Abstract
The genus Octadecabacter is a member of the ubiquitous marine Roseobacter clade. The two described species of this genus, Octadecabacter arcticus and Octadecabacter antarcticus, are psychrophilic and display a bipolar distribution. Here we provide the manually annotated and finished genome sequences of the type strains O. arcticus 238 and O. antarcticus 307, isolated from sea ice of the Arctic and Antarctic, respectively. Both genomes exhibit a high genome plasticity caused by an unusually high density and diversity of transposable elements. This could explain the discrepancy between the low genome synteny and high 16S rRNA gene sequence similarity between both strains. Numerous characteristic features were identified in the Octadecabacter genomes, which show indications of horizontal gene transfer and may represent specific adaptations to the habitats of the strains. These include a gene cluster encoding the synthesis and degradation of cyanophycin in O. arcticus 238, which is absent in O. antarcticus 307 and unique among the Roseobacter clade. Furthermore, genes representing a new subgroup of xanthorhodopsins as an adaptation to icy environments are present in both Octadecabacter strains. This new xanthorhodopsin subgroup differs from the previously characterized xanthorhodopsins of Salinibacter ruber and Gloeobacter violaceus in phylogeny, biogeography and the potential to bind 4-keto-carotenoids. Biochemical characterization of the Octadecabacter xanthorhodopsins revealed that they function as light-driven proton pumps.
Introduction
The Roseobacter clade, a subclade of the Rhodobacteraceae belonging to the Alphaproteobacteria, is a phylogenetically coherent but physiologically and morphologically diverse group of predominantly marine bacteria [1]. The members of this clade comprise more than 38 genera and are present ubiquitously in marine habitats worldwide [2], [3]. Based on multi-locus sequence analysis (MLSA), this clade can be divided into at least 5 subclades [4], [5]. It has been suggested that horizontal gene transfer (HGT) has a large influence on the physiological heterogeneity and genomic diversity of this clade [5].
In sea ice microbial communities, Roseobacters are represented by the genus Octadecabacter. O. antarcticus constitutes up to 1% of the total bacterial community in the Southern Ocean, and O. arcticus up to 23% of the total bacterial community in Arctic sea ice [6]. These psychrophilic, heterotrophic and gas vacuole-containing bacteria were originally isolated from the lower 20 cm of annual sea ice of the Arctic and the Antarctic, respectively [7]. O. arcticus 238 and O. antarcticus 307 are the type strains of the genus Octadecabacter [8]. They are of particular interest for the comprehensive description of the Roseobacter clade, because of their bipolar distribution and unique sea ice habitat [9]. Sea ice is an extreme environment, which is characterized by strong gradients of salinity and temperature as well as low nutrient availability [10], [11]. Despite their geographical separation, both strains share >99% identity on 16S rRNA gene sequence level. However, DNA/DNA hybridization assays revealed an overall genome similarity of only 42% [8]. Since this value is well below the species threshold [12], these strains were classified as two separate species.
In a preliminary genome comparison, presence of rhodopsin genes affiliated to the xanthorhodopsin group [13], [14] was reported for the Octadecabacter strains [5]. Rhodopsins are highly diverse retinal-binding and photoactive membrane proteins [15], [16]. Bacteriorhodopsins, halorhodopsins and sensory rhodopsins function as light-driven proton pumps, chloride pumps, and signal transducers, respectively. They are predominantly of archaeal origin, but closely related to fungal rhodopsins [17]. Proteorhodopsins, however, are predominantly of bacterial origin and assumed to function as proton pumps [18]. Proton-pumping xanthorhodopsins were first described in Salinibacter ruber [13] and Gloeobacter violaceus [14]. The unique feature of the xanthorhodopsins of these organisms is binding of 4-keto-carotenoids as antenna pigments.
The aim of this study was a comprehensive genome analysis of the Octadecabacter type strains to elucidate relationships between these geographically separated type strains and identify genomic features linked to sea ice habitats. Ice-associated features include the xanthorhodopsin gene products of the Octadecabacter strains. These products represent a new subgroup of xanthorhodopsins that is functionally and phylogenetically distinct from previously described xanthorhodopsins.
Materials and Methods
Cultivation
Octadecabacter and Gloeobacter strains were obtained from the Centre de Ressources Biologiques de l'Institut Pasteur (CRBIP, Paris, France). A recombinant Escherichia coli clone harboring the subcloned proteorhodopsin gene of the environmental clone EBAC31A08 [19] was kindly provided by Edward F. DeLong (MIT, Cambridge, MA, USA). All strains used in this study are listed in Table 1.
Table 1. List of strains used in this study.
| Strain | Description | Source |
| Octadecabacter arcticus 238 | Type strain of O. arcticus | CRBIP, Paris, France |
| Octadecabacter antarcticus 307 | Type strain of O. antarcticus | CRBIP, Paris, France |
| Gloeobacter violaceus PCC 7421 | Type strain of G. violaceus | CRBIP, Paris, France |
| pBAD_EBAC31A08 in E. coli UT5600 | Opsin gene of EBAC31A08 subcloned into expression vector pBAD in host E. coli UT5600 | DeLong, E.F., MIT, Cambridge, MA, USA |
| pET24D_protRho2 in E. coli C43 | Opsin insert of pBAD_EBAC31A08 subcloned into expression vector pET24D in host E. coli C43 | This study |
| pET24D_oarRho2 in E. coli C43 | Opsin gene of O. arcticus 238 cloned in expression vector pET24D in host E. coli C43 | This study |
| pET24D_oanRho2 in E. coli C43 | Opsin gene of O. antarcticus cloned in expression vector pET24D in host E. coli C43 | This study |
| pET24D_gviolRho2 in E. coli C43 | Opsin gene of G. violaceus PCC 7421 in expression vector pET24D in host E. coli C43 | This study |
| pET24D in E. coli C43 | Expression vector pET24D in host E. coli C43 | This study |
Octadecabacter cells were grown in marine broth medium MB2216 [20] at 8°C. Gloeobacter violaceus cells were grown in BG-11 medium at 22°C. Recombinant E. coli cells were grown in LB medium supplemented with kanamycin (30 µg/mL) or ampicillin (100 µg/mL) at 37°C. All cultures were incubated under constant shaking.
Sequencing and Annotation
The genomes of O. arcticus 238 and O. antarcticus 307 were sequenced using the Sanger approach (https://moore.jcvi.org/moore/). Gap closure and polishing were done using the Staden software package [21] and PCR-based techniques. Open reading frames (ORFs) were identified using YACOP [22] and GLIMMER [23], and manually corrected. Functional annotation was initially performed with the ERGO software tool [24] and manually corrected by comparison to the Swissprot, TrEMBL (http://kr.expasy.org/), and Interpro databases [25]. Genes associated with transposable elements (TEs), were classified via BLAST comparisons with the ISFinder database (http://www-is.biotoul.fr) [26].
The complete sequences of O. arcticus 238 and O. antarcticus 307 chromosomes and plasmids have been deposited in GenBank under accession numbers CP003742 (O. arcticus chromosome), CP003743 (pOAR118), CP003744 (pOAR160), CP003740 (O. antarcticus chromosome), and CP003741 (pOAN63).
Determination of Orthologs
Orthologous protein sequences were identified by bidirectional best-hit analyses (BBH; often also referred to as reciprocal best-hit method, RBH) [27], [28] using BLAST (http://www.ncbi.nlm.nih.gov/). Only bidirectional best-hits with e-values lower than 1e-10 were considered. In order to filter and remove false hits based on short local alignments of conserved protein domains, sequence identities were determined by performing global alignments for each bidirectional best-hit using the Needleman-Wunsch algorithm [29]. As in previous comparative studies [5], [30], [31], a cutoff value of 30% sequence identity was chosen to identify orthologs. An additional cutoff value of 60% was used to determine the number of orthologs with highly conserved sequences, as the probability for equivalent functions is considerably higher at sequence identities >50% [32]. The Octadecabacter pan-genome was determined as the sum of all genes in both Octadecabacter strains.
Genome Sequence Comparisons
Genome alignment-based synteny plots were done using the NUCmer tool of the MUMmer suite [33] implemented in the Integrated Microbial Genomes (IMG) system (http://img.jgi.doe.gov) [34]. Only genome sequences consisting of ten or less scaffolds were used to compare genome synteny. BLAST-based average nucleotide identities (ANIb) were determined using the JSpecies software (www.imedea.uib.es/jspecies/) [35].
Phylogenetic Analyses
Genome sequences were obtained from the NCBI GenBank sequence database (www.ncbi.nlm.nih.gov) or from the J. Craig Venter Institute (http://www.jcvi.org). Reference protein and nucleotide sequences were obtained from the NCBI non-redundant (nr) database. For Multilocus Sequence Analysis (MLSA), protein-sequences of genes for which one ortholog but no paralog was found in every comparison strain were concatenated. Sequences were aligned using clustalW [36]. Neighbor-joining and maximum-likelihood trees were constructed using ARB v5.1 [37]. To calculate tree backbones only 16S sequences >1200 bp and complete rhodopsin protein sequences were used. Short partial sequences were added to the tree backbones using Parsimony. For phylogenetic analysis of 16S rRNA gene sequences a dataset was assembled, which contains type strains representing the Roseobacter clade. Methylococcus capsulatus ACM1292 and Thiotrix nivea JP2 were employed as outgroups. For the analyses of microbial rhodopsins and cyanophycin ligases, datasets containing representatives of all described subgroups were assembled (Supplementary Table S1 and Supplementary Table S2). For MLSA, a filter was employed to remove gapped positions prior to tree calculation.
Screening of Metagenomic Databases
Several metagenomic datasets derived from freshwater, marine, hypersaline, thermophilic, and ice-associated habitats [38]–[49] (Table S3) available at the CAMERA (http://camera.calit2.net/) and MG-RAST (http://metagenomics.anl.gov/) databases were screened for rhodopsins via batched BLAST comparisons. Only samples from the surface region to 30 m depth were analyzed. To prevent bias caused by small survey sizes only metagenomes with more than 100 000 reads were evaluated. Query sequences were selected from a phylogenetic dataset based on rhodopsin sequences available at NCBI. To ensure sensitivity at least two representatives of each main group (proteorhodopsins, xanthorhodopsins, fungal rhodopsins, bacteriorhodopsins, halorhodopsins and sensory rhodopsins, Supplementary Table S1) were used as query sequences in primary tBLASTn analyses of the metagenome datasets using a non-stringent e-value cutoff of 1.
The resulting hits were verified and classified by secondary BLASTx comparisons using stringent e-value and alignment cutoffs. As short reads generally yield lower e-values than long reads, different e-value cutoffs depending on the average read length of the respective dataset were chosen. A cutoff of 1e-20 was used at average read lengths >150 bp and a cutoff of 1e-10 at average read length <150 bp. The resulting alignments had to cover at least 60% of the reference sequence or 80% of the query sequence. The NCBI RefSeq and the self-produced phylogenetic dataset were used as reference databases for verification and classification of the rhodopsin sequences. Total rhodopsin abundances were normalized against the total number of reads of the respective metagenome.
