C).
Figure 5.
General strategy for detecting protein cysteine sulfenylation (RSOH) in cells. (A) Chemoselective reaction between sulfenic acid and 5,5-dimethyl-1,3-cyclohexanedione (dimedone, 1). (B) Azide and alkyne-functionalized small-molecule probes for trapping and tagging protein sulfenic acids include DAz-2 (2) and DYn-2 (3). (C) Detection of protein sulfenic acids in living cells. Target cells are incubated with cell-permeable probes to trap and tag protein sulfenic acids in situ. In subsequent steps, lysates are prepared and tagged proteins are further elaborated by attachment of biotin or fluorescence labels via click chemistry and enables detection by Western blot or in-gel fluorescence. Alternatively, biotinylated proteins may be enriched for proteomic analysis.
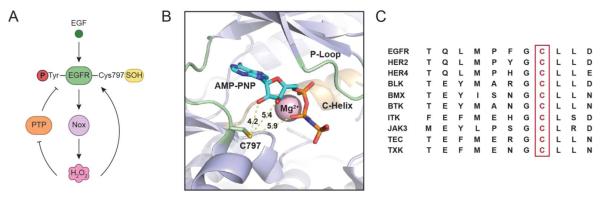
Employing this method, we demonstrated that EGFR undergoes sulfenylation in response to EGF addition, even at low-nanomolar concentrations of growth factor (71) (Fig. 6A). Reciprocal immunoprecipitation analysis also showed that EGFR and Nox2 became associated in an EGF-dependent fashion (71). The intracellular kinase domain of EGFR contains six cysteine residues, one of which (Cys797) is located in the ATP-binding pocket (Fig. 6B) and is conserved among nine additional receptor and non-receptor tyrosine kinases (Fig. 6C) (111). Given its active site location and conservation, we hypothesized that Cys797 might be preferentially targeted by endogenous H2O2. Of particular relevance, this residue is selectively targeted by irreversible EGFR inhibitors, such as afatinib, extensively used in basic research and clinical trials for breast and non-small cell lung cancers (112, 113). Consistent with this proposal, pre-treatment of cells with inhibitors that irreversibly modify Cys797 prevented sulfenylation of EGFR. Mass spectrometry was subsequently used to verify Cys797 as the specific site of oxidation. Finally, oxidation of Cys797 increased EGFR kinase activity by approximately five-fold. To put these findings into context, a comparable degree of stimulation is observed for L858R and T790M oncogenic EGFR mutations (114-116). Beyond the A431 cell line model, recent studies in our group also indicate that wild type and several activated mutant EGFR kinases undergo sulfenylation in both lung and breast tumors (T.H.T. and K.S.C., unpublished data). Hence, it appears that oxidation of specific residues in PTPs (catalytic Cys) and EGFR (Cys797) both contribute to an increase in downstream signaling (Fig. 6A). Current work is directed toward understanding the molecular mechanism by which sulfenylation of EGFR Cys797 enhances kinase activity. The proximity of Cys797 to the ATP ligand as well as the C-helix and activation segment raises the possibility of transition state stabilization and/or destabilization of its auto inhibited conformation (116).
Figure 6.
Model for H2O2-dependent regulation of EGFR tyrosine kinase activity. (A) Binding of EGF induces production of H2O2 through Nox2. Nox-derived H2O2 directly modifies EGFR cysteine (Cys797) to sulfenic acid in the active site, which enhances its tyrosine kinase activity. Endogenous H2O2 can also oxidize and deactivate localized PTPs, leading to a net increase in EGFR phosphorylation. (B) Crystal structure of the EGFR kinase domain (PDB 3GT8) bound to AMP-PNP, a hydrolysis resistant ATP analog, and Mg2+. Dashed yellow lines and accompanying numbers indicate the distance (Å) between the γ-sulfur atom of Cys797 and key substrate functional groups. Note also that Cys797 can adopt different rotamers and sulfenylation of this residue may enhance its ability to participate in electrostatic and hydrogen-bonding interactions with its substrate. (C) Abbreviated sequence alignment of EGFR and nine other kinases that harbor a cysteine at the structural position that corresponds to Cys797 (adapted from ref. 112).
We have also applied our chemical biology approach to monitor global changes in EGF-dependent protein sulfenylation (71). Interestingly, growth factor addition led to widespread changes in protein sulfenylation within cells. EGF induced cysteine oxidation in a dose- and time-dependent manner and was accompanied by concomitant fluxes in intracellular H2O2. Additional experiments showed that treatment with cell-permeable PEGylated catalase attenuated EGF associated changes in protein sulfenylation, underscoring the importance of endogenous H2O2. Although sulfenylation of EGFR (and several PTPs) were the focus our recent study (71), many protein targets of EGF-induced endogenous H2O2 remain to be identified and will necessitate large-scale proteomic analysis.
Remarkably, a number of studies indicate that EGFR may also undergo modulation by RNS at cysteine residues distinct from Cys797. Reaction of cysteine thiols with RNS, such as nitric oxide (NO), generates nitrosothiols (S-NO). This process, known as S-nitrosylation, is a well-established reversible post-translational cysteine modification and has been implicated in proliferative and anti-proliferative cellular effects (117). The approach most often used to identify protein nitrosothiols is known as the biotin switch technique (BST) (118). The BST is an indirect method whose success relies heavily on the alkylation of free thiols and the selectivity/efficiency of the ascorbate reducing agent towards nitrosothiols (119, 120). Nonetheless, this method was utilized in two separate studies reporting S-nitrosylation of EGFR. Treatment of several cell types with one millimolar of an exogenous nitric oxide (NO) donor 1,1-diethyl-2-hydroxy-2-nitrosohydrazine (DEA-NO) inhibited EGFR autophosphorylation (121, 122). Serine mutation of EGFR Cys166, located in the extracellular domain, rendered the receptor resistant to NO-induced inhibition. Alternatively, another cysteine residue located at the extracellular EGFR ligand-binding interface undergoes nitrosylation after exposure to one millimolar of exogenous NO donor S-nitroso-L-cysteine (Cys-NO) (123). S-nitrosylation of these residues may inhibit EGFR-mediated signaling by interfering with the ligand interaction site, although this proposal awaits evaluation. By contrast, a more recent study demonstrates that S-nitrosylation induced by the chemical NO donor (Z)-1-[N-2-aminoethyl)-N-(2-ammonioethyl)amino] diazen-1-ium-1,2-diolate known as DETA-NO can upregulate EGFR signaling and correlates with a transformed breast cancer phenotype (124, 125). An important area for future research is to determine whether EGFR undergoes direct S-nitrosylation in response to physiological stimuli.
