Abstract
Background
Clostridium difficile infection (CDI) is a major health problem. Epidemiological evidence suggests that there is an association between acid suppression therapy and development of CDI.
Purpose
We sought to systematically review the literature that examined the association between histamine 2 receptor antagonists (H2RAs) and CDI.
Data source
We searched Medline, Current Contents, Embase, ISI Web of Science and Elsevier Scopus from 1990 to 2012 for all analytical studies that examined the association between H2RAs and CDI.
Study selection
Two authors independently reviewed the studies for eligibility.
Data extraction
Data about studies characteristics, adjusted effect estimates and quality were extracted.
Data synthesis
Thirty-five observations from 33 eligible studies that included 201834 participants were analyzed. Studies were performed in 6 countries and nine of them were multicenter. Most studies did not specify the type or duration of H2RAs therapy. The pooled effect estimate was 1.44, 95% CI (1.22–1.7), I2 = 70.5%. This association was consistent across different subgroups (by study design and country) and there was no evidence of publication bias. The pooled effect estimate for high quality studies was 1.39 (1.15–1.68), I2 = 72.3%. Meta-regression analysis of 10 study-level variables did not identify sources of heterogeneity. In a speculative analysis, the number needed to harm (NNH) with H2RAs at 14 days after hospital admission in patients receiving antibiotics or not was 58, 95% CI (37, 115) and 425, 95% CI (267, 848), respectively. For the general population, the NNH at 1 year was 4549, 95% CI (2860, 9097).
Conclusion
In this rigorous systematic review and meta-analysis, we observed an association between H2RAs and CDI. The absolute risk of CDI associated with H2RAs is highest in hospitalized patients receiving antibiotics.
Introduction
Clostridium difficile infection (CDI) is considered a major health problem with a point prevalence of 13.1/1000 in-patient [1] and is increasing in incidence and mortality [2]–[5]. The CDI cost in the United States of America (USA) alone was conservatively estimated to exceed $1.1 billion annually [6]. Risk factors associated with CDI acquisition are numerous and traditionally have included exposure to antibiotics, advanced age, comorbidities, enteral feeding, prolonged hospitalization, endoscopy and antineoplastic medications [7]–[10].
The role of gastric acid suppression therapy has gained interest recently as a risk factor for CDI. Four recently published meta-analyses have suggested an association between gastric acid suppression therapy with proton pump inhibitors (PPI) and CDI [11]–[14]. The United States Food and Drug Administration (FDA) recently warned the public about a possible association between CDI and PPI use [15]. However, to date; there is no systematic review dedicated to evaluate the potential association between histamine 2 receptors antagonists (H2RAs) use and risk of CDI.
H2RAs are popular over-the-counter (OTC) drugs worldwide [16]. Off -label use of H2RAs and substitution for physician care were reported in 46 % and 34% of the adult consumer, respectively [15]. Masking serious conditions, missed diagnosis, and the potential for inappropriate use by patients are concerns about OTC use of H2RAs [17]. Nonetheless, the implications of OTC H2RAs use are not yet well defined.
Given the high prevalence of prescription use and OTC use of H2RAs and the increasing incidence and severity of CDI, we sought to systematically review the published literature that examined the association between H2RAs use and development of CDI following the MOOSE [18] and PRISMA [19] guidelines. We use the Grades of Recommendation, Assessment, Development and Evaluation (GRADE) framework [20] to interpret our findings.
Methods
Search strategy
The search strategy and subsequent literature searches were performed by a medical reference librarian (PJE) with 37 years of experience. The initial strategy was developed in Ovid MEDLINE (1990 through January 2012), using MeSH (Medical Subject Headings) controlled vocabulary, and then modified for Ovid EMBASE (1990 through January 2012). Primary terms were: enterocolitis, pseudomembranous/ AND the therapeutic agents of interest: explode omeprazole, explode proton pump inhibitors, anti-ulcer agents, and explode histamine H2 antagonists (Explode allows including all of the specific drugs, without having to use all of the various terms, synonyms, brands and generic names.) Articles were limited to randomized controlled trials, cohort studies, and or case-control studies. The same process was used with Ovid EMBASE with alterations as necessary to accommodate EMBASE's more granular subject headings. ISI Web of Science and Elsevier Scopus use text words: (difficile OR pseudomembranous OR pseudo-membranous) AND (omeprazole OR “proton pump” OR ranitidine OR h2 OR h-2 OR “acid suppression” OR antacid*)) AND (random* OR trial* OR blind* OR cohort* OR controlled OR prospective). Moreover, bibliographic references of all articles and previous meta-analyses were searched for eligible studies. We have designed the search strategy to capture any association between gastric acid suppression therapy and development of CDI.
There was no restriction to language. All results were downloaded into EndNote 7.0 (Thompson ISI Research soft, Philadelphia, Pennsylvania), a bibliographic database manager, and duplicate citations were identified and removed. Two authors (A.B.A. and F.A.) independently assessed the eligibility of identified studies.
Study selection
To be included, a study had to: (1) be an analytical study; and (2) examine the association between H2RAs use and incidence of CDI in adult population.
Data collection
A data collection form was developed and used to retrieve information on relevant features and results of pertinent studies. Two reviewers (A.B.A. and F.A.) independently extracted and recorded data in a predefined checklist. Disagreements among reviewers were discussed with two other reviewers (I.M.T. and M.A.A.), and agreement was reached by consensus. We collected adjusted effect estimates and 95% confidence intervals (CI) based on the multivariable regression model used in each study.
We used the Newcastle-Ottawa Quality Assessment Scale for cohort and case-control studies [21] which is intended to rate selection bias, comparability of the exposed and unexposed groups of each cohort, outcome assessment, and attrition bias. Two reviewers (M.A.G and F.A.) independently assessed the methodological quality of selected. Disagreement among reviewers was discussed with 2 other reviewers (I.M.T. and M.A.A.), and agreement was reached by consensus.
We used the GRADE framework to interpret our findings. The Cochrane Collaboration has adopted the principles of the GRADE system [20] for evaluating the quality of evidence for outcomes reported in systematic reviews.
For purposes of systematic reviews, the GRADE approach defines the quality of a body of evidence as the extent to which one can be confident that an estimate of effect or association is close to the quantity of specific interest. Quality of a body of evidence involves consideration of within-study risk of bias (methodological quality), directness of evidence, heterogeneity, precision of effect estimates and risk of publication bias.
Statistical Analyses
Meta-analyses
The primary effect measures used in the meta-analysis were Odds Ratios (OR), Hazard Ratios (HR) and Relative Risks (RR) which were assumed to reasonably estimate the same association between CDI and H2RAs given the low incidence of CDI and thus were pooled together. Adjusted effect estimates were primarily used for this analysis. Unadjusted effect estimates were used as alternatives if studies did not pursue adjustment because of absence of association on univariate comparison.
Effect estimates from all included studies were pooled in a meta-analysis weighing individual studies according to their log-transformed inverse variance. The DerSimonian and Laird random effects model [22] was used to calculate the pooled effect estimates.
We extracted data on the proportion of CDI cases that were exposed to antibiotics from all studies that reported these data. We then performed a meta-analysis for the proportion on logit scale using random effects model weighing the individual studies according to their log-transformed inverse variance.
Exploring heterogeneity
Homogeneity among studies was tested by means of Cochran's Q test and calculation of the variation across studies attributable to heterogeneity rather than chance (I2). The influence of a range of a-priori selected study-level and aggregated individual-level parameters on the observed effect estimate was investigated by means of meta-regressions. In these analyses, the log odds ratio from each study was regressed on the potential confounders in univariate and multivariate weighted linear regressions, weighted according to the inverse standard error and the residual between-study variance. Ten potential confounders were considered. Seven variables were categorical: design of the study (case-control vs. cohort), country of publication, setting (single center vs. multicenter), method of ascertainment of antibiotic use, method of effect measure (OR vs. RR/HR), effect estimate (adjusted vs. unadjusted) and quality of included studies (high score vs. low score). Three continuous variables were: the impact factor of the journal where the study was published, number of variables the effect measure was adjusted for and proportion of cases that were exposed to antibiotics.
Publication bias
The possible influence of publication bias was graphically assessed with the novel method of contour-enhanced funnel plot where log-transformed odds ratios were plotted against standard errors. This method examines whether any funnel plot asymmetry is likely to be due to publication bias compared with other underlying causes of funnel plot asymmetry. The contours help to indicate whether areas of the plot, where studies are perceived to be missing, are where studies would have statistically significant effect sizes or not and thus decrease or increase the evidence that the asymmetry is due to publication bias. The presence of funnel plot asymmetry was also assessed using Egger's test [23].
