Abstract
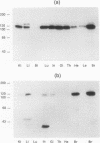
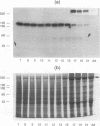
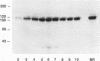
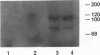
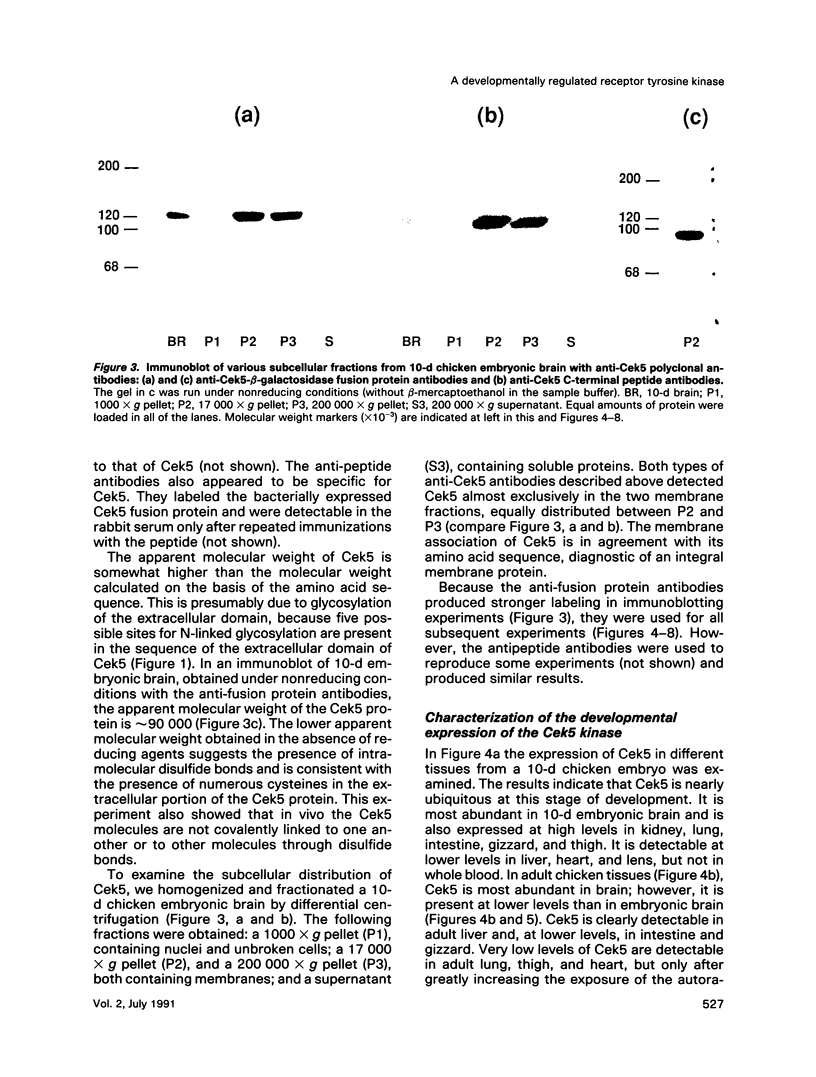
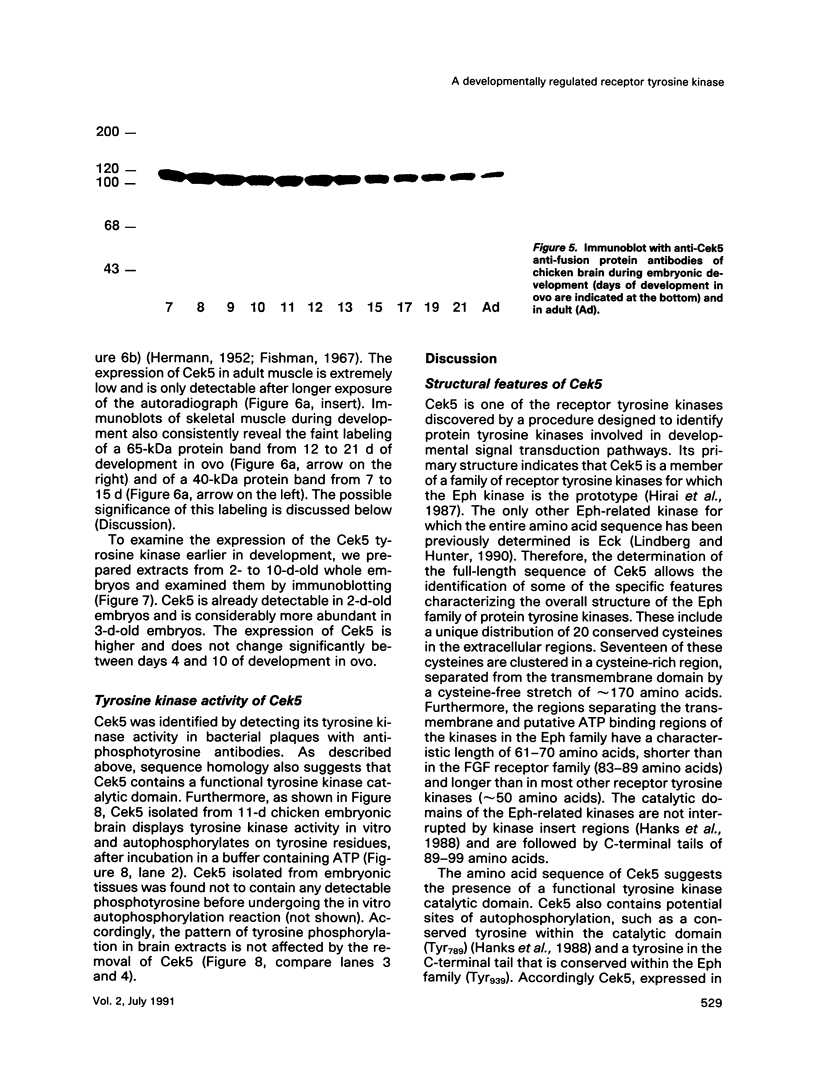
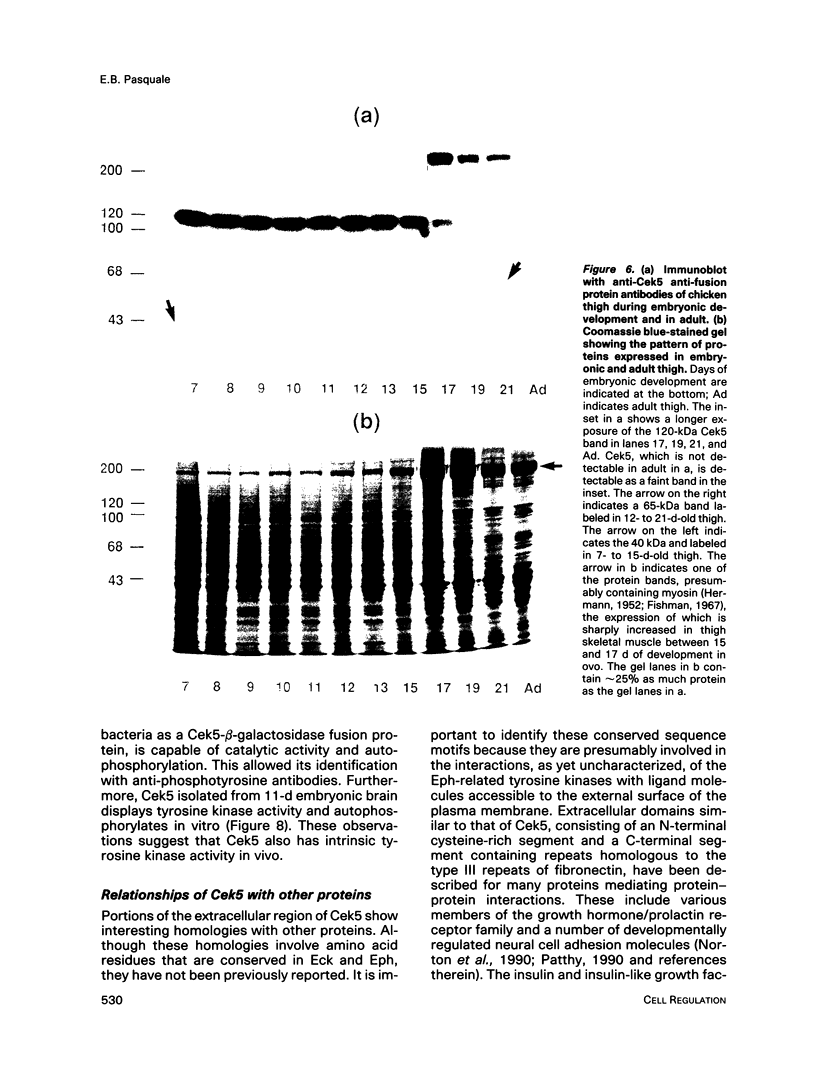
Chicken embryo kinase 5 (Cek5) is a transmembrane tyrosine kinase of the Eph family that was identified by screening a 10-d chicken embryo cDNA expression library with anti-phosphotyrosine antibodies. The extracellular region of Cek5 contains a cysteine rich N-terminal subdomain and a C-terminal subdomain mostly devoid of cysteines and comprising two repeats similar to fibronectin type III repeats. Immunoblotting experiments with anti-Cek5 polyclonal antibodies indicated that Cek5 is a membrane-associated 120-kDa protein containing intramolecular (but not intermolecular) disulfide bonds. Cek5 is already expressed in 2-d-old chicken embryos and is also expressed, at higher levels, later in development. In 10-d-old chicken embryos, Cek5 is expressed at substantial levels in nearly all the tissues examined, whereas in adult it is expressed predominantly in the brain. The expression of Cek5 in the brain gradually diminishes during embryonic development, whereas in the skeletal muscle of the thigh a sharp decrease in Cek5 expression was detected at the time of terminal muscle differentiation. Its wide tissue distribution throughout development and its sustained expression in adult brain suggest that Cek5 is an important component of signal transduction pathways, likely to interact with a widely distributed and important ligand, which is as yet unknown.
Full text
PDF








Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Burgess W. H., Maciag T. The heparin-binding (fibroblast) growth factor family of proteins. Annu Rev Biochem. 1989;58:575–606. doi: 10.1146/annurev.bi.58.070189.003043. [DOI] [PubMed] [Google Scholar]
