Abstract
The t(12;21) translocation which generates the ETV6-RUNX1 (TEL-AML1) fusion gene, is the most common chromosomal rearrangement in childhood cancer and is exclusively associated with B-cell precursor acute lymphoblastic leukemia (BCP-ALL). The translocation arises in utero and is necessary but insufficient for the development of leukemia. SNP array analysis of ETV6-RUNX1 patient samples have identified multiple additional genetic alterations, however the role of these lesions in leukemogenesis remains undetermined. Moreover, murine models of ETV6-RUNX1 ALL that faithfully recapitulate the human disease are lacking. To identify novel genes that co-operate with ETV6-RUNX1 in leukemogenesis, we generated a mouse model that uses the endogenous Etv6 locus to co-express the ETV6-RUNX1 fusion and Sleeping Beauty (SB) transposase. An insertional mutagenesis screen was performed by intercrossing these mice with those carrying a SB transposon array. In contrast to previous models, a substantial proportion (20%) of the offspring developed BCP-ALL. Isolation of the transposon insertion sites identified genes known to be associated with BCP-ALL, including Ebf1 and Epor, in addition to other novel candidates. This is the first mouse model of ETV6-RUNX1 to develop BCP-ALL and provides important insights into the cooperating genetic alterations in ETV6-RUNX1 leukemia.
Keywords: ETV6-RUNX1, leukemia, precursor-B cell, insertional mutagenesis
Introduction
The ETV6-RUNX1 fusion gene generated by the t(12;21)(p13;q22) chromosomal translocation,1 is the most prevalent fusion gene in childhood acute lymphoblastic leukemias (ALL), the most common malignancy of childhood. It occurs in around 20% of cases, and is almost exclusively associated with the common B-cell precursor subset of ALL (also known as common ALL, cALL).2 The fusion gene arises during fetal hematopoiesis in a B-cell precursor,3 giving rise to a preleukemic cALL-propagating cell that was recently identified by the surface phenotype CD34+CD38−/lowCD19+.4-5 However, the frequency of individuals carrying the ETV6-RUNX1 fusion gene at birth considerably exceeds the number of patients presenting with clinically overt ALL,6 and twin studies and retrospective analysis of neonatal blood spots from ALL patients indicate that ETV6-RUNX1-expressing fetal clones expand and can persist in a clinically covert state for more than a decade.5 Taken together, this indicates the requirement for additional secondary (postnatal) genetic events (‘hits’) for the development of ALL.
ETV6-RUNX1+ ALL at diagnosis is characterized by multiple copy number alterations (4-7 per case). These commonly include deletions in genes regulating B cell differentiation or cell cycling7 and studies on identical twins with concordant ALL indicate that these genetic events are secondary to ETV6-RUNX1 fusion, and probably occur post-natally.8 In agreement with these clinical observations, animal models of ETV6-RUNX1 in both mice and zebrafish have shown that expression of the ETV6-RUNX1 fusion alone is insufficient for leukemogenesis, yet similar to the RUNX1-RUNX1T1 fusion,9 leukemia may occur following the acquisition of co-operating mutations.10-14 However, these models of ETV6-RUNX1 leukemia have not been suitable for the identification of co-operating mutations for two reasons. First, they do not accurately recapitulate the precursor B-cell phenotype associated with expression of the ETV6-RUNX1 fusion. Secondly, the models either utilize mutations already known to co-occur in ETV6-RUNX1 expressing ALL (such as deletion of the CDKN2A [p16] and p19 genes10) which provide no further pathogenetic information, or use agents such as N-ethyl-N-nitrosourea to induce secondary mutations, that are difficult to identify. To overcome these limitations, we have developed a new mouse model of ETV6-RUNX1 ALL, in which expression of the fusion gene is driven from the endogenous Etv6 promoter, and is linked to expression of the Sleeping Beauty (SB) transposase. This not only allows for expression of the fusion gene at endogenous levels, but also recapitulates expression of the fusion gene in the pattern of endogenous Etv6, and allows forward mutagenesis to occur in the same cellular compartment.
Our model demonstrates that in the presence of mutations induced by the SB transposon, mice carrying the ETV6-RUNX1 allele can develop B-cell precursor ALL (BCP-ALL). Furthermore, transposon insertions can be used to identify gene mutations that co-operate with ETV6-RUNX1 in leukemogenesis. This model therefore represents a unique tool for both studying the biology of this common disease and for identifying mutations that mediate development of ALL in cooperation with ETV6-RUNX1.
Materials and methods
Generation of ETV6-RUNX1 mice
A 10.1 kb fragment of 129S5/SvEVBrd genomic DNA (NCBI m37: Chr6; 134200205-134210356) from BAC clone bMQ-66f22 was captured by recombineering into pBluescript. This fragment contained most of intron 5-6, based on the longest protein-coding transcript of Etv6 (Ensembl ID: ENSMUST00000081028). Into the captured genomic fragment a cassette was inserted containing: a splice acceptor, exons 1-6 of human RUNX1, an EMCV IRES, a hyperactive variant of the SB transposase (HSB5),15 and an FRT-flanked PGK-PuroΔ TK drug selection marker.16 This entire cassette was synthesized by GENEART (GENEART AG, Regensburg, Germany) to ensure fidelity, and was flanked by Lox66/Lox71 sites (to allow Cre-mediated inactivation of the ETV6-RUNX1 fusion gene) and HpaI sites (to allow cloning into the two StuI sites of the captured genomic fragment). The final targeting construct was termed pAML-IRES-SB-puro. Targeting was performed in E14Tg2a ES cells (129P2Ola/Hsd) and correctly targeted clones identified by southern blotting on StuI digested gDNA using a 5′ Probe A (generated by PCR on gDNA using FOR: 5′-GGG CAC CTA CGA TGC ACA GAT AAA TAC ATC CGC-3′ and REV: 5′-GAG ATA GGG TCA GTC TGG AAC TGG TCG CAG GGA-3′) and 3′ Probe B (generated by PCR on gDNA using FOR 5′-TCC TCC TTG GTC CTC ATA TAA AGG TGG TA-3′ and REV: 5′-TGA GTT CTG CTG AGT TAA TGG GCC CCG GA-3′). Targeted ES cells were injected into C57BL/6J blastocysts to generate chimaeras, which were bred to 129S5/SvEvBrd or C57BL/6J mice for germline transmission of the allele (Etv6+/RUNX1). Mice were housed in accordance with Home Office regulations (UK). ES cell targeting of the RUNX1-Flag was performed in the same way (Etv6+/RUNX1-Flag).
Immunoprecipitation of FLAG tagged proteins and Western blotting
Flag-tagged proteins were immunoprecipitated from whole-cell ES cell lysates using Dynabead protein G (Invitrogen, Carlsbad, CA) according to the manufacturer’s instructions and Western blot analysis performed using monoclonal anti-Flag M2 antibody (Sigma-Aldrich, St. Louis, MO). Western blot was performed on whole-cell mouse bone marrow lysates using anti-SB transposase antibody (R&D Systems, Minneapolis, MN) and actin (C-11) polyclonal antibody (Santa Cruz Biotechnology, Santa Cruz, CA). The ETV6-RUNX1-Flag expression plasmid,17 which was used as a positive control for immunoprecipitation and Western blotting, was kindly provided by Dr. O Williams, University College London.
Quantitative PCR
Total RNA from mouse tissue (spleen, thymus and bone marrow) was isolated using TRIzol reagent (Invitrogen) and cDNA reverse transcribed using SuperScript First-Strand RT-PCR kit (Invitrogen), according to the manufacturer’s instructions. Quantitative PCR (qPCR) was performed using ABsolute™ qPCR ROX Mix kit on an ABI PRISM 7900HT sequence detection system (Applied Biosystems, Carlsbad, CA). qPCR probes with 5′ FAM and 3′ TAMRA modifications (MWG Operon, Ebersberg, Germany) were as follows: Etv6 probe: 5′-CAC GCC ATG CCC ATT GGG AGA A-3′ (FWD primer: 5′-TCT CTA TGT CCC CAC CGG AAG-3′; REV primer: 5′-CAT AAT CCC AAA GCA GTC TAC AGT CT-3′), Etv6-RUNX1 probe: 5′-AGC ACG CCA TGC CCA TTG GG-3′ (FWD primer: 5′-CTT GAA CCA CAT CAT GGT CTC TAT G-3′; REV primer: 5′-TCG TGC TGG CAT CTG CTAT T-3′), and β-Actin probe: 5′-TTT GAG ACC TTC AAC ACC CCA GCC A-3′ (FWD primer: 5′-CGT GAA AAG ATG ACC CAG ATC A-3′ and REV primer: 5′-CAC AGC CTG GAT GGC TAC GT-3′).
Embryonic lethality and leukemogenesis studies
Embryonic lethality studies were performed by timed matings of Etv6+/RUNX1 mice and the embryos collected at day 10.5 of gestation. The embryos were genotyped by PCR using primers to detect the wildtype Etv6 allele (FOR 5′-AGG CAT TGT GCA AAG AAT GAG AGA C-3′ and REV 5′-GAC CAA CCA AAC CAA ACA AAC AAA A-3′; with a 65°C annealing temperature to produce a 360 bp band) and the Etv6+/RUNX1 allele (FOR 5′-AAG AAG CCA CTG CTC CAA AA-3′ and REV 5′-GCA CTT GCT CTC CCA AAG TC-3′; with a 60°C annealing temperature to produce a 376 bp band). Mice carrying the SB transposon array (T2Onc)18 were kindly provided by Lara Collier (University of Wisconsin). Genotyping for the T2Onc allele and the excision PCR was performed as described previously.16 Mice carrying the Etv6+/RUNX1 and T2Onc+/Tg alleles were intercrossed and their offspring placed on tumor watch. These mice were examined twice daily for signs of disease, at which time they were sacrificed and a full necropsy performed.
