Abstract
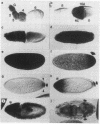
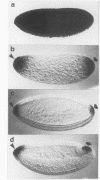
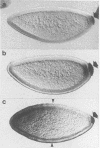
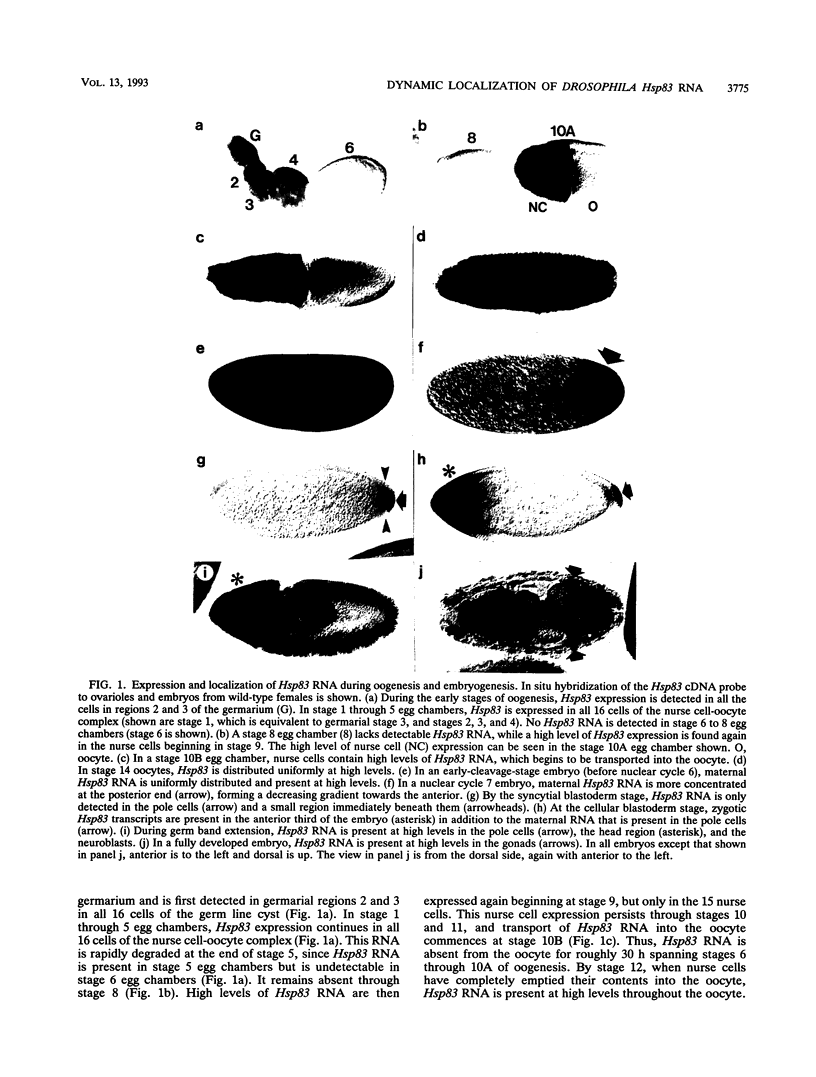
Hsp83 is the Drosophila homolog of the mammalian Hsp90 family of regulatory molecular chaperones. We show that maternally synthesized Hsp83 transcripts are localized to the posterior pole of the early Drosophila embryo by a novel mechanism involving a combination of generalized RNA degradation and local protection at the posterior. This protection of Hsp83 RNA occurs in wild-type embryos and embryos produced by females carrying the maternal effect mutations nanos and pumilio, which eliminate components of the posterior polar plasm without disrupting polar granule integrity. In contrast, Hsp83 RNA is not protected at the posterior pole of embryos produced by females carrying maternal mutations that disrupt the posterior polar plasm and the polar granules--cappuccino, oskar, spire, staufen, tudor, valois, and vasa. Mislocalization of oskar RNA to the anterior pole, which has been shown to result in induction of germ cells at the anterior, leads to anterior protection of maternal Hsp83 RNA. These results suggest that Hsp83 RNA is a component of the posterior polar plasm that might be associated with polar granules. In addition, we show that zygotic expression of Hsp83 commences in the anterior third of the embryo at the syncytial blastoderm stage and is regulated by the anterior morphogen, bicoid. We consider the possible developmental significance of this complex control of Hsp83 transcript distribution.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Berleth T., Burri M., Thoma G., Bopp D., Richstein S., Frigerio G., Noll M., Nüsslein-Volhard C. The role of localization of bicoid RNA in organizing the anterior pattern of the Drosophila embryo. EMBO J. 1988 Jun;7(6):1749–1756. doi: 10.1002/j.1460-2075.1988.tb03004.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blackman R. K., Meselson M. Interspecific nucleotide sequence comparisons used to identify regulatory and structural features of the Drosophila hsp82 gene. J Mol Biol. 1986 Apr 20;188(4):499–515. doi: 10.1016/s0022-2836(86)80001-8. [DOI] [PubMed] [Google Scholar]
- Borkovich K. A., Farrelly F. W., Finkelstein D. B., Taulien J., Lindquist S. hsp82 is an essential protein that is required in higher concentrations for growth of cells at higher temperatures. Mol Cell Biol. 1989 Sep;9(9):3919–3930. doi: 10.1128/mcb.9.9.3919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bossy B., Hall L. M., Spierer P. Genetic activity along 315 kb of the Drosophila chromosome. EMBO J. 1984 Nov;3(11):2537–2541. doi: 10.1002/j.1460-2075.1984.tb02169.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boswell R. E., Mahowald A. P. tudor, a gene required for assembly of the germ plasm in Drosophila melanogaster. Cell. 1985 Nov;43(1):97–104. doi: 10.1016/0092-8674(85)90015-7. [DOI] [PubMed] [Google Scholar]
- Cohen S., Jürgens G. Drosophila headlines. Trends Genet. 1991 Aug;7(8):267–272. doi: 10.1016/0168-9525(91)90327-M. [DOI] [PubMed] [Google Scholar]