Heterologous Expression of Rhodopsins
Rhodopsin gene sequences were amplified via PCR using rhodopsin-specific primers and the PCR extender system (5 PRIME Inc., Gaithersburg, USA) at annealing temperatures of 60°C according to the recommendations of the manufacturer. A poly-guanosine tail and NcoI and XhoI restriction sites were added to the 5′ ends of the primers to allow directed ligation into the expression vector. The resulting primer sequences were as follows (poly-guanosine tail and restriction sites are underlined): oanrho2f, (5′-GGGGGCCATGGAAACTTTATCACTGGTCAG-3′); oanrho2r, (5′-GGGGGCTCGAGTTACTCGGCGGGGACCGTCTTGGTGTTTTTGTCC-3′); oarrho2f, (5′-GGGGGCCATGGAAACATTATCATTGGGTCAATATG-3′); oarrho2r, (5′-GGGGGCTCGAGTTATTCAGCAGGGACTGCTGTCTTTATGGAATCGTTG-3′); gviolrho2f, (5′-GGGGGCCATGGGGATGTTGATGACCGTATTTTCTTCTGC-3′); gviolrho2r, (5′-GGGGCTCGAGCTAGGAGATAAGACTGCCTC CCGATTTATTTGC-3′); protrho2f, (5′-GGGGGGCCATGGATGAAATTATTACTGATATTAGGTAGTG TTATTGCACTTCCTACATTTGC-3′); and protrho2r, (5′-GGGGGCTCGAGTTAAGCATT AGAAGATTCTTTAACAGCAACATTCCA-3′). The resulting PCR products were cloned into the expression vector pET24D and subsequently used to transform E. coli Top10 cells as recommended by the manufacturer (Invitrogen, Carlsbad CA, USA). Fidelity of PCR products and constructs was verified by sequencing. For expression of the rhodopsin genes, E. coli C43 [DE3] (Lucigen Cooperation, Middleton WI, USA) was used as host. The resulting recombinant E. coli strains are listed in Table 1.
Reconstitution of Rhodopsins with Chromophores
All-trans retinal was obtained from Sigma-Aldrich (Steinheim, Germany). Salinixanthin was extracted from S. ruber cultures according to Imasheva et al. [14] and Lutnaes et al. [50].
To isolate membrane fragments carrying heterologously produced rhodopsins, cells were lysed by sonication. Subsequently, cell debris was precipitated by centrifugation (3000 g, 8°C, 10 min) followed by precipitation of membrane fragments via ultracentrifugation of the supernatant (40,000 g, 8°C, 2 h). The membrane fragment-containing pellet was then suspended in 50 mM Tris buffer (pH 8) containing 5 mM MgCl2 [19]. The chromophores retinal and salinixanthin were stored as stock solutions in ethanol (10 mM and 1 mM, respectively) and were added to final concentrations of 10 µM. Absorbance spectra were recorded before and after addition of chromophores using a Lambda25 UV/Vis Spectrometer (PerkinElmer, Rodgau, Germany).
Light-induced Proton Translocation
Proton-pumping function of heterologously expressed rhodopsins was detected directly in suspensions of host cells according to Beja et al. [19]. The cells were washed twice by centrifugation (5,000 g, 8°C, 10 min) with a non-buffered nutrient-free salt solution (10 mM NaCl, 10 mM MgSO4, 100 µM CaCl2). The nutrient-free cell suspensions were kept at room temperature for at least 15 minutes before measurement. Light-dependent proton translocation was detected via temporary fluxes in the acidification rate of the cell suspensions, using a WTW pH330i pH-meter and a Sentix81 pH electrode (WTW, Weilheim, Germany). A 500 W tungsten Halogen lamp was used as light source. The light was filtered through 15 cm of ice-cold water in order to block heat emitted from the light source. Additionally, the suspensions were kept at room temperature by partial submersion of the culture bottles in water. The temperature of the suspensions was monitored throughout the experiment.
Results and Discussion
General Genome Comparisons
The genome of O. antarcticus 307 consists of a 4.8 Mb chromosome and a 63 kb plasmid (pOAN63) whereas O. arcticus 238 harbors a 5.2 Mb chromosome and two plasmids of 118 kb and 160 kb (pOAR118 and pOAR160, respectively). The GC-content of both genomes is 55%, which is at the lower end of the typical GC-content of Roseobacter genomes (Table 2). The number of predicted protein-encoding genes is 4,683 in O. arcticus and 4,492 in O. antarcticus.
Table 2. General genome comparisons of Roseobacter clade members.
| Number of genes | |||||||
| Organism | GC- content | Genome Size [Mb] | Protein- coding | Pseudo- genes | RNA genesa | TEs/Mb | Sub- groupc |
| Phaeobacter gallaeciensis 2.10 | 60% | 4.16 | 3875 | 16 | 69 | 4 | 1 |
| Phaeobacter gallaeciensis DSM17395 | 60% | 4.23 | 3875 | 16 | 69 | 10 | 1 |
| Phaeobacter arcticus DSM 23566 | 59% | 5.05 | 4726 | 102 | 81 | 10 | 1 |
| Phaeobacter caeruleus 13 | 63% | 5.35 | 5146 | 81 | 108 | 13 | 1 |
| Phaeobacter daeponensis DSM 23529 | 64% | 4.64 | 4284 | 69 | 78 | 9 | 1 |
| Phaeobacter inhibensT5 | 60% | 4.13 | 3884 | 39 | 63 | 7 | 1 |
| Phaeobacter sp. Y4I | 64% | 4.34 | 4132 | 1 | 69 | 13 | 1 |
| Nautella italica R11 | 60% | 3.82 | 3655 | 1 | 69 | 3 | 1 |
| Rhodobacterales sp. MED193 | 57% | 4.65 | 4535 | 0 | 70 | 15 | 1 |
| Rhodobacterales sp. SK209-2-6 | 57% | 4.56 | 4537 | 0 | 73 | 20 | 1 |
| Ruegeria sp. TrichCH4B | 59% | 4.69 | 4734 | 1 | 79 | 20 | 1 |
| Ruegeria sp. TM1040 | 60% | 4.15 | 3864 | 6 | 94 | 7 | 1 |
| Ruegeria lacuscaerulensis ITI-1157 | 63% | 3.52 | 3608 | 3 | 63 | 14 | 1 |
| Ruegeria sp. KLH11 | 58% | 4.49 | 4269 | 5 | 64 | 24 | 1 |
| Ruegeria sp. TW15 | 56% | 4.49 | 4380 | 0 | 45 | 8 | 1 |
| Ruegeria pomeroyi DSS-3 | 64% | 4.60 | 4252 | 31 | 72 | 5 | 1 |
| Rhodobacterales sp. R2A57 | 51% | 4.14 | 4386 | 0 | 43 | 16 | 1 |
| Sulfitobacter sp. NAS-14.1 | 60% | 4.00 | 3962 | 0 | 64 | 23 | 2 |
| Sulfitobacter sp. EE-36 | 60% | 3.55 | 3474 | 0 | 68 | 11 | 2 |
| Sulfitobacter sp. GAI101 | 59% | 4.53 | 4202 | 1 | 55 | 15 | 2 |
| Oceanibulbus indolifex HEL-45 | 60% | 4.11 | 4153 | 0 | 55 | 16 | 2 |
| Roseobacter denitrificans OCh 114 | 59% | 4.33 | 4129 | 17 | 55 | 8 | 2 |
| Roseobacter litoralis Och 149 | 57% | 4.75 | 4537 | 0 | 40 | 14 | 2 |
| Rhodobacterales sp. HTCC2083 | 53% | 4.02 | 4177 | 2 | 47 | 32 | 2 |
| Citreicella sp. SE45 | 67% | 5.52 | 5425 | 2 | 72 | 18 | 3 |
| Citreicella sp. 357 | 64% | 4.60 | 4528 | 0 | 45 | 31 | 3 |
| Pelagibaca bermudensis HTCC2601 | 66% | 5.43 | 5452 | 0 | 62 | 23 | 3 |
| Sagittula stellata E-37 | 65% | 5.26 | 5067 | 0 | 54 | 17 | 3 |
| Oceanicola batsensis HTCC2597 | 66% | 4.44 | 4212 | 0 | 49 | 12 | 3 |
| Roseovarius sp. 217 | 61% | 4.76 | 4772 | 0 | 51 | 23 | 3 |
| Roseovarius sp. TM1035 | 61% | 4.21 | 4102 | 0 | 56 | 9 | 3 |
| Roseobacter sp. AzwK-3b | 62% | 4.18 | 4145 | 0 | 52 | 37 | 3 |
| Roseovarius nubinhibens ISM | 64% | 3.67 | 3547 | 0 | 58 | 2 | 3 |
| Loktanella sp. CCS2 | 55% | 3.50 | 3660 | 0 | 43 | 3 | 4 |
| Loktanella vestfoldensis SKA53 | 60% | 3.06 | 3068 | 0 | 49 | 9 | 4 |
| Loktanella sp. SE62 | 62% | 4.58 | 4596 | 0 | 43 | 8 | 4 |
| Octadecabacter antarcticus 307 | 55% | 4.88 | 4492 | 361 | 48 | 74 | 4 |
| Octadecabacter arcticus 238 | 55% | 5.20 | 4683 | 411 | 49 | 175 | 4 |
| Thalassiobium sp. R2A62 | 55% | 3.49 | 3696 | 0 | 48 | 34 | 4 |
| Oceanicola granulosus HTCC2516 | 70% | 4.04 | 3792 | 0 | 63 | 6 | 4 |
| Wenxinia marina DSM 24838 | 71% | 4.18 | 4045 | 0 | 59 | 10 | 4 |
| Ketogulonicigenium vulgare Y25 | 62% | 3.29 | 3213 | 0 | 74 | 5 | 4 |
| Ketogulonigenium vulgarum WSH-001 | 62% | 3.28 | 3054 | 0 | 71 | 6 | 4 |
| Dinoroseobacter shibae DFL-12 | 66% | 4.42 | 4186 | 33 | 52 | 22 | 5 |
| Jannaschiasp. CCS1 | 62% | 4.40 | 4283 | 0 | 56 | 6 | 5 |
| Maritimibacter alkaliphilus HTCC2654 | 64% | 4.53 | 4712 | 0 | 48 | 9 | - |
| Rhodobacterales sp. HTCC2150 | 49% | 3.58 | 3667 | 0 | 46 | 20 | - |
| Rhodobacterales sp. HTCC2255b | 39%b | 4.81b | 4507b | 0 | 86b | 4b | - |
Bidirectional BLAST analyses showed that 76% of the genes in the Octadecabacter pan-genome have orthologs in at least one other Roseobacter clade member. These genes represent part of the shared Roseobacter pan-genome (core and flexible genome without singletons). The predicted gene products of most of these orthologs (85%) exhibit >60% sequence identity, indicating identical or equivalent function. The remaining orthologs may have adapted to new functions in the respective strains through mutation and genetic drift. Although the overall shared pan-genome comprises most of the Octadecabacter genes, only 32–50% of the Octadecabacter pan-genome is shared by individual Roseobacter strains. This reflects the overall diversity of the Roseobacter clade.
The two analyzed Octadecabacter strains shared approximately 2% of the genome content exclusively with each other. This indicates that on genomic level the genus Octadecabacter is less defined by unique features, but more by unique composition of shared features from the Roseobacter pan-genome. This module-like assembly of a Roseobacter genome is in accordance with previous observations. Characteristic phenotypical and selected genomic features occur in a “patchy” distribution along distantly related Roseobacter phylogenetic groups [2].