Histology and immunohistochemistry
Tissues and tumors were fixed in 10% neutral-buffered formalin (NBF) at room temperature overnight. Samples were then transferred to 50% ethanol, embedded in paraffin, sectioned and stained with hematoxylin and eosin (H&E). For samples where no bone marrow was available for flow cytometric analysis of CD antigen expression, immunophenotyping was performed by immunohistochemical on formalin-fixed, paraffin-embedded tissue sections that had undergone antigen retrieval (microwaving in citrate buffer pH 6 for 20 min) using antibodies for CD3 (clone SP7; Abcam, Cambridge, UK), B220 (clone RA3-6B2, R&D systems) and MPO (DAKO, Ely, UK). Immunohistochemical signal was detected by secondary biotinylated goat anti-rabbit antibody (Vector Laboratories, Burlingame, CA), followed by Vectorstain Elite ABC kit (Vector Laboratories) according to the manufacturer’s instructions.
Flow cytometric analysis
For analysis of CD antigen expression, single-cell suspensions from spleen or bone marrow were stained with combinations of Mac-1-FITC, Gr-1-PE/Cy7, Ter-119-PE, B220-AlexaFluor 647, CD4-PE/Cy7, CD19-PE, B220-PE/Cy7, CD43-PE, IgM-FITC, IgD-AlexaFluor 647, c-Kit-PE, AA4.1-PE, CD24-FITC, BP1-PE and IL7Ra-PE/Cy7 anti-mouse antibodies, obtained from BioLegend (San Diego, CA), according to the supplier’s recommendations. FACS analysis was performed with a Cyan ADP Analyser (Beckman Coulter, High Wycombe UK). For the IgH rearrangement analysis, cells were sorted on a MoFlo Cell Sorter (Beckman Coulter).
IgH rearrangement analysis
PCR analysis was performed using Phusion High Fidelity DNA polymerase (Finnzymes, Thermo Fisher Scientific, Surrey, UK), amplifying from upstream of DQ52 (5′DQ52 FOR: TTG GGT CAC TTT CCT GCT GT) to JH4 (J4 REV: AGA CC TGG AGA GGC CAT TCT), or from upstream of DFL/DSP genes (5′D FOR: GCA TGT CTC AAA GCA CAA TG) to JH4 (J4 REV2: CTG AGG AGA CGG TGA CTG AG). PCR conditions used a 65°C annealing temperature and 90 sec extension time for 40 cycles. PCR products were purified (Qiagen Gel Extraction Kit, Qiagen, West Sussex, UK), cloned into pGEM-T Easy plasmid (Promega, Madison, WI), transformed into subcloning efficiency DH5α chemically competent cells (Invitrogen) and clones sequenced using either the appropriate D primer or a primer in the vector. For some samples it was necessary to perform whole genome amplification prior to IgH rearrangement analysis and this was performed using the GenomiPhi DNA Amplification Kit (GE Healthcare, Chalfont St Giles, UK), according to the manufacturer’s instructions.
Whole genome expression profiling
Total RNA extracted from the spleens of 15 ETV6-RUNX1 BCP-ALL cases and 3 ETV6-RUNX1 non-diseased mice using TRIzol, according to the manufacturer’s instructions. Expression profiling of the RNA was performed using a MouseWG-6 v2.0 expression beadchip kit (Illumina, Essex UK), according to the manufacturer’s instructions. Data were quantile normalised19 and analyzed using the bioconductor packages limma and lumi [http://www.bioconductor.org/]. Data were P-value adjusted to yield a sorted list of differentially expressed genes.20 Gene annotations were further refined using ReMOAT [http://remoat.sysbiol.cam.ac.uk/]. Gene set enrichment analysis (GSEA) was performed on the differentially expressed genes using Ingenuity Pathway Analysis (IPA) software (Ingenuity Systems, Redwood City, CA).
Isolation & statistical analysis of transposon insertion sites
Isolation of the transposon insertion sites from the leukemias was performed using splinkerette PCR to produce barcoded PCR products that were pooled and sequenced as described previously.21 The pooled PCRs were sequenced on 454 GS-FLX sequencers (Roche) platform) over four separate lanes, with one lane per restriction enzyme and a maximum of 48 leukemias per lane. Processing of 454 reads, identification of insertion sites, and Gaussian kernel convolution statistical methods used to identify common insertion sites (CIS) have been described previously.21 The P-value for each CIS was adjusted by chromosome and a cut-off of P<0.05 was used. Any CIS on mouse chromosome 1 are not reported as this is the ‘donor chromosome’ where the transposon array is located. Data from this chromosome is automatically excluded from analysis due to the phenomenon of ‘local hopping’.18 The insertion sites have been made publically available by submission to both the Retrovirus Tagged Cancer Gene Database (RTCGD; http://rtcgd.ncifcrf.gov/) and the Insertional Mutagenesis Database (http://imdb.nki.nl/).
Results
Generation of ETV6-RUNX1 mice
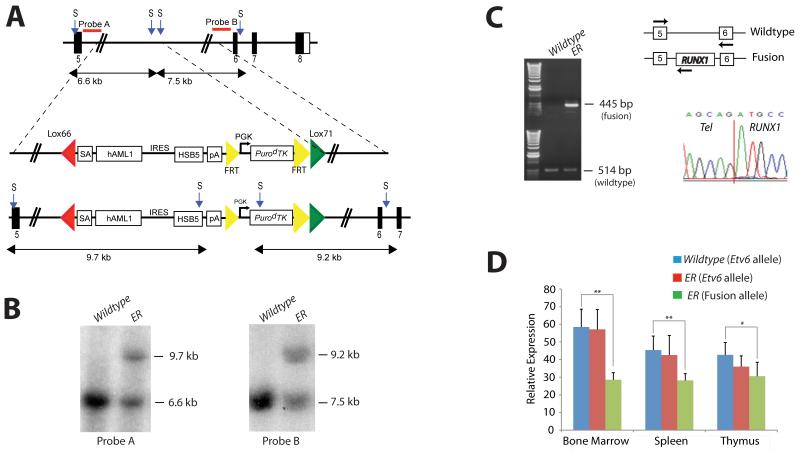
The endogenous Etv6 locus was targeted to introduce a Lox66/Lox71-flanked cassette containing a splice acceptor (SA), exons 1-6 of human RUNX1, an IRES, a hyperactive variant of the Sleeping Beauty (SB) transposase (HSB5), and an FRT flanked PGK-PuroΔ TK drug selection marker into Etv6 intron 5, as shown in Figure 1A. The targeting vector was introduced into ES cells and correctly targeted clones, Etv6+/RUNX1 (ER), were identified by Southern blotting using a 5′ and 3′ external probe on StuI-digested genomic DNA (Figure 1B). These ES cell clones were used to generate chimeric mice that transmitted the mutated allele through the germline. All offspring were genotyped by PCR to detect the presence of the transposase. The PuroΔ TK drug marker was removed by breeding the mice to a Flpe-deleter strain (FLPeR mice)22 prior to tumor watch experiments.
Figure 1. Generation of the Etv6-RUNX1 transposase knockin allele (Etv6+/RUNX1).
A. The endogenous Etv6 locus was targeted to introduce a splice acceptor (SA), exons 1-6 of human RUNX1, an IRES followed by a hyperactive variant of the Sleeping Beauty transposase (HSB5), and an FRT-flanked PGK-PurodTK drug selection marker between exons 5 and 6. The entire cassette was flanked by Lox66 and Lox71 sites for potential Cre-mediated inactivation of the Etv6-RUNX1 fusion gene. The PurodTK drug marker was removed by breeding mice to a Flpe deleter strain prior to tumor watch experiments. Probe A (5′) and Probe B (3′) are shown. S, StuI. B. Southern blot analysis on StuI digested tail DNA confirmed germline transmission and homologous recombination of the 5′ and 3′ targeting vector arms. C. RT-PCR showing expression of Etv6-RUNX1 fusion transcripts in bone marrow from Etv6+/RUNX1 mice, using primers located as indicated by the arrows on the schematic of the Etv6 locus. Lower panel shows a sequence trace showing splicing of the Etv6 and RUNX1 transcripts to produce an in-frame fusion transcript. D. qPCR of Etv6 and Etv6-RUNX1 fusion transcripts (using primers as mentioned above). RNA was extracted from bone marrow, spleen and thymus. Relative Etv6 gene expression in wildtype (blue) and Etv6+/RUNX1 tissues (red), and relative Etv6-RUNX1 fusion transcript expression in Etv6+/RUNX1 tissues (green) is shown. The data was collected from the analysis of 5 littermate mice of each genotype (±SD). ** P<0.005, * P<0.05.
RT-PCR performed on ER bone marrow cDNA confirmed the presence of the fusion transcript (not seen in Etv6+/+ (wildtype) bone marrow cDNA; Figure 1C). Sequencing the RT-PCR product confirmed faithful splicing of the Etv6 and RUNX1 transcripts to produce an in-frame fusion transcript (Figure 1C). Quantitative-RT-PCR performed on RNA extracted from bone marrow, spleen and thymus showed that the relative expression level of the wildtype Etv6 gene to be comparable between wildtype and ER mice, whereas the relative expression level of the ETV6-RUNX1 fusion transcript was less than that of the wildtype Etv6 transcript in ER tissues (Figure 1D). Immunoprecipitation followed by Western blot analysis confirmed the presence of the fusion protein (Figure S1).