Future Perspectives
Of the ~95 receptor and non-receptor PTKs in the human genome, nine additional members harbor a cysteine residue that is structurally homologous to EGFR Cys797 (Fig. 6C), including two EGFR subfamily members, HER2 and HER4. Although speculative at this time, it is possible that this group of kinases is regulated by oxidation of this residue. EGFR Cys797 and its structural analogues are located at the N-terminal end of an alpha helix, also known as the Ncap position. Interestingly, cysteine is the most sparsely occurring Ncap residue in natural proteins, comprising less than 1% of all these positions (126). Interaction of a cysteine located at the Ncap position with the helix dipole can drastically lower thiol pKa and increases its reactivity (127, 128). Of note, the Ncap effect has also been attributed to the reactivity of the human PrxI catalytic cysteine (98). Another subfamily of PTKs, which includes cytoplasmic Src and FGFR1, contains a cysteine located within a glycine rich loop that interacts with the γ-phosphate of ATP (129). Oxidation of this residue inhibits the kinase activity of c-Src and FGFR1 in vitro; however neither kinase has been confirmed as a direct target of endogenous H2O2 in cells. Over 150 kinases have a cysteine in or around the nucleotide-binding site, some of which may play similar regulatory roles. However, much more work will be required to define the scope and molecular details underlying the redox-regulated kinome.
EGFR is mutated or amplified in a number of human carcinomas including breast and lung cancers, which has motivated the development of selective kinase inhibitors, including analogs that covalently modify Cys797 and are currently in Phase II and III clinical trials (130). The recent finding that EGFR Cys797 undergoes sulfenic acid modification (71) and that elevated EGFR and HER2 in cancer cells correlates with a substantial increase in global protein sulfenylation (71), raises several fundamental questions vis-à-vis cysteine oxidation and thiol-targeted irreversible inhibitors. For example, the acrylamide moiety of irreversible EGFR inhibitors undergoes Michael addition with Cys797 in its thiol form, but these inhibitors would not react with the sulfenic acid or other cysteine chemotypes (Fig. 7A). Thus, oxidation of Cys797 could affect the potency of these inhibitors, particularly under conditions of oxidative stress often associated with cancer. On the other hand, the sulfenic acid moiety represents an entirely new opportunity in covalent inhibitor design whereby the electrophilic S-atom is targeted using a nucleophilic warhead (Fig. 7B). In this approach, the propensity for specific cysteine residues in kinases, and other therapeutically important proteins, to undergo sulfenylation could be exploited for the development of inhibitors that target this unique modification, similar to the proof-of-concept compounds we have recently reported to target oxidized PTPs (131).
Figure 7.
Covalent cysteine-based protein targeting strategies. (A) Conventional covalent inhibitors of kinases inactivate their target through covalent attachment to the cysteine thiol functional group. However, the electrophilic center (e.g., acrylamide, haloacetamide, vinyl sulfonamide) that reacts with the cysteine can exhibit nonspecific reactivity toward other cellular thiols, including glutathione present at millimolar concentrations inside mammalian cells. The electrophile may also react with other nucleophilic functionalities present in biological systems (amino and imidazole groups of amino acids, various reactive sites in nucleic acid bases, water). (B) Orthogonal strategy as one potential mechanism to address issues associated with employing an electrophilic functional group to target one cysteine among a sea of biological nucleophiles. According to this approach, active site-directed small-molecule inhibitors containing a reactive nucleophilic substituent form a covalent bond with a sulfenic acid-modified cysteine side chain. Such modifications form transiently in specific proteins during H2O2-mediated signal transduction in normal cells, but form constitutively in diseases associated with chronically elevated levels of H2O2, including cancer. In the sulfenic acid oxidation state, the electron-deficient sulfur exhibits enhanced electrophilic character that can be selectively targeted by certain nucleophilic compounds. Because sulfenic acid is a unique chemical moiety in biochemistry, this strategy might decrease the potential for off-target activity while retaining the advantages gained by covalent targeting.
Conclusions
The perspectives presented here highlight the emerging and rapidly expanding role of redox regulation during EGFR signaling. These studies point to the unique chemistry of reactive cysteine residues within specific target proteins, including EGFR. These redox reactions enable covalent regulation of protein function, much like phosphorylation. The expanding array of modifications that target cysteine suggests that we are just beginning to understand the molecular basis for specificity of redox signaling. One theme that has consistently emerged in numerous studies is the co-localization of the oxidant sources with the redox-regulated target protein. As case in point, ligand activation triggers the association of EGFR and the NADPH oxidase, Nox2. We have also tried to emphasize the value of selective and cell-permeable chemical approaches to elucidate regulatory mechanisms that govern H2O2-mediated sulfenylation of proteins. In addition, we are just beginning to appreciate that different biological oxidants may target distinct cysteine residues in the same protein and thus lead to unique regulatory outcomes. Given that aberrant sulfenylation of proteins is linked to aggressive cancer phenotypes and that genetic lesions in H2O2-metabolizing enzymes can contribute to tumorigenesis, defining mechanisms that control reversible protein sulfenylation will be vital to understanding both human physiology and disease. Finally, it is hoped that the discovery of EGFR as a direct target of H2O2 will lead to a broader examination of the redox-regulated kinome and the development of an orthogonal nucleophilic strategy for covalent inhibition of therapeutically important proteins.
Acknowledgements
The authors acknowledge funding from the Camille Henry Dreyfus Teacher Scholar Award (to K.S.C.) and the National Institutes of Health grant GM102187 (to K.S.C.).
Abbreviations
- RTK
receptor tyrosine kinase
- PTK
protein tyrosine kinase
- PTP
protein tyrosine phosphatase
- EGF
epidermal growth factor
- EGFR
EGF receptor
- MAPK
mitogen activated protein kinase
- PI3K
phosphatidylinositol 3′ kinase
- PTEN
phosphatase and tensin homologue
- PIP3
phosphatidylinositol (3,4,5) triphosphate
- H2O2
hydrogen peroxide
- ROS
reactive oxygen species
- Tyr
tyrosine
- Ser
serine
- Thr
threonine
- Nox
NADPH oxidase
- NADPH
nicotinamide adenine dinucleotide phosphate
- SOD
superoxide dismutase
- Prx
peroxiredoxin
- Gpx
glutathione peroxidase
- Grx
glutaredoxin
- GSH
glutathione
- GSR
glutathione reductase
- Trx
thioredoxin
- TrxR
thioredoxin reductase
- ER
endoplasmic reticulum
- DTT
dithiothreitol
- IAA
iodoacetic acid
- IAM
iodoacetamide
- NEM
N-ethylmaleimide
- DCFH-DA
2′, 7′-dichlorofluorescein diacetate
- DN
dominant-negative
- RNS
reactive nitrogen species
- NO
nitric oxide
Footnotes
This work was supported by National Institutes of Health grant GM102187 (K.S.C.) and funding from the Camille Henry Dreyfus Teacher Scholar Award (K.S.C).
