Abstract
Two forms of activated BCR/ABL proteins, P210 and P185, that differ in BCR-derived sequences, are associated with Philadelphia chromosome-positive leukemias. One of these diseases is chronic myelogenous leukemia, an indolent disease arising in hematopoietic stem cells that is almost always associated with the P210 form of BCR/ABL. Acute lymphocytic leukemia, a more aggressive malignancy, can be associated with both forms of BCR/ABL. While it is virtually certain that BCR/ABL plays a central role in both of these diseases, the features that determine the association of a particular form with a given disease have not been elucidated. We have used the bone marrow reconstitution leukemogenesis model to test the hypothesis that BCR sequences influence the ability of activated ABL to transform different types of hematopoietic cells. Our studies reveal that both P185 and P210 induce a similar spectrum of hematological diseases, including granulocytic, myelomonocytic, and lymphocytic leukemias. Despite the similarity of the disease patterns, animals given P185-infected marrow developed a more aggressive disease after a shorter latent period than those given P210-infected marrow. These data demonstrate that the structure of the BCR/ABL oncoprotein does not affect the type of disease induced by each form of the oncogene but does control the potency of the oncogenic signal.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bergold P. J., Blumenthal J. A., D'Andrea E., Snyder H. W., Lederman L., Silverstone A., Nguyen H., Besmer P. Nucleic acid sequence and oncogenic properties of the HZ2 feline sarcoma virus v-abl insert. J Virol. 1987 Apr;61(4):1193–1202. doi: 10.1128/jvi.61.4.1193-1202.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Champlin R. E., Golde D. W. Chronic myelogenous leukemia: recent advances. Blood. 1985 May;65(5):1039–1047. [PubMed] [Google Scholar]
- Chan L. C., Karhi K. K., Rayter S. I., Heisterkamp N., Eridani S., Powles R., Lawler S. D., Groffen J., Foulkes J. G., Greaves M. F. A novel abl protein expressed in Philadelphia chromosome positive acute lymphoblastic leukaemia. Nature. 1987 Feb 12;325(6105):635–637. doi: 10.1038/325635a0. [DOI] [PubMed] [Google Scholar]
