Abstract
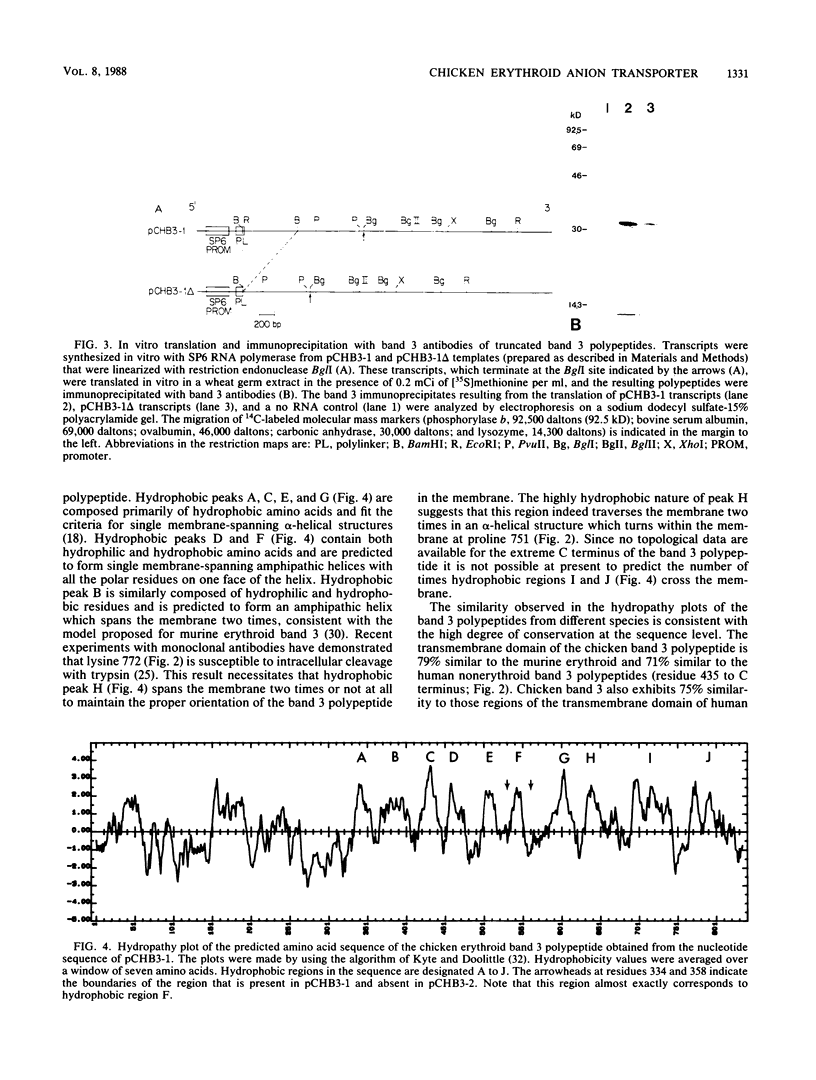
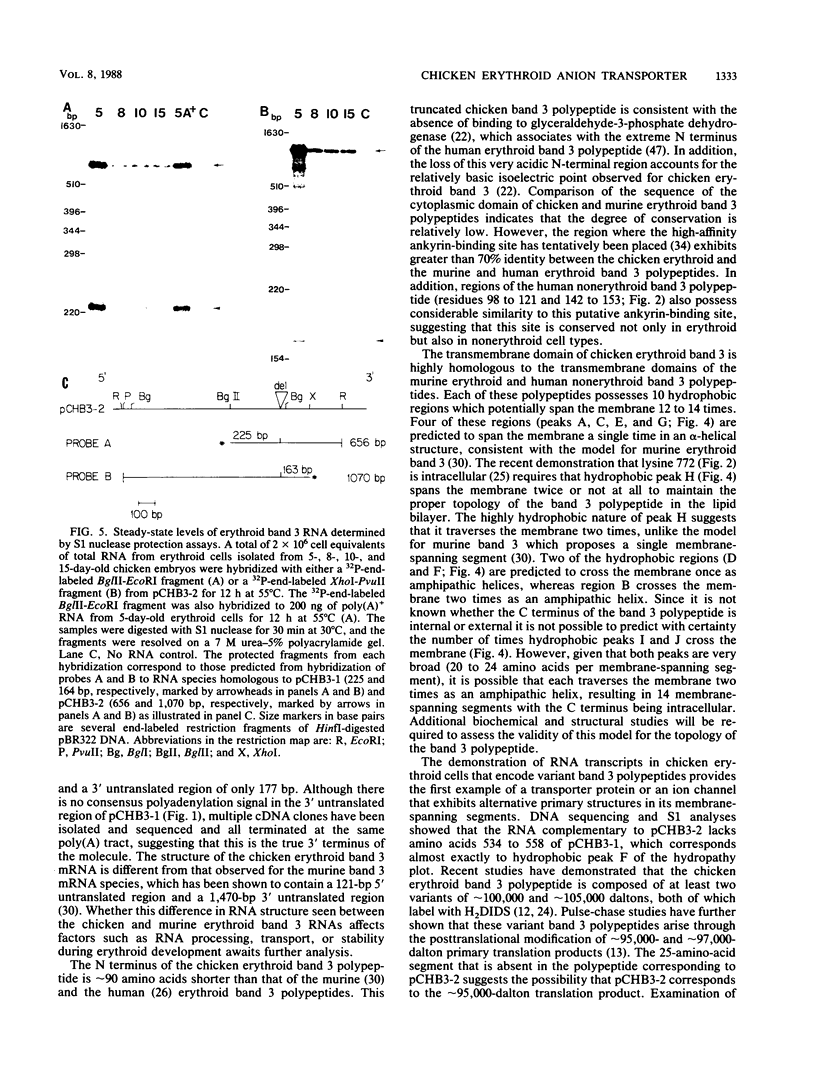
Isolation and characterization of the chicken erythroid anion transporter (band 3) cDNA clone, pCHB3-1, revealed that the chicken erythroid band 3 polypeptide is 844 amino acids in length with a predicted mass of 109,000 daltons. This polypeptide is composed of a hydrophilic N-terminal cytoplasmic domain and a hydrophobic C-terminal transmembrane domain. The approximately 90 N-terminal amino acids of the human and murine erythroid band 3 polypeptides are absent in the predicted sequence of the chicken erythroid band 3 polypeptide. The absence of this very acidic N-terminal region is consistent with the lack of binding of glyceraldehyde-3-phosphate dehydrogenase to chicken erythroid band 3, as well as the relatively basic isoelectric point observed for this molecule. The remainder of the cytoplasmic domain shows little similarity to the cytoplasmic domain of the murine and human erythroid band 3, with the exception of the putative ankyrin-binding site, which is highly conserved. In contrast, the transmembrane domain of the chicken band 3 polypeptide is very similar to that of the murine erythroid and human nonerythroid band 3 polypeptides. The transmembrane domain contains 10 hydrophobic regions that could potentially traverse the membrane 12 to 14 times. In addition, a variant of chicken erythroid band 3, pCHB3-2, was cloned in which one of the hydrophobic regions of pCHB3-1 is lacking. The transcript complementary to pCHB3-2 accumulated in chicken erythroid cells in a similar manner as the transcript complementary to pCHB3-1 during embryonic development. This is the first example of a transporter protein or ion channel with alternative primary structures in its membrane-spanning segments.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Appell K. C., Low P. S. Partial structural characterization of the cytoplasmic domain of the erythrocyte membrane protein, band 3. J Biol Chem. 1981 Nov 10;256(21):11104–11111. [PubMed] [Google Scholar]
- Aviv H., Leder P. Purification of biologically active globin messenger RNA by chromatography on oligothymidylic acid-cellulose. Proc Natl Acad Sci U S A. 1972 Jun;69(6):1408–1412. doi: 10.1073/pnas.69.6.1408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bennett V., Stenbuck P. J. The membrane attachment protein for spectrin is associated with band 3 in human erythrocyte membranes. Nature. 1979 Aug 9;280(5722):468–473. doi: 10.1038/280468a0. [DOI] [PubMed] [Google Scholar]
- Benton W. D., Davis R. W. Screening lambdagt recombinant clones by hybridization to single plaques in situ. Science. 1977 Apr 8;196(4286):180–182. doi: 10.1126/science.322279. [DOI] [PubMed] [Google Scholar]