Residual confounding
Finally, the possible influence of unknown confounders (residual confounding) was investigated with a rule-out approach described by Schneeweiss [24]. This approach stipulates the influence of a hypothetical confounder and determines what characteristics this confounder must have to fully account for the observed association between use of H2RAs and occurrence of CDI. The hypothetical confounder is characterized by its association to H2RAs use (OREC, odds ratio of exposure to the confounder) and its association to the outcome (RRCO, relative risk of outcome in individuals exposed to the confounder vs. non-exposed). For this analysis, the absolute risk in the pooled non-exposed group was used for conversion of odds ratio to relative risk using the method described by Zhang and Yu [25]. Separate analyses were performed to demonstrate what levels of OREC and RRCO would be required to fully explain the observed association between H2RAs and CDI for different hypothetical prevalence of the unknown confounder (i.e. PC = 0.2, PC = 0.4) before and after adjustment for publication bias as described above.
In all analyses, results associated with p-values <0.05 (two-sided test) were considered statistically significant. All statistical analyses were performed using Stata version 12 statistical software (StataCorp, College Station, Texas).
Results
Search results
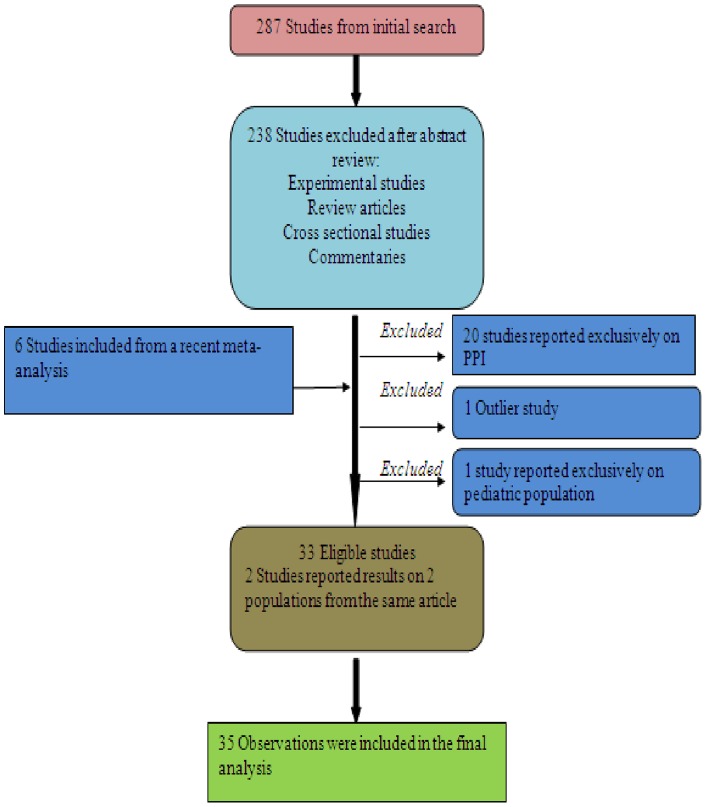
The search yielded 27 eligible studies after excluding 260 citations. Six more studies were retrieved from recent review articles and added to the total eligible studies. Kutty [26] et al and Jayatilaka [27] et al, each reported 2 different observations for different participants. Thus, a total of 33 articles met our inclusion criteria representing 35 observations that included 201834 participants. There was excellent agreement for the inclusion of the studies, data abstraction and quality assessment between the reviewers (kappa statistic being 1.0, 1.0 and 0.91 respectively).
The study selection process is illustrated in Figure 1 and the main characteristics of the included studies are summarized in Table 1. Twenty-four case control studies [26]–[37], [45], [49]–[51], [53]–[58] and 11 cohort studies [38]–[44], [46]–[48], [52] reported data on both community-acquired and hospital-acquired CDI (8 observations were from community-acquired CDI, 23 from hospital-acquired CDI and 4 representing both type of CDI). Six studies [26], [39], [47], [51], [53], [54] were from multiple centers; two from UK general practice research database [28], [30], and the remaining were from single centers. The included studies were performed in 6 countries (17 studies from USA, 9 from Canada, 6 from United Kingdom, 1 from Netherlands, 1 from Israel, and one from Korea). Most studies did not specify the type or duration of therapy with H2RAs. Tables 2 and 3 summarized the case ascertainment, control or non-exposed group selection method for case control and cohort studies, respectively. Among all citations, seventeen studies reported the proportion of cases exposed to antibiotics. Eight studies used antibiotics exposure as inclusion criteria. Three studies did not provide either the absolute number of exposed or unexposed groups thus were not included in this pooled proportion analysis.
Figure 1. Flow diagram of eligible studies.
Table 1. Characteristics of the included studies.
| Source | Country | Centers | Setting | Condition | Study Design | Inclusion Criteria | Acid Suppression Therapy |
| Kutty et al (VA),262010 | US | Multicenter | Community | Gen Pop | Case-control | Age: ≥18 yr; Communityonset CDAD | H2RAs: exposure 3mo prior to test |
| Kutty et al (D),26 2010 | US | Multicenter | Community | Gen Pop | Case-control | Age: ≥18 yr; Communityonset CDAD | H2RAs: exposure 3mo prior to test |
| Nath et al, 281994 | CA | Single | Hospital | Hem-onco pts | Case-control | Adult; In-patient >3d | Acid suppression therapy |
| Jayatilaka et al, 27 2007 | US | Single | Hospital | Gen In-patient | Case-control | Age >18 | H2RAs: pre admission |
| Jayatilaka et al,272007 | US | Single | Hospital | Gen In-patient | Case-control | Age >18 | H2RAs: post admission |
| Shah et al (D),29 2000 | UK | Single | Hospital | Gen In-patient | Case-control | Age >65 yr; Gen medical/elderlycare wards | H2RAs: upto 16 wk before diarrhea |
| Dial et al, 30 2005 | UK | GPRD | Community | Gen Pop | Case-control | Age ≥18 yr; At least 2 yrs of recordsin the GPR; firstoccurrence of CDAD | H2RAs: 90 d prior to the index date |
| Debast et al, 31 2009 | NL | Single | Hospital | Gen In-patient | Case control | Age:≥18 yr; CDAD | H2RAs: exposure |
| Lowe et al, 32 2006 | CA | Single | Community | Gen Pop | Case-control (R) | 1 hospital admission for CDAD; Age ≥ 66yr; CDAD diagnosis within 60d of ABX therapy | H2RAs: exposure |
| Dial et al, 33 2006 | UK | GPRD | Community | General pop | Case-control | First prescription oral Vancomycin; No previous admission 1yr before index date | H2RAs: 90d prior to index date |
| Aseeri et al, 34 2008 | US | Single | Hospital | Gen In-patient | Case-control | Age ≥18 Yr; Inpt for ≥3 d | H2RAs: 3d before CDAD |
| Dubberke et al,35 2007 | US | Single | Hospital | Gen In-patient | Case- Control | Pts admitted for >48 hrbetween study period | H2RAs |
| Loo et al, 36 2005 | UK | Single | Hospital | Gen In-patient | Case-control | Hospital Acquired CDAD; | H2RAs: 6wk before diagnosis |
| Sundram et al,37 2009 | UK | Single | Hospital | Gen In-patient | Case-control | Adult HospitalAcquired CDAD | H2RAs: 6wk prior to onset |
| Howell et al,38 2010 | US | Single | Hospital | Gen In-patient | Cohort | Age ≥18 yr; LOS ≥3 d;Only first diagnosis | H2RAs |
| Dalton et al, 39 2009 | CA | Multicenter | Hospital | Med/Surgical Subspecialty | Cohort, (R) | Age: ≥18 yr; Minimum 7-dLOS; ABX exposure | H2RAs |
| Dubberk et al,402007 | US | Single | Hospital | Gen In-patient | Cohort, (R) | All pts admitted to BJHfor more than 48 hours | H2RAs |
| Pepin et al,41 2005 | CA | Single | Hospital | Gen In-patient | Cohort, (R) | Adult In-patient | H2RAs |
| Beaulieu et al, 42 2007 | CA | Single | Hospital | Medical ICU | Cohort | ICU LOS>24hr; Diarrhea >24 hr and positive CD toxin (2d to 2monthspost discharge) | H2RAs |
| Peled et al, 43 2007 | IL | Single | Hospital | Gen In-patient | Cohort, (P) | CD testing during 4m period; ABX within 40d prior to diarrhea | H2RAs |
| Dial et al, 44 2004 | CA | Single | Hospital | Med/CT/Surgical wards | Cohort | Pharmacy database; ABX during studyperiod; positive toxin in theinfection control registry | H2RAs |
| Novell et al, 45, 2010 | US | Single | Hospital | Gen Inpatients | Case-control, (R) | Age ≥18 yr; CDAD | H2RAs |
| Netland et al, 46 2011 | US | Single | Both | Gen Pop | Cohort, (R) | Recurrent CDI | H2RAs |
| Jung et al, 47 2010 | Korea | Single | Hospital | Gen Inpatients | Cohort study, (R) | Recurrent CDAD ortreatment failure cases | H2RAs |
| Loo et al, 48 2011 | CA | Multicenter | Hospital | Gen Inpatients | Cohort study(P) | Age ≥18, Health CareAssociated CDAD | H2RAs |
| Manges et al, 49 2010 | CA | Single | Hospital | Gen Inpatients | Case control | Nosocomial CDAD | H2RAs |
| Kuntz et a,l50 2011 | US | Single | Community | Gen Pop | Case control, (R) | CommunityAssociated CDAD | Acid suppression therapy |
| Naggie et al,51 2011 | US | Multicenter | Community | Gen Pop | Case control | Age≥18 yr | Acid suppression therapy |
| Stevens et al,52, 2011 | US | Single | Hospital | Gen Inpatients | Cohort, (R) | Age ≥18 yr,Hospital acquired | H2RAs |
| Dial et al, 53 2008 | CA | Multicenter | Community | Elderly patients | Case control | Age ≥65, CommunityAssociated CDAD | H2RAs |
| McFarland et al,54 2007 | US | Multicenter | Both | Gen Pop | Case control | CDAD Diagnosis | H2RAs |
| Kazakova et al,55 2012 | US | Single | Both | Gen Pop | Case control | CDAD Diagnosis, onset duringthe pre-outbreak or outbreakperiods, hospitalization | H2RAs |
| Modena et al,56 2005 | US | Single | Both | Gen Pop | Case control | Received at least 5 daysof antibiotics prior to diagnosis of CDAD | H2RAs |
| Muto et al,57 2005 | US | Single | Hospital | Gen Inpatients | Case control | Nosocomial CDAD | H2RAs: During the 4 weeks before detection of CDAD |
| Yip et al, 58 2001 | CA | Single | Hospital | Gen Inpatients | Case control | Nosocomial CDAD | H2RAs |
Abbreviations: US, United States;UK, United Kingdom; BMT, Bone Marrow Transplant; ESRD, End Stage Renal Disease; GPRD – general practice research database; IBD, Inflammatory Bowel Disease; CD, Clostridium Difficile; CDAD, Clostridium difficile associated diarrhea; LOS, Length of Stay; LTCF, Long Term Care Facility; Gen, General.; Pop, Population; d, day/days; mo, month/months; yr, year/year; wk, week/week; Pts, Patients; Pt, Patient; Med, Medical; CT, Cardio-thoracic; NL, Netherland; CA, Canada; IL, Israel; Abd, Abdominal; (P), prospective; (R), Retrospective.*, Mostly hospital.
