Abstract
Elevated IOP in clinical practice is usually seen in glaucoma or ocular hypertension. Glaucoma affects 60 million people worldwide and 8.4 million are bilaterally blind from this chronic disease.1 Options for reducing IOP rely on pharmacological agents, laser treatments and surgery which may be penetrating or non-penetrating. The last twenty years has seen significant changes in all of these strategies. This review aims to cover these clinical options and introduce some of the new technologies currently in development for the clinical lowering of IOP.
Keywords: reducing IOP, pharmacological agents, laser, surgery, glaucoma, ocular hypertension
Introduction
In 2002, an international consensus panel published definitions of open-angle glaucoma (OAG) and angle-closure glaucoma (ACG) that are now widely accepted.2 In OAG the IOP may, but need not necessarily be elevated, the drainage angles are open and there are characteristic structural and functional changes manifest as optic disc damage and visual field loss.1,2 IOP lowering may take weeks or months to reach target. ACG, where the drainage angle is occluded, may present acutely with very elevated IOP requiring urgent and immediate lowering or sub-acutely where the management may be similar to OAG. The existence of OAG, ACG and secondary causes of glaucoma reinforces the need for a variety of IOP lowering treatment options with differing mechanisms of action and speed of onset. Although the aetiology of glaucoma is multifactorial the only modifiable risk factor is IOP and numerous studies have shown that reducing IOP prevents glaucoma or delays progression of established disease.3–12
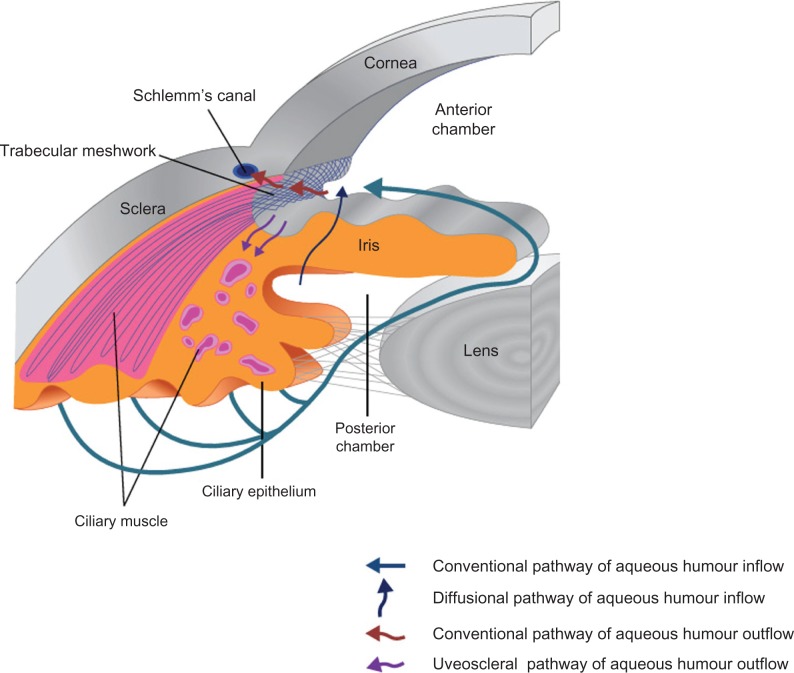
Physiology of Aqueous Humor (Fig. 1)
Figure 1.
Schematic diagram of physiology of aqueous humor.
Notes: Aqueous humor is produced by the epithelium of the ciliary body and passes from the posterior chamber through the pupil into the anterior chamber. There is also some evidence for a diffusional pathway through the iris root thus bypassing the pupil.245 The conventional outflow pathway for the aqueous humor includes trabecular meshwork, Schlemm’s canal, collector channels and episcleral venous system and 80% of flow is via this route. The uveoscleral pathway is for the remaining 20% flows directly through the iris root and interstitial spaces of the ciliary muscle into the suprachoroidal space.
IOP is determined by the rate of aqueous humor production (inflow) and exit from the eye (outflow). Aqueous humor is produced by the nonpigmented epithelium of the ciliary body and exits the eye through either the trabecular meshwork (conventional pathway) or via the uveoscleral (unconventional pathway). The conventional pathway consists of the sieve like trabecular meshwork, Schlemms’ canal and 360° collector channels for aqueous humor that lead to the episcleral venous system and 80% of flow is via this route. The remaining 20% flows directly through the iris root and interstitial spaces of the ciliary muscle into the suprachoroidal space (uveoscleral pathway). This pathway was discovered later, in the 1960s, but is an important target for newer drug treatments.13 The relative percentages change with age and presence of disease.14–17 Episcleral venous pressure has an impact on trabecular outflow so it also influences IOP. The relationship is described as follows:
where F is aqueous humor formation (μL/min), U is uveoscleral outflow (μL/min), C is trabecular meshwork outflow facility (μL/min/mmHg) and Pv is the episcleral venous pressure mmHg. Pharmacological modulation of carbonic anhydrase, adenosine triphosphatases and adrenoreceptors located in the nonpigmented ciliary epithelium can reduce aqueous production and thus lower IOP. Similarly, pharmacological modulation of adrenoreceptors and prostanoid receptors located in the trabecular meshwork or ciliary body can increase aqueous outflow through both pathways and lower IOP.
Pharmacotherapy (Table 1)
Table 1.
Pharmacological IOP lowering agents: efficacy, side effects and mechanism of action.
| Agent | Dose | Action | IOP reduction | Local side effects | Systemic side effects |
|---|---|---|---|---|---|
| Prostaglandin analogues | |||||
| Bimatoprost | Potentiate uveoscleral pathway and conventional pathway30–36 | IOP reduction of 20%–35%31,37–53 | Hyperaemia (often transient) | Few systemic side effects | |
| Lumigan® 0.01% and 0.03% | Once daily preferably nocte | Periocular skin pigmentation (reversible) | Dyspnoea | ||
| Latanoprost Xalatan® 0.005% | Unoprostone twice daily | Hypertrichosis eyelashes (Reversible) | |||
| Tafluprost Saflutan® 0.0015% | Reactivation of herpes keratitis231 | ||||
| Travoprost Travatan® 0.004% | Anterior uveitis232 | ||||
| Unoprostone Rescula® 0.12% and 0.15% | Iris pigmentation, non-reversible233,234 Cystoid macular oedema (less than previously thought)235 |
||||
| β blockers | |||||
| Beta 1 selective | Twice daily (Once daily in gel form) | Decrease aqueous production17,66 | IOP reduction 20%–27%56–60,63,66 | Topical side effects uncommon | Bronchospasm |
| Betaxolol 0.25%, 0.5% | Epithelial keratopathy | Bradycardia, contraindicated in heart block and heart failure | |||
| Betoptic® Betoptic S® Betoptima® | |||||
| Non selective | Hypotension, especially nocturnal | ||||
| Befunolol 0.5% Betaclar® | |||||
| Levobunolol 0.25%, 0.5% | |||||
| Betagan® Vistagan® | |||||
| Metipranolol 0.1%, 0.3% | |||||
| Betaman® Beta-ophtiole®, | |||||
| Optipranolol®, Turoptin® | |||||
| Timolol 0.1%, 0.25%, 0.5% | |||||
| Aquanil® Arutimol® Cusimolol® | |||||
| Nyogel® Opimol® Oftamolol® | |||||
| Timoptic® Timoptic-XE® | |||||
| Timoptol® Timabak® Timogel® | |||||
| Timolabak® Timosine XE® | |||||
| Timosan® | |||||
| With intrinsic sympathomimetic activity | |||||
| Carteolol 0.5%, 1%, 2% Carteol® Carteabak® | |||||
| Teoptic® Arteoptic® Ocupress® | |||||
| Pindolol 2% Pindoptic® | |||||
| α Agonists | |||||
| Non selective | Dipivefrin twice daily | Decrease aqueous production and increase aqueous outflow | IOP reduction 15%–25%14,236 | Pupil dilation (contraindicated in occludable angles), Hyperaemia | Headache palpitations, anxiety, hypertension |
| Dipivefrin 0.1% | Epinephrine 3× daily | Apraclonidine 25%–39%17,70,237 | |||
| Propine® Epinal® d-Epifrin® | Brimonidine 25%–28%12,14,56,238,239 | MAOI inhibitor reaction | |||
| Glaucothil® 2× daily | Cystoid macular oedema in aphakic eyes | CNS depression in children | |||
| Epinephrine 0.25% 0.5% 1.0% 2.0% | Fatigue | ||||
| 3× daily | Allergy to Brimonidine allergy (25% patients within 2 weeks)69 | ||||
| Alpha-2 selective | Apraclonidine 2–3 times daily | Decrease aqueous production | |||
| Apraclonidine 0.5% 1.0% Iopidine® 2–3× daily | Contact dermatitis Follicular conjunctivitis | ||||
| Brimonidine 0.2% Alphagan® 2× daily | Brimonidine twice daily | Brimonidine also increases uveoscleral ouflow | |||
| Clonidine 0.125% 0.25% 0.5% | Clonidine 3 times daily | ||||
| Isoglaucon® Catapres® Glaucopres® Aruclonin® | |||||
| Carbonic anhydrase inhibitors | |||||
| Topical | 3× daily as monotherapy | Decrease aqueous production | IOP reduction 17%–20%56 | Burning, stinging | Sulphonamide allergy: Anaphylaxis |
| Brinzolamide 1% Azopt® | 2× daily in combination | Acetazolamide | Steven Johnson syndrome | ||
| Dorzolamide 2% Trusopt® | Acetazolamide | 20%–30% IP reduction242,243 | Bone marrow depression, thrombotic thrombocytopenic purpura, haemolytic anaemia, leukopenia, pancytopenia and agranulocytosis | ||
| Systemic | 250 mg 4× daily | ||||
| Acetazolamide Diamox® Diamox SR® | 500 mg capsule slow release twice daily | Metallic taste | |||
| Diamox Retard® Odemin® | Metabolic acidosis | ||||
| Dichlorphenamide Antidrasi® Daranide® | Dichlorphenamide 50 mg 1–3 times daily | Hypokalaemia Paraesthesia | |||
| Glaumid® Oralcon® | Tinnitus | ||||
| Methazolamide Neptazane® | Methazolamide 50–100 mg 2–3 times daily | Renal calculi, depression | |||
| Parasympathomimetics | |||||
| Direct acting | 4× daily | Increase aqueous ouflow | IOP reduction with topical pilocarpine 20%–30%76 | Meiosis | Gastro intestinal upset |
| Pilocarpine 0.5% 1% 2% 4% | Gel formulations 1–3× daily | Direct action on longitudinal ciliary muscle | Accommodative spasm | ||
| E-Pilo® Isopto Carpine® Pilagan® Pilocar® | Acetylcholine 1:100 solution intracameral route in ocular surgery | Pseudomyopia | |||
| Pilogel® Pilomann® Pilopine® Pilopine HS gel® | Brow ache | ||||
| Pilostat® Spersacarpine® | Retinal detachment | ||||
| Aceclidine 2% | Ciliary spasm and increased pupil block | ||||
| Glaucostat®Glaunorm® | |||||
| Carbachol 0.75%–3% | |||||
| Isopto Carbachol® | |||||
| Acetylcholine 1% | |||||
| Miochol® | |||||
| Indirect acting | |||||
| Demecarium bromide 0.125% 0.25% | |||||
| Humorsol® Tosmilen® | |||||
| Ecothiophate iodide 0.03% 0.25% Echodide® |
Topical IOP treatments have been available since the 1870s with Pilocarpine introduced in 1875 and epinephrine drops in 1925. Modern day ocular hypotensives that are still in clinical use arrived in 1978 when timolol maleate was first marketed in the United States. A number of effective topical beta adrenergic blocking agents then followed. They gained widespread acceptance in the 1980s because for the first time topical medications had few visual or ocular side effects.18 For the next 25 years timolol became the gold standard to which all other topical IOP medications were compared.18 The 1990s saw the next phase of new and clinically important IOP lowering agents with the selective α2-adrenergic receptor agonist apraclonidine (Iopidine) released in 1988 and the more selective α2-agonist brimonidine tartrate (Alphagan) in 1996. Topical carbonic anhydrase inhibitors became commercially available in 1995 with dorzolamide (Trusopt) and brinzolamide (Azopt) in 1998. Prostaglandin analogues heralded a new era in topical IOP lowering therapy with improved efficacy, tolerability and a once daily dosing regimen. Table 1 demonstrates the main IOP lowering agents in current clinical practice.
Prostaglandin derivatives
Currently the most effective drugs for pressure reduction are Prostaglandin F2alpha analogues. This class now includes latanoprost, bimatoprost, travaprost, tafluprost and unoprostone.
Unoprostone (Rescula; Ciba vision) was the first commercially available prostaglandin analogue and became available in Japan in 1994 and in the United States in 2000. However, it is little used as it has limited efficacy and requires twice daily topical administration.19–25 There is some newer evidence that it may have a neuroprotective effect and interest in it has been renewed.20
Latanoprost was introduced to the US market in 1996, travoprost and bimatoprost in 2001. Latanaprost and Travaprost are ester prodrugs of PGF 2 alpha and Bimatoprost is the amide prodrug of 17-phenyl-PGF 2alpha. Bimatoprost, latanoprost and travoprost have received American (FDA) and European (EMEA) approval as first line agents in patients where IOP reduction is desired, specifically OAG and OHT. The decline in the rate of glaucoma surgeries since the 1990s has in part been attributed to these agents.26–29 They are more effective than beta blockers and have comparable tolerability on a once daily dosing regimen with less systemic side effects. The PG analogues share similar modes of action by potentiating both the trabecular meshwork route and uveoscleral pathway for aqueous drainage.30–36
Latanoprost,37,54,55 bimatoprost56–58 and travoprost56,59–61 have been shown to be equal or similar to timolol in lowering pressure in patients with open angle glaucoma or ocular hypertension and typically reduce IOP by 20%–35%.31,37–53 Pooled evidence suggests that latanoprost reduces diurnal variation in IOP significantly more than timolol by an average of 1.2 mmHg (P < 0.001).62
Beta blockers
Topical Beta blockers reduce aqueous production and have a peak effect in 2 hours. They typically reduce IOP by 20%–27% from baseline.56
Bronchospasm and bradycardia remain the main concerns with use of topical β blockers. Substantial systemic absorption can occur with all topical ocular hypotensives and one drop of 0.5% timolol may lead to a serum concentration of timolol equal to a 10 mg oral dose.64 Systemic absorption is thought to be responsible for the nocturnal hypotension seen in some patients and may be a risk factor in progression of glaucomatous optic nerve damage.65
Carbonic Anhydrase Inhibitors (CAI)
CAIs are available in topical and systemic preparations. Topical carbonic anhydrase inhibitors such as brinzolamide and dorzolamide reduce IOP by reducing aqueous production and are typically instilled 3× daily as monotherapy or twice daily when used in combination, usually with a topical beta blocker. They typically reduce IOP by 17%–20% from baseline.56 The European Glaucoma Prevention Study (EGPS)66 was a randomised multicentre double-masked placebo-controlled clinical trial evaluating the efficacy of IOP reduction by dorzolamide in preventing or delaying POAG in patients with ocular hypertension. The mean IOP reduction was 15% after 6 months and 22% after 5 years in the dorzolamide treated group, however there was also a 9% reduction in IOP at 6 months and 19% at 5 years in the placebo group. This may in part be explained by regression to the mean however it also reinforces the belief that there are other factors besides IOP which determine progression in glaucoma.
Oral CAIs such as acetazolamide are used in refractory cases of raised IOP or where topical treatment is not feasible as a temporising measure or while definitive treatment, usually laser or surgical, is being planned.
Adrenergic agonists
In the eye alpha-agonists activate alpha-1 receptors that stimulate contraction of iris dilators and Muller muscles leading to mydriasis and lid retraction. Their effect on IOP is predominantly by inducing vasoconstriction in blood vessels supplying the ciliary body thus decreasing aqueous production.67 They also stimulate post junctional alpha-2 receptors in the ciliary body and episcleral vessles causing reduced aqueous production and increased uveoscleral outflow.14,65–68
Non-selective α adrenergic agonists such as dipivefrin 0.1% and epinephrine 0.25%–2.0% are no longer used in modern ophthalmic clinical practice as they have significant side effects (Table 1) and there are better α2 selective agonists now available apraclonidine 0.5%–1.0% (Iopidine) and brimonidine (Alphagan). Both decrease aqueous production. Brimonidine also has a secondary action on uveoscleral outflow.14,65,53 The hypotensive effect lasts 12 hours with a peak effect at 2 hours hence it’s twice daily dosing.65 Allergic reactions to brimonidine can occur in up to 25% of patients and usually develops within two weeks of the beginning of treatment.66 In patients who can tolerate it, it is a useful ocular hypotensive agent but is rarely used first line and is reserved instead for use in combination, usually with timolol, or as monotherapy as a third line agent.