- Berk A. J., Sharp P. A. Sizing and mapping of early adenovirus mRNAs by gel electrophoresis of S1 endonuclease-digested hybrids. Cell. 1977 Nov;12(3):721–732. doi: 10.1016/0092-8674(77)90272-0. [DOI] [PubMed] [Google Scholar]
- Blikstad I., Nelson W. J., Moon R. T., Lazarides E. Synthesis and assembly of spectrin during avian erythropoiesis: stoichiometric assembly but unequal synthesis of alpha and beta spectrin. Cell. 1983 Apr;32(4):1081–1091. doi: 10.1016/0092-8674(83)90292-1. [DOI] [PubMed] [Google Scholar]
- Brock C. J., Tanner M. J., Kempf C. The human erythrocyte anion-transport protein. Partial amino acid sequence, conformation and a possible molecular mechanism for anion exchange. Biochem J. 1983 Sep 1;213(3):577–586. doi: 10.1042/bj2130577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Capetanaki Y. G., Ngai J., Flytzanis C. N., Lazarides E. Tissue-specific expression of two mRNA species transcribed from a single vimentin gene. Cell. 1983 Dec;35(2 Pt 1):411–420. doi: 10.1016/0092-8674(83)90174-5. [DOI] [PubMed] [Google Scholar]
- Cassoly R., Salhany J. M. Spectral and oxygen-release kinetic properties of human hemoglobin bound to the cytoplasmic fragment of band 3 protein in solution. Biochim Biophys Acta. 1983 Jun 15;745(2):134–139. doi: 10.1016/0167-4838(83)90041-9. [DOI] [PubMed] [Google Scholar]
- Cox J. V., Moon R. T., Lazarides E. Anion transporter: highly cell-type-specific expression of distinct polypeptides and transcripts in erythroid and nonerythroid cells. J Cell Biol. 1985 May;100(5):1548–1557. doi: 10.1083/jcb.100.5.1548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cox J. V., Stack J. H., Lazarides E. Erythroid anion transporter assembly is mediated by a developmentally regulated recruitment onto a preassembled membrane cytoskeleton. J Cell Biol. 1987 Sep;105(3):1405–1416. doi: 10.1083/jcb.105.3.1405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crandall E. D., Mathew S. J., Fleischer R. S., Winter H. I., Bidani A. Effects of inhibition of RBC HCO3-/Cl- exchange on CO2 excretion and downstream pH disequilibrium in isolated rat lungs. J Clin Invest. 1981 Oct;68(4):853–862. doi: 10.1172/JCI110340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Demuth D. R., Showe L. C., Ballantine M., Palumbo A., Fraser P. J., Cioe L., Rovera G., Curtis P. J. Cloning and structural characterization of a human non-erythroid band 3-like protein. EMBO J. 1986 Jun;5(6):1205–1214. doi: 10.1002/j.1460-2075.1986.tb04348.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Drenckhahn D., Zinke K., Schauer U., Appell K. C., Low P. S. Identification of immunoreactive forms of human erythrocyte band 3 in nonerythroid cells. Eur J Cell Biol. 1984 May;34(1):144–150. [PubMed] [Google Scholar]