- Feng D. F., Doolittle R. F. Progressive sequence alignment as a prerequisite to correct phylogenetic trees. J Mol Evol. 1987;25(4):351–360. doi: 10.1007/BF02603120. [DOI] [PubMed] [Google Scholar]
- Fischman D. A. An electron microscope study of myofibril formation in embryonic chick skeletal muscle. J Cell Biol. 1967 Mar;32(3):557–575. doi: 10.1083/jcb.32.3.557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Foster D. A., Levy J. B., Daley G. Q., Simon M. C., Hanafusa H. Isolation of chicken cellular DNA sequences with homology to the region of viral oncogenes that encodes the tyrosine kinase domain. Mol Cell Biol. 1986 Jan;6(1):325–331. doi: 10.1128/mcb.6.1.325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Furey W. F., Robbins A. H., Clancy L. L., Winge D. R., Wang B. C., Stout C. D. Crystal structure of Cd,Zn metallothionein. Science. 1986 Feb 14;231(4739):704–710. doi: 10.1126/science.3945804. [DOI] [PubMed] [Google Scholar]
- Garofalo R. S., Rosen O. M. Insulin and insulinlike growth factor 1 (IGF-1) receptors during central nervous system development: expression of two immunologically distinct IGF-1 receptor beta subunits. Mol Cell Biol. 1989 Jul;9(7):2806–2817. doi: 10.1128/mcb.9.7.2806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Girbau M., Bassas L., Alemany J., de Pablo F. In situ autoradiography and ligand-dependent tyrosine kinase activity reveal insulin receptors and insulin-like growth factor I receptors in prepancreatic chicken embryos. Proc Natl Acad Sci U S A. 1989 Aug;86(15):5868–5872. doi: 10.1073/pnas.86.15.5868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HERRMANN H. Studies of muscle development. Ann N Y Acad Sci. 1952 Aug 8;55(2):99–108. doi: 10.1111/j.1749-6632.1952.tb26525.x. [DOI] [PubMed] [Google Scholar]
- Hanks S. K., Quinn A. M., Hunter T. The protein kinase family: conserved features and deduced phylogeny of the catalytic domains. Science. 1988 Jul 1;241(4861):42–52. doi: 10.1126/science.3291115. [DOI] [PubMed] [Google Scholar]