- Chen S. J., Flandrin G., Daniel M. T., Valensi F., Baranger L., Grausz D., Bernheim A., Chen Z., Sigaux F., Berger R. Philadelphia-positive acute leukemia: lineage promiscuity and inconsistently rearranged breakpoint cluster region. Leukemia. 1988 May;2(5):261–273. [PubMed] [Google Scholar]
- Chung S. W., Wong P. M., Durkin H., Wu Y. S., Petersen J. Leukemia initiated by hemopoietic stem cells expressing the v-abl oncogene. Proc Natl Acad Sci U S A. 1991 Feb 15;88(4):1585–1589. doi: 10.1073/pnas.88.4.1585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clark S. S., Crist W. M., Witte O. N. Molecular pathogenesis of Ph-positive leukemias. Annu Rev Med. 1989;40:113–122. doi: 10.1146/annurev.me.40.020189.000553. [DOI] [PubMed] [Google Scholar]
- Clark S. S., McLaughlin J., Crist W. M., Champlin R., Witte O. N. Unique forms of the abl tyrosine kinase distinguish Ph1-positive CML from Ph1-positive ALL. Science. 1987 Jan 2;235(4784):85–88. doi: 10.1126/science.3541203. [DOI] [PubMed] [Google Scholar]
- Cook W. D., Fazekas de St Groth B., Miller J. F., MacDonald H. R., Gabathuler R. Abelson virus transformation of an interleukin 2-dependent antigen-specific T-cell line. Mol Cell Biol. 1987 Jul;7(7):2631–2635. doi: 10.1128/mcb.7.7.2631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cook W. D., Metcalf D., Nicola N. A., Burgess A. W., Walker F. Malignant transformation of a growth factor-dependent myeloid cell line by Abelson virus without evidence of an autocrine mechanism. Cell. 1985 Jul;41(3):677–683. doi: 10.1016/s0092-8674(85)80048-9. [DOI] [PubMed] [Google Scholar]
- Daley G. Q., Baltimore D. Transformation of an interleukin 3-dependent hematopoietic cell line by the chronic myelogenous leukemia-specific P210bcr/abl protein. Proc Natl Acad Sci U S A. 1988 Dec;85(23):9312–9316. doi: 10.1073/pnas.85.23.9312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Daley G. Q., McLaughlin J., Witte O. N., Baltimore D. The CML-specific P210 bcr/abl protein, unlike v-abl, does not transform NIH/3T3 fibroblasts. Science. 1987 Jul 31;237(4814):532–535. doi: 10.1126/science.2440107. [DOI] [PubMed] [Google Scholar]
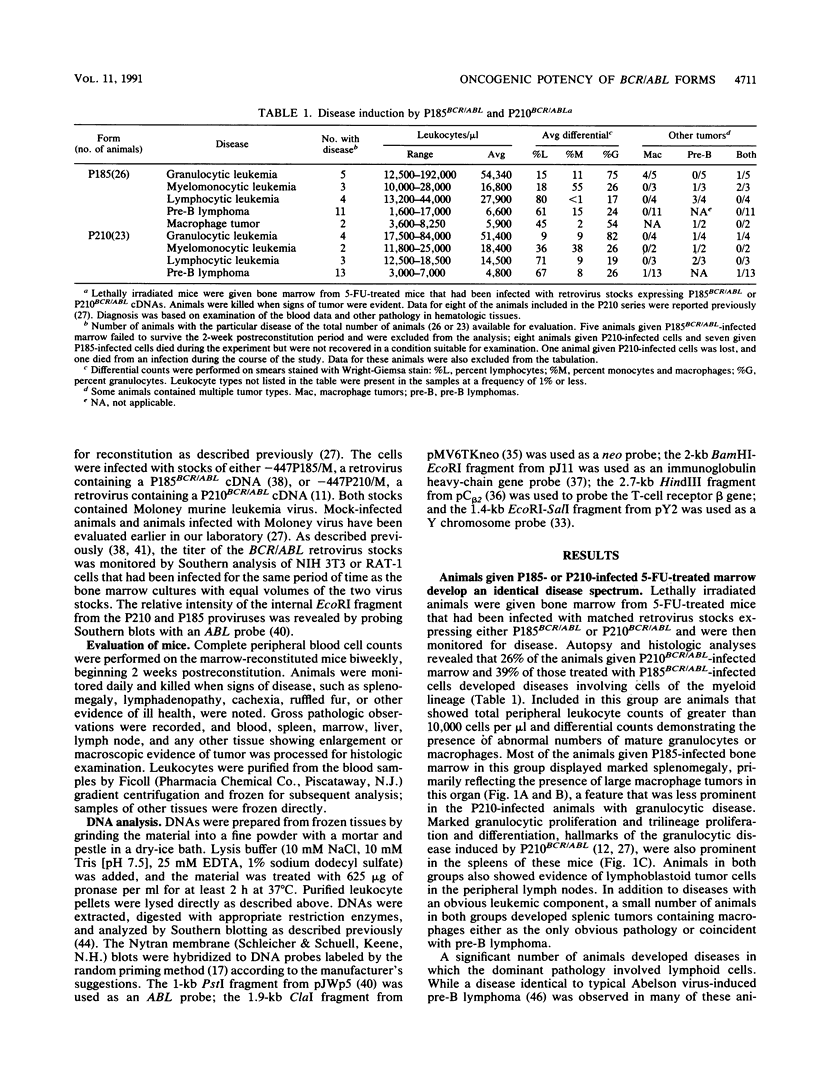
- Daley G. Q., Van Etten R. A., Baltimore D. Induction of chronic myelogenous leukemia in mice by the P210bcr/abl gene of the Philadelphia chromosome. Science. 1990 Feb 16;247(4944):824–830. doi: 10.1126/science.2406902. [DOI] [PubMed] [Google Scholar]