- Drickamer L. K. Fragmentation of the 95,000-dalton transmembrane polypeptide in human erythrocyte membranes. J Biol Chem. 1976 Sep 10;251(17):5115–5123. [PubMed] [Google Scholar]
- Eisenberg D. Three-dimensional structure of membrane and surface proteins. Annu Rev Biochem. 1984;53:595–623. doi: 10.1146/annurev.bi.53.070184.003115. [DOI] [PubMed] [Google Scholar]
- Finer-Moore J., Stroud R. M. Amphipathic analysis and possible formation of the ion channel in an acetylcholine receptor. Proc Natl Acad Sci U S A. 1984 Jan;81(1):155–159. doi: 10.1073/pnas.81.1.155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fukuda M., Eshdat Y., Tarone G., Marchesi V. T. Isolation and characterization of peptides derived from the cytoplasmic segment of band 3, the predominant intrinsic membrane protein of the human erythrocyte. J Biol Chem. 1978 Apr 10;253(7):2419–2428. [PubMed] [Google Scholar]
- Hargreaves W. R., Giedd K. N., Verkleij A., Branton D. Reassociation of ankyrin with band 3 in erythrocyte membranes and in lipid vesicles. J Biol Chem. 1980 Dec 25;255(24):11965–11972. [PubMed] [Google Scholar]
- Jay D. G. Characterization of the chicken erythrocyte anion exchange protein. J Biol Chem. 1983 Aug 10;258(15):9431–9436. [PubMed] [Google Scholar]
- Jay D. G. Glycosylation site of band 3, the human erythrocyte anion-exchange protein. Biochemistry. 1986 Feb 11;25(3):554–556. doi: 10.1021/bi00351a006. [DOI] [PubMed] [Google Scholar]
- Jay D., Cantley L. Structural aspects of the red cell anion exchange protein. Annu Rev Biochem. 1986;55:511–538. doi: 10.1146/annurev.bi.55.070186.002455. [DOI] [PubMed] [Google Scholar]
- Jennings M. L., Anderson M. P., Monaghan R. Monoclonal antibodies against human erythrocyte band 3 protein. Localization of proteolytic cleavage sites and stilbenedisulfonate-binding lysine residues. J Biol Chem. 1986 Jul 5;261(19):9002–9010. [PubMed] [Google Scholar]
- Kaul R. K., Murthy S. N., Reddy A. G., Steck T. L., Kohler H. Amino acid sequence of the N alpha-terminal 201 residues of human erythrocyte membrane band 3. J Biol Chem. 1983 Jul 10;258(13):7981–7990. [PubMed] [Google Scholar]
- Kay M. M., Tracey C. M., Goodman J. R., Cone J. C., Bassel P. S. Polypeptides immunologically related to band 3 are present in nucleated somatic cells. Proc Natl Acad Sci U S A. 1983 Nov;80(22):6882–6886. doi: 10.1073/pnas.80.22.6882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kopito R. R., Andersson M., Lodish H. F. Structure and organization of the murine band 3 gene. J Biol Chem. 1987 Jun 15;262(17):8035–8040. [PubMed] [Google Scholar]
