Abstract
Revertants of Kirsten sarcoma virus transformed nonproducer BALB/3T3 cells (KA31 cells) were isolated after exposing the transformed cells to 5-fluorodeoxyuridine at high cell density, or when suspended in methylcellulose. Revertants were also isolated by treating KA31 cells with the lectin, concanavalin A, which is manyfold more toxic to transformed cells than for normal cells. The revertants resemble BALB/3T3 cells in their morphology and growth characteristics in that they have a low saturation density, fail to grow in 1% calf serum or when suspended in methylcellulose, and cease to synthesize DNA after reaching their saturation density. Infection by murine leukemia virus rescues Kirsten sarcoma virus from only the concanavalin-A-selected variants, though all the revertants are susceptible to infection by leukemia virus. The concanavalin A revertants also become transformed after infection with murine leukemia virus. All the revertants can be transformed by Kirsten sarcoma virus but not by simian virus 40.
Full text
PDF


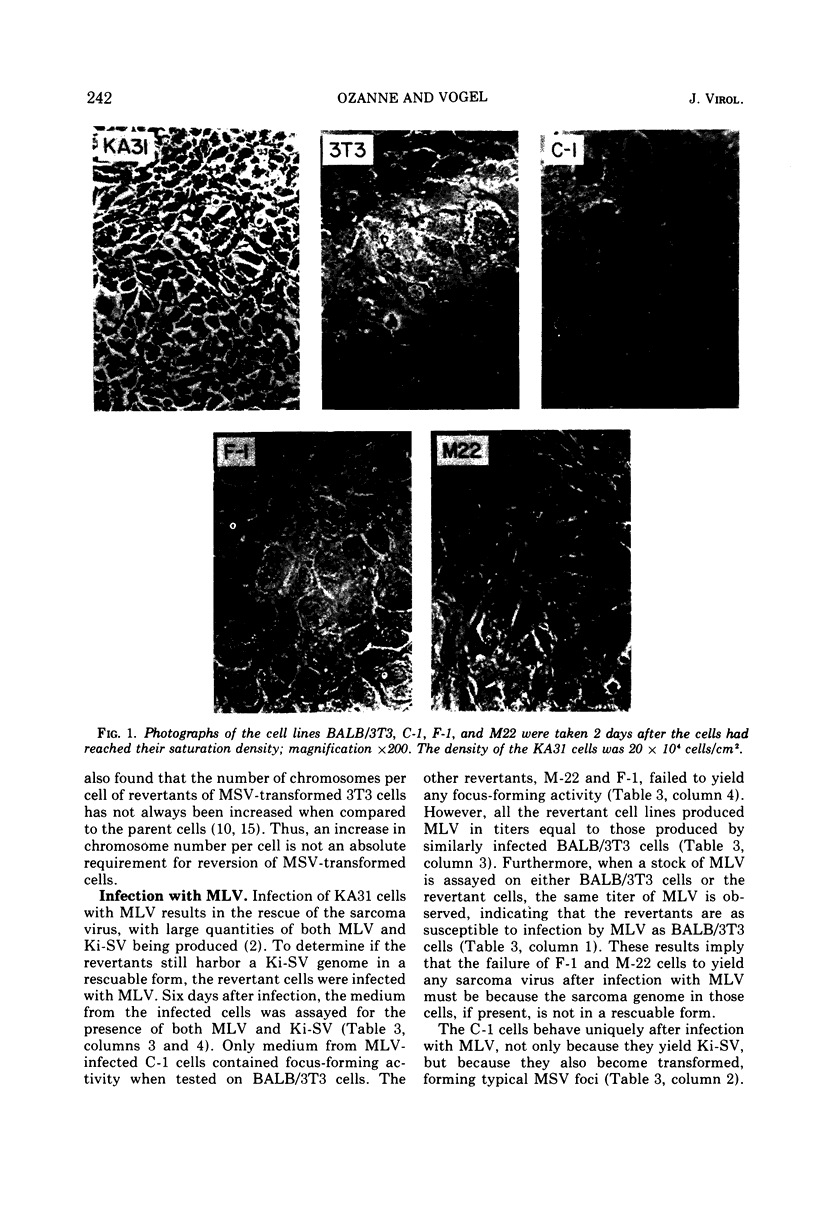
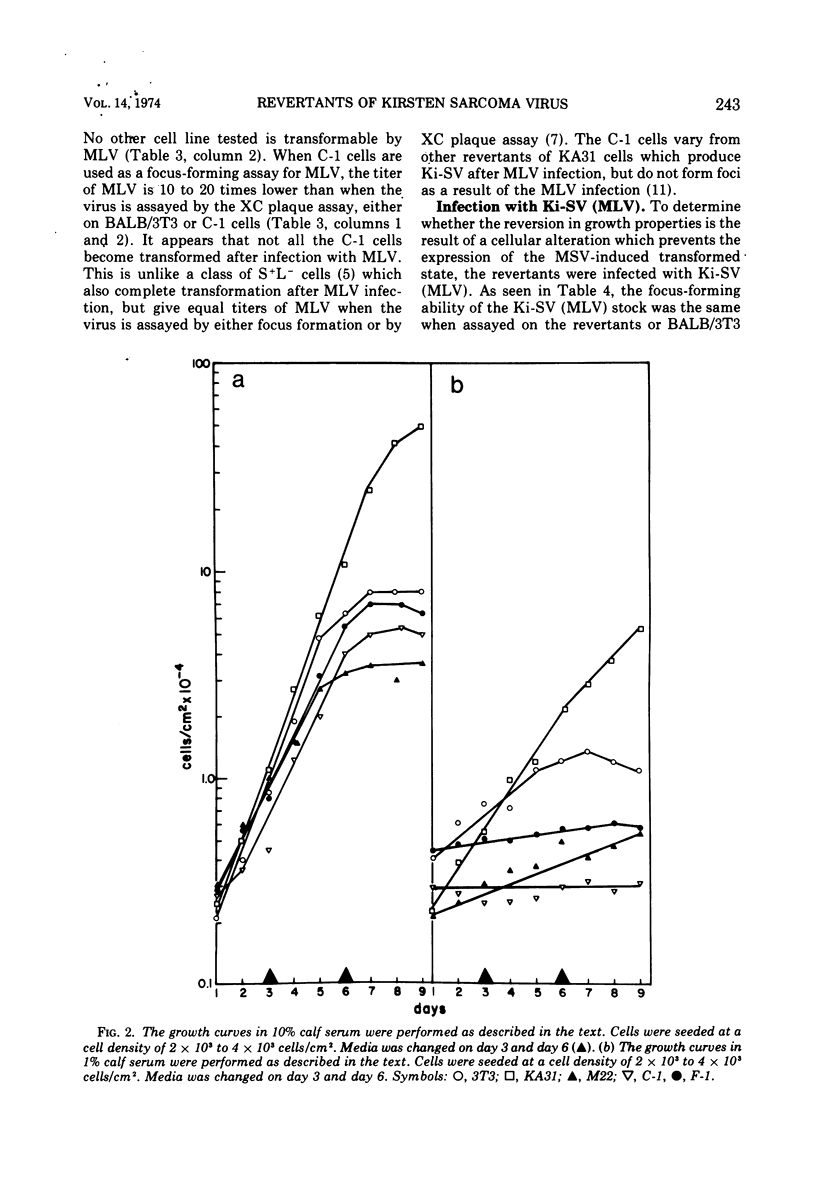
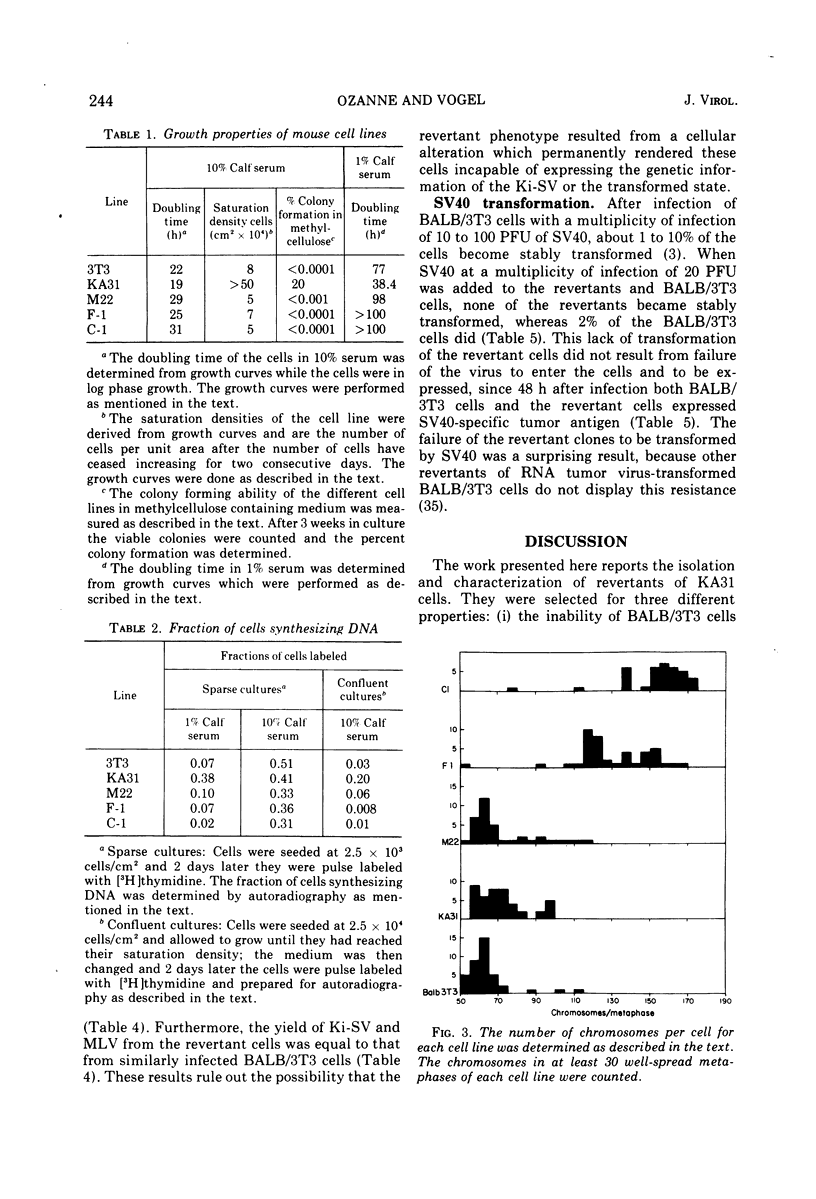




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aaronson S. A. Chemical induction of focus-forming virus from nonproducer cells transformed by murine sarcoma virus. Proc Natl Acad Sci U S A. 1971 Dec;68(12):3069–3072. doi: 10.1073/pnas.68.12.3069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aaronson S. A., Rowe S. P. Nonproducer clones of murine sarcoma virus transformed BALB-3T3 cells. Virology. 1970 Sep;42(1):9–19. doi: 10.1016/0042-6822(70)90233-3. [DOI] [PubMed] [Google Scholar]
- Aaronson S. A., Todaro G. J. Development of 3T3-like lines from Balb-c mouse embryo cultures: transformation susceptibility to SV40. J Cell Physiol. 1968 Oct;72(2):141–148. doi: 10.1002/jcp.1040720208. [DOI] [PubMed] [Google Scholar]
- Ball J., McCarter J. A., Sunderland S. M. Evidence for helper independent murine sarcoma virus. I. Segregation of replication-defective and transformation-defective viruses. Virology. 1973 Nov;56(1):268–284. doi: 10.1016/0042-6822(73)90305-x. [DOI] [PubMed] [Google Scholar]