- Elefanty A. G., Hariharan I. K., Cory S. bcr-abl, the hallmark of chronic myeloid leukaemia in man, induces multiple haemopoietic neoplasms in mice. EMBO J. 1990 Apr;9(4):1069–1078. doi: 10.1002/j.1460-2075.1990.tb08212.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Erikson J., Griffin C. A., ar-Rushdi A., Valtieri M., Hoxie J., Finan J., Emanuel B. S., Rovera G., Nowell P. C., Croce C. M. Heterogeneity of chromosome 22 breakpoint in Philadelphia-positive (Ph+) acute lymphocytic leukemia. Proc Natl Acad Sci U S A. 1986 Mar;83(6):1807–1811. doi: 10.1073/pnas.83.6.1807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fainstein E., Marcelle C., Rosner A., Canaani E., Gale R. P., Dreazen O., Smith S. D., Croce C. M. A new fused transcript in Philadelphia chromosome positive acute lymphocytic leukaemia. 1987 Nov 26-Dec 2Nature. 330(6146):386–388. doi: 10.1038/330386a0. [DOI] [PubMed] [Google Scholar]
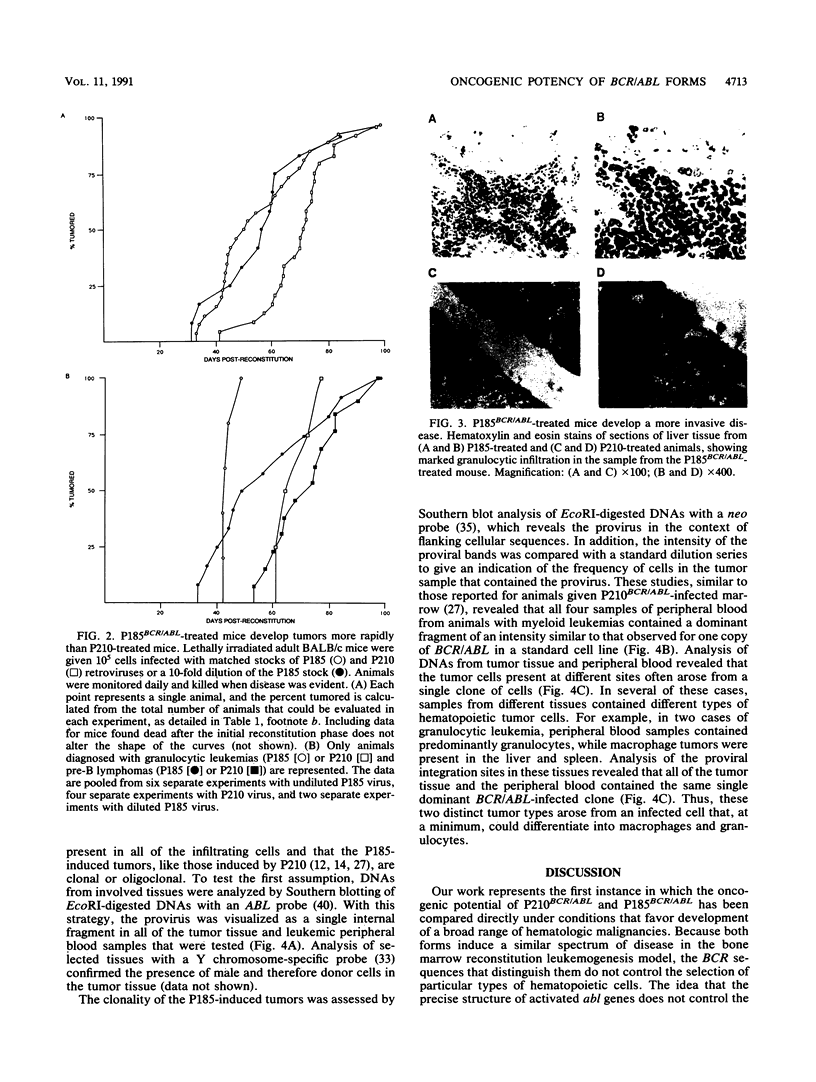
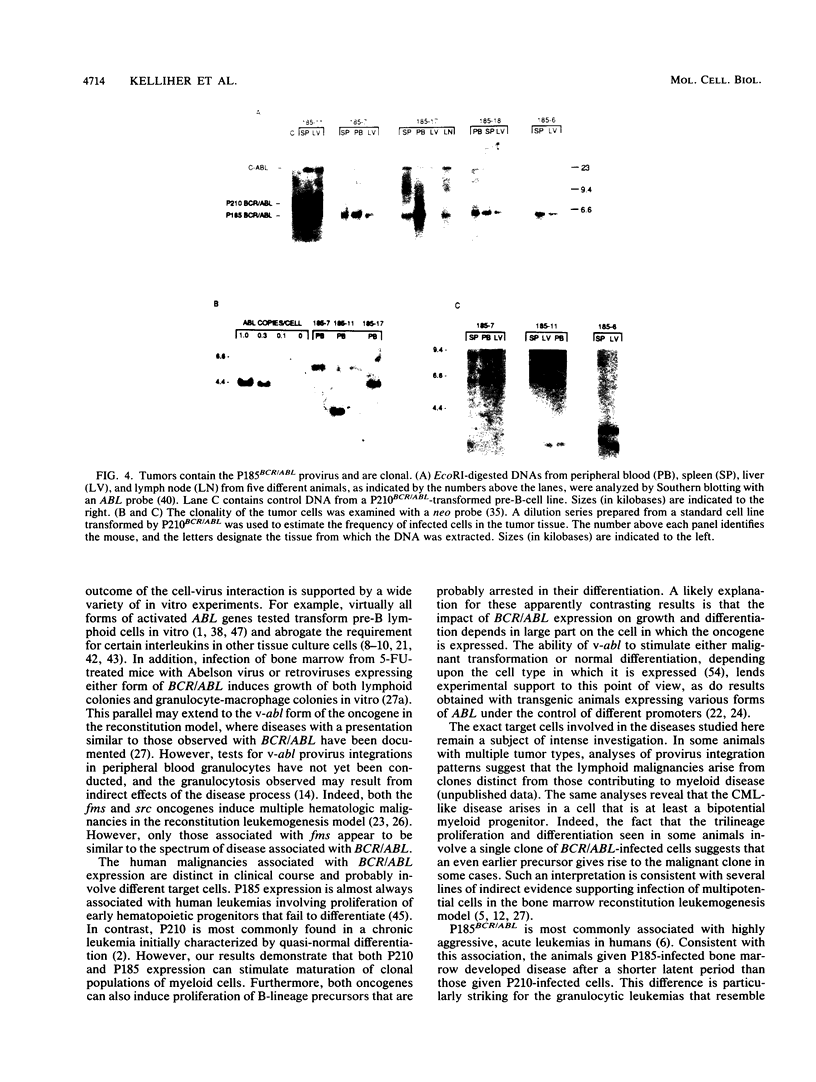
- Feinberg A. P., Vogelstein B. A technique for radiolabeling DNA restriction endonuclease fragments to high specific activity. Anal Biochem. 1983 Jul 1;132(1):6–13. doi: 10.1016/0003-2697(83)90418-9. [DOI] [PubMed] [Google Scholar]
- Fialkow P. J., Jacobson R. J., Papayannopoulou T. Chronic myelocytic leukemia: clonal origin in a stem cell common to the granulocyte, erythrocyte, platelet and monocyte/macrophage. Am J Med. 1977 Jul;63(1):125–130. doi: 10.1016/0002-9343(77)90124-3. [DOI] [PubMed] [Google Scholar]
- Groffen J., Stephenson J. R., Heisterkamp N., de Klein A., Bartram C. R., Grosveld G. Philadelphia chromosomal breakpoints are clustered within a limited region, bcr, on chromosome 22. Cell. 1984 Jan;36(1):93–99. doi: 10.1016/0092-8674(84)90077-1. [DOI] [PubMed] [Google Scholar]
- Hariharan I. K., Adams J. M., Cory S. bcr-abl oncogene renders myeloid cell line factor independent: potential autocrine mechanism in chronic myeloid leukemia. Oncogene Res. 1988;3(4):387–399. [PubMed] [Google Scholar]