Apraclonidine 0.5% and 1% are equally effective in preventing IOP spikes post YAG iridotomy, 0.5% is more effective than 1% in IOP control post YAG capsulotomy and less effective for post trabeculoplasty IOP control.240 Apraclonidine can induce a dramatic fall in IOP but there has always been concern about tachyphylaxis and the high rate of follicular conjunctivitis with prolonged use.241,242
In addition to their IOP lowering effects there is some evidence that α agonists may have potential as neuroprotective agents. Animal studies have shown that brimonidine may prevent retinal ganglion cell death independent of its IOP lowering effect.71–73 Human studies suggest that there may be less retinal nerve fibre layer loss (RNFL) in patients treated with brimonidine compared to timolol over 12 months.74
Parasympathomimetics
Parasympathomimetics, also known as cholinergics, work by directly stimulating the muscarinic receptors of the ciliary muscle that widen the anterior chamber angle resulting in increased outflow through the trabecular meshwork. They also have a direct action on the longitudinal ciliary muscle.
Pilocarpine is the most commonly prescribed cholinergic. It is short acting and is usually prescribed 4 times daily in drop form or once daily in the 4% gel formulation. It is also used pre laser treatment and in trabeculectomy surgery to constrict the pupil thus stretching the iris facilitating the creation of an iridotomy or iridectomy. It has been shown to decrease uveoscleral outflow and this may be of particular significance in eyes with a compromised trabecular meshwork.75 Pilocarpine typically reduces the IOP by 20%–30%.73 Pilocarpine’s use is limited by the frequency of its application and by ocular side effects Table 1.
Osmotic agents
Osmotic agents such as mannitol 20% (1.0–1.5 g/Kg intravenously) and glycerol (1.0–1.5 g/Kg orally) are very effective in controlling acutely raised IOP. They must be used with caution as they increase blood volume and therefore increase cardiac preload. Careful assessment of renal and cardiac status is advised. They may also alter blood glucose levels.
Combination therapies
Despite advances in pharmacotherapy for raised IOP monotherapy fails to reduce IOP to a satisfactory level in 40%–75% of patients after more than 2 years of therapy.66,77 Switching within or between IOP lowering drug classes is usually attempted before adding a second agent. Adding a second drop has disadvantages of increased exposure to preservative and the risk of washing the first agent out prematurely when the second drop is instilled.
The introduction of fixed combination therapies brought hope for improved compliance and persistence with treatment as two drugs could be prescribed but in one preparation typically administered once or twice daily.
Use of β blocker preparations with either a prostaglandin analogue/prostamide, a CAI, pilocarpine or brimonidine have been shown to be more effective at lowering IOP than their use singly.75–90 As previously mentioned topical β blockers and specifically timolol were the gold standard treatment for many years but with increasing popularity of PGA as first line agents the use of both these classes in fixed combination therapy has been popular. Webers et al published a systematic review of combination therapies in 2010.91 They make the point that there are many possible combinations to assess with use of timolol 0.25% and 0.5% alongside bimatoprost, travaprost and latanoprost and using these combinations either in the morning or the evening making assessment of combination therapies not quite as straight forward as one might expect. Most of the eligible studies used timolol 0.5% and latanoprost in fixed combination. Diurnal variation is also important in fixed combination therapies with Weber et al reporting that the mean additional decrease in IOP when using latanoprost 0.005%/timolol 0.5% fixed combination in the morning was less (3.8 mmHg) than when used in the evening (6.3 mmHg).91 It is important to note that the reported additional IOP lowering of latanoprost when added to timolol is higher when used concomitantly rather than when used as a fixed combination.91 This may be partly explained by the fact that the combination therapy uses timolol 0.5% only once daily.
The CAI/β blocker fixed combination, Cosopt (trusopt and timolol 0.5%) has been shown to be equally effective at reducing IOP as its components used separately.92 and as brimonidine and timolol 0.5% used separately93 thus offering equivalent clinical effect on a simpler regime with its associated advantages.
Pharmacological treatments to lower IOP have improved dramatically. The newer topical ocular hypotensives have changed the paradigm by which clinically raised IOP is treated. The improved efficacy and tolerability of these drugs is in part responsible for the reduction in the number of glaucoma operations carried out since the 1990s.26–29
Laser
An alternative or additive treatment to the use of pharmacotherapy is laser treatment. Laser may be applied to the trabecular meshwork (trabeculoplasty) or the ciliary body (Table 2).
Table 2.
Laser treatments to lower IOP.
| Laser to the outflow pathway: Laser trabeculoplasty | Argon laser trabeculoplasty (ALT)94–99,104,105,109,112–114 |
| Green 514 nm, blue/green 488–514 nm | |
| Spot size 50 μm | |
| Exposure 0.1 s | |
| Power 500–1200 mW | |
| Placement between anterior pigmented TM and the non-pigmented TM over 180°–360° | |
| Selective laser trabeculoplasty (SLT)94,98,99,104,105,110 | |
| Nd:YAG 532 nm | |
| Spot size 400 μm | |
| Exposure 3 ns | |
| Power 0.4–1.2 mJ | |
| Placement as per ALT | |
| Diode laser trabeculoplasty (DLT)94,100–103,105 | |
| 810 nm | |
| Spot size 100 μm | |
| Exposure 0.2 s | |
| Power 800–1200 mW continuous wave | |
| 50 burns per 180° | |
| Micropulse diode laser trabeculoplasty (MDLT)94,105,133,246 | |
| 810 nm diode laser | |
| Spot size 200 μm | |
| Exposure 0.2 s | |
| Power 2 W; 15% duty cycle gives 100 laser pulses at 300 μs ON and 1700 μs | |
| OFF per 0.2 s application | |
| 70–80 applications (no visible ‘burn’) per 180° | |
| Titanium sapphire laser trabeculoplasty (TSLT)94,105,134 | |
| 790 nm | |
| Spot size 200 μm | |
| Exposure 7 μs | |
| Power 25–100 mJ | |
| Laser to the inflow pathway: Cyclophotocoagulation | Trans-scleral Nd-YAG laser (contact)143,145–149,152–155 |
| 1064 nm | |
| Applications 8–25 per 180° (avoid 3 and 9 o’clock positions to spare long posterior ciliary nerves) | |
| Placement 1–3 mm from limbus facilitated with transillumination | |
| Exposure 0.5–1 s | |
| Power 7–9 W | |
| Trans-scleral diode laser (contact)143,156–168 | |
| 810 nm | |
| Applications 10–20 per 180° (avoid 3 and 9 o’clock positions to spare long posterior ciliary nerves) | |
| Placement as per Nd:YAG aim to treat 270° | |
| Exposure 1.5–2.5 s | |
| Power 1–2 W | |
| Endoscopic cyclophotocoagulation (ECP)143,169,170 | |
| Diode 810 nm | |
| Application endoscopically viewed (via limbal or pars plana incision) to ciliary processes and intervening bridges to achieve visible whitening and shrinkage 180°–360° | |
| Exposure continuous | |
| Power 0.3–0.9 W |
Laser trabeculoplasty
Argon Laser Trabeculoplasty (ALT)
Trabeculoplasty describes multiple laser burns applied to the trabecular meshwork.94 Krasnov first reported on this technique using a ruby laser in 1972.95 Following Hager’s report of using argon laser to create what he thought was trabeculopuncture,96 Wise and Witter97 conducted a pilot study on the use of argon laser trabeculoplasty (ALT) reporting in 1979 that it lowered IOP. Subsequent studies have demonstrated ALT to be a safe and effective method of lowering IOP.
Selective Laser Trabeculoplasty (SLT)
In 1995, Latina and Park described SLT as an alternative laser modality applied to the trabecular meshwork to lower IOP.98 This is an Nd:YAG laser that selectively targets the pigmented trabecular meshwork cells and uses several thousand times less energy than a standard ALT treatment. Histological studies have shown that is induces less structural damage change in the meshwork.99
Diode Laser Trabeculoplasty (DLT)
The diode laser is a solid state laser and more compact and portable than an argon laser. It is traditionally used trans-sclerally to treat the ciliary body as will be discussed, however studies have reported its use in trabeculoplasty.100–103
Micropulse-Diode Laser Trabeculoplasty (MDLT)
MDLT is a large spot, low irradiance treatment that uses an 810 nm diode laser to emit repetitive short near infrared laser pulses to confine the laser induced thermal effect spatially.94 Theoretically each micropulse induces less thermal damage than ALT without compromising clinical effect.
Titanium-Sapphire Laser Trabeculoplasty (TSLT)
The titanium-sapphire laser has a longer wavelength than that used in ALT and SLT at 790 nm. It is thought that this near infrared wavelength may allow deeper penetration to the juxta canalicular meshwork which is thought to be the primary site of outflow resistance of aqueous humor.94
Three recent reviews have been published on this technology.94,104,105
Mechanism of action of laser trabeculoplasty
The exact mechanism by which trabeculoplasty works is unknown but there are three main theories that are not necessarily mutually exclusive.
Mechanical theory
The mechanical theory suggests that electromagnetic energy produced by the argon laser is converted to heat energy when applied to the trabecular meshwork causing a burn which contracts trabecular meshwork tissue thus stretching adjacent portions of tissue. This is then purported to reduce resistance to flow in the trabecular meshwork and Schlemm’s canal thus potentiating the conventional outflow pathway and reducing IOP.104 Histopathological evidence and tracer studies supporting this theory have been provided in monkey eyes,106,107 however controversy still exists as Van Buskirk et al108 failed to demonstrate a change in the cross sectional area of Schlemm’s canal in enucleated human eyes after ALT. SLT does not induce contraction or scarring of the trabecular meshwork and yet still lowers IOP and therefore the mechanical theory applies more to ALT than SLT.
Biologic theory
This theory is based on the evidence that thermal energy stimulates cellular activity in the trabecular meshwork increasing the number and recruitment of macrophages thus allegedly remodelling the meshwork and increasing aqueous outflow.107 Both ALT and SLT have been shown to release chemotactic and vasoactive agents such as interleukin-1 (IL-1), tumour necrosis factor (TNF-α). These molecules up regulate matrix metalloproteinase (MMP) expression triggering remodelling of the meshwork presumably decreasing resistance and potentiating outflow.107 As SLT causes very little mechanical damage to the trabecular meshwork tissue,99 unlike ALT, the favoured theory for SLT’s mode of action is biological rather than mechanical.
Repopulation theory
Bylsma et al111 suggested that laser energy stimulates increased cell division and trabecular meshwork repopulation. They demonstrated an increase in DNA replication 2 days after ALT treatment with increased cell division initially in the non-filtering portion of the meshwork migrating out to the trabecular beams where filtration does occur.
It appears that the trabecular meshwork has a multitude of responses to injury that are not necessarily stimulus specific.
Effectiveness of ALT
Evidence suggests laser trabeculoplasty provides clinically significant IOP reduction in more than 75% of initial treatments of previously unoperated eyes.11,112–118 When reviewing the literature, besides differences in methodology, follow up times and use of adjunctive treatments one must remember that many of the studies were conducted in the time when PG analogues were not available and thus the medical treatment arms of these studies usually rely on topical β blocker therapy. The Glaucoma Laser Trial compared the IOP lowering effects of 360 degree ALT vs. Timolol 0.5% as primary therapy for patients with open angle glaucoma.113 The major findings were that ALT effectively lowered the IOP by 9 mmHg vs. 7 mmHg in the Timolol group. After 2 years 44% of the ALT group did not require additional interventions for glaucoma compared with 30% in the Timolol only group. After 7 years of follow up, the ALT treated eyes continued to demonstrate lower IOP and less subjective visual field loss progression compared with the eyes receiving timolol leading the authors to conclude that ALT was at least as effective as timolol in this scenario.
Effectiveness of SLT
Several studies have verified the IOP lowering effect of SLT119–131 the magnitude of which has been reported as 11%–40% from baseline. A study by McIlraith et al132 compared SLT to latanoprost and demonstrated an average IOP reduction of 8.3 mmHg in the SLT group vs. 7.7 mmHg in the latanoprost group. Overall, SLT’s effectiveness is thought to be comparable to a single potent topical anti-glaucoma medication with the added benefit of easier compliance.104,126
The Cochrane systematic review of laser trabeculoplasty concluded that there was some evidence demonstrating similar effects in IOP control for diode and SLT compared with ALT at 6 months and one year follow up.105 Detry-Morel et al133 reported 3 months data on MDLT vs. ALT and found that the mean IOP decrease was 2.5 ± 2.6 mmHg in the MDLT group and 4.9 ± 3.4 mmHg in the ALT group (P = 0.04) Goldenfeld et al134 reported mean follow up data at 15 months on titanium-sapphire laser trabeculoplasty vs. ALT and found similar reduction in IOP in both groups. The literature supports similar complication rates across all modalities of laser with transient IOP rise the most common.101,113 Iritis, corneal burns and reflux bleeding from the meshwork have also been noted.94
Repeat treatments
Repeat ALT beyond 360 degrees is generally not recommended as it has relatively low efficacy but has increased risk of IOP spikes.135–139 Evidence also suggests that eyes that receive repeat ALT within 12 months of the initial treatment will require further intervention more quickly than eyes with an initial success of more than 12 months.135 Conversely, there is some evidence for repeat SLT therapy with Shah et al140 reporting 70% success rate of repeat SLT at one year and 53% at 2 years.
A major advantage of laser is that it does not rely on adherence and persistence with topical medical treatment longterm and studies suggest that laser trabeculoplasty is less costly when compared with medical and filtering-surgery groups.141,142
Laser ciliary body ablation
Cyclodestructive procedures are traditionally reserved for cases of raised IOP that are refractory to pharmacotherapy and outflow surgery and in eyes that have little or no visual potential. Cyclodestruction has been achieved by various methods including surgical excision, diathermy, ultrasound, cryotherapy and laser.143 Laser to the ciliary body may be employed via a contact or non-contact approach through the sclera using the Nd:YAG laser(Surgical Laser Technologies Inc., Malvern, PA) or the more portable semi-conductor diode laser (IRIS Oculight SLx, Iris Medical Inc., Mountain View, CA). An endoscopic approach where the ciliary processes are lasered under direct vision (ECP) has gained in popularity particularly in eyes where there is good visual potential.143
Trans-scleral Cyclophotocoagulation (TCP)
TCP administered with a Diode or Nd:YAG laser is usually carried out with a contact approach under regional anaesthesia (peri bulbar or retro bulbar technique) as the procedure is painful. Both diode and Nd:YAG offer comparable IOP lowering and safety profiles but the diode laser is more portable. Both contact and non-contact TCP have been shown to be effective therapies for reducing IOP in which medications and/or other surgeries have failed with success rates of 34%–94% at mean follow up 5.85 years.144–168 Traditionally, concerns over side effects such as pain, hyphaema, uveitis, cataract progression, hypotony, phthisis and severe visual loss have meant that TCP has not been used earlier in the treatment schedule of raised IOP. Reported visual loss and adverse events are partially due to the underlying disease and not just due to laser cyclodestruction. The Diode Laser Ciliary Ablation Study Group157 reported vision loss of 2 or more lines in 30% of treated eyes. In more recent studies where TCP has been used as primary surgery the rates of serious complications are less and may be due to lower energy settings and less severe forms of glaucoma than in previous studies.163–168
Endoscopic Cyclophotocoagulation (ECP)
ECP causes localised shrinkage of the ciliary processes and this can be visualised as the treatment is applied. This is thought to cause an initial reduction in blood flow and aqueous producing capacity in the ciliary body with partial reperfusion at 1 month hence the lower associated rates of hypotony and phthisis.143 ECP can be undertaken in conjunction with cataract surgery or in pseudophakic or aphakic eyes. As it is an intraocular procedure there is the associated risk of endophthalmitis not applicable to the trans-scleral approach. The largest series on ECP by Chen et al 1997169 reports a mean IOP reduction of 34% from baseline at 12.9 months. Glaucoma medication usage was reduced from an average of 3 agents preoperatively to 2 postoperatively indicating that ECP is frequently an adjunctive treatment to medical therapy. As there is relative sparing of the adjacent tissues when compared to TCP, namely the iris root, pars plicata, pars plana and sclera there is a relatively lower incidence of vision threatening complications. Complications reported in the largest series included fibrin exudate in 24%, hyphaema 12%, cystoid macular oedema 10% and vision loss of 2 lines or greater in 6%.169,170
Surgery
When pharmacological and laser treatments have been unsuccessful in reducing IOP adequately surgery is usually indicated.
