Abstract
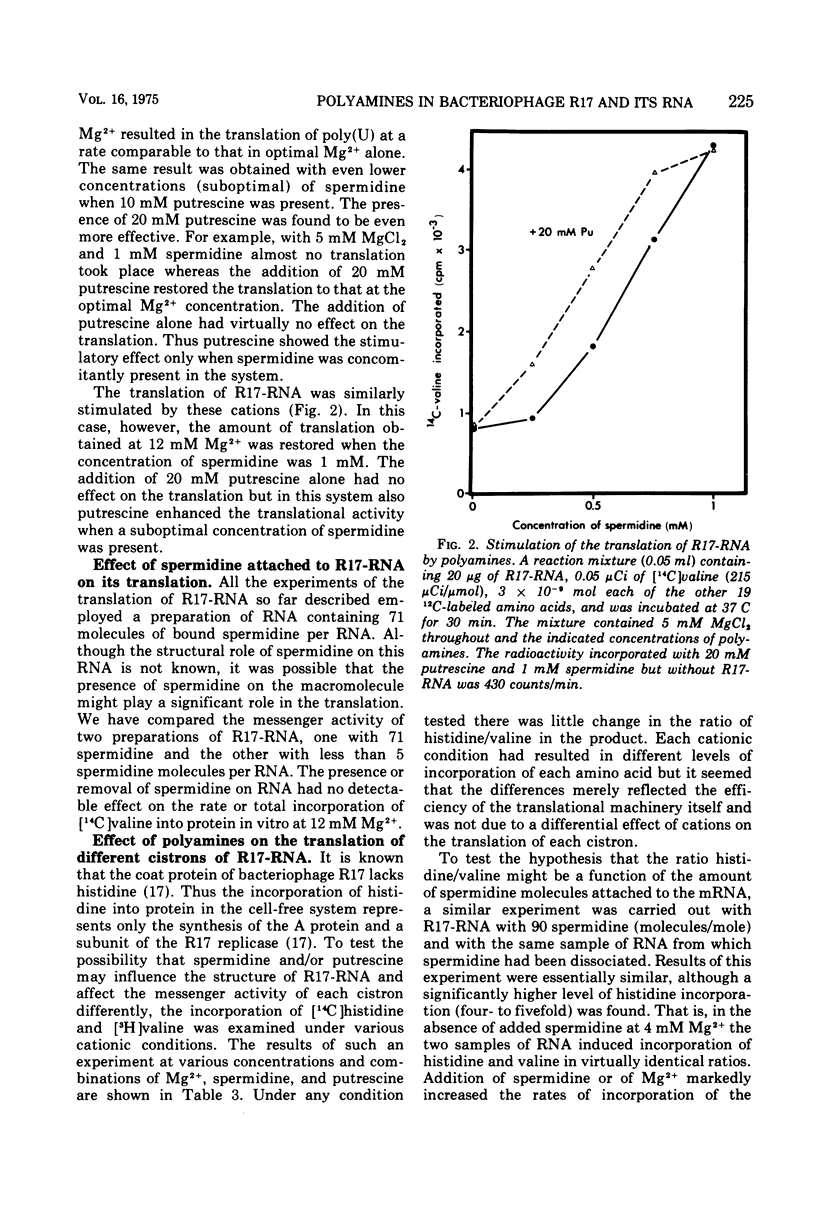
Bacteriophage R17 and its RNA were found to contain significant amounts of spermidine but not of putrescine. When isolated at 0.01 M KCl, up to 1,000 molecules of spermidine were associated with the virion. The phage RNA isolated with phenol plus sodium lauryl sulfate contained approximately 70 to 90 molecules of spermidine. The association appeared to be ionic because the bound spermidine could be dissociated by KCl, MgCl2, or both. Effects of polyamines on in vitro translation were studied using both poly(U) and phage R17-RNA as mRNA. Addition of spermidine to the system at suboptimal concentrations of Mg2+ resulted in marked stimulations of the rate of protein synthesis. Putrescine alone had no effect but stimulated the incorporation in the presence of suboptimal concentrations of spermidine plus Mg2+. The isolated amino acid-incorporating system contained suboptimal soluble and bound polyamines. A comparison of incorporation was made in this system using R17-RNA with and without bound spermidine. No effects of these bound cations were detected on the rate or extent of incorporation of valine. The ratio of incorporation of histidine (present in non-coat proteins) to valine (total protein) revealed little difference as a functions of cation in the system or a function of the spermidine present in R17-RNA.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- AMES B. N., DUBIN D. T. The role of polyamines in the neutralization of bacteriophage deoxyribonucleic acid. J Biol Chem. 1960 Mar;235:769–775. [PubMed] [Google Scholar]
- Adams J. M., Cory S., Spahr P. F. Nucleotide sequences of fragments of R17 bacteriophage RNA from the region immediately preceding the coat-protein cistron. Eur J Biochem. 1972 Sep 25;29(3):469–479. doi: 10.1111/j.1432-1033.1972.tb02011.x. [DOI] [PubMed] [Google Scholar]
- Bachrach U., Don S., Wiener H. Occurrence of polyamines in myxoviruses. J Gen Virol. 1974 Mar;22(3):451–454. doi: 10.1099/0022-1317-22-3-451. [DOI] [PubMed] [Google Scholar]
- Beer S. V., Kosuge T. Spermidine and spermine--polyamine components of turnip yellow mosaic virus. Virology. 1970 Apr;40(4):930–938. doi: 10.1016/0042-6822(70)90139-x. [DOI] [PubMed] [Google Scholar]
- Cohen S. S., Morgan S., Streibel E. The polyamine content of the tRNA of E. coli. Proc Natl Acad Sci U S A. 1969 Oct;64(2):669–676. doi: 10.1073/pnas.64.2.669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deutscher M. P., Chambon P., Konberg A. Biochemical studies of bacterial sporulation and germination. XI. Protein-synthesizing systems from vegetative cells and spores of Bacillus megaterium. J Biol Chem. 1968 Oct 10;243(19):5117–5125. [PubMed] [Google Scholar]