- Hariharan I. K., Adams J. M. cDNA sequence for human bcr, the gene that translocates to the abl oncogene in chronic myeloid leukaemia. EMBO J. 1987 Jan;6(1):115–119. doi: 10.1002/j.1460-2075.1987.tb04727.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hariharan I. K., Harris A. W., Crawford M., Abud H., Webb E., Cory S., Adams J. M. A bcr-v-abl oncogene induces lymphomas in transgenic mice. Mol Cell Biol. 1989 Jul;9(7):2798–2805. doi: 10.1128/mcb.9.7.2798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heard J. M., Roussel M. F., Rettenmier C. W., Sherr C. J. Multilineage hematopoietic disorders induced by transplantation of bone marrow cells expressing the v-fms oncogene. Cell. 1987 Nov 20;51(4):663–673. doi: 10.1016/0092-8674(87)90135-8. [DOI] [PubMed] [Google Scholar]
- Heisterkamp N., Jenster G., ten Hoeve J., Zovich D., Pattengale P. K., Groffen J. Acute leukaemia in bcr/abl transgenic mice. Nature. 1990 Mar 15;344(6263):251–253. doi: 10.1038/344251a0. [DOI] [PubMed] [Google Scholar]
- Hermans A., Heisterkamp N., von Linden M., van Baal S., Meijer D., van der Plas D., Wiedemann L. M., Groffen J., Bootsma D., Grosveld G. Unique fusion of bcr and c-abl genes in Philadelphia chromosome positive acute lymphoblastic leukemia. Cell. 1987 Oct 9;51(1):33–40. doi: 10.1016/0092-8674(87)90007-9. [DOI] [PubMed] [Google Scholar]
- Keller G., Wagner E. F. Expression of v-src induces a myeloproliferative disease in bone-marrow-reconstituted mice. Genes Dev. 1989 Jun;3(6):827–837. doi: 10.1101/gad.3.6.827. [DOI] [PubMed] [Google Scholar]
- Kelliher M. A., McLaughlin J., Witte O. N., Rosenberg N. Induction of a chronic myelogenous leukemia-like syndrome in mice with v-abl and BCR/ABL. Proc Natl Acad Sci U S A. 1990 Sep;87(17):6649–6653. doi: 10.1073/pnas.87.17.6649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Konopka J. B., Clark S., McLaughlin J., Nitta M., Kato Y., Strife A., Clarkson B., Witte O. N. Variable expression of the translocated c-abl oncogene in Philadelphia-chromosome-positive B-lymphoid cell lines from chronic myelogenous leukemia patients. Proc Natl Acad Sci U S A. 1986 Jun;83(11):4049–4052. doi: 10.1073/pnas.83.11.4049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Konopka J. B., Watanabe S. M., Singer J. W., Collins S. J., Witte O. N. Cell lines and clinical isolates derived from Ph1-positive chronic myelogenous leukemia patients express c-abl proteins with a common structural alteration. Proc Natl Acad Sci U S A. 1985 Mar;82(6):1810–1814. doi: 10.1073/pnas.82.6.1810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Konopka J. B., Watanabe S. M., Witte O. N. An alteration of the human c-abl protein in K562 leukemia cells unmasks associated tyrosine kinase activity. Cell. 1984 Jul;37(3):1035–1042. doi: 10.1016/0092-8674(84)90438-0. [DOI] [PubMed] [Google Scholar]