Surgery for raised IOP is divided into penetrating and non-penetrating groups. Surgical techniques have evolved over the last 40 years from the traditionally less predictable full thickness procedures to safer guarded antiproliferative enhanced trabeculectomy, implantation of glaucoma drainage devices and a variety of non-penetrating techniques such as viscocanalostomy and deep sclerectomy. For lowering IOP and preventing visual field loss, a recent Cochrane review171 comparing the effectiveness of surgery vs. medical IOP lowering treatments found there was evidence of slightly better IOP control with surgery though no current evidence to support any difference in visual field progression.
Trabeculectomy
Trabeculectomy is the still the most widely used surgical procedure for glaucoma and is more effective in lowering IOP than medical or laser treatment.114 A trabeculectomy creates a fistula between the anterior chamber of the eye and the subconjunctival space to allow controlled release of aqueous. The conjunctiva is reflected in either a fornix based or limbus based approach and a full thickness sclerostomy is made under a pre-fashioned partial thickness scleral flap. The conjunctiva is then resutured above the scleral flap. Aqueous flows through the sclerostomy and under the scleral flap. This fluid forms a conjunctival bleb and is thought to be carried away by vessels within the conjunctiva and also by evaporating across the conjunctival surface. The final IOP lowering effect is determined by the size of the bleb, downstream drainage from the conjunctival vessels and the scarring response around the bleb. Since its original description by Cairns in 1968172 advances in operating microscopes, instruments, suture material and the introduction of antimetabolites have improved the success rates of trabeculectomy alongside a marked reduction in complications.5,171,173,174
The Collaborative Initial Glaucoma Treatment Study (CIGTS)5 was a randomised controlled trial involving 607 patients with newly diagnosed open-angle glaucoma randomised to medication or trabeculectomy with or without 5-fluorouracil (5FU). The IOP was lower in the surgical group (mean 14–15 mmHg) than in the medical treatment group (17–18 mmHg) however as is common with most penetrating surgery the rate of cataract progression and incidence of cataract surgery was increased (6% medication group vs. 17% trabeculectomy group). In the hands of experienced surgeons the success rate of filtering surgery (alone, or with adjunctive medical therapy) in a previously un operated eye is reported to be up to 90% at 2 years, however as with any meta-analysis there are large differences in the criteria used to define success.77,171,175 Optimum success rates are achieved when the eye has not been exposed to previous interventions either surgical or medical.176 Risk factors for failure of trabeculectomy to control IOP include previous exposure to topical medication (especially sympathomimetics), previous surgical manipulation of the conjunctiva or other injury. Age is inversely related to risk.177 The success rate is lower in black Africans, people with previous ocular inflammation or vascularisation and children.174 The commonest cause for failure of trabeculectomy is subconjunctival fibrosis178 resulting in occlusion of the fistula with an encapsulated bleb, commonly known as the ‘ring of steel’. Various agents have been used to minimise scarring with mitomycin C (MMC) the most commonly used intra-operative agent. 5FU is commonly used to influence bleb morphology post operatively. Beta irradiation has also been used as an anti-scarring agent.174 It may be particularly useful in developing countries due to the technical simplicity of application of the probe and lower cost.
MMC was first introduced in 1983 although its widespread use did not come until the 1990’s. MMC’s alkylating properties inhibit DNA replication and inhibit or kill fibroblasts in in-vitro and in-vivo models of trabeculectomy thus increasing the success rates of filtration surgery.175,179–181 MMC is typically administered intraoperatively in concentrations of 0.1–0.5 mg/mL for 1–5 minutes depending on personal surgical preference. The main concerns about MMC use revolve around the thinning of the conjunctiva overlying the guarded sclerostomy which may cause late bleb leaks, hypotony, bleb related infection and endophthalmitis. The Cochrane review of MMC use in filtering surgery181 found the risk of cataract to be the only side effect significantly increased and failed to confirm many ophthalmologists’ anecdotal view of increased complications of hypotony and endophthalmitis. This may, in part, be due to low event rates and increasing success in preventing these complications being mindful of the potential risks inducing ever more meticulous care and attention when handling the conjunctiva and sclera.
Aqueous shunts
Aqueous shunts, also known as glaucoma drainage devices, tube implants and setons are alternatives to trabeculectomy surgery. Aqueous shunts have evolved over more than one hundred years and 4 main shunts are used in clinical practice today; Ahmed (New World Medical Inc., Rancho Cucamonga, CA), Baerveldt (Advanced Medical Optics, Inc Santa Ana, CA 250 mm2 and 3500 mm2 devices), Krupin (Eagle Vision Inc., Memphis TN) and Molteno (Molteno Ophthalmic Ltd., Dunedin, New Zealand, single and double plated devices).182,190
They follow the same biological principles using an explant plate sutured to the sclera, typically in the superotemporal part of the globe that when encapsulated provides a potential space into which aqueous can drain via a connecting silicone-rubber tube which is placed into the anterior chamber taking care not to abut the corneal endothelium or phakic lens. The devices differ in explant surface areas, shape, plate thickness, presence or absence of a valve and the absolute details of surgical implantation.182 Aqueous flows from the eye through the tube to the encapsulated plate and then diffuses into the surrounding tissues by passive diffusion.183
These shunts were traditionally thought not to be as effective as standard and augmented trabeculectomy182 with typical IOPs of 18 mmHg vs. 14–16 mmHg and 8–10 mmHg respectively, however the recent Tube vs. Trabeculectomy study184 challenges this with a mean IOP of 13 mmHg and IOP of 14 mmHg or less in 62% of patients at 3 years. They used a 350-mm2 Baerveldt shunt. It also reported fewer complications in the tube group compared with the MMC trabeculectomy group (39% vs. 60% respectively during the first 3 years of follow up P = 0.004) but more adjunctive medications at 1 year in the tube group however this had ceased to be statistically significant at 3 years. Cataract progression was similar in both groups at 3 years (58% Tube group vs. 57% Trabeculectomy group) Recently published expert opinion make the point that the complication rates of augmented trabeculectomy in the study were higher than other published data and the debate regarding superiority is ongoing.244 Studies published before 2005 were included in the Cochrane review of aqueous shunts.185 Comparative outcomes of current shunts remain insufficient to conclude that one device is more effective than another, but several studies indicate that devices with larger equatorial plates control IOP better than smaller plates.185,186 There is no definitive evidence to support the use of antifibrotic agents as adjuncts to aqueous shunt surgery.187,188
Aqueous shunts tend to be reserved for complicated cases of raised IOP, after pharmacotherapy, laser and filtering surgery has failed and in eyes with traumatic, inflammatory and neovascular glaucoma.182,185 Medicare data between 1995 and 2004189 suggests a trend towards earlier and increased usage of aqueous shunts. This may in part be explained by increasing surgical familiarity and confidence in these devices to deliver lower IOP with fewer side effects.
The principal concept of non-penetrating glaucoma surgery (NPGS) is to create filtration through a naturally occurring membrane that acts as an outflow resistance site, allowing a progressive IOP drop and avoiding postoperative ocular hypotony.191 The rate of aqueous drainage is controlled at the level of the trabecular meshwork and Descemet’s membrane rather than a guarded sclerostomy. The proposed advantage of NPGS is that uncomplicated surgery is virtually an extraocular procedure192 and some believe it to be safer than trabeculectomy with quicker visual recovery, less intraocular inflammation and is astigmatically neutral.193–195 The basic premise underlying NPGS is that the greatest resistance to aqueous flow is at the juxtacanalicular trabecular meshwork and Schlemm’s canal. The first published report was in 1964 when Krasnov196,197 described a procedure, (sinusostomy) which involved removing a lamellar band of the sclera thus opening up Schlemm’s canal over 120 degrees. Various modifications then ensued until Fydorov stressed the importance of removing the corneal stroma behind the anterior trabeculum and Descemet’s membrane calling it deep sclerectomy.198
The two main NPGS procedures in current clinical use are Deep Sclerectomy (DS) and Viscocanalostomy.
Deep sclerectomy
DS involves making a 5 × 5 mm superficial scleral flap of approximately one third scleral thickness. A second scleral flap 4 × 4 mm beneath the original is then fashioned and removed to leave a residual scleral bed of 50–100 microns. Schlemm’s canal is de-roofed in the most challenging part of the surgery due to the risk of inadvertent perforation of the anterior chamber or removing insufficient tissue in efforts to avoid perforation. Some surgeons then peel the Schlemm’s canal endothelium and juxtacanalicular trabeculum membrane and this additional step corresponds to abexterno trabeculectomy.198 The superficial scleral flap is then repositioned and sutured with or without an implant to help keep the scleral space open. After aqueous passage through the residual trabeculo-Descemet’s membrane there are four hypothetical mechanisms for resorption; a subconjunctival filtering bleb, an intrascleral filtering bleb, a suprachoroidal filtering bleb and an episcleral vein outflow via Schlemm’s canal.198–200
Efficacy of deep sclerectomy
Clinical IOP lowering with deep sclerectomy without the adjunctive use of antimetabolite or implant has been reported as a mean drop of 50% at 46 months in a retrospective study on eyes newly diagnosed with POAG and no prior treatment to success rates of 61.4%, 36.6% and 18.9% at 21, 24 and 30 months respectively in a prospective study of 43 eyes where surgical success was deemed to be IOP of less than 22 mmHg and 20% reduction from baseline without the use of medication.201
Recent evidence suggests that success rates are improved with antimetabolites and implants.192,198 Using an implant is thought to help maintan a low pressure intrascleral lake between the external scleral flap and trabecular meshwork acting as a spacer in the time of maximal healing and hence scar formation. In one RCT deep sclerectomy with implant (DSCI) achieved IOP of <21 mmHg without adjunctive medical treatment in 69.2% at 48 months vs. deep sclerectomy without implant (DS) 38.5%. The IOP at 48 months was 10 (±4 mmHg DSCI) compared with 16 ± 3.0 mmHg (DS) (P = 0.005).201 Various authors report prospective and retrospective case series demonstrating enhanced success with the use of implants.198,202–204 however there are some comparative studies where implants have not improved success rates.192,205,206 Some discrepancy may be due to the confounding influence of using 5FU in the control group but not the DSCI group. Several commercially available implants have been developed (Aqua-flow collagen drainage device, T flux, SKGEL and PMMA implant) Interestingly, Ultrasound Biomicroscopy (UBM) performed 1 year after deep sclerectomy without implant demonstrates the presence of an intrascleral space in over 90% of cases207 and the presence and volume of the intrascleral space correlates poorly with level of IOP.
Antiproliferatives and deep sclerectomy
Both 5FU and MMC have been used to try to augment the success rate of DS and both appear to increase success rates.205,206,208 An RCT of DS with and without MMC (0.2 mg/mL for 2.5 minutes) demonstrated a greater IOP reduction in the MMC group at 36 months (11.7 mmHg or 42.3% vs. 7.1 mmHg or 27.6% P = 0.05).209 Neudorfer et al also reported an increased success rate210,211 however, Anand et al212 report a RCT studying the effect of DS with low dose MMC in a Nigerian population and MMC did not deliver better IOP lowering and success rates were low in both groups (13% MMC DS vs. 24% DS at 18 months P = 0.5).
Comparing DS with trabeculectomy213–217 is important as many specialist surgeons use one or other technique predominantly. Randomised controlled trials comparing NPGS to trabeculectomy213,218–222 suggest an improved safety profile of NPSG but are not in agreement when it comes to efficacy even in RCTs on paired eyes. El Sayyad et al213 randomly assigned 39 patients to DS or trabeculectomy in the first eye followed by the opposite procedure in the fellow eye reporting mean postoperative IOP of 15.6 mmHg ± 4.2 DS group vs. 14.1 mmHg ± 4.6 in the trabeculectomy group; an IOP reduction of 44% and 50% respectively from baseline. Chiselita214 in a similar study of 34 eyes in 17 patients found a statistically significant lower IOP in the trabeculectomy group throughout the 18 month follow up period (17.3 ± 1.2 mmHg vs. 20.9 ± 4.0 mmHg DS group) but with significantly fewer complications in the DS group. This reduced complication rate was echoed by Ambresin et al216 who also reported similar success rates between DS and trabeculectomy. Cilino et al215 found no significant difference in outcome between DS and trabeculectomy although the trend was towards lower IOP in the trabeculectomy group.
Viscocanalostomy
Stegmann et al described a variant of NPGS and termed it viscocanalostomy to emphasise the importance of injecting high viscosity sodium hyaluronate (Healon GV) into Schlemm’s canal as a means of improving aqueous drainage.223 In viscocanalostomy the superficial scleral flap is tightly sutured so that only aqueous regressing through trabeculo-Descemet’s membrane can reach the two surgically created ostia of Schlemm’s canal, travel circumferentially within the canal and enter the collector channel s and then the aqueous veins. In a post mortem eye model, injecting viscoelastic into Schlemm’s canal enlarged its area adjacent to and 6 mm from the flap significantly more than if it was not injected.224 In vivo primate225 and human eye223 studies show that not only is Schlemm’s canal dilated but so too are the collector channels and there is disruption of the inner wall endothelium of the canal with disorganisation of the juxtacanalicular zone resulting in direct communication of the juxtacanalicular space with the lumen of Schlemm’s canal. This may enhance conventional aqueous outflow.223 Disruption of the posterior wall of Schlemm’s canal may also allow direct communication between the lumen and the ciliary body thus potentiating uveoscleral outflow. The viscoelastic material is also postulated to have an anti-inflammatory effect and may inhibit cellular migration, phagocytosis and cytokine production thus interfering with wound healing.198
Stegmann reported the results of viscocanalastomy in 214 eyes from 157 black African patients with average follow up time of 35 months.223 82.7% of eyes achieved an IOP of 22 mmHg or less without medical therapy and an average IOP reduction of 64% with few complications. In the mainly caucasian series of Sunaric-Megevand and Leuenberger the mean IOP reduction was 38.3% with complete success in 68% at 1 year, 60% at 2 years and 59% at 3 years.226 Yarangameli et al227 compared a prospective group undergoing viscocanalostomy with MMC with a retrospective viscocanalostomy alone group and reported no statistically significant difference in IOP although they observed a trend towards more IOP reduction and higher success rate in the MMC group. A prospective RCT of viscocanalostomy with and without the SKGEL implant found 40% success rate at 1 year in both groups (IOP < 22 mmHg and no medication) with equally low complication rates.228
Viscocanalostomy compared with trabeculectomy
O’Brart et al conducted 2 RCTs one comparing MMC trabeculectomy with standard viscocanalostomy and the other comparing augmented trabeculectomy with augmented viscocanalostomy.220,221 In the RCT of 50 eyes from 48 POAG patients randomised to MMC augmented trabeculectomy or standard viscocanalostomy 100% of eyes in the trabeculectomy group achieved IOP < 21 mmHg at one year compared with 64% in the viscocanalostomy group (P < 0.001) The trabeculectomy group however had significantly more postoperative interventions (64% vs. 23% in the viscocanalostomy group) When both procedures were augmented success rates were 68% in the MMC trabeculectomy group vs. 34% in the MMC viscocanalostomy group after mean follow up 1 year. Most of the evidence suggests that augmented viscocanalostomy tends not to achieve the same IOP reduction as MMC augmented trabeculectomy but does result in fewer complications.218–222,227,228 However, Mendrinos et al in their review on NPGS make the point that there is a significant learning curve to NPGS and one cannot compare one’s first few cases of NPGS to their last few cases of trabeculectomy.198
There are some studies using laser assisted ablation of deep scleral tissues and this step is generally felt to be the most difficult to do successfully. An erbium-YAG laser has a wavelength near the maximum absorption of water hence the ablation depth is self-limited by the percolation of aqueous and may reduce the risk of inadvertent anterior chamber perforation.229 Excimer and CO2 lasers have also been tested experimentally and may be viable.198
NPGS continues to evolve and IOP lowering appears to have improved with modifications in technique just as trabeculectomy and aqueous shunt surgery have however the degree and longevity of IOP lowering between augmented trabeculectomy and NPGS are still uncertain.