ETV6-RUNX1 in embryonic development
Mice heterozygous for the ETV6-RUNX1 allele (EV) were grossly normal at birth and fertile (data not shown). However, homozygosity resulted in embryonic lethality at day E10.5 (Figure S2A), similar to that seen in homozygous null Etv6 mice (Etv6−/−).23 This result confirms that our ETV6-RUNX1 allele does not produce a functional ETV6 transcript. This is important when modeling human leukemia, as loss of the second ETV6 allele is one of the most common genetic alterations in ETV6-RUNX1 ALL patients.7, 24
These studies were performed using ER mice on a mixed genetic background (129;C57), and intercrossing of ER mice resulted in 54 of the 94 (57%) offspring being TA, which is within normal Mendelian ratios for homozygous lethal alleles (P=0.2285; Chi-squared test, 2-tailed). This suggests that ETV6-RUNX1 does not have a dominant negative function over the remaining fusion partner alleles during embryogenesis, unlike the RUNX1-RUNX1T1 fusion.9 However, when the proportion of C57 genetic background was increased by breeding, ER mice were produced at sub-Mendelian ratios (Figure S2B) – a phenotype similar to that observed in a recently described conditional knockin ETV6-RUNX1 mouse generated on a C57 background,14 suggesting the C57 strain may contain a genetic modifier of the function of ETV6-RUNX1 in embryonic development.
ETV6-RUNX1 expression predisposes to hematologic malignancies
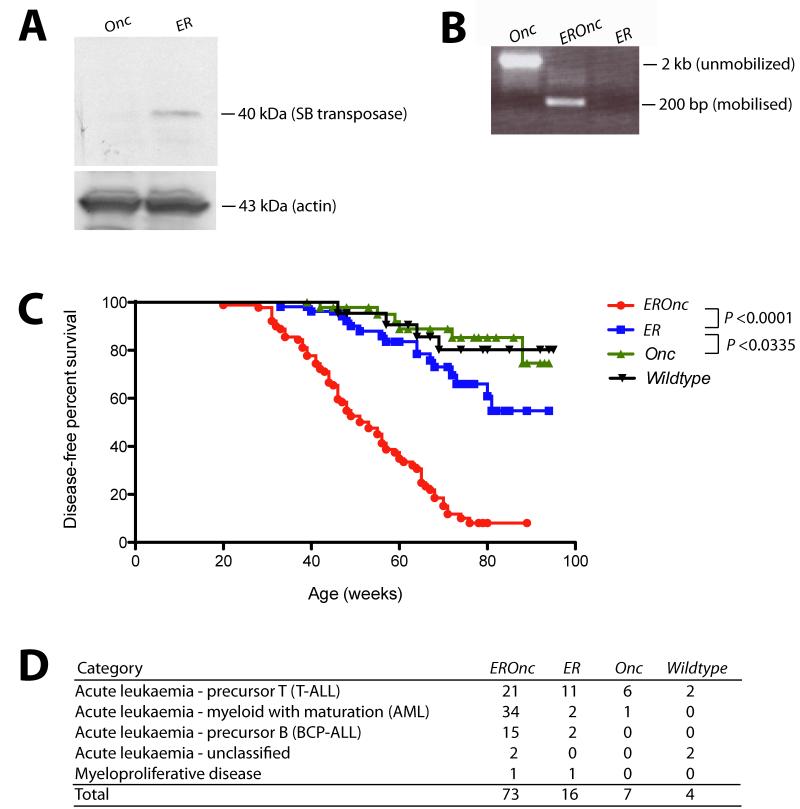
Consistent with previous reports that ETV6-RUNX1 expression does not grossly affect hematopoiesis in the post-natal period,14 we did not observe significant gross perturbation of hematopoietic homeostasis in ETV6-RUNX1 mice (as assessed by immunophenotyping of bone marrow at 3, 6 and 12 months of age; Figure S3). To model the ‘second hit’ event(s) that occurs in ETV6-RUNX1 patients facilitating the development of overt leukemia, we intercrossed ETV6-RUNX1 (Etv6+/RUNX1; ER) mice with mice carrying the T2Onc transposon array (T2Onc+/Tg).18 To confirm that the SB system was functioning in these mice we firstly analyzed bone marrow cell lysates for the presence of SB transposase protein. Western blotting of these lysates using an anti-SB transposase antibody showed the presence of a 40 kDa band in mice carrying the Etv6-RUNX1 allele, consistent with the molecular weight of the SB transposase (Figure 2A). Secondly, we performed ‘excision PCR’ on genomic tail DNA from the offspring (Figure 2B). A small PCR product (~200 bp) is only present in mice carrying both the transposon and transposase (Etv6+/RUNX1; T2Onc+/Tg, hereafter referred to as EROnc mice), indicative of ‘jumping’ of single transposons out of the array (into the genome) whereas mice carrying the transposon array alone (Etv6+/+; T2Onc+/Tg, hereafter referred to as Onc mice) show only a large PCR product (~2 kb) representing the transposon concatamer.
Figure 2. Activation of transposon mutagenesis in Etv6+/RUNX1 mice.
A. Western blot showing expression of the Sleeping Beauty transposase in the bone marrow of Etv6+/RUNX1 (ER) mice, but not mice carrying the T2Onc transposon (Onc). B. ‘Excision’ PCR showing mobilization of the transposon in bone marrow from Etv6+/RUNX1; T2Onc+/Tg (EROnc) mice, as evidenced by the 200 bp ‘excision’ band (instead of the 2 kb band seen in mice carrying the unmobilized transposon array, i.e., Etv6+/+; T2Onc+/Tg (Onc) mice). C. Kaplan-Meier curves showing the tumor latency of ‘jumping’ Etv6+/RUNX1; T2Onc+/Tg (EROnc) mice and ‘non-jumping’ control mice (ER, Onc and wildtype). D. Categorization of the malignancies developed by mice shown in the Kaplan-Meier curve according to the Bethesda criteria for lymphoid and non-lymphoid murine malignancies.25-26
Mice of all four genotypes that arose from the intercrossing of Etv6+/RUNX1 mice with T2Onc+/Tg mice: EROnc (n=90), Etv6+/RUNX1; T2Onc+/+ (hereafter refereed to as ER; n=54), Onc (n=50), and Etv6+/+; T2Onc+/+ (hereafter referred to as wildtype; n=22) were placed on tumor watch, and observed twice daily for signs of illness. Although mice of all genotypes developed hematological malignancies, EROnc mice showed a statistically significant increased incidence of leukemia and decreased survival compared with the other genotypes (P<0.0001; Figure 2C). ER mice (which carry the Etv6RUNX1 allele without the T2Onc transposon allele, hence no mutagenesis) also showed decreased survival compared to Onc mice carrying just the T2Onc allele (i.e., no Etv6-RUNX1 or transposase expression hence no mutagenesis).
This decreased survival in both cohorts was due to the development of hematological malignancies, which were classified according to the Bethesda criteria for murine malignancies using standard morphological, histological and immunophenotypic analysis (Figure 2D).25-26 Occasional mice (11/72 mice, 14%) in the Onc and WT cohorts presented with long latency predominantly T-cell acute leukemias. However the incidence of leukemia was significantly increased in the cohorts expressing Etv6-RUNX1 (73/90 mice, 81% for the EROnc cohort and 16/54, 29% for ER). The presence of Etv6-RUNX1 expression not only increased the incidence of acute leukemias, but also modulated the phenotype. In the EROnc cohort, acute myeloid leukemia (AML) predominated (34/73 cases, 46%), with T-cell ALL also seen (21/73, 28%). T-cell ALL predominated in the ER mice (11/16 cases, 68%), with an occasional case of AML seen. Importantly, 15 of 73 (21%) of EROnc mice developed B-cell precursor (BCP-ALL) as did a slightly smaller proportion of the ER cohort (2/16 cases, 13%). Most importantly, BCP-ALL was only seen in mice carrying the ETV6-RUNX1 allele. This is the only mouse model of ETV6-RUNX1 to develop BCP-ALL, either through spontaneous or mutagenesis-driven acquisition of ‘second hits’.