References
- 1.Schlessinger J. Cell signaling by receptor tyrosine kinases. Cell. 2000;103:211–225. doi: 10.1016/s0092-8674(00)00114-8. [DOI] [PubMed] [Google Scholar]
- 2.Schlessinger J. Ligand-induced, receptor-mediated dimerization and activation of EGF receptor. Cell. 2002;110:669–672. doi: 10.1016/s0092-8674(02)00966-2. [DOI] [PubMed] [Google Scholar]
- 3.Cohen S. Isolation of a mouse submaxillary gland protein accelerating incisor eruption and eyelid opening in the new-born animal. J. Biol. Chem. 1962;237:1555–1562. [PubMed] [Google Scholar]
- 4.Cohen S, Carpenter G, King L., Jr. Epidermal growth factor-receptor-protein kinase interactions. Co-purification of receptor and epidermal growth factor-enhanced phosphorylation activity. J. Biol. Chem. 1980;255:4834–4842. [PubMed] [Google Scholar]
- 5.Carpenter G, King L, Jr., Cohen S. Epidermal growth factor stimulates phosphorylation in membrane preparations in vitro. Nature. 1978;276:409–410. doi: 10.1038/276409a0. [DOI] [PubMed] [Google Scholar]
- 6.Cohen S. Origins of growth factors: NGF and EGF. J. Biol. Chem. 2008;283:33793–33797. doi: 10.1074/jbc.X800008200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Macias A, Azavedo E, Hagerstrom T, Klintenberg C, Perez R, Skoog L. Prognostic significance of the receptor for epidermal growth factor in human mammary carcinomas. Anticancer Res. 1987;7:459–464. [PubMed] [Google Scholar]
- 8.Yarden Y, Sliwkowski MX. Untangling the ErbB signalling network. Nat. Rev. Mol. Cell Biol. 2001;2:127–137. doi: 10.1038/35052073. [DOI] [PubMed] [Google Scholar]
- 9.Herbst RS. Review of epidermal growth factor receptor biology. Int. J. Radiat. Oncol. Biol. Phys. 2004;59:21–26. doi: 10.1016/j.ijrobp.2003.11.041. [DOI] [PubMed] [Google Scholar]
- 10.Bae YS, Kang SW, Seo MS, Baines IC, Tekle E, Chock PB, Rhee SG. Epidermal growth factor (EGF)-induced generation of hydrogen peroxide. Role in EGF receptor-mediated tyrosine phosphorylation. J. Biol. Chem. 1997;272:217–221. [PubMed] [Google Scholar]
- 11.Miller EW, Tulyathan O, Isacoff EY, Chang CJ. Molecular imaging of hydrogen peroxide produced for cell signaling. Nat. Chem. Biol. 2007;3:263–267. doi: 10.1038/nchembio871. [DOI] [PubMed] [Google Scholar]
- 12.Lo YY, Cruz TF. Involvement of reactive oxygen species in cytokine and growth factor induction of c-fos expression in chondrocytes. J. Biol. Chem. 1995;270:11727–11730. doi: 10.1074/jbc.270.20.11727. [DOI] [PubMed] [Google Scholar]
- 13.Sundaresan M, Yu ZX, Ferrans VJ, Irani K, Finkel T. Requirement for generation of H2O2 for platelet-derived growth factor signal transduction. Science. 1995;270:296–299. doi: 10.1126/science.270.5234.296. [DOI] [PubMed] [Google Scholar]
- 14.Colavitti R, Pani G, Bedogni B, Anzevino R, Borrello S, Waltenberger J, Galeotti T. Reactive oxygen species as downstream mediators of angiogenic signaling by vascular endothelial growth factor receptor-2/KDR. J. Biol. Chem. 2002;277:3101–3108. doi: 10.1074/jbc.M107711200. [DOI] [PubMed] [Google Scholar]
- 15.May JM, de Haen C. Insulin-stimulated intracellular hydrogen peroxide production in rat epididymal fat cells. J. Biol. Chem. 1979;254:2214–2220. [PubMed] [Google Scholar]
- 16.Meier B, Radeke HH, Selle S, Younes M, Sies H, Resch K, Habermehl GG. Human fibroblasts release reactive oxygen species in response to interleukin-1 or tumour necrosis factor-alpha. Biochem. J. 1989;263:539–545. doi: 10.1042/bj2630539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Griendling KK, Minieri CA, Ollerenshaw JD, Alexander RW. Angiotensin II stimulates NADH and NADPH oxidase activity in cultured vascular smooth muscle cells. Circ. Res. 1994;74:1141–1148. doi: 10.1161/01.res.74.6.1141. [DOI] [PubMed] [Google Scholar]
- 18.Rhee SG. Cell signaling. H2O2, a necessary evil for cell signaling. Science. 2006;312:1882–1883. doi: 10.1126/science.1130481. [DOI] [PubMed] [Google Scholar]
- 19.D’Autreaux B, Toledano MB. ROS as signalling molecules: mechanisms that generate specificity in ROS homeostasis. Nat. Rev. Mol. Cell Biol. 2007;8:813–824. doi: 10.1038/nrm2256. [DOI] [PubMed] [Google Scholar]
- 20.Dickinson BC, Chang CJ. Chemistry and biology of reactive oxygen species in signaling or stress responses. Nat. Chem. Biol. 2011;7:504–511. doi: 10.1038/nchembio.607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Finkel T. Signal transduction by reactive oxygen species. J. Cell. Biol. 2011;194:7–15. doi: 10.1083/jcb.201102095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Reddie KG, Carroll KS. Expanding the functional diversity of proteins through cysteine oxidation. Curr. Opin. Chem. Biol. 2008;12:746–754. doi: 10.1016/j.cbpa.2008.07.028. [DOI] [PubMed] [Google Scholar]
- 23.Paulsen CE, Carroll KS. Orchestrating redox signaling networks through regulatory cysteine switches. ACS Chem. Biol. 2010;5:47–62. doi: 10.1021/cb900258z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Jacob C, Battaglia E, Burkholz T, Peng D, Bagrel D, Montenarh M. Control of oxidative posttranslational cysteine modifications: from intricate chemistry to widespread biological and medical applications. Chem. Res. Toxicol. 2012;25:588–604. doi: 10.1021/tx200342b. [DOI] [PubMed] [Google Scholar]