- Curci A., Bevilacqua A., Fiorenza M. T., Mangia F. Developmental regulation of heat-shock response in mouse oogenesis: identification of differentially responsive oocyte classes during Graafian follicle development. Dev Biol. 1991 Apr;144(2):362–368. doi: 10.1016/0012-1606(91)90428-6. [DOI] [PubMed] [Google Scholar]
- Ding D., Parkhurst S. M., Lipshitz H. D. Different genetic requirements for anterior RNA localization revealed by the distribution of Adducin-like transcripts during Drosophila oogenesis. Proc Natl Acad Sci U S A. 1993 Mar 15;90(6):2512–2516. doi: 10.1073/pnas.90.6.2512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Driever W., Nüsslein-Volhard C. A gradient of bicoid protein in Drosophila embryos. Cell. 1988 Jul 1;54(1):83–93. doi: 10.1016/0092-8674(88)90182-1. [DOI] [PubMed] [Google Scholar]
- Driever W., Nüsslein-Volhard C. The bicoid protein determines position in the Drosophila embryo in a concentration-dependent manner. Cell. 1988 Jul 1;54(1):95–104. doi: 10.1016/0092-8674(88)90183-3. [DOI] [PubMed] [Google Scholar]
- Ephrussi A., Dickinson L. K., Lehmann R. Oskar organizes the germ plasm and directs localization of the posterior determinant nanos. Cell. 1991 Jul 12;66(1):37–50. doi: 10.1016/0092-8674(91)90137-n. [DOI] [PubMed] [Google Scholar]
- Ephrussi A., Lehmann R. Induction of germ cell formation by oskar. Nature. 1992 Jul 30;358(6385):387–392. doi: 10.1038/358387a0. [DOI] [PubMed] [Google Scholar]
- Finkelstein R., Perrimon N. The molecular genetics of head development in Drosophila melanogaster. Development. 1991 Aug;112(4):899–912. doi: 10.1242/dev.112.4.899. [DOI] [PubMed] [Google Scholar]
- Golumbeski G. S., Bardsley A., Tax F., Boswell R. E. tudor, a posterior-group gene of Drosophila melanogaster, encodes a novel protein and an mRNA localized during mid-oogenesis. Genes Dev. 1991 Nov;5(11):2060–2070. doi: 10.1101/gad.5.11.2060. [DOI] [PubMed] [Google Scholar]
- Gottlieb E. Messenger RNA transport and localization. Curr Opin Cell Biol. 1990 Dec;2(6):1080–1086. doi: 10.1016/0955-0674(90)90159-c. [DOI] [PubMed] [Google Scholar]
- Gruppi C. M., Zakeri Z. F., Wolgemuth D. J. Stage and lineage-regulated expression of two hsp90 transcripts during mouse germ cell differentiation and embryogenesis. Mol Reprod Dev. 1991 Mar;28(3):209–217. doi: 10.1002/mrd.1080280302. [DOI] [PubMed] [Google Scholar]
- Hackett R. W., Lis J. T. Localization of the hsp83 transcript within a 3292 nucleotide sequence from the 63B heat shock locus of D. melanogaster. Nucleic Acids Res. 1983 Oct 25;11(20):7011–7030. doi: 10.1093/nar/11.20.7011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hay B., Ackerman L., Barbel S., Jan L. Y., Jan Y. N. Identification of a component of Drosophila polar granules. Development. 1988 Aug;103(4):625–640. doi: 10.1242/dev.103.4.625. [DOI] [PubMed] [Google Scholar]