ETV6-RUNX1-expressing mice develop a B-cell precursor acute lymphoblastic leukemia
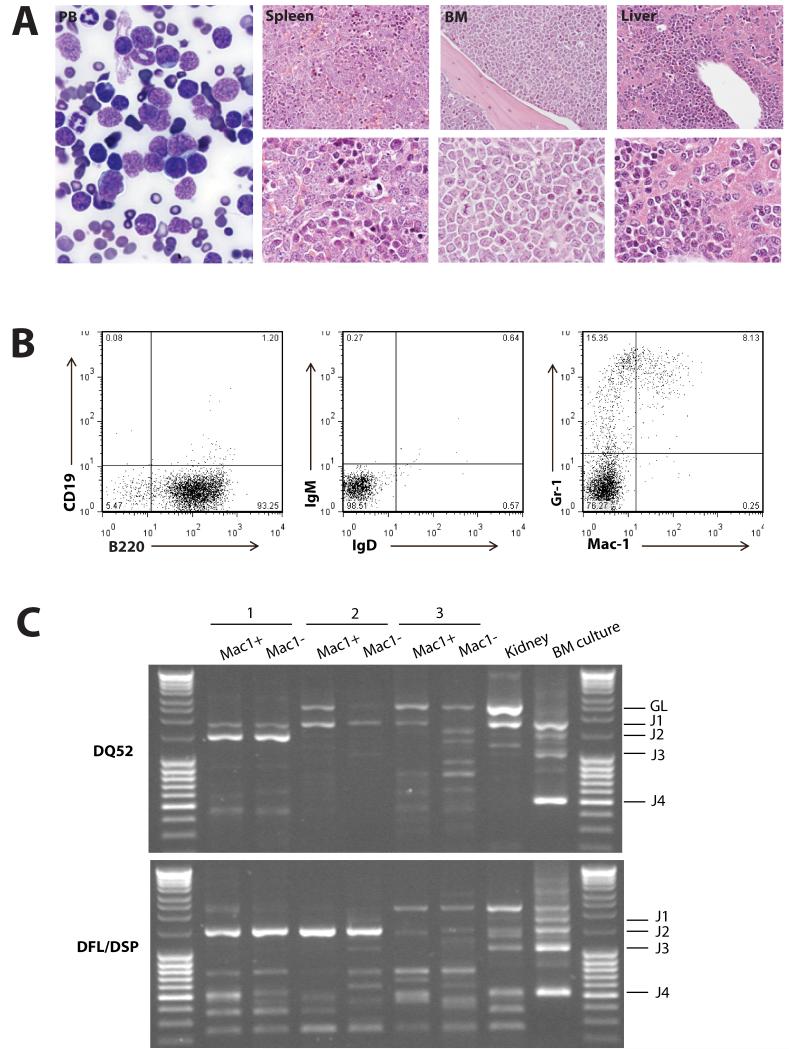
Mice with the BCP-ALL phenotype demonstrated the obvious presence of lymphoblasts in the peripheral blood and bone marrow, and heavy infiltration of lymphoblasts in the spleen and liver, resulting in effacement of the normal cellular architecture and replacement with predominantly nucleolated blasts (Figure 3A). Immunophenotypic analysis of the bone marrow by FACS from the 17 cases of BCP-ALL showed the majority of tumors to be of early B cell progenitor immunophenotype (B220+ CD19−, IgM−, IgD−) (Figure 3B and S4). ETV6-RUNX1 patients typically have a surface phenotype reminiscent of pre-B cells (equivalent to Hardy Fraction C).27 More detailed immunophenotyping of a subset of 7 cases predominantly showed a phenotypic profile (AA4.1+, CD24+, CD43+, BP1−, IL7Ra−; Figure S4) consistent with early pro-B cells (Hardy Fraction B) suggesting transformation of a slightly earlier lymphoid progenitor. In addition, a number of cases also showed co-expression of myeloid (Mac-1+) markers on lymphoblasts (Figure S4). Co-expression of myeloid antigens (particularly CD13 and CD33), is well described for patients with ETV6-RUNX1 BCP-ALL.27
Figure 3. Etv6+/RUNX1 mice with ‘second hits’ develop a pre-B acute lymphoblastic leukemia recapitulating features of the human disease.
A. Peripheral blood (PB; ×2000 magnification), spleen, bone marrow and liver (×400 magnification in upper panels and ×1000 magnification in lower panels) from a representative mouse with BCP-ALL. The presence of lymphoblasts in the peripheral blood is obvious, as is the infiltration of the spleen, bone marrow and liver (2nd-4th panels), with effacement of the normal cellular architecture and replacement by nucleolated blasts. B. FACS plots from the bone marrow of a representative mouse demonstrate only background Gr-1/Mac1 myeloid cells, with the majority of cells having a B220+/CD19−/sIg- phenotype in keeping with BCP-ALL. C. D-J PCR rearrangement studies (involving DQ52 and DFL/DSP genes) were performed on spleen or bone marrow gDNA of three mice with phenotypic B-ALL. Negative control was gDNA from C57BL/6J mouse kidney (known to be unrearranged), and positive control was gDNA from a C57BL/6J primary pro-B cell bone marrow culture (known to contain a lot of DJ-rearranged alleles). Sample 1 shows two different rearrangement events (DQ52-J2 and DFL/DSP-J2 rearrangements), in keeping with a clonal B-cell population with both alleles D-J recombined. Sample 2 shows both a germline band and an identical DFL/DSP-J2 band, in keeping with a clonal B-cell population with rearrangement of one allele and germline configuration of the other. Sample 3 shows an absence of recombination.
To further demonstrate clonality and determine the differentiation stage of the tumor the VDJH recombination status of three BCP-ALLs (B220+/CD19−/sIg− phenotype) was analyzed. Clonal DJH rearrangements were detected in two samples, however, both alleles were in germline configuration in the third (Figure 3C). Thus, taking into account the results of the IgH recombination and immunophenotyping analysis, these murine leukemias are B-cell precursor in origin, however, show an earlier arrest in B-cell ontogeny than typically found in ETV6-RUNX1 BCP-ALL patients (CD10+/CD19+ and evidence of VDJH recombination).
Gene expression analysis to look at transcripts differentially expressed between BCP-ALL spleen and normal splenic tissue (EROnc mice with no disease, n=3), found statistically significant differential expression (typically down-regulation) of 125 genes (P<0.05; Table S1), several of which are known to be important for transition through pro-B and pre-B cell stages of development (including Blk, Blnk, CD19, Ebf1, Foxo1, Irf4, Spib, Vpreb3i and Zhx2.) Gene set enrichment analysis (GSEA) performed on these differentially expressed genes found the top 5 canonical pathways involved B-cell signalling or development (Table S2). There was also decreased expression of the chemokine receptors Ccr6 (CD196) and Cxcr5 (CD185), reported to be down-regulated in BCP-ALL patients.28 Importantly, we also found decreased expression in genes found in deleted regions of human ETV6-RUNX1 patients (including Ebf1, Blnk, and Btg)7, 24 and decreased expression of Ms4a1 (CD20), a B-lymphocyte surface antigen reported to be of lower expression in t(12;21)+ than t(12;21)− BCP-ALL cases.29 Thus the molecular profiling of these leukemias in part mirrors human ETV6-RUNX1 positive BCP-ALL.
Identification of co-operating mutations in ETV6-RUNX1-expressing mice using insertional mutagenesis
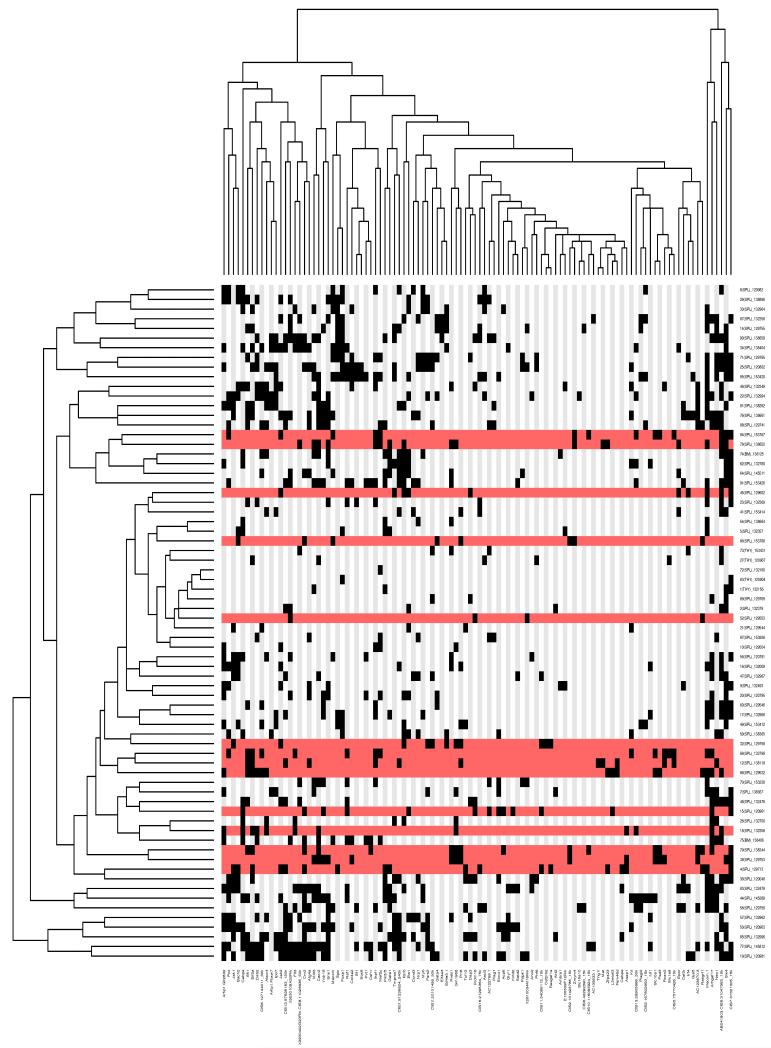
Genomic DNA extracted from leukemic tissue of 73 Etv6+/RUNX1; T2Onc+/Tg (EROnc) mice was used in a splinkerette PCR reaction to produce barcoded PCR products that were subsequently pooled and directly sequenced on the 454 GS-FLX platform.21 This generated 695,504 sequence reads, of which 51.5% unambiguously aligned to the mouse genome (see Figure S5). Using a previously developed computational pipeline to trim, map, and annotate each sequence read,21 we were able to identify 23,529 unique (non-redundant) insertion sites. We used the Gaussian kernel convolution (GKC) algorithm to determine statistically significant common insertion sites (CIS; genomic regions with a higher density of insertion sites than expected by chance),30 and CIS were assigned to genes as described previously.21 In total, 71 leukemias (34 AML, 15 BCP-ALL, 19 T-ALL, 2 unclassified and 1 myeloproliferative) contributed to generate a total of 101 unique GKC CIS regions/genes, with an average of 15 ± 7 CIS/leukemia (listed in Figure 4 and Table S3). To identify potential co-occurring CIS and common patterns of mutation between each of the leukemias, a matrix of the leukemias against the unique CIS regions was constructed and hierarchical clustering performed to aggregate those with similar patterns of insertions (Figure 4). Interestingly, many of the CIS that were found in the BCP-ALLs were also found in the AML and T-ALLs (see Figure S6), suggesting that these CIS/genes may predispose to tumorigenesis in multiple hematopoietic lineages, or may affect HSC function before individual lineages are specified. However, the possibility cannot be excluded that since many of our BCP-ALLs also showed a population of cells with aberrant expression of myeloid markers (Mac-1 and Gr-1; Figures 3B and S4), mutation of genes involved in the myeloid pathway would be expected or that these tumors were polyclonal and contained a separate myeloid clone.