- Bassin R. H., Phillips L. A., Kramer M. J., Haapala D. K., Peebles P. T., Nomura S., Fischinger P. J. Transformation of mouse 3T3 cells by murine sarcoma virus: release of virus-like particles in the absence of replicating murine leukemia helper virus. Proc Natl Acad Sci U S A. 1971 Jul;68(7):1520–1524. doi: 10.1073/pnas.68.7.1520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bassin R. H., Tuttle N., Fischinger P. J. Isolation of murine sarcoma virus-transformed mouse cells which are negative for leukemia virus from agar suspension cultures. Int J Cancer. 1970 Jul 15;6(1):95–107. doi: 10.1002/ijc.2910060114. [DOI] [PubMed] [Google Scholar]
- Bassin R. H., Tuttle N., Fischinger P. J. Rapid cell culture assay technic for murine leukaemia viruses. Nature. 1971 Feb 19;229(5286):564–566. doi: 10.1038/229564b0. [DOI] [PubMed] [Google Scholar]
- Duc-Nguyen H. Enhancing effect of diethylaminoethyl-dextran on the focus-forming titer of a murine sarcoma virus (Harvey strain). J Virol. 1968 Jun;2(6):643–644. doi: 10.1128/jvi.2.6.643-644.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fischinger P. J., Nomura S., Peebles P. T., Haapala D. K., Bassin R. H. Reversion of murine sarcoma virus transformed mouse cells: variants without a rescuable sarcoma virus. Science. 1972 Jun 2;176(4038):1033–1035. doi: 10.1126/science.176.4038.1033. [DOI] [PubMed] [Google Scholar]
- Gazdar A. F., Stull H. B., Chopra H. C., Ikawa Y. Properties of flat variants of murine sarcoma virus transformed non-producer cells isolated after high-temperature passage. Int J Cancer. 1974 Feb 15;13(2):219–226. doi: 10.1002/ijc.2910130209. [DOI] [PubMed] [Google Scholar]
- Greenberger J. S., Aaronson S. A. Morphologic revertants of murine sarcoma virus transformed nonproducer BALB-3T3: selective techniques for isolation and biologic properties in vitro and in vivo. Virology. 1974 Feb;57(2):339–346. doi: 10.1016/0042-6822(74)90173-1. [DOI] [PubMed] [Google Scholar]