- Hirai H., Maru Y., Hagiwara K., Nishida J., Takaku F. A novel putative tyrosine kinase receptor encoded by the eph gene. Science. 1987 Dec 18;238(4834):1717–1720. doi: 10.1126/science.2825356. [DOI] [PubMed] [Google Scholar]
- Kojima Y., Berger C., Vallee B. L., Kägi J. H. Amino-acid sequence of equine renal metallothionein-1B. Proc Natl Acad Sci U S A. 1976 Oct;73(10):3413–3417. doi: 10.1073/pnas.73.10.3413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kozak M. The scanning model for translation: an update. J Cell Biol. 1989 Feb;108(2):229–241. doi: 10.1083/jcb.108.2.229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lai C., Lemke G. An extended family of protein-tyrosine kinase genes differentially expressed in the vertebrate nervous system. Neuron. 1991 May;6(5):691–704. doi: 10.1016/0896-6273(91)90167-x. [DOI] [PubMed] [Google Scholar]
- Lee P. L., Johnson D. E., Cousens L. S., Fried V. A., Williams L. T. Purification and complementary DNA cloning of a receptor for basic fibroblast growth factor. Science. 1989 Jul 7;245(4913):57–60. doi: 10.1126/science.2544996. [DOI] [PubMed] [Google Scholar]
- Letwin K., Yee S. P., Pawson T. Novel protein-tyrosine kinase cDNAs related to fps/fes and eph cloned using anti-phosphotyrosine antibody. Oncogene. 1988 Dec;3(6):621–627. [PubMed] [Google Scholar]
- Lhoták V., Greer P., Letwin K., Pawson T. Characterization of elk, a brain-specific receptor tyrosine kinase. Mol Cell Biol. 1991 May;11(5):2496–2502. doi: 10.1128/mcb.11.5.2496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maher P. A., Pasquale E. B. Tyrosine phosphorylated proteins in different tissues during chick embryo development. J Cell Biol. 1988 May;106(5):1747–1755. doi: 10.1083/jcb.106.5.1747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maru Y., Hirai H., Takaku F. Overexpression confers an oncogenic potential upon the eph gene. Oncogene. 1990 Mar;5(3):445–447. [PubMed] [Google Scholar]