Figure 4. Hierarchical clustering of the leukemias and their mutated genes based on common insertion sites.
A matrix of 71 leukemias against the 102 common insertion site (CIS) genes identified by analysis of transposon insertions by the Gaussian kernel convolution method was constructed. Although 73 leukemias were subjected to LM-PCR, sample 120992 did not have any insertions that were mapped and sample 138641 had only 44 insertions sites that were mapped but none of these contributed to any CIS, thus these two samples do not appear in the heatmap as the heatmap is based on the co-occurrence of insertions in CIS. Hierarchical clustering was performed on a binarized version of the matrix using the Hamming distance metric along both the rows and columns, to aggregate leukemias and CIS genes with similar insertions patterns. The two resulting dendrograms were visualized as a heatmap, where black squares indicate the presence of at least one insertion in the tumor, and grey no insertion site. Leukemia rows highlighted in red indicate a BCP-ALL phenotype (n=15; only 14 rows highlighted in red because no insertions were found in the called CIS for sample 138895). The clustering and visualization were performed using the R programming language (version 2.10.0) and Bioconductor (version 2.5).
Restricting our analysis to the BCP-ALL cases (Table 1 and Figure S7), the most frequently targeted genes/loci were an intragenic region on mouse chromosome 7 (CIS peak location at 37320759), translation initiation factor 2 (Eif2a), the erythropoietin receptor (Epor), Ikaros family zinc finger 1 (Ikzf1), CCAAT/enhancer binding protein, alpha (Cebpa) and the cannabinoid receptor 2 (Cnr2). In some cases the transposons had inserted themselves in the forward orientation and were clustered in the non-coding introns upstream of the ATG start site, suggesting they act as activating mutations to induce over-expression of the gene (such as Raf1 and Mef2c). In other cases, the transposons had inserted themselves in both the forward and reverse orientations and were scattered throughout the gene, suggesting they act as inactivating mutations, to produce a truncated transcript (such as Ikzf1). Although some of these genes/loci were also found as CIS in AML and T-ALL cases, 66% (22/33) of these CIS were exclusive to the BCP-ALL cases (including Eif2a and Ikzf1). Intriguingly, these B-cell specific genes did not appear to cluster together (Figure 4), suggesting that there is significant heterogeneity in the genes that can contribute to the development of acute lymphoblastic leukemia in this model.
Table 1. Gaussian kernel convolution (GKC) common insertion sites (CIS) identified in all B-cell precursor acute lymphoblastic leukemia (BCP-ALL) cases.
CIS shown in bold are only found in the BCP-ALLs (n=15), not the other ALL cases. CIS that are not located within ± 150K base pairs of a gene are given the label ‘CIS’ followed by the chromosome and the peak location of the Gaussian kernel.
| Gene | Chr. | CIS leak location |
GKC scale (Kbps) |
Tumours with CIS |
Unique insertions |
Other genes in the CIS interval |
|---|---|---|---|---|---|---|
| CIS7:37320759 | 7 | 37320759 | 15 | 6 | 12 | - |
| Eif2a | 3 | 58350745 | 240 | 6 | 10 |
2810407C02Rik,
AC111080.1, AC119873.1, AC132302.1, AC142228.1, Commd2, Gm410, Pfn2, Rnf13, Serp1, Siah2, Tsc22d2, U6, Wwtr1 |
| Ikzf1 | 11 | 11652893 | 15 | 4 | 9 | - |
| Epor | 9 | 21769506 | 15 | 2 | 9 | - |
| Cebpa | 7 | 35899198 | 15 | 2 | 8 | 5S_rRNA |
| Cnr2 | 4 | 135467435 | 15 | 4 | 7 | - |
| Prr8 | 5 | 28716988 | 75 | 5 | 6 |
AC121843.1,
AC134530.1, Shh |
| Rcan2 | 17 | 44101905 | 75 | 3 | 6 | 7SK, CT025668.2 |
| CIS6:31099937 | 6 | 31099937 | 30 | 5 | 5 |
AB041803,
AC153820.1/4, RP23-459L15.3 |
| Rasgrf1 | 9 | 89867811 | 15 | 5 | 5 | - |
| Slc10a1 | 12 | 82095071 | 15 | 5 | 5 | Sfrs5 |
| Gm1968 | 16 | 29983555 | 30 | 5 | 5 |
CT025592.1,
CT030736.1 |
| Mecom | 3 | 30047667 | 15 | 4 | 5 | - |
| Psd3 | 8 | 70258943 | 30 | 4 | 5 |
AC109142.1,
AC109142.2 |
| Sfi1 | 11 | 3065855 | 50 | 4 | 5 |
AL671968.1/2/4/5,
Drg1, Eif4enif1 |
| CIS5:75770426 | 5 | 75770426 | 15 | 3 | 5 | U6 |
| Raf1 | 6 | 115605310 | 120 | 2 | 5 | - |
| Kctd2 | 11 | 115288407 | 15 | 1 | 5 | - |
| CIS11:54066112 | 11 | 54066112 | 15 | 3 | 4 | AL596103.1, Il3 |
| Asap1 | 15 | 64063966 | 30 | 3 | 4 | - |
| Met | 6 | 17424888 | 15 | 2 | 4 | - |
| Fam46d | X | 105018631 | 15 | 2 | 4 | 2610002M06Rik |
| Csf2rb2 | 15 | 78135922 | 15 | 1 | 4 | - |
| CIS2:131423786 | 2 | 131423786 | 15 | 3 | 3 | - |
| Zmym4 | 4 | 126594535 | 15 | 3 | 3 | - |
| Zfp423 | 8 | 90421110 | 15 | 3 | 3 | - |
| Mef2c | 13 | 83672435 | 15 | 3 | 3 | - |
| Il2rb | 15 | 78328305 | 15 | 3 | 3 | - |
| Rasgef1a | 6 | 118007445 | 15 | 2 | 3 | - |
| L3mbtl3 | 10 | 26098605 | 15 | 2 | 3 | - |
| CIS10:116095924 | 10 | 116095924 | 15 | 2 | 3 | - |
| Thg1l | 11 | 45761354 | 15 | 1 | 3 | - |
| Gata1 | X | 7537541 | 15 | 2 | 2 | - |
Insertional mutagenesis in ETV6-RUNX1-expressing mice models human ALL
By performing cross-species comparisons we found an overlap between some of the CIS in our BCP-ALL cases and genes recurrently found to be altered in patients with ETV6-RUNX1 positive ALL (Table 1), specifically insertions in the coding region of Ikzf1 and Epor. IKZF1 has a central role in the pathogenesis of BCP-ALL. Deletions involving IKZF1 are common (30%) in high-risk/poor prognosis B-cell precursor ALL31 (although not in ETV6-RUNX1 ALL) and EPOR is consistently highly expressed in ETV6-RUNX1 ALL.32 Compared to control spleens (n=3), those spleens with insertions in Epor (n=2) showed a log-fold-change of 0.35 for Epor expression (which is ~27% increase in expression)20 whereas those without insertions in Epor (n=13) showed a log-fold-change of −0.23 (which is ~15% decrease in expression). This suggests that the insertions in the promoter region of Epor upregulate Epor expression, a finding consistent with ETV6-RUNX1 ALL patients. In addition, we found Met to be a CIS, and c-MET activation specifically enhances FAS-mediated apoptosis in ETV6-RUNX1 cells. Indeed, the c-MET/FAS complex is only found in normal B lymphocytes and ETV6-RUNX1 leukemias and not B-ALLs that lack the t(12;21) translocation, an observation which may account for their high sensitivity to chemotherapeutic regimens.33 We also found CIS in genes mutated in AML, specifically Gata1 (Gata-1 mutation is found in AMKL associated with Downs syndrome)34 and Cebpa (mutations in C/EBPA are seen in AML).35
Seven of the CIS genes in our BCP-ALL samples had genomic locations syntenic with human chromosomal locations known to be regions of recurrent copy number change in ETV6-RUNX1 patient samples, including Fam46d, Gata1 and L3mbtl3 (n ≥ 3/50 patients; Table 2). Although the genomic size of the lesions was quite large in some cases, it is possible that our CIS genes can serve as a focal point for identification of the causative gene in these regions.
Table 2. CIS identified in the BCP-ALL samples whose homologous human chromosomal location was found to be a recurrent region of copy number (CN) change in human ETV6-RUNX1 leukemias.
A total of 50 ETV6-RUNX1 patient samples were analyzed by SNP array, and the CN changes for the relevant lesions shown. Deletions correspond to CN<2 and amplifications to CN>2. The LiftOver tool (http://genome.ucsc.edu/cgi-bin/hgLiftOver) was used to find the associated human chromosomal region for each mouse CIS gene (using the GRCh37/hg19 assembly). Statistical significance for each CIS was calculated (using 1,000 randomly selected Refseq genes) and a cut-off of P<0.2 was used.
| Mouse CIS (no. insertions) |
Homologous human region (chr: basepair) |
No. patients |
CN changes in patient samples |
CN lesion size (Mb): mean ± SD (median) |
|---|---|---|---|---|
|
Fam46d (4) |
X: 79589304- 79616318 |
10 | 1.4, 1.34, 1.36, 1.28, 1.53, 1.11, 1, 2.99, 0.85, 1.23 |
124 ± 43 (150) |
|
Gata1 (2) |
X: 48645231- 48659263 |
9 | 2.47, 1.34, 1.36, 1.28, 1.11, 1, 2.99, 0.85, 1.23 |
132 ± 34 (151) |
|
L3mbtl3 (3) |
6: 130336865- 130339827 |
5 | 0.88, 1.28, 0.84, 1.55, 3.17 |
110 ± 57 (87) |
|
CIS7:37320759 (12) |
19: 31962394- 32030766 |
3 | 1.68, 1.65, 1.06 | 23 ± 29 (7) |
|
Cebpa (8) |
19: 33765960- 33835715 |
3 | 1.68, 1.65, 1.06 | 23 ± 29 (7) |
|
Gm1968 (5) |
3: 193728382- 193781161 |
3 | 0.98, 0.94, 1.59 | 37 ± 42 (20) |
|
Psd3 (5) |
8: 18414769- 18459953 |
3 | 1.16, 0.93, 2.6 | 58 ± 37 (37) |
Given the small number of BCP-ALL cases in this cohort (n=15), we were statistically underpowered to generate a large number of CIS. Therefore for the BCP-ALL cases we looked at the 4,787 unique/non-redundant insertion sites in addition to the CIS, and found that many of them were of interest because of their location relative to genes known to be dysregulated in ETV6-RUNX1 positive ALL patients. These genes included Lef1, Dpf3, Tbl1xr1, Ebf1 and Nr3c1 (Table 3). Although the insertion frequency was too low, or the genomic distribution of the insertions too broad to be classified as a CIS, a frequency of 1-3 cases from a total of 15 represents 6-20%, and echoes the frequency reported in ETV6-RUNX1 positive ALL patients.7, 24
Table 3. Human-mouse comparison of the insertion sites in BCP-ALL cases.
