Abstract
Recent epidemiologic data show that low serum cholesterol level as well as statin use is associated with a decreased risk of developing aggressive or advanced prostate cancer, suggesting a role for cholesterol in aggressive prostate cancer development. Intracellular cholesterol promotes prostate cancer progression as a substrate for de novo androgen synthesis and through regulation of AKT signaling. By performing next-generation sequencing-based DNA methylome analysis, we have discovered marked hypermethylation at the promoter of the major cellular cholesterol efflux transporter, ABCA1, in LNCaP prostate cancer cells. ABCA1 promoter hypermethylation renders the promoter unresponsive to trans-activation and leads to elevated cholesterol levels in LNCaP. ABCA1 promoter hypermethylation is enriched in intermediate to high grade prostate cancers and not detectable in benign prostate. Remarkably, ABCA1 down-regulation is evident in all prostate cancers examined, and expression levels are inversely correlated with Gleason grade. Our results suggest cancer-specific ABCA1 hypermethylation and loss of protein expression direct high intracellular cholesterol levels and hence contribute to an environment conducive to tumor progression.
Keywords: ABCA1, cholesterol, prostate cancer, DNA methylation, statins
Introduction
Despite detection of prostate cancer at earlier stages and advances in the treatment of local as well as metastatic disease, there will still be an estimated 28,170 deaths due to prostate cancer in 2012 (1). Men who die of prostate cancer have cancers with aggressive pathologic features that increase the risk of tumor progression and metastasis, even if they undergo radical therapy with curative intent. As such, the investigation of novel strategies to prevent the development of aggressive or advanced prostate cancer will be critical to lowering the morbidity and mortality attributed to this disease.
Epidemiologic studies have described a positive correlation between high serum cholesterol level and prostate cancer aggressiveness (2, 3) as well as a protective effect of statin use in lowering the risk of advanced prostate cancer (2, 4–7). These reports, along with the discovery of de novo androgen synthesis in castration resistant prostate cancer (CRPC), have fueled a renewed interest in intratumoral cholesterol homeostasis due to the central role of cholesterol in steroidogenesis (8–10). Furthermore, excess intracellular cholesterol is incorporated into membrane lipid rafts, thereby stabilizing the raft structure and enhancing AKT signaling in prostate cancer cells (11–15). Therefore, examining how prostate cancer cells manipulate intracellular cholesterol content is important for understanding prostate cancer biology.
To understand the role that altered DNA methylation patterns play in prostate cancer development, we used MBD-isolated Genome Sequencing (MiGS) (16) to construct genome-wide DNA methylation profiles in the common prostate cell line models, PrEC, LNCaP, and DU 145. Using unbiased global analysis, we discovered dense hypermethylation in the 5’ regulatory regions of the cholesterol efflux transporter, ATP-binding cassette, sub-family A, member 1 (ABCA1), in LNCaP cells but not in PrEC or DU 145 cells. Given data suggesting that loss of cellular cholesterol homeostasis is important in prostate cancer, we proceeded to delineate the biological relevance of this epigenetic modification. We report that DNA hypermethylation at ABCA1 promoter in LNCaP cells effectively suppresses basal expression and prevents full induction by a trans-activator. Loss of ABCA1 expression results in retention of intracellular cholesterol. Furthermore, ABCA1 hypermethylation is exclusively detected in intermediate and high grade prostate cancers, suggesting that epigenetic inactivation of ABCA1 is involved in prostate cancer progression. Finally, significant decrease and complete loss of ABCA1 protein expression are evident in all prostate cancers examined in our study. Together, these findings indicate that ABCA1 is an important regulator of intracellular cholesterol levels in prostate epithelial cells and that its pervasive inactivation in prostate cancers likely provides a milieu favorable for tumor progression by permitting the accumulation of intracellular cholesterol.
Materials and methods
DNA methylation analysis
DNA methylome profiles for PrEC, LNCaP, and DU 145 were generated using MiGS as previously described (16). The sequencing reads generated and used in this manuscript are deposited in the NCBI Sequence Read Archive (SRA) under the accession number SRA049689.1. The raw sequencing reads for each sample were mapped to the reference human genome (UCSC Hg18) using Bowtie (17). Bisulfite sequencing and methylation-specific PCR (MSP) of the ABCA1 promoter was performed as previously described (18), and primers used are listed in Supplementary Table S1.
Cell culture, transfection, and luciferase assay
PrEC (Lonza, Walkersville, MD) was cultured in PrEGM according to the manufacturer’s instructions. LNCaP and DU 145 (ATCC, Manassas, VA) were cultured in RPMI 1640 supplemented with 10% FBS. All three cell lines were obtained directly from the cell banks, and the identities of the cell lines were verified per the cell banks’ protocols. Cells were treated with 10 µM T0901317 (Sigma, St. Louis, MO) for 24 hours or 5 µM 5-aza-2’-deoxycytidine (Sigma, St. Louis, MO) for 7 days, or a combination of the two compounds. ABCA1 promoter (−1132 to +112 relative to the transcription start site) was amplified by PCR and subcloned into the pGL4.20 (Promega, Madison, WI). Methylated ABCA1 promoter was in vitro DNA methylated using SssI (NEB, Ipswich, MA) and ligated into pGL4.20 prior to transfection. The reporter construct was co-transfected with pGL 4.74 vector into DU 145 cells using Nucleofection (Lonza, Walkersville, MD). Reporter luciferase activity was measured and normalized to control renilla luciferase activity for each sample. The mean ± SEM from triplicate experiments for each experimental group was plotted for comparisons. The different groups were compared using one way ANOVA with Bonferroni’s multiple comparison test.