- Hay B., Jan L. Y., Jan Y. N. A protein component of Drosophila polar granules is encoded by vasa and has extensive sequence similarity to ATP-dependent helicases. Cell. 1988 Nov 18;55(4):577–587. doi: 10.1016/0092-8674(88)90216-4. [DOI] [PubMed] [Google Scholar]
- Hay B., Jan L. Y., Jan Y. N. Localization of vasa, a component of Drosophila polar granules, in maternal-effect mutants that alter embryonic anteroposterior polarity. Development. 1990 Jun;109(2):425–433. doi: 10.1242/dev.109.2.425. [DOI] [PubMed] [Google Scholar]
- Hoffmann T., Hovemann B. Heat-shock proteins, Hsp84 and Hsp86, of mice and men: two related genes encode formerly identified tumour-specific transplantation antigens. Gene. 1988 Dec 30;74(2):491–501. doi: 10.1016/0378-1119(88)90182-5. [DOI] [PubMed] [Google Scholar]
- Holmgren R., Livak K., Morimoto R., Freund R., Meselson M. Studies of cloned sequences from four Drosophila heat shock loci. Cell. 1979 Dec;18(4):1359–1370. doi: 10.1016/0092-8674(79)90246-0. [DOI] [PubMed] [Google Scholar]
- Jongens T. A., Hay B., Jan L. Y., Jan Y. N. The germ cell-less gene product: a posteriorly localized component necessary for germ cell development in Drosophila. Cell. 1992 Aug 21;70(4):569–584. doi: 10.1016/0092-8674(92)90427-e. [DOI] [PubMed] [Google Scholar]
- Katzen A. L., Kornberg T., Bishop J. M. Diverse expression of dsrc29A, a gene related to src, during the life cycle of Drosophila melanogaster. Development. 1990 Dec;110(4):1169–1183. doi: 10.1242/dev.110.4.1169. [DOI] [PubMed] [Google Scholar]
- Kim-Ha J., Smith J. L., Macdonald P. M. oskar mRNA is localized to the posterior pole of the Drosophila oocyte. Cell. 1991 Jul 12;66(1):23–35. doi: 10.1016/0092-8674(91)90136-m. [DOI] [PubMed] [Google Scholar]
- Kurtz S., Rossi J., Petko L., Lindquist S. An ancient developmental induction: heat-shock proteins induced in sporulation and oogenesis. Science. 1986 Mar 7;231(4742):1154–1157. doi: 10.1126/science.3511530. [DOI] [PubMed] [Google Scholar]
- Lai B. T., Chin N. W., Stanek A. E., Keh W., Lanks K. W. Quantitation and intracellular localization of the 85K heat shock protein by using monoclonal and polyclonal antibodies. Mol Cell Biol. 1984 Dec;4(12):2802–2810. doi: 10.1128/mcb.4.12.2802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lantz V., Ambrosio L., Schedl P. The Drosophila orb gene is predicted to encode sex-specific germline RNA-binding proteins and has localized transcripts in ovaries and early embryos. Development. 1992 May;115(1):75–88. doi: 10.1242/dev.115.1.75. [DOI] [PubMed] [Google Scholar]
- Lasko P. F., Ashburner M. Posterior localization of vasa protein correlates with, but is not sufficient for, pole cell development. Genes Dev. 1990 Jun;4(6):905–921. doi: 10.1101/gad.4.6.905. [DOI] [PubMed] [Google Scholar]
- Lasko P. F., Ashburner M. The product of the Drosophila gene vasa is very similar to eukaryotic initiation factor-4A. Nature. 1988 Oct 13;335(6191):611–617. doi: 10.1038/335611a0. [DOI] [PubMed] [Google Scholar]