- Fukuma I., Cohen S. S. Polyamine synthesis and accumulation in Escherichia coli infected with bacteriophage R17. J Virol. 1973 Dec;12(6):1259–1264. doi: 10.1128/jvi.12.6.1259-1264.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gibson W., Roizman B. Compartmentalization of spermine and spermidine in the herpes simplex virion. Proc Natl Acad Sci U S A. 1971 Nov;68(11):2818–2821. doi: 10.1073/pnas.68.11.2818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HERSHEY A. D. Some minor components of bacteriophage T2 particles. Virology. 1957 Oct;4(2):237–264. doi: 10.1016/0042-6822(57)90061-2. [DOI] [PubMed] [Google Scholar]
- HOLLEY R. W., APGAR J., EVERETT G. A., MADISON J. T., MARQUISEE M., MERRILL S. H., PENSWICK J. R., ZAMIR A. STRUCTURE OF A RIBONUCLEIC ACID. Science. 1965 Mar 19;147(3664):1462–1465. doi: 10.1126/science.147.3664.1462. [DOI] [PubMed] [Google Scholar]
- Martin R. G., Ames B. N. THE EFFECT OF POLYAMINES AND OF POLY U SIZE ON PHENYLALANINE INCORPORATION. Proc Natl Acad Sci U S A. 1962 Dec;48(12):2171–2178. doi: 10.1073/pnas.48.12.2171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- NATHANS D., LIPMANN F. Amino acid transfer from aminoacyl-ribonucleic acids to protein on ribosomes of Escherichia coli. Proc Natl Acad Sci U S A. 1961 Apr 15;47:497–504. doi: 10.1073/pnas.47.4.497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- OHTAKA Y., SPIEGELMAN S. TRANSLATIONAL CONTROL OF PROTEIN SYNTHESIS IN A CELL-FREE SYSTEM DIRECTED BY A POLYCISTRONIC VIRAL RNA. Science. 1963 Oct 25;142(3591):493–497. doi: 10.1126/science.142.3591.493. [DOI] [PubMed] [Google Scholar]
- Pochon F., Cohen S. S. 4-Thiouridine and the conformation of E. coli tRNA induced by spermidine. Biochem Biophys Res Commun. 1972 May 26;47(4):720–726. doi: 10.1016/0006-291x(72)90551-7. [DOI] [PubMed] [Google Scholar]
- Raina A., Cohen S. S. Polyamines and RNA synthesis in a polyauxotrophic strain of E. coli. Proc Natl Acad Sci U S A. 1966 Jun;55(6):1587–1593. doi: 10.1073/pnas.55.6.1587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seiler N., Werner G., Fischer H. A., von Bernd Knötgen M., Hinz H. Der Einfluss von Nahrungsentzug, Phenobarbital und Perfluorbuttersäure auf den Nucleinsäuren-, Spermin- und Spermidingehalt der Mäuseleber. Hoppe Seylers Z Physiol Chem. 1969 Jun;350(6):676–682. [PubMed] [Google Scholar]
- Shortridge K. F., Stevens L. Spermine and spermidine--polyamine components of human type 5 adenovirions. Microbios. 1973;7(25):61–68. [PubMed] [Google Scholar]
- Slater D. W., Spiegelman S. An estimation of genetic messages in the unfertilized echinoid egg. Proc Natl Acad Sci U S A. 1966 Jul;56(1):164–170. doi: 10.1073/pnas.56.1.164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takeda Y. Polyamines and protein synthesis. II. The shift in optimal concentration of Mg2+ by polyamines in the MS2 phage RNA-directed polypeptide synthesis. Biochim Biophys Acta. 1969 Mar 18;179(1):232–234. doi: 10.1016/0005-2787(69)90140-3. [DOI] [PubMed] [Google Scholar]
- de Wachter R., Merregaert J., Vandenberghe A., Contreras R., Fiers W. Studies on the bacteriophage MS2. The untranslated 5'-terminal nucleotide sequence preceding the first cistron. Eur J Biochem. 1971 Oct 14;22(3):400–414. doi: 10.1111/j.1432-1033.1971.tb01558.x. [DOI] [PubMed] [Google Scholar]


