Abstract
Transfer of phospholipid from the envelope of hemagglutinating virus of Japan (HVJ) to erythrocyte (RBC) membrane and the virus-induced transfer of phospholipid between RBC membranes were studied using spin-labeled phosphatidylcholine (PC). The transfer of PC from membranes labeled densely with PC to unlabeled membranes was followed by the peak height increase in the electron spin resonance spectrum. The two kinds of transfer reactions took place very rapidly as reported previously. To obtain further details, the transfer reactions were studied with HVJ, HVJ inactivated by trypsin, HVJ harvested early, HVJ grown in fibroblast cells, the fibroblast HVJ activated by trypsin, influenza virus, and glutaraldehyde-treated RBCs. The results demonstrated that the viral F glycoprotein played a crucial role in the transmembrane phospholipid movements as well as in the fusion and hemolysis of RBCs. The transfer from HVJ to RBC's occurred partially through an exchange mechanism not accompanying the envelope fusion. This was shown by a decrease in the exchange broadening of the electron spin resonance spectrum of released spin-labeled HVJ (HVJ) and also by an increase in the ratio of PC to viral proteins incorporated into RBC membranes. HVJ modified RBC membrane so as to be able to exchange its phospholipids with those of inactive membranes such as fibroblast HVJ, influenza virus, glutaraldehyde-treated RBC'S, and phosphatidylcholine vesicles. HVJ affected the fluidity of RBC membranes markedly, the environments around PC being much fluidized. The virus-induced fusion was discussed based on close apposition of the membranes by HANA proteins and on the destabilization and fluidization of RBC membranes by F glycoproteins.
Full text
PDF

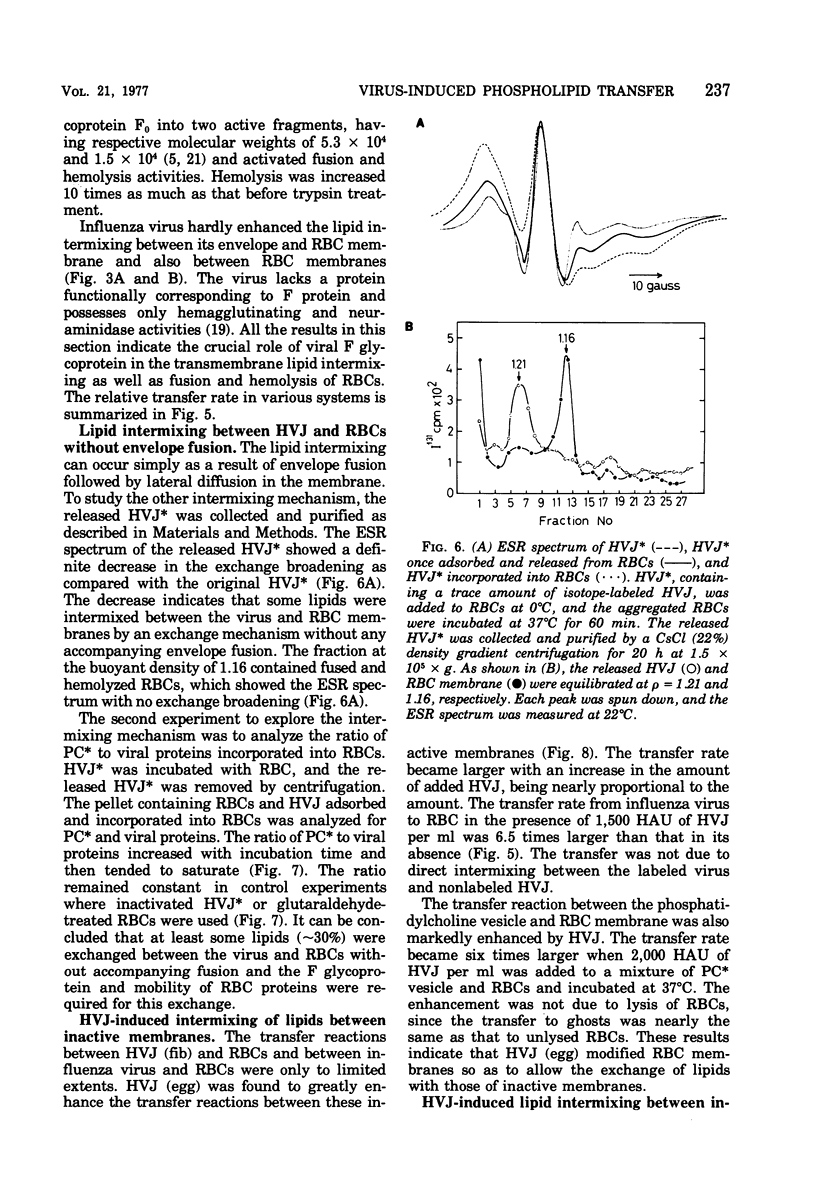
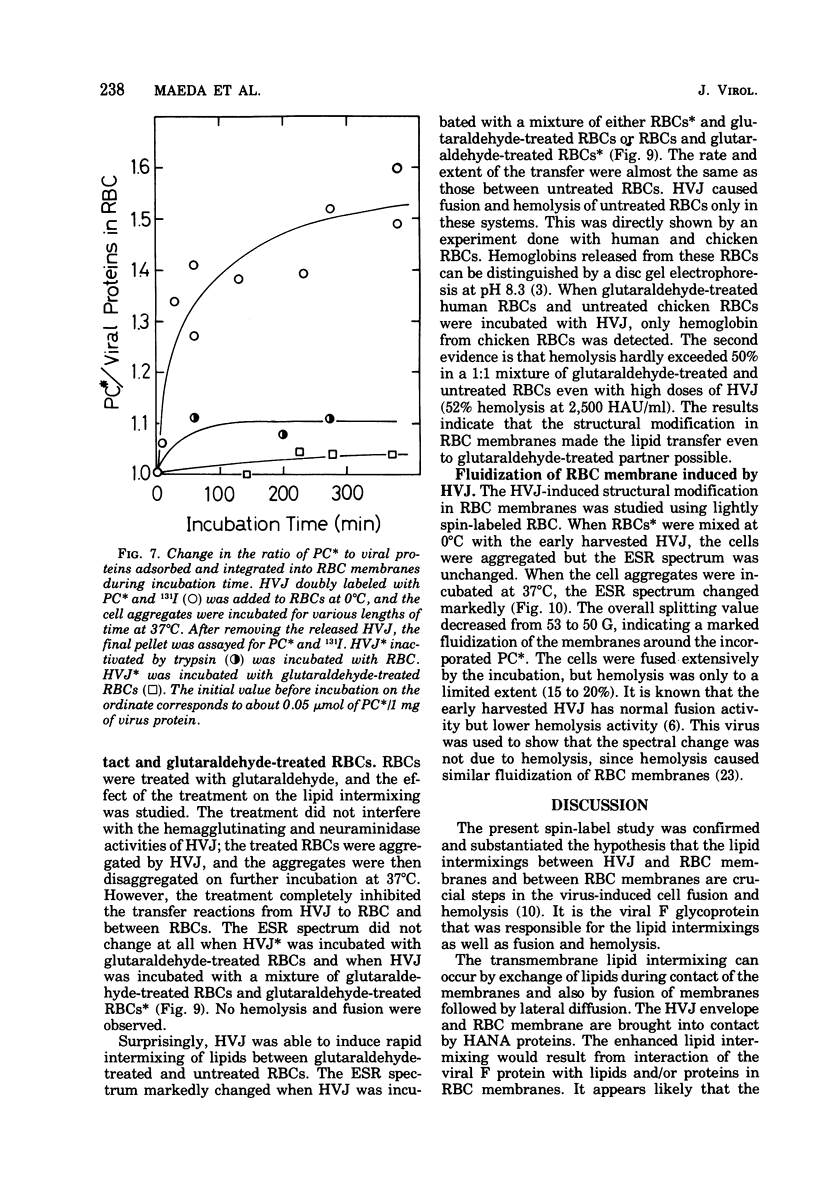
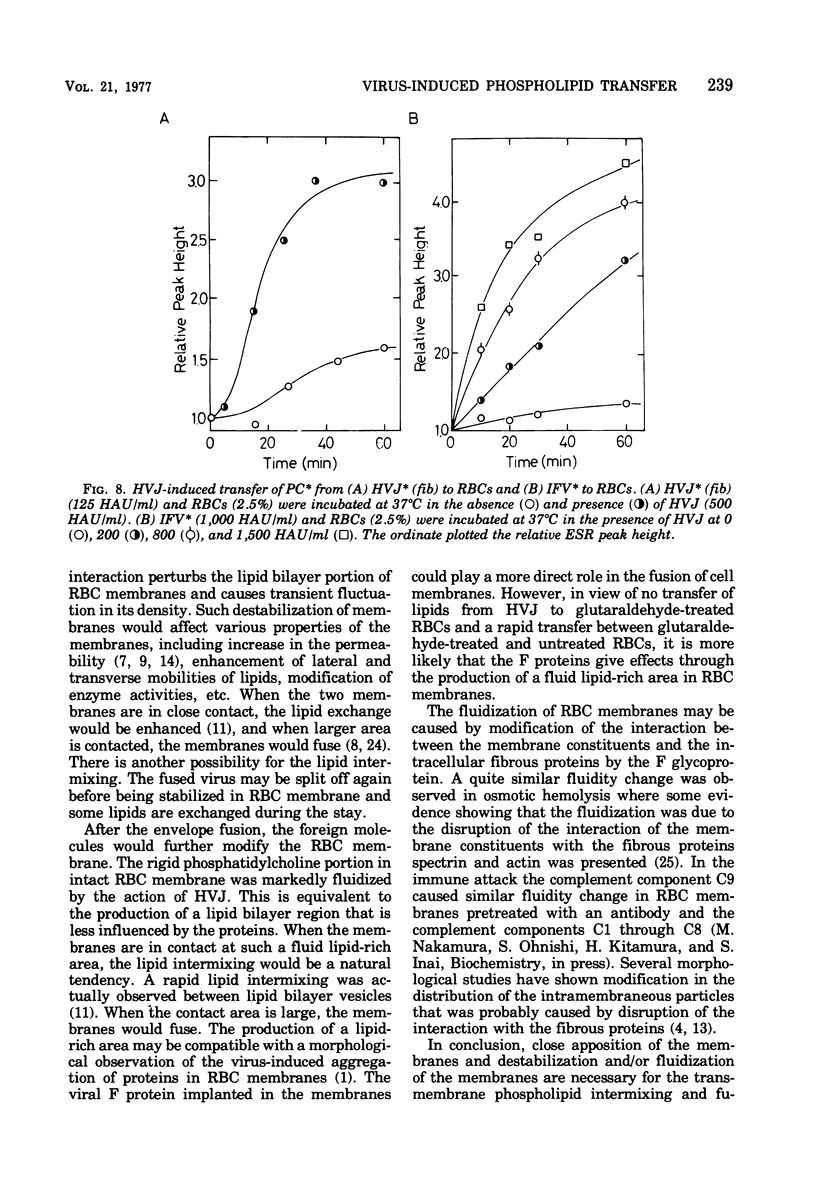
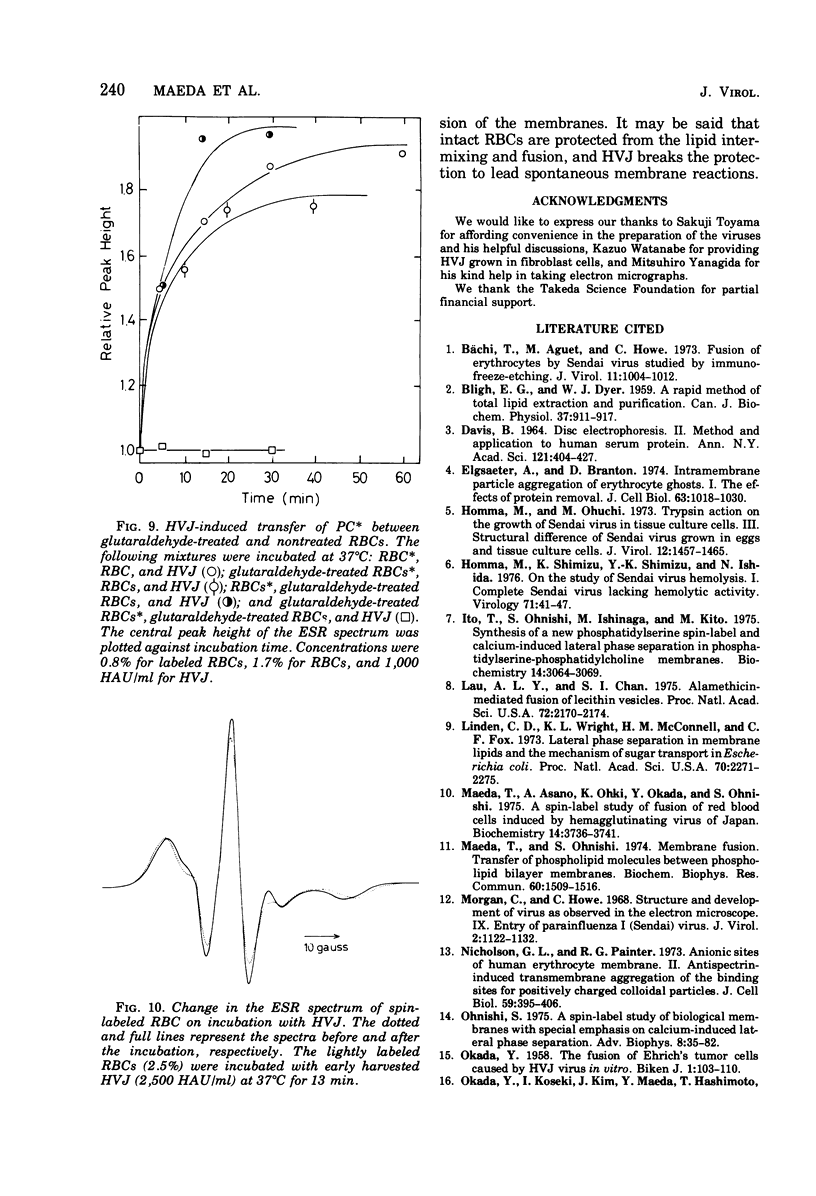

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- BLIGH E. G., DYER W. J. A rapid method of total lipid extraction and purification. Can J Biochem Physiol. 1959 Aug;37(8):911–917. doi: 10.1139/o59-099. [DOI] [PubMed] [Google Scholar]
- Bächi T., Aguet M., Howe C. Fusion of erythrocytes by Sendai virus studied by immuno-freeze-etching. J Virol. 1973 Jun;11(6):1004–1012. doi: 10.1128/jvi.11.6.1004-1012.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DAVIS B. J. DISC ELECTROPHORESIS. II. METHOD AND APPLICATION TO HUMAN SERUM PROTEINS. Ann N Y Acad Sci. 1964 Dec 28;121:404–427. doi: 10.1111/j.1749-6632.1964.tb14213.x. [DOI] [PubMed] [Google Scholar]