- 25.Zheng M, Aslund F, Storz G. Activation of the OxyR transcription factor by reversible disulfide bond formation. Science. 1998;279:1718–1721. doi: 10.1126/science.279.5357.1718. [DOI] [PubMed] [Google Scholar]
- 26.Chen CY, Willard D, Rudolph J. Redox regulation of SH2-domain-containing protein tyrosine phosphatases by two backdoor cysteines. Biochemistry. 2009;48:1399–1409. doi: 10.1021/bi801973z. [DOI] [PubMed] [Google Scholar]
- 27.Aslan M, Ozben T. Oxidants in receptor tyrosine kinase signal transduction pathways. Antioxid. Redox. Signal. 2003;5:781–788. doi: 10.1089/152308603770380089. [DOI] [PubMed] [Google Scholar]
- 28.Tonks NK. Redox redux: revisiting PTPs and the control of cell signaling. Cell. 2005;121:667–670. doi: 10.1016/j.cell.2005.05.016. [DOI] [PubMed] [Google Scholar]
- 29.Finkel T. From sulfenylation to sulfhydration: what a thiolate needs to tolerate. Sci. Signal. 2012;5:pe10. doi: 10.1126/scisignal.2002943. [DOI] [PubMed] [Google Scholar]
- 30.Gamou S, Shimizu N. Hydrogen peroxide preferentially enhances the tyrosine phosphorylation of epidermal growth factor receptor. FEBS Lett. 1995;357:161–164. doi: 10.1016/0014-5793(94)01335-x. [DOI] [PubMed] [Google Scholar]
- 31.Stone JR, Yang S. Hydrogen peroxide: a signaling messenger. Antioxid. Redox. Signal. 2006;8:243–270. doi: 10.1089/ars.2006.8.243. [DOI] [PubMed] [Google Scholar]
- 32.Bass DA, Parce JW, Dechatelet LR, Szejda P, Seeds MC, Thomas M. Flow cytometric studies of oxidative product formation by neutrophils: a graded response to membrane stimulation. J. Immunol. 1983;130:1910–1917. [PubMed] [Google Scholar]
- 33.Giorgio M, Trinei M, Migliaccio E, Pelicci PG. Hydrogen peroxide: a metabolic by-product or a common mediator of ageing signals? Nat. Rev. Mol. Cell. Biol. 2007;8:722–728. doi: 10.1038/nrm2240. [DOI] [PubMed] [Google Scholar]
- 34.Halliwell B, Gutteridge JMC. Free Radicals in Biology and Medicine. Oxford University Press; Oxford: 1999. [Google Scholar]
- 35.Goldkorn T, Balaban N, Matsukuma K, Chea V, Gould R, Last J, Chan C, Chavez C. EGF-Receptor phosphorylation and signaling are targeted by H2O2 redox stress. Am. J. Respir. Cell. Mol. Biol. 1998;19:786–798. doi: 10.1165/ajrcmb.19.5.3249. [DOI] [PubMed] [Google Scholar]
- 36.Rao GN. Hydrogen peroxide induces complex formation of SHC-Grb2-SOS with receptor tyrosine kinase and activates Ras and extracellular signal-regulated protein kinases group of mitogen-activated protein kinases. Oncogene. 1996;13:713–719. [PubMed] [Google Scholar]
- 37.McCubrey JA, Lahair MM, Franklin RA. Reactive oxygen species-induced activation of the MAP kinase signaling pathways. Antioxid. Redox. Signal. 2006;8:1775–1789. doi: 10.1089/ars.2006.8.1775. [DOI] [PubMed] [Google Scholar]
- 38.Guyton KZ, Liu Y, Gorospe M, Xu Q, Holbrook NJ. Activation of mitogen-activated protein kinase by H2O2. Role in cell survival following oxidant injury. J. Biol. Chem. 1996;271:4138–4142. doi: 10.1074/jbc.271.8.4138. [DOI] [PubMed] [Google Scholar]
- 39.Wang X, Martindale JL, Liu Y, Holbrook NJ. The cellular response to oxidative stress: influences of mitogen-activated protein kinase signalling pathways on cell survival. Biochem. J. 1998;333(Pt 2):291–300. doi: 10.1042/bj3330291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Leslie NR. The redox regulation of PI 3-kinase-dependent signaling. Antioxid. Redox. Signal. 2006;8:1765–1774. doi: 10.1089/ars.2006.8.1765. [DOI] [PubMed] [Google Scholar]
- 41.Stokoe D, Stephens LR, Copeland T, Gaffney PR, Reese CB, Painter GF, Holmes AB, McCormick F, Hawkins PT. Dual role of phosphatidylinositol-3,4,5-trisphosphate in the activation of protein kinase B. Science. 1997;277:567–570. doi: 10.1126/science.277.5325.567. [DOI] [PubMed] [Google Scholar]
- 42.Franke TF, Kaplan DR, Cantley LC, Toker A. Direct regulation of the Akt proto-oncogene product by phosphatidylinositol-3,4-bisphosphate. Science. 1997;275:665–668. doi: 10.1126/science.275.5300.665. [DOI] [PubMed] [Google Scholar]
- 43.Park HS, Lee SH, Park D, Lee JS, Ryu SH, Lee WJ, Rhee SG, Bae YS. Sequential activation of phosphatidylinositol 3-kinase, beta Pix, Rac1, and Nox1 in growth factor-induced production of H2O2. Mol. Cell. Biol. 2004;24:4384–4394. doi: 10.1128/MCB.24.10.4384-4394.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Wang X, McCullough KD, Franke TF, Holbrook NJ. Epidermal growth factor receptor-dependent Akt activation by oxidative stress enhances cell survival. J. Biol. Chem. 2000;275:14624–14631. doi: 10.1074/jbc.275.19.14624. [DOI] [PubMed] [Google Scholar]
- 45.Huang X, Begley M, Morgenstern KA, Gu Y, Rose P, Zhao H, Zhu X. Crystal structure of an inactive Akt2 kinase domain. Structure. 2003;11:21–30. doi: 10.1016/s0969-2126(02)00937-1. [DOI] [PubMed] [Google Scholar]
- 46.Murata H, Ihara Y, Nakamura H, Yodoi J, Sumikawa K, Kondo T. Glutaredoxin exerts an antiapoptotic effect by regulating the redox state of Akt. J. Biol. Chem. 2003;278:50226–50233. doi: 10.1074/jbc.M310171200. [DOI] [PubMed] [Google Scholar]