Table 2. The Association between H2RAs use and development of Clostridium difficile infection from case-control studies.
| Source | Case Ascertainment | Selection of Controls | Sample size | Adjusted Effect Estimates |
| Kutty et al,26 (VA) | Non-formed stool,Positive CD toxin | Randomly selectedfrom the same geographicaloutpatients territory | Exposed group; cases:7, controls: 13 | Crude OR, 1.8 (0.6–4.8) |
| Non-exposed group;cases: 29, controls: 95 | ||||
| Kutty et al,26 (D) | Non-formed stool,Positive CD toxin | Randomly selected fromthe same geographical outpatients territory | Exposed group; cases:6, controls: 3 | Crude OR, 1.3 (0.3–5.6) |
| Non-exposed group;cases: 67, controls: 45 | ||||
| Sundram et al,37 2009 | Diarrhea, Positive stoolfor CD toxin, ribotyped | Inpatients, No diarrhea, Nevertested positive for CD | Exposed group; cases:65, controls: 52 | Crude OR for PPI/ H2RAs :1.7, P 0.456 |
| Non-exposed group;cases: 32, controls: 45 | ||||
| Jayatilaka et al,27 2007 | Diarrhea, Positive toxin | Age and sex matched,Same period of time | H2RAs use pre andduring admission | H2RAs use pre andduring admission |
| Exposed group; cases:9, controls: 17 | OR: 0.95(0.39-2.34) | |||
| Non-exposed group;cases: 6, controls: 14 | ||||
| Jayatilaka et al,27 2007 | Diarrhea, Positivetoxin | Age and sex matched,Same period of time | H2RAsuse post admission | H2RAsuse post admission |
| Exposed group; cases:133, controls: 227 | OR: 0.73(0.26-2.06) | |||
| Non-exposed group;cases: 116, controls: 230 | ||||
| Loo et al,36 2005 | Diarrhea/positive CD, Endoscopicdiagnosis, histological evidence | Matched to Age,Charlson index, date ofadmission, ward, LOS | Exposed group; cases:47, controls: 47 | Diarrhea/positive CD, Endoscopicdiagnosis, histological evidence |
| Non-exposed group;cases: 190, controls: 190 | ||||
| Shah et al,29 2000 | Diarrhea Positivestool for CD toxin | Negative stool toxins,Similar age, Hospitalward, Same time | Exposed group; cases:22, controls: 22 | Diarrhea |
| Non-exposed group;cases: 104, controls: 104 | Positive stoolfor CD toxin | |||
| Dial et al,33 2006 | Patients with first prescriptionof oral Vancomycin | Age matched,Same ward | Exposed group; cases:23, controls: 112 | Patients with first prescriptionof oral Vancomycin |
| Non-exposed group;cases: 294, controls: 2055 | ||||
| Asseri et al,34 2008 | Diarrhea | Matched to date ofadmission, antibiotic use, gender, age group, patient location, room type | Exposed group; cases:17, controls: 9 | Diarrhea |
| Positive stoolfor CD toxin | Non-exposed group;cases: 77, controls: 85 | Positive stoolfor CD toxin | ||
| Dial et al,30 2005 | Positive CD toxin | Same general practice,Not hospitalized in the yearprior to index date, Negative CD toxin, No diagnosis of CDI | Exposed group;cases: 83, controls: 367 | Positive CD toxin |
| Clinical diagnosismade by GP | Non-exposed gp;cases: 1150, controls: 11963 | Clinical diagnosismade by GP | ||
| Lowe et al,32 2006 | CDAD | Matched to age, sex,and antibiotic use | Exposed group; cases: 213, controls: 1846Non-exposed gp; cases: 1176, controls: 10457 | Exposed group; cases: 213, controls: 1846Non-exposed gp; cases: 1176, controls: 10457 |
| Debast et al,31 2009 | Diarrhea | Randomly selected frothe same time and same wards asCDI cases | Exposed group; cases:2, controls: 2 | Exposed group; cases:2, controls: 2 |
| Positive stoolfor CD toxin | Non-exposed group;cases: 43, controls: 88 | Non-exposed group;cases: 43, controls: 88 | ||
| Nath et al,28 1994 | Diarrhea | Age matched,Same hospital unit | Exposed group; cases:51, controls: 32 | Exposed group; cases:51, controls: 32 |
| Positive stoolfor CD toxin | Non-exposed group;cases:29, controls: 48 | Non-exposed group;cases:29, controls: 48 | ||
| Dubberke et al,35 2007 | Diarrhea | Randomly selectedDuring the study period | Exposed group; cases:206, controls: 426 | Exposed group; cases:206, controls: 426 |
| Positive stoolfor CD toxin | Non-exposed group;cases: 176, controls: 1102 | Non-exposed group;cases: 176, controls: 1102 | ||
| Novell et al,45 2010 | New diarrhea | Matched to in-patient unit,age, gender, date of admission | Exposed group; cases:12, controls: 07 | Exposed group; cases:12, controls: 07 |
| Positive stoolfor CD toxin | Non-exposed group;cases: 162, controls: 167 | Non-exposed group;cases: 162, controls: 167 | ||
| Manges et al,49 2010 | Diarrhea/positive CD, Endoscopicdiagnosis, histological evidence | Matched to Age, gender,date of hospitalization | Exposed group; cases:09, controls: 12 | Exposed group; cases:09, controls: 12 |
| Non-exposed group;cases: 16, controls: 38 | Non-exposed group;cases: 16, controls: 38 | |||
| Kuntz et al,50 2011 | ICD-9 code, CDAD | Randomly selected | Exposed group; cases:55, controls: 157 | Exposed group; cases:55, controls: 157 |
| Non-exposed group;cases: 249, controls: 2883 | Non-exposed group;cases: 249, controls: 2883 | |||
| Naggie et al,51 2011 | Diarrhea | Matched by geographiclocation | Exposed group; cases:22, controls: 44 | Exposed group; cases:22, controls: 44 |
| Positive stoolfor CD toxin | Non-exposed group;cases: 44, controls: 70 | Non-exposed group;cases: 44, controls: 70 | ||
| Dial et al,53 2008 | ICD-9 code008.45, CDAD | Randomly selected, matchedto index date and date of first hospital admission | NR | RR:1.60 (0.90-2.20) |
| McFarland et al,54 2007 | Acute diarrhea Culture positive or positive C.D toxins | Matched to time ofCDAD, Age, Ward | Exposed group; cases:24, controls: 160 | NR |
| No other causefor the diarrhea | Non-exposed group;cases: 23, controls: 161 | |||
| Kazakova et al,55 2012 | Diarrhea, positiveCD toxin A | Matched to Sex, Age,admission date | Exposed group;cases:19, controls: 49 | OR:2.69(1.22-5.97) |
| Non-exposed group;cases: 18, controls: 109 | ||||
| Modena et al,56 2005 | Diarrhea | Inpatients, Receivedantibiotics for at least 5 days | Exposed group;cases:32, controls:18 | NR |
| Positive stoolfor CD toxins | Non-exposed group;cases: 98, controls: 102 | |||
| Muto et al,57 2005 | Diarrhea | Matched to admission date,Type of medical service, Length of hospital stay | Exposed group;cases:159, controls:44 | OR:2.00(1.10-3.50) |
| Positive stoolfor CD toxin | Non-exposed group; cases: 141, controls: 62 | |||
| Yip et al,58 2001 | Diarrhea | Matched to Age,Gender, admission date | Exposed group;cases:14, controls:13 | OR:2.70(0.71–10.10) |
| Positive stoolfor CD toxin | Non-exposed group;cases: 9, controls: 18 |
Table 3. The Association between H2RAs use and development of Clostridium difficile infection from cohort studies.