- Elgsaeter A., Branton D. Intramembrane particle aggregation in erythrocyte ghosts. I. The effects of protein removal. J Cell Biol. 1974 Dec;63(3):1018–1036. doi: 10.1083/jcb.63.3.1018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Homma M., Ouchi M. Trypsin action on the growth of Sendai virus in tissue culture cells. 3. Structural difference of Sendai viruses grown in eggs and tissue culture cells. J Virol. 1973 Dec;12(6):1457–1465. doi: 10.1128/jvi.12.6.1457-1465.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Homma M., Shimizu K., Shimizu Y. K., Ishida N. On the study of Sendai virus hemolysis. I. Complete Sendai virus lacking in hemolytic activity. Virology. 1976 May;71(1):41–47. doi: 10.1016/0042-6822(76)90092-1. [DOI] [PubMed] [Google Scholar]
- Lau A. L., Chan S. I. Alamethicin-mediated fusion of lecithin vesicles. Proc Natl Acad Sci U S A. 1975 Jun;72(6):2170–2174. doi: 10.1073/pnas.72.6.2170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Linden C. D., Wright K. L., McConnell H. M., Fox C. F. Lateral phase separations in membrane lipids and the mechanism of sugar transport in Escherichia coli. Proc Natl Acad Sci U S A. 1973 Aug;70(8):2271–2275. doi: 10.1073/pnas.70.8.2271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maeda T., Asano A., Oki K., Okada Y., Onishi S. A spin-label study on fusion of red blood cells induced by hemagglutinating virus of Japan. Biochemistry. 1975 Aug 26;14(17):3736–3741. doi: 10.1021/bi00688a003. [DOI] [PubMed] [Google Scholar]