- 47.Wani R, Qian J, Yin L, Bechtold E, King SB, Poole LB, Paek E, Tsang AW, Furdui CM. Isoform-specific regulation of Akt by PDGF-induced reactive oxygen species. Proc. Natl. Acad. Sci. U S A. 2011;108:10550–10555. doi: 10.1073/pnas.1011665108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Ushio-Fukai M, Griendling KK, Becker PL, Hilenski L, Halleran S, Alexander RW. Epidermal growth factor receptor transactivation by angiotensin II requires reactive oxygen species in vascular smooth muscle cells. Arterioscler. Thromb. Vasc. Biol. 2001;21:489–495. doi: 10.1161/01.atv.21.4.489. [DOI] [PubMed] [Google Scholar]
- 49.Chen K, Vita JA, Berk BC, Keaney JF., Jr. c-Jun N-terminal kinase activation by hydrogen peroxide in endothelial cells involves SRC-dependent epidermal growth factor receptor transactivation. J. Biol. Chem. 2001;276:16045–16050. doi: 10.1074/jbc.M011766200. [DOI] [PubMed] [Google Scholar]
- 50.Giannoni E, Buricchi F, Grimaldi G, Parri M, Cialdai F, Taddei ML, Raugei G, Ramponi G, Chiarugi P. Redox regulation of anoikis: reactive oxygen species as essential mediators of cell survival. Cell Death. Differ. 2008;15:867–878. doi: 10.1038/cdd.2008.3. [DOI] [PubMed] [Google Scholar]
- 51.Reynolds AR, Tischer C, Verveer PJ, Rocks O, Bastiaens PI. EGFR activation coupled to inhibition of tyrosine phosphatases causes lateral signal propagation. Nat. Cell Biol. 2003;5:447–453. doi: 10.1038/ncb981. [DOI] [PubMed] [Google Scholar]
- 52.Tonks NK. Protein tyrosine phosphatases: from genes, to function, to disease. Nat. Rev. Mol. Cell Biol. 2006;7:833–846. doi: 10.1038/nrm2039. [DOI] [PubMed] [Google Scholar]
- 53.Blume-Jensen P, Hunter T. Oncogenic kinase signalling. Nature. 2001;411:355–365. doi: 10.1038/35077225. [DOI] [PubMed] [Google Scholar]
- 54.Denu JM, Tanner KG. Specific and reversible inactivation of protein tyrosine phosphatases by hydrogen peroxide: evidence for a sulfenic acid intermediate and implications for redox regulation. Biochemistry. 1998;37:5633–5642. doi: 10.1021/bi973035t. [DOI] [PubMed] [Google Scholar]
- 55.Zhang ZY, Dixon JE. Active site labeling of the Yersinia protein tyrosine phosphatase: the determination of the pKa of the active site cysteine and the function of the conserved histidine 402. Biochemistry. 1993;32:9340–9345. doi: 10.1021/bi00087a012. [DOI] [PubMed] [Google Scholar]
- 56.Lohse DL, Denu JM, Santoro N, Dixon JE. Roles of aspartic acid-181 and serine-222 in intermediate formation and hydrolysis of the mammalian protein-tyrosine-phosphatase PTP1. Biochemistry. 1997;36:4568–4575. doi: 10.1021/bi963094r. [DOI] [PubMed] [Google Scholar]
- 57.Winterbourn CC, Metodiewa D. Reactivity of biologically important thiol compounds with superoxide and hydrogen peroxide. Free Radic. Biol. Med. 1999;27:322–328. doi: 10.1016/s0891-5849(99)00051-9. [DOI] [PubMed] [Google Scholar]
- 58.Sohn J, Rudolph J. Catalytic and chemical competence of regulation of cdc25 phosphatase by oxidation/reduction. Biochemistry. 2003;42:10060–10070. doi: 10.1021/bi0345081. [DOI] [PubMed] [Google Scholar]
- 59.Flint AJ, Tiganis T, Barford D, Tonks NK. Development of “substrate-trapping” mutants to identify physiological substrates of protein tyrosine phosphatases. Proc. Natl. Acad. Sci. U S A. 1997;94:1680–1685. doi: 10.1073/pnas.94.5.1680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Liu F, Chernoff J. Protein tyrosine phosphatase 1B interacts with and is tyrosine phosphorylated by the epidermal growth factor receptor. Biochem. J. 1997;327(Pt 1):139–145. doi: 10.1042/bj3270139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Wu Y, Kwon KS, Rhee SG. Probing cellular protein targets of H2O2 with fluorescein-conjugated iodoacetamide and antibodies to fluorescein. FEBS Lett. 1998;440:111–115. doi: 10.1016/s0014-5793(98)01415-x. [DOI] [PubMed] [Google Scholar]
- 62.Lou YW, Chen YY, Hsu SF, Chen RK, Lee CL, Khoo KH, Tonks NK, Meng TC. Redox regulation of the protein tyrosine phosphatase PTP1B in cancer cells. FEBS J. 2008;275:69–88. doi: 10.1111/j.1742-4658.2007.06173.x. [DOI] [PubMed] [Google Scholar]
- 63.Haj FG, Markova B, Klaman LD, Bohmer FD, Neel BG. Regulation of receptor tyrosine kinase signaling by protein tyrosine phosphatase-1B. J. Biol. Chem. 2003;278:739–744. doi: 10.1074/jbc.M210194200. [DOI] [PubMed] [Google Scholar]
- 64.Haj FG, Verveer PJ, Squire A, Neel BG, Bastiaens PI. Imaging sites of receptor dephosphorylation by PTP1B on the surface of the endoplasmic reticulum. Science. 2002;295:1708–1711. doi: 10.1126/science.1067566. [DOI] [PubMed] [Google Scholar]
- 65.Lee SR, Kwon KS, Kim SR, Rhee SG. Reversible inactivation of protein-tyrosine phosphatase 1B in A431 cells stimulated with epidermal growth factor. J. Biol. Chem. 1998;273:15366–15372. doi: 10.1074/jbc.273.25.15366. [DOI] [PubMed] [Google Scholar]
- 66.Leslie NR, Bennett D, Lindsay YE, Stewart H, Gray A, Downes CP. Redox regulation of PI 3-kinase signalling via inactivation of PTEN. EMBO J. 2003;22:5501–5510. doi: 10.1093/emboj/cdg513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Maehama T, Taylor GS, Dixon JE. PTEN and myotubularin: novel phosphoinositide phosphatases. Annu. Rev. Biochem. 2001;70:247–279. doi: 10.1146/annurev.biochem.70.1.247. [DOI] [PubMed] [Google Scholar]
- 68.Lee SR, Yang KS, Kwon J, Lee C, Jeong W, Rhee SG. Reversible inactivation of the tumor suppressor PTEN by H2O2. J. Biol. Chem. 2002;277:20336–20342. doi: 10.1074/jbc.M111899200. [DOI] [PubMed] [Google Scholar]