| Source | Case Ascertainment | Selection of Controls | Sample size | Adjusted Effect Estimates |
| Howell et al,38 2010 | Positive CD toxin | A nearest-neighbor–matching algorithmwas applied | Exposed group; cases: 66,controls: 10619 | OR : 1.53 (1.12–2.10) |
| Non-exposed group; cases:599,controls: 90512 | ||||
| Dalton et al,39 2009 | Positive stool toxins or colonoscopy-confirmed psudomembraneous colitis | Age, ≥ 18 years, Minimum7d LOS, Antibiotic exposure | Exposed group; cases:28 controls: 2135 | OR, 1.70 (1.09 2.64) |
| Non-exposed group;cases:121, controls: 12435 | ||||
| Dubberk et al,40 2007 | Positive stool for CD | In-patient, No positivestool toxin assay during the period(60d before startof study to the end) | Exposed group; cases: 206,controls: 998 Non-exposed group;cases: 176, controls: 25716 | OR, 2.0 (1.6-2.6) |
| Pepin et al, 41 2005 | Diarrhea, Positive toxin, provenpseudomembranous colitis | Unclear | Exposed group; cases:1199, controls: NR | HR, 1.07 (0.8-1.43) |
| Non-exposed gp; cases:6222, controls: NR | ||||
| Beaulieu et al,42 2007 | DiarrheaPositive stool for CD toxin | Unclear | Exposed group; cases:470, controls: NR | HR, 0.78 (0.5 – 1.23) |
| Non-exposed group;cases: 357, controls: NR | ||||
| Peled et al,43 2007 | DiarrheaPositive stool for CD toxin | Diarrhea with negativestool for CD, sameinstitution | Exposed group; cases:22, controls: 45 | OR, 3.1 P value : 0.024 |
| Non-exposed group;cases: 30, controls: 120 | ||||
| Dial et al,44 2004 | Positive stoolfor CD toxins | Unclear | Exposed group; cases:NR, controls: NR | OR : 1.1 (0.4-3.4) |
| Non-exposed group;cases: NR, controls: NR | ||||
| Netland et al, 46 2011 | Diarrhea between 5–60 daysafter antibiotic therapy for CDAD | Patients with CDAD inthe same institution | Exposed group; cases:05, controls: 50 | OR, 0.49 P value : 0.33 |
| Non-exposed group;cases: 50, controls: 99 | ||||
| Jung et al,47 2010 | Diarrhea or pseudomembranouscolitis, Positive toxin | Same institution | Exposed group; cases:06, controls: 31 | OR, 1.59 P value : 0.367 |
| Non-exposed group;cases: 08, controls: 66 | ||||
| Loo et al,48 2011 | Diarrhea and: positive CD, histological evidenceor pseudomembranous colitis | Frequency matchingapproach | Exposed group; cases:NR, controls: NR | OR : 0.55 (0.21 – 1.49) |
| Non-exposed group;cases: 190, controls: 190 | ||||
| Stevens et al,52 2011 | Diarrhea Positivestool for CD toxin | Same institution | Exposed group; cases:23, controls: 1060 | HR, 1.7 (0.7 – 3.9), P value 0.25 |
| Non-exposed group;cases: 218, controls: 8853 |
Quality assessment
Quality assessment of all included studies was done using the validated Newcastle-Ottawa Quality Assessment Scale [21] for cohort and case control studies (Tables 4 and 5). Included studies were scored based on the sum number of the stars given to each study. Among case-control studies, Loo et al 2011, Manges et al 2010, McFarland et al 2007, Modena et al 2005 and Dial et al 2008 scored the lowest. While Beaulieu et al 2005 scored the lowest among cohort studies. Most studies were of good quality with no evidence of selection bias, and with good comparability of the exposed and unexposed groups of each cohort, and outcome assessment.
Table 4. Modified Newcastle-Ottawa quality assessment scale for case-control studies included in the meta-analysis.
| Selection* | Exposure0 | ||||||||
| Included Studies | Adequacy of Case Definition | Representativeness of the Cases | Selection of Controls | Definition of Controls | Comparability• | Ascertainment of Exposure | Same Method of Ascertainment for Cases and Controls | Non- ResponseRate | Total No. of stars |
| Kutty et al,26 2010. | A* | A* | A* | A* | A* | A* | A* | C | 7 |
| Nath et al,281994 | A* | A* | B | A* | A** | A* | A* | C | 7 |
| Jayatilaka et al,27 2007 | B | A* | B | A* | A** | A* | A* | C | 6 |
| Shah et al,29 2000 | A* | A* | B | A* | A* | A* | A* | C | 6 |
| Lowe et al,32 2006 | A* | A* | A* | A* | A* | A* | A* | C | 7 |
| Dial et al,30 2005 | A* | A* | A* | A* | A* | A** | A* | C | 8 |
| Dial et al,33 2006 | B | A | A* | A | A* | A** | A* | C | 5 |
| Aseeri et al,34 2008 | A* | A* | B | A* | A** | E | B* | C | 6 |
| Dubberke et al,35 2007 | A* | B | B | A* | A** | A* | A* | C | 6 |
| Loo et al,36 2005 | A* | A* | B | A* | A* | E | A* | C | 5 |
| Sundram et al,37 2009 | A* | A* | B | A* | A* | A* | A* | C | 6 |
| Novell et al,45 2010 | A* | A* | B | A* | A** | A* | A* | C | 7 |
| Debast et al,31 2009 | A* | A* | B | A* | A* | A* | A* | C | 6 |
| Kuntz et al,50 2011 | A* | A* | A* | A* | A* | A* | A* | C | 7 |
| Manges et al,49 2010 | A* | A* | B | B | A* | A* | A* | C | 5 |
| Naggie et al,51 2011 | A* | A* | A* | A* | A* | C | A* | C | 6 |
| McFarland et al,54 2007 | B | A* | C | A* | A* | A* | A* | C | 6 |
| Modena et al,56 2005 | B | A* | B | A* | A** | A* | A* | C | 5 |
| Muto et al,57 2005 | B | A* | B | A* | A** | A* | A* | C | 6 |
| Yip, et al,58 2001 | B | A* | B | A* | A** | A* | A* | C | 6 |
| Dial et al,53 2008 | B | A* | B | A* | A* | A* | A* | C | 5 |
| Kazakova et al,55 2006 | A* | A* | B | A* | A** | D | A* | C | 6 |
Selection:
(1)Is this case definition adequate? A, yes, with independent validation; B, yes, eg record linkage or based on self reports C, no description.
(2) Representativeness of the cases: A, Consecutive or obviously representative series of cases; B, Potential for selection biases or not stated.
(3) Selection of controls: A, Community controls; B, Hospital controls; C, No description.
(4) Definition of controls: A, No history of disease; B, No description of source.
Comparability: Comparability of cases and controls on the basis of the design or analysis: A, study controls for co-morbidities; B, study controls for any additional factor (e.g., age and severity of illness).
Exposure:
Ascertainment of exposure: A, Secured records; B, Structured interview where blind to case/control status; C, Interview not blinded to case/control status; D, written self report or medical record only.
Same method of ascertainment for cases and controls; A, yes; B, no.
Non-response rate: A, Same for both groups; B, Non-respondents described; C, Rate different and no designation.
Table 5. Modified Newcastle-Ottawa Quality Assessment Scale for Cohort studies included in the Meta-analysis.
| Selection* | Outcome0 | ||||||||
| Included Studies | Representativeness of the exposed cohort | Selection of the Non-exposed Cohort | Ascertainment of Exposure | Incident Disease | Comparability• | Assessment of Outcome | Length of Follow-up | Adequacy of Follow-up | Total number of stars |
| Howell et al,2010 | A* | A* | A* | A* | A** | B* | A* | A* | 9 |
| Dalton et al, 2009 | A* | A* | A* | A* | A** | B* | A* | A* | 9 |
| Dubberke et al, 2007 | A* | A* | A* | A* | A** | B* | A* | A* | 9 |
| Pepin et al, 2005 | A* | A* | A* | A* | A* | B* | A* | A* | 8 |
| Beaulieu et al, 2007 | B | A* | A* | A* | A* | A* | A* | A* | 7 |
| Peled et al, 2007 | A* | A* | B | A* | A** | A* | A* | A* | 8 |
| Loo et al 2011 | A* | A* | B | A* | A** | A* | A* | A* | 8 |
| Netland et al, 2011 | A* | A* | A* | A* | A** | B* | A* | A* | 9 |
| Jung et al, 2010 | A* | A* | A* | A* | A** | B* | A* | A* | 9 |
| Stevens et al, 2011 | A* | A* | A* | A* | A** | B* | A* | A* | 9 |
| Dial et al 2004 | A* | A* | A* | A* | A* | A* | A* | A* | 8 |
Selection:
(1) Representativeness of the exposed cohort: A, truly representative; B, somewhat representative; C, selected group; D, no description of the derivation of the cohort.