- Maeda T., Ohnishi S. Membrane fusion. Transfer of phospholipid molecules between phospholipid bilayer membranes. Biochem Biophys Res Commun. 1974 Oct 23;60(4):1509–1516. doi: 10.1016/0006-291x(74)90368-4. [DOI] [PubMed] [Google Scholar]
- Morgan C., Howe C. Structure and development of viruses as observed in the electron microscope. IX. Entry of parainfluenza I (Sendai) virus. J Virol. 1968 Oct;2(10):1122–1132. doi: 10.1128/jvi.2.10.1122-1132.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nicolson G. L., Painter R. G. Anionic sites of human erythrocyte membranes. II. Antispectrin-induced transmembrane aggregation of the binding sites for positively charged colloidal particles. J Cell Biol. 1973 Nov;59(2 Pt 1):395–406. doi: 10.1083/jcb.59.2.395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohnishi S. I. A spin-label study of biological membranes with special emphasis on calcium-induced lateral phase separation. Adv Biophys. 1976;8:35–82. [PubMed] [Google Scholar]
- Poste G., Allison A. C. Membrane fusion. Biochim Biophys Acta. 1973 Dec 28;300(4):421–465. doi: 10.1016/0304-4157(73)90015-4. [DOI] [PubMed] [Google Scholar]
- Scheid A., Choppin P. W. Identification of biological activities of paramyxovirus glycoproteins. Activation of cell fusion, hemolysis, and infectivity of proteolytic cleavage of an inactive precursor protein of Sendai virus. Virology. 1974 Feb;57(2):475–490. doi: 10.1016/0042-6822(74)90187-1. [DOI] [PubMed] [Google Scholar]