- 69.Kwon J, Lee SR, Yang KS, Ahn Y, Kim YJ, Stadtman ER, Rhee SG. Reversible oxidation and inactivation of the tumor suppressor PTEN in cells stimulated with peptide growth factors. Proc. Natl. Acad. Sci. U S A. 2004;101:16419–16424. doi: 10.1073/pnas.0407396101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Agazie YM, Hayman MJ. Molecular mechanism for a role of SHP2 in epidermal growth factor receptor signaling. Mol. Cell Biol. 2003;23:7875–7886. doi: 10.1128/MCB.23.21.7875-7886.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Paulsen CE, Truong TH, Garcia FJ, Homann A, Gupta V, Leonard SE, Carroll KS. Peroxide-dependent sulfenylation of the EGFR catalytic site enhances kinase activity. Nat. Chem. Biol. 2012;8:57–64. doi: 10.1038/nchembio.736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Persson C, Kappert K, Engstrom U, Ostman A, Sjoblom T. An antibody-based method for monitoring in vivo oxidation of protein tyrosine phosphatases. Methods. 2005;35:37–43. doi: 10.1016/j.ymeth.2004.07.006. [DOI] [PubMed] [Google Scholar]
- 73.Lambeth JD. NOX enzymes and the biology of reactive oxygen. Nat. Rev. Immunol. 2004;4:181–189. doi: 10.1038/nri1312. [DOI] [PubMed] [Google Scholar]
- 74.McCord JM, Fridovich I. Superoxide dismutase. An enzymic function for erythrocuprein (hemocuprein) J. Biol. Chem. 1969;244:6049–6055. [PubMed] [Google Scholar]
- 75.Hsu JL, Hsieh Y, Tu C, O’Connor D, Nick HS, Silverman DN. Catalytic properties of human manganese superoxide dismutase. J. Biol. Chem. 1996;271:17687–17691. doi: 10.1074/jbc.271.30.17687. [DOI] [PubMed] [Google Scholar]
- 76.Babior BM, Lambeth JD, Nauseef W. The neutrophil NADPH oxidase. Arch. Biochem. Biophys. 2002;397:342–344. doi: 10.1006/abbi.2001.2642. [DOI] [PubMed] [Google Scholar]
- 77.Suh YA, Arnold RS, Lassegue B, Shi J, Xu X, Sorescu D, Chung AB, Griendling KK, Lambeth JD. Cell transformation by the superoxide-generating oxidase Mox1. Nature. 1999;401:79–82. doi: 10.1038/43459. [DOI] [PubMed] [Google Scholar]
- 78.Cheng G, Cao Z, Xu X, van Meir EG, Lambeth JD. Homologs of gp91phox: cloning and tissue expression of Nox3, Nox4, and Nox5. Gene. 2001;269:131–140. doi: 10.1016/s0378-1119(01)00449-8. [DOI] [PubMed] [Google Scholar]
- 79.De Deken X, Wang D, Many MC, Costagliola S, Libert F, Vassart G, Dumont JE, Miot F. Cloning of two human thyroid cDNAs encoding new members of the NADPH oxidase family. J. Biol. Chem. 2000;275:23227–23233. doi: 10.1074/jbc.M000916200. [DOI] [PubMed] [Google Scholar]
- 80.Oh H, Jung HY, Kim J, Bae YS. Phosphorylation of serine282 in NADPH oxidase activator 1 by Erk desensitizes EGF-induced ROS generation. Biochem. Biophys. Res. Commun. 2010;394:691–696. doi: 10.1016/j.bbrc.2010.03.053. [DOI] [PubMed] [Google Scholar]
- 81.Kroviarski Y, Debbabi M, Bachoual R, Perianin A, Gougerot-Pocidalo MA, El-Benna J, Dang PM. Phosphorylation of NADPH oxidase activator 1 (NOXA1) on serine 282 by MAP kinases and on serine 172 by protein kinase C and protein kinase A prevents NOX1 hyperactivation. FASEB J. 2010;24:2077–2092. doi: 10.1096/fj.09-147629. [DOI] [PubMed] [Google Scholar]
- 82.Chen K, Kirber MT, Xiao H, Yang Y, Keaney JF. Regulation of ROS signal transduction by NADPH oxidase 4 localization. J. Cell. Biol. 2008;181:1129–1139. doi: 10.1083/jcb.200709049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Fridovich I. Superoxide radical and superoxide dismutases. Annu. Rev. Biochem. 1995;64:97–112. doi: 10.1146/annurev.bi.64.070195.000525. [DOI] [PubMed] [Google Scholar]
- 84.Gardner PR, Raineri I, Epstein LB, White CW. Superoxide radical and iron modulate aconitase activity in mammalian cells. J. Biol. Chem. 1995;270:13399–13405. doi: 10.1074/jbc.270.22.13399. [DOI] [PubMed] [Google Scholar]
- 85.Juarez JC, Manuia M, Burnett ME, Betancourt O, Boivin B, Shaw DE, Tonks NK, Mazar AP, Donate F. Superoxide dismutase 1 (SOD1) is essential for H2O2-mediated oxidation and inactivation of phosphatases in growth factor signaling. Proc. Natl. Acad. Sci. U S A. 2008;105:7147–7152. doi: 10.1073/pnas.0709451105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.DeYulia GJ, Jr., Carcamo JM, Borquez-Ojeda O, Shelton CC, Golde DW. Hydrogen peroxide generated extracellularly by receptor-ligand interaction facilitates cell signaling. Proc. Natl. Acad. Sci. U S A. 2005;102:5044–5049. doi: 10.1073/pnas.0501154102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.DeYulia GJ, Jr., Carcamo JM. EGF receptor-ligand interaction generates extracellular hydrogen peroxide that inhibits EGFR-associated protein tyrosine phosphatases. Biochem. Biophys. Res. Commun. 2005;334:38–42. doi: 10.1016/j.bbrc.2005.06.056. [DOI] [PubMed] [Google Scholar]
- 88.Bienert GP, Schjoerring JK, Jahn TP. Membrane transport of hydrogen peroxide. Biochim. Biophys. Acta. 2006;1758:994–1003. doi: 10.1016/j.bbamem.2006.02.015. [DOI] [PubMed] [Google Scholar]
- 89.Bienert GP, Moller AL, Kristiansen KA, Schulz A, Moller IM, Schjoerring JK, Jahn TP. Specific aquaporins facilitate the diffusion of hydrogen peroxide across membranes. J. Biol. Chem. 2007;282:1183–1192. doi: 10.1074/jbc.M603761200. [DOI] [PubMed] [Google Scholar]