Novel Glaucoma Procedures
The American Academy of Ophthalmology recently issued a report summarising the exciting new developments in surgical procedures for IOP lowering.230 These have FDA approval or are currently in phase III clinical trials in the USA. The majority of novel procedures seek to avoid bleb formation and its associated disadvantages and instead rely on augmentation of the physiological outflow pathways. There are three main categories of new devices; (1) increasing outflow by creating a new outflow channel) Ex-PRESS glaucoma implant (Alcon, Inc., Hunenberg, Switzerland)247–251 and Fugo blade transciliary filtration (Medisurg Ltd., Norristown, PA);252,253 (2) augment the conventional (trabecular meshwork) pathway, trabecular by passstent (iStent, Glaukos Corporation, Laguna Hills, CA),254–256 trabeculectomy by internal approach with the Trabectome (NeoMedix, Inc., Tustin, CA)257–259 Fugo blade goniotomy,260 excimer laser trabeculoplasty (AIDA, Glautec AG Nurnberg, Germany)261,262 and canaloplasty (iScience Interventional Corp., Menlo Park, CA)263–265 and (3) enhancing uveoscleral flow, the SOLX Gold Shunt (SOLX Ltd., Boston, MA).266–268
Conclusions
The ideal scenario in clinical IOP lowering would be a cost effective procedure or pharmacological agent which lowered IOP significantly and predictably with minimum side effects or complications and minimal reliance on adherence and persistence with treatment regimes. This review comments on current clinical options for IOP lowering and there is no doubt that refinement and modifications of existing medical and surgical options have provided better IOP lowering for patients but continued innovation and pursuit of the perfect ocular hypotensive are to be encouraged.
Footnotes
Author Contributions
Wrote the first draft of the manuscript: LC. Contributed to the writing of the manuscript: SZ, MC, LG. Agree with manuscript results and conclusions: MC, LG. Jointly developed the structure and arguments for the paper: LC, MC, LG. Made critical revisions and approved final version: MC, LG. All authors reviewed and approved of the final manuscript.
Disclosures and Ethics
As a requirement of publication author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest. Provenance: the authors were invited to submit this paper.
References
- 1.Quigley HA. Glaucoma. Lancet. 2011;377:1367–77. doi: 10.1016/S0140-6736(10)61423-7. [DOI] [PubMed] [Google Scholar]
- 2.Foster PJ, Buhrmann R, Quigley HA, Johnson GJ. The definition and classification of glaucoma in prevalence surveys. Br J Ophthalmol. 2002;86:238–42. doi: 10.1136/bjo.86.2.238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Freidman DS, Wilson MR, Liebmann JM, Fechtner RD, Weinreb RN. An evidence based assessment of risk factors for the progression of ocular hypertension and glaucoma. Am J Ophthalmol. 2004;138(Suppl 3):S19–31. doi: 10.1016/j.ajo.2004.04.058. [DOI] [PubMed] [Google Scholar]
- 4.AGIS Investigators The advanced glaucoma intervention study (AGIS) (12. Baseline risk factors for sustained loss of visual field and visual acuity in patients with advanced glaucoma) Am J Ophthalmol. 2002;134:499–512. doi: 10.1016/s0002-9394(02)01659-8. [DOI] [PubMed] [Google Scholar]
- 5.Lichter PR, Musch DC, Gillespie BW, Guire KE, Janz NK, Wren PA, et al. CIGTS Study Group Interim clinical outcomes in the Collaborative Initial Glaucoma Treatment Study comparing initial treatment randomised to medications or surgery. Ophthalmology. 2001;108:1943–53. doi: 10.1016/s0161-6420(01)00873-9. [DOI] [PubMed] [Google Scholar]
- 6.Leske MC, Heijl A, Hussein M, Early Manifest Glaucoma Trial Group Factors for glaucoma progression and the effect of treatment: the Early Manifest Glaucoma Trial. Arch Ophthalmol. 2003;121:48–56. doi: 10.1001/archopht.121.1.48. [DOI] [PubMed] [Google Scholar]
- 7.Collaborative Normal Tension Glaucoma Study Group Comparison of glaucomatous progression between untreated patients with normal tension glaucoma and patients with therapeutically reduced intraocular pressures. Am J Ophthalmol. 1998;126:487–97. doi: 10.1016/s0002-9394(98)00223-2. [DOI] [PubMed] [Google Scholar]
- 8.Heijl A, Leske MC, Hyman L, Yang Z, Bengtsson B, for the EMGT Group Intraocular pressure reduction with a fixed treatment protocol in the early manifest glaucoma trial. Ophthalmology. 2009;116(12):2271–6. doi: 10.1111/j.1755-3768.2009.01852.x. [DOI] [PubMed] [Google Scholar]
- 9.Kass MA, Heuer DK, Higginbotham EJ, et al. The Ocular Hypertension Treatment Study: a randomised trial determines that topical ocular hypotensive medication delays or prevents the onset of primary open angle glaucoma. Arch Ophthalmol. 2002;120:701–13. doi: 10.1001/archopht.120.6.701. [DOI] [PubMed] [Google Scholar]
- 10.Kass MA, Gordon MO, Gao F, et al. Ocular Hypertension Treatment Study Group Delaying treatment of ocular hypertension: the ocular hypertension treatment study. Arch Ophthalmol. 2010;128:276–87. doi: 10.1001/archophthalmol.2010.20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hoyng PF, Kitazawa Y. Medical treatment of normal tension glaucoma. Ophthalmology. 2002;47(Suppl 1):S116–24. doi: 10.1016/s0039-6257(02)00322-3. [DOI] [PubMed] [Google Scholar]
- 12.Arthur S, Cantor LB. Update on the role of alpha-agonists in glaucoma management. Experimental Eye Research. 2011 doi: 10.1016/jexer.2011.04.002. [DOI] [PubMed] [Google Scholar]
- 13.Weinreb RN. Uveoscleral outflow: the other outflow pathway. J Glaucoma. 2000 Oct;9(5):343–5. doi: 10.1097/00061198-200010000-00001. [DOI] [PubMed] [Google Scholar]
- 14.Becker B. The decline in aqueous secretion and outflow facility with age. Am J Ophthalmol. 1958 Nov;46(5):731–6. doi: 10.1016/0002-9394(58)91272-8. [DOI] [PubMed] [Google Scholar]
- 15.Bill A. Blood circulation and fluid dynamics of the eye. Physiol Rev. 1975 Jul;55(3):383–417. doi: 10.1152/physrev.1975.55.3.383. [DOI] [PubMed] [Google Scholar]
- 16.Toris CB, Pederson JE. Aqueous humor dynamics in experimental iridocyclitis. Invest Ophthalmol Vis Sci. 1987 Mar;28(3):477–81. [PubMed] [Google Scholar]
- 17.Marquis RE, Whitson JT. Management of glaucoma: Focus on pharmacological therapy. Drugs Aging. 2005;22(1):1–21. doi: 10.2165/00002512-200522010-00001. [DOI] [PubMed] [Google Scholar]
- 18.Bean GW, Camras CB. Commercially available prostaglandin analogs for the reduction of intraocular pressure: Similarities and differences. Surv Ophthalmol. 2008;53(Suppl 1):S69–84. doi: 10.1016/j.survophthal.2008.08.012. [DOI] [PubMed] [Google Scholar]
- 19.Aung T, Chew PT, Yip CC, et al. A randomised double masked crossover study comparing latanoprost 0.005% with unoprostone 0.12% in patients with primary open angle glaucoma and ocular hypertension. Am J Ophthalmol. 2001;131:636–42. doi: 10.1016/s0002-9394(00)00943-0. [DOI] [PubMed] [Google Scholar]
- 20.Yamagishi R, Aihara M, Araie M. Neuroprotective effects of prostaglandin analogues on retinal ganglion cell death independent of intraocular pressure reduction. Exp Eye Res. 2011;93:265–70. doi: 10.1016/j.exer.2011.06.022. [DOI] [PubMed] [Google Scholar]
- 21.Jampel HD, Bacharach J, Sheu WP, et al. Randomised clinical trial of latanoprost and unoprostone in patients with elevated intraocular pressure. Am J Ophthalmol. 2002;134:863–71. doi: 10.1016/s0002-9394(02)01820-2. [DOI] [PubMed] [Google Scholar]
- 22.Nordman JP, Mertz B, Yannoulis NC, et al. A double masked randomised comparison of the efficacy and safety of unoprostone with timolol and betaxolol in patients with primary open angle glaucoma including pseudoexfoliation glaucoma or ocular hypertension. 6 months data. Am J Ophthalmol. 2002;133:1–10. doi: 10.1016/s0002-9394(01)01337-x. [DOI] [PubMed] [Google Scholar]
- 23.Susanna R, Jr, Chew P, Kitazawa Y. Current status of prostaglandin therapy: latanoprost and unoprostone. Surv Ophthalmol. 2002;47(Suppl 1):S97–104. doi: 10.1016/s0039-6257(02)00308-9. [DOI] [PubMed] [Google Scholar]
- 24.Susanna R, Jr, Giampani J, Jr, Borges AS, et al. A double masked randomised clinical trial comparing latanoprost with unoprostone in patients with open angle glaucoma or ocular hypertension. Ophthalmology. 2001;108:259–63. doi: 10.1016/s0161-6420(00)00495-4. [DOI] [PubMed] [Google Scholar]
- 25.Tsukamoto H, Yokoyama T, Okada K, et al. Substituting latanoprost (Xalatan) for isopropyl unoprostone (Rescula) in monotherapy and combination therapy. Acta Ophthalmol Scand. 2000;78:604–5. doi: 10.1034/j.1600-0420.2000.078005604.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Fraser SG, Wormwald RP. Hospital episode statistics and changing trends in glaucoma surgery. Eye. 2008;22:3–7. doi: 10.1038/sj.eye.6702458. [DOI] [PubMed] [Google Scholar]
- 27.Kenigsberg PA. Changes in medical and surgical treatments of glaucoma between 1997 and 2003 in France. Eur J Ophthalmol. 2007;17:521–7. doi: 10.1177/112067210701700407. [DOI] [PubMed] [Google Scholar]
- 28.Strutton DR, Walt JG. Trends in glaucoma surgery before and after the introduction of new topical glaucoma pharmacotherapies. J Glaucoma. 2004;13:221–6. doi: 10.1097/00061198-200406000-00008. [DOI] [PubMed] [Google Scholar]
- 29.Van der Valk R, Schouoten JS, Webers CA, et al. The impact of a nationwide introduction of new drugs and a treatment protocol for glaucoma on the number of glaucoma surgeries. J Glaucoma. 2005;14:239–42. doi: 10.1097/01.ijg.0000159121.11371.5f. [DOI] [PubMed] [Google Scholar]
- 30.Bahler CK, Howell KG, Hann CR, et al. Prostaglandins increase trabecular meshwork outflow facility in cultured human anterior segments. Am J Ophthalmol. 2008;145:114–9. doi: 10.1016/j.ajo.2007.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Brubaker RF, Schoff EO, Nau CB, et al. Effects of AGN 192024, a new ocular hypotensive agent, on aqueous dynamics. Am J Ophthalmol. 2001;131:19–24. doi: 10.1016/s0002-9394(00)00843-6. [DOI] [PubMed] [Google Scholar]
- 32.Toris CB, Camras CB, Yablonski ME, Brubaker RF. Effects of exogenous prostaglandins on aqueous humor dynamics and blood-aqueous barrier function. Surv Ophthalmol. 1997;41(Suppl 2):S69–75. doi: 10.1016/s0039-6257(97)80010-0. [DOI] [PubMed] [Google Scholar]
- 33.Toris CB, Gabelt BT, Kaufman PL. Update on the mechanism of action of topical prostaglandins for intraocular pressure reduction. Surv Ophthalmol. 2008;53(Suppl 1):S107–20. doi: 10.1016/j.survophthal.2008.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Wan Z, Woodward DF, Cornell CL, et al. Bimatoprost prostamide activity and conventional drainage. Invest Ophthalmol Vis Sci. 2007;48:4107–15. doi: 10.1167/iovs.07-0080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Weinreb RN, Toris CB, Gabelt BT, et al. Effects of prostaglandins on aqueous humor outflow pathways. Surv Ophthalmol. 2002;47(Suppl 1):S53–64. doi: 10.1016/s0039-6257(02)00306-5. [DOI] [PubMed] [Google Scholar]
- 36.Ziai N, Dolan JW, Kacere RD, Brubaker RF. The effects on aqueous dynamics of PhXA41, a new prostaglandin F2α analogue, after topical application in normal and ocular hypertensive human eyes. Arch Ophthalmol. 1993;111:1351–8. doi: 10.1001/archopht.1993.01090100059027. [DOI] [PubMed] [Google Scholar]
- 37.Watson P, Stjernschantz J. A six-month randomised double blind study comparing the effects of latanoprost and timolol on primary open angle glaucoma and ocular hypertension. The Latanoprost Study Group. Ophthalmology. 1996;103(1):126–37. doi: 10.1016/s0161-6420(96)30750-1. [DOI] [PubMed] [Google Scholar]
- 38.Waewar RE, Bullock JD, Ballal D. Cystoid macular oedema and anterior uveitis associated with latanoprost use. Ophthalmology. 1998;105:263–8. doi: 10.1016/s0161-6420(98)92977-3. [DOI] [PubMed] [Google Scholar]
- 39.Gandolfi SA, Cimino L. Effect of bimatoprost on patients with primary open angle glaucoma or ocular hypertension who are non-responders to latanoprost. Ophthalmology. 2003;110(3):609–14. doi: 10.1016/S0161-6420(02)01891-2. [DOI] [PubMed] [Google Scholar]
- 40.Hayreh SS, Podhajsky P, Zimmerman MB. Beta-blocker eyedrops and nocturnal arterial hypotension. Am J Ophthalmol. 1999;128:301–9. doi: 10.1016/s0002-9394(99)00160-9. [DOI] [PubMed] [Google Scholar]
- 41.Higginbotham BJ, Schuman JS, Goldberg I, et al. Bimatoprost Study Group 1 and 2 One-year randomised study comparing Bimatoprost and Timolol in glaucoma and ocular hypertension. Arch Ophthalmol. 2002;120:1286–9. doi: 10.1001/archopht.120.10.1286. [DOI] [PubMed] [Google Scholar]
- 42.Alm A, Camras CB, Watson PG. Phase III latanoprost studies in Scandinavia, the United Kingdom and the United States. Surv Ophthalmol. 1997;41(Suppl 2):S105–10. doi: 10.1016/s0039-6257(97)80016-1. [DOI] [PubMed] [Google Scholar]
- 43.Netland PA, Landry T, Sullivan EK, et al. Travoprost compared with latanoprost and timolol in patients with open angle glaucoma or ocular hypertension. Am J Ophthalmol. 2001;132(4):472–84. doi: 10.1016/s0002-9394(01)01177-1. [DOI] [PubMed] [Google Scholar]
- 44.Sherwood M, Brandt J. Six-month comparison of bimatoprost once daily and twice daily with timolol twice daily in patients with elevated intraocular pressure. Surv Ophthalmol. 2001;45(Suppl 4):S361–8. doi: 10.1016/s0039-6257(01)00219-3. [DOI] [PubMed] [Google Scholar]
- 45.Noecker RS, Dirk MS, Choplin NT, Bernstein P, Batoosingh AL, Whitcup SM, for the Bimatoprost/Latanoprost Study Group A six-month randomised clinical trial comparing the IOP lowering efficacy of bimatoprost and latanoprost in patients with ocular hypertension or glaucoma. Am J ophthalmol. 2003;135(1):55–63. doi: 10.1016/s0002-9394(02)01827-5. [DOI] [PubMed] [Google Scholar]