- Schulze I. T. The structure of influenza virus. II. A model based on the morphology and composition of subviral particles. Virology. 1972 Jan;47(1):181–196. doi: 10.1016/0042-6822(72)90251-6. [DOI] [PubMed] [Google Scholar]
- Sefton B. M., Wickus G. G., Burge B. W. Enzymatic iodination of Sindbis virus proteins. J Virol. 1973 May;11(5):730–735. doi: 10.1128/jvi.11.5.730-735.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimizu K., Isida N. The smallest protein of Sendi virus: its candidate function of binding nucleocaspsid to envelope. Virology. 1975 Oct;67(2):427–437. [PubMed] [Google Scholar]
- Tanaka K. I., Ohnishi S. Heterogeneity in the fluidity of intact erythrocyte membrane and its homogenization upon hemolysis. Biochim Biophys Acta. 1976 Mar 5;426(2):218–231. doi: 10.1016/0005-2736(76)90333-3. [DOI] [PubMed] [Google Scholar]
- Weber K., Osborn M. The reliability of molecular weight determinations by dodecyl sulfate-polyacrylamide gel electrophoresis. J Biol Chem. 1969 Aug 25;244(16):4406–4412. [PubMed] [Google Scholar]
- van der Bosch J., McConnell M. Fusion of dipalmitoylphosphatidylcholine vesicle membranes induced by concanavalin A. Proc Natl Acad Sci U S A. 1975 Nov;72(11):4409–4413. doi: 10.1073/pnas.72.11.4409. [DOI] [PMC free article] [PubMed] [Google Scholar]