- 90.Miller EW, Dickinson BC, Chang CJ. Aquaporin-3 mediates hydrogen peroxide uptake to regulate downstream intracellular signaling. Proc. Natl. Acad. Sci. U S A. 2010;107:15681–15686. doi: 10.1073/pnas.1005776107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Wood ZA, Schroder E, Robin Harris J, Poole LB. Structure, mechanism and regulation of peroxiredoxins. Trends Biochem. Sci. 2003;28:32–40. doi: 10.1016/s0968-0004(02)00003-8. [DOI] [PubMed] [Google Scholar]
- 92.Kang SW, Chae HZ, Seo MS, Kim K, Baines IC, Rhee SG. Mammalian peroxiredoxin isoforms can reduce hydrogen peroxide generated in response to growth factors and tumor necrosis factor-alpha. J. Biol. Chem. 1998;273:6297–6302. doi: 10.1074/jbc.273.11.6297. [DOI] [PubMed] [Google Scholar]
- 93.Handy DE, Lubos E, Yang Y, Galbraith JD, Kelly N, Zhang YY, Leopold JA, Loscalzo J. Glutathione peroxidase-1 regulates mitochondrial function to modulate redox-dependent cellular responses. J. Biol. Chem. 2009;284:11913–11921. doi: 10.1074/jbc.M900392200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Halvey PJ, Watson WH, Hansen JM, Go YM, Samali A, Jones DP. Compartmental oxidation of thiol-disulphide redox couples during epidermal growth factor signalling. Biochem. J. 2005;386:215–219. doi: 10.1042/BJ20041829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Winterbourn CC. Reconciling the chemistry and biology of reactive oxygen species. Nat. Chem. Biol. 2008;4:278–286. doi: 10.1038/nchembio.85. [DOI] [PubMed] [Google Scholar]
- 96.Parsonage D, Karplus PA, Poole LB. Substrate specificity and redox potential of AhpC, a bacterial peroxiredoxin. Proc. Natl. Acad. Sci. U S A. 2008;105:8209–8214. doi: 10.1073/pnas.0708308105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Peskin AV, Low FM, Paton LN, Maghzal GJ, Hampton MB, Winterbourn CC. The high reactivity of peroxiredoxin 2 with H2O2 is not reflected in its reaction with other oxidants and thiol reagents. J. Biol. Chem. 2007;282:11885–11892. doi: 10.1074/jbc.M700339200. [DOI] [PubMed] [Google Scholar]
- 98.Woo HA, Yim SH, Shin DH, Kang D, Yu DY, Rhee SG. Inactivation of peroxiredoxin I by phosphorylation allows localized H2O2 accumulation for cell signaling. Cell. 2010;140:517–528. doi: 10.1016/j.cell.2010.01.009. [DOI] [PubMed] [Google Scholar]
- 99.Buhrow SA, Cohen S, Staros JV. Affinity labeling of the protein kinase associated with the epidermal growth factor receptor in membrane vesicles from A431 cells. J. Biol. Chem. 1982;257:4019–4022. [PubMed] [Google Scholar]
- 100.Clark S, Konstantopoulos N. Sulphydryl agents modulate insulin- and epidermal growth factor (EGF)-receptor kinase via reaction with intracellular receptor domains: differential effects on basal versus activated receptors. Biochem. J. 1993;292(Pt 1):217–223. doi: 10.1042/bj2920217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Woltjer RL, Staros JV. Effects of sulfhydryl modification reagents on the kinase activity of the epidermal growth factor receptor. Biochemistry. 1997;36:9911–9916. doi: 10.1021/bi963007v. [DOI] [PubMed] [Google Scholar]
- 102.Seo YH, Carroll KS. Profiling protein thiol oxidation in tumor cells using sulfenic acid-specific antibodies. Proc. Natl. Acad. Sci. U S A. 2009;106:16163–16168. doi: 10.1073/pnas.0903015106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Reddie KG, Seo YH, Muse WB, III, Leonard SE, Carroll KS. A chemical approach for detecting sulfenic acid-modified proteins in living cells. Mol. Biosyst. 2008;4:521–531. doi: 10.1039/b719986d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Leonard SE, Reddie KG, Carroll KS. Mining the thiol proteome for sulfenic acid modifications reveals new targets for oxidation in cells. ACS Chem. Biol. 2009;4:783–799. doi: 10.1021/cb900105q. [DOI] [PubMed] [Google Scholar]
- 105.Seo YH, Carroll KS. Quantification of Protein Sulfenic Acid Modifications Using Isotope-Coded Dimedone and Iododimedone. Angew. Chem. Int. Ed. Engl. 2011;50:1342–1345. doi: 10.1002/anie.201007175. [DOI] [PubMed] [Google Scholar]
- 106.Truong TH, Garcia FJ, Seo YH, Carroll KS. Isotope-coded chemical reporter and acid-cleavable affinity reagents for monitoring protein sulfenic acids. Bioorg. Med. Chem. Lett. 2011;21:5015–5020. doi: 10.1016/j.bmcl.2011.04.115. [DOI] [PubMed] [Google Scholar]
- 107.Leonard SE, Carroll KS. Chemical ‘omics’ approaches for understanding protein cysteine oxidation in biology. Curr. Opin. Chem. Biol. 2011;15:88–102. doi: 10.1016/j.cbpa.2010.11.012. [DOI] [PubMed] [Google Scholar]
- 108.Benitez LV, Allison WS. The inactivation of the acyl phosphatase activity catalyzed by the sulfenic acid form of glyceraldehyde 3-phosphate dehydrogenase by dimedone and olefins. J. Biol. Chem. 1974;249:6234–6243. [PubMed] [Google Scholar]
- 109.Saxon E, Bertozzi CR. Cell surface engineering by a modified Staudinger reaction. Science. 2000;287:2007–2010. doi: 10.1126/science.287.5460.2007. [DOI] [PubMed] [Google Scholar]
- 110.Rostovtsev VV, Green LG, Fokin VV, Sharpless KB. A stepwise huisgen cycloaddition process: copper(I)-catalyzed regioselective “ligation” of azides and terminal alkynes. Angew. Chem. Int. Ed. Engl. 2002;41:2596–2599. doi: 10.1002/1521-3773(20020715)41:14<2596::AID-ANIE2596>3.0.CO;2-4. [DOI] [PubMed] [Google Scholar]