(2) Selection of the non-exposed cohort: A, drawn from the same community as the exposed cohort; B, drawn from a different source; C, no description of the derivation of the non-exposed cohort.
(3) Ascertainment of exposure: A, secure record; B, structured interview; C, written self-report; D, no description.
(4) For demonstration that the outcome of interest was not present at start of study: A, yes; B, no.
Comparability: For comparability of cohorts on the basis of the design or analysis: A, study controls for co-morbidities; B, study controls for any additional factor (e.g., age and severity of illness); C, not done.
Outcome:
(1) Assessment of outcome: A, independent blind assessment; B, record linkage; C, self-report; D, no description.
(2) Was follow-up long enough for outcomes to occur? A, yes, (i.e. in-hospital or up to 30 d); B, no.
(3) Adequacy of follow-up of cohorts: A, complete follow-up and all subjects accounted for; B, subjects lost to follow-up was unlikely to introduce bias, because a small number were lost or a description was provided of those lost; C, follow-up rate 90% or lower (select an adequate percentage) and no description of those lost; D, no statement.
Meta-analysis
Thirty-five observations from 33 eligible studies were pooled using a random effect model meta-analysis. We excluded the study by Jenkins et al. as an outlier due to its large standard error. The pooled effect estimate was 1.44, 95% CI (1.22–1.7), I2 = 70.5%. The pooled effect estimate for high quality studies was 1.39 (1.15–1.68), I2 = 72.3%.
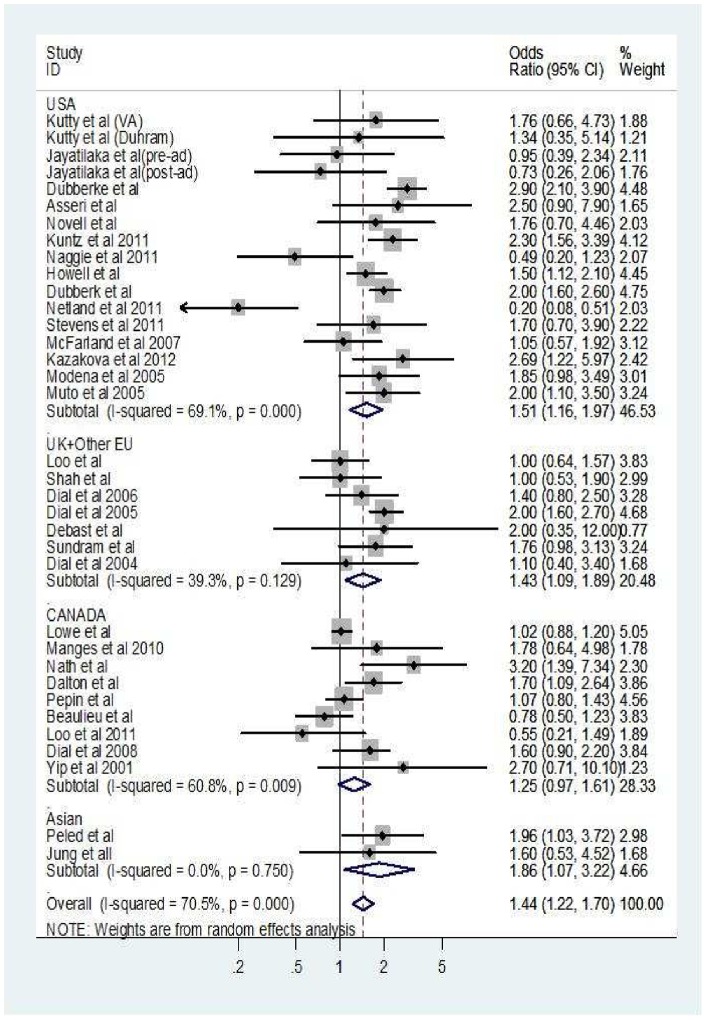
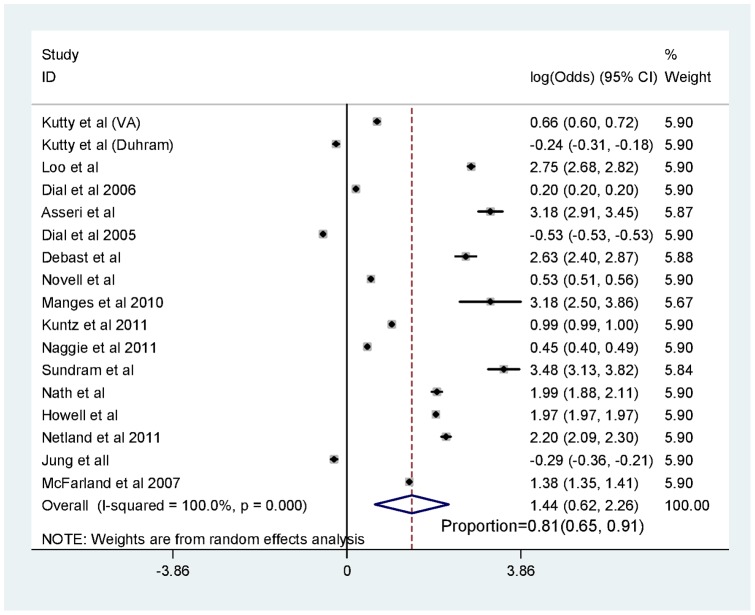
Although the heterogeneity between the analyzed studies was moderate, the majority of studies pointed towards a positive association. Figure 2 shows the forest plot and the pooled effect estimate for all studies stratified by country. Table 6 summarizes the pooled estimates and associated heterogeneity across different subgroups. The pooled proportion of CDI cases that were exposed to antibiotics was 0.81, 95% CI (0.65–0.91) as shown in Figure 3.
Figure 2. Forest plot-random effect model meta-analysis of the association between CDI and H2RAs based on 35 observations stratified by country.
Error bars indicate confidence interval.
Table 6. Influence of study type and country on the pooled effect estimate and its associated heterogeneity.
| Group | Pooled Effect Estimate (95 % CI) | I2 % | Number of Observations |
| All studies | 1.44 (1.22, 1.70) | 70.5 | 35 |
| Case-control studies | 1.58 (1.28, 1.95) | 68.9 | 24 |
| Cohort studies | 1.19 (0.87, 1.62) | 75.6 | 11 |
| Asia | 1.86 (1.07, 3.22) | 0 | 2 |
| Canada | 1.25 (0.97, 1.61) | 60.8 | 9 |
| Europe | 1.43 (1.09, 1.89) | 39.3 | 7 |
| USA | 1.51 (1.16, 1.95) | 65.1 | 17 |
Figure 3. Forest plot of the pooled proportion of Clostridium difficile cases that were exposed to antibiotics.
Exploring heterogeneity
The influence of a range of a-priori selected study-level and aggregated individual-level parameters on the observed effect estimate was investigated by means of meta-regressions. Table 7 summarizes the meta-regression analyses for all 35 results. Heterogeneity could not be explained by any of the 10 considered variables.
Table 7. Meta-regression analysis to explore sources of heterogeneity.
| Univariate Analyses | ||
| Study Characteristics | Coefficient | p-values |
| Study Design | −.27729 | 0.137 |
| Low score study | .194575 | 0.389 |
| Country where the study is conducted | ||
| United States | Reference | |
| Canada | −.1738854 | 0.431 |
| European countries | −.0849204 | 0.726 |
| Asian Countries | .1809134 | 0.686 |
| Setting | −.0286546 | 0.893 |
| No of variables adjusted for | .0251339 | 0.175 |
| Method of measuring effect estimate | −.2540725 | 0.325 |
| Impact factor of the journal | −.0067289 | 0.380 |
| Method of ascertainment of antibiotic | ||
| Patient chart | Reference | |
| Pharmacy record | −.0139199 | 0.955 |
| Interview | .3666586 | 0.517 |
| Questionnaire | .2703275 | 0.703 |
| Combined | .0368821 | 0.905 |
| Not reported | .2469137 | 0.381 |
| Proportion of antibiotic use | −.0023797 | 0.588 |
Publication bias
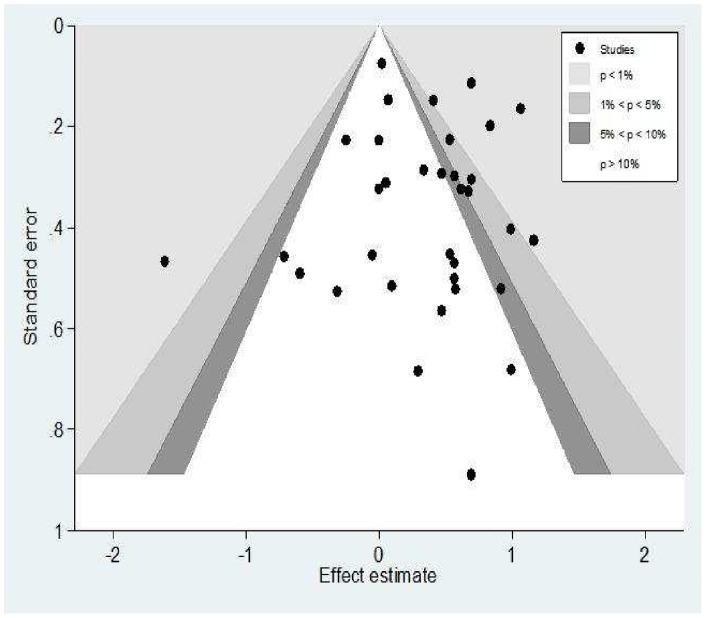
Figure 4 displays the contour enhanced funnel plot which showed no evidence of publication bias. This was confirmed by the Egger's test (P = 0.905).