- 46.Parrish RK, Palmberg P, Sheu WP, for the XLT Study Group A comparison of latanoprost, bimatoprost and travoprost in patients with elevated intraocular pressure: A 12-week randomised masked-evaluator multicentre study. Am J Ophthalmol. 2003;135(5):688–703. doi: 10.1016/s0002-9394(03)00098-9. [DOI] [PubMed] [Google Scholar]
- 47.Azuma I, Masuda K, Kitazawa Y, Yamamura H. Double masked comparative study of UF-021 and timolol ophthalmic solutions in patients with primary open angle glaucoma or ocular hypertension. Jpn J Ophthalmol. 1993;37:514–25. [PubMed] [Google Scholar]
- 48.Takagi Y, Nakajima T, Shimazaki A, et al. Pharmacological characteristics of AFP-168 (tafluprost), a new prostanoid FP receptor agonist, as an ocular hypotensive drug. Exp Eye Res. 2004;78(4):767–76. doi: 10.1016/j.exer.2003.12.007. [DOI] [PubMed] [Google Scholar]
- 49.Ishida N, Odani-Kawabata N, Shimazaki A, Hara H. Prostanoids in the therapy of glaucoma. Cardiovasc Drug Rev. 2006;24(1):1–10. doi: 10.1111/j.1527-3466.2006.00001.x. [DOI] [PubMed] [Google Scholar]
- 50.Sutton A, Gilvarry A, Ropo A. A comparative placebo controlled study of prostanoid fluoroprostaglandin receptor agonists tafluprost and latanprost in healthy males. J Ocul Pharmacol Ther. 2007;23(4):359–65. doi: 10.1089/jop.2006.0061. [DOI] [PubMed] [Google Scholar]
- 51.Brasnu E, Brignole-Baudouin F, Riancho L, Guenon JM, Warnet JM, Baudouin C. In vitro effects of preservative free tafluprost and preserved latanoprost, travoprost and bimatoprost in a conjunctival epithelial cell line. Curr Eye Res. 2008;33(4):303–12. doi: 10.1080/02713680801971857. [DOI] [PubMed] [Google Scholar]
- 52.Baudouin C. Detrimental effects of preservative in eye drops: implications for the treatment of glaucoma. Acta Ophthalmol. 2008;86(7):716–26. doi: 10.1111/j.1755-3768.2008.01250.x. [DOI] [PubMed] [Google Scholar]
- 53.Sutton A, Gouws P, Ropo A. Tafluprost, a new potent prostanoid FP-receptor agonist: a dose response study on pharmacodynamics and tolerability in healthy volunteers. Int J Clin Pharmacol Ther. 2008;46(8):400–6. doi: 10.5414/cpp46400. [DOI] [PubMed] [Google Scholar]
- 54.Alm A, Stjernschantz J. Effects on intraocular pressure and side effects of 0.005% latanoprost applied once daily, evening or morning. A comparison with timolol. Scandinavian Latanoprost Study Group. Ophthalmology. 1995;102(12):1743–52. doi: 10.1016/s0161-6420(95)30798-1. [DOI] [PubMed] [Google Scholar]
- 55.Camras CB. Comparison of latanoprost and timolol in patients with ocular hypertension and glaucoma: a six-month masked multicentre trial in the United States. The United States Latanoprost Study Group. Ophthalmology. 1996;103(1):138–47. doi: 10.1016/s0161-6420(96)30749-5. [DOI] [PubMed] [Google Scholar]
- 56.van der Valk R, Webers CA, Schouten JS, Zeegers MP, Hendrikse F, Prins MH. Intraocular pressure lowering effects of all commonly used glaucoma drugs. Ophthalmology. 2005;112:1177–85. doi: 10.1016/j.ophtha.2005.01.042. [DOI] [PubMed] [Google Scholar]
- 57.Brandt JD, VanDenburgh AM, Chen K, Whitcup SM. Comparison of once or twice daily bimatoprost with twice daily timolol in patients with elevated IOP; a 3-month clinical trial. Ophthalmology. 2001;108:1023–31. doi: 10.1016/s0161-6420(01)00584-x. [DOI] [PubMed] [Google Scholar]
- 58.Whitcup SM, Cantor LB, VanDenburgh AM, Chen K. A randomised double masked multicentre clinical trial comparing bimatoprost and timolol for the treatment of glaucoma and ocular hypertension. Br J Ophthalmol. 2003;87:57–62. doi: 10.1136/bjo.87.1.57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Fellman RL, Sullivan EK, Ratliff M, et al. Comparison of travaprost 0.0015% and 0.004% with timolol 0.5% in patients with elevated intraocular pressure: a 6-month masked multicentre trial. Ophthalmology. 2002;109:998–1008. doi: 10.1016/s0161-6420(02)01010-2. [DOI] [PubMed] [Google Scholar]
- 60.Goldberg I, Cunha-Vaz J, Jakobsen JE, et al. Comparison of topical travoprost eye drops given once daily and timolol 0.5% given twice daily in patients with open angle glaucoma or ocular hypertension. J Glaucoma. 2001;10:414–22. doi: 10.1097/00061198-200110000-00010. [DOI] [PubMed] [Google Scholar]
- 61.Netland PA, Landry T, Sullivan EK, et al. Travoprost compared with latanoprost and timolol in patients with open angle glaucoma or ocular hypertension. Am J Ophthalmol. 2001;132:472–84. doi: 10.1016/s0002-9394(01)01177-1. [DOI] [PubMed] [Google Scholar]
- 62.Hedman K, Alm A. A pooled-data analysis of three randomised double masked six-month clinical studies comparing the intraocular pressure reducing effect of latanoprost and timolol. Eur J Ophthalmol. 2000;10:95–104. doi: 10.1177/112067210001000201. [DOI] [PubMed] [Google Scholar]
- 63.Schlect LP, Brubaker RF. The effects of withdrawal of timolol in chronically treated glaucoma patients. Ophthalmology. 1998;95(9):1212–6. doi: 10.1016/s0161-6420(88)33026-5. [DOI] [PubMed] [Google Scholar]
- 64.Korte JM, Kaila T, Saari KM. Systemic bioavailability and cardiopulmonary effects of 0.5% timolol eye drops. Graefes Arch Clin Exp Ophthalmol. 2002;240(6):430–5. doi: 10.1007/s00417-002-0462-2. [DOI] [PubMed] [Google Scholar]
- 65.Stamper RL, Wigginton SA, Higginbottom EJ. Primary drug treatment for glaucoma: Beta blockers versus other medications. Surv Ophthalmol. 2002;47(1):63–73. doi: 10.1016/s0039-6257(01)00286-7. [DOI] [PubMed] [Google Scholar]
- 66.European Glaucoma Prevention Study Group (EGPS) Ophthalmology. 2002;109:1612–21. doi: 10.1016/s0161-6420(02)01167-3. [DOI] [PubMed] [Google Scholar]
- 67.Docherty JR. Subtypes of functional alpha1-adrenoreceptor. Cell Mol Life Sci. 2010;67:405–17. doi: 10.1007/s00018-009-0174-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Adkins JC, Balfour JA. Brimonidine. A review of its pharmacological properties and clinical potential in the management of open angle glaucoma and ocular hypertension. Drugs & Aging. 1998;12(3):225–41. doi: 10.2165/00002512-199812030-00005. [DOI] [PubMed] [Google Scholar]
- 69.Blondeau P, Rousseau JA. Allergic reactions to brimonidine in patients treated for glaucoma. Can J Ophthalmol. 2002;37:21–6. doi: 10.1016/s0008-4182(02)80094-1. [DOI] [PubMed] [Google Scholar]
- 70.Chung HS, Shin DH, Bir CM, et al. Chronic use of apraclonidine decreases its moderation of post laser intraocular pressure spikes. Ophthalmology. 1997;104:1921–5. doi: 10.1016/s0161-6420(97)30006-2. [DOI] [PubMed] [Google Scholar]
- 71.Kalapesi FB, Coroneo MT, Hill MA. Human ganglion cells express the alpha-2 adrenergic receptor: relevance to neuroprotection. Br J Ophthalmol. 2005;89:758–63. doi: 10.1136/bjo.2004.053025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Donello JE, Padillo EU, Webster ML, Wheeler LA, Gill DW. Alpha 2-adrenoreceptor agonists inhibit vitreal glutamate and aspartate accumulation and preserve retinal function after transient ischaemia. J Pharmacol Exp Ther. 2001;296:216–23. [PubMed] [Google Scholar]
- 73.Yoles E, Wheeler LA, Scwartz M. Alpha2-adrenoreceptor agonists are neuroprotective in a rat model of optic nerve degeneration. Invest Ophthalmol Vis Sci. 1999;40:65–73. [PubMed] [Google Scholar]
- 74.Krupin T, Liebman JN, Greenfield DS, Ritch R, Gardiner S, Low-Pressure Glaucoma Study Group A Randomised trial of brimonidine versus timolol in preserving visual function: results from the low pressure glaucoma treatment study. Am J Ophthalmol. 2011;151(4):671–81. doi: 10.1016/j.ajo.2010.09.026. [DOI] [PubMed] [Google Scholar]
- 75.Crawford K, Kaufman PL. Pilocarpine antagonises prostaglandin F2 alpha-induced ocular hypotension in monkeys: evidence for enhancement of uveoscleral outflow by prostaglandin F2 alpha. Arch Ophthalmol. 1987;105(8):1112–6. doi: 10.1001/archopht.1987.01060080114039. [DOI] [PubMed] [Google Scholar]
- 76.Drance SM, Nash PA. The dose response of human intraocular pressure to pilocarpine. Can J Ophthalmol. 1971;6(1):9–13. [PubMed] [Google Scholar]
- 77.European Glaucoma Society . Terminology and Guidelines for Glaucoma. 3rd edition. Savona, Italy: Dogma; 2008. [Google Scholar]
- 78.Hutzlemann J, Owens S, Shedden A, Adamsons I, Vargas E. Comparison of the safety and efficacy of fixed combination of dorzolamide/timolol and the concomitant administration dorzolamide and timolol: a clinical equivalence study. International Clinical Equivalence Study Group. Br J Ophthalmol. 1998;82(11):1249–53. doi: 10.1136/bjo.82.11.1249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Clineschmidt CM, Williams RD, Snyder E, Adamsons IA. A randomised trial in patients inadequately controlled with timolol alone comparing the dorzolamide-timolol combination to monotherapy with timolol or dorzolamide. Dorzolamide-Timolol Combination Study Group. Ophthalmology. 1998;105(10):1952–9. doi: 10.1016/s0161-6420(98)91047-8. [DOI] [PubMed] [Google Scholar]
- 80.Sherwood MB, Craven ER, Chou C, et al. Twice daily 0.2% brimonidine-0.5% timolol fixed combination therapy vs. monotherapy with timolol or brimonidine in patients with glaucoma or ocular hypertension: a 12-month randomised trial. Arch Ophthalmol. 2006;124(9):1230–8. doi: 10.1001/archopht.124.9.1230. [DOI] [PubMed] [Google Scholar]
- 81.Goni FJ. Brimonidine/Timolol fixed combination study group. 12-week study comparing the fixed combination of brimonidine and timolol with concomitant use of the individual components in patients with glaucoma and ocular hypertension. Eur J Ophthalmol. 2005;15(5):581–90. [PubMed] [Google Scholar]
- 82.Konstas AG, Katsimpris IE, Kaltsos K, et al. Twenty-four hour efficacy of the brimonidine/timolol fixed combination versus therapy with the unfixed components. Eye. 2008;22(11):1391–7. doi: 10.1038/sj.eye.6702906. [DOI] [PubMed] [Google Scholar]
- 83.Konstas AG, Quaranta L, Yan DB, et al. Twenty-four hour efficacy with the dorzolamide/timolol fixed combination compared with the brinzolamide/timolol fixed combination in primary open angle glaucoma. Eye. 2011 Sep 30; doi: 10.1038/eye.2011.239. [Epub ahead of print.] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Pfeiffer N, European Latanoprost Fixed Combination Study Group A comparison of the fixed combination of latanoprost and timolol with its individual components. Graefes Arch Clin Exp Ophthalmol. 2002;240:893–9. doi: 10.1007/s00417-002-0553-0. [DOI] [PubMed] [Google Scholar]
- 85.Higginbotham EJ, Diestelhorst M, Pfeiffer N, et al. The efficacy and safety of unfixed and fixed combinations of latanoprost and other antiglaucoma medications. Surv Ophthalmol. 2002;47(Suppl 1):S133–40. doi: 10.1016/s0039-6257(02)00295-3. [DOI] [PubMed] [Google Scholar]
- 86.Barneby H, Orengo-Nania S, Flowers BE, et al. The safety and efficacy of travoprost 0.004%/Timolol 0.5% fixed combination ophthalmic solution. Am J Ophthalmol. 2005;140:1–7. doi: 10.1016/j.ajo.2005.02.043. [DOI] [PubMed] [Google Scholar]
- 87.Diestelhorst M, Larsson LI, European Latanoprost Fixed Combination Study Group A 12-week study comparing the fixed combination of latanoprost and timolol with the concomitant use of the individual components in patients with open angle glaucoma and ocular hypertension. Br J Ophthalmol. 2004;88:199–203. doi: 10.1136/bjo.2003.018234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Hughes BA, Bacharach J, Craven ER, et al. A three-month multi centre double masked study of the safety and efficacy of travoprost 0.004%/timolol 0.5% ophthalmic solution compared to travoprost 0.004% ophthalmic solution and timolol 0.5% dosed concomitantly in subjects with open angle glaucoma and ocular hypertension. J Glaucoma. 2005;14:392–9. doi: 10.1097/01.ijg.0000176935.08392.14. [DOI] [PubMed] [Google Scholar]
- 89.Brandt JD, Cantor LB, Katz LJ, Batoosingh AL, Chou C, Bossowska I, Ganfort Investigators Group II Bimatoprost/timolol fixed combination: a 3-month double masked randomised parallel comparison to its individual components in patients with glaucoma or ocular hypertension. J Glaucoma. 2008;17(3):211–6. doi: 10.1097/IJG.0b013e3181507313. [DOI] [PubMed] [Google Scholar]
- 90.Hommer A, Ganfort Investigators Group I A double masked randomised parallel comparison of a fixed combination of bimatoprost 0.03%/timolol 0.5% with non-fixed combination use in patients with glaucoma or ocular hypertension. Eur J Ophthalmol. 2007;17(1):53–62. doi: 10.1177/112067210701700108. [DOI] [PubMed] [Google Scholar]
- 91.Webers CA, Becjers HJ, Zeegers MP, Nuijts RM, Hendrikse F, Schouten JS. The intraocular pressure lowering effect of prostaglandin analogues combined with topical β blocker therapy: a systematic review and meta-analysis. Ophthalmology. 2010;117(11):2067–74. doi: 10.1016/j.ophtha.2010.03.024. [DOI] [PubMed] [Google Scholar]
- 92.Boyle JE, Ghosh K, Gieser DK, et al. A randomised trial comparing dorzolamide/timolol combination given twice daily to monotherapy with timolol and dorzolamide. Ophthalmology. 1998;105(10):1945–51. doi: 10.1016/s0161-6420(98)91046-6. [DOI] [PubMed] [Google Scholar]
- 93.Sall KN, Greff LJ, Johnson-Pratt LR, et al. Dorzolamide/timolol 51 combination versus concomitant administration of brimonidine and timolol: a six-month comparison of efficacy and tolerability. Ophthalmology. 2003;110(3):615–24. doi: 10.1016/S0161-6420(02)01900-0. [DOI] [PubMed] [Google Scholar]
- 94.Samples JR, Singh K, Lin SC, et al. Laser trabeculoplasty for open angle glaucoma; A report by the American Academy of Ophthalmology. Ophthalmology. 2011 doi: 10.1016/j.ophtha.2011.04.037. [Epub ahead of print.] [DOI] [PubMed] [Google Scholar]