- Lee S. J. Expression of HSP86 in male germ cells. Mol Cell Biol. 1990 Jun;10(6):3239–3242. doi: 10.1128/mcb.10.6.3239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lehmann R., Nüsslein-Volhard C. Abdominal segmentation, pole cell formation, and embryonic polarity require the localized activity of oskar, a maternal gene in Drosophila. Cell. 1986 Oct 10;47(1):141–152. doi: 10.1016/0092-8674(86)90375-2. [DOI] [PubMed] [Google Scholar]
- Lehmann R., Nüsslein-Volhard C. The maternal gene nanos has a central role in posterior pattern formation of the Drosophila embryo. Development. 1991 Jul;112(3):679–691. doi: 10.1242/dev.112.3.679. [DOI] [PubMed] [Google Scholar]
- Lehmann R., Nüsslein-Volhard C. hunchback, a gene required for segmentation of an anterior and posterior region of the Drosophila embryo. Dev Biol. 1987 Feb;119(2):402–417. doi: 10.1016/0012-1606(87)90045-5. [DOI] [PubMed] [Google Scholar]
- Lehner C. F., O'Farrell P. H. The roles of Drosophila cyclins A and B in mitotic control. Cell. 1990 May 4;61(3):535–547. doi: 10.1016/0092-8674(90)90535-m. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lindquist S., Craig E. A. The heat-shock proteins. Annu Rev Genet. 1988;22:631–677. doi: 10.1146/annurev.ge.22.120188.003215. [DOI] [PubMed] [Google Scholar]
- Macdonald P. M. The Drosophila pumilio gene: an unusually long transcription unit and an unusual protein. Development. 1992 Jan;114(1):221–232. doi: 10.1242/dev.114.1.221. [DOI] [PubMed] [Google Scholar]
- Mahowald A. P. Polar granules of Drosophila. 3. The continuity of polar granules during the life cycle of Drosophila. J Exp Zool. 1971 Mar;176(3):329–343. doi: 10.1002/jez.1401760308. [DOI] [PubMed] [Google Scholar]
- Mahowald A. P. Polar granules of Drosophila. II. Ultrastructural changes during early embryogenesis. J Exp Zool. 1968 Feb;167(2):237–261. doi: 10.1002/jez.1401670211. [DOI] [PubMed] [Google Scholar]
- Mahowald A. P. Polar granules of drosophila. IV. Cytochemical studies showing loss of RNA from polar granules during early stages of embryogenesis. J Exp Zool. 1971 Mar;176(3):345–352. doi: 10.1002/jez.1401760309. [DOI] [PubMed] [Google Scholar]
- Manseau L. J., Schüpbach T. The egg came first, of course! Anterior-posterior pattern formation in Drosophila embryogenesis and oogenesis. Trends Genet. 1989 Dec;5(12):400–405. doi: 10.1016/0168-9525(89)90198-4. [DOI] [PubMed] [Google Scholar]
- Manseau L. J., Schüpbach T. cappuccino and spire: two unique maternal-effect loci required for both the anteroposterior and dorsoventral patterns of the Drosophila embryo. Genes Dev. 1989 Sep;3(9):1437–1452. doi: 10.1101/gad.3.9.1437. [DOI] [PubMed] [Google Scholar]
- Moore S. K., Kozak C., Robinson E. A., Ullrich S. J., Appella E. Murine 86- and 84-kDa heat shock proteins, cDNA sequences, chromosome assignments, and evolutionary origins. J Biol Chem. 1989 Apr 5;264(10):5343–5351. [PubMed] [Google Scholar]
- Palazzolo M. J., Hamilton B. A., Ding D. L., Martin C. H., Mead D. A., Mierendorf R. C., Raghavan K. V., Meyerowitz E. M., Lipshitz H. D. Phage lambda cDNA cloning vectors for subtractive hybridization, fusion-protein synthesis and Cre-loxP automatic plasmid subcloning. Gene. 1990 Mar 30;88(1):25–36. doi: 10.1016/0378-1119(90)90056-w. [DOI] [PubMed] [Google Scholar]