Figure 4. Contour-enhanced funnel plot.
of the association between the estimated effect size and its standard error in all studies comparing those exposed and unexposed to H2RA displays areas of statistical significance on a funnel plot. Contours represent conventional “milestone” levels of statistical significance (e.g., <0.01, <0.05, <0.1). This funnel plot is symmetrical as it is not missing studies in the white area excluding the possibility of publication bias (Egger's test, p = 0.905).
Residual confounding
The results of the residual confounding analysis are presented in Figure 5. Panel A refers to a confounder with a prevalence of 0.20; at this prevalence level, a strong confounder causing a two-fold increased risk of CDI would have to be severely imbalanced between H2 blockers users and non users (OREC = 8.87) in order to fully account for the observed adjusted RR of 1.40. For a very common confounder with a prevalence of 0.40, stronger associations with acid-suppression use and/or CDI would be needed to explain the observed association between acid-suppression use and CDI. At this prevalence level, the confounder would have to be both imbalanced (OREC = 5.87) and increase the CDI risk (2.5-fold) to account for the observed OR, after taking publication bias into account.
Figure 5. Influence of a hypothetical dichotomous confounder present in 20% (panel A) and 40% (panel B) of the study population, unaccounted for in prior adjustments performed in individual studies.
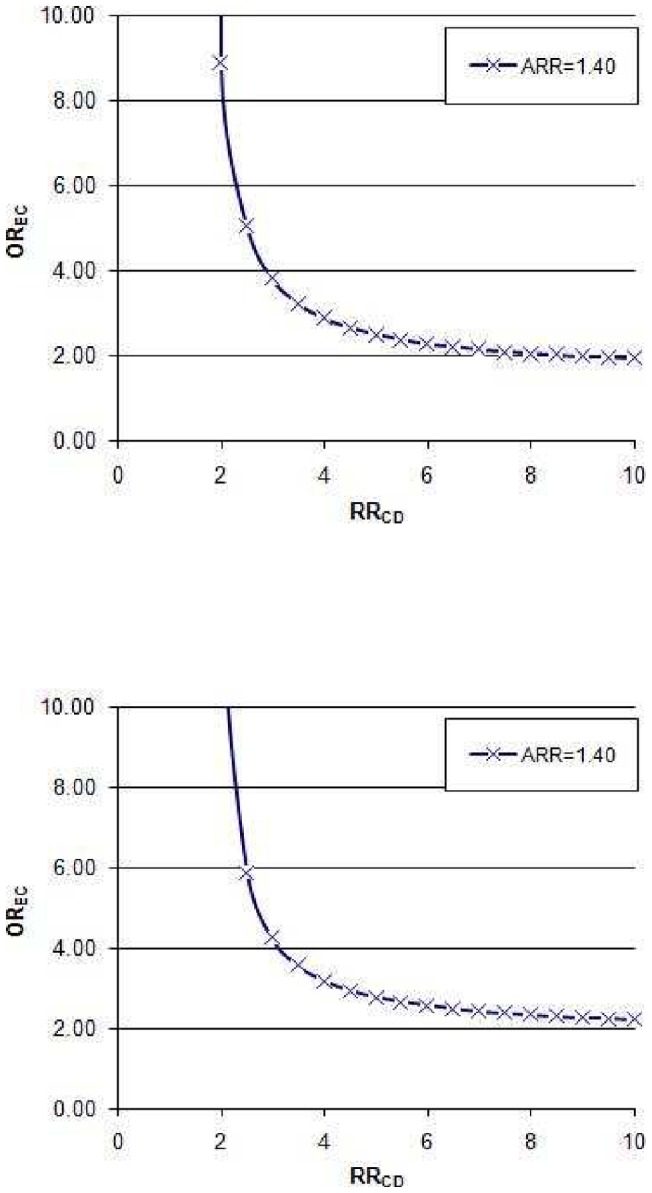
The graphs depict what combinations of OREC and RR would be necessary for the confounder to fully account for the observed association between H2RA use and CDI acquisition. Abbreviations: OREC, odds ratio of exposure to the confounder in H2RA non-users vs. H2RA users; RRCD, relative risk of CDAD in individuals exposed to the confounder vs. non-exposed.
Number needed to harm
The number needed to harm (NNH) was estimated by using the pooled OR from the meta-analysis [59]. A recent large prospective hospital cohort [48] reported the incidence of CDI at 14 days after hospital admission in patients receiving antibiotics or not: which was 42/1,000 and 5.4/1000, respectively. Based on these reported baseline risks, the number needed to harm (NNH) was 58, 95% CI (37, 115) and 425, 95% CI (267, 848), respectively. For the general population, the NNH at 1 year was 4549, 95% CI (2860, 9097) at 1 year, based on a baseline incidence of CDI of 48/100,000 person-years [60].
Discussion
Findings
In this rigorously conducted systematic review and meta-analysis, we observed an association between H2RAs use and development of CDI. Using the GRADE framework, the evidence supporting this association is considered of moderate quality. Although evidence from observational studies is considered of weak quality, we have ruled out a strong effect of an unmeasured confounder and, therefore, have upgraded its quality to moderate evidence in favor of this association.
The absolute risk of CDI was highest in hospitalized patients receiving antibiotics with an estimated NNH of 58 at 2 weeks. In contrast, the risk was very low (4549) in the general population. We also observed that, on average, 19% of CDI cases had not been recently exposed to antibiotics.
These findings add to previous subgroup analyses of a limited number of H2RA studies performed in a recent systematic review of the association between PPI and CDI. In this review, Kwok [11] et al conducted a subgroup analysis of 15 H2RA studies and reported a pooled effect estimate of 1.50, 95% CI (1.23–1.83). Similarly, Leonard et al [61] reported in 2007 an analysis based on 12 studies that showed H2RAs use was also associated with risk of CDI with a pooled OR 1.40, 95% CI (0.85–2.29).
Biologic plausibility
The pathogenic mechanisms operative in H2RAs therapy causing an increased risk of CDI acquisition are unclear, because gastric acid does not kill gastric C. difficile spores. One potential explanation for the association between CDI and gastric acid suppression therapies could be that the vegetative form of C. difficile, which is killed by acid, plays a role in pathogenesis. Vegetative forms survive on surfaces and could be ingested by patients [62]. Survival of acid-sensitive vegetative forms in the stomach could be facilitated by two primary factors: (1) suppression of gastric acid production by acid-suppressive medications; and (2) presence of bile salts in gastric contents of patients on acid-suppressive therapy. Bile salts, which are mainly found in the small intestine, are present in gastric contents, particularly among patients with gastro-esophageal reflux disease (GERD).
The extent of gastric acid suppression could play an important role in potentiating the risk of infection. Kwok [11] et at compared the risk of CDI with gastric acid suppression from 15 studies that reported on estimates of both PPI and H2RAs independently on their sample of participants and found that PPI is associated with higher risk of infection in comparison to H2RAs though both increase the risk.
Implications
Our findings have global implications both on the inappropriate use of acid-suppression therapy and on the increasing incidence of CDI.
Given the relatively low NNH (58 patients) needed to cause a case of CDI in hospitalized patients receiving antibiotics it becomes necessary to judiciously use H2RAs in these patients. In addition, reducing the inappropriate use of acid-suppression medications in this patient population could lead to a significant reduction in the incidence of CDI.
On the other hand, our findings are re-assuring to the public that H2RAs use in the general population as over-the-counter medications do not pose significant CDI risk and is associated with a high NNH.
Strengths
Our study has several important strengths. This review is the first systematic evaluation dedicated to examine the association between H2RAs and risk of CDI. It includes a comprehensive, up-to-date literature search and formal assessment of the methodological quality of pertinent studies with the largest number of relevant studies as compared to previous reviews 11,61. In addition, our pooled estimates are based on multivariate ORs of studies adjusting for several important CDI risk factors. We also performed subgroup analyses and sensitivity analyses that confirmed the robustness of our main results. There was no statistical evidence of publication bias and the effect of residual confounding on the observed association was examined. Finally, the NNH in different risk groups was calculated to aid physicians and patients in making a decision to use H2RA or not.
