Abstract
Prostate cancer is the most commonly diagnosed malignancy in men. While tumors initially respond to androgen-deprivation therapy, the standard care for advanced or metastatic disease, tumors eventually recur as castration-resistant prostate cancer. Upregulation of the insulin-like growth factor receptor type 1 (IGF-1R) signaling axis drives growth and progression of prostate cancer by promoting proliferation, survival, and angiogenesis. Ganitumab (formerly AMG 479) is a fully human antibody that inhibits binding of IGF-1 and IGF-2 to IGF-1R. We evaluated the therapeutic value of ganitumab in several pre-clinical settings including androgen-dependent prostate cancer, castration-resistant prostate cancer, and in combination with androgen-deprivation therapy. Ganitumab inhibited IGF-1 induced phosphorylation of the downstream effector AKT and reduced proliferation of multiple androgen-dependent and castration-resistant human prostate cancer cell lines in vitro. Ganitumab inhibited androgen-dependent VCaP xenograft growth and increased tumor doubling time from 2.3±0.4 weeks to 6.4±0.4 weeks. Ganitumab blocked growth of castration-resistant VCaP xenografts for over 11.5 weeks of treatment. In contrast, ganitumab did not have appreciable effects on the castration-resistant CWR-22Rv1 xenograft model. Ganitumab was most potent against VCaP xenografts when combined with complete androgen-deprivation therapy (castration). Tumor volume was reduced by 72% after 4 weeks of treatment and growth suppression was maintained over 16 weeks of treatment. These data suggest that judicious use of ganitumab particularly in conjunction with androgen-deprivation therapy may be beneficial in the treatment of prostate cancer.
Keywords: Prostate Cancer, Growth Factors and Receptors, Ganitumab, IGF-1R, androgen-deprivation therapy
Introduction
Prostate cancer is the most commonly diagnosed cancer in men and is the second leading cause of cancer-related deaths in men each year (1). Androgen-deprivation therapy is and has been the gold standard of care for advanced or metastatic prostate cancer for decades. While this treatment strategy initially shows benefit, eventually tumors recur as castration-resistant prostate cancer (CRPC) for which there are limited treatment options with only modest survival benefit.
The insulin-like growth factor receptor type I (IGF-1R) and native ligands, IGF-1 and IGF-2, are dysregulated in a variety of cancers including prostate cancer (2–5). Signaling through the IGF-1R plays an important role in many cellular processes including: mitogenesis, apoptosis, proliferation, and angiogenesis (Supplementary Figure 1). The downstream effects of IGF-1R activation include PI3K/AKT and MEK/ERK signaling cascades (6, 7). Elevated levels of serum IGF-1, the most potent activator of IGF-1R, has been correlated with an increased risk of prostate cancer (8, 9). IGF-1 and IGF-1R are increased with progression to castration-resistance in vivo (10). In clinical specimens, IGF-1R expression is elevated in primary prostate cancer versus benign prostatic epithelium (11). Studies with an IGF-1R receptor inhibitor, the antibody cixutumumab (formerly IMC-A12), showed positive results against LuCaP 35 prostate cancer xenografts alone and in conjunction with androgen-deprivation therapy or docetaxel (12–14). A recently completed phase 2 clinical trial with figitumumab (formerly CP-751,871), an IgG2 IGF-1R antibody inhibitor that blocks IGF-1 binding and promotes receptor internalization, in patients with localized prostate cancer showed a decrease in serum levels of prostate specific antigen (PSA) (15, 16). These studies support further investigation of inhibition of IGF-1R signaling with ganitumab in robust models of human prostate cancer.
Ganitumab (formerly AMG 479) is a fully human antibody (IgG1) against IGF-1R. Ganitumab reduces IGF-1R activation by binding the L2 domain (an extracellular leucine-rich domain that contributes to ligand binding) of IGF-1R thereby preventing both IGF-1 and IGF-2 interaction with the receptor (17, 18). Binding of ganitumab to IGF-1R also induces internalization and degradation of IGF-1R in vivo (18). Ganitumab does not interact with the closely related insulin receptor (INSR) but does inhibit hybrid IGF-1R/INSR receptors (18). Ganitumab has been studied in phase II trials for a variety of advanced tumors (19, 20). In phase I/Ib escalation studies, ganitumab was generally well-tolerated when administered to patients with advanced solid tumors both as a single agent and in combination with either the multi-kinase inhibitor sorafenib, either of the EGFR inhibitors panitumumab or erlotinib, or gemcitabine. These combination therapies as well as ganitumab as a single agent displayed promising anti-tumor effects in patients and resulted in few dose-limiting toxicities, and generally mild adverse events including fatigue, nausea, vomiting or chills (21, 22). To date, ganitumab has shown signs of efficacy against pancreatic cancer and Ewing’s sarcoma, yet there has been minimal evaluation of ganitumab for prostate cancer.
In this study, we tested the effect of ganitumab on several human prostate cancer cell lines both in vitro and in vivo. VCaP is an androgen-dependent human prostate cancer cell line that like ~50% of prostate cancers is wild-type for PTEN (a negative regulator for AKT activator PI3K) (23, 24). As observed in human prostate cancer, VCaP cells progress to castration-resistance in vivo (25, 26). VCaP also expresses the TMPRSS2:ERG fusion gene, which is seen in ~50% of primary prostate cancer specimens and may promote prostate cancer progression (27). Another androgen-dependent human prostate cancer cell line, LNCaP, does not express active PTEN (28). CWR-22Rv1 (22Rv1) and CWR-R1 are castration-resistant, PTEN wild-type and express both full length androgen receptor (AR) as well as constitutively active AR splice variants, which lack the ligand binding domain (termed ARΔLBDs) (29, 30). Utilizing these established models of prostate cancer, we assessed the effectiveness of ganitumab alone and in conjunction with complete androgen-deprivation therapy (castration) as a treatment for androgen-dependent prostate cancer, advanced CRPC, and progression to CRPC. We show that ganitumab inhibits growth of both androgen-dependent and castration-resistant VCaP xenografts. Ganitumab does not affect growth of aggressive castration-resistant 22Rv1 xenografts. Lastly, we found that ganitumab is highly effective against VCaP xenografts when combined with androgen-deprivation therapy.
Materials and Methods
Cell culture and chemical reagents
The human prostate cancer cell lines LNCaP.FGC (CRL 1740; batch F-11701) and CWR-22Rv1 (CRL-2505, batch 4484055) were obtained from American Type Culture Collection (ATCC). LNCaP and 22Rv1 cells were authenticated and certified by ATCC. ATCC ensures each cell line is negative for mycoplasma, bacteria, and fungi contamination; confirms species identity; and performs DNA profiling and cytogenetic analysis to authenticate each cell line. CWR-R1 cells were provided by Dr. Elizabeth Wilson (University of North Carolina, Chapel Hill, NC) in July 2011 and tested for prostate and cell line specific characteristics including AR and AR splice variants (31, 32). VCaP cells were provided in March 2009 by Dr. Kenneth Pienta (University of Michigan, Ann Arbor, MI), VCaP cells were tested for prostate and cell line specific characteristics including AR, TMPRSS:ERG fusion, and PSA. VCaP cells were negative for mycoplasma, human T-lymphotropic virus, hepatitis (A, B, and C), and HIV. All cell lines were used within 6 months of resuscitation. VCaP, LNCaP, 22Rv1, and CWR-R1 cells were maintained as previously described (31, 33). Ganitumab was supplied by Amgen Inc. Recombinant IGF-1 was obtained from Peprotech.
In Vitro AKT Phosphorylation Studies
VCaP and 22Rv1 were plated in medium containing 10% FBS. At ~70% confluence, medium were replaced with fresh medium supplemented with 5% charcoal-stripped serum and cells cultured for an additional 24 hours. Ganitumab (0–1000 nM) was added 90 minutes prior to treatment with 1nM IGF-1. Cells were harvested in RIPA buffer 30 minutes after IGF-1 administration and immunoblotted.
In Vitro Proliferation Assays
LNCaP, 22Rv1, or CWR-R1 cells were seeded in 24-well plates (BD Falcon) (2x104 per well) in RPMI/10% FBS. VCaP cells were seeded in 24-well plates (6x104 per well) in DMEM/10% FBS. The following day, cells were washed with PBS, and media supplemented with 2% FBS and ganitumab was added and incubated for 7 days. Cells were trypsinized (Cellgro), mixed with trypan blue (Gibco), and live cells were counted using a hemocytometer. Data represent at least three independent experiments performed in triplicate.
In Vitro Apoptosis Studies
6.5x105 VCaP, 22Rv1, CWR-R1, or LnCaP were plated in the appropriate medium containing 10% FBS in 60mm plates. The following day, cell monolayers were washed with PBS, and medium supplemented with 2%FBS containing 500nM ganitumab or vehicle was added. Plates were incubated for 3 days, harvested in RIPA buffer and immunoblotted. Data represent at least two independent experiments.
Western Blots
Western blots were performed as previously described (31, 33). Antibodies against phospho-AKT (S473), total AKT, IGF-1R, and cleaved PARP were obtained from Cell Signaling. INSR, actin (Santa Cruz), HRP-conjugated secondary antibodies were obtained from (Santa Cruz). Densitometry was performed using Adobe Photoshop CS3.
Xenograft studies
Studies involving animals were conducted in a manner approved by the University of Miami Animal Care and Use Committee. VCaP xenografts were established as previously described (31, 33). 22Rv1 cells (2x106) were injected subcutaneously into both hind flanks of castrated nude mice (Harlan) with Matrigel (BD Biosciences). Tumor volumes were assessed as previously described (33). Mice were euthanized when tumors reached a maximal volume of 1000mm3 or 10 weeks post-treatment for androgen-replete VCaP xenografts; 1000 mm3 or 17 weeks post-treatment for castration-resistant VCaP xenografts; and 1000 mm3 or 7 weeks post-treatment for castration-resistant 22Rv1 xenografts; upon which tumors were excised, and flash frozen. Levels of circulating PSA were quantified from serum samples by an enzyme-linked immunosorbent assay (Biocheck Inc.). Levels of IGF-1R, INSR, phospho-AKT and total AKT were measured using either MSD multiplex assays (Meso Scale Discovery) as previously described (5, 18) or densitometry as described above. Ganitumab (Amgen) and control anti-streptavidin IgG1 antibody (Amgen) were diluted in sterile PBS and administered via intraperitoneal injections at a dosage of 300μg twice weekly as specified.
Results
Ganitumab inhibits IGF-1R signaling in androgen-dependent and castration-resistant prostate cancer cells in vitro
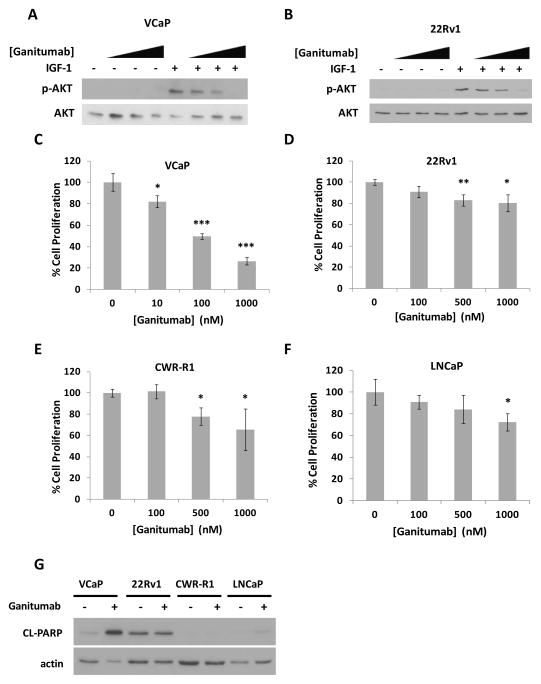
To assess the potential use of ganitumab for treatment of both androgen-dependent and castration-resistant prostate cancer, we determined whether androgen-dependent VCaP and castration-resistant 22Rv1 were sensitive to IGF-1 stimulation. Serum deprivation decreased AKT phosphorylation while IGF-1 treatment induced phosphorylation of AKT, a downstream effector of IGF-1/IGF-1R signaling in both VCaP and 22Rv1 cell lines (Figure 1a–b). Administration of ganitumab reduced IGF-1-induced phosphorylation of AKT in both cell lines. These results indicate that ganitumab inhibited IGF-1/IGF-1R signaling in both androgen-dependent and castration-resistant prostate cancer cells.
Figure 1. Analysis of ganitumab effects on IGF-1R signaling and prostate cancer cell proliferation in vitro.
VCaP and 22Rv1 were cultured in serum deprived media for 24 hours, and treated with ganitumab [0 nM, 10 nM, 100 nM, 1000 nM] for 90 minutes prior to treatment with 1nM IGF-1 for 30 minutes. Lysates were probed by western blot for phospho-AKT and total-AKT in VCaP (A) and 22Rv1 (B). The effects of seven day ganitumab treatment (0–1000 nM) on prostate cancer cell proliferation in 2% FBS containing media [(C), 22Rv1 (D), CWR-R1 (E), and LNCaP (F)] were determined. (G) VCaP, 22Rv1, CWR-R1 and LNCaP were cultured in medium containing 2%FBS for 72 hours with 500 nM ganitumab or control. Lysates were immunoblotted for cleaved PARP and actin. (C–E) are representative of at least 3 experiments performed in triplicate. (F) Represents 3 combined experiments. (G) is representative of 2 independent experiments. All data are shown ± SD. (*p<.05, **<.01, ***<.001, Two-tailed Student’s t-test)
Ganitumab inhibits growth of androgen-dependent and castration-resistant prostate cancer in vitro
To assess the effect of ganitumab on prostate cancer cell proliferation, we administered ganitumab to several prostate cancer cell line models in vitro. VCaP cells are wild type for PTEN and androgen-dependent; 22Rv1 and CWR-R1 are castration-resistant, express wild-type PTEN and full length AR as well as constitutively active AR splice variants; LNCaP do not express active PTEN and are androgen-dependent. The proliferation of VCaP, 22Rv1, CWR-R1 and LNCaP cells was inhibited by ganitumab to varying degrees (Figure 1c–f). VCaP cells were the most sensitive to ganitumab. Ganitumab increased the level of cleaved PARP, a late-stage marker of apoptosis, in VCaP cells, but had no effect on 22Rv1, CWR-R1 or LNCaP cell lines (Figure 1g). We chose an androgen-dependent (VCaP) and a castration-resistant (22Rv1) model for further studies in vivo.
Ganitumab inhibits androgen-dependent prostate cancer growth in vivo
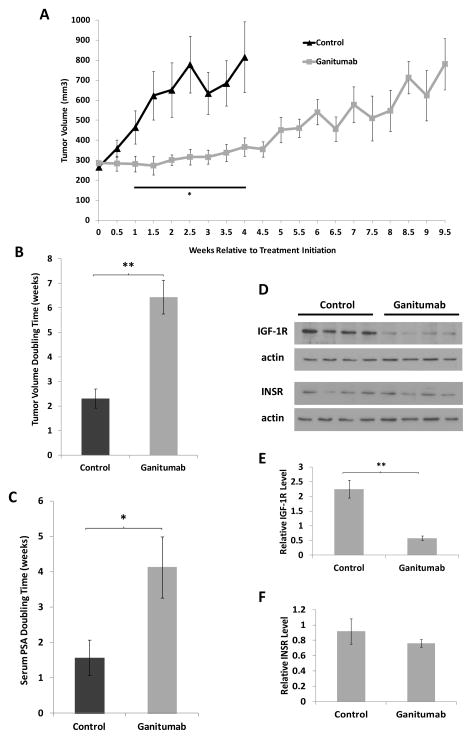
We utilized androgen-dependent VCaP xenografts to evaluate ganitumab treatment in a more physiological setting. VCaP human prostate cancer cells were injected subcutaneously into intact mice. When tumors reached an average volume of 260–320 mm3, mice were randomized into two groups and treatment with ganitumab or control antibody at a dose of 300μg twice weekly via intraperitoneal injections commenced. The ganitumab dosage was based on safety and efficacy shown in previous mouse xenograft studies (5, 18). Ganitumab inhibition of tumor growth was observed beginning one week after treatment initiation. Growth inhibition was maintained as shown by the tumor doubling time, which was strikingly increased from 2.3±0.4 weeks in control-treated mice to 6.4±0.4 weeks in ganitumab-treated mice (Figure 2b).
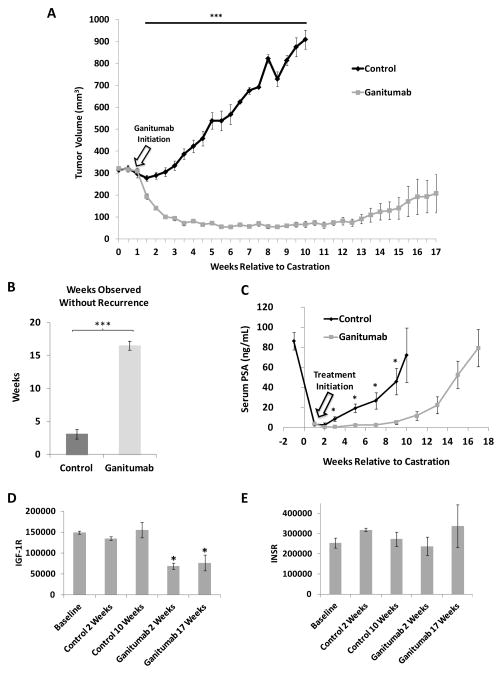
Figure 2. Ganitumab inhibits the growth of androgen-dependent VCaP xenografts.
VCaP xenografts were established in intact (androgen-replete) mice. When tumors reached an average volume of 260–320 mm3, treatment with ganitumab or control antibody was initiated. (A) Tumor volumes (mm3) are shown relative to treatment initiation (n=3–4 for control; n=4–8 for ganitumab). (B) Tumor doubling times for control and ganitumab treated mice are shown (n=4 for control; n=8 for ganitumab). (C) Serum PSA doubling times are shown (n=4 for control; n=8 for ganitumab). (D) Xenograft lysates were immunoblotted for IGF-1R, INSR, and actin for control-treated and ganitumab treated tumors. Western blots were quantified by densitometry and relative IGF-1R (E) and relative INSR (F) normalized to actin are shown. All data are displayed ± SEM. (*p<.05, **<.01, Two-tailed Student’s t-test)
The clinically relevant biomarker prostate-specific antigen (PSA) was also assessed throughout the duration of the experiment. The PSA gene is regulated by AR and circulating PSA is used to assess biochemical recurrence in patients (34). Serum PSA doubling time was increased from 1.6±0.5 weeks in control-treated mice to 4.1±0.9 weeks ganitumab-treated mice (Figure 2c) which paralleled changes in tumor volume. Levels of IGF-1R were significantly decreased in ganitumab-treated mice compared to control-treated mice, and INSR levels were unaffected by ganitumab treatment (Figure 2d–f). Ganitumab had no effect on levels of AR (Supplementary Figure 2a). These data show that ganitumab slows the growth of this androgen-dependent prostate cancer model.
Effect of ganitumab treatment on castration-resistant prostate cancer in vivo
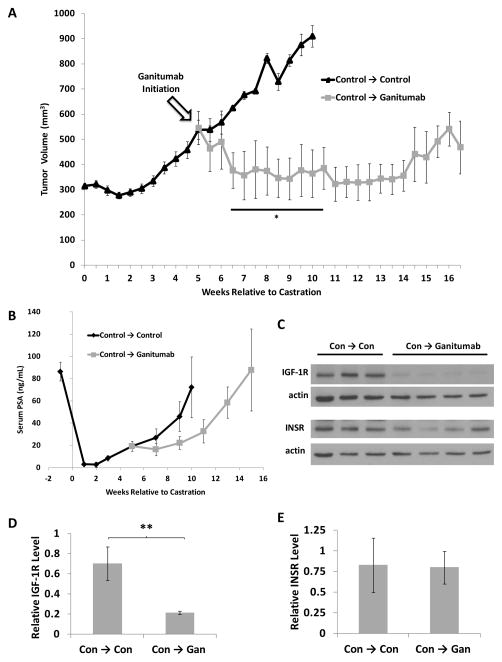
To test whether ganitumab may be effective for CRPC, we utilized two xenograft models. VCaP xenografts are initially androgen-dependent, but recur as castration-resistant xenografts after murine castration (26, 33). VCaP cells were injected into the flanks of intact SCID mice and mice were castrated when average tumor volumes reached 260~320 mm3. Castrated mice with tumors averaging 538 mm3 were randomized into 2 groups and treatment with ganitumab or continued control antibody was initiated. This experimental design allowed evaluation of ganitumab effectiveness for tumors that had recently progressed to castration-resistance.
Ganitumab halted tumor growth for the remainder of the experiment (11.5 weeks) whereas the tumors of the control-treated mice continued to grow rapidly (Figure 3a). The ganitumab-treated mice showed decreased serum PSA relative to the control-treated mice at all comparable time points (Figure 3b). Although tumor volume did not significantly increase in the ganitumab-treated mice, the serum PSA increased at later time points (15 weeks post-castration). Levels of IGF-1R were significantly decreased in ganitumab-treated mice compared to control-treated mice (Figure 3c,d). Levels of INSR, AR, and ARΔLBD were unaffected by ganitumab treatment (Figure 3c,d) (Supplementary Figure 2b).
Figure 3. Ganitumab inhibits the growth of castration-resistant VCaP xenografts.
Castration-resistant VCaP xenografts were established and mice were treated with control antibody beginning one week after castration. Five weeks after castration with an average tumor volume of 538 mm3, mice were randomized into 2 groups and treatment with ganitumab was initiated (Control → Ganitumab) or control antibody was continued (Control → Control). (A) Tumor volumes (mm3) are shown relative to time of castration (n=4–5 for control; n=3–4 for ganitumab). (B) Serum PSA (ng/mL) levels are shown (n=18 for pre-treatment; n=3–4 for control; n=3 for ganitumab). (C) Xenograft lysates were immunoblotted for IGF-1R, INSR, and actin for Control → Control and Control → Ganitumab tumors. Blots were quantified by densitometry and relative IGF-1R (D) and relative INSR (E) normalized to actin are shown. All data are displayed ± SEM. (*p<.05, Two-tailed Student’s t-test)
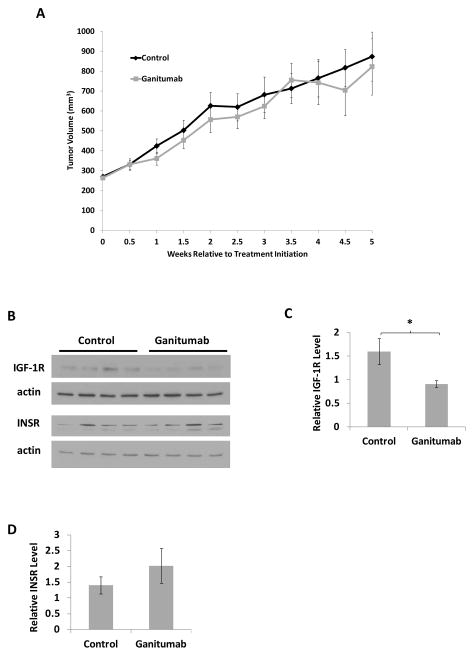
22Rv1 is a castration-resistant model of prostate cancer that grows readily in androgen-deprived environments including xenografts in castrated mice (29). 22Rv1 cells were injected into the hind flanks of castrated nude mice. Once tumors reached an average volume of 260~320 mm3, treatment with ganitumab or control antibody commenced for 5 weeks. In this highly aggressive model of prostate cancer, ganitumab had no appreciable effect on tumor growth (Figure 4a). There was no difference in weight between control and ganitumab-treated mice (Supplementary Table 1a). Protein levels of IGF-1R were significantly decreased in ganitumab-treated mice compared to control treated mice (Figure 4b–c). INSR, AR, and ARΔLBD levels were not significantly changed after treatment with ganitumab (Figure 4b,d) (Supplementary Figure 2c).
Figure 4. Ganitumab does not affect the growth of well-established castration-resistant 22Rv1 xenografts.
22Rv1 xenografts were established in castrated nude mice. When tumors reached an average volume of 260–320 mm3 treatment with ganitumab or control antibody was initiated. (A) Tumor volumes are shown for ganitumab and control treated mice (n=4–14 for control, n=3–14 for ganitumab). (B) Xenograft lysates were immunoblotted for IGF-1R, INSR, and actin. (C) Blots were quantifed by densitometry and relative IGF-1R levels (C) and relative INSR levels (D) normalized to actin are shown. All data are displayed ± SEM. (*p<.05, Two-tailed Student’s t-test)
Ganitumab combined with androgen-deprivation is a highly effective treatment regimen
The standard of care for advanced or metastatic prostate cancer is androgen-deprivation therapy. Since ganitumab showed inhibitory effects on both androgen-dependent and castration-resistant VCaP prostate cancer xenografts, we tested whether combining ganitumab with androgen-deprivation therapy would show added benefits. VCaP cells were implanted into the hind flanks of SCID mice, mice were castrated when average tumor volume reached 260–320 mm3. One week later, treatment with ganitumab or control antibody was initiated. A single dose of ganitumab resulted in an immediate decrease in tumor volume compared to control-treated mice after castration (Figure 5a). Tumors of ganitumab-treated mice remained significantly smaller than those of control-treated mice throughout the remainder of the experiment (16 weeks). In addition to decreased tumor volume, combining androgen-deprivation therapy with ganitumab lengthened time to recurrence in the 17 week experiment from 3.1±0.7 weeks (control-treated) to 16.5±0.7 weeks (ganitumab-treated) with recurrence defined as tumor volume surpassing that at time of castration as determined for each tumor individually (Figure 5b). Serum PSA decreased to a greater extent with androgen-deprivation and ganitumab when compared to androgen-deprivation combined with control antibody (Figure 5c). Between weeks 11 and 15 there was a sharp increase in serum PSA which was not reflected in an increase in tumor volume. Ganitumab-treated mice lost a small proportion of body weight (~10%) whereas control-treated mice did not lose weight during the experiment (Supplementary table 1b). Total levels of IGF-1R protein in xenografts were decreased in the ganitumab-treated mice compared to intact mice and control-treated mice at 2 and 17 weeks post-castration (Figure 5d), which is consistent with previous studies in pancreatic cancer and Ewing’s sarcoma (5, 18). INSR levels remained unchanged under all conditions tested (Figure 5e). Levels of phosphorylated AKT and total AKT were also unaffected by androgen-deprivation, ganitumab treatment, or combined androgen-deprivation and ganitumab treatment (Supplementary Figure 3). AR and constitutively active splice variant AR3 (AR-V7) mRNA levels were increased in end stage ganitumab-treated tumors (10 weeks post-castration) compared to control-treated tumors (17 weeks post-castration) (Supplementary Figure 4). These data show that combination of ganitumab and androgen-deprivation substantially decreased tumor growth and delayed progression to castration resistance.
Figure 5. Combining ganitumab with androgen-deprivation therapy most effectively inhibits VCaP xenografts.
VCaP xenografts were established in intact mice, and mice were castrated when tumors reached an average volume of 260–320 mm3. One week after castration mice were randomized into 2 groups and treatment with ganitumab or control antibody was initiated. (A) Tumor volumes (mm3) are shown relative to time of castration for control and ganitumab treated mice (n=4–14 for control; n=5–16 for ganitumab) (control-treated tumor volumes are those from figure 3a). (B) Weeks observed without recurrences were calculated and are shown above (n=11 for control; n=5 for ganitumab) (C) Serum PSA (ng/mL) is shown (n=3–13 for control; n=5–11 for ganitumab). Relative IGF-1R (D) INSR (E) protein levels detected by MSD multiplex analysis are shown for intact (pre-castrate baseline), control-treated, and ganitumab-treated mice relative to time of castration (n=3~5 per group). All data are displayed ± SEM. (*p<.05, ***<.001, Two-tailed Student’s t-test)
Reversibility and durability of treatment with ganitumab combined with androgen-deprivation therapy
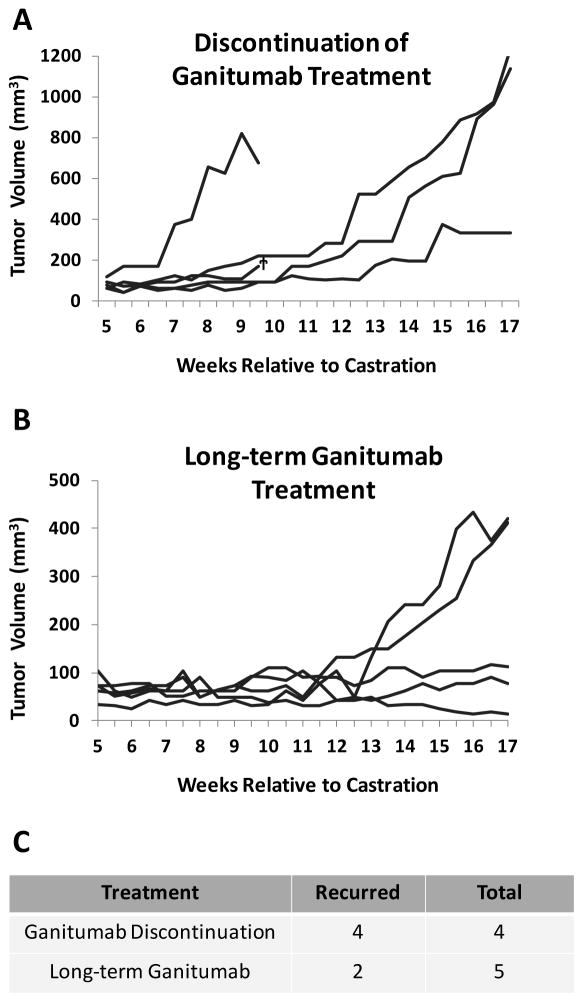
We evaluated the durability of combining androgen-deprivation therapy with ganitumab treatment by examining long-term ganitumab treatment. Reversibility of ganitumab treatment was also assessed. VCaP xenografts were established and mice castrated when tumors reached an average volume of 260–320 mm3. Treatment with ganitumab commenced one week after castration. Five weeks after castration, mice were randomized into two groups. One group remained on ganitumab (to evaluate durability of response) and ganitumab was discontinued in the other group (to evaluate reversibility of response). In the ganitumab discontinued cohort, all tumors (n=4) recurred within 11 weeks (Figure 6a). Two of 5 tumors recurred in the long-term ganitumab treatment arm (Figure 6b). Thus, while combination therapy with androgen-deprivation and ganitumab is highly effective, tumors recur after ganitumab cessation and some tumors recur during long-term ganitumab treatment.
Figure 6. Evaluation of durability and reversibility of combined ganitumab and androgen-deprivation therapy on VCaP xenografts.
VCaP xenografts were established in intact mice, and mice were castrated when tumors reached an average volume of 260–320 mm3. Beginning one week after castration mice were treated with ganitumab for four weeks. Mice were randomized into 2 groups and treatment with control antibody was initiated or ganitumab was continued. (A) Discontinuation of ganitumab after 4 weeks (5 weeks post castration) resulted in recurrence of all VCaP tumors (n=4) († – deceased mouse due to causes unrelated to treatment). (B) Long-term treatment with ganitumab resulted in recurrence in 2 of 5 tumors. (Average tumor volume is reflected in figure 5a.) (C) Quantification of recurrence after ganitumab discontinuation (reversibility) and long-term ganitumab treatment (durability) is shown.
Discussion
A major problem in the care of advanced prostate cancer is the lack of a durable response to androgen-deprivation therapy. Ganitumab is a fully human antibody that targets and inhibits IGF-1R by blocking ligand binding that has shown promising efficacy in the treatment of pancreatic cancer and Ewing’s sarcoma (19, 21). Here we show that ganitumab represents a new therapeutic strategy for the treatment of prostate cancer particularly when combined with the standard of care, androgen-deprivation. Ganitumab effectively inhibited the IGF-1R signaling axis in multiple human prostate cancer cell lines by blocking IGF-1 induced AKT phosphorylation. Ganitumab also decreased proliferation in vitro, albeit to varying extents, of both androgen-dependent and castration-resistant human prostate cancer cell lines. VCaP cells exhibited the highest sensitivity to ganitumab of all tested cell lines. Ganitumab increased apoptosis in VCaP cells but had minimal effects on cleaved PARP in LNCaP, CWR-R1, and 22Rv1 cells.
PTEN has been suspected to play a role in the efficacy of IGF-1R inhibitors as PTEN is a negative regulator of the PI3K/AKT pathway, which is major downstream effector of IGF-1R (7, 23). PTEN deficiencies lead to highly active, hyperphosphorylated AKT in the absence of exogenous signaling (35). PTEN is not expressed or is inactive in a significant number of prostate cancers (28, 36). The prostate cancer cell model LAPC-4, which harbors a wild-type PTEN, was recently shown to be insensitive to ganitumab in vitro (37). Harboring wild-type PTEN does not appear to be sufficient for sensitivity to ganitumab as also evidenced by a lack of response of 22Rv1 xenografts in castrated mice. LuCaP 35, a PTEN-null prostate cancer cell model, is inhibited through blockade of the IGF-1R signaling axis (12–14). These findings are in line with results from a recent phase Ib clinical trial in which PTEN status in advanced solid tumors did not correlate with response to ganitumab (22). Thus, the inhibitory effect of ganitumab does not appear to be solely due to blockade of the IGF-1R/AKT signaling cascade and PTEN status alone is unlikely to serve as a biomarker for ganitumab response.
The case-control study ProtecT associated changes in the circulating IGF-1 signaling axis that promote increased IGF signaling with prostate cancer detected through PSA screening. This large-scale study conducted in the United Kingdom showed that PSA-diagnosed prostate cancer is positively associated with circulating levels of IGFBP-2, IGFBP-3 and possibly with IGF-2 but not with IGF-1 (38). Thus the ProtecT study provides support for the use of IGF-1R inhibitors, such as ganitumab, which inhibits binding of both IGF-1 and IGF-2 to IGF-1R, as a treatment for prostate cancer.
A recent phase II study of treatment-naïve patients with localized prostate cancer showed that preoperative administration of figitumumab, an IGF-1R inhibitor, decreased serum PSA and AR expression (15). Similarly, we found that ganitumab inhibited VCaP xenograft growth and decreased serum PSA under androgen-replete conditions. However, we did not observe differences in AR or AR splice variant levels in end stage tumors of ganitumab-treated compared to control mice. VCaP cells express high levels of AR that do not appear to be regulated by IGF-1R signaling, but nonetheless ganitumab decreased PSA indicative of diminished AR activity. These data suggest that ganitumab may be clinically beneficial prior to androgen-deprivation therapy in the treatment of prostate cancer, although a role for AR is currently unclear.
Castration-resistant VCaP xenograft growth was also inhibited by ganitumab treatment. Ganitumab prevented significant increases in tumor volume in these castration-resistant xenografts throughout the duration of treatment (11.5 weeks). Since there are limited treatment options for established castration resistant tumors, blocking IGF-1R with ganitumab may be an effective strategy in this setting.
Ganitumab treatment did not inhibit castration-resistant growth of 22Rv1 xenografts (Fig. 2) or growth of 22Rv1 tumors in intact (androgen-replete) mice (37). Thus, 22Rv1 cells exhibit de novo resistance to ganitumab in vivo. In contrast, VCaP xenografts were strikingly growth inhibited in the presence or absence of androgen in vivo. These cell lines are distinct. 22Rv1 are an aggressive CRPC line developed by serial passage of androgen-dependent CWR22 xenografts in castrated mice (29). 22Rv1 proliferate in the absence of serum (31) whereas VCaP cells rely on serum for growth and are initially androgen-dependent taking several weeks to achieve castration-resistance in vivo (33). VCaP express higher levels of IGF-1R compared to 22Rv1. VCaP and 22Rv1 also express different levels of AR (wild type in VCaP, mutated in 22Rv1) and ARΔLBDs. VCaP express higher levels of ERG than 22Rv1 and express the TMPRSS2:ERG fusion protein, seen in ~50% of prostate cancer specimens, whereas 22Rv1 do not contain the TMPRSS2:ERG fusion (39, 40). These data suggest that 22Rv1 and VCaP rely on different signals for survival and proliferation, which may underlie differential response to ganitumab. Thus, more mechanism-based research is needed in order to stratify patients based on molecular and genetic features of their prostate tumors.
In our preclinical study, ganitumab was most effective when combined with a more complete androgen-deprivation therapy. To date, no clinical trials have combined complete androgen-deprivation therapy (using abiraterone or the AR antagonist enzalutamide) with IGF-1R inhibition. Based on clinical trials, ganitumab is also suitable and well-tolerated alone and in combination with a variety of chemotherapeutic agents including the multi-kinase inhibitor sorafenib, EGF-R inhibitors panitumumab and erlotinib, or the nucleoside analogue gemcitabine (22). In fact, the only prostate cancer patient who participated in this study showed a partial response (~60% maximum change from baseline tumor size) in response to ganitumab plus gemcitabine.
PSA is a clinical biomarker used extensively to evaluate incidence and recurrence of prostate cancer. Recent studies have shown wide variability in PSA levels and suggest that PSA may not be suitable as a surrogate end point in clinical trials involving non-cytotoxic drugs such as ganitumab (41). We found that PSA levels were increased with no parallel increase in tumor volume, AR expression, or ARΔLBD expression in ganitumab-treated mice harboring castration-resistant VCaP xenografts. Mice treated with both androgen-deprivation therapy and long-term ganitumab showed increased AR and AR3 mRNA levels compared to control-treated mice. However, our studies were only able to assess transcript levels in this setting, which are not necessarily indicative of protein expression or function (transcriptional activity). The molecular basis for the discrepancy between PSA levels and tumor volume in our preclinical models is not known but suggests that serum PSA may not be an ideal biomarker for clinical evaluation of recurrence during treatment with ganitumab.
Some VCaP xenografts acquired resistance to the combined treatment of ganitumab and androgen-deprivation (castration). Development of resistance to therapy is a major and common problem in cancer care. In models of Ewing’s sarcoma, upregulation of signaling by the closely related INSR is an important mechanism of resistance to ganitumab (5). However, we saw no increase in levels of INSR in any xenograft experiments or increased phospho-AKT levels when ganitumab was combined with androgen deprivation, thus it is unlikely that upregulation of INSR is a mechanism of resistance to ganitumab in either VCaP or 22Rv1 prostate cancer cell models. Since mTOR can potentiate compensatory pathways, co-administering ganitumab with an mTOR inhibitor may be effective in combating drug resistance. Further investigation into the mechanism of acquired resistance to ganitumab in prostate cancer is needed.
Our data suggest that ganitumab may be beneficial in the treatment of both androgen-dependent and some forms of CRPC. Importantly, ganitumab plus complete androgen-deprivation therapy was highly effective. Ganitumab is currently under investigation for the treatment of several tumor types either alone or in conjunction with targeted or cytotoxic agents. Here we showed one example of de novo resistance to ganitumab with the CRPC model of 22Rv1 cells and evidence of acquired resistance to ganitumab utilizing VCaP xenografts. The characteristics that give rise to de novo resistance and acquired resistance to ganitumab must be understood in order to identify prostate cancer patients who will have the greatest likelihood of response and to develop strategies to prevent or abrogate acquired resistance to ganitumab therapy.
Supplementary Material
Acknowledgments
We thank Dr. Maria Mudyri (University of California – Davis) for her expertise with the CWR-R1 cell line and Dr. Wayne Balkan (University of Miami) for instruction and assistance with mouse xenografts and surgery. We also thank Drs. Young-Ah Chung, Elaina Cajulis, and Frank Calzone of Amgen Inc. for technical assistance, expertise and support.
Grant Support: Funding for this research was provided by Amgen Inc and NIH grant R01CA132200 (KLB). CDF was supported by NIH training grant T32-HL007188.
Footnotes
Disclosure of Potential Conflicts of Interest: Pedro J. Beltran is an employee of and owns stock in Amgen, Inc.
Works Cited
- 1.Jemal A, Siegel R, Xu J, Ward E. Cancer statistics, 2010. CA Cancer J Clin. 2010;60:277–300. doi: 10.3322/caac.20073. [DOI] [PubMed] [Google Scholar]
- 2.Breuhahn K, Longerich T, Schirmacher P. Dysregulation of growth factor signaling in human hepatocellular carcinoma. Oncogene. 2006;25:3787–800. doi: 10.1038/sj.onc.1209556. [DOI] [PubMed] [Google Scholar]
- 3.Mendivil A, Zhou C, Cantrell LA, Gehrig PA, Malloy KM, Blok LJ, et al. AMG 479, a novel IGF-1-R antibody, inhibits endometrial cancer cell proliferation through disruption of the PI3K/Akt and MAPK pathways. Reprod Sci. 2011;18:832–41. doi: 10.1177/1933719111398501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Grothey A, Voigt W, Schober C, Muller T, Dempke W, Schmoll HJ. The role of insulin-like growth factor I and its receptor in cell growth, transformation, apoptosis, and chemoresistance in solid tumors. J Cancer Res Clin Oncol. 1999;125:166–73. doi: 10.1007/s004320050259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Beltran PJ, Chung YA, Moody G, Mitchell P, Cajulis E, Vonderfecht S, et al. Efficacy of ganitumab (AMG 479), alone and in combination with rapamycin, in Ewing’s and osteogenic sarcoma models. J Pharmacol Exp Ther. 2011;337:644–54. doi: 10.1124/jpet.110.178400. [DOI] [PubMed] [Google Scholar]
- 6.Yin M, Guan X, Liao Z, Wei Q. Insulin-like growth factor-1 receptor-targeted therapy for non-small cell lung cancer: a mini review. Am J Transl Res. 2009;1:101–14. [PMC free article] [PubMed] [Google Scholar]
- 7.Riedemann J, Macaulay VM. IGF1R signalling and its inhibition. Endocr Relat Cancer. 2006;13 (Suppl 1):S33–43. doi: 10.1677/erc.1.01280. [DOI] [PubMed] [Google Scholar]
- 8.Pollak MN, Schernhammer ES, Hankinson SE. Insulin-like growth factors and neoplasia. Nat Rev Cancer. 2004;4:505–18. doi: 10.1038/nrc1387. [DOI] [PubMed] [Google Scholar]
- 9.Woodson K, Tangrea JA, Pollak M, Copeland TD, Taylor PR, Virtamo J, et al. Serum insulin-like growth factor I: tumor marker or etiologic factor? A prospective study of prostate cancer among Finnish men. Cancer Res. 2003;63:3991–4. [PubMed] [Google Scholar]
- 10.Nickerson T, Chang F, Lorimer D, Smeekens SP, Sawyers CL, Pollak M. In vivo progression of LAPC-9 and LNCaP prostate cancer models to androgen independence is associated with increased expression of insulin-like growth factor I (IGF-I) and IGF-I receptor (IGF-IR) Cancer Res. 2001;61:6276–80. [PubMed] [Google Scholar]
- 11.Hellawell GO, Turner GD, Davies DR, Poulsom R, Brewster SF, Macaulay VM. Expression of the type 1 insulin-like growth factor receptor is up-regulated in primary prostate cancer and commonly persists in metastatic disease. Cancer Res. 2002;62:2942–50. [PubMed] [Google Scholar]
- 12.Plymate SR, Haugk K, Coleman I, Woodke L, Vessella R, Nelson P, et al. An antibody targeting the type I insulin-like growth factor receptor enhances the castration-induced response in androgen-dependent prostate cancer. Clin Cancer Res. 2007;13:6429–39. doi: 10.1158/1078-0432.CCR-07-0648. [DOI] [PubMed] [Google Scholar]
- 13.Wu JD, Haugk K, Coleman I, Woodke L, Vessella R, Nelson P, et al. Combined in vivo effect of A12, a type 1 insulin-like growth factor receptor antibody, and docetaxel against prostate cancer tumors. Clin Cancer Res. 2006;12:6153–60. doi: 10.1158/1078-0432.CCR-06-0443. [DOI] [PubMed] [Google Scholar]
- 14.Wu JD, Odman A, Higgins LM, Haugk K, Vessella R, Ludwig DL, et al. In vivo effects of the human type I insulin-like growth factor receptor antibody A12 on androgen-dependent and androgen-independent xenograft human prostate tumors. Clin Cancer Res. 2005;11:3065–74. doi: 10.1158/1078-0432.CCR-04-1586. [DOI] [PubMed] [Google Scholar]
- 15.Chi KN, Gleave ME, Fazli L, Goldenberg SL, So A, Kollmannsberger C, et al. A Phase 2 Pharmacodynamic Study of Pre-operative Figitumumab in Patients with Localized Prostate Cancer. Clin Cancer Res. 2012 doi: 10.1158/1078-0432.CCR-12-0482. [DOI] [PubMed] [Google Scholar]
- 16.Cohen BD, Baker DA, Soderstrom C, Tkalcevic G, Rossi AM, Miller PE, et al. Combination therapy enhances theinhibition of tumor growth with the fully human anti-type 1 insulin-like growth factor receptor monoclonal antibody CP-751,871. Clin Cancer Res. 2005;11:2063–73. doi: 10.1158/1078-0432.CCR-04-1070. [DOI] [PubMed] [Google Scholar]
- 17.Ward CW, Garrett TP, McKern NM, Lou M, Cosgrove LJ, Sparrow LG, et al. The three dimensional structure of the type I insulin-like growth factor receptor. Mol Pathol. 2001;54:125–32. doi: 10.1136/mp.54.3.125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Beltran PJ, Mitchell P, Chung YA, Cajulis E, Lu J, Belmontes B, et al. AMG 479, a fully human anti-insulin-like growth factor receptor type I monoclonal antibody, inhibits the growth and survival of pancreatic carcinoma cells. Mol Cancer Ther. 2009;8:1095–105. doi: 10.1158/1535-7163.MCT-08-1171. [DOI] [PubMed] [Google Scholar]
- 19.Tap WD, Demetri G, Barnette P, Desai J, Kavan P, Tozer R, et al. Phase II Study of Ganitumab, a Fully Human Anti-Type-1 Insulin-Like Growth Factor Receptor Antibody, in Patients With Metastatic Ewing Family Tumors or Desmoplastic Small Round Cell Tumors. J Clin Oncol. 2012 doi: 10.1200/JCO.2011.37.2359. [DOI] [PubMed] [Google Scholar]
- 20.Kindler HL, Richards DA, Garbo LE, Garon EB, Stephenson JJ, Jr, Rocha-Lima CM, et al. A randomized, placebo-controlledphase 2 study of ganitumab (AMG 479) or conatumumab (AMG 655) in combination with gemcitabine in patients with metastatic pancreatic cancer. Ann Oncol. 2012 doi: 10.1093/annonc/mds142. [DOI] [PubMed] [Google Scholar]
- 21.Tolcher AW, Sarantopoulos J, Patnaik A, Papadopoulos K, Lin CC, Rodon J, et al. Phase I, pharmacokinetic, and pharmacodynamic study of AMG 479, a fully human monoclonal antibody to insulin-like growth factor receptor 1. J Clin Oncol. 2009;27:5800–7. doi: 10.1200/JCO.2009.23.6745. [DOI] [PubMed] [Google Scholar]
- 22.Rosen LS, Puzanov I, Friberg G, Chan E, Hwang Y, Deng H, et al. Safety and Pharmacokinetics of Ganitumab (AMG 479) Combined With Sorafenib, Panitumumab, Erlotinib, or Gemcitabine in Patients With Advanced Solid Tumors. Clin Cancer Res. 2012 doi: 10.1158/1078-0432.CCR-11-3369. [DOI] [PubMed] [Google Scholar]
- 23.Cantley LC, Neel BG. New insights into tumor suppression: PTEN suppresses tumor formation by restraining the phosphoinositide 3-kinase/AKT pathway. Proc Natl Acad Sci U S A. 1999;96:4240–5. doi: 10.1073/pnas.96.8.4240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Jiang X, Chen S, Asara JM, Balk SP. Phosphoinositide 3-kinase pathway activation in phosphate and tensin homolog (PTEN)-deficient prostate cancer cells is independent of receptor tyrosine kinases and mediated by the p110beta and p110delta catalytic subunits. J Biol Chem. 2010;285:14980–9. doi: 10.1074/jbc.M109.085696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Korenchuk S, Lehr JE, L MC, Lee YG, Whitney S, Vessella R, et al. VCaP, a cell-based model system of human prostate cancer. In Vivo. 2001;15:163–8. [PubMed] [Google Scholar]
- 26.Loberg RD, St John LN, Day LL, Neeley CK, Pienta KJ. Development of the VCaP androgen-independent model of prostate cancer. Urol Oncol. 2006;24:161–8. doi: 10.1016/j.urolonc.2005.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Yu J, Mani RS, Cao Q, Brenner CJ, Cao X, Wang X, et al. An integrated network of androgen receptor, polycomb, and TMPRSS2-ERG gene fusions in prostate cancer progression. Cancer Cell. 2010;17:443–54. doi: 10.1016/j.ccr.2010.03.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.McMenamin ME, Soung P, Perera S, Kaplan I, Loda M, Sellers WR. Loss of PTEN expression in paraffin-embedded primary prostate cancer correlates with high Gleason score and advanced stage. Cancer Res. 1999;59:4291–6. [PubMed] [Google Scholar]
- 29.Sramkoski RM, Pretlow TG, 2nd, Giaconia JM, Pretlow TP, Schwartz S, Sy MS, et al. A new human prostate carcinoma cell line, 22Rv1. In Vitro Cell Dev Biol Anim. 1999;35:403–9. doi: 10.1007/s11626-999-0115-4. [DOI] [PubMed] [Google Scholar]
- 30.Dehm SM, Schmidt LJ, Heemers HV, Vessella RL, Tindall DJ. Splicing of a novel androgen receptor exon generates a constitutively active androgen receptor that mediates prostate cancer therapy resistance. Cancer Res. 2008;68:5469–5477. doi: 10.1158/0008-5472.CAN-08-0594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Peacock SO, Fahrenholtz CD, Burnstein KL. Vav3 Enhances Androgen Receptor Splice Variant Activity and Is Critical for Castration-Resistant Prostate Cancer Growth and Survival. Molecular Endocrinology. 2012 doi: 10.1210/me.2012-1165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Gregory CW, Johnson RT, Jr, Mohler JL, French FS, Wilson EM. Androgen receptor stabilization in recurrent prostate cancer is associated with hypersensitivity to low androgen. Cancer Res. 2001;61:2892–8. [PubMed] [Google Scholar]
- 33.Rao S, Lyons LS, Fahrenholtz CD, Wu F, Farooq A, Balkan W, et al. A novel nuclear role for the Vav3 nucleotide exchange factor in androgen receptor coactivation in prostate cancer. Oncogene. 2012;31:716–27. doi: 10.1038/onc.2011.273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Sengupta S, Blute ML, Bagniewski SM, Myers RP, Bergstralh EJ, Leibovich BC, et al. Increasing prostate specific antigen following radical prostatectomy and adjuvant hormonal therapy: doubling time predicts survival. J Urol. 2006;175:1684–90. doi: 10.1016/S0022-5347(05)00978-X. discussion 90. [DOI] [PubMed] [Google Scholar]
- 35.Cully M, You H, Levine AJ, Mak TW. Beyond PTEN mutations: the PI3K pathway as an integrator of multiple inputs during tumorigenesis. Nat Rev Cancer. 2006;6:184–92. doi: 10.1038/nrc1819. [DOI] [PubMed] [Google Scholar]
- 36.McCall P, Witton CJ, Grimsley S, Nielsen KV, Edwards J. Is PTEN loss associated with clinical outcome measures in human prostate cancer? Br J Cancer. 2008;99:1296–301. doi: 10.1038/sj.bjc.6604680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Konijeti R, Koyama S, Gray A, Barnard RJ, Said JW, Castor B, et al. Effect of a low-fat diet combined with IGF-1 receptor blockade on 22Rv1 prostate cancer xenografts. Mol Cancer Ther. 2012 doi: 10.1158/1535-7163.MCT-11-1003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Rowlands MA, Holly JM, Gunnell D, Donovan J, Lane JA, Hamdy F, et al. Circulating insulin-like growth factors and IGF-binding proteins inPSA-detected prostate cancer: the large case-control study ProtecT. Cancer Res. 2012;72:503–15. doi: 10.1158/0008-5472.CAN-11-1601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Cai C, Wang H, Xu Y, Chen S, Balk SP. Reactivation of androgen receptor-regulated TMPRSS2:ERG gene expression in castration-resistant prostate cancer. Cancer Res. 2009;69:6027–32. doi: 10.1158/0008-5472.CAN-09-0395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Mertz KD, Setlur SR, Dhanasekaran SM, Demichelis F, Perner S, Tomlins S, et al. Molecular characterization of TMPRSS2-ERG gene fusion in the NCI-H660 prostate cancer cell line: a new perspective for an old model. Neoplasia. 2007;9:200–6. doi: 10.1593/neo.07103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Colloca G. Prostate-specific antigen kinetics as a surrogate endpoint in clinical trials of metastatic castration-resistant prostate cancer: A review. Cancer Treat Rev. 2012 doi: 10.1016/j.ctrv.2012.03.008. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.