The Ensembl mouse genome annotation database (version 59) was used to find the associated mouse gene for the human genes relevant to ETV6-RUNX1 patients (genes that did not map were hand-curated). The location of insertion sites was scored as being in the coding region of the gene (‘coding’) or ≤ 5K upstream of the gene (‘promoter’).
| Mouse gene with insertion |
Insertion location (relative to gene) |
No. unique insertions |
Dysregulation of gene in human ETV6-RUNX1 ALL lesions |
Reference |
|---|---|---|---|---|
| Ikzf1 | coding | 4 | In a deletion region | 36*, 31** |
| C20orf94 | coding | 3 | In a deletion region | 7 |
| Ebf1 | coding | 3 | In a deletion region | 7-8 |
| Fhit | coding | 3 | In a deletion region | 7 |
| Chn2 | coding | 2 | Down-regulated expression | 37 |
| Epor | promoter | 2 | Up-regulated expression | 37-39 |
| Flt3 | promoter | 2 | Activating mutations | 40 |
| Mllt3 | coding | 2 | In a deletion region | 7 |
| Nf1 | coding | 2 | In a deletion region | 7 |
| Smarcc1 | coding | 2 | In a deletion region | 24 |
| Tbl1xr1 | coding | 2 | In a deletion region | 7-8 |
| Tcf4 (E2-2) | coding | 2 | In a deletion region | 36, 41 |
| Ankrd27 | coding | 1 | In a deletion region | 24 |
| Arid1b | coding | 1 | In a deletion region | 42 |
| Atf2 | coding | 1 | Down-regulated expression | 37 |
| Atp10a | coding | 1 | In a deletion region | 42 |
| B4galt1 | coding | 1 | Down-regulated expression | 37 |
| Birc7 | promoter | 1 | Up-regulated expression | 38 |
| Cd44 | coding | 1 | Down-regulated expression | 37-39 |
| Clic5 | coding | 1 | Up-regulated expression | 38 |
| Dpf3 | coding | 1 | In a deletion region | 7 |
| Eif2s1 | coding | 1 | Down-regulated expression | 37 |
| Fbn2 | coding | 1 | Up-regulated expression | 38 |
| Flnb | coding | 1 | Down-regulated expression | 37 |
| Fyn | coding | 1 | In a deletion region | 36 * |
| Grik2 | coding | 1 | In a deletion region | 7 |
| Hmg20a | coding | 1 | Up-regulated expression | 38 |
| Jak1 | coding | 1 | Expression up-regulated | 37 |
| Lef1 | coding | 1 | In a deletion region | 7 |
| Lztfl1 | coding | 1 | In a deletion region | 24 |
| Myo10 | coding | 1 | Up-regulated expression | 38 |
| Nr3c1 | coding | 1 | In a deletion region | 7 |
| Nr3c2 | coding | 1 | In a deletion region | 7 |
| Pbef1 (Nampt) | coding | 1 | In a deletion region | 36 |
| Pde4b | coding | 1 | In a deletion region | 41 |
| Pdlim1 | coding | 1 | Down-regulated expression | 37 |
| Ptpn2 | coding | 1 | Down-regulated expression | 37 |
| Ptprk | coding | 1 | Up-regulated expression | 37-38 |
| Rap1b | coding | 1 | In a deletion region | 42 |
| Rb1 | coding | 1 | In a deletion region | 7-8 |
| Rhpn2 | promoter | 1 | In a deletion region | 24 |
| Tcf3 (E2A) | promoter | 1 | In a deletion region | 36 * |
| Tcf12 | coding | 1 | In a deletion region | 42 |
| Thada | coding | 1 | In a deletion region | 7 |
| Usp9x | coding | 1 | Down-regulated expression | 37 |
| Yipf7 | coding | 1 | In a deletion region | 41 |
| Ywhaq | coding | 1 | In a deletion region | 41 |
| Zcchc7 | coding | 1 | In a deletion region | 8, 24 |
reported in childhood ALL (not necessarily ETV6-RUNX1 positive patients).
IKZF1 deletion outside of the BCR-ABL context.
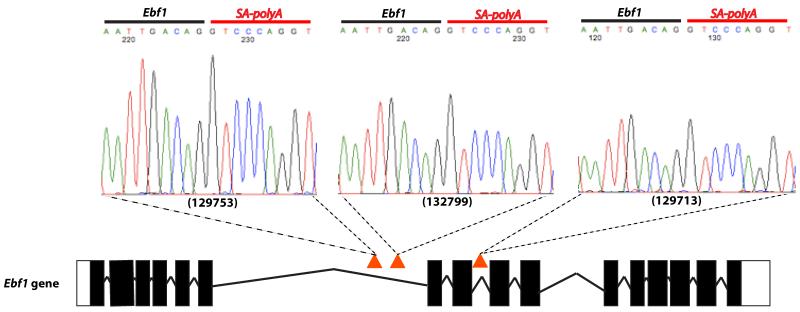
Of particular importance was Ebf1. EBF1 can activate expression of PAX5,43 and analysis of cDNA from the 3 cases which carried Ebf1 insertions showed splicing of Ebf1 directly onto the splice acceptor/polyA of the transposon (Figure 5). This resulted in premature truncation of the gene at exon 6 and thus presumably an inability to activate Pax5. As well as Ebf1, we also had insertions in many other key regulators of B-cell development including Ikzf1 (Figure S8), Lef1, Tcf3 and Blnk, providing strong evidence that genetic alterations resulting in a block in B-lymphoid development are key events in the pathogenesis of B-ALL.
Figure 5. Insertions in the Ebf1 gene.
Three Etv6+/RUNX1; T2Onc+/Tg (EROnc) mice (129753, 132799 and 129713) that developed BCP-ALL carried insertions in the Ebf1 gene (indicated by the red triangles in introns 6 and 8). Sequencing of the insertion-genome junction from splenic cDNA of these mice showed the splicing of Ebf1 directly onto the splice acceptor (SA)-polyA from the transposon. Although sample 129713 carried an insertion in intron 8, we detected splicing onto the transposon directly from exon 6.
Discussion
ETV6-RUNX1 is the most common fusion oncogene associated with childhood ALL, and although generally associated with favorable prognosis, no faithful mouse models exist to provide mechanistic or potential therapeutic insights into this disease. Previous models have been hampered by potential overexpression of the fusion oncogene from transgenic or retroviral promoters, by a failure to generate leukemia or by generation of a leukemia of a differing phenotype.10-14 In this report, we describe the generation of the first mouse model of ETV6-RUNX1 that develops B-cell precursor (BCP)-ALL. In addition, through the use of the SB transposon system, we identify cooperating mutations that facilitate development of ALL, thus providing an excellent resource to further dissect the biology of this disease.
We identified a total of 102 unique statistically significant common insertion sites (CIS) regions/genes from the 71 leukemias analyzed (Table S3 and Figure 4). Interestingly, many of the CIS that were found in the BCP-ALLs (Table 1) were also found in the AML and T-ALL cases. Since expression of the fusion in our model occurs in hematopoietic stem cells (HSCs) following the expression pattern of endogenous Etv6, it likely that the fusion ‘primes’ cells to become leukemic with the actual phenotype driven by the nature of the cooperating mutations and the hematopoietic lineage/compartment in which they are acquired (before specification of the B-cell lineage). However, over-lapping CIS from the BCP-ALL and AML cases may reflect the fact that several of these BCP-ALL cases also expressed myeloid markers (Figure S4), as the co-expression of myeloid antigens has been described in ETV6-RUNX1 positive ALL patients.27
Cross-species analysis of the BCP-ALL cases identified insertions/CIS in genes known to be dysregulated in human ETV6-RUNX1 positive ALL patients, including Btg1, Dpf3, Ebf1, Epor, Fhit, Grik2, Ikzf1, Lef1, Mllt3, Nf1, Nr3c1/2, Rap1b, Rb1, Tbl1xr1, Tcf12 and Zcchc7 (Table 3). Previous studies that have performed genome-wide profiling of genetic alterations in ETV6-RUNX1 BCP-ALL have shown that although a small number of mutations predominate, the mutational spectrum is highly heterogeneous, with numerous recurrent lesions generating mutations or copy number alterations in only a few patients.7, 41 For example, in 47 cases of ETV6-RUNX1 positive ALL, deletions at chromosome 12p13.2 (ETV6) and 9p13.2 (PAX5) were found in as many as 33 and 13 patients (70% and 27% of cases) respectively. However, the majority of chromosomal gains/losses were found in less than eight (14%) patients, including deletions at 3q26.32 (TBL1XR1), 5q33.3 (EBF1) and 5q31.3 (NR3C1), with 8 of the genomic rearrangements only being found in 1 (2%) patient, including deletions at 4q25 (LEF1), 10q24.1 (BLNK) and 14q24.2 (DPF3).7 Thus although our insertions in these genes do not classify as “CIS”, a ‘hit’ frequency of 6-20% echoes the 2-14% frequency reported in ETV6-RUNX1 positive ALL patients.