- Kurzrock R., Shtalrid M., Talpaz M., Kloetzer W. S., Gutterman J. U. Expression of c-abl in Philadelphia-positive acute myelogenous leukemia. Blood. 1987 Nov;70(5):1584–1588. [PubMed] [Google Scholar]
- Lamar E. E., Palmer E. Y-encoded, species-specific DNA in mice: evidence that the Y chromosome exists in two polymorphic forms in inbred strains. Cell. 1984 May;37(1):171–177. doi: 10.1016/0092-8674(84)90312-x. [DOI] [PubMed] [Google Scholar]
- Lugo T. G., Pendergast A. M., Muller A. J., Witte O. N. Tyrosine kinase activity and transformation potency of bcr-abl oncogene products. Science. 1990 Mar 2;247(4946):1079–1082. doi: 10.1126/science.2408149. [DOI] [PubMed] [Google Scholar]
- Maddon P. J., Littman D. R., Godfrey M., Maddon D. E., Chess L., Axel R. The isolation and nucleotide sequence of a cDNA encoding the T cell surface protein T4: a new member of the immunoglobulin gene family. Cell. 1985 Aug;42(1):93–104. doi: 10.1016/s0092-8674(85)80105-7. [DOI] [PubMed] [Google Scholar]
- Malissen M., Minard K., Mjolsness S., Kronenberg M., Goverman J., Hunkapiller T., Prystowsky M. B., Yoshikai Y., Fitch F., Mak T. W. Mouse T cell antigen receptor: structure and organization of constant and joining gene segments encoding the beta polypeptide. Cell. 1984 Jul;37(3):1101–1110. doi: 10.1016/0092-8674(84)90444-6. [DOI] [PubMed] [Google Scholar]
- Marcu K. B., Banerji J., Penncavage N. A., Lang R., Arnheim N. 5' flanking region of immunoglobulin heavy chain constant region genes displays length heterogeneity in germlines of inbred mouse strains. Cell. 1980 Nov;22(1 Pt 1):187–196. doi: 10.1016/0092-8674(80)90167-1. [DOI] [PubMed] [Google Scholar]