Limitations
Our review has certain limitations. There was moderate between-study heterogeneity; however, this is often the case in meta-analyses of large observational studies [63]–[65]. Moreover the majority of studies pointed towards a positive association. There was virtually no qualitative heterogeneity, and subgroup and sensitivity analyses showed results consistent with the main analysis. There are many patient level parameters which may have led to substantial heterogeneity. Nevertheless, investigating these variables is only possible with individual patient data meta-analysis.
Conclusions
In this rigorous systematic review and meta-analysis, we observed an association between H2RAs and CDI. The absolute risk of CDI associated with H2RAs was highest in hospitalized patients receiving antibiotics. On the other hand, our findings are re-assuring that H2RAs use in the general population as over-the-counter medications do not pose a significant CDI risk.
Supporting Information
PRISMA 2009 flow diagram.
(DOC)
PRISMA checklist.
(DOC)
Funding Statement
The authors have no support or funding to report.
References
- 1. Jarvis WR, Schlosser J, Jarvis AA, Chinn RY (2009) National point prevalence of Clostridium difficile in US health care facility inpatients, 2008. Am J Infect Control 37: 263–270. [DOI] [PubMed] [Google Scholar]
- 2. Archibald LK, Banerjee SN, Jarvis WR (2004) Secular trends in hospital-acquired Clostridium difficile disease in the United States, 1987–2001. J Infect Dis 189: 1585–1589. [DOI] [PubMed] [Google Scholar]
- 3. McDonald LC, Owings M, Jernigan DB (2006) Clostridium difficile infection in Patients discharged from US short-stay hospitals, 1996–2003. Emerg Infect Dis 12: 409–415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Elixhauser A, Jhung M. Clostridium difficile-associated disease in US hospitals, 1993–2005. Statistical Brief #50: Clostridium difficile-Associated Disease in U.S. Hospitals, 1993–2005.Available at: http://www.hcupus.ahrq.gov/reports/statbriefs/sb50.jsp. Last accessed Oct 21/2012.
- 5. Redelings MD, Sorvillo F, Mascola L (2007) Increase in Clostridium difficile-related mortality rates, United States, 1999–2004. Emerg Infect Dis 13: 1417–1419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Kyne L, Hamel MB, Polavaram R, Kelly CP (2002) Health care costs and mortality associated with nosocomial diarrhea due to Clostridium difficile . Clin Infect Dis 34: 346–353. [DOI] [PubMed] [Google Scholar]
- 7. Schuller I, Saha V, Lin L, Kingston J, Eden T, et al. (1995) Investigation and Management of Clostridium difficile colonisation in a paediatric oncology unit. Arch Dis Child 72: 219–222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Emoto M, Kawarabayashi T, Hachisuga MD, Eguchi F, Shirakawa K (1996) Clostridium difficile colitis associated with cisplatin-based chemotherapy in ovarian cancer patients.Gynecol Oncol. 61: 369–372. [DOI] [PubMed] [Google Scholar]
- 9. Bliss DZ, Johnson S, Savik K, Clabots CR, Willard K, et al. (1998) Acquisition of Clostridium difficile and Clostridium difficile-associated diarrhea in hospitalized Patients receiving tube feeding. Ann Intern Med 129: 1012–1019. [DOI] [PubMed] [Google Scholar]
- 10. Brown E, Talbot GH, Axelrod P, Provencher M, Hoegg C (1990) Risk factors for Clostridium difficile toxin-associated diarrhea. Infect Control Hosp Epidemiol 11: 283–290. [DOI] [PubMed] [Google Scholar]
- 11. Kwok CS, Arthur AK, Anibueze CI, Singh S, Cavallazzi R, et al. (2012) Risk of Clostridium difficile infection with acid suppressing drugs and antibiotics: meta-analysis. Am J Gastroenterol 107: 1011–1019. [DOI] [PubMed] [Google Scholar]
- 12. Deshpande A, Pant C, Pasupuleti V, Rolston DD, Jain A, et al. (2012) Association between proton pump inhibitor therapy and Clostridium difficile infection in a meta-analysis. Clin Gastroenterol Hepatol 10: 225–233. [DOI] [PubMed] [Google Scholar]
- 13. Bavishi C, Dupont HL (2011) Systematic review: the use of proton pump inhibitors and increased susceptibility to enteric infection. Aliment Pharmacol Ther 34: 1269–1281. [DOI] [PubMed] [Google Scholar]
- 14. Tleyjeh IM, Bin Abdulhak AA, Riaz M, Alasmari FA, Garbati MA, et al. (2012) Association between Proton Pump Inhibitor Therapy and Clostridium difficile Infection: A Contemporary Systematic Review and Meta-Analysis. PLoS ONE 7(12): e50836 doi:10.1371/journal.pone.0050836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.U.S food and drug administration. Proton Pump Inhibitors (PPIs) – Drug Safety Communication: Clostridium difficile-Associated Diarrhea (CDAD) Can be Associated With Stomach Acid Drugs. Available: http://www.fda.gov/Safety/MedWatch/SafetyInformation/SafetyAlertsforHumanMedicalProducts/ucm290838.htm. Accessed May, 31, 2012.
- 16. Shi CW, Gralnek IM, Dulai GS, Towfigh A, Asch S (2004) Consumer usage patterns of nonprescription histamine2-receptor antagonists. Am J Gastroenterol 99 (4): 606–10. [DOI] [PubMed] [Google Scholar]
- 17. Erwin J, Britten N, Jones R (1997) General practitioners' views on the over-the-counter availability of H2-antagonists. Br J Gen Pract 47: 99–102. [PMC free article] [PubMed] [Google Scholar]
- 18. Stroup DF, Berlin JA, Morton SC, Olkin I, Williamson GD, et al. (2000) Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA 283: 2008–2012. [DOI] [PubMed] [Google Scholar]
- 19. Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, et al. (2009) The PRISMA Statement for Reporting Systematic Reviews and Meta-Analyses of Studies That Evaluate Health Care Interventions: Explanation and Elaboration. PLoS Med 6(7): e1000100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Guyatt GH, Oxman AD, Vist GE, Kunz R, Falck-Ytter Y, et al. (2008) GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ 336: 924–926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wells G, Shea B, O'Connell D, et al. The Newcastle- Ottawa scale (NOS) for assessing the quality of non-randomized studies in meta-analysis. http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp. Last accessed May31.2012.