- Maru Y., Hirai H., Yoshida M. C., Takaku F. Evolution, expression, and chromosomal location of a novel receptor tyrosine kinase gene, eph. Mol Cell Biol. 1988 Sep;8(9):3770–3776. doi: 10.1128/mcb.8.9.3770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Musci T. J., Amaya E., Kirschner M. W. Regulation of the fibroblast growth factor receptor in early Xenopus embryos. Proc Natl Acad Sci U S A. 1990 Nov;87(21):8365–8369. doi: 10.1073/pnas.87.21.8365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Norton P. A., Hynes R. O., Rees D. J. sevenless: Seven found? Cell. 1990 Apr 6;61(1):15–16. doi: 10.1016/0092-8674(90)90209-w. [DOI] [PubMed] [Google Scholar]
- Olwin B. B., Hauschka S. D. Cell surface fibroblast growth factor and epidermal growth factor receptors are permanently lost during skeletal muscle terminal differentiation in culture. J Cell Biol. 1988 Aug;107(2):761–769. doi: 10.1083/jcb.107.2.761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olwin B. B., Hauschka S. D. Fibroblast growth factor receptor levels decrease during chick embryogenesis. J Cell Biol. 1990 Feb;110(2):503–509. doi: 10.1083/jcb.110.2.503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pasquale E. B. A distinctive family of embryonic protein-tyrosine kinase receptors. Proc Natl Acad Sci U S A. 1990 Aug;87(15):5812–5816. doi: 10.1073/pnas.87.15.5812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pasquale E. B., Singer S. J. Identification of a developmentally regulated protein-tyrosine kinase by using anti-phosphotyrosine antibodies to screen a cDNA expression library. Proc Natl Acad Sci U S A. 1989 Jul;86(14):5449–5453. doi: 10.1073/pnas.86.14.5449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patthy L. Homology of a domain of the growth hormone/prolactin receptor family with type III modules of fibronectin. Cell. 1990 Apr 6;61(1):13–14. doi: 10.1016/0092-8674(90)90208-v. [DOI] [PubMed] [Google Scholar]