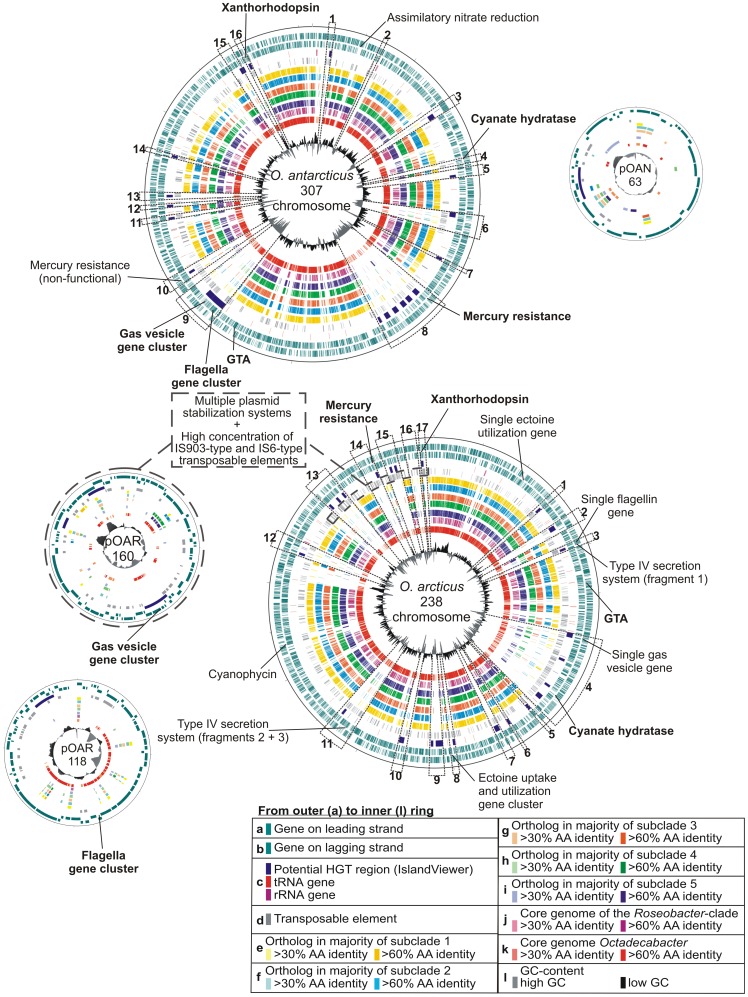
The unique genes of each strain comprise 20% of the genome in O. antarcticus and 23% in O. arcticus, indicating a high potential for individual adaptations. The majority of the unique and rare genes found in the Octadecabacter genomes are located in distinct regions of the chromosomes, thereby forming potential genomic islands (Figure 1) of which several were also indicated by IslandViewer [51] predictions. Regions containing multiple indicators for HGT such as IslandViewer predictions, divergent GC-content and low numbers of orthologs in closely related reference genomes were defined as “regions of enhanced genome plasticity” (RGP, Figure 1). These regions represent recombinatorial hot spots and were numbered Oar-RGP 1–17 on the chromosome of O. arcticus and Oan-RGP 1–16 on the chromosome of O. antarcticus. Plasmids were entirely defined as RGPs. Many of the below-described characteristic gene clusters are located in these regions (a general overview of RGP regions is provided in Supplementary Table S4).
Figure 1. Circular representations of the Octadecabacter genomes.
Regions of enhanced genome plasticity (RGP) are marked by dotted lines and numbered on the chromosomes of both strains. The location of protein-encoding genes, IslandViewer [49] predictions of potential genomic islands, rRNA genes, transposable elements (TEs) and orthologs to genes in strains of the different Roseobacter subclades as well as variations in GC-content are displayed. The comparison strains were grouped in subclades according to Newton et al. [5]. Several selected features indicative for horizontal gene transfer or intra-genomic recombination are labelled. Features that are present in both strains are marked in bold.
Phylogeny and Biogeography of the Genus Octadecabacter
Despite their close relationship on 16S rRNA gene sequence level, the genomes of O. arcticus 238 and O. antarcticus 307 exhibit significant differences in organization and content. This has been indicated by previously reported low DNA/DNA-hybridization values [8] and relatively high phylogenetic distances on MLSA level [5] (Supplementary Figure S1). The low resolution of 16S rRNA gene-based phylogeny with respect to closely related species has often been reported [52], [53]. Nevertheless, this approach has been proven to present reliable phylogenetic backbones, which are comparable to MLSA-based approaches [54], [55]. Thus, despite the differences of both Octadecabacter strains on genomic level, the 16S rRNA gene sequences indicate that Arctic and Antarctic strains are phylogenetically closely linked. Moreover, all Octadecabacter 16S rRNA gene sequences obtained from northern and southern polar habitats form a single distinct phylogenetic cluster (Figure 2). Only three sequences from non-polar habitats fall into this cluster, which originate from uncultivated organisms obtained from low temperature habitats: one from deep sea sediments (AB094833) and two from ciliates sampled in the Atlantic Ocean in winter (FN999980 + FN999956). The remaining non-polar sequences form separate clusters, indicating that psychrophilic strains of the opposite polar regions are more related to each other than to mesophilic strains from warmer regions between the two poles (Figure 2). Therefore, a direct link seems to exist between Arctic and Antarctic Octadecabacter populations.
Figure 2. Phylogeny of Octadecabacter strains based on 16S rRNA gene sequences.
Depicted is a subtree of a neighbor-joining tree of the complete Roseobacter clade, based on 16S rRNA gene sequences available at NCBI. The respective NCBI accession numbers are given in parentheses behind the individual clone or isolate designations. Sequences derived from clones are marked by an asterisk (*). Sequences derived from the type strain of a species are marked with a “T”. Neighbor-joining bootstrap values above 50% are given without parentheses at the respective nodes. For nodes that could be reproduced with maximum-likelihood calculation, the bootstrap values above 50% of the maximum-likelihood tree are given in parentheses. The 16S rRNA gene sequences of the type strains of Methylococcus capsulatus and Thiothrix nivea (NCBI-Accession-numbers AJ563935 and L40993, respectively) were used as outgroup (not shown).
Genome Plasticity and Genetic Variability
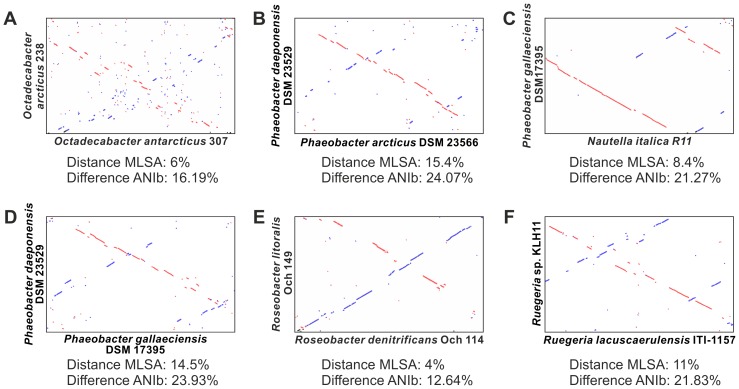
Whole-genome alignments of O. arcticus and O. antarcticus reveal only short continuous regions of sequence homology with a high frequency of inversions and strongly divergent organization along the genomes (Figure 3). This high divergence is in accordance with the low DNA/DNA-hybridization values of the Octadecabacter strains, which originally led to their classification as separate species [8]. However, it is not consistent with the genetic distances between these two organisms derived from MLSA-phylogeny or BLAST-based average nucleotide identity (ANIb). Other Roseobacter species with similar or higher genetic distances, including isolates from globally opposite locations [31], [56]–[58], exhibit much longer continuous stretches of sequence homology and less inversions in pairwise genome alignments (Figure 3). This implies that rearrangements occur more frequently in the genomes of the Octadecabacter strains than in other Roseobacters. Indications for intra-genomic rearrangements were mainly found in the genome of O. arcticus 238, which contained the most transposable elements of both Octadecabacter strains. Fragments of several gene clusters are scattered across multiple RGPs and even multiple replicons in this genome. Examples include the fragmentation of the type IV secretion system into three partial clusters found in Oar-RGP 3 and 11, and the presence of single flagella and gas vesicle genes in Oar-RGP 3 and 4 in addition to the complete gene clusters on plasmids pOAR118 and pOAR160 (Figure 1). Furthermore, an exchange of genetic material between the plasmid pOAR160 and the chromosomal area between Oar-RGP 13 and 17 is indicated by several shared features such as related plasmid stabilization systems and high concentrations of certain IS elements (Figure 1). The high frequency of recombination events is probably caused by the unusually large number and high diversity of transposable elements (TEs) such as IS elements or transposons in both Octadecabacter genomes (Tables 2 and 3). Multiple copies of TEs in a genome are known to indirectly facilitate inversion, deletion and translocation of large genomic areas via homologous recombination events [59], [60]. Accordingly, a higher number of TEs should result in a higher probability of recombination events. Based on the number of TE-associated mobility genes per megabase, the average density of these genes in all sequenced Roseobacter genomes is approximately 21 per megabase, whereas it is 4–8 times higher in the genomes of both Octadecabacter strains (Table 2). The TE-associated genes of O. arcticus and O. antarcticus fall into at least 21 and 16 families of IS elements, respectively (Table 3). The families IS3 and IS5 can be further divided into distinct subgroups [61], of which several are represented in the Octadecabacter genomes. This high diversity of IS elements in the Octadecabacter genomes is indicative for multiple independent HGT events from different sources. By acting as recombinational anchors for illegitimate recombination events [62], [63], TEs can also enhance the probability of HGT [64]. An enhanced potential for incorporation of new genetic material would explain the large number and size of RGPs in the Octadecabacter genomes. This might be of relevance in sea ice microbial communities, as Collins and Demming have described sea ice as rich in extracellular DNA and as a potential hot spot for HGT [65], [66]. Type IV secretion systems are known to be involved in transfer and uptake of DNA [67], but in O. arcticus the corresponding gene cluster is fragmented due to intra-genomic recombination (Figure 1 and Supplementary Figure S2A). New genomic features can be efficiently spread throughout Octadecabacter populations via gene transfer agents (GTAs). GTAs are phage-like particles that package and transfer random fragments of the host genome. GTAs are conserved in most members of the Roseobacter clade, including both Octadecabacter strains [5]. The function of GTAs as a mechanism for HGT between closely related Roseobacter strains has been demonstrated for Silicibacter pomeroyi DSS-3 [68].
Figure 3. Synteny plots of the genomes of the Octadecabacter strains and other selected Roseobacter clade members.
Synteny plots based on pairwise genome-alignments using MUMmer [33]. Linear regions indicate coherent regions of sequence homology. Homolog regions with identical orientation are displayed in red, inversions in blue. For reference the distance values obtained via multi locus sequence analysis (MLSA, Supplementary Figure S1) and the BLAST-based average nucleotide identities (ANIb) calculated using the JSpecies software [35] are given for each genome pair. Despite their close relationship on MLSA level and ANIb level, the Octadecabacter strains show a low genome synteny and many inversions (A). Other strains show much higher syntenies, despite similar oder more distant relationships on MLSA level and ANIb level (B–F). This includes strains isolated from globally opposite locations (C, D).