- McLaughlin J., Chianese E., Witte O. N. Alternative forms of the BCR-ABL oncogene have quantitatively different potencies for stimulation of immature lymphoid cells. Mol Cell Biol. 1989 May;9(5):1866–1874. doi: 10.1128/mcb.9.5.1866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McWhirter J. R., Wang J. Y. Activation of tyrosinase kinase and microfilament-binding functions of c-abl by bcr sequences in bcr/abl fusion proteins. Mol Cell Biol. 1991 Mar;11(3):1553–1565. doi: 10.1128/mcb.11.3.1553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mes-Masson A. M., McLaughlin J., Daley G. Q., Paskind M., Witte O. N. Overlapping cDNA clones define the complete coding region for the P210c-abl gene product associated with chronic myelogenous leukemia cells containing the Philadelphia chromosome. Proc Natl Acad Sci U S A. 1986 Dec;83(24):9768–9772. doi: 10.1073/pnas.83.24.9768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muller A. J., Young J. C., Pendergast A. M., Pondel M., Landau N. R., Littman D. R., Witte O. N. BCR first exon sequences specifically activate the BCR/ABL tyrosine kinase oncogene of Philadelphia chromosome-positive human leukemias. Mol Cell Biol. 1991 Apr;11(4):1785–1792. doi: 10.1128/mcb.11.4.1785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oliff A., Agranovsky O., McKinney M. D., Murty V. V., Bauchwitz R. Friend murine leukemia virus-immortalized myeloid cells are converted into tumorigenic cell lines by Abelson leukemia virus. Proc Natl Acad Sci U S A. 1985 May;82(10):3306–3310. doi: 10.1073/pnas.82.10.3306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pierce J. H., Di Fiore P. P., Aaronson S. A., Potter M., Pumphrey J., Scott A., Ihle J. N. Neoplastic transformation of mast cells by Abelson-MuLV: abrogation of IL-3 dependence by a nonautocrine mechanism. Cell. 1985 Jul;41(3):685–693. doi: 10.1016/s0092-8674(85)80049-0. [DOI] [PubMed] [Google Scholar]
- Ramakrishnan L., Rosenberg N. Novel B-cell precursors blocked at the stage of DJH recombination. Mol Cell Biol. 1988 Dec;8(12):5216–5223. doi: 10.1128/mcb.8.12.5216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ribeiro R. C., Abromowitch M., Raimondi S. C., Murphy S. B., Behm F., Williams D. L. Clinical and biologic hallmarks of the Philadelphia chromosome in childhood acute lymphoblastic leukemia. Blood. 1987 Oct;70(4):948–953. [PubMed] [Google Scholar]