- 95.Krasnov MM. Laser puncture of the anterior chamber angle in glaucoma (a preliminary report) [In Russian] Vest Oftalmol. 1972;3:27–31. [PubMed] [Google Scholar]
- 96.Hager H. Special microsurgical interventions 2. First experiences with the argon laser apparatus 800 [in German] Klin Monbl Augenheilkd. 1973;162:437–50. [PubMed] [Google Scholar]
- 97.Wise JB, Witter SL. Argon laser therapy for open angle glaucoma: a pilot study. Arch Ophthalmol. 1979;97:319–22. doi: 10.1001/archopht.1979.01020010165017. [DOI] [PubMed] [Google Scholar]
- 98.Latina MA, Park C. Selective targeting of trabecular meshwork cells: in vitro studies of pulsed and CW laser interactions. Exp Eye Res. 1995;60:359–71. doi: 10.1016/s0014-4835(05)80093-4. [DOI] [PubMed] [Google Scholar]
- 99.Kramer TR, Noecker RJ. Comparison of the morphologic changes after selective laser trabeculoplasty and argon laser trabeculoplasty in human eye bank eyes. Ophthalmology. 2001;108:773–9. doi: 10.1016/s0161-6420(00)00660-6. [DOI] [PubMed] [Google Scholar]
- 100.Brancato R, Carassa R, Trabucchi G. Diode laser compared with argon laser for trabeculoplasty. Am J Ophthalmol. 1991;112:50–5. doi: 10.1016/s0002-9394(14)76212-9. [DOI] [PubMed] [Google Scholar]
- 101.Chung PY, Schuman JS, Netland PA, et al. Five year results of a randomised prospective clinical trial of diode vs. argon laser trabeculoplasty for open angle glaucoma. Am J Ophthalmol. 1998;126:185–90. doi: 10.1016/s0002-9394(98)00151-2. [DOI] [PubMed] [Google Scholar]
- 102.McHugh D, Marshall J, Ffytche TJ, et al. Diode laser trabeculoplasty (DLT) for primary open angle glaucoma and ocular hypertension. Br J Ophthalmol. 1990;74:743–7. doi: 10.1136/bjo.74.12.743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Moriarty AP, McHugh JD, Ffytche TJ, et al. Longterm follow up of diode laser trabeculoplasty for primary open angle glaucoma and ocular hypertension. Ophthalmology. 1993;100:1614–8. doi: 10.1016/s0161-6420(93)31428-4. [DOI] [PubMed] [Google Scholar]
- 104.Stein JD, Challa P. Mechanisms of action and efficacy of argon laser trabeculoplasty and selective laser trabeculoplasty. Curr Opin Ophthalmol. 2007;18:140–5. doi: 10.1097/ICU.0b013e328086aebf. [DOI] [PubMed] [Google Scholar]
- 105.Rolim de Moura CR, Paranhos A, Jr, Wormald R. Laser trabeculoplasty for open angle glaucoma. Cochrane Database of Systematic Reviews. 2007 doi: 10.1002/12651858.CD003919.pub2. Issue 4 Art No.: CD003910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Melamed S, Pei J, Epstein DL. Delayed response to argon laser trabeculoplasty in monkeys: morphological and morphometric analysis. Arch Ophthalmol. 1986;104:10778–83. doi: 10.1001/archopht.1986.01050190136053. [DOI] [PubMed] [Google Scholar]
- 107.Melamed S, Pei J, Epstein DL. Short term effect of argon laser trabeculoplasty in monkeys. Arch Ophthalmol. 1985;103:1546–62. doi: 10.1001/archopht.1985.01050100122032. [DOI] [PubMed] [Google Scholar]
- 108.Van Buskirk EM, Pond V, Rosenquist RC, Acott TS. Argon laser trabeculoplasty: studies of mechanism of action. Ophthalmology. 1984;91:1005–10. doi: 10.1016/s0161-6420(84)34197-5. [DOI] [PubMed] [Google Scholar]
- 109.Ruddat MS, Alexander JR, Samples JR, et al. Early changes in trabecular metalloproteinase mRNA levels in response to argon laser trabeculoplasty are induced by media borne factors. Invest Ophthalmol Vis Sci. 30(Suppl):280. 198. [Google Scholar]
- 110.Latina MA, DeLeon JM. Selective laser trabeculoplasty. Ophthalmol Clin N Am. 2005;18:409–19. doi: 10.1016/j.ohc.2005.05.005. [DOI] [PubMed] [Google Scholar]
- 111.Bylsma SS, Samples JR, Acott TS, Van Buskirk EM. Trabecular cell division after argon laser trabeculoplasty. Arch Ophthalmol. 1988;106:544–7. doi: 10.1001/archopht.1988.01060130590044. [DOI] [PubMed] [Google Scholar]
- 112.Glaucoma Laser trial Research Group The Glaucoma Laser Trial (GLT) and Glaucoma Laser Trial Follow-up Study:7. Results. Am J Ophthalmol. 1995;120:718–31. doi: 10.1016/s0002-9394(14)72725-4. [DOI] [PubMed] [Google Scholar]
- 113.Glaucoma Laser Trial Research Group The Glaucoma Laser Trial (GLT) 2. Results of argon laser trabeculoplasty versus topical medicines. Ophthalmology. 1990;97:1403–3. [PubMed] [Google Scholar]
- 114.Migdal C, Gregory W, Hitchings R. Long term functional outcome after early surgery compared with laser and medicine in open angle glaucoma. Ophthalmology. 1994;101:1651–6. doi: 10.1016/s0161-6420(94)31120-1. [DOI] [PubMed] [Google Scholar]
- 115.Leske MC, Heijl A, Hussein M, Early Manifest Glaucoma Trial Group Predictors of long term progression in the Early Manifest Glaucoma Trial. Ophthalmology. 2007;114:1965–72. doi: 10.1016/j.ophtha.2007.03.016. [DOI] [PubMed] [Google Scholar]
- 116.Heijl A, Leske MC, Bengtsson B, et al. Reduction of intraocular pressure and glaucoma progression: results from the Early Manifest Glaucoma Trial. Arch Ophthalmol. 2002;120:1268–79. doi: 10.1001/archopht.120.10.1268. [DOI] [PubMed] [Google Scholar]
- 117.AGIS Investigators The Advanced Glaucoma Intervention Study (AGIS):7. The relationship between control of intraocular pressure and visual field deterioration. Am J Ophthalmol. 2000;130:429–40. doi: 10.1016/s0002-9394(00)00538-9. [DOI] [PubMed] [Google Scholar]
- 118.AGIS Investigators The Advanced Glaucoma Intervention Study (AGIS): 13. Comparison of treatment outcomes within race: 10-year results. Ophthalmology. 2004;111:651–4. doi: 10.1016/j.ophtha.2003.09.025. [DOI] [PubMed] [Google Scholar]
- 119.Cvenkel B. One-year follow up of selective laser trabeculoplasty in open angle glaucoma. Ophthalmologica. 2004;218:20–5. doi: 10.1159/000074562. [DOI] [PubMed] [Google Scholar]
- 120.Gracner T. Intraocular pressure reduction after selective laser trabeculoplasty in primary open angle glaucoma. Collegium Antropologicum. 2001;25(Suppl):S111–5. [PubMed] [Google Scholar]
- 121.Gracner T. Intraocular pressure response of capsular glaucoma and primary open angle glaucoma to selective Nd:YAG laser trabeculoplasty: a prospective comparative clinical trial. Eur J Ophthalmol. 2002;12:287–92. doi: 10.1177/112067210201200406. [DOI] [PubMed] [Google Scholar]
- 122.Johnson PB, Katz LJ, Rhee DJ. Selective laser trabeculoplasty: predictive value of early intraocular pressure measurements for success at 3 months. Br J Ophthalmol. 2006;90:741–3. doi: 10.1136/bjo.2005.086363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Kajiya S, Hayakawa K, Sawaguchi S. Clinical results of selective laser trabeculoplasty. Nippon Ganka Gakkai Zasshi Japonicae. 2000;104:160–4. [PubMed] [Google Scholar]
- 124.Kano K, Kuwayama Y, Mizoue S, Ito N. Clinical results of selective laser trabeculoplasty. Nippon Ganka Gakkai Zasshi Japonicae. 1999;103:612–6. [PubMed] [Google Scholar]
- 125.Kim YJ, Moon CS. One year follow up of laser trabeculoplasty using Q-switched frequency doubled Nd-YAG laser of 532 nm wavelength. Ophthalmic Surg Lasers. 2000;31:394–9. [PubMed] [Google Scholar]
- 126.Lai JS, Chua JK, Tham CC, Lam DS. Five year selective laser trabeculoplasty in Chinese eyes. Clin Exp Ophthalmol. 2004;32:368072. doi: 10.1111/j.1442-9071.2004.00839.x. [DOI] [PubMed] [Google Scholar]
- 127.Lanzetta P, Menchini U, Virgili G. Immediate intraocular pressure response to selective laser trabeculoplasty. Br J Ophthalmol. 1999;83:29–32. doi: 10.1136/bjo.83.1.29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Latina MA, Sibayan SA, Shin DH, et al. Q-switched 532 nm Nd-YAG laser trabeculoplasty (selective laser trabeculoplasty): a multicentre pilot clinical study. Ophthalmology. 1998;105:2082–8. doi: 10.1016/S0161-6420(98)91129-0. [DOI] [PubMed] [Google Scholar]
- 129.Melamed S, Ben Simon GJ, Levkovitch-Verbin H. Selective laser trabeculoplasty as a primary treatment for open angle glaucoma; a prospective non randomised pilot study. Arch Ophthalmol. 2003;121:957–60. doi: 10.1001/archopht.121.7.957. [DOI] [PubMed] [Google Scholar]
- 130.Rozsival P, Kana V, Hovorkova M. Selective laser trabeculoplasty. Ceska a Slovenska. Oftalmologie. 2004;60:267–74. [PubMed] [Google Scholar]
- 131.Song J, Lee PP, Epstein DL, et al. High failure rate associated with 180 degrees selective laser trabeculoplasty. J Glaucoma. 2005;14:400–8. doi: 10.1097/01.ijg.0000176939.43681.c2. [DOI] [PubMed] [Google Scholar]
- 132.McIlraith I, Strasfield M, Colev G, Hutnik CM. Selective laser trabeculoplasty as initial and adjunctive treatment for open angle glaucoma. J Glaucoma. 2006;15:124–30. doi: 10.1097/00061198-200604000-00009. [DOI] [PubMed] [Google Scholar]
- 133.Detry-Morel M, Muschart F, Pourjavan S. Micropulse diopde laser (810 nm) versus argon laser trabeculoplasty in the treatment of open angle glaucoma: comparative short-term safety and efficacy profile. Bull Soc Belge Ophthalmol. 2008;308:21–8. [PubMed] [Google Scholar]
- 134.Goldenfeld M, Melamed S, Simon G, Ben Simon GJ. Titanium:sapphire laser trabeculoplasty versus argon laser trabeculoplasty in patients with open angle glaucoma. Ophthalmic Surg Lasers Imaging. 2009;40:264–9. doi: 10.3928/15428877-20090430-07. [DOI] [PubMed] [Google Scholar]
- 135.Feldman RM, Katz LJ, Spaeth GL, et al. Long-term efficacy of repeat argon laser trabeculoplasty. Ophthalmology. 1991;98:1061–65. doi: 10.1016/s0161-6420(91)32176-6. [DOI] [PubMed] [Google Scholar]
- 136.Richter CU, Shingleton BJ, Bellows Ar, et al. Retreatment with argon laser trabeculoplasty. Ophthalmology. 1987;94:1085–9. doi: 10.1016/s0161-6420(87)33319-6. [DOI] [PubMed] [Google Scholar]
- 137.Weber PA, Burton GD, Epitropoulos AT. Laser trabeculoplasty retreatment. Ophthalmic Surg. 1989;20:702–6. [PubMed] [Google Scholar]
- 138.Grayson DK, Camras CB, Podos SM, Lustgarten JS. Long term reduction of intraocular pressure after repeat argon laser trabeculoplasty. Am J Ophthalmol. 1988;106:312–21. doi: 10.1016/0002-9394(88)90367-4. [DOI] [PubMed] [Google Scholar]
- 139.Starita RJ, Fellman RL, Spaeth GL, et al. The effect of repeating full circumference argon laser trabeculoplasty. Ophthalmic Surg. 1984;15:41–3. [PubMed] [Google Scholar]
- 140.Shah N, Yadav R, Nagar M. Selective laser trabeculoplasty: the effect of enhancement and retreatment on IOP control. Paper presented at the XXIV Congress of the European Cataract and Refractive Surgeons (ESCRS); London. 2006. [Google Scholar]
- 141.Lee R, Hutnik CM. Projected cost comparison of selective laser trabeculoplasty versus glaucoma medication in the Ontario Health Insurance Plan. Can J Ophthalmol. 2006;41(4):449–56. doi: 10.1016/S0008-4182(06)80006-2. [DOI] [PubMed] [Google Scholar]
- 142.Cantor LB, Katz LJ, Cheng JW, et al. Economic evaluation of medication, laser trabeculoplasty and filtering surgeries in treating patients with glaucoma in the US. Curr Med Res Opin. 2008;24:2905–18. doi: 10.1185/03007990802379996. [DOI] [PubMed] [Google Scholar]
- 143.Lin SC. Endoscopic and transscleral cyclophotocoagulation for the treatment of refractory glaucoma. J Glaucoma. 2008;17:238–47. doi: 10.1097/IJG.0b013e31815f2539. [DOI] [PubMed] [Google Scholar]
- 144.Beckman H, Kinoshita A, Rota AN, et al. Transscleral ruby laser irradiation of the ciliary body in the treatment of intractable glaucoma. Trans Am Acad Ophth Otol. 1972;76:423–36. [PubMed] [Google Scholar]
- 145.Beckman H, Sugar HS. Neodymium laser cyclocoagulation. Arch Ophthalmol. 1973;90:27–8. doi: 10.1001/archopht.1973.01000050029006. [DOI] [PubMed] [Google Scholar]
- 146.Hampton C, Shields MB, Miller KN, et al. Evaluation of a protocol for transscleral Nd-YAG cyclophotocoagulation in one hundred patients. Ophthalmology. 1990;97:910–7. doi: 10.1016/s0161-6420(90)32482-x. [DOI] [PubMed] [Google Scholar]
- 147.Wright MM, Grajewski AI, Feuer WJ. YAG cyclophotocoagulation: outcome of treatment for uncontrolled glaucoma. Ophthalmic Surg. 1991;22:279–83. [PubMed] [Google Scholar]
- 148.Simmons RB, Shields MB, Blasini M, et al. Transcleral Nd:YAG laser cyclophotocoagulation with a contact lens. Am J Ophthalmol. 1991;112:671–7. doi: 10.1016/s0002-9394(14)77273-3. [DOI] [PubMed] [Google Scholar]
- 149.Noureddin BN, Wilson-Holt N, Lavin M, et al. Advanced uncontrolled glaucoma: Nd:YAG cyclophotocoagulation or tube surgery. Ophthalmology. 1992;99:430–7. [PubMed] [Google Scholar]
- 150.Shields MB, Shields SE. Noncontact transscleral Nd:YAG cyclophotocoagulation: a long term follow up of 500 patients. Trans Am Ophthalmol Soc. 1994;92:271–87. [PMC free article] [PubMed] [Google Scholar]
- 151.Dickens CJ, Hguyen N, Mora JS, et al. Long-term results of non-contact transscleral neodymium:YAG cyclophotocoagulation. Ophthalmology. 1995;102:1777–81. doi: 10.1016/s0161-6420(95)30794-4. [DOI] [PubMed] [Google Scholar]
- 152.Brancato R, Giovanni L, Trabucchi G, et al. Contact transscleral cyclophotocoagulation with Nd:YAG laser in uncontrolled glaucoma. Ophthalmic Surg. 1989;20:547–51. [PubMed] [Google Scholar]
- 153.Schuman JS, Puliafito CA, Allingham RR, et al. Contact transscleral continuous wave neodymium:YAG laser cyclophotocoagulation. Ophthalmology. 1990;97:571–80. doi: 10.1016/s0161-6420(90)32550-2. [DOI] [PubMed] [Google Scholar]
- 154.Schuman JS, Bellows AR, Shingleton BJ, et al. Contact transscleral Nd:YAG laser cyclophotocoagulation: midterm results. Ophthalmology. 1992;99:1089–95. doi: 10.1016/s0161-6420(92)31846-9. [DOI] [PubMed] [Google Scholar]
- 155.Lin P, Wollstein G, Glavas IP, et al. Contact transscleral Nd:YAG laser cyclophotocoagulation: long-term outcome. Ophthalmology. 2004;111:2137–43. doi: 10.1016/j.ophtha.2004.05.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Bloom PA, Tsai JC, Sharma K, et al. Cyclodiode: transscleral diode laser cyclophotocoagulation in the treatment of advanced refractory glaucoma. Ophthalmology. 1997;104:1508–20. doi: 10.1016/s0161-6420(97)30109-2. [DOI] [PubMed] [Google Scholar]
- 157.Spencer AF, Vernon SA. Cyclodiode: results of a standard protocol. Br J Ophthalmol. 1999;83:311–6. doi: 10.1136/bjo.83.3.311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Pucci V, Tappainer F, Borin S, et al. Long-term follow up after transscleral diode laser photocoagulation in refractory glaucoma. Ophthalmologica. 2003;217:279–83. doi: 10.1159/000070635. [DOI] [PubMed] [Google Scholar]
- 159.Murphy CC, Burnett CA, Spry PG, et al. A two centre study of the dose response relation for transscleral diode laser cyclophotocoagulation in refractory glaucoma. Br J Ophthalmol. 2003;87:1252–7. doi: 10.1136/bjo.87.10.1252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Hauber FA, Scherer WJ. Influence of total energy delivery on success rate after contact diode laser transscleral cyclophotocoagulation : a retrospective case review and meta-analysis. J Glaucoma. 2002;11:329–33. doi: 10.1097/00061198-200208000-00009. [DOI] [PubMed] [Google Scholar]
- 161.Noureddin BN, Zein W, Haddad C, et al. Diode laser transscleral cyclophotocoagulation for refractory glaucoma: a 1 year follow up of patients treated using an aggressive protocol. Eye. 2006;20:329–35. doi: 10.1038/sj.eye.6701875. [DOI] [PubMed] [Google Scholar]
- 162.Kosoko O, Gaasterland DE, Pollack IP, et al. Longterm outcome of initial ciliary ablation with contact diode laser transscleral cyclophotocoagulation for severe glaucoma. Ophthalmology. 1996;103:1294–302. doi: 10.1016/s0161-6420(96)30508-3. [DOI] [PubMed] [Google Scholar]
- 163.Egbert PR, Fiadoyor S, Budenz DL, et al. Diode laser transscleral cyclophotocoagulation as a primary surgical treatment for primary open angle glaucoma. Arch Ophthalmol. 2001;119:345–50. doi: 10.1001/archopht.119.3.345. [DOI] [PubMed] [Google Scholar]
- 164.Kramp K, Vick HP, Guthoff R. Transscleral diode laser contact cyclophotocoagulation in the treatment of different glaucomas, also as primary surgery. Graefes Arch Clin Exp Ophthalmol. 2002;240:698–703. doi: 10.1007/s00417-002-0508-5. [DOI] [PubMed] [Google Scholar]
- 165.Lai JS, Than CC, Chan JC, et al. Diode laser transscleral cyclophotocoagulation as a primary surgical treatment for medically uncontrolled chronic angle closure glaucoma: longterm clinical outcomes. J Glaucoma. 2005;14:114–9. doi: 10.1097/01.ijg.0000151890.41239.c5. [DOI] [PubMed] [Google Scholar]
- 166.Grueb M, Rohrbach JM, Bart-Schmidt KU, et al. Transscleral diode laser contact cyclophotocoagulation as primary and secondary surgical treatment in primary open angle glaucoma and pseudoexfoliative glaucoma. Longterm clinical outcomes. Graefes Arch Clin Exp Ophthalmol. 2006;244:1293–9. doi: 10.1007/s00417-006-0280-z. [DOI] [PubMed] [Google Scholar]
- 167.Ansari E, Gandhewar J. Longterm efficacy and visual acuity following transscleral diode laser contact cyclophotocoagulation in cases of refractory and non-refractory glaucoma. Eye. 2007;21:936–40. doi: 10.1038/sj.eye.6702345. [DOI] [PubMed] [Google Scholar]
- 168.Youn J, Cox TA, Herndon LW, et al. A clinical comparison of transscleral diode laser contact cyclophotocoagulation with Nd:YAG and semiconductor diode lasers. Am J Ophthalmol. 1998;126:640–7. doi: 10.1016/s0002-9394(98)00228-1. [DOI] [PubMed] [Google Scholar]
- 169.Chen J, Cohn RA, Lin SC, et al. Endoscopic cyclophotocoagulation of the ciliary body for treatment of refractory glaucomas. Am J Ophthalmol. 1997;124:787–96. doi: 10.1016/s0002-9394(14)71696-4. [DOI] [PubMed] [Google Scholar]
- 170.Lin S. Endoscopic cyclophotocoagulation. Br J Ophthalmol. 2002;86:1434–8. doi: 10.1136/bjo.86.12.1434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Burr J, Azuara-Blanco A, Avenell A. Medical versus surgical interventions for open angle glaucoma. Cochrane Database of Systematic Reviews. 2004;(2) doi: 10.1002/14651858.CD004399.pub2. Art No.: CD004399. [DOI] [PubMed] [Google Scholar]
- 172.Cairns JE. Preliminary report of a new method. Am J Ophthalmol. 1968;66:673–9. Trabeculectomy. [PubMed] [Google Scholar]
- 173.Migdal C, Hitchings R. Control of chronic simple glaucoma with primary medical, surgical and laser treatment. Trans Ophthalmol Soc U K. 1986;105(Pt 6):653–6. [PubMed] [Google Scholar]
- 174.Kirwan JF, Rennie C, Evans JR. Beta radiation for glaucoma surgery. Cochrane Database of Systematic Reviews. 2009;(2) doi: 10.1002/14651858.CD003433.pub2. Art No.: CD003433. 1.1002/14651858.CD003433.pub2. [DOI] [PubMed] [Google Scholar]
- 175.Jones E, Clarke J, Khaw PT. Recent advances in trabeculectomy technique. Curr Opin Ophthalmol. 2005;16:107–13. doi: 10.1097/01.icu.0000156138.05323.6f. [DOI] [PubMed] [Google Scholar]
- 176.Lanvin MJ, Wormald RP, Migdal CS, Hitchings RA. The influence of prior therapy on the success of trabeculectomy. Arch Ophthalmol. 1990;108(11):1543–8. doi: 10.1001/archopht.1990.01070130045027. [DOI] [PubMed] [Google Scholar]
- 177.Wormald R, Wilkins M, Bunce C. Postoperative 5-Fluorouracil for glaucoma surgery. Cochrane Database of Systematic Reviews. 2001;(3) doi: 10.1002/14651858.CD001132. Art. No.: CD001132. [DOI] [PubMed] [Google Scholar]
- 178.Hitchings RA, Grierson I. Clinicopathological correlation in eyes with failed fistulising surgery. Trans Ophth Soc U K. 1983;103(Pt 1):84–8. [PubMed] [Google Scholar]
- 179.Palmer SS. Mitomycin as an adjunct chemotherapy with trabeculectomy. Ophthalmology. 1991;98(3):317–21. doi: 10.1016/s0161-6420(91)32293-0. [DOI] [PubMed] [Google Scholar]
- 180.Khaw PT. Advances in glaucoma surgery: evolution of antimetabolite adjunctive therapy. J Glaucoma. 2001;10(5 Suppl 1):S81–4. doi: 10.1097/00061198-200110001-00029. [DOI] [PubMed] [Google Scholar]
- 181.Wilkins M, Indar A, Wormald R. Intraoperative Mitomycin C for glaucoma surgery. Cochrane Database of Systematic Reviews. 2005;(4) doi: 10.1002/14651858.CD002897.pub.2. Art No.: CD002897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Minckler DS, Francis BA, Hodapp EA, et al. Aqueous shunts in Glaucoma: A report by the American Academy of Ophthalmology. Ophthalmology. 2003;115:1089–98. doi: 10.1016/j.ophtha.2008.03.031. [DOI] [PubMed] [Google Scholar]
- 183.Minckler DS, Shammas A, Wilcox M, Ogden TE. Experimental studies of aqueous filtration using the Molteno implant. Trans Am Ophthalmol Soc. 1987;85:368–92. [PMC free article] [PubMed] [Google Scholar]
- 184.Gedde SJ, Heuer DK, Parrish RK, II, The Tube Versus Trabeculectomy Study Group Review of results from the tube versus trabeculectomy study. Curr Opin Ophthalmol. 2010;21:123–8. doi: 10.1097/ICU.0b013e3283360b68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Minckler DS, Vedula SS, Li TJ, et al. Aqueous shunts for glaucoma. Cochrane Database of Systematic Reviews. 2006;(2) doi: 10.1002/14651858. Art. No.: CD004918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Heuer DK, Lloyd MA, Abrams DA, et al. Which is better? One or two? A randomised clinical trial of single-plate versus double-plate Molteno implantation for glaucomas in aphakia and pseudophakia. Ophthalmology. 1992;99:1512–9. doi: 10.1016/s0161-6420(92)31772-5. [DOI] [PubMed] [Google Scholar]
- 187.Cantor L, Burgoyne J, Sanders S, et al. The effect of mitomycin C on Molteno implant surgery: a 1 year randomised masked prospective study. J Glaucoma. 1998;7:240–6. [PubMed] [Google Scholar]
- 188.Costa VP, Azuara-Blanco A, Netland PA, et al. Efficacy and safety of adjunctive mitomycin C during Ahmed glaucoma valve implantation: a prospective randomised clinical trial. Ophthalmology. 2004;111:1071–6. doi: 10.1016/j.ophtha.2003.09.037. [DOI] [PubMed] [Google Scholar]
- 189.Ramulu PY, Corcoran KJ, Corcoran SL, Robin AL. Utilisation of various glaucoma surgeries and procedures in Medicare beneficiaries from 1995 to 2004. Ophthalmology. 2007;114:2265–70. doi: 10.1016/j.ophtha.2007.02.005. [DOI] [PubMed] [Google Scholar]
- 190.Hong CH, Arosemena A, Zurakowski D, Ayyala RS. Glaucoma drainage devices: a systematic literature review and current controversies. Surv Ophthalmol. 2005;50:48–60. doi: 10.1016/j.survophthal.2004.10.006. [DOI] [PubMed] [Google Scholar]
- 191.Ravinet E, Tritten JJ, Roy S, et al. Descemet membrane detachment after nonpenetrating filtering surgery. J Glaucoma. 2002;11:244–52. doi: 10.1097/00061198-200206000-00014. [DOI] [PubMed] [Google Scholar]
- 192.Sarodia U, Shaarawy T, Barton K. Nonpenetrating glaucoma surgery: a critical evaluation. Curr Opin Ophthalmol. 2007;18:152–8. doi: 10.1097/ICU.0b013e328091c1ae. [DOI] [PubMed] [Google Scholar]
- 193.Chiou AGY, Mermoud A, Jewelewicz DA. Postoperative inflammation following deep sclerectomy with collagen implant versus standard trabeculectomy. Graefes Arch Clin Exp Ophthalmol. 1998;236:593–6. doi: 10.1007/s004170050127. [DOI] [PubMed] [Google Scholar]
- 194.Egrilmez S, Ates H, Nalcaci S, et al. Surgically induced corneal refractive change following glaucoma surgery: nonpenetrating trabecular surgeries versus trabeculectomy. J Cataract Refract Surg. 2004;30:1232–9. doi: 10.1016/j.jcrs.2003.11.055. [DOI] [PubMed] [Google Scholar]
- 195.Corcostegui J, Rebolleda G, Munoz-Negrete FJ. Refractive changes after phacoemulsification combined with deep sclerectomy assisted by corneal topography. J Cataract Refract Surg. 2004;30:2391–6. doi: 10.1016/j.jcrs.2004.05.012. [DOI] [PubMed] [Google Scholar]
- 196.Krasnov MM. Sinusotomy in glaucoma. Vestn Oftalmol. 1964;77:37–41. [PubMed] [Google Scholar]
- 197.Krasnov MM. Externalisation of Schlemm’s canal in glaucoma. Br J Ophthalmol. 1968;52:157–61. doi: 10.1136/bjo.52.2.157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Mendrinos E, Mermoud A, Shaarawy T. Nonpenetrating glaucoma surgery. Surv Ophth. 2008;53(6):592–630. doi: 10.1016/j.survophthal.2008.08.023. [DOI] [PubMed] [Google Scholar]
- 199.Marchini G, Marraffa M, Brunelli C, et al. Ultrasound biomicroscopy and intraocular pressure lowering mechanisms of deep sclerectomy with reticulated hyaluronic acid implant. J Cataract Refract Surg. 2001;27:507–17. doi: 10.1016/s0886-3350(00)00857-9. [DOI] [PubMed] [Google Scholar]
- 200.Mermoud A. Sinusotomy and deep sclerectomy. Eye. 2000;14:531–5. doi: 10.1038/eye.2000.140. [DOI] [PubMed] [Google Scholar]
- 201.Dahan E, Drusedau MU. Nonpenetrating filtration surgery for glaucoma: control by surgery only. J Cataract Refract Surg. 2000;26:695–701. doi: 10.1016/s0886-3350(00)00366-7. [DOI] [PubMed] [Google Scholar]
- 202.Shaarawy T, Mermoud A. Deep sclerectomy in one eye versus deep sclerectomy with implant in the contralateral eye of the same patient: long term follow up. Eye. 2005;19:298–302. doi: 10.1038/sj.eye.6701469. [DOI] [PubMed] [Google Scholar]
- 203.Dahan E, Ravinet E, Ben-Simon GJ, Mermoud A. Comparison of the efficacy and longevity of nonpenetrating glaucoma surgery with and without a new nonabsorbable hydrophilic implant. Ophthalmic Surg Lasers Imaging. 2003;34:457–63. [PubMed] [Google Scholar]
- 204.Shaarawy T, Nguyen C, Schnyder C, Mermoud A. Comparative study between deep sclerectomy with and without collagen implant: long term follow up. Br J Ophthalmol. 2004;88:95–8. doi: 10.1136/bjo.88.1.95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Demailly P, Lavat P, Kretz G, Jeanteur-Lunel MN. Nonpenetrating deep sclerectomy (NPDS) with or without collagen device (CD) in primary open angle glaucoma: middle term retrospective study. Int Ophthalmol. 1996;20:131–40. doi: 10.1007/BF00212959. [DOI] [PubMed] [Google Scholar]
- 206.Lachkar Y, Neverauskiene J, Jeanteur-Lunel MN, et al. Nonpenetrating deep sclerectomy: a 6 year retrospective study. Eur J Ophthalmol. 2004;14:26–36. doi: 10.1177/112067210401400105. [DOI] [PubMed] [Google Scholar]
- 207.Khairy HA, Atta HR, Green FD, et al. Ultrasound biomicroscopy in deep sclerectomy. Eye. 2005;19:555–60. doi: 10.1038/sj.eye.6701558. [DOI] [PubMed] [Google Scholar]
- 208.Hamard P, Lachkar Y. Nonpenetrating filtering surgery, evolution and results. J Fr Ophthalmol. 2002;225:527–36. doi: 10.1097/00055735-200204000-00010. [DOI] [PubMed] [Google Scholar]
- 209.Kozobolis VP, Christodoulakis EV, Tzanakis N, et al. Primary deep sclerectomy versus primary deep sclerectomy with the use of mitomycin C in primary open angle glaucoma. J Glaucoma. 2002;11:287–93. doi: 10.1097/00061198-200208000-00003. [DOI] [PubMed] [Google Scholar]
- 210.Neurdorfer M, Sadetzki S, Anisimova S, Geyer O. Nonpenetrating deep sclerectomy with the use of adjunctive mitomycin C. Ophthalmic Surg Lasers Imaging. 2004;35:6–12. [PubMed] [Google Scholar]
- 211.Anand N, Atherly C. Deep sclerectomy augmented with mitomycin C. Eye. 2005;19:442–50. doi: 10.1038/sj.eye.6701403. [DOI] [PubMed] [Google Scholar]
- 212.Mielke C, Dawda VK, Anand N. Deep sclerectomy and low dose mitomycin C: a randomised prospective trial in West Africa. Br J Ophthalmol. 2006;90:310–3. doi: 10.1136/bjo.2005.079483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.El Sayyad F, Helal M, El-Kholify H, et al. Nonpenetrating deep sclerectomy versus trabeculectomy in bilateral primary open angle glaucoma. Ophthalmology. 2000;107:1671–4. doi: 10.1016/s0161-6420(00)00263-3. [DOI] [PubMed] [Google Scholar]