- Hatanaka M., Klein R., Lomg C. W., Gilden R. Mutants of nonproducer cell lines transformed by murine sarcoma virus. II. Relationship of tumorigenicity to presence of viral markers and rescuable sarcoma genome. J Exp Med. 1973 Aug 1;138(2):364–372. doi: 10.1084/jem.138.2.364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hatanaka M., Klein R., Toni R., Walker J., Gilden R. Mutants of nonproducer cell lines transformed by murine sarcoma viruses. I. Induction, isolation, particle production, and tumorigenicity. J Exp Med. 1973 Aug 1;138(2):356–363. doi: 10.1084/jem.138.2.356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hitotsumachi S., Rabinowitz Z., Sachs L. Ciromosomal control of reversion in transformed cells. Nature. 1971 Jun 25;231(5304):511–514. doi: 10.1038/231511a0. [DOI] [PubMed] [Google Scholar]
- Jainchill J. L., Aaronson S. A., Todaro G. J. Murine sarcoma and leukemia viruses: assay using clonal lines of contact-inhibited mouse cells. J Virol. 1969 Nov;4(5):549–553. doi: 10.1128/jvi.4.5.549-553.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jainchill J. L., Todaro G. J. Stimulation of cell growth in vitro by serum with and without growth factor. Relation to contact inhibition and viral transformation. Exp Cell Res. 1970 Jan;59(1):137–146. doi: 10.1016/0014-4827(70)90632-4. [DOI] [PubMed] [Google Scholar]
- Nomura S., Fischinger P. J., Mattern C. F., Gerwin B. I., Dunn K. J. Revertants of mouse cells transformed by murine sarcoma virus. II. Flat variants induced by fluordeoxyuridine and colcemid. Virology. 1973 Nov;56(1):152–163. doi: 10.1016/0042-6822(73)90294-8. [DOI] [PubMed] [Google Scholar]
- Nomura S., Fischinger P. J., Mattern C. F., Peebles P. T., Bassin R. H., Friedman G. P. Revertants of mouse cells transformed by murine sarcoma virus. I. Characterization of flat and transformed sublines without a rescuable murine sarcoma virus. Virology. 1972 Oct;50(1):51–64. doi: 10.1016/0042-6822(72)90345-5. [DOI] [PubMed] [Google Scholar]
- Ozanne B., Sharp P. A., Sambrook J. Transcription of simian virus 40. II. Hybridization of RNA extracted from different lines of transformed cells to the separated strands of simian virus 40 DNA. J Virol. 1973 Jul;12(1):90–98. doi: 10.1128/jvi.12.1.90-98.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ozanne B. Variants of simian virus 40-transformed 3T3 cells that are resistant to concanavalin A. J Virol. 1973 Jul;12(1):79–89. doi: 10.1128/jvi.12.1.79-89.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pollack R. E., Green H., Todaro G. J. Growth control in cultured cells: selection of sublines with increased sensitivity to contact inhibition and decreased tumor-producing ability. Proc Natl Acad Sci U S A. 1968 May;60(1):126–133. doi: 10.1073/pnas.60.1.126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pollack R., Vogel A. Isolation and characterization of revertant cell lines. II. Growth control of a polyploid revertant line derived from SV40-transformed 3T3 mouse cells. J Cell Physiol. 1973 Aug;82(1):93–100. doi: 10.1002/jcp.1040820111. [DOI] [PubMed] [Google Scholar]
- Pollack R., Wolman S., Vogel A. Reversion of virus-transformed cell lines: hyperploidy accompanies retention of viral genes. Nature. 1970 Dec 5;228(5275):938–passim. doi: 10.1038/228938a0. [DOI] [PubMed] [Google Scholar]
- Renger H. C. Retransformation of ts SV40 transformants by murine sarcoma virus at non-permissive temperature. Nat New Biol. 1972 Nov 1;240(96):19–21. doi: 10.1038/newbio240019a0. [DOI] [PubMed] [Google Scholar]
- Rowe W. P., Pugh W. E., Hartley J. W. Plaque assay techniques for murine leukemia viruses. Virology. 1970 Dec;42(4):1136–1139. doi: 10.1016/0042-6822(70)90362-4. [DOI] [PubMed] [Google Scholar]
- Salzberg S., Green M. Surface alterations of cells carrying RNA tumour virus genetic information. Nat New Biol. 1972 Nov 22;240(99):116–118. doi: 10.1038/newbio240116a0. [DOI] [PubMed] [Google Scholar]
- Sambrook J. Transformation by polyoma virus and simian virus 40. Adv Cancer Res. 1972;16:141–180. [PubMed] [Google Scholar]
- Scher C. D., Nelson-Rees W. A. Direct isolation and characterization of "flat" SV40-transformed cells. Nat New Biol. 1971 Oct 27;233(43):263–265. doi: 10.1038/newbio233263a0. [DOI] [PubMed] [Google Scholar]
- Scolnick E. M., Rands E., Williams D., Parks W. P. Studies on the nucleic acid sequences of Kirsten sarcoma virus: a model for formation of a mammalian RNA-containing sarcoma virus. J Virol. 1973 Sep;12(3):458–463. doi: 10.1128/jvi.12.3.458-463.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scolnick E. M., Stephenson J. R., Aaronson S. A. Isolation of temperature-sensitive mutants of murine sarcoma virus. J Virol. 1972 Oct;10(4):653–657. doi: 10.1128/jvi.10.4.653-657.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stephenson J. R., Aaronson S. A. Antigenic properties of murine sarcoma virus-transformed BALB-3T3 nonproducer cells. J Exp Med. 1972 Mar 1;135(3):503–515. doi: 10.1084/jem.135.3.503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stephenson J. R., Aaronson S. A. Murine sarcoma and leukemia viruses: genetic differences determined by RNA-DNA hybridization. Virology. 1971 Nov;46(2):480–484. doi: 10.1016/0042-6822(71)90048-1. [DOI] [PubMed] [Google Scholar]
- Stephenson J. R., Reynolds R. K., Aaronson S. A. Characterization of morphologic revertants of murine and avian sarcoma virus-transformed cells. J Virol. 1973 Feb;11(2):218–222. doi: 10.1128/jvi.11.2.218-222.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stephenson J. R., Scolnick E. M., Aaronson S. A. Genetic stability of the sarcoma viruses in murine and avian sarcoma virus-transformed nonproducer cells. Int J Cancer. 1972 May 15;9(3):577–583. doi: 10.1002/ijc.2910090314. [DOI] [PubMed] [Google Scholar]
- Stoker M. Abortive transformation by polyoma virus. Nature. 1968 Apr 20;218(5138):234–238. doi: 10.1038/218234a0. [DOI] [PubMed] [Google Scholar]
- Vogel A., Pollack R. Isolation and characterization of revertant cell lines. IV. Direct selection of serum-revertant sublines of SV40-transformed 3T3 mouse cells. J Cell Physiol. 1973 Oct;82(2):189–198. doi: 10.1002/jcp.1040820207. [DOI] [PubMed] [Google Scholar]
- Vogel A., Risser R., Pollack R. Isolation and characterization of revertant cell lines. 3. Isolation of density-revertants of SV40-transformed 3T3 cells using colchicine. J Cell Physiol. 1973 Oct;82(2):181–188. doi: 10.1002/jcp.1040820206. [DOI] [PubMed] [Google Scholar]
- Wyke J. A method of isolating cells incapable of multiplication in suspension culture. Exp Cell Res. 1971 May;66(1):203–208. doi: 10.1016/s0014-4827(71)80030-7. [DOI] [PubMed] [Google Scholar]