Gene expression and Western blot
Expression of ABCA1 and GAPDH mRNA was measured by realtime RT-PCR as previously described (18), and primers used are listed in Supplementary Table S1. The relative fold change in expression was calculated using the 2−ΔΔCT method by normalizing to GAPDH mRNA expression in each sample and compared to LNCaP mock treated cells. The mean ± SEM from triplicate experiments for each experimental group was plotted, and comparisons between each sample group against LNCaP mock treated cells were performed using one way ANOVA with Bonferroni’s multiple comparison test. For Western blot analysis of ABCA1 and β actin (ACTB), 15 µg cell lysate per sample were resolved in 4–12% Bis-Tris gel (Life Technologies, Grand Island, NY), transferred onto nylon membranes, and probed with rabbit anti-ABCA1 (Novus Biologicals, Littleton, CO) and mouse anti- ACTB (Sigma, St. Louis, MO). For gene expression microarray analysis, total RNA was extracted with TRIzol (Life Technologies, Grand Island, NY), followed by DNase I treatment. The RNA samples were labeled and hybridized according to the manufacture’s protocol to the Illumina HumanRef-8 v3.0 expression beadchips (Illumina, San Diego, CA) in triplicates. The expression results generated and used in this manuscript are deposited with the Gene Expression Omnibus under the accession number GSE35401. Differential gene expression analysis was performed using the Illumina GenomeStudio v2009.1 (Illumina, San Diego, CA).
Cellular cholesterol analysis
For filipin staining, LNCaP and DU 145 cells were grown on glass coverslips, fixed in 3% paraformaldehyde, and stained with 50 µg/mL filipin (Sigma, St. Louis, MO). Images were acquired using QCapturePro software (QImaging, Surrey, Canada) at the designated magnifications and fixed aperture and exposure time for both cell lines. Biochemical quantification of intracellular cholesterol was performed as previously described (19). The different groups were compared using one way ANOVA with Bonferroni correction. For analysis of cholesterol efflux, cells were labeled with 0.5 µCi/mL [3H]-cholesterol in RPMI containing 1% FBS for 16 hours at 37°C. After labeling, cells were chased for 4 hours at 37°C in RPMI with or without acceptors (10 µg/mL APOA1 or 100 µg/mL HDL). At the end of this chase period, the radioactivity in the medium and cells was determined by liquid scintillation counting, and the percent efflux was calculated as 100 × (medium dpm)/(medium dpm + cell dpm). Percent efflux to acceptors was calculated as (percent efflux to acceptor)-(percent efflux to no acceptor). LNCaP treatment groups were compared to the mock treated sample using one way ANOVA with Dunnett’s correction. Unpaired t test with Welch’s correction was used to compare the T0901317-treated with the mock treated DU 145 cells.
Human tissue specimens
Prostate cancer tissues were obtained from patients treated with radical prostatectomy at Cleveland Clinic (Cleveland, OH). Benign prostate tissues were obtained from patients treated with radical cystoprostatectomy for either malignant or benign bladder disease at Cleveland Clinic (Cleveland, OH). All study specimens were collected under an approved Cleveland Clinic IRB protocol. All sections were retrieved and reviewed by dedicated genitourinary pathologists (C.M.G and S.M.F.) to confirm the original diagnosis. For MSP, formalin fixed paraffin embedded sections were de-paraffinized using xylene and rehydrated prior to genomic DNA extraction. 2 µg genomic DNA from each sample was bisulfite treated using the EpiTect bisulfite conversion kit (Qiagen, Hilden, Germany) in 3 independent experiments. Only samples that show consistent methylation in all 3 experiments were deemed to harbor ABCA1 promoter methylation. Immunohistochemistry was performed on 4 µm sections. Antigen retrieval was performed prior to incubation with a custom anti-ABCA1 rabbit polyclonal antibody raised against AA 104–125 in NP_005493.2 (Thermo, Rockford, IL), OmniMap secondary antibody (Ventana, Tucson, AZ), and ChromoMap DAB (Ventana, Tucson, AZ). ABCA1 staining patterns were evaluated by C.M.G. and S.M.F. The specificity of this custom antibody was tested using both Western blotting and immunohistochemistry staining of DU 145 and LNCaP cells (Supplementary Fig. S1). Scoring of ABCA1 staining was performed using the H-score system as previously described (20) with the scale set from 0 to 3. H-score comparisons were performed using the Mann-Whitney U-test and Kruskal-Wallis test, with p < 0.05 considered to be statistically significant. ABCA1 staining was independently analyzed by comparing the percentage of cancer cells stained positively for ABCA1 using the Kruskal-Wallis test, with p < 0.05 considered to be statistically significant.
Results
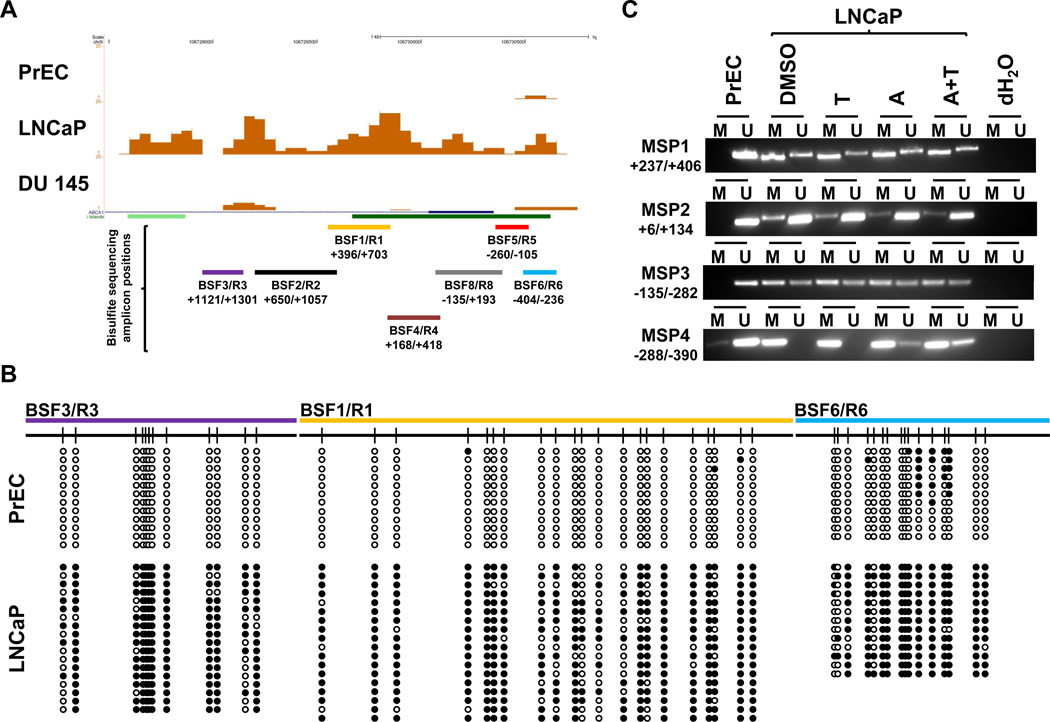
We assembled DNA methylome profiles for normal prostate epithelial cells, PrEC, and prostate cancer cell lines, LNCaP and DU 145, using MiGS. Unambiguously mapped sequencing reads were used for generating the individual DNA methylome profiles. At a false discovery rate of 5%, we identified the major cellular cholesterol efflux transporter, ABCA1, to be densely methylated in its 5’ regulatory region in LNCaP cells but not in PrEC or DU 145 cells (Fig. 1A).