- Pawson T., Bernstein A. Receptor tyrosine kinases: genetic evidence for their role in Drosophila and mouse development. Trends Genet. 1990 Nov;6(11):350–356. doi: 10.1016/0168-9525(90)90276-c. [DOI] [PubMed] [Google Scholar]
- Ruoslahti E. Fibronectin and its receptors. Annu Rev Biochem. 1988;57:375–413. doi: 10.1146/annurev.bi.57.070188.002111. [DOI] [PubMed] [Google Scholar]
- Singer S. J. The structure and insertion of integral proteins in membranes. Annu Rev Cell Biol. 1990;6:247–296. doi: 10.1146/annurev.cb.06.110190.001335. [DOI] [PubMed] [Google Scholar]
- Skorstengaard K., Jensen M. S., Sahl P., Petersen T. E., Magnusson S. Complete primary structure of bovine plasma fibronectin. Eur J Biochem. 1986 Dec 1;161(2):441–453. doi: 10.1111/j.1432-1033.1986.tb10464.x. [DOI] [PubMed] [Google Scholar]
- Ullrich A., Gray A., Tam A. W., Yang-Feng T., Tsubokawa M., Collins C., Henzel W., Le Bon T., Kathuria S., Chen E. Insulin-like growth factor I receptor primary structure: comparison with insulin receptor suggests structural determinants that define functional specificity. EMBO J. 1986 Oct;5(10):2503–2512. doi: 10.1002/j.1460-2075.1986.tb04528.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ullrich A., Schlessinger J. Signal transduction by receptors with tyrosine kinase activity. Cell. 1990 Apr 20;61(2):203–212. doi: 10.1016/0092-8674(90)90801-k. [DOI] [PubMed] [Google Scholar]
- Yarden Y., Ullrich A. Growth factor receptor tyrosine kinases. Annu Rev Biochem. 1988;57:443–478. doi: 10.1146/annurev.bi.57.070188.002303. [DOI] [PubMed] [Google Scholar]
- von Heijne G. A new method for predicting signal sequence cleavage sites. Nucleic Acids Res. 1986 Jun 11;14(11):4683–4690. doi: 10.1093/nar/14.11.4683. [DOI] [PMC free article] [PubMed] [Google Scholar]