- Rosenberg N., Baltimore D., Scher C. D. In vitro transformation of lymphoid cells by Abelson murine leukemia virus. Proc Natl Acad Sci U S A. 1975 May;72(5):1932–1936. doi: 10.1073/pnas.72.5.1932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saglio G., Guerrasio A., Tassinari A., Ponzetto C., Zaccaria A., Testoni P., Celso B., Rege Cambrin G., Serra A., Pegoraro L. Variability of the molecular defects corresponding to the presence of a Philadelphia chromosome in human hematologic malignancies. Blood. 1988 Oct;72(4):1203–1208. [PubMed] [Google Scholar]
- Sawyers C. L., Denny C. T., Witte O. N. Leukemia and the disruption of normal hematopoiesis. Cell. 1991 Jan 25;64(2):337–350. doi: 10.1016/0092-8674(91)90643-d. [DOI] [PubMed] [Google Scholar]
- Sawyers C. L., Timson L., Kawasaki E. S., Clark S. S., Witte O. N., Champlin R. Molecular relapse in chronic myelogenous leukemia patients after bone marrow transplantation detected by polymerase chain reaction. Proc Natl Acad Sci U S A. 1990 Jan;87(2):563–567. doi: 10.1073/pnas.87.2.563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Secker-Walker L. M., Cooke H. M., Browett P. J., Shippey C. A., Norton J. D., Coustan-Smith E., Hoffbrand A. V. Variable Philadelphia breakpoints and potential lineage restriction of bcr rearrangement in acute lymphoblastic leukemia. Blood. 1988 Aug;72(2):784–791. [PubMed] [Google Scholar]
- Selleri L., von Lindern M., Hermans A., Meijer D., Torelli G., Grosveld G. Chronic myeloid leukemia may be associated with several bcr-abl transcripts including the acute lymphoid leukemia-type 7 kb transcript. Blood. 1990 Mar 1;75(5):1146–1153. [PubMed] [Google Scholar]
- Walker L. C., Ganesan T. S., Dhut S., Gibbons B., Lister T. A., Rothbard J., Young B. D. Novel chimaeric protein expressed in Philadelphia positive acute lymphoblastic leukaemia. 1987 Oct 29-Nov 4Nature. 329(6142):851–853. doi: 10.1038/329851a0. [DOI] [PubMed] [Google Scholar]
- Waneck G. L., Keyes L., Rosenberg N. Abelson virus drives the differentiation of Harvey virus-infected erythroid cells. Cell. 1986 Jan 31;44(2):337–344. doi: 10.1016/0092-8674(86)90768-3. [DOI] [PubMed] [Google Scholar]
- de Klein A., van Kessel A. G., Grosveld G., Bartram C. R., Hagemeijer A., Bootsma D., Spurr N. K., Heisterkamp N., Groffen J., Stephenson J. R. A cellular oncogene is translocated to the Philadelphia chromosome in chronic myelocytic leukaemia. Nature. 1982 Dec 23;300(5894):765–767. doi: 10.1038/300765a0. [DOI] [PubMed] [Google Scholar]