- 22. DerSimonian R, Laird N (1986) Meta-analysis in clinical trials. Control Clin Trials 7: 177–188. [DOI] [PubMed] [Google Scholar]
- 23. Egger M, Davey Smith G, Schneider M, Minder C (1997) Bias in meta-analysis detected by a simple, graphical test. BMJ 315: 629–634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Schneeweiss S (2006) Sensitivity analysis and external adjustment for unmeasured confounders in epidemiologic database studies of therapeutics. Pharmacoepidemiol Drug Saf 15: 291–303. [DOI] [PubMed] [Google Scholar]
- 25. Zhang J, Yu KF (1998) What's the relative risk? A method of correcting the odds ratio in cohort studies of common outcomes. JAMA 280: 1690–1691. [DOI] [PubMed] [Google Scholar]
- 26.Kutty PK, Woods CW, Sena AC, Benoit SR, Naggie S, et al. (2010) Risk factors for and estimated incidence of community-associated Clostridium difficile infection, North Carolina, USA. Emerg Infect Dis 16: 197 204. [DOI] [PMC free article] [PubMed]
- 27. Jayatilaka S, Shakov R, Eddi R, Bakaj G, Baddoura WJ, et al. (2007) Clostridium difficile infection in an urban medical center: five-year analysis of infection rates among adult admissions and association with the use of proton pump inhibitors. Ann Clin Lab Sci 37: 241–247. [PubMed] [Google Scholar]
- 28. Nath SK, Salama S, Persaud D, Thornley JH, Smith I, et al. (1994) Drug risk factors associated with a sustained outbreak of Clostridium difficile diarrhea in a teaching hospital. Can J Infect Dis 5: 270–275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Shah S, Lewis A, Leopold D, Dunstan F, Woodhouse K (2000) Gastric acid suppression does not promote clostridial diarrhoea in the elderly. QJM 93: 175–181. [DOI] [PubMed] [Google Scholar]
- 30. Dial S, Delaney JA, Barkun AN, Suissa S (2005) Use of gastric acid-suppressive agents and the risk of community-acquired Clostridium difficile-associated disease. JAMA 294: 2989–2995. [DOI] [PubMed] [Google Scholar]
- 31. Debast SB, Vaessen N, Choudry A, Wiegers-Ligtvoet EA, van den Berg RJ, et al. (2009) Successful combat of an outbreak due to Clostridium difficile PCR ribotype 027 and recognition of specific risk factors. Clin Microbiol Infect 15: 427–434. [DOI] [PubMed] [Google Scholar]
- 32. Lowe DO, Mamdani MM, Kopp A, Low DE, Juurlink DN (2006) Proton pump inhibitors and hospitalization for Clostridium difficile-associated disease: a population-based study. Clin Infect Dis 43: 1272–1276. [DOI] [PubMed] [Google Scholar]
- 33. Dial S, Delaney JA, Schneider V, Suissa S (2006) Proton pump inhibitor use and risk of community acquired Clostridium difficile-associated disease defined by prescription for oral vancomycin therapy.CMAJ. 175: 745–748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Aseeri M, Schroeder T, Kramer J, Zackula R (2008) Gastric acid suppression by proton pump inhibitors as a risk factor for Clostridium difficile-associated diarrhea in hospitalized patients. Am J Gastroenterol 103: 2308–2313. [DOI] [PubMed] [Google Scholar]
- 35. Dubberke ER, Reske KA, Olsen MA, McMullen KM, Mayfield JL, et al. (2007) Evaluation of Clostridium difficile-associated disease pressure as a risk factor for C difficile-associated disease. Arch Intern Med (167) 1092–1097. [DOI] [PubMed] [Google Scholar]
- 36. Loo VG, Poirier L, Miller MA, Oughton M, Libman MD, et al. (2005) A predominantly clonal multi-institutional outbreak of Clostridium difficile-associated diarrhea with high morbidity and mortality. N Engl J Med 353: 2442–2449. [DOI] [PubMed] [Google Scholar]
- 37. Sundram F, Guyot A, Carboo I, Green S, Lilaonitkul M, et al. (2009) Clostridium difficile ribotypes 027 and 106: clinical outcomes and risk factors. J Hosp Infect 72: 111–118. [DOI] [PubMed] [Google Scholar]
- 38. Howell MD, Novack V, Grgurich P, Soulliard D, Novack L, et al. (2010) Iatrogenic gastric acid suppression and the risk of nosocomial Clostridium difficile infection. Arch Intern Med 170: 784–790. [DOI] [PubMed] [Google Scholar]
- 39. Dalton BR, Lye-Maccannell T, Henderson EA, Maccannell DR, Louie TJ (2009) Proton pump inhibitors increase significantly the risk of Clostridium difficile infection in a low-endemicity, non-outbreak hospital setting. Aliment Pharmacol Ther 29: 626–634. [DOI] [PubMed] [Google Scholar]
- 40. Dubberke ER, Reske KA, Yan Y, Olsen MA, McDonald LC, et al. (2007) Clostridium difficile – associated disease in a setting of endemicity: identification of novel risk factors. Clin Infect Dis 45: 1543–1549. [DOI] [PubMed] [Google Scholar]
- 41. Pepin J, Saheb N, Coulombe MA, Alary ME, Corriveau MP, et al. (2005) Emergence of fluoroquinolones as the predominant risk factor for Clostridium difficile-associated diarrhea: a cohort study during an epidemic in Quebec. Clin Infect Dis 41: 1254–1260. [DOI] [PubMed] [Google Scholar]
- 42. Beaulieu M, Williamson D, Pichette G, Lachaine J (2007) Risk of Clostridium difficile-associated disease among patients receiving proton-pump inhibitors in a Quebec medical intensive care unit. Infect Control Hosp Epidemiol 28: 1305–1307. [DOI] [PubMed] [Google Scholar]
- 43. Peled N, Pitlik S, Samra Z, Kazakov A, Bloch Y, et al. (2007) Predicting Clostridium difficile toxin in hospitalized patients with antibiotic-associated diarrhea. Infect Control Hosp Epidemiol 28: 377–381. [DOI] [PubMed] [Google Scholar]
- 44. Dial S, Alrasadi K, Manoukian C, Huang A, Menzies D (2004) Risk of Clostridium difficile diarrhea among hospital inpatients prescribed proton pump inhibitors: cohort and case-control studies. CMAJ 171: 33–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Novell MJ, Morreale CA (2010) The relationship between inpatient fluoroquinolone use and Clostridium difficile-associated diarrhea. Ann Pharmacother : 826–831. [DOI] [PubMed]
- 46. Netland PJ, Brochardt SM, Koo JM, Lo TS, Krenz T (2011) A Retrospective Analysis of Risk factors and Therapeutic Outcomes for Clostridium difficile Infection. Hosp Pham 46(5): 336–340. [Google Scholar]
- 47. Jung KS, Park JJ, Chon YE, Jung ES, Lee HJ, et al. (2010) Risk Factors for Treatment Failure and Recurrence after Metronidazole Treatment for Clostridium difficile-associated Diarrhea. Gut Liver 4: 332–337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Loo VG, Bourgault AM, Poirier L, Lamothe F, Michaud S, et al. (2011) Host and pathogen factors for Clostridium difficile infection and colonization. N Engl J Med 365: 1693–1703. [DOI] [PubMed] [Google Scholar]
- 49. Manges AR, Labbe A, Loo VG, Atherton JK, Behr MA, et al. (2010) Comparative metagenomic study of alterations to the intestinal microbiota and risk of nosocomial Clostridum difficile-associated disease. J Infect Dis 202: 1877–1884. [DOI] [PubMed] [Google Scholar]
- 50. Kuntz JL, Chrischilles EA, Pendergast JF, Herwaldt LA, Polgreen PM (2011) Incidence of and risk factors for community-associated Clostridium difficile infection: a nested case-control study. BMC Infect Dis 11: 194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Naggie S, Miller BA, Zuzak KB, Pence BW, Mayo AJ, et al. (2011) A case control study of community associated Clostridium difficile infection: no role for proton pump inhibitors. Am J Med 124: 276 e271–277. [DOI] [PubMed]
- 52. Stevens V, Dumyati G, Brown J, Wijngaarden E (2011) Differential risk of Clostridium difficile infection with proton pump inhibitor use by level of antibiotic exposure. Pharmacoepidemiol Drug Saf 20: 1035–1042. [DOI] [PubMed] [Google Scholar]
- 53. Dial S, Kezouh A, Dascal A, Barkun A, Suissa S (2008) Patterns of antibiotic use and risk of hospital admission because of Clostridium difficile infection. CMAJ 179: 767–772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. McFarland LV, Clarridge JE, Beneda HW, Raugi GJ (2007) Fluoroquinolone use and risk factors for Clostridium difficile-associated disease within a Veterans Administration health care system. Clin Infect Dis 45: 1141–1151. [DOI] [PubMed] [Google Scholar]
- 55. Kazakova SV, Ware K, Baughman B, Bilukha O, Paradis A, et al. (2006) A hospital outbreak of diarrhea due to an emerging epidemic strain of Clostridium difficile . Arch Intern Med 166: 2518–2524. [DOI] [PubMed] [Google Scholar]
- 56. Modena S, Bearelly D, Swartz K, Friedenberg FK (2005) Clostridium difficile among hospitalized patients receiving antibiotics: a case-control study. Infect Control Hosp Epidemiol 26: 685–690. [DOI] [PubMed] [Google Scholar]
- 57. Muto CA, Pokrywka M, Shutt K, Mendelsohn AB, Nouri K, et al. (2005) A large outbreak of Clostridium difficile-associated disease with an unexpected proportion of deaths and colectomies at a teaching hospital following increased fluoroquinolone use. Infect Control Hosp Epidemiol 26: 273–280. [DOI] [PubMed] [Google Scholar]
- 58. Yip C, Loeb M, Salama S, Moss L, Olde J (2001) Quinolone use as a risk factor for nosocomial Clostridium difficile-associated diarrhea. Infect Control Hosp Epidemiol 22: 572–575. [DOI] [PubMed] [Google Scholar]
- 59. Cates CJ (2002) Simpson's paradox and calculation of number needed to treat from meta-analysis. BMC Med Res Methodol 2: 1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Khanna S, Pardi DS, Aronson SL, Kammer PP, Orenstein R, et al. (2012) The epidemiology of community-acquired Clostridium difficile infection: a population based study. Am J Gastroenterol 107: 89–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Leonard J, Marshall JK, Moayyedi P (2007) Systematic review of the risk of enteric infection in patients taking acid suppression. Am J Gastroenterol 102: 2047–2056. [DOI] [PubMed] [Google Scholar]
- 62. Jump RL, Pultz MJ, Donskey CJ (2007) Vegetative Clostridium difficile survives in room air on moist surfaces and in gastric contents with reduced acidity: a potential mechanism to explain the association between proton pump inhibitors and C. difficile associated diarrhea? Antimicrob Agents Chemother 51: 2883–2887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Coory MD (2010) Comment on: Heterogeneity in meta-analysis should be expected and appropriately quantified. Int J Epidemiol 39: 932; author reply 933. [DOI] [PubMed]
- 64. Higgins JP (2008) Commentary: Heterogeneity in meta-analysis should be expected and appropriately quantified. Int J Epidemiol 37: 1158–1160. [DOI] [PubMed] [Google Scholar]
- 65. Higgins JP, Thompson SG, Deeks JJ, Altman DG (2003) Measuring inconsistency in meta analyses. BMJ 327: 557–560. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
PRISMA 2009 flow diagram.
(DOC)
PRISMA checklist.
(DOC)