- 111.Zhang J, Yang PL, Gray NS. Targeting cancer with small molecule kinase inhibitors. Nat. Rev. Cancer. 2009;9:28–39. doi: 10.1038/nrc2559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Singh J, Petter RC, Kluge AF. Targeted covalent drugs of the kinase family. Curr. Opin. Chem. Biol. 2010;14:475–480. doi: 10.1016/j.cbpa.2010.06.168. [DOI] [PubMed] [Google Scholar]
- 113.Singh J, Petter RC, Baillie TA, Whitty A. The resurgence of covalent drugs. Nat. Rev. Drug Discov. 2011;10:307–317. doi: 10.1038/nrd3410. [DOI] [PubMed] [Google Scholar]
- 114.Yun CH, Mengwasser KE, Toms AV, Woo MS, Greulich H, Wong KK, Meyerson M, Eck MJ. The T790M mutation in EGFR kinase causes drug resistance by increasing the affinity for ATP. Proc. Natl. Acad. Sci. USA. 2008;105:2070–2075. doi: 10.1073/pnas.0709662105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Yun CH, Boggon TJ, Li Y, Woo MS, Greulich H, Meyerson M, Eck MJ. Structures of lung cancer-derived EGFR mutants and inhibitor complexes: mechanism of activation and insights into differential inhibitor sensitivity. Cancer Cell. 2007;11:217–227. doi: 10.1016/j.ccr.2006.12.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Zhang X, Gureasko J, Shen K, Cole PA, Kuriyan J. An allosteric mechanism for activation of the kinase domain of epidermal growth factor receptor. Cell. 2006;125:1137–1149. doi: 10.1016/j.cell.2006.05.013. [DOI] [PubMed] [Google Scholar]
- 117.Jaffrey SR, Erdjument-Bromage H, Ferris CD, Tempst P, Snyder SH. Protein S-nitrosylation: a physiological signal for neuronal nitric oxide. Nat. Cell Biol. 2001;3:193–197. doi: 10.1038/35055104. [DOI] [PubMed] [Google Scholar]
- 118.Jaffrey SR, Snyder SH. The biotin switch method for the detection of S-nitrosylated proteins. Sci. STKE 2001. 2001:l1. doi: 10.1126/stke.2001.86.pl1. [DOI] [PubMed] [Google Scholar]
- 119.Wang H, Xian M. Chemical methods to detect S-nitrosation. Curr. Opin. Chem. Biol. 2011;15:32–37. doi: 10.1016/j.cbpa.2010.10.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Giustarini D, Dalle-Donne I, Colombo R, Milzani A, Rossi R. Is ascorbate able to reduce disulfide bridges? A cautionary note. Nitric Oxide. 2008;19:252–258. doi: 10.1016/j.niox.2008.07.003. [DOI] [PubMed] [Google Scholar]
- 121.Estrada C, Gomez C, Martin-Nieto J, De Frutos T, Jimenez A, Villalobo A. Nitric oxide reversibly inhibits the epidermal growth factor receptor tyrosine kinase. Biochem. J. 1997;326(Pt 2):369–376. doi: 10.1042/bj3260369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Murillo-Carretero M, Torroglosa A, Castro C, Villalobo A, Estrada C. S-Nitrosylation of the epidermal growth factor receptor: a regulatory mechanism of receptor tyrosine kinase activity. Free Radic. Biol. Med. 2009;46:471–479. doi: 10.1016/j.freeradbiomed.2008.10.048. [DOI] [PubMed] [Google Scholar]
- 123.Lam YW, Yuan Y, Isaac J, Babu CV, Meller J, Ho SM. Comprehensive identification and modified-site mapping of S-nitrosylated targets in prostate epithelial cells. PLoS One. 2010;5:e9075. doi: 10.1371/journal.pone.0009075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Glynn SA, Boersma BJ, Dorsey TH, Yi M, Yfantis HG, Ridnour LA, Martin DN, Switzer CH, Hudson RS, Wink DA, Lee DH, Stephens RM, Ambs S. Increased NOS2 predicts poor survival in estrogen receptor-negative breast cancer patients. J. Clin. Invest. 2010;120:3843–3854. doi: 10.1172/JCI42059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Switzer CH, Glynn SA, Cheng RY, Ridnour LA, Green JE, Ambs S, Wink DA. S-Nitrosylation of EGFR and Src Activates an Oncogenic Signaling Network in Human Basal-Like Breast Cancer. Mol. Cancer Res. 2012 doi: 10.1158/1541-7786.MCR-12-0124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Penel S, Hughes E, Doig AJ. Side-chain structures in the first turn of the alpha-helix. J. Mol. Biol. 1999;287:127–143. doi: 10.1006/jmbi.1998.2549. [DOI] [PubMed] [Google Scholar]
- 127.Anderson TA, Sauer RT. Role of an N(cap) residue in determining the stability and operator-binding affinity of Arc repressor. Biophys. Chem. 2003;100:341–350. doi: 10.1016/s0301-4622(02)00291-0. [DOI] [PubMed] [Google Scholar]
- 128.Miranda JJ. Position-dependent interactions between cysteine residues and the helix dipole. Protein Sci. 2003;12:73–81. doi: 10.1110/ps.0224203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Kemble DJ, Sun G. Direct and specific inactivation of protein tyrosine kinases in the Src and FGFR families by reversible cysteine oxidation. Proc. Natl. Acad. Sci. U S A. 2009;106:5070–5075. doi: 10.1073/pnas.0806117106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Singh J, Dobrusin EM, Fry DW, Haske T, Whitty A, McNamara DJ. Structure-based design of a potent, selective, and irreversible inhibitor of the catalytic domain of the erbB receptor subfamily of protein tyrosine kinases. J. Med. Chem. 1997;40:1130–1135. doi: 10.1021/jm960380s. [DOI] [PubMed] [Google Scholar]
- 131.Leonard SE, Garcia FJ, Goodsell DS, Carroll KS. Redox-based probes for protein tyrosine phosphatases. Angew. Chem. Int. Ed. Engl. 2011;50:4423–4427. doi: 10.1002/anie.201007871. [DOI] [PubMed] [Google Scholar]