- Kopito R. R., Lodish H. F. Primary structure and transmembrane orientation of the murine anion exchange protein. Nature. 1985 Jul 18;316(6025):234–238. doi: 10.1038/316234a0. [DOI] [PubMed] [Google Scholar]
- Kozak M. Point mutations define a sequence flanking the AUG initiator codon that modulates translation by eukaryotic ribosomes. Cell. 1986 Jan 31;44(2):283–292. doi: 10.1016/0092-8674(86)90762-2. [DOI] [PubMed] [Google Scholar]
- Kyte J., Doolittle R. F. A simple method for displaying the hydropathic character of a protein. J Mol Biol. 1982 May 5;157(1):105–132. doi: 10.1016/0022-2836(82)90515-0. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Low P. S., Westfall M. A., Allen D. P., Appell K. C. Characterization of the reversible conformational equilibrium of the cytoplasmic domain of erythrocyte membrane band 3. J Biol Chem. 1984 Nov 10;259(21):13070–13076. [PubMed] [Google Scholar]
- Mawby W. J., Findlay J. B. Characterization and partial sequence of di-iodosulphophenyl isothiocyanate-binding peptide from human erythrocyte anion-transport protein. Biochem J. 1982 Sep 1;205(3):465–475. doi: 10.1042/bj2050465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Melton D. A., Krieg P. A., Rebagliati M. R., Maniatis T., Zinn K., Green M. R. Efficient in vitro synthesis of biologically active RNA and RNA hybridization probes from plasmids containing a bacteriophage SP6 promoter. Nucleic Acids Res. 1984 Sep 25;12(18):7035–7056. doi: 10.1093/nar/12.18.7035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moon R. T., Ngai J., Wold B. J., Lazarides E. Tissue-specific expression of distinct spectrin and ankyrin transcripts in erythroid and nonerythroid cells. J Cell Biol. 1985 Jan;100(1):152–160. doi: 10.1083/jcb.100.1.152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murthy S. N., Liu T., Kaul R. K., Köhler H., Steck T. L. The aldolase-binding site of the human erythrocyte membrane is at the NH2 terminus of band 3. J Biol Chem. 1981 Nov 10;256(21):11203–11208. [PubMed] [Google Scholar]
- Ngai J., Stack J. H., Moon R. T., Lazarides E. Regulated expression of multiple chicken erythroid membrane skeletal protein 4.1 variants is governed by differential RNA processing and translational control. Proc Natl Acad Sci U S A. 1987 Jul;84(13):4432–4436. doi: 10.1073/pnas.84.13.4432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pasternack G. R., Anderson R. A., Leto T. L., Marchesi V. T. Interactions between protein 4.1 and band 3. An alternative binding site for an element of the membrane skeleton. J Biol Chem. 1985 Mar 25;260(6):3676–3683. [PubMed] [Google Scholar]