- Raff J. W., Whitfield W. G., Glover D. M. Two distinct mechanisms localise cyclin B transcripts in syncytial Drosophila embryos. Development. 1990 Dec;110(4):1249–1261. doi: 10.1242/dev.110.4.1249. [DOI] [PubMed] [Google Scholar]
- Schüpbach T., Wieschaus E. Female sterile mutations on the second chromosome of Drosophila melanogaster. I. Maternal effect mutations. Genetics. 1989 Jan;121(1):101–117. doi: 10.1093/genetics/121.1.101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- St Johnston D., Beuchle D., Nüsslein-Volhard C. Staufen, a gene required to localize maternal RNAs in the Drosophila egg. Cell. 1991 Jul 12;66(1):51–63. doi: 10.1016/0092-8674(91)90138-o. [DOI] [PubMed] [Google Scholar]
- St Johnston D., Driever W., Berleth T., Richstein S., Nüsslein-Volhard C. Multiple steps in the localization of bicoid RNA to the anterior pole of the Drosophila oocyte. Development. 1989;107 (Suppl):13–19. doi: 10.1242/dev.107.Supplement.13. [DOI] [PubMed] [Google Scholar]
- Struhl G., Struhl K., Macdonald P. M. The gradient morphogen bicoid is a concentration-dependent transcriptional activator. Cell. 1989 Jun 30;57(7):1259–1273. doi: 10.1016/0092-8674(89)90062-7. [DOI] [PubMed] [Google Scholar]
- Suter B., Romberg L. M., Steward R. Bicaudal-D, a Drosophila gene involved in developmental asymmetry: localized transcript accumulation in ovaries and sequence similarity to myosin heavy chain tail domains. Genes Dev. 1989 Dec;3(12A):1957–1968. doi: 10.1101/gad.3.12a.1957. [DOI] [PubMed] [Google Scholar]
- Tautz D., Pfeifle C. A non-radioactive in situ hybridization method for the localization of specific RNAs in Drosophila embryos reveals translational control of the segmentation gene hunchback. Chromosoma. 1989 Aug;98(2):81–85. doi: 10.1007/BF00291041. [DOI] [PubMed] [Google Scholar]
- Vincent W. S., 3rd, Gregory R. J., Wadsworth S. C. Embryonic expression of a Drosophila src gene: alternate forms of the protein are expressed in segmental stripes and in the nervous system. Genes Dev. 1989 Mar;3(3):334–347. doi: 10.1101/gad.3.3.334. [DOI] [PubMed] [Google Scholar]
- Wang C., Lehmann R. Nanos is the localized posterior determinant in Drosophila. Cell. 1991 Aug 23;66(4):637–647. doi: 10.1016/0092-8674(91)90110-k. [DOI] [PubMed] [Google Scholar]
- Wohlwill A. D., Bonner J. J. Genetic analysis of chromosome region 63 of Drosophila melanogaster. Genetics. 1991 Aug;128(4):763–775. doi: 10.1093/genetics/128.4.763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiao H., Lis J. T. Heat shock and developmental regulation of the Drosophila melanogaster hsp83 gene. Mol Cell Biol. 1989 Apr;9(4):1746–1753. doi: 10.1128/mcb.9.4.1746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zimmerman J. L., Petri W., Meselson M. Accumulation of a specific subset of D. melanogaster heat shock mRNAs in normal development without heat shock. Cell. 1983 Apr;32(4):1161–1170. doi: 10.1016/0092-8674(83)90299-4. [DOI] [PubMed] [Google Scholar]