- 214.Chiselita D. Nonpenetrating deep sclerectomy versus trabeculectomy in primary open angle glaucoma surgery. Eye. 2001;15:197–201. doi: 10.1038/eye.2001.60. [DOI] [PubMed] [Google Scholar]
- 215.Cilino S, Di Pace F, Casuccio A, et al. Deep sclerectomy versus punch trabeculectomy with and without phacoemulsification: a randomised clinical trial. J Glaucoma. 2004;13:500–6. doi: 10.1097/01.ijg.0000137869.18156.81. [DOI] [PubMed] [Google Scholar]
- 216.Ambresin A, Shaarawy T, Mermoud A. Deep sclerectomy with collagen implant in one eye compared with trabeculectomy in the other eye of the same patient. J Glaucoma. 2002;11:214–20. doi: 10.1097/00061198-200206000-00009. [DOI] [PubMed] [Google Scholar]
- 217.Mermoud A, Schnyder CC, Sickenberg M, et al. Comparison of deep sclerectomy with collagen implant and trabeculectomy in open angle glaucoma. J Cataract Refract Surg. 1999;25:323–31. doi: 10.1016/s0886-3350(99)80079-0. [DOI] [PubMed] [Google Scholar]
- 218.Carassa RG, Bettin P, Fiori M, Brancato R. Viscocanalostomy versus trabeculectomy in white adults affected by open angle glaucoma: a 2-year randomised controlled trial. Ophthalmology. 2003;110:882–7. doi: 10.1016/S0161-6420(03)00081-2. [DOI] [PubMed] [Google Scholar]
- 219.Kobayashi H, Kobayashi K. Randomised comparison of the intraocular pressure lowering effect of phacotrabeculectomy. Ophthalmology. 2007;114:909–14. doi: 10.1016/j.ophtha.2006.12.032. [DOI] [PubMed] [Google Scholar]
- 220.O’Brart DP, Rowlands E, Islam N, Noury AM. A randomised prospective study comparing trabeculectomy augmented with antimetabolites with a viscocanalstomy technique for the management of open angle glaucoma uncontrolled by medical therapy. Br J Ophthalmol. 2002;86:748–54. doi: 10.1136/bjo.86.7.748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.O’Brart DP, Shiew M, Edmunds B. A randomised prospective study comparing trabeculectomy with viscocanalostomy with adjunctive antimetabolite usage for the management of open angle glaucoma uncontrolled by medical management. Br J Ophthalmol. 2002;88:1012–7. doi: 10.1136/bjo.2003.037432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Yalvac IS, Sahin M, Eksioglu U, et al. Primary viscocanalostomy versus trabeculectomy for primary open angle glaucoma: three year prospective randomised clinical trial. J Cataract Refract Surg. 2004;30:2050–7. doi: 10.1016/j.jcrs.2004.02.073. [DOI] [PubMed] [Google Scholar]
- 223.Stegmann R, Pienaar A, Miller D. Viscocanalostomy for open angle glaucoma in black African patients. J Cataract Refract Surg. 1999;25:316–22. doi: 10.1016/s0886-3350(99)80078-9. [DOI] [PubMed] [Google Scholar]
- 224.Wild GJ, Kent AR, Peng Q. Dilation of Schlemm’s canal in viscocanalostomy: comparison of 2 viscoelastic substances. J Cataract Refract Surg. 2001;27:1294–7. doi: 10.1016/s0886-3350(01)00867-7. [DOI] [PubMed] [Google Scholar]
- 225.Tamm ER, Carassa RG, Albert DM, et al. Viscocanalostomy in rhesus monkeys. Arch Ophthalmol. 2004;122:1826–38. doi: 10.1001/archopht.122.12.1826. [DOI] [PubMed] [Google Scholar]
- 226.Sunaric-Megevand G, Leuenberger P. Results of viscocanalostomy for primary open angle glaucoma. Am J Ophthalmol. 2001;132:221–8. doi: 10.1016/s0002-9394(01)00972-2. [DOI] [PubMed] [Google Scholar]
- 227.Yarangumeli A, Gurboz Koz O, Alp MN, et al. Viscocanalostomy with mitomycin C: a preliminary study. Eur J Ophthalmol. 2005;15:202–8. doi: 10.1177/112067210501500204. [DOI] [PubMed] [Google Scholar]
- 228.Luke C, Dietlein TS, Jacobi PC, et al. A prospective randomised trial of viscocanalostomy with and without implantation of a reticulated hyaluronic acid implant (SKGEL) in open angle glaucoma. Br J Ophthalmol. 2003;87:599–603. doi: 10.1136/bjo.87.5.599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229.Klink T, Lieb W, Grehn F. erbium-YAG laser assisted preparation of deep sclerectomy. Graefes Arch Exp Ophthalmol. 2000;238:792–6. doi: 10.1007/s004170000169. [DOI] [PubMed] [Google Scholar]
- 230.Francis BA, Singh K, Lin SC. Novel glaucoma procedures: A report by the American Academy of Ophthalmology. Ophthalmology. 2011;118:1466–80. doi: 10.1016/j.ophtha.2011.03.028. [DOI] [PubMed] [Google Scholar]
- 231.Wand M, Gilbert CM, Liesegang TJ. Latanoprost and herpes simplex keratitis. Am J Ophthalmol. 1999;127:602–4. doi: 10.1016/s0002-9394(99)00050-1. [DOI] [PubMed] [Google Scholar]
- 232.Warwar RE, Bullock JD. Latanoprost-induced uveitis. Surv Ophthalmol. 1999;43:466–8. [PubMed] [Google Scholar]
- 233.Wistrand PJ, Stjernschantz J, Olsson K. The incidence and time-course of latanoprost induced iridial pigmentation as a function of eye colour. Surv Ophthalmol. 1997;41(Suppl 2):S129–38. doi: 10.1016/s0039-6257(97)80020-3. [DOI] [PubMed] [Google Scholar]
- 234.Yamamoto T, Kitazawa Y. Iris colour change developed after topical isopropyl unoprostone treatment. J Glaucoma. 1997;6:430–2. [PubMed] [Google Scholar]
- 235.Horsley MB, Chen TC. The use of prostaglandin analogues in the uveitic patient. Semin Ophthalmol. 2011;26(4–5):285–9. doi: 10.3109/08820538.2011.588650. [DOI] [PubMed] [Google Scholar]
- 236.Becker B, Pettit TH, Gay AJ. Topical epinephrine therapy of open angle glaucoma. Arch Ophthalmol. 1961;66(2):219–5. doi: 10.1001/archopht.1961.00960010221012. [DOI] [PubMed] [Google Scholar]
- 237.Robin AL. Short term effects of unilateral 1% apraclonidine therapy. Arch Ophthalmol. 1988;106(7):912–5. doi: 10.1001/archopht.1988.01060140058024. [DOI] [PubMed] [Google Scholar]
- 238.Schuman JS. Clinical experience with brimonidine 0.2% and timolol 0.5% in glaucoma and ocular hypertension. Surv Ophthalmol. 1996;41(Supp1):S27–37. doi: 10.1016/s0039-6257(96)82029-7. [DOI] [PubMed] [Google Scholar]
- 239.Schuman JS, Horowitz B, Choplin NT, et al. A 1 year study of brimonidine twice daily in glaucoma and ocular hypertension: a controlled randomised multicentre clinical trial. Chronic Brimonidine Study Group. Arch Ophthalmol. 1997;115(7):847–52. doi: 10.1001/archopht.1997.01100160017002. [DOI] [PubMed] [Google Scholar]
- 240.Rosenberg LF, Krupin T, Ruderman J, et al. Apraclonidine and anterior segment laser surgery: Comparison of 0.5% vs. 1.0% apraclonidine for prevention of postoperative intraocular pressure rise. Ophthalmology. 1995;102:1312–8. doi: 10.1016/s0161-6420(95)30869-x. [DOI] [PubMed] [Google Scholar]
- 241.Butler P, Mannschreck M, Lin S, Hwang I, Alvarado J. Clinical experience with the longterm use of 1% apraclonidine. Incidence of allergic reactions. Arch Ophthalmol. 1995;113:293–6. doi: 10.1001/archopht.1995.01100030047020. [DOI] [PubMed] [Google Scholar]
- 242.Dailey RA, Brubaker RF, Bourne WM. The effects of timolol maleate and acetazolamide on the rate of aqueous formation in normal human subjects. Am J Ophthalmol. 1982;93(2):232–7. doi: 10.1016/0002-9394(82)90419-6. [DOI] [PubMed] [Google Scholar]
- 243.Granero GE, Longhi MR. Promising complexes of acetazolamide for topical ocular administration. Expert Opin Drug Deliv. 2010;7(8):943–53. doi: 10.1517/17425247.2010.497536. [DOI] [PubMed] [Google Scholar]
- 244.Caprioli J. The Tube versus Trabeculectomy study: Why its findings may not change clinical practice. Am J Ophthalmol. 2011 May;151(5):742–4e1. doi: 10.1016/j.ajo.2011.01.020. [DOI] [PubMed] [Google Scholar]
- 245.Bert RJ, Caruthers SD, Jara H, et al. Demonstration of an anterior diffusional pathway for solutes in the normal human eye with high spatial resolution contrast-enhanced dynamic MR imaging. Invest Ophthalmol Vis Sci. 2006;47:5153–62. doi: 10.1167/iovs.05-0372. [DOI] [PubMed] [Google Scholar]
- 246.Fea AM, Bosone A, Rolle T, Broglialli B, Grignolo FM. Micropulse diode laser trabeculoplasty (MDLT) A Phase II clinical study with 12 months follow up. Clin Ophthalmol. 2008;2(2):247–52. doi: 10.2147/opth.s2303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 247.Wamsley S, Moster MR, Rai S, et al. Results of the use of the ExPRESS miniature glaucoma implant in technically challenging, advanced glaucoma cases: a clinical pilot study. Am J Ophthalmol. 2004;138:1049–51. doi: 10.1016/j.ajo.2004.06.024. [DOI] [PubMed] [Google Scholar]
- 248.Dahan E, Carmichael TR. Implantation of a miniature glaucoma device under a scleral flap. J Glaucoma. 2005;14:98–102. doi: 10.1097/01.ijg.0000151688.34904.b7. [DOI] [PubMed] [Google Scholar]
- 249.Maris PJ, Jr, Ishida K, Netland PA. Comparison of trabeculectomy with Ex-PRESS miniature glaucoma device implanted under scleral flap. J Glaucoma. 2007;16:14–9. doi: 10.1097/01.ijg.0000243479.90403.cd. [DOI] [PubMed] [Google Scholar]
- 250.Traverso CE, De Feo F, Messas-Kaplan A, et al. Long term effect on IOP of a stainless steel glaucoma drainage implant (Ex-PRESS) in combined surgery with phacoemulsification. Br J Ophthalmol. 2005;89:425–9. doi: 10.1136/bjo.2004.058032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 251.Rivier D, Roy S, Mermoud A. Ex-PRESS R-50 miniature glaucoma implant insertion under the conjunctiva combined with cataract extraction. J Cataract Refract Surg. 2007;33:1946–52. doi: 10.1016/j.jcrs.2007.06.069. [DOI] [PubMed] [Google Scholar]
- 252.Dow CT, deVenecia G. Transciliary filtration (Singh filtration) with the Fugo plasma blade. Ann Ophthalmol (Skokie) 2008;40:8–14. [PubMed] [Google Scholar]
- 253.Singh D, Singh K. Transciliary filtration usinf the Fugo Blade. Ann Ophthalmol (Skokie) 2002;34:183–7. [Google Scholar]
- 254.Spiegel D, Wetzel W, Haffner DS, Hill RA. Initial clinical experience with the trabecular micro-bypass stent in patients with glaucoma. Adv Ther. 2007;24:161–70. doi: 10.1007/BF02850004. [DOI] [PubMed] [Google Scholar]
- 255.Spiegel D, Garcia-Feijoo J, Garcia-Sanchez J, Lamielle H. Coexistent primary open angle glaucoma and cataract: preliminary analysis of treatment by cataract surgery and the iStent trabecular micro-bypass stent and concurrent cataract surgery. Adv Ther. 2008;25:453–64. doi: 10.1007/s12325-008-0062-6. [DOI] [PubMed] [Google Scholar]
- 256.Spiegel D, Wetzel W, Neuhann T, et al. Coexistent primary open angle glaucoma and cataract: interim analysis of treatment by cataract surgery and the iStent trabecular micro-bypass stent and concurrent cataract surgery. Eur J Ophthalmol. 2009;19:393–9. doi: 10.1177/112067210901900311. [DOI] [PubMed] [Google Scholar]
- 257.Minckler DS, Baerveldt G, Alfaro MR, Francis BA. Clinical results with the Trabectome for treatment of open-angle glaucoma. Ophthalmology. 2005;112:962–7. doi: 10.1016/j.ophtha.2004.12.043. [DOI] [PubMed] [Google Scholar]
- 258.Minckler DS, Baerveldt G, Ramirez MA, et al. Clinical results with the Trabectome, a novel surgical device for treatment of open angle glaucoma. Trans Am Ophthalmol Soc. 2006;104:40–50. [PMC free article] [PubMed] [Google Scholar]
- 259.Francis VA, Minckler D, Dustin L, et al. Trabectome Study Group. Combined cataract extraction and trabeculotomy by the internal approach for coexisting cataract and open angle glaucoma: initial results. J Cataract Refract Surg. 2008;34:1096–103. doi: 10.1016/j.jcrs.2008.03.032. [DOI] [PubMed] [Google Scholar]
- 260.Singh D, Bundela R, Agarwal A, et al. Goniotomy ab interno “a glaucoma filtering surgery” using the Fugo Plasma Blade. Ann Ophthalmol (Skokie) 2002;34:183–7. doi: 10.1007/s12009-006-0007-0. [DOI] [PubMed] [Google Scholar]
- 261.Wilmsmeyer S, Philippin H, Funk J. Excimer laser trabeculotomy: a new, minimally invasive procedure for patients with glaucoma. Graefes Arch Clin Exp Ophthalmol. 2006;244:670–6. doi: 10.1007/s00417-005-0136-y. [DOI] [PubMed] [Google Scholar]
- 262.Babighian S, Rapizzi E, Galan A. Efficacy and safety of ab interno excimer laser trabeculotomy in primary open angle glaucoma: two years of follow-up. Ophthalmologica. 2006;220:285–90. doi: 10.1159/000094616. [DOI] [PubMed] [Google Scholar]
- 263.Lewis RA, von Wolff K, Tetz M, et al. Canaloplasty: circumferential viscodilatation and tensioning of Schlemm’s canal using a flexible microcatheter for the treatment of open angle glaucoma in adults: interim clinical study analysis. J Cataract Refract Surg. 2007;33:1217–26. doi: 10.1016/j.jcrs.2007.03.051. [DOI] [PubMed] [Google Scholar]
- 264.Lewis RA, von Wolff K, Tetz M, et al. Canaloplasty: circumferential viscodilatation and tensioning of Schlemm’s canal using a flexible microcatheter for the treatment of open angle glaucoma in adults: two year interim clinical study results. J Cataract Refract Surg. 2009;35:814–24. doi: 10.1016/j.jcrs.2009.01.010. [DOI] [PubMed] [Google Scholar]
- 265.Shingleton B, Tetz M, Korber N. Circumferential viscodilation and tensioning of Schlemm canal (canaloplasty) with temporal clear corneal phacoemulsification cataract surgery for open-angle glaucoma and visually significant cataract: one year results. J Cataract Refract Surg. 2008;34:433–40. doi: 10.1016/j.jcrs.2007.11.029. [DOI] [PubMed] [Google Scholar]
- 266.Melamed S, Ben Simon GJ, Goldenfeld M, Simon G. Efficacy and safety of gold micro shunt implantation to the supraciliary space in patients with glaucoma: a pilot study. Arch Ophthalmol. 2009;127:264–9. doi: 10.1001/archophthalmol.2008.611. [DOI] [PubMed] [Google Scholar]
- 267.Figus M, Lazzeri S, Fogagnolo P, Lester M, Martinelli P, Nardi M. Supraciliary shunts in refractory glaucoma. Br J Ophthalmol. 2011;95:1537–41. doi: 10.1136/bjophthalmol-2011-300308. [DOI] [PubMed] [Google Scholar]
- 268.Agnifili L, Costagliola C, Figus M, et al. Histological findings of failed gold micro shunts in primary open angle glaucoma. Graefes Arch Clin Exp Ophthalmol. 2011 Aug 23; doi: 10.1007/s00417-11-1778-6. epublication ahead of print. [DOI] [PubMed] [Google Scholar]