- Reithmeier R. A. Fragmentation of the band 3 polypeptide from human erythrocyte membranes. Size and detergent binding of the membrane-associated domain. J Biol Chem. 1979 Apr 25;254(8):3054–3060. [PubMed] [Google Scholar]
- Rigby P. W., Dieckmann M., Rhodes C., Berg P. Labeling deoxyribonucleic acid to high specific activity in vitro by nick translation with DNA polymerase I. J Mol Biol. 1977 Jun 15;113(1):237–251. doi: 10.1016/0022-2836(77)90052-3. [DOI] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steck T. L., Koziarz J. J., Singh M. K., Reddy G., Köhler H. Preparation and analysis of seven major, topographically defined fragments of band 3, the predominant transmembrane polypeptide of human erythrocyte membranes. Biochemistry. 1978 Apr 4;17(7):1216–1222. doi: 10.1021/bi00600a013. [DOI] [PubMed] [Google Scholar]
- Steck T. L., Ramos B., Strapazon E. Proteolytic dissection of band 3, the predominant transmembrane polypeptide of the human erythrocyte membrane. Biochemistry. 1976 Mar 9;15(5):1153–1161. doi: 10.1021/bi00650a030. [DOI] [PubMed] [Google Scholar]
- Steck T. L. The organization of proteins in the human red blood cell membrane. A review. J Cell Biol. 1974 Jul;62(1):1–19. doi: 10.1083/jcb.62.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsai I. H., Murthy S. N., Steck T. L. Effect of red cell membrane binding on the catalytic activity of glyceraldehyde-3-phosphate dehydrogenase. J Biol Chem. 1982 Feb 10;257(3):1438–1442. [PubMed] [Google Scholar]
- Weinstein R. S., Khodadad J. K., Steck T. L. Fine structure of the band 3 protein in human red cell membranes: freeze-fracture studies. J Supramol Struct. 1978;8(3):325–335. doi: 10.1002/jss.400080310. [DOI] [PubMed] [Google Scholar]
- Wieslander L. A simple method to recover intact high molecular weight RNA and DNA after electrophoretic separation in low gelling temperature agarose gels. Anal Biochem. 1979 Oct 1;98(2):305–309. doi: 10.1016/0003-2697(79)90145-3. [DOI] [PubMed] [Google Scholar]
- Wieth J. O., Bjerrum P. J., Borders C. L., Jr Irreversible inactivation of red cell chloride exchange with phenylglyoxal, and arginine-specific reagent. J Gen Physiol. 1982 Feb;79(2):283–312. doi: 10.1085/jgp.79.2.283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wieth J. O., Bjerrum P. J. Titration of transport and modifier sites in the red cell anion transport system. J Gen Physiol. 1982 Feb;79(2):253–282. doi: 10.1085/jgp.79.2.253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zaki L. Inhibition of anion transport across red blood cells with 1,2-cyclohexanedione. Biochem Biophys Res Commun. 1981 Mar 16;99(1):243–251. doi: 10.1016/0006-291x(81)91738-1. [DOI] [PubMed] [Google Scholar]