Table 3. Families of transposable elements in the Octadecabacter strains.
| O. arcticus 238 | O. antarcticus 307 | |||||
| Family | Genesa | Pseudo-genes | Totalb | Genesa | Pseudo-genes | Totalb |
| IS3c | 177 | 30 | 207 | 137 | 49 | 186 |
| IS4 | 0 | 0 | 0 | 1 | 1 | 2 |
| IS5d | 44 | 19 | 63 | 34 | 8 | 42 |
| IS6 | 25 | 4 | 29 | 12 | 3 | 15 |
| IS10 | 7 | 8 | 15 | 0 | 0 | 0 |
| IS21 | 11 | 0 | 11 | 13 | 4 | 17 |
| IS30 | 45 | 17 | 62 | 8 | 0 | 8 |
| IS66 | 66 | 15 | 81 | 2 | 1 | 3 |
| IS91 | 22 | 5 | 27 | 12 | 0 | 12 |
| IS110 | 17 | 4 | 21 | 33 | 39 | 72 |
| IS200/IS605 | 38 | 2 | 40 | 14 | 0 | 14 |
| IS204/IS1001/IS1096/IS1165 | 0 | 2 | 2 | 0 | 0 | 0 |
| IS256 | 72 | 21 | 93 | 9 | 3 | 12 |
| IS481 | 33 | 3 | 36 | 1 | 16 | 17 |
| IS630 | 73 | 10 | 83 | 2 | 3 | 5 |
| IS116/IS110/IS902 | 1 | 0 | 1 | 0 | 0 | 0 |
| IS1182 | 0 | 0 | 0 | 1 | 14 | 15 |
| IS1380 | 1 | 0 | 1 | 2 | 0 | 2 |
| IS1595 | 33 | 1 | 34 | 20 | 3 | 23 |
| ISAs1 | 28 | 2 | 30 | 0 | 0 | 0 |
| ISL3 | 29 | 10 | 39 | 0 | 0 | 0 |
| P4-integrase | 1 | 0 | 1 | 0 | 0 | 0 |
| ISNCY | 1 | 2 | 3 | 0 | 0 | 0 |
| unclassified | 188 | 108 | 296 | 60 | 121 | 181 |
| Total TEs | 912 | 263 | 1175 | 361 | 265 | 626 |
High genome plasticity mediated by TEs could explain the divergence in genome content and genome organization of both Octadecabacter strains. The strong discrepancy between phylogenetic distances obtained by 16S rRNA gene-based (Figure 2) and MLSA-based approaches (Supplementary Figure S1) may result from an enhanced mutagenic activity. The 16S rRNA gene sequence exhibits a significantly lower rate of evolutionary substitution than protein-encoding genes [52], [69]. Consequently, evolutionary changes manifest earlier and more significantly in protein-encoding genes. In the Octadecabacter genomes, a high mutagenicity is indicated by an unusually large number of pseudogenes (411 in O. arcticus and 361 in O. antarcticus; Table 2). Most of these pseudogenes are TE-associated or carry an insertion of a TE (339 in O. arcticus and 282 in O. antarcticus). However, approximately 20% of the pseudogenes in both strains are not directly linked to TEs and comprise approximately 1.5% of the total genes in each Octadecabacter strain. This degree is above average for members of the Roseobacter clade (Table 2). Thus, polar Octadecabacter populations seem to be subjected to mutagenic influences other than TEs.
Characteristic Features of the Octadecabacter Strains
The Octadecabacter strains are characterized by several features that are rare or even unique among the Roseobacter clade. Many of these show indications of HGT, such as location in a region of enhanced genome plasticity (RGP), flanking transposases and sequence similarities to organisms of different taxa. The Arctic and the Antarctic strain are distinguished by the presence of a gene cluster encoding synthesis and degradation of cyanophycin, which is only present in O. arcticus (Supplementary Figure S2B). Cyanophycin is a non-ribosomally synthesized branched polypeptide [70], which functions as a nitrogen storage compound in diverse bacteria [71], [72]. Ten phylogenetically distinct groups of cyanophycin ligases (Groups I–X) were established by Füser and Steinbüchel [71] based on protein sequences that were publicly available in 2007. Due to the increase of publicly available protein sequences at least 9 additional groups can be distinguished today (Groups XI–XIX, Supplementary Figure S3), but the groups previously described by Füser and Steinbüchel remain valid. The cyanophycin ligase of O. arcticus is affiliated with group IV [71], which consists of 31 mostly gammaproteobacterial sequences (Supplementary Figure S3). The Octadecabacter ligase is most closely related to the cyanophycin ligase of Colwellia psychrerythraea 34H, which is also an Arctic sea ice bacterium [73], [74]. Diverse cyanophycin ligases, including members of group VI, were found in Antarctic marine metagenomes. Therefore the absence of this trait in O. antarcticus cannot be directly attributed to differences in northern and southern polar habitats. Although this feature is not located within a RGP region in O. arcticus, its relationship to mostly gammaproteobacterial cyanophycin ligases indicates an origin via HGT. Instead of a cyanophycin pathway, O. antarcticus possesses genes for assimilatory nitrate and nitrite reduction pathways (Supplementary Figure S2C), which are present in 20 other Roseobacter clade members but not in O. arcticus. Like the cyanophycin genes in O. arcticus, these genes are also not located within an RGP. However, in O. antarcticus the corresponding gene cluster is closely flanked by TEs. The distribution of this feature among Roseobacter clade members is not linked with phylogeny [5], indicating that this feature may be frequently transferred via HGT. Another feature involved in nitrate metabolism are genes encoding cyanate hydratases. Cyanate is a toxic substance, which can accumulate in organisms as a byproduct of metabolic pathways such as the urea cycle [75], [76]. Cyanate hydratases allow the detoxification of cyanate as well as its utilization as a nitrogen source [77]. Corresponding gene clusters can be found in both Octadecabacter chromosomes (Oar-RGP 4 and Oan-RGP 4, Supplementary Figure S4A) but are absent in most other members of the Roseobacter clade.
Mercury resistance gene clusters [78] are also present in both Octadecabacter chromosomes (Supplementary Figure S4B). O. arcticus has one copy of this gene cluster in region Oar-RGP 14 whereas O. antarcticus possesses two copies, one without mutations in Oan-RGP 8 and one with a frameshift mutation in merT in Oan-RGP 10. Bidirectional BLAST analyses revealed similar gene clusters only in 10 of the 46 Roseobacter clade reference organisms.
A gene cluster encoding the formation of gas vesicles, a main characteristic of polar Octadecabacter strains, is located in Oan-RGP 9 of the O. antarcticus chromosome and the O. arcticus plasmid pOAR160 (Supplementary Figure S4C). Homologs of all 8 genes (gvpAJM, gvpLF, gvpG, gvpO and gvpK) that are essential for gas vesicle formation in Halobacterium salinarum PHH1 [79] were found. In addition, the Octadecabacter gene cluster contains genes encoding the chaperone GvpN and a conserved protein of unknown function. Heterotrophic bacteria containing gas vesicles are not common in marine habitats, except for polar sea ice microbial communities [7] in which they may function as dispersal mechanisms [9]. Gas vesicle genes can also be found in two Loktanella strains. L. sp. CCS2 harbors a gvpK gene but no complete gene cluster, whereas L. sp. SE-62 contains several gas vesicle genes in a coherent gene cluster. BLAST comparisons showed that these genes show low similarities to the Octadecabacter gas vesicle genes and are closer related to a gene cluster of Rhodobacter capsulatus SB1003. Furthermore, the corresponding gene clusters are located close to a photoactive yellow protein in L. sp. SE-62 as well as in R. capsulatus SB1003 [80]. This indicates that gas vesicles of the Loktanella strains are not directly related to those of the Octadecabacter strains but may share the same phylogenetic history as the gas vesicles of R. capsulatus.
The genomes of both Octadecabacter strains contain three large gene clusters encoding flagella synthesis (Supplementary Figure S4D), which show only low similarities to the majority of Roseobacter flagella gene clusters. The flagella gene clusters are organized almost identically in both Octadecabacter strains, although they are located in different replicons. In O. arcticus they are located on plasmid pOAR118 whereas in O. antarcticus they are present in Oan-RGP 9 of the chromosome. The Octadecabacter strains have been described as non-motile [8] and showed no swimming or swarming under various conditions (data not shown). Thus, the function of the flagella genes remains to be elucidated. Similar flagella gene clusters were only found in L. vestfoldensis SKA53, Rhodobacterales bacterium HTCC2083, and Roseovarius sp. TM1035.
A gene cluster encoding an ectoine uptake and utilization pathway is present in region Oar-RGP 8 of O. arcticus (Supplementary Figure S2D). Ectoine is a compatible solute involved in osmoprotection [81], and might be of importance for survival in the hypersaline brine channels of sea ice. The ectoine gene cluster is flanked by IS3 family TEs and consists of the ABC-transporter genes ehuABCD and the ectoine utilization genes eutABCDE. A paralog of eutD also flanked by IS3 family elements can be found near Oar-RGP 1 (Figure 1), indicating a TE mediated intra-genomic recombination event.
A Novel Xanthorhodopsin Subgroup
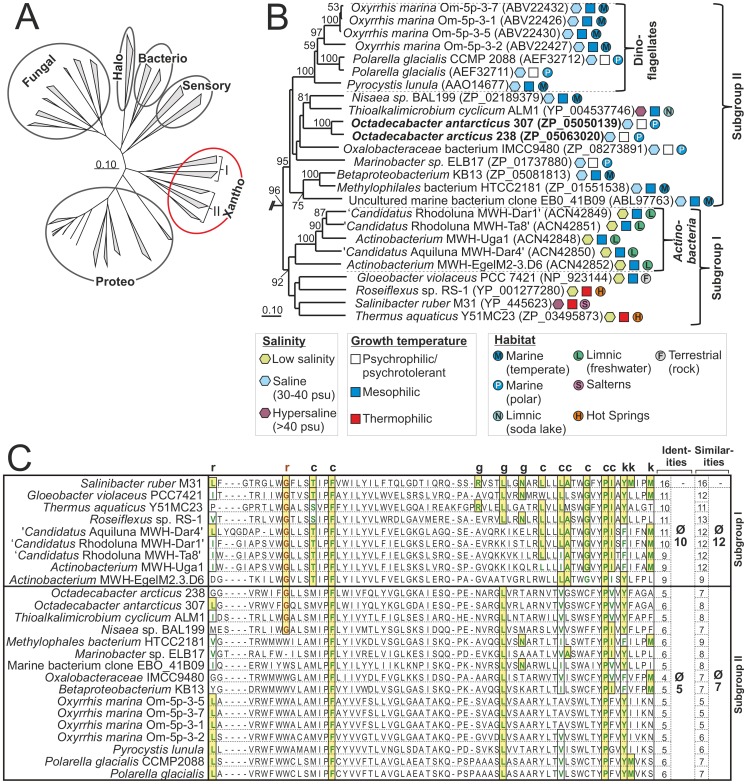
Gene clusters encoding a rhodopsin affiliated to the xanthorhodopsin group [14] were identified on the chromosomes of O. arcticus and O. antarcticus [5]. Several rhodopsin sequences available at the NCBI database are affiliated to this phylogenetically distinct and coherent group (Figure 4A). The Rhodobacteraceae strain HTCC2255 also possesses a rhodopsin gene cluster [5], however this cluster is affiliated to the proteorhodopsin group and not directly related to the Octadecabacter xanthorhodopsins. Based on sequence similarities this group can be divided in two main subgroups, here designated subgroup I and subgroup II (Figure 4A and B). This classification is supported by high bootstrap values of 95% (subgroup I) and 92% (subgroup II) and robust Hidden-Markov models (data not shown). Such a differentiation of xanthorhodopsins has not been previously reported. Both of the functionally characterized xanthorhodopsins, the rhodopsins of Salinibacter ruber and Gloeobacter violaceus fall into subgroup I. This subgroup also includes a noteworthy cluster of rhodopsins, designated actinorhodopsins, which is found exclusively in Actinobacteria [82]. The xanthorhodopsins of O. arcticus and O. antarcticus are affiliated to subgroup II and the first members of subgroup II that are functionally described (see below). Also included in this subgroup is a cluster of rhodopsins found in marine eukaryotes [83].
Figure 4. Comparison of the Octadecabacter rhodopsins with other microbial rhodopsins.
(A) Unrooted neighbor-joining tree based on the amino acid sequence of representative members of all known groups of microbial rhodopsins (fungal, sensory, halo-, bacterio-, proteo-, and xanthorhodopsins). The two subgroups of xanthorhodopsins are indicated by the roman letters I and II. (B) Rooted detailed view of the xanthorhodopsin-branch of the same tree. Fungal rhodopsins served as outgroup. Bootstrap-values >50 are given at the respective nodes. The subgroups I and II as well as the Actinobacteria and Dinoflagellate subclusters are indicated by brackets. The lifestyle of the associated organisms is indicated by different symbols. For each rhodopsin the NCBI accession number is given in parentheses. (C) Alignment of the putative keto-carotenoid-binding region of xanthorhodopsins. Residues that interact with the keto-carotenoid in Salinibacter xanthorhodopsin as identified by Imasheva et al. [14] are marked by the letters c, g, k and r, which indicate contact with the chain, glucoside, keto group and ring of the carotenoid, respectively. Similarities are marked by solid boxes and colored letters. Identities are additionally marked by background shading. Amino acids corresponding to Gly156 of Salinibacter xanthorhodopsin are highlighted in red. The number of identities and similarities are given for each xanthorhodopsin and as an average for each subgroup (excluding Salinibacter xanthorhodopsin).
One difference between members of subgroup I and subgroup II is the organization of the corresponding gene clusters. All subgroup II xanthorhodopsins are organized in conserved gene clusters (Supplementary Figure S5), which are similar to the majority of proteorhodopsin gene clusters. These gene clusters consist of an opsin-encoding gene and several genes for retinal synthesis [15], [84], [85]. The gene clusters of subgroup I xanthorhodopsins are highly divergent and lack orthologs to conserved retinal synthesis genes found in subgroup II xanthorhodopsin and proteorhodopsin gene clusters. Several retinal synthesis genes have been identified in separate locations of the S. ruber genome [86], but these genes were more similar to the corresponding genes of halophilic archaea than to genes of other rhodopsin-harboring bacteria [15], [86]. Based on bidirectional best-hit analyses, most of these genes were not orthologous to retinal synthesis genes of subgroup II xanthorhodopsin-harboring organisms. Correspondingly, no orthologs of the β-carotene oxygenase gene crtO, which is associated with the subgroup I xanthorhodopsins of S. ruber [86] and T. aquaticus (Supplementary Figure S5), were identified in subgroup II xanthorhodopsin-harboring organisms.
Ecology of Xanthorodopsin Subgroups
Like most rhodopsins, closely related xanthorhodopsins can be found in diverse bacterial taxa [14], [15], indicating that this feature is often transmitted via HGT. Almost all subgroup II xanthorhodopsin sequences available from the NCBI nr database were obtained from mesophilic to psychrophilic marine microorganisms (Figure 4B). The only exception is the xanthorhodopsin from Thioalkalimicrobium cyclicum ALM1, isolated from a limnic hypersaline environment (Mono Lake, California, USA) [87]. More than a third of the subgroup II xanthorhodopsins (six sequences) was found in psychrophilic organisms from polar regions. In contrast, all subgroup I xanthorhodopsin sequences originate from mesophilic to thermophilic organisms derived from non-marine environments. The xanthorhodopsins of the actinorhodopsin cluster were obtained from freshwater habitats [82] whereas Gloeobacter violaceus was isolated from limestone rock [88]. The remaining three subgroup I members originate from thermotolerant [89] and thermophilic organisms [90] that have been isolated from saltern ponds and hot springs, respectively. This indicates that the xanthorhodopsin subgroups differ in their habitat distribution. To test this assumption several metagenomes from various environments were analyzed for the relative abundances of the different rhodopsin types (Figure 5 and Supplementary Figure S6).
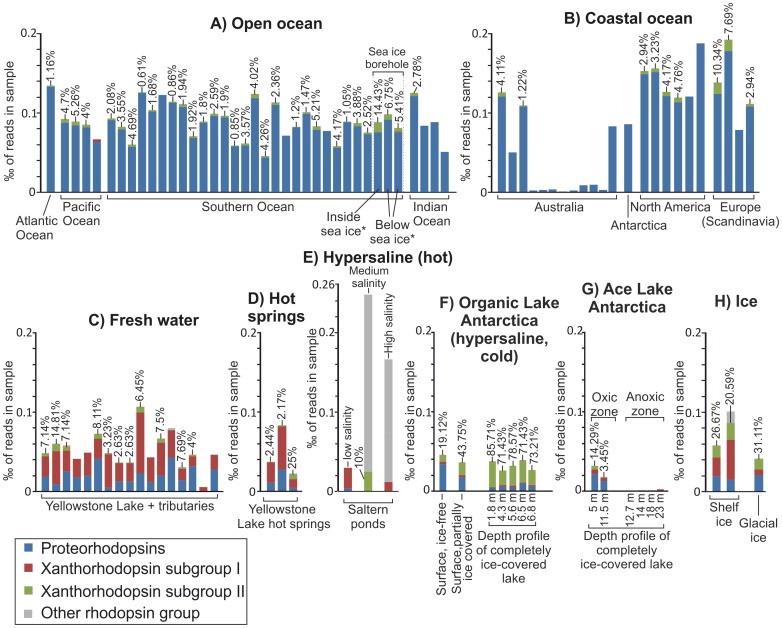
Figure 5. Abundance and diversity of rhodopsins in 454 sequencing-based metagenomes.
The height of each bar indicates the normalized total abundance of rhodopsins, given in ? of reads in the respective metagenomic sample. The relative abundances of different rhodopsin groups are indicated by the relative color proportions in each bar. The relative abundances of subgroup II xanthorhodopsins are given in percent above the respective bars. The results for Sanger sequencing-based metagenomes are shown in a separate figure (Supplementary Figure S6). The represented metagenomes are listed in Supplementary Table S3.
The most abundant rhodopsin type in all marine metagenome samples was proteorhodopsin. Xanthorhodopsin sequences of both subgroups were present but only in low abundances. On average they constitute approximately 1–2% of the total rhodopsin sequences in marine environments. In 454 sequencing-based marine metagenomes xanthorhodopsins were mainly represented by subgroup II (Figure 5A and B), whereas in Sanger sequencing-based marine metagenomes they were more often represented by subgroup I (Supplementary Figure 6A and B). Both xanthorhodopsin subgroups were generally more abundant in freshwater, hot springs, and hypersaline habitats than in marine environments. This indicates that none of the subgroups is a characteristic trait of marine microorganisms. The fact that almost all subgroup II xanthorhodopsins originate from marine isolates (Figure 4) may be due to cultivation bias.
Subgroup I members were on average the most abundant rhodopsin type in freshwater and hot spring habitats, and comprise more than 50% of the total rhodopsin sequences in most samples (Figure 5C and D, Supplementary Figure S6C and D). Subgroup II xanthorhodopsins were also present in these samples and comprised 0–14% of the total rhodopsin sequences in freshwater habitats and 0–25% in hot springs. In warm hypersaline habitats such as solar salterns or tropical hypersaline lagoons xanthorhodopsins constituted varying but substantial fractions of the total rhodopsins. These fractions were dominated by subgroup I in three of the four analyzed hypersaline samples (Figure 5E, Supplementary Figure S6E).
Xanthorhodopsins of both subgroups were identified as the major type of rhodopsin in icy environments such as glacial ice and shelf ice (Figure 5H). This was also the case for subgroup II xanthorhodopsins in Organic Lake, Antarctica. Organic Lake is a shallow, eutrophic, and hypersaline lake with extremely low water temperatures [91], [92]. The relative abundance of subgroup II xanthorhodopsins was lowest (approximately 19%) when the lake was free of ice, increased (approximately 44%) when it was partially covered by ice, and highest (>70%) when it was completely covered by ice (Figure 5F). This shows that xanthorhodopsins, especially those of subgroup II, are mostly associated with psychrophilic organisms, indicating a possible evolutionary advantage over other rhodopsins in icy environments. A depth profile of the marine-derived Ace Lake (Antarctica) during complete ice coverage [38] yielded lower relative xanthorhodopsin abundances than Organic Lake (3–0% at depths >10 m). However, the uppermost sample (5 m depth) of Ace Lake still showed a higher relative abundance (approximately 14%) of subgroup II than any non-ice associated marine sample (Figure 5G). The generally lower rhodopsin abundances compared to Organic Lake may be due to the larger depth-range of the samples in Ace Lake (5–24 m compared to 0–7 m), but also to several factors that differentiate these two environments such as water temperature and salinity [91], [93]. Nonetheless, the importance of subgroup II xanthorhodopsins in icy environments is supported by the fact that the relative abundance of this subgroup was higher (approximately 14%) in seawater sampled directly from inside a borehole drilled through a 3 m thick sheet of pack ice than in the underlying water body (5–7%) or in any other marine sample (0–10%, Figure 5A).
Functional Characterization of Xanthorhodopsin Subgroups
The Octadecabacter opsins were heterologously expressed in E. coli cells. Subsequently, the resulting recombinant E. coli strains displayed a characteristic pink color after addition of retinal (data not shown). Spectral analysis of membrane fragment suspensions revealed absorption maxima at 533(±1) nm and 535(±1) nm for the xanthorhodopsins of O. arcticus and O. antarcticus, respectively (Supplementary Figure S7). These values are higher than the absorption maximum of the reference proteorhodopsin from EBAC31A08 (521±1 nm), but lower than those of the subgroup I xanthorhodopsins from Gloeobacter (540±1 nm) and Salinibacter (560 nm) [94].
The rhodopsins were also analyzed for their potential to bind keto-carotenoids. Based on the crystal structure of Salinibacter xanthorhodopsin [95], Imasheva et al. [14] identified 16 amino acid residues that form the keto-carotenoid binding site and are positioned around the E and F helices of the protein. Gloeobacter xanthorhodopsin, which shares the ability to bind 4-keto-carotenoids [14], [96], harbors 11 identical amino acids at the corresponding positions (Figure 4C). This indicates that not all of the residues predicted in Salinibacter xanthorhodopsin are essential for this function. The residues forming this keto-carotenoid-binding site are conserved among subgroup I. On average 10 identical amino acids were located in the corresponding positions, whereas subgroup II xanthorhodopsins show only five correlating amino acid residues (Figure 4C). The functionally most important difference between subgroup I and subgroup II proteins is the amino acid residue in position 156 with respect to Salinibacter xanthorhodopsin [96]. All subgroup I members contain a glycine residue at this position whereas the majority of subgroup II harbors a tryptophan. It has been shown for the subgroup I xanthorhodopsins of Salinibacter and Gloeobacter that this residue is part of the keto-ring binding pocket. Due to its relatively small size, a glycine at this position provides space for a keto-ring, but substitution with a bulky tryptophan abolishes binding of keto-carotenoids [14]. Thus, most of the xanthorhodopsins belonging to subgroup II lack this requirement for keto-carotenoid binding, except the proteins from O. arcticus, O. antarcticus, T. cyclicum and Nisaea sp. BAL199. However, in all subgroup II xanthorhodopsins differences also occur at positions corresponding to Thr160, Leu194, Leu197, Ala198, Gly201 and Ile205 of Salinibacter xanthorhodopsin. These positions are part of a binding slot along helix F, which harbors the polyene chain of the keto-carotenoid. The absence of this slot in all subgroup II xanthorhodopsins affects keto-carotenoid-binding. This is supported by analysis of difference spectra after addition of crude salinixanthin extracts to membrane fragments harboring heterologously expressed xanthorhodopsins (Supplementary Figure S7). Vibrionic bands at 456, 480 and 521 nm that are typical for binding of salinixanthin [14], [96] were observed in Gloeobacter xanthorhodopsin, but not in Octadecabacter xanthorhodopsin. This shows that the presence of an equivalent to Gly156 is not sufficient to enable binding of salinixanthin in poorly conserved keto-carotenoid binding sites and indicates that subgroup II members cannot bind keto-carotenoids.
The subgroup II xanthorhodopsins were predicted to be light-driven proton pumps like their subgroup I relatives, as they possess conserved residues that are indicative for this function (Supplementary Figure S8) [97]. To confirm this assumption the efflux of protons in xanthorhodopsin-producing E. coli cells was assessed via the acidification rate in a non-buffered, nutrient-free cell suspension according to Béjà et al. [19]. Suspensions of recombinant cells expressing Octadecabacter xanthorhodopsin showed a light-induced acidification, which was absent in suspensions without rhodopsins (Supplementary Figure S7). This result indicates that Octadecabacter xanthorhodopsins function as light-driven proton pumps. However, no growth advantage was observed for Octadecabacter cells in light compared to dark (data not shown). In several species, the function of proteorhodopsins is only supporting survival during periods of starvation [98]–[100]. Thus the activity of the Octadecabacter xanthorhodopsins might be required during transition periods with rapidly changing nutrient availabilities such as formation and melting of sea ice, or to overcome low diffusion rates of nutrients in sea ice.
Conclusions
The genome analyses of the Octadecabacter strains emphasize the importance of horizontal gene transfer among members of the Roseobacter clade. This clade exhibits a large and accessible pan-genome, which seems to be more characteristic than its core-genome. GTAs enable individual Roseobacters to access this pan-genome [68], resulting in a high ability to adapt to changing environmental conditions. In both analyzed Octadecabacter strains, the adaptability is enhanced through TE-mediated genome plasticity, which is much higher than that of other Roseobacter clade members. This is indicated by the large number and size of RGPs in the Octadecabacter genomes (Figure 1), and the numerous genomic rearrangements that are evident in genome alignments (Figure 3). A linkage of this trait to the sea ice habitat of the Octadecabacter strains is in accordance with Collins and Deming [65], [66], who suggested that sea ice environments are hotspots for HGT in marine ecosystems. Thus, polar Octadecabacter strains may be a driving force of the genomic diversity in marine Roseobacters. In order to address whether this is a specific trait of polar Octadecabacter strains or not it is demanding to perform a corresponding analysis of non-polar Octadecabacter isolates in future experiments.
Many of the characteristic features found in the Octadecabacter genomes may represent adaptations to polar habitats. For example, the cyanophycin ligase of O. arcticus could present an advantage to polar marine and sea ice organisms, as nitrogen can be a significant limiting factor to prokaryotic heterotrophic production in polar surface waters during summer [101]. Furthermore, storage compounds in general can enhance the survival of organisms in rapidly changing environments such as sea ice [102]. Mercury reduction can be found in various organisms from polar and non-polar habitats [76], [103]. However, it could be of special significance in polar habitats because the Arctic and Antarctic region both act as sinks for atmospheric mercury leading to seasonal accumulations of mercury in these ecosystems [104]–[106]. An importance of mercury resistance in polar habitats is also indicated by the fact that the corresponding gene clusters are conserved in the Arctic and the Antarctic Octadecabacter strain but absent in many other Roseobacter clade members. The xanthorhodopsins of the Octadecabacter strains were also linked to sea ice habitats. Comparative analysis of these rhodopsins revealed the presence of two distinct xanthorhodopsin subgroups. Functional differences of the two subgroups were indicated by the observed habitat preference, organization of the gene clusters, and the potential for keto-carotenoid-binding. Metagenome analyses showed that none of the xanthorhodopsin subgroups represent a characteristic marine trait. The xanthorhodopsin-encoding genes found in the Octadecabacter-genomes are more typical for ice-associated rather than marine organisms.
The 16S rRNA sequence relationships of polar and non-polar Octadecabacter strains indicate a direct connection between bacterial populations of both poles (Figure 2). Results of the genome analyses support this hypothesis. Most characteristic gene clusters are remarkably conserved in both Octadecabacter strains, despite the fact that they seem to originate from HGT. The most prominent examples are the xanthorhodopsin, flagella and gas vesicle gene clusters. Variations of these features found in mesophilic Roseobacter clade members are often highly divergent in sequence and/or organization (Supplementary Figures S4), indicating that the Arctic and the Antarctic Octadecabacter strains share a common distinct gene pool. Due to the psychrophilic lifestyle of the polar Octadecabacter strains, a transit over the warm surface waters of the equator is unlikely. However, cold deep-water currents could be a possible vector for transport of bacteria between both poles [9]. This assumption remains to be validated by community analyses along deep-water currents and polar surface waters.
Supporting Information
Neighbor-joining tree based on multi locus sequence analyses (MLSA).
(PDF)
Selected gene clusters that represent differences between the Octadecabacter strains.
(PDF)
Neighbor-Joining tree of cyanophycin ligases and cyanophycin ligase-like proteins.
(PDF)
Selected gene clusters shared by both Octadecabacter strains.
(PDF)
Conservation of subgroup I and subgroup II xanthorhodopsin gene clusters.
(PDF)
Abundance and diversity of rhodopsins in Sanger sequencing-based metagenomes.
(PDF)
Functional characterization of the O ctadecabacter xanthorhodopsins.
(PDF)
Functional residues important for proton translocation in different rhodopsins.
(PDF)
Rhodopsin sequences used for phylogenetic and metagenomic analyses.
(PDF)
Cyanophycin ligase and cyanophycin ligase-like protein sequences used for phylogenetic analyses.
(PDF)
List of metagenome projects used for the analyses of biogeography and diversity of microbial rhodopsins.
(PDF)
Overview of selected features in regions of enhanced genome plasticity in the genomes of Octadecabacter arcticus (Oar-RGP1-17, pOAR118, pOAR160) and Octadecabacter antarcticus (Oan-RGP1-16, pOAN63).
(PDF)
Acknowledgments
We thank Renate Gahl-Janßen (Oldenburg) for culturing the Octadecabacter strains and Antje Wollherr (Göttingen) for providing bioinformatical support.
Funding Statement
This study was supported by the Niedersächsisches VW-Vorab “Comparative and functional genome analysis of representative members of the Roseobacter clade” (ZN2235), Germany, the Collaborative Research Center Roseobacter (TRR 51) funded by Deutsche Forschungsgemeinschaft, Germany, and the Marine Microbiology Initiative of the Moore Foundation, USA. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Buchan A, Gonzalez JM, Moran MA (2005) Overview of the marine Roseobacter lineage. Appl Environ Microbiol 71: 5665–5677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Wagner-Döbler I, Biebl H (2006) Environmental biology of the marine Roseobacter lineage. Annu Rev Microbiol 60: 255–280. [DOI] [PubMed] [Google Scholar]
- 3. Brinkhoff T, Giebel H, Simon M (2008) Diversity, ecology, and genomics of the Roseobacter clade: a short overview. Arch Microbiol 189: 531–539. [DOI] [PubMed] [Google Scholar]
- 4. Luo H, Löytynoja A, Moran MA (2011) Genome content of uncultivated marine Roseobacters in the surface ocean. Environ Microbiol 14: 41–51. [DOI] [PubMed] [Google Scholar]
- 5. Newton RJ, Griffin LE, Bowles KM, Meile C, Gifford S, et al. (2010) Genome characteristics of a generalist marine bacterial lineage. ISME J 4: 784–798. [DOI] [PubMed] [Google Scholar]
- 6. Brinkmeyer R, Knittel K, Jurgens J, Weyland H, Amann R, et al. (2003) Diversity and structure of bacterial communities in arctic versus antarctic pack ice. Appl Environ Microbiol 69: 6610–6619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Gosink JJ, Staley JT (1995) Biodiversity of gas vacuolate bacteria from Antarctic sea ice and water. Appl Environ Microbiol 61: 3486–3489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Gosink JJ, Herwig RP, Staley JT (1997) Octadecabacter arcticus gen. nov., sp. nov., and O. antarcticus, sp. nov., nonpigmented, psychrophilic gas vacuolate bacteria from polar sea ice and water. Syst Appl Microbiol 20: 356–365. [Google Scholar]
- 9. Staley JT, Gosink JJ (1999) Poles apart: Biodiversity and biogeography of sea ice bacteria. Annu Rev Microbiol 53: 189–215. [DOI] [PubMed] [Google Scholar]
- 10. Mock T, Thomas DN (2005) Recent advances in sea-ice microbiology. Environ Microbiol 7: 605–619. [DOI] [PubMed] [Google Scholar]
- 11. Thomas DN, Dieckmann GS (2002) Antarctic sea ice – a habitat for extremophiles. Science 295: 641–644. [DOI] [PubMed] [Google Scholar]
- 12. Wayne LG, Brenner DJ, Colwell RR, Grimont PAD, Kandler O, et al. (1987) Report of the Ad hoc committee on reconciliation of approaches to bacterial systematics. Int J Syst Bacteriol 37: 463–464. [Google Scholar]
- 13. Balashov SP, Imasheva ES, Boichenko VA, Anton J, Wang JM, et al. (2005) Xanthorhodopsin: a proton pump with a light-harvesting carotenoid antenna. Science 309: 2061–2064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Imasheva ES, Balashov SP, Choi AR, Jung KH, Lanyi JK (2009) Reconstitution of Gloeobacter violaceus rhodopsin with a light-harvesting carotenoid antenna. Biochemistry 48: 10948–10955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. McCarren J, DeLong EF (2007) Proteorhodopsin photosystem gene clusters exhibit co-evolutionary trends and shared ancestry among diverse marine microbial phyla. Environ Microbiol 9: 846–858. [DOI] [PubMed] [Google Scholar]
- 16. Spudich JL, Yang CS, Jung KH, Spudich EN (2000) Retinylidene proteins: Structures and functions from archaea to humans. Annu Rev Cell Dev Biol 16: 365–392. [DOI] [PubMed] [Google Scholar]
- 17. Brown LS (2004) Fungal rhodopsins and opsin-related proteins: eukaryotic homologues of bacteriorhodopsin with unknown functions. Photochem Photobiol Sci 3: 555–565. [DOI] [PubMed] [Google Scholar]
- 18. Fuhrman JA, Schwalbach MS, Stingl U (2008) Opinion – Proteorhodopsins: an array of physiological roles? Nat Rev Microbiol 6: 488–494. [DOI] [PubMed] [Google Scholar]
- 19. Beja O, Aravind L, Koonin EV, Suzuki MT, Hadd A, et al. (2000) Bacterial rhodopsin: Evidence for a new type of phototrophy in the sea. Science 289: 1902–1906. [DOI] [PubMed] [Google Scholar]
- 20. Zobell CE (1941) Studies on marine bacteria. I. The cultural requirements of heterotrophic aerobes. J Mar Res 4: 42–75. [Google Scholar]
- 21. Staden R (1996) The Staden sequence analysis package. Mol Biotechnol 5: 233–241. [DOI] [PubMed] [Google Scholar]
- 22. Tech M, Merkl R (2003) YACOP: Enhanced gene prediction obtained by a combination of existing methods. In Silico Biol 3: 441–451. [PubMed] [Google Scholar]
- 23. Delcher AL, Bratke KA, Powers EC, Salzberg SL (2007) Identifying bacterial genes and endosymbiont DNA with Glimmer. Bioinformatics 23: 673–679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Overbeek R, Larsen N, Walunas T, D'Souza M, Pusch G, et al. (2003) The ERGO genome analysis and discovery system. Nucleic Acids Res 31: 164–171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Zdobnov EM, Apweiler R (2001) InterProScan – an integration platform for the signature-recognition methods in InterPro. Bioinformatics 17: 847–848. [DOI] [PubMed] [Google Scholar]
- 26. Siguier P, Perochon J, Lestrade L, Mahillon J, Chandler M (2006) ISfinder: the reference centre for bacterial insertion sequences. Nucleic Acids Res 34: D32–D36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Altenhoff AM, Dessimoz C (2009) Phylogenetic and Functional Assessment of Orthologs Inference Projects and Methods. PLoS Comp Biol 5: e1000262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Hulsen T, Huynen MA, de Vlieg J, Groenen PMA (2006) Benchmarking ortholog identification methods using functional genomics data. Genome Biol 7: R31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Needleman SB, Wunsch CD (1970) A general method applicable to search for similarities in amino acid sequence of 2 proteins. J Mol Biol 48: 443–453. [DOI] [PubMed] [Google Scholar]
- 30. Kalhöfer D, Thole S, Voget S, Lehmann R, Liesegang H, et al. (2011) Comparative genome analysis and genome-guided physiological analysis of Roseobacter litoralis . BMC Genomics 12: 324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Thole S, Kalhöfer D, Voget S, Berger M, Engelhardt T, et al. (2012) Phaeobacter gallaeciensis genomes from globally opposite locations reveal high similarity of adaptation to surface life. ISME J 6: 2229–2244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Sangar V, Blankenberg D, Altman N, Lesk AM, Sangar V, et al. (2007) Quantitative sequence-function relationships in proteins based on gene ontology. BMC Bioinformatics 8: 294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Kurtz S, Phillippy A, Delcher AL, Smoot M, Shumway M, et al. (2004) Versatile and open software for comparing large genomes. Genome Biology 5: R12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Markowitz VM, Chen IM, Palaniappan K, Chu K, Szeto E, et al. (2012) IMG: the Integrated Microbial Genomes database and comparative analysis system. Nucleic Acids Res 40: D115–122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Richter M, Rosselló-Móra R (2009) Shifting the genomic gold standard for the prokaryotic species definition. Proc Natl Acad Sci U S A 106: 19126–19131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Larkin MA, Blackshields G, Brown NP, Chenna R, McGettigan PA, et al. (2007) Clustal W and clustal X version 2.0. Bioinformatics 23: 2947–2948. [DOI] [PubMed] [Google Scholar]
- 37. Ludwig W, Strunk O, Westram R, Richter L, Meier H, et al. (2004) ARB: a software environment for sequence data. Nucleic Acids Res 32: 1363–1371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Lauro FM, DeMaere MZ, Yau S, Brown MV, Ng C, et al. (2011) An integrative study of a meromictic lake ecosystem in Antarctica. ISME J 5: 879–895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Varin T, Lovejoy C, Jungblut AD, Vincent WF, Corbeil J (2010) Metagenomic profiling of Arctic microbial mat communities as nutrient scavenging and recycling systems. Limnol Oceanogr 55: 1901–1911. [Google Scholar]
- 40. Thomas T, Rusch D, DeMaere MZ, Yung PY, Lewis M, et al. (2010) Functional genomic signatures of sponge bacteria reveal unique and shared features of symbiosis. ISME J 4: 1557–1567. [DOI] [PubMed] [Google Scholar]
- 41. Rusch DB, Halpern AL, Sutton G, Heidelberg KB, Williamson S, et al. (2007) The Sorcerer II Global Ocean Sampling expedition: Northwest Atlantic through Eastern Tropical Pacific. PLoS Biol 5: 398–431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Simon C, Wiezer A, Strittmatter AW, Daniel R (2009) Phylogenetic diversity and metabolic potential revealed in a glacier ice metagenome. Appl Environ Microbiol 75: 7519–7526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Hewson I, Paerl RW, Tripp HJ, Zehr JP, Karl DM (2009) Metagenomic potential of microbial assemblages in the surface waters of the central Pacific Ocean tracks variability in oceanic habitat. Limnol Oceanograph 54: 1981–1994. [Google Scholar]
- 44. Gilbert JA, Thomas S, Cooley NA, Kulakova A, Field D, et al. (2009) Potential for phosphonoacetate utilization by marine bacteria in temperate coastal waters. Environ Microbiol 11: 111–125. [DOI] [PubMed] [Google Scholar]
- 45. Havig JR, Raymond J, Meyer-Dombard DR, Zolotova N, Shock EL (2011) Merging isotopes and community genomics in a siliceous sinter-depositing hot spring. J Geophys Res 116: G01005. [Google Scholar]
- 46. Martinez A, Tyson GW, DeLong EF (2009) Widespread known and novel phosphonate utilization pathways in marine bacteria revealed by functional screening and metagenomic analyses. Environ Microbiol 12: 222–238. [DOI] [PubMed] [Google Scholar]
- 47. Rich VI, Pham VD, Eppley J, Shi Y, DeLong EF (2011) Time-series analyses of Monterey Bay coastal microbial picoplankton using a ‘genome proxy’ microarray. Environ Microbiol 13: 116–134. [DOI] [PubMed] [Google Scholar]
- 48. Dinsdale EA, Edwards RA, Hall D, Angly D, Breitbart M, et al. (2008) Functional metagenomic profiling of nine biomes. Nature 452: 629–632. [DOI] [PubMed] [Google Scholar]
- 49. Kan J, Clingenpeel S, Macur RE, Inskeep WP, Lovalvo D, et al. (2011) Archaea in Yellowstone Lake. ISME J 5: 1784–1795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Lutnaes BF, Oren A, Liaaen-Jensen S (2002) New C-40-carotenoid acyl glycoside as principal carotenoid in Salinibacter ruber, an extremely halophilic eubacterium. J Nat Prod 65: 1340–1343. [DOI] [PubMed] [Google Scholar]
- 51. Langille MGI, Brinkman FSL (2009) IslandViewer: an integrated interface for computational identification and visualization of genomic islands. Bioinformatics 25: 664–665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Palys T, Berger E, Mitrica I, Nakamura LK, Cohan FM (2000) Protein-coding genes as molecular markers for ecologically distinct populations: the case of two Bacillus species. Int J Syst Evol Microbiol 50: 1021–1028. [DOI] [PubMed] [Google Scholar]
- 53. Thompson FL, Gevers D, Thompson CC, Dawyndt P, Naser S, et al. (2005) Phylogeny and molecular identification of Vibrios on the basis of multilocus sequence analysis. Appl Environ Microbiol 71: 5107–5115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Adekambi T, Drancourt M (2004) Dissection of phylogenetic relationships among 19 rapidly growing Mycobacterium species by 16S rRNA, hsp65, sodA, recA and rpoB gene sequencing. Int J Syst Evol Microbiol 54: 2095–2105. [DOI] [PubMed] [Google Scholar]
- 55. Soria-Carrasco V, Valens-Vadell M, Pena A, Anton J, Amann R, et al. (2007) Phylogenetic position of Salinibacter ruber based on concatenated protein alignments. Syst Appl Microbiol 30: 171–179. [DOI] [PubMed] [Google Scholar]
- 56. Zhang DC, Li HR, Xin YH, Liu HC, Chi ZM, et al. (2008) Phaeobacter arcticus sp. nov., a psychrophilic bacterium isolated from the Arctic. Int J Syst Evol Microbiol 58: 1384–1387. [DOI] [PubMed] [Google Scholar]
- 57. Yoon JH, Kang SJ, Lee SY, Oh TK (2007) Phaeobacter daeponensis sp. nov., isolated from a tidal flat of the Yellow Sea in Korea. Int J Syst Evol Microbiol 57: 856–861. [DOI] [PubMed] [Google Scholar]
- 58. Fernandes N, Case RJ, Longford SR, Seyedsayamdost MR, Steinberg PD, et al. (2011) Genomes and virulence factors of novel bacterial pathogens causing bleaching disease in the marine red alga Delisea pulchra . PLoS One 6: e27387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Gray HMY (2000) It takes two transposons to tango: transposable-element-mediated chromosomal rearrangements. Trends Genet 16: 461–468. [DOI] [PubMed] [Google Scholar]
- 60. Petrosino JF, Xiang Q, Karpathy SE, Jiang H, Yerrapragada S, et al. (2006) Chromosome rearrangement and diversification of Francisella tularensis revealed by the type B (OSU18) genome sequence. J Bacteriol 188: 6977–6985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Siguier P, Filee J, Chandler M (2006) Insertion sequences in prokaryotic genomes. Curr Opin Microbiol 9: 526–531. [DOI] [PubMed] [Google Scholar]
- 62. de Vries J, Wackernagel W (2002) Integration of foreign DNA during natural transformation of Acinetobacter sp. by homology-facilitated illegitimate recombination. Proc Natl Acad Sci U S A 99: 2094–2099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Prudhomme M, Libante V, Claverys J-P (2002) Homologous recombination at the border: Insertion-deletions and the trapping of foreign DNA in Streptococcus pneumoniae . Proc Natl Acad Sci U S A 99: 2100–2105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Thomas CM, Nielsen KM (2005) Mechanisms of, and barriers to, horizontal gene transfer between bacteria. Nat Rev Microbiol 3: 711–721. [DOI] [PubMed] [Google Scholar]
- 65. Collins RE, Deming JW (2011) Abundant dissolved genetic material in Arctic sea ice Part I: Extracellular DNA. Polar Biol 34: 1819–1830. [Google Scholar]
- 66. Collins RE, Deming JW (2011) Abundant dissolved genetic material in Arctic sea ice Part II: Viral dynamics during autumn freeze-up. Polar Biol 34: 1831–1841. [Google Scholar]
- 67. Wallden K, Rivera-Calzada A, Waksman G (2010) Type IV secretion systems: versatility and diversity in function. Cell Microbiol 12: 1203–1212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Biers EJ, Wang K, Pennington C, Belas R, Chen F, et al. (2008) Occurrence and expression of gene transfer agent genes in marine bacterioplankton. Appl Environ Microbiol 74: 2933–2939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Palys T, Nakamura LK, Cohan FM (1997) Discovery and classification of ecological diversity in the bacterial world: The role of DNA sequence data. Int J Syst Bacteriol 47: 1145–1156. [DOI] [PubMed] [Google Scholar]
- 70. Berg H, Ziegler K, Piotukh K, Baier K, Lockau W, et al. (2000) Biosynthesis of the cyanobacterial reserve polymer multi-L-arginyl-poly-L-aspartic acid (cyanophycin) – Mechanism of the cyanophycin synthetase reaction studied with synthetic primers. Eur J Biochem 267: 5561–5570. [DOI] [PubMed] [Google Scholar]
- 71. Füser G, Steinbüchel A (2007) Analysis of genome sequences for genes of cyanophycin metabolism: Identifying putative cyanophycin metabolizing prokaryotes. Macromol Biosci 7: 278–296. [DOI] [PubMed] [Google Scholar]
- 72. Krehenbrink M, Oppermann-Sanio FB, Steinbüchel A (2002) Evaluation of non-cyanobacterial genome sequences for occurrence of genes encoding proteins homologous to cyanophycin synthetase and cloning of an active cyanophycin synthetase from Acinetobacter sp. strain DSM 587. Arch Microbiol 177: 371–380. [DOI] [PubMed] [Google Scholar]
- 73. Bowman JP, Gosink JJ, McCammon SA, Lewis TE, Nichols DS, et al. (1998) Colwellia demingiae sp. nov., Colwellia hornerae sp. nov., Colwellia rossensis sp. nov. and Colwellia psychrotropica sp. nov.: psychrophilic Antarctic species with the ability to synthesize docosahexaenoic acid (22:ω63). Int J Syst Bacteriol 48: 1171–1180. [Google Scholar]
- 74. Methé BA, Nelson KE, Deming JW, Momen B, Melamud E, et al. (2005) The psychrophilic lifestyle as revealed by the genome sequence of Colwellia psychrerythraea 34H through genomic and proteomic analyses. Proc Natl Acad Sci U S A 102: 10913–10918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Scanlan DJ, Ostrowski M, Mazard S, Dufresne A, Garczarek L, et al. (2009) Ecological genomics of marine picocyanobacteria. Microbiol Mol Biol Rev 73: 249–299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Qian D, Jiang L, Lu L, Wei CH, Li Y (2011) Biochemical and structural properties of cyanases from Arabidopsis thaliana and Oryza sativa . PloS One 6: e18300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Kamennaya NA, Post AF (2011) Characterization of cyanate metabolism in marine Synechococcus and Prochlorococcus spp. Appl Environ Microbiol 77: 291–301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. Osborn AM, Bruce KD, Strike P, Ritchie DA (1997) Distribution, diversity and evolution of the bacterial mercury resistance (mer) operon. FEMS Microbiol Rev 19: 239–262. [DOI] [PubMed] [Google Scholar]
- 79. Offner S, Hofacker A, Wanner G, Pfeifer F (2000) Eight of fourteen gvp genes are sufficient for formation of gas vesicles in halophilic archaea. J Bacteriol 182: 4328–4336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. Kyndt JA, Hurley JK, Devreese B, Meyer TE, Cusanovich MA, et al. (2004) Rhodobacter capsulatus photoactive yellow protein: genetic context, spectral and kinetics characterization, and mutagenesis. Biochemistry 43: 1809–1820. [DOI] [PubMed] [Google Scholar]
- 81. Severin J, Wohlfarth A, Galinski EAI (1992) The predominant role of recently discovered tetrahydropyrimidines for the osmoadaptation of halophilic eubacteria. J Gen Microbiol 138: 1629–1638. [Google Scholar]
- 82. Sharma AK, Sommerfeld K, Bullerjahn GS, Matteson AR, Wilhelm SW, et al. (2009) Actinorhodopsin genes discovered in diverse freshwater habitats and among cultivated freshwater Actinobacteria. ISME J 3: 726–737. [DOI] [PubMed] [Google Scholar]
- 83. Slamovits CH, Okamoto N, Burri L, James ER, Keeling PJ (2011) A bacterial proteorhodopsin proton pump in marine eukaryotes. Nat Commun 2: 183. [DOI] [PubMed] [Google Scholar]
- 84. Armstrong GA (1997) Genetics of eubacterial carotenoid biosynthesis: A colorful tale. Annu Rev Microbiol 51: 629–659. [DOI] [PubMed] [Google Scholar]
- 85.Sabehi G, Loy A, Jung K, Partha R, Spudich JL, et al.. (2005) New insights into metabolic properties of marine bacteria encoding proteorhodopsins. PLoS Biol: 1409–1417. [DOI] [PMC free article] [PubMed]
- 86. Mongodin EF, Nelson KE, Daugherty S, DeBoy RT, Wister J, et al. (2005) The genome of Salinibacter ruber: Convergence and gene exchange among hyperhalophilic bacteria and archaea. Proc Nat Acad Sci U S A 102: 18147–18152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87. Sorokin DY, Gorlenko VM, Tourova TP, Tsapin A, Nealson KH, et al. (2002) Thioalkalimicrobium cyclicum sp. nov and Thioalkalivibrio jannaschii sp. nov., novel species of haloalkaliphilic, obligately chemolithoautotrophic sulfur-oxidizing bacteria from hypersaline alkaline Mono Lake (California). Int J Syst Evol Microbiol 52: 913–920. [DOI] [PubMed] [Google Scholar]
- 88. Rippka R, Waterbury J, Cohen-Bazire G (1974) A cyanobacterium which lacks thylakoids. Arch Microbiol 100: 419–436. [Google Scholar]
- 89. Bowers KJ, Mesbah NM, Wiegel J (2009) Biodiversity of poly-extremophilic bacteria: Does combining the extremes of high salt, alkaline pH and elevated temperature approach a physico-chemical boundary for life? Saline Systems 5: 9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90. Brock TD, Freeze H (1969) Thermus aquaticus gen. n. and sp. n., a nonsporulating extreme thermophile. J Bacteriol 98: 289–297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Franzmann PD, Deprez PP, Burton HR, van den Hoff J (1987) Limnology of Organic Lake, Antarctica, a meromictic lake that contains high concentrations of dimethyl sulfide. Austral J Mar Fresh Res 38: 409–417. [Google Scholar]
- 92. Yau S, Lauro FM, DeMaere MZ, Brown MV, Thomas T, et al. (2011) Virophage control of antarctic algal host–virus dynamics. Proc Natl Acad Sci U S A 108: 6163–6168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93. Rankin LM, Gibson JAE, Franzmann PD, Burton HR (1996) The chemical stratification and microbial communities of Ace Lake, Antarctica: A review of the characteristics of a marine-derived meromictic lake. Polarforschung 66: 33–52. [Google Scholar]
- 94. Lanyi JK, Balashov SP (2008) Xanthorhodopsin: A bacteriorhodopsin-like proton pump with a carotenoid antenna. Biochim Biophys Acta Bioenerg 1777: 684–688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. Luecke H, Schobert B, Stagno J, Imasheva ES, Wang JM, et al. (2008) Crystallographic structure of xanthorhodopsin, the light-driven proton pump with a dual chromophore. Proc Natl Acad Sci U S A 105: 16561–16565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96. Balashov SP, Imasheva ES, Choi AR, Jung KH, Liaaen-Jensen S, et al. (2010) Reconstitution of Gloeobacter rhodopsin with echinenone: Role of the 4-Keto group. Biochemistry 49: 9792–9799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97. Balashov SP (2000) Protonation reactions and their coupling in bacteriorhodopsin. Biochim Biophys Acta Bioenerg 1460: 75–94. [DOI] [PubMed] [Google Scholar]
- 98. DeLong EF, Beja O (2010) The light-driven proton pump proteorhodopsin enhances bacterial survival during tough times. PLoS Biol 8: e1000359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99. Gomez-Consarnau L, Gonzalez JM, Coll-Llado M, Gourdon P, Pascher T, et al. (2007) Light stimulates growth of proteorhodopsin-containing marine Flavobacteria . Nature 445: 210–213. [DOI] [PubMed] [Google Scholar]
- 100. Gomez-Consarnau L, Akram N, Lindell K, Pedersen A, Neutze R, et al. (2010) Proteorhodopsin phototrophy promotes survival of marine bacteria during starvation. PLoS Biol 8: e1000358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101. Ortega-Retuerta E, Jeffrey WH, Joux F (2012) Evidence of heterotrophic prokaryotic activity limitation by nitrogen in the Western Arctic Ocean during summer. Polar Biol 35: 785–794. [Google Scholar]
- 102. Ayub ND, Pettinari MJ, Ruiz JA, López NI (2004) A polyhydroxybutyrate-producing Pseudomonas sp. isolated from Antarctic environments with high stress resistance. Curr Microbiol 49: 170–174. [DOI] [PubMed] [Google Scholar]
- 103. Møller AK, Barkay T, Al-Soud WA, Sørensen SJ, Skov H, et al. (2012) Diversity and characterization of mercury-resistant bacteria in snow, freshwater and sea-ice brine from the High Arctic. FEMS Microbiol Ecol 75: 390–401. [DOI] [PubMed] [Google Scholar]
- 104. Ariya PA, Dastoor AP, Amyot M, Schroeder WH, Barrie L, et al. (2004) The Arctic: a sink for mercury. Tellus B 56: 397–403. [Google Scholar]
- 105. Poulain AJ, Ní Chadhain SM, Ariya PA, Amyot M, Garcia E, et al. (2007) Potential for mercury reduction by microbes in the high arctic. Appl Environ Microbiol 73: 2230–2238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106. Ebinghaus R, Kock HH, Temme C, Einax JW, Löwe AG, et al. (2002) Antarctic springtime depletion of atmospheric mercury. Environ Sci Technol 36: 1238–1244. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Neighbor-joining tree based on multi locus sequence analyses (MLSA).
(PDF)
Selected gene clusters that represent differences between the Octadecabacter strains.
(PDF)
Neighbor-Joining tree of cyanophycin ligases and cyanophycin ligase-like proteins.
(PDF)
Selected gene clusters shared by both Octadecabacter strains.
(PDF)
Conservation of subgroup I and subgroup II xanthorhodopsin gene clusters.
(PDF)
Abundance and diversity of rhodopsins in Sanger sequencing-based metagenomes.
(PDF)
Functional characterization of the O ctadecabacter xanthorhodopsins.
(PDF)
Functional residues important for proton translocation in different rhodopsins.
(PDF)
Rhodopsin sequences used for phylogenetic and metagenomic analyses.
(PDF)
Cyanophycin ligase and cyanophycin ligase-like protein sequences used for phylogenetic analyses.
(PDF)
List of metagenome projects used for the analyses of biogeography and diversity of microbial rhodopsins.
(PDF)
Overview of selected features in regions of enhanced genome plasticity in the genomes of Octadecabacter arcticus (Oar-RGP1-17, pOAR118, pOAR160) and Octadecabacter antarcticus (Oan-RGP1-16, pOAN63).
(PDF)