In addition, it is interesting that some genes in recurrent regions of chromosomal imbalance were not involved, including Etv6, Cdkn2a/Cdkn2b (encoding Ink4a/Arf) and Pax5.7, 24 This could be due to our small sample size of 15 BCP-ALL cases. Alternatively, it is possible that these genes were not available for insertion, through prior inactivation by other means such as focal deletion or point mutation (potentially through illegitimate RAG-mediated recombination). It is also recognized that the mutation mechanism itself can contribute to (bias) the selection of target genes. For example, AID expression in CML cells has been suggested to promote overall genetic instability by hypermutation of tumor suppressor and DNA repair genes.44 Moreover, malignancies of progenitor B-cells have also been associated with aberrant RAG mediated recombination.45 Both of these mechanisms are sequence specific and may contribute a different bias to our experimental system in the selection of specific targets. Finally it is also possible that this reflects the fact that our disease involves an earlier cell type (pro-B) than typically seen in ETV6-RUNX1 positive ALL patients (which are typically pre-B).
However, although Pax5 was not involved in our screen, we identified mutations in Ebf1 and Tcf3 (E2A), which cooperatively regulate PAX5,46 and Tcf4, Lef1, and CD44, which are PAX5-activated genes.47-48 These “complementary” lesions therefore potentially having the same signaling impact as if we had directly mutated Pax5. Interestingly, a study recently found that many PAX5-regulated genes lie within 100 kb of an EBF1-bound region, suggesting coordinated regulation of common target genes by these transcription factors.49
Of the CIS regions/genes that were identified in the BCP-ALLs (Table 1), Epor and Met are known to show altered expression in ETV6-RUNX1 positive ALL patients (and Ikzf in some cases). Although the other regions/genes have not been associated with ETV6-RUNX1 ALL, some have been shown to play a role in lymphocyte biology. For example, Cebpa belongs to the CEBP family, which have recently been found to show dysregulated expression in B-cell precursors and can contribute to their malignant transformation50 and Rasgrf1 has been shown to be over-expressed in B-cell chronic lymphocytic leukemia (B-CLL) compared to normal B lymphocytes.51 Other regions/genes have previously been implicated as players in tumorigenesis, such as Cnr2, a proto-oncogene that has been postulated to offer a growth advantage in leukemia cells52 and Psd3, reduced expression of which is believed to contribute to malignant progression.53 Thus these genes are all attractive candidates to co-operate with ETV6-RUNX1 in promoting ALL, and as new technologies emerge that allow further interrogation of cancer biology (such as RNA sequencing, exome sequencing, etc) new genes will constantly be added to the list of those found to be dysregulated in ETV6-RUNX1 ALL.
In summary, we present the first mouse model of ETV6-RUNX1 that exhibits BCP-ALL. This model will prove a valuable resource to both further understanding of the disease and in the development of therapeutics in ETV6-RUNX1 positive ALL.
Supplementary Material
Acknowledgements
The authors wish to thank the staff of the Research Support Facility at the Wellcome Trust Sanger Institute for looking after the mice and Mahrokh Nohadani for performing the immunohistochemistry. LvdW is supported by a Fellowship from the Kay Kendall Leukemia Fund. DJA is supported by Cancer Research-UK and the Wellcome Trust. BJH and GG are supported by an MRC-UK Senior Fellowship (BJH) and Cancer Research-UK. FWvD and MG are supported by the Kay Kendall Leukaemia Fund and Leukaemia & Lymphoma Research UK. AEC and LSM are supported by the BBSRC. CGM is supported by the American Lebanese Syrian Associated Charities of St Jude Children’s Research Hospital, and is a Pew Scholar in the Biomedical Sciences.
Footnotes
Conflict-of-interest disclosure: The authors declare no competing financial interests.
References
- 1.Romana SP, Mauchauffé M, Le Coniat M, et al. The t(12;21) of acute lymphoblastic leukemia results in a TEL-AML1 gene fusion. Blood. 1995;85:3662–3670. [PubMed] [Google Scholar]
- 2.Shurtleff SA, Buijs A, Behm FG, et al. TEL/AML1 fusion resulting from a cryptic t(12;21) is the most common genetic lesion in pediatric ALL and defines a subgroup of patients with an excellent prognosis. Leukemia. 1995;9:1985–1989. [PubMed] [Google Scholar]
- 3.Greaves MF, Wiemels J. Origins of chromosome translocations in childhood leukaemia. Nat Rev Cancer. 2003;3:639–649. doi: 10.1038/nrc1164. [DOI] [PubMed] [Google Scholar]
- 4.Hong D, Gupta R, Ancliff P, et al. Initiating and cancer-propagating cells in TEL-AML1-associated childhood leukemia. Science. 2008;319:336–339. doi: 10.1126/science.1150648. [DOI] [PubMed] [Google Scholar]
- 5.Castor A, Nilsson L, Astrand-Grundström I, et al. Distinct patterns of hematopoietic stem cell involvement in acute lymphoblastic leukemia. Nat Med. 2005;11:630–637. doi: 10.1038/nm1253. [DOI] [PubMed] [Google Scholar]
- 6.Mori H, Colman SM, Xiao Z, et al. Chromosome translocations and covert leukemic clones are generated during normal fetal development. Proc Natl Acad Sci USA. 2002;99:8242–8247. doi: 10.1073/pnas.112218799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mullighan CG, Goorha S, Radtke I, et al. Genome-wide analysis of genetic alterations in acute lymphoblastic leukaemia. Nature. 2007;446:758–764. doi: 10.1038/nature05690. [DOI] [PubMed] [Google Scholar]
- 8.Bateman CM, Colman SM, Chaplin T, et al. Acquisition of genome-wide copy number alterations in monozygotic twins with acute lymphoblastic leukemia. Blood. 2010;115:3553–3558. doi: 10.1182/blood-2009-10-251413. [DOI] [PubMed] [Google Scholar]
- 9.Higuchi M, O’Brien D, Kumaravelu P, Lenny N, Yeoh EJ, Downing JR. Expression of a conditional AML1-ETO oncogene bypasses embryonic lethality and establishes a murine model of human t(8;21) acute myeloid leukemia. Cancer Cell. 2002;1:63–74. doi: 10.1016/s1535-6108(02)00016-8. [DOI] [PubMed] [Google Scholar]
- 10.Bernardin F, Yang Y, Cleaves R, et al. TEL-AML1, expressed from t(12;21) in human acute lymphocytic leukemia, induces acute leukemia in mice. Cancer Res. 2002;62:3904–3908. [PubMed] [Google Scholar]
- 11.Tsuzuki S, Seto M, Greaves M, Enver T. Modeling first-hit functions of the t(12;21) TEL-AML1 translocation in mice. Proc Natl Acad Sci U S A. 2004;101:8443–8448. doi: 10.1073/pnas.0402063101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Fischer M, Schwieger M, Horn S, et al. Defining the oncogenic function of the TEL/AML1 (ETV6/RUNX1) fusion protein in a mouse model. Oncogene. 2005;24:7579–7591. doi: 10.1038/sj.onc.1208931. [DOI] [PubMed] [Google Scholar]
- 13.Sabaawy HE, Azuma M, Embree LJ, Tsai HJ, Starost MF, Hickstein DD. TEL-AML1 transgenic zebrafish model of precursor B cell acute lymphoblastic leukemia. Proc Natl Acad Sci U S A. 2006;103:15166–15171. doi: 10.1073/pnas.0603349103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Schindler JW, Van Buren D, Foudi A, et al. TEL-AML1 corrupts hematopoietic stem cells to persist in the bone marrow and initiate leukemia. Cell Stem Cell. 2009;5:43–53. doi: 10.1016/j.stem.2009.04.019. [DOI] [PubMed] [Google Scholar]
- 15.Yant SR, Huang Y, Akache B, Kay MA. Site-directed transposon integration in human cells. Nucleic Acids Res. 2007;35:e50. doi: 10.1093/nar/gkm089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.van der Weyden L, Adams DJ, Harris LW, Tannahill D, Arends MJ, Bradley A. Null and conditional semaphorin 3B alleles using a flexible puroDeltatk loxP/FRT vector. Genesis. 2005;41:171–178. doi: 10.1002/gene.20111. [DOI] [PubMed] [Google Scholar]
- 17.Morrow M, Samanta A, Kioussis D, Brady HJ, Williams O. TEL-AML1 preleukemic activity requires the DNA binding domain of AML1 and the dimerization and corepressor binding domains of TEL. Oncogene. 2007;26:4404–4414. doi: 10.1038/sj.onc.1210227. [DOI] [PubMed] [Google Scholar]
- 18.Collier LS, Carlson CM, Ravimohan S, Dupuy AJ, Largaespada DA. Cancer gene discovery in solid tumours using transposon-based somatic mutagenesis in the mouse. Nature. 2005;436:272–276. doi: 10.1038/nature03681. [DOI] [PubMed] [Google Scholar]
- 19.Yang YH, Dudoit S, Luu P, et al. Normalization for cDNA microarray data: a robust composite method addressing single and multiple slide systematic variation. Nucleic Acids Res. 2002;30:e15. doi: 10.1093/nar/30.4.e15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Benjamini Y, Hochberg Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J Roy Statist Soc Ser B. 1995;57:289–300. [Google Scholar]