Figure 1. DNA methylation analysis of ABCA1 5’ regulatory sequences in prostate cell lines.
(A) UCSC genome browser snapshot displaying the DNA methylation sequencing signals in PrEC, LNCaP, and DU 145 cells at the ABCA1 promoter region (UCSC Hg18, chr9:106,728,482–106,730,800). (B) Bisulfite sequencing validation in PrEC and LNCaP cells. Black circles represent methylated CpG sites, and white circles represent unmethylated CpG sites. (C) Methylation-specific PCR (MSP) results in PrEC and LNCaP cells either mock treated (DMSO), or treated with 10µM T0901317 for 24 hours (T), 5 µM 5-aza-2’-deoxycytidine for 7 days (A), or a combination of 5-aza-2’-deoxycytidine and T0901317 (A+T).
While the ABCA1 promoter region is one of several thousand novel differentially methylated loci among the three prostate cell lines, we focused on this gene because of its central role in intracellular cholesterol homeostasis. We verified this differential DNA methylation at the ABCA1 promoter by targeted bisulfite (BSF) sequencing in PrEC and LNCaP cells (Fig. 1B and Supplementary Fig. S2). We also assayed this region using methylation-specific PCR (MSP) (Fig. 1C). Both BSF sequencing and MSP results corroborated the robust DNA methylation at the ABCA1 promoter in LNCaP cells as detected by MiGS.
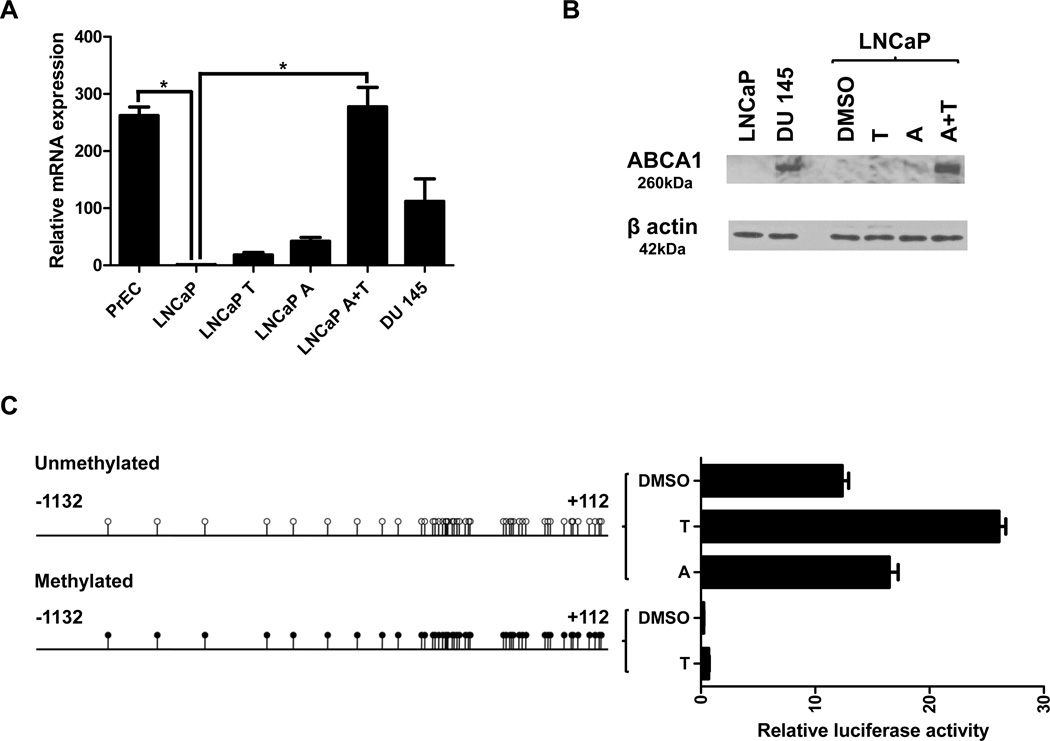
To examine the functional consequence of ABCA1 promoter methylation, we used RT-PCR to quantify ABCA1 mRNA expression in PrEC, LNCaP, and DU 145 cells (Fig. 2A). Compared with LNCaP cells, ABCA1 mRNA levels are at least 100-fold higher in PrEC and DU 145 cells where the ABCA1 promoter is not DNA methylated. This finding is consistent with transcriptional repression caused by ABCA1 promoter hypermethylation in LNCaP cells. We treated LNCaP cells with T0901317, a synthetic liver-x-receptor (LXR) α agonist known to induce ABCA1 transcription, and found that ABCA1 transcription was only modestly induced above baseline. Treatment of LNCaP cells with 5-aza-2’-deoxycytidine (5-aza), a demethylating agent, also did not strongly induce ABCA1 transcription. However, when the ABCA1 promoter in LNCaP is first demethylated with 5-aza, treatment with T0901317 resulted in robust activation of ABCA1 transcription to levels comparable to PrEC cells. We confirmed demethylation of ABCA1 promoter by 5-aza using MSP (Fig. 1C). These data demonstrate that ABCA1 promoter hypermethylation renders it unresponsive to trans-activation. Not surprisingly, the lack of messenger RNA corresponds to a lack of ABCA1 protein expression in LNCaP cells while DU 145 cells clearly express ABCA1 (Fig. 2B). Again, demethylation of the ABCA1 promoter with 5-aza followed by treatment with T0901317 resulted in robust ABCA1 protein expression in LNCaP. It is worth noting that minimal ABCA1 expression and severely limited induction by trans-activators in LNCaP cells have been independently reported by other groups without a mechanistic explanation (14, 21, 22).
Figure 2. Effects of promoter DNA methylation on basal expression and inducibility of ABCA1.
(A) Relative mRNA expression of ABCA1 in prostate cells. LNCaP cells were treated identically as in Figure 1. Data are represented as mean ± SEM from triplicate experiments and * indicates p < 0.05. (B) Western blot analysis of ABCA1 and β actin (ACTB) in LNCaP and DU 145 cells. LNCaP cells were treated identically as in Figure 1. (C) Luciferase reporter assay of the unmethylated and the methylated ABCA1 promoter in DU 145 cells. The cells containing the indicated reporter construct were either mock treated (DMSO) or treated with 10µM T0901317 (T) or 5 µM 5-aza-2’-deoxycytidine (A) for 24 hours. Data are represented as mean ± SEM from triplicate experiments. All pair-wise comparisons were statistically significant (p < 0.05) except for between the two methylated treatment groups.
Next, we ascertained the effect of ABCA1 promoter hypermethylation on transcriptional activity (Fig. 2C). An ABCA1 promoter/luciferase reporter construct was generated with sequences surrounding the ABCA1 transcription start site. A fully methylated version was produced by treating the ABCA1 promoter fragment with Sss I methylase and ligating it into the luciferase reporter construct prior to transfection. These vectors were transfected into DU 145 cells, which have the ability to express endogenous ABCA1. The unmethylated promoter expressed the luciferase reporter, and as expected, treatment with T0901317 resulted in a significant induction of reporter expression. Conversely, reporter activity from the methylated ABCA1 promoter was almost undetectable, and it was not induced by T0901317. These data show that promoter hypermethylation of ABCA1 is directly responsible for transcriptional repression and loss of responsiveness to activation by LXR agonist.
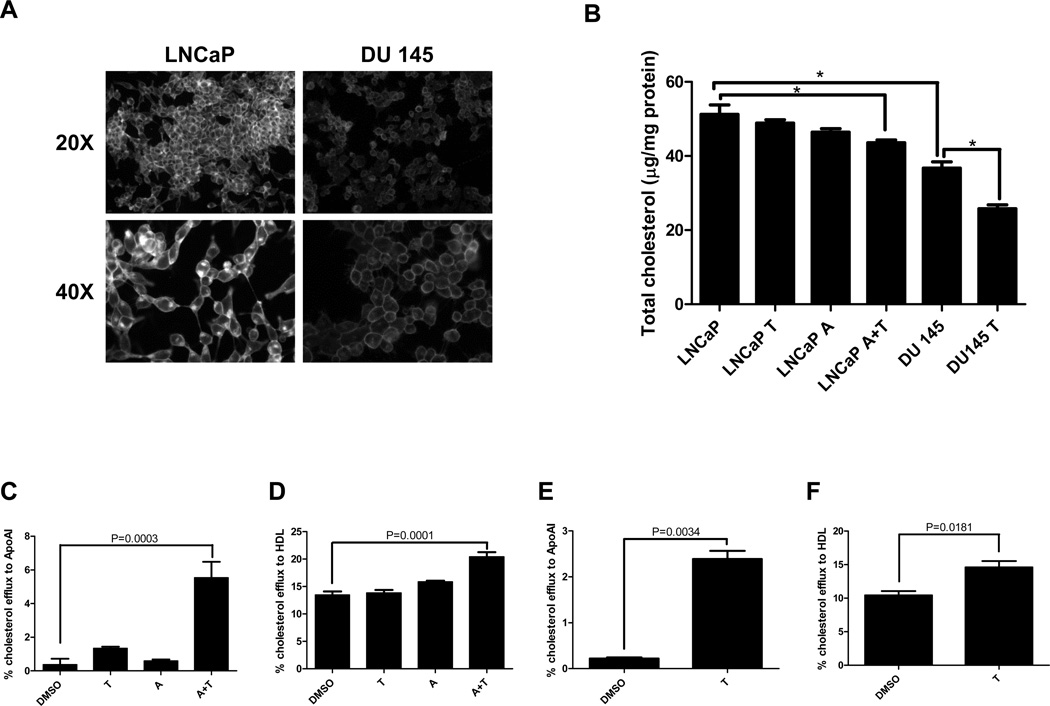
We examined the functional consequence of ABCA1 promoter hypermethylation and transcriptional silencing. LNCaP and DU 145 cells were subjected to filipin staining, which allows visualization of free cholesterol, the major unesterified sterol in mammalian cells (Fig. 3A). Fluorescent microscopy showed that LNCaP cells have significantly elevated basal intracellular cholesterol levels when compared with DU 145. Total cellular cholesterol content was quantified biochemically (Fig. 3B), which confirmed that LNCaP has a higher basal level of intracellular cholesterol than DU 145 (51.2 ± 4.4 µg/mg protein vs. 36.7 ± 3.0 µg/mg protein). When LNCaP was treated with either T0901317 or 5-aza alone, intracellular cholesterol did not decrease significantly. However, when LNCaP was treated with 5-aza prior to T0901317, intracellular cholesterol was significantly lower than untreated LNCaP cells (43.6 ± 1.4 µg/mg protein vs. 51.2 ± 4.4 µg/mg protein). As expected, treatment of DU 145 with only T0901317 resulted in decreased intracellular cholesterol. We assessed whether ABCA1 reactivation in LNCaP was responsible for the decrease in intracellular cholesterol content (Fig. 3C and D). We measured cholesterol efflux to apolipoprotein A-I (APOA1), which can accept cellular cholesterol only via ABCA1, and to HDL, which can accept cholesterol from both ABCA1 and other transporters such as SR-B1 and ABCG1. Using APOA1 as an acceptor, treatment of LNCaP with either T0901317 or 5-aza did not result in robust increases in cholesterol efflux. However, treatment with 5-aza followed by T0901317 led to a 2.5-fold increase in cholesterol efflux to APOA1. When HDL was used as an acceptor, the same overall trend was observed in cholesterol efflux in LNCaP after treatment with T0901317, 5-aza-, or the two drugs combined. However, the magnitude of increase in cholesterol efflux after treatment with 5-aza followed by T0901317 was significantly lower when compared with APOA1 as an acceptor. These data suggest that the decrease in cholesterol after treatment of LNCaP with 5-aza followed by T0901317 is mainly due to re-activation of ABCA1. Conversely, treatment of DU 145 with T0901317 alone led to a significant increase in cholesterol efflux to APOA1, suggesting that ABCA1 was readily inducible in the absence of promoter methylation (Fig. 3E and F). Treatment of DU 145 with T0901317 also resulted in a small, but statistically significant increase in cholesterol efflux to HDL. Since ABCG1 is the other major contributor of cellular cholesterol efflux, we also examined ABCG1 promoter methylation and expression in these cells. By MiGS analysis, the ABCG1 promoter is free of DNA methylation in PrEC, LNCaP, and DU 145 cells (Supplementary Fig. S3). All three cell lines expressed ABCG1 mRNA robustly with no statistically significant differences among them while ABCA1 expression showed previously validated differences by microarray analysis (Supplementary Table S2). Altogether, these data support the notion that hypermethylation and consequent loss of expression of ABCA1 in LNCaP cells contribute to the aberrant accumulation of intracellular cholesterol in these cancer cells.
Figure 3. Functional analysis of ABCA1 expression in prostate cancer cells.
(A) Representative fields of filipin staining for LNCaP and DU145. (B) Total cellular cholesterol content for LNCaP and DU 145 cells. LNCaP cells were treated identically as in Figure 1 while DU 145 cells were either mock treated (DMSO) or treated with 10 µM T0901317. (C–F) Cellular cholesterol efflux to APOA1 and HDL in LNCaP (C and D respectively) and DU 145 (E and F respectively) cells. Data are represented as mean ± SEM from triplicate experiments, and * indicates p < 0.05.
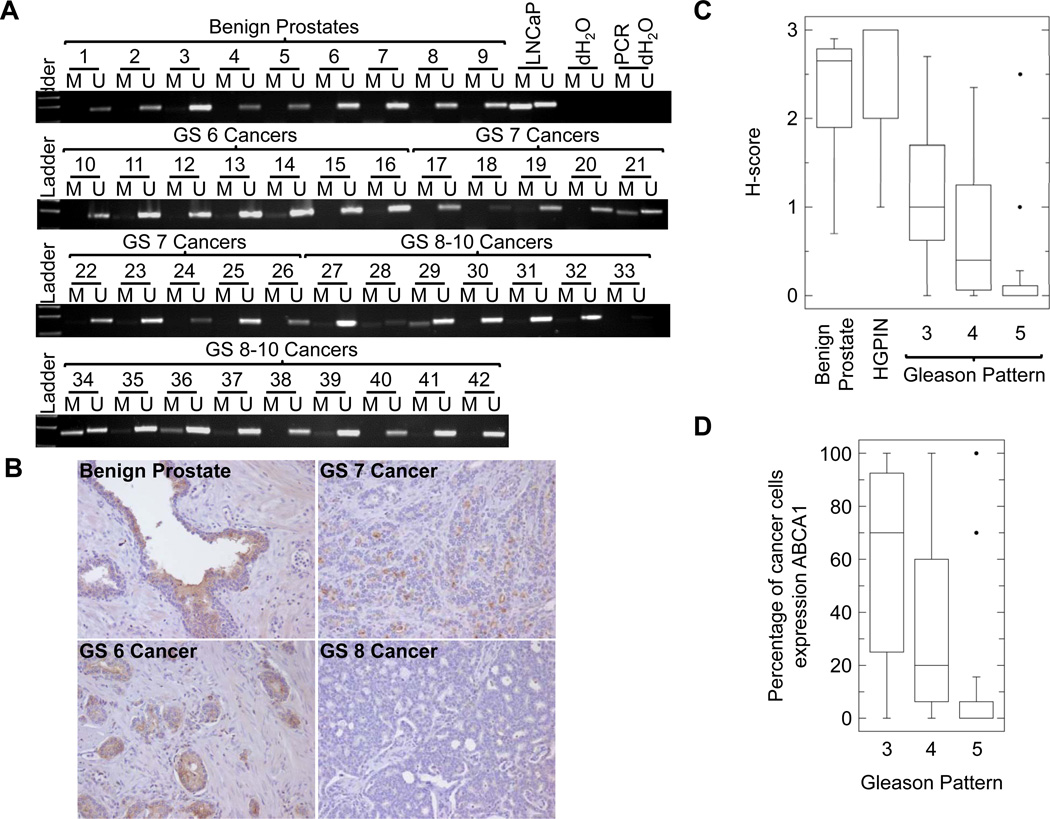
To assess the prevalence of ABCA1 hypermethylation in human prostate cancer, we performed MSP on DNA extracted from 9 benign prostatic tissue samples from cystoprostatectomy specimens and 33 prostate cancers. Of the 33 prostate cancer samples, 30 yielded high quality bisulfite converted DNA for this analysis. We did not find ABCA1 hypermethylation in any of the benign prostatic tissue samples; however, 4 of 30 prostate cancers (samples 21, 29, 34, and 36) showed ABCA1 hypermethylation (Fig. 4A). Notably, ABCA1 hypermethylation was only seen in men with intermediate and high risk prostate cancer (1/9 of Gleason score (GS) 7 cancers and 3/14 of GS 8–10 cancers). Biochemical recurrence after radical therapy was documented in all of these men. Furthermore, we investigated the expression of ABCA1 in prostate tissue by developing a custom antibody to the protein and performing immunohistochemistry on individual radical prostatectomy specimens and tissue microarrays containing prostate cancers (Fig. 4B). We used the H-score method to evaluate the ABCA1 expression in a semiquantitative fashion. There was significant heterogeneity of ABCA1 staining in each cancer specimen due to differences in tumor pattern. Thus, we determined the H-score of each of the following patterns observed: benign prostatic tissues (n=8), high grade prostatic intraepithelial neoplasia (HGPIN) (n=27), and Gleason patterns 3 (n=13), 4 (n=24), and 5 (n=14) (Fig. 4C). No significant difference was observed in ABCA1 expression between benign prostatic tissues and HGPIN (median H-score 2.65 vs. 3.00). ABCA1 expression was significantly lower for prostate cancers when compared with benign prostatic tissues (median H-score 0.35 vs. 3.00; p < 0.001; Mann-Whitney test). Moreover, there was an inverse correlation between ABCA1 expression and Gleason pattern. Both Gleason pattern 4 and 5 cancers had a lower median H-score when compared with Gleason pattern 3 cancer (0.40 and 0.00 vs. 1.00; p = 0.0017; Kruskal-Wallis test), and 71% of Gleason pattern 5 cancers completely lost ABCA1 expression. When we examined the percentage of cancer cells staining positively for ABCA1, we observed a statistically significant difference (p = 0.0013; Kruskal-Wallis test) among the Gleason patterns (Fig. 4D). Specifically, ranking by the percentage of cells expressing ABCA1, Gleason pattern 3 was the highest, Gleason pattern 4 was second, and Gleason pattern 5 was the lowest (median percentages 70%, 20%, and 0% respectively). These results demonstrate that ABCA1 hypermethylation is specific to prostate cancer, and decrease in ABCA1 expression is associated with tumor aggressiveness.
Figure 4. Analysis of ABCA1 promoter methylation and expression in radical prostatectomy specimens.
(A) Methylation-specific PCR (MSP) was performed using MSP1 primer set (Supplementary Table S1) on bisulfite converted genomic DNA extracted from benign prostates (1–9), GS 6 (10–16), GS 7 (17–26), and GS 8–10 (27–42) prostate cancer specimens. LNCaP DNA was included as a positive control. Samples 18, 28, and 33 did not yield sufficient bisulfite converted DNA to produce successful PCR results and therefore were excluded from further analysis. (B) ABCA1 immunohistochemistry on benign prostate, GS 6, GS 7, and GS 8 prostate cancers. (C) Box plots of H-scores for ABCA1 staining in benign prostatic tissues (n=8), high grade prostatic intraepithelial neoplasia (HGPIN) (n=27), Gleason pattern 3 (n=13), 4 (n=24), and 5 (n=14) tumors. The box shows the first quartile, median, and third quartile values. The whiskers show the minimum and maximum values. For Gleason pattern 5, outlier values, defined as three times the interquartile range, are present, and the whiskers denote 1.5 times the interquartile range with outliers plotted as individual black circles. The difference among the H-scores of Gleason patterns 3, 4, and 5 is statistically significant (p = 0.0017; Kruskal-Wallis test). (D) Box plots of percentages of cancer cells expressing ABCA1. Graphical representation is identical to Figure 4c. The difference among the percentages of cells staining positive for ABCA1 in Gleason patterns 3, 4, and 5 is statistically significant (p = 0.0013; Kruskal-Wallis test).
Discussion
Perturbation in cholesterol homeostasis is a well-known characteristic of cancer that was described more than fifty years ago (23, 24). Subsequently, anecdotal reports described a beneficial effect of cholesterol lowering agents in the management of prostate cancer (25). The introduction and widespread use of statins as cholesterol lowering agents in the prevention of heart disease allowed the collection of epidemiological data correlating prostate cancer risk and statin use. Although meta-analyses showed that statins had no effect on the overall risk of prostate cancer (26–29), other studies have shown that statin use is associated with a decreased risk of aggressive or advanced prostate cancer (6, 29–31). Importantly, these cancers are potentially life threatening even after radical treatment. Thus, focusing on preventing the development or progression of aggressive prostate cancer is of utmost importance, and cholesterol may provide an opportune target. Indeed, recent reports suggest that statin use protects against prostate cancer with adverse pathologic characteristics (32) and improves progression free survival in men undergoing radiation therapy (33, 34).
Based on these observations, after compiling the methylomes for the three prostate cell lines, we initially focused on candidates involved in cholesterol homeostasis. As discussed previously, cholesterol has two proposed roles in the development of advanced prostate cancer: serving as a substrate in de novo androgen synthesis in CRPC and enhancing AKT signaling by stabilizing lipid raft structure. However, the exact mechanism by which cholesterol accumulates inside the cancer cells is not clearly defined. In this study, we have identified ABCA1 promoter hypermethylation and subsequent transcriptional silencing as one mechanism that prostate cancer cells can use to maintain elevated intracellular cholesterol levels. Since intracellular cholesterol level is the net sum of uptake, synthesis, and efflux, disruption of a major transporter involved in efflux will result in intracellular cholesterol accumulation. We have demonstrated that this is the case through fluorescence microscopy as well as cholesterol quantification. When mechanisms responsible for cholesterol homeostasis are intact, excess cholesterol is converted to oxysterols which bind to LXR’s so that ABCA1 transcription is activated (35). We showed that in LNCaP cells, ABCA1 promoter hypermethylation prevents ABCA1 activation by the synthetic LXR agonist, T0901317, and demethylation of the promoter by 5-aza restores responsiveness to T0901317. As a result, cholesterol levels are significantly decreased when compared with untreated LNCaP cells or those treated with either agent alone.
When we examined human prostate tissue, we found that ABCA1 promoter hypermethylation was seen in prostate cancer but not benign prostatic tissue. Interestingly, this epigenetic alteration has a higher prevalence in intermediate and high grade cancers when compared with low grade cancers. Importantly, immunohistochemistry revealed that loss of ABCA1 expression is more prevalent in higher grade tumors than can be explained by promoter hypermethylation alone. These data suggest that ABCA1 inactivation may be important in the development of or progression to aggressive and/or advanced prostate cancer. Identifying the exact mechanism underlying ABCA1 inactivation is important, since promoter hypermethylation will render the gene unresponsive to LXR agonists but other mechanisms may not. Although it is presumed that statins protect against aggressive and advanced prostate cancers by inhibiting cholesterol synthesis, it would be interesting to see whether their use would be successful in prostate cancers with ABCA1 inactivation.
In summary, ABCA1 promoter hypermethylation and gene inactivation leads to the accumulation of cholesterol in prostate cancer cells. Thus, this cellular cholesterol efflux pathway may be an important determinant of prostate cancer aggressiveness and a potential therapeutic target.
Supplementary Material
Acknowledgements
We thank Paula Carver for her assistance in retrieving and sectioning the archival patient specimens and acknowledge the use of Cleveland Clinic Lerner Research Institute Genomics core facility.
Grant support
This work was partially funded by the Cleveland Clinic Research Program Committee (B.H.L.) and the National Institutes of Health (CA154356 for A.H.T. and HL098055 for J.D.S. and P.R.).
References
- 1.Siegel R, Naishadham D, Jemal A. Cancer statistics, 2012. CA Cancer J Clin. 2012;62(1):10–29. doi: 10.3322/caac.20138. [DOI] [PubMed] [Google Scholar]
- 2.Platz EA, Till C, Goodman PJ, Parnes HL, Figg WD, Albanes D, et al. Men with low serum cholesterol have a lower risk of high-grade prostate cancer in the placebo arm of the prostate cancer prevention trial. Cancer Epidemiol Biomarkers Prev. 2009;18(11):2807–2813. doi: 10.1158/1055-9965.EPI-09-0472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Batty GD, Kivimaki M, Clarke R, Davey Smith G, Shipley MJ. Modifiable risk factors for prostate cancer mortality in London: forty years of follow-up in the Whitehall study. Cancer Causes Control. 2011;22(2):311–318. doi: 10.1007/s10552-010-9691-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Solomon KR, Freeman MR. The complex interplay between cholesterol and prostate malignancy. The Urologic clinics of North America. 2011;38(3):243–259. doi: 10.1016/j.ucl.2011.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Mondul AM, Selvin E, De Marzo AM, Freedland SJ, Platz EA. Statin drugs, serum cholesterol, and prostate-specific antigen in the National Health and Nutrition Examination Survey 2001–2004. Cancer Causes Control. 2010;21(5):671–678. doi: 10.1007/s10552-009-9494-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Platz EA, Leitzmann MF, Visvanathan K, Rimm EB, Stampfer MJ, Willett WC, et al. Statin drugs and risk of advanced prostate cancer. J Natl Cancer Inst. 2006;98(24):1819–1825. doi: 10.1093/jnci/djj499. [DOI] [PubMed] [Google Scholar]
- 7.Farwell WR, D'Avolio LW, Scranton RE, Lawler EV, Gaziano JM. Statins and prostate cancer diagnosis and grade in a veterans population. J Natl Cancer Inst. 2011;103(11):885–892. doi: 10.1093/jnci/djr108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Dillard PR, Lin MF, Khan SA. Androgen-independent prostate cancer cells acquire the complete steroidogenic potential of synthesizing testosterone from cholesterol. Mol Cell Endocrinol. 2008;295(1–2):115–120. doi: 10.1016/j.mce.2008.08.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Locke JA, Guns ES, Lubik AA, Adomat HH, Hendy SC, Wood CA, et al. Androgen levels increase by intratumoral de novo steroidogenesis during progression of castration-resistant prostate cancer. Cancer Res. 2008;68(15):6407–6415. doi: 10.1158/0008-5472.CAN-07-5997. [DOI] [PubMed] [Google Scholar]
- 10.Montgomery RB, Mostaghel EA, Vessella R, Hess DL, Kalhorn TF, Higano CS, et al. Maintenance of intratumoral androgens in metastatic prostate cancer: a mechanism for castration-resistant tumor growth. Cancer Res. 2008;68(11):4447–4454. doi: 10.1158/0008-5472.CAN-08-0249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Adam RM, Mukhopadhyay NK, Kim J, Di Vizio D, Cinar B, Boucher K, et al. Cholesterol sensitivity of endogenous and myristoylated Akt. Cancer Res. 2007;67(13):6238–6246. doi: 10.1158/0008-5472.CAN-07-0288. [DOI] [PubMed] [Google Scholar]
- 12.Cinar B, Mukhopadhyay NK, Meng G, Freeman MR. Phosphoinositide 3-kinase-independent non-genomic signals transit from the androgen receptor to Akt1 in membrane raft microdomains. J Biol Chem. 2007;282(40):29584–29593. doi: 10.1074/jbc.M703310200. [DOI] [PubMed] [Google Scholar]
- 13.Oh HY, Lee EJ, Yoon S, Chung BH, Cho KS, Hong SJ. Cholesterol level of lipid raft microdomains regulates apoptotic cell death in prostate cancer cells through EGFR-mediated Akt and ERK signal transduction. Prostate. 2007;67(10):1061–1069. doi: 10.1002/pros.20593. [DOI] [PubMed] [Google Scholar]
- 14.Pommier AJ, Alves G, Viennois E, Bernard S, Communal Y, Sion B, et al. Liver X Receptor activation downregulates AKT survival signaling in lipid rafts and induces apoptosis of prostate cancer cells. Oncogene. 2010;29(18):2712–2723. doi: 10.1038/onc.2010.30. [DOI] [PubMed] [Google Scholar]
- 15.Zhuang L, Lin J, Lu ML, Solomon KR, Freeman MR. Cholesterol-rich lipid rafts mediate akt-regulated survival in prostate cancer cells. Cancer Res. 2002;62(8):2227–2231. [PubMed] [Google Scholar]
- 16.Serre D, Lee BH, Ting AH. MBD-isolated Genome Sequencing provides a high-throughput and comprehensive survey of DNA methylation in the human genome. Nucleic Acids Res. 2010;38(2):391–399. doi: 10.1093/nar/gkp992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Langmead B, Trapnell C, Pop M, Salzberg SL. Ultrafast and memory-efficient alignment of short DNA sequences to the human genome. Genome Biol. 2009;10(3):R25. doi: 10.1186/gb-2009-10-3-r25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Yan H, Choi AJ, Lee BH, Ting AH. Identification and functional analysis of epigenetically silenced microRNAs in colorectal cancer cells. PLoS ONE. 2011;6(6):e20628. doi: 10.1371/journal.pone.0020628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Robinet P, Wang Z, Hazen SL, Smith JD. A simple and sensitive enzymatic method for cholesterol quantification in macrophages and foam cells. J Lipid Res. 2010;51(11):3364–3369. doi: 10.1194/jlr.D007336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.McCarty KS, Jr, Szabo E, Flowers JL, Cox EB, Leight GS, Miller L, et al. Use of a monoclonal anti-estrogen receptor antibody in the immunohistochemical evaluation of human tumors. Cancer Res. 1986;46(8 Suppl):4244s–4248s. [PubMed] [Google Scholar]
- 21.Leon CG, Locke JA, Adomat HH, Etinger SL, Twiddy AL, Neumann RD, et al. Alterations in cholesterol regulation contribute to the production of intratumoral androgens during progression to castration-resistant prostate cancer in a mouse xenograft model. Prostate. 2010;70(4):390–400. doi: 10.1002/pros.21072. [DOI] [PubMed] [Google Scholar]
- 22.Trasino SE, Kim YS, Wang TT. Ligand, receptor, and cell type-dependent regulation of ABCA1 and ABCG1 mRNA in prostate cancer epithelial cells. Mol Cancer Ther. 2009;8(7):1934–1945. doi: 10.1158/1535-7163.MCT-09-0020. [DOI] [PubMed] [Google Scholar]
- 23.Potter VR. The biochemical approach to the cancer problem. Fed Proc. 1958;17(2):691–697. [PubMed] [Google Scholar]
- 24.Siperstein MD, Fagan VM. Deletion of the Cholesterol-Negative Feedback System in Liver Tumors. Cancer Res. 1964;24:1108–1115. [PubMed] [Google Scholar]
- 25.Addleman W. Cancer, cholesterol and cholestyramine. N Engl J Med. 1972;287(20):1047. [PubMed] [Google Scholar]
- 26.Browning DR, Martin RM. Statins and risk of cancer: a systematic review and metaanalysis. Int J Cancer. 2007;120(4):833–843. doi: 10.1002/ijc.22366. [DOI] [PubMed] [Google Scholar]
- 27.Dale KM, Coleman CI, Henyan NN, Kluger J, White CM. Statins and cancer risk: a meta-analysis. Jama. 2006;295(1):74–80. doi: 10.1001/jama.295.1.74. [DOI] [PubMed] [Google Scholar]
- 28.Kuoppala J, Lamminpaa A, Pukkala E. Statins and cancer: A systematic review and meta-analysis. Eur J Cancer. 2008;44(15):2122–2132. doi: 10.1016/j.ejca.2008.06.025. [DOI] [PubMed] [Google Scholar]
- 29.Murtola TJ, Tammela TL, Lahtela J, Auvinen A. Cholesterol-lowering drugs and prostate cancer risk: a population-based case-control study. Cancer Epidemiol Biomarkers Prev. 2007;16(11):2226–2232. doi: 10.1158/1055-9965.EPI-07-0599. [DOI] [PubMed] [Google Scholar]
- 30.Jacobs EJ, Rodriguez C, Bain EB, Wang Y, Thun MJ, Calle EE. Cholesterol-lowering drugs and advanced prostate cancer incidence in a large U. S. cohort. Cancer Epidemiol Biomarkers Prev. 2007;16(11):2213–2217. doi: 10.1158/1055-9965.EPI-07-0448. [DOI] [PubMed] [Google Scholar]
- 31.Shannon J, Tewoderos S, Garzotto M, Beer TM, Derenick R, Palma A, et al. Statins and prostate cancer risk: a case-control study. Am J Epidemiol. 2005;162(4):318–325. doi: 10.1093/aje/kwi203. [DOI] [PubMed] [Google Scholar]
- 32.Mondul AM, Han M, Humphreys EB, Meinhold CL, Walsh PC, Platz EA. Association of statin use with pathological tumor characteristics and prostate cancer recurrence after surgery. J Urol. 2011;185(4):1268–1273. doi: 10.1016/j.juro.2010.11.089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Gutt R, Tonlaar N, Kunnavakkam R, Karrison T, Weichselbaum RR, Liauw SL. Statin use and risk of prostate cancer recurrence in men treated with radiation therapy. J Clin Oncol. 2010;28(16):2653–2659. doi: 10.1200/JCO.2009.27.3003. [DOI] [PubMed] [Google Scholar]
- 34.Kollmeier MA, Katz MS, Mak K, Yamada Y, Feder DJ, Zhang Z, et al. Improved biochemical outcomes with statin use in patients with high-risk localized prostate cancer treated with radiotherapy. Int J Radiat Oncol Biol Phys. 2011;79(3):713–718. doi: 10.1016/j.ijrobp.2009.12.006. [DOI] [PubMed] [Google Scholar]
- 35.Schmitz G, Langmann T. Transcriptional regulatory networks in lipid metabolism control ABCA1 expression. Biochim Biophys Acta. 2005;1735(1):1–19. doi: 10.1016/j.bbalip.2005.04.004. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.