Abstract
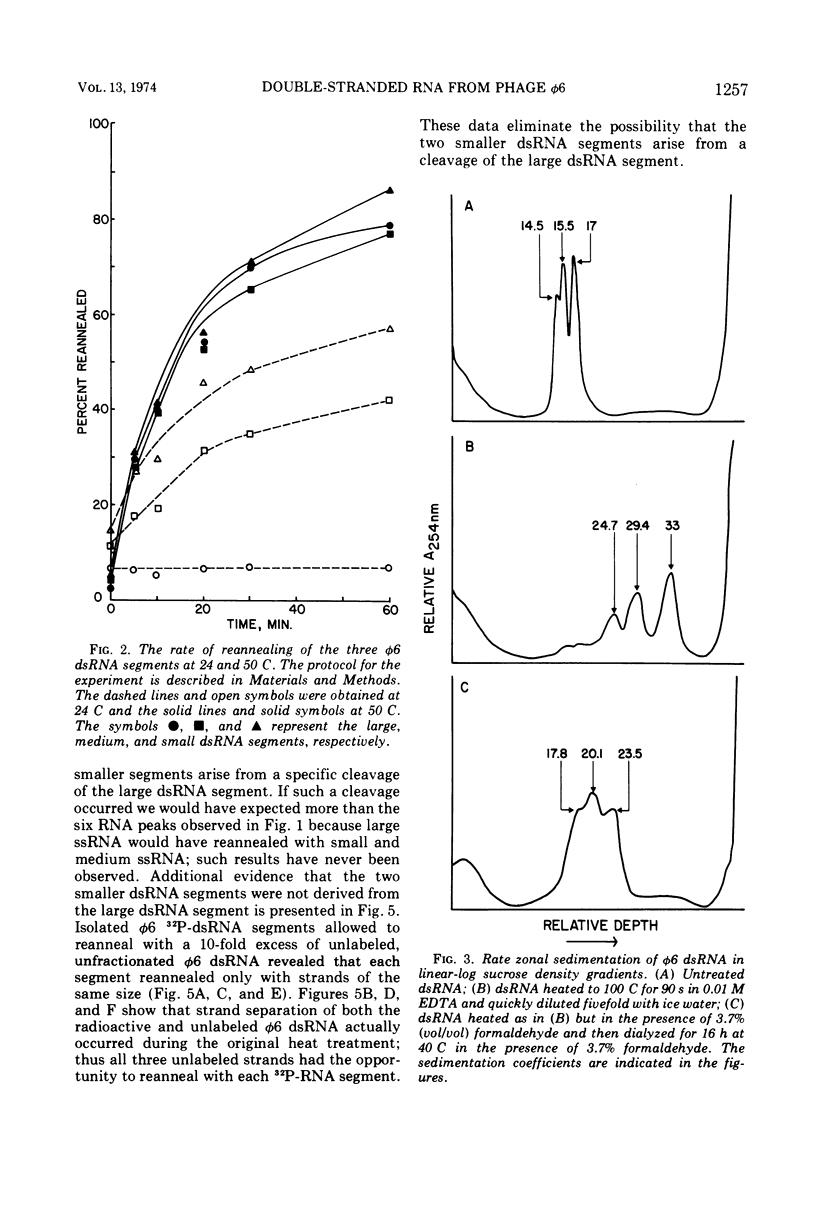
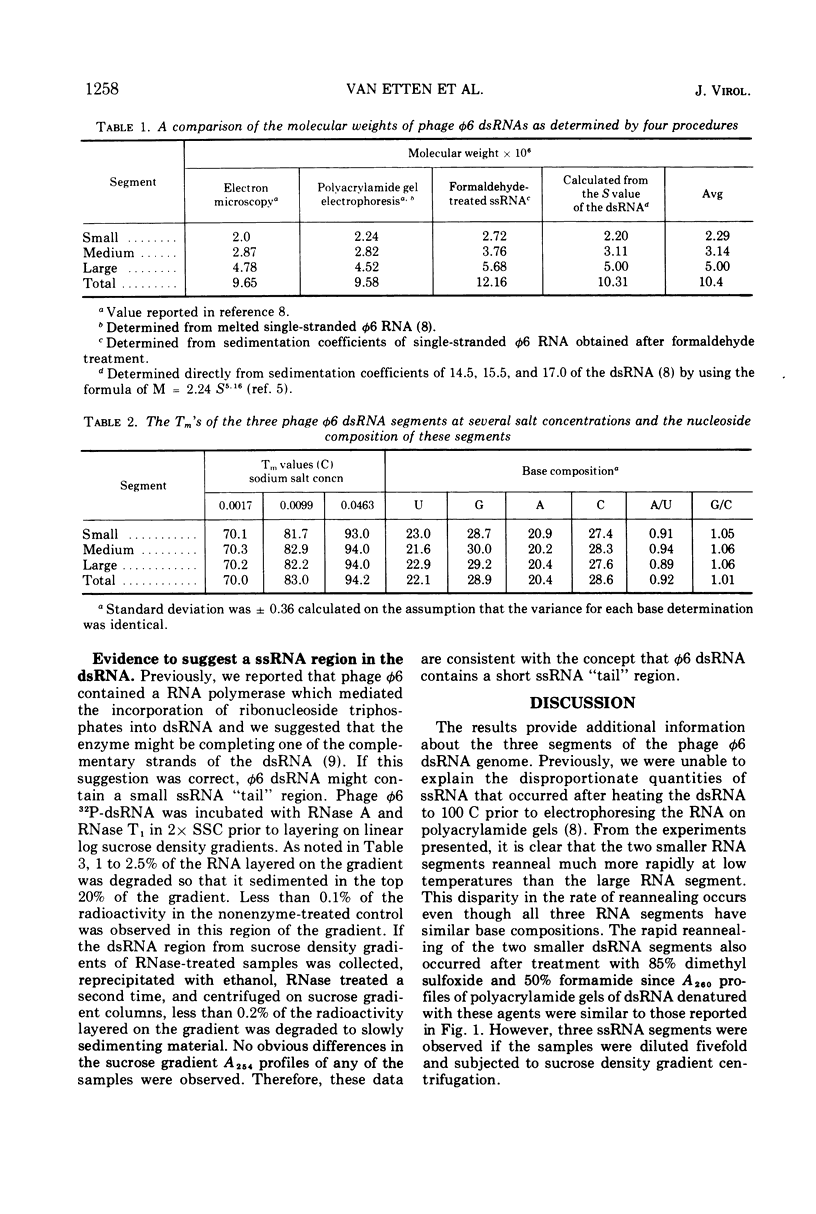
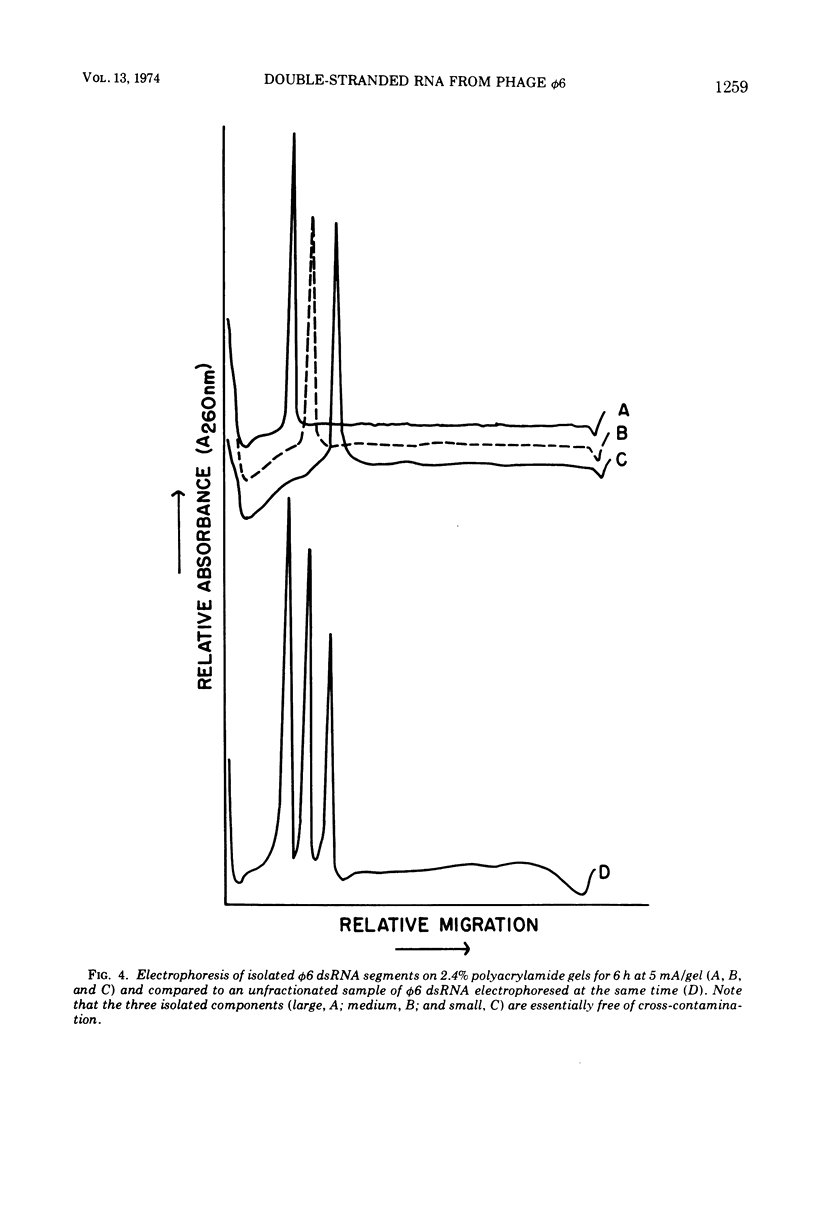
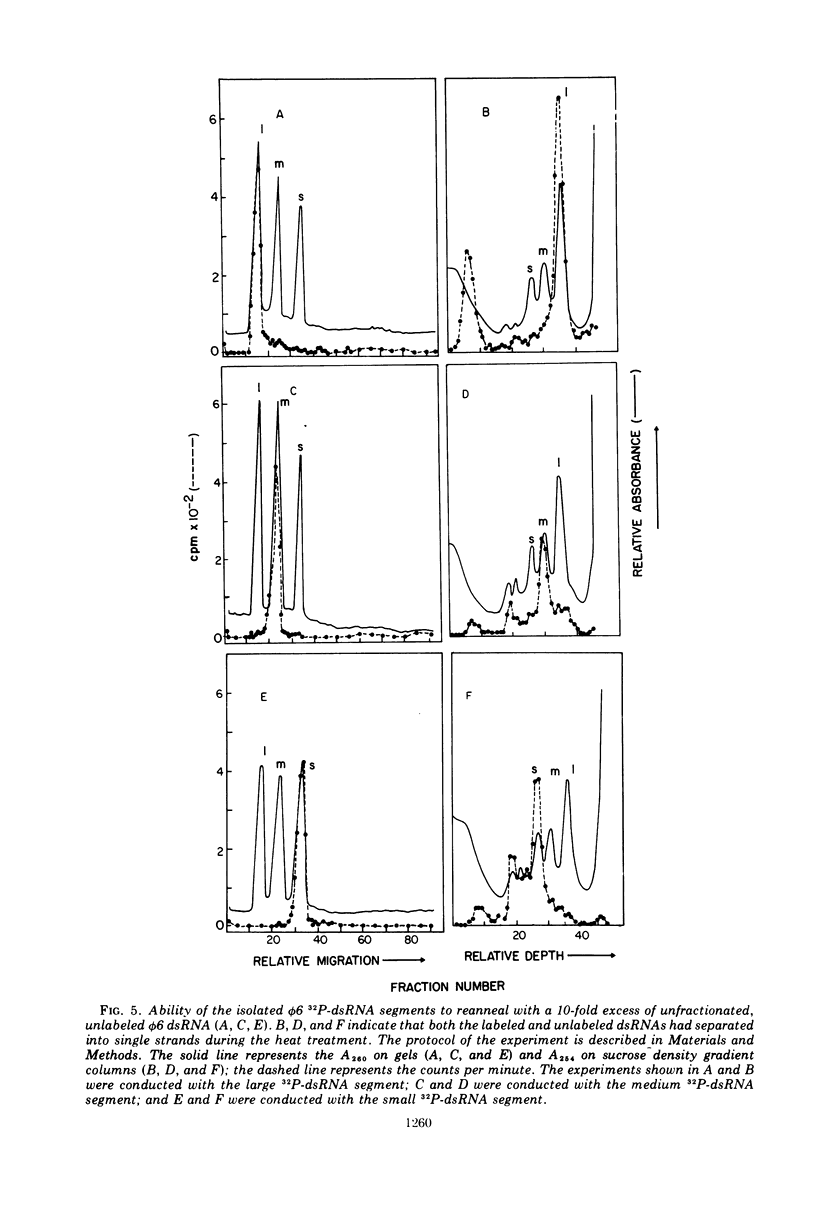
The three double-stranded ribonucleic acid (dsRNA) segments of the bacteriophage φ6 were isolated and shown to have similar melting temperatures and base compositions. RNA:RNA hybridization experiments with the isolated segments eliminate the possibility that the two smaller dsRNA segments arise from a cleavage of the large dsRNA segment. The two smaller RNA segments reanneal rapidly even at low temperatures; in contrast, the large dsRNA reannealed only at higher temperatures. Evidence is also presented which suggests that the dsRNAs may contain a short single-stranded RNA tail.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bishop D. H., Claybrook J. R., Spiegelman S. Electrophoretic separation of viral nucleic acids on polyacrylamide gels. J Mol Biol. 1967 Jun 28;26(3):373–387. doi: 10.1016/0022-2836(67)90310-5. [DOI] [PubMed] [Google Scholar]
- Brakke M. K., Van Pelt N. Linear-log sucrose gradients for estimating sedimentation coefficients of plant viruses and nucleic acids. Anal Biochem. 1970 Nov;38(1):56–64. doi: 10.1016/0003-2697(70)90155-7. [DOI] [PubMed] [Google Scholar]
- Brakke M. K., Van Pelt N. Properties of infectious ribonucleic acid from wheat streak mosaic virus. Virology. 1970 Nov;42(3):699–706. doi: 10.1016/0042-6822(70)90315-6. [DOI] [PubMed] [Google Scholar]
- Diener T. O., Schneider I. R. Virus degradation and nucleic acid release in single-phase phenol systems. Arch Biochem Biophys. 1968 Mar 20;124(1):401–412. doi: 10.1016/0003-9861(68)90344-5. [DOI] [PubMed] [Google Scholar]
- Franklin R. M. Replication of bacteriophage ribonucleic acid: some physical properties of single-stranded, double-stranded, and branched viral ribonucleic acid. J Virol. 1967 Feb;1(1):64–75. doi: 10.1128/jvi.1.1.64-75.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Katz L., Penman S. The solvent denaturation of double-stranded RNA from poliovirus infected HeLa cells. Biochem Biophys Res Commun. 1966 May 25;23(4):557–560. doi: 10.1016/0006-291x(66)90765-0. [DOI] [PubMed] [Google Scholar]
- Semancik J. S., Vidaver A. K., Van Etten J. L. Characterization of segmented double-helical RNA from bacteriophage phi6. J Mol Biol. 1973 Aug 25;78(4):617–625. doi: 10.1016/0022-2836(73)90283-0. [DOI] [PubMed] [Google Scholar]
- Van Etten J. L., Vidaver A. K., Koski R. K., Semancik J. S. RNA polymerase activity associated with bacteriophage phi 6. J Virol. 1973 Sep;12(3):464–471. doi: 10.1128/jvi.12.3.464-471.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vidaver A. K., Koski R. K., Van Etten J. L. Bacteriophage phi6: a Lipid-Containing Virus of Pseudomonas phaseolicola. J Virol. 1973 May;11(5):799–805. doi: 10.1128/jvi.11.5.799-805.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wood H. A. Viruses with double-stranded RNA genomes. J Gen Virol. 1973 Jun;20(Suppl):61–85. doi: 10.1099/0022-1317-20-Supplement-61. [DOI] [PubMed] [Google Scholar]
- Zaitlin M., Hariharasubramanian V. An improvement in a procedure for counting tritium and carbon-14 in polyacrylamide gels. Anal Biochem. 1970 May;35(1):296–297. doi: 10.1016/0003-2697(70)90038-2. [DOI] [PubMed] [Google Scholar]