- 21.March HN, Rust AG, Wright NA, et al. Insertional mutagenesis reveals multiple networks of co-operating genes driving intestinal mutagenesis. Submitted to Nature Genetics. 2010 Jul; doi: 10.1038/ng.990. Manuscript ID: NG-A28875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Farley FW, Soriano P, Steffen LS, Dymecki SM. Widespread recombinase expression using FLPeR (flipper) mice. Genesis. 2000;28:106–110. [PubMed] [Google Scholar]
- 23.Wang LC, Kuo F, Fujiwara Y, Gilliland DG, Golub TR, Orkin SH. Yolk sac angiogenic defect and intra-embryonic apoptosis in mice lacking the Ets-related factor TEL. EMBO J. 1997;16:4374–4383. doi: 10.1093/emboj/16.14.4374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Tsuzuki S, Karnan S, Horibe K, et al. Genetic abnormalities involved in t(12;21) TEL-AML1 acute lymphoblastic leukemia: analysis by means of array-based comparative genomic hybridization. Cancer Sci. 2007;98:698–706. doi: 10.1111/j.1349-7006.2007.00443.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Morse HC, 3rd, Anver MR, Fredrickson TN, et al. Bethesda proposals for classification of lymphoid neoplasms in mice. Blood. 2002;100:246–58. doi: 10.1182/blood.v100.1.246. [DOI] [PubMed] [Google Scholar]
- 26.Kogan SC, Ward JM, Anver MR, et al. Bethesda proposals for classification of nonlymphoid hematopoietic neoplasms in mice. Blood. 2002;100:238–245. doi: 10.1182/blood.v100.1.238. [DOI] [PubMed] [Google Scholar]
- 27.Romana SP, Poirel H, Leconiat M, et al. High frequency of t(12;21) in childhood B-lineage acute lymphoblastic leukemia. Blood. 1995;86:4263–4269. [PubMed] [Google Scholar]
- 28.Wong S, Fulcher D. Chemokine receptor expression in B-cell lymphoproliferative disorders. Leuk Lymphoma. 2004;45:2491–2496. doi: 10.1080/10428190410001723449. [DOI] [PubMed] [Google Scholar]
- 29.De Zen L, Orfao A, Cazzaniga G, et al. Quantitative multiparametric immunophenotyping in acute lymphoblastic leukemia: correlation with specific genotype. I. TEL/AML1 ALLs identification. Leukemia. 2000;14:1225–1231. doi: 10.1038/sj.leu.2401824. [DOI] [PubMed] [Google Scholar]
- 30.de Ridder J, Uren A, Kool J, Reinders M, Wessels L. Detecting statistically significant common insertion sites in retroviral insertional mutagenesis screens. PLoS Comput Biol. 2006;2:e166. doi: 10.1371/journal.pcbi.0020166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Mullighan CG, Su X, Zhang J, et al. Deletion of IKZF1 and prognosis in acute lymphoblastic leukemia. N. Engl. J. Med. 2009;360:470–480. doi: 10.1056/NEJMoa0808253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Fine BM, Stanulla M, Schrappe M, Ho M, Viehmann S, Harbott J, Boxer LM. Gene expression patterns associated with recurrent chromosomal translocations in acute lymphoblastic leukemia. Blood. 2004;103:1043–1049. doi: 10.1182/blood-2003-05-1518. [DOI] [PubMed] [Google Scholar]
- 33.Accordi B, Pillozzi S, Dell’Orto MC, et al. Hepatocyte growth factor receptor c-MET is associated with FAS and when activated enhances drug-induced apoptosis in pediatric B acute lymphoblastic leukemia with TEL-AML1 translocation. J Biol Chem. 2007;282:29384–29393. doi: 10.1074/jbc.M706314200. [DOI] [PubMed] [Google Scholar]
- 34.Wechsler J, Greene M, McDevitt MA, et al. Acquired mutations in GATA1 in the megakaryoblastic leukemia of Down syndrome. Nat Genet. 2002;32:148–152. doi: 10.1038/ng955. [DOI] [PubMed] [Google Scholar]
- 35.Lu Y, Chen W, Chen W, Stein A, Weiss LM, Huang Q. C/EBPA gene mutation and C/EBPA promoter hypermethylation in acute myeloid leukemia with normal cytogenetics. Am J Hematol. 2010;85:426–430. doi: 10.1002/ajh.21706. [DOI] [PubMed] [Google Scholar]
- 36.Kuiper RP, Schoenmakers EF, van Reijmersdal SV, et al. High-resolution genomic profiling of childhood ALL reveals novel recurrent genetic lesions affecting pathways involved in lymphocyte differentiation and cell cycle progression. Leukemia. 2007;21:1258–1266. doi: 10.1038/sj.leu.2404691. [DOI] [PubMed] [Google Scholar]
- 37.Yeoh EJ, Ross ME, Shurtleff SA, et al. Classification, subtype discovery, and prediction of outcome in pediatric acute lymphoblastic leukemia by gene expression profiling. Cancer Cell. 2002;1:133–143. doi: 10.1016/s1535-6108(02)00032-6. [DOI] [PubMed] [Google Scholar]
- 38.Ross ME, Zhou X, Song G. Classification of pediatric acute lymphoblastic leukemia by gene expression profiling. Blood. 2003;102:2951–2959. doi: 10.1182/blood-2003-01-0338. [DOI] [PubMed] [Google Scholar]
- 39.Fine BM, Stanulla M, Schrappe M. Gene expression patterns associated with recurrent chromosomal translocations in acute lymphoblastic leukemia. Blood. 2004;103:1043–1049. doi: 10.1182/blood-2003-05-1518. [DOI] [PubMed] [Google Scholar]
- 40.Armstrong SA, Mabon ME, Silverman LB. FLT3 mutations in childhood acute lymphoblastic leukemia. Blood. 2004;103:3544–3546. doi: 10.1182/blood-2003-07-2441. [DOI] [PubMed] [Google Scholar]
- 41.Lilljebjörn H, Soneson C, Andersson A. The correlation pattern of acquired copy number changes in 164 ETV6/RUNX1-positive childhood acute lymphoblastic leukemias. Hum Mol Genet. 2010;19:3150–3158. doi: 10.1093/hmg/ddq224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Mullighan CG, Phillips LA, Su X. Genomic analysis of the clonal origins of relapsed acute lymphoblastic leukemia. Science. 2008;322:1377–1380. doi: 10.1126/science.1164266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Zandi S, Mansson R, Tsapogas P, Zetterblad J, Bryder D, Sigvardsson M. EBF1 is essential for B-lineage priming and establishment of a transcription factor network in common lymphoid progenitors. J Immunol. 2008;181:3364–3372. doi: 10.4049/jimmunol.181.5.3364. [DOI] [PubMed] [Google Scholar]
- 44.Klemm L, Duy C, Iacobucci I, Kuchen S, von Levetzow G, Feldhahn N, Henke N, Li Z, Hoffmann TK, Kim YM, Hofmann WK, Jumaa H, Groffen J, Heisterkamp N, Martinelli G, Lieber MR, Casellas R, Müschen M. The B cell mutator AID promotes B lymphoid blast crisis and drug resistance in chronic myeloid leukemia. Cancer Cell. 2009;16:232–245. doi: 10.1016/j.ccr.2009.07.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Tsai AG, Lu H, Raghavan SC, Muschen M, Hsieh CL, Lieber MR. Human chromosomal translocations at CpG sites and a theoretical basis for their lineage and stage specificity. Cell. 2008;135:1130–1142. doi: 10.1016/j.cell.2008.10.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Busslinger M. Transcriptional control of early B cell development. Annu Rev Immunol. 2004;22:55–7. doi: 10.1146/annurev.immunol.22.012703.104807. [DOI] [PubMed] [Google Scholar]
- 47.Pridans C, Holmes ML, Polli M. Identification of Pax5 target genes in early B cell differentiation. J Immunol. 2008;180:1719–1728. doi: 10.4049/jimmunol.180.3.1719. [DOI] [PubMed] [Google Scholar]
- 48.Schebesta A, McManus S, Salvagiotto G, Delogu A, Busslinger GA, Busslinger M. Transcription factor Pax5 activates the chromatin of key genes involved in B cell signaling, adhesion, migration, and immune function. Immunity. 2007;27:49–63. doi: 10.1016/j.immuni.2007.05.019. [DOI] [PubMed] [Google Scholar]
- 49.Treiber T, Mandel EM, Pott S, Györy I, Firner S, Liu ET, Grosschedl R. Early B cell factor 1 regulates B cell gene networks by activation, repression, and transcription-independent poising of chromatin. Immunity. 2010;32:714–725. doi: 10.1016/j.immuni.2010.04.013. [DOI] [PubMed] [Google Scholar]
- 50.Akasaka T, Balasas T, Russell LJ. Five members of the CEBP transcription factor family are targeted by recurrent IGH translocations in B-cell precursor acute lymphoblastic leukemia (BCP-ALL) Blood. 2007;109:3451–3461. doi: 10.1182/blood-2006-08-041012. [DOI] [PubMed] [Google Scholar]
- 51.Jelinek DF, Tschumper RC, Stolovitzky GA. Identification of a global gene expression signature of B-chronic lymphocytic leukemia. Mol Cancer Res. 2003;1:346–361. [PubMed] [Google Scholar]
- 52.Valk PJ, Delwel R. The peripheral cannabinoid receptor, Cb2, in retrovirally-induced leukemic transformation and normal hematopoiesis. Leuk Lymphoma. 1998;32:29–43. doi: 10.3109/10428199809059244. [DOI] [PubMed] [Google Scholar]
- 53.van den Boom J, Wolter M, Blaschke B, Knobbe CB, Reifenberger G. Identification of novel genes associated with astrocytoma progression using suppression subtractive hybridization and real-time reverse transcription-polymerase chain reaction. Int J Cancer. 2006;119:2330–2338. doi: 10.1002